Abstract
Case series summary
Chronic diarrhea is a common and recurring problem in feline medicine. Intestinal trichomonads have been reported as causative agents of diarrhea in cats. While Pentatrichomonas hominis is considered commensal, Tritrichomonas foetus has been found to cause feline large bowel diarrhea in cats. In our report, two young cats infected with the feline leukemia virus (FeLV) and presenting with chronic diarrhea were identified as having trichomonads in their feces, based on direct examination and fecal culture. Molecular assays (PCR and DNA sequencing) revealed that the parasite involved was P hominis, not T foetus, as was suspected. The animals had already been subjected to ineffective therapy with metronidazole, and after the use of ronidazole, their feces became dry and formed.
Relevance and novel information
This case report describes P hominis infection as a possible cause of chronic diarrhea in two young cats also infected by FeLV. The parasite was probably resistant to metronidazole, the drug of choice in the literature, and sensitive to ronidazole. Although considered commensal, P hominis infection should be evaluated as a differential diagnosis in cats with chronic diarrhea, especially those that are immunocompromised. Moreover, a combination of techniques such as direct examination and/or fecal culture and PCR is essential for an accurate diagnosis of P hominis infection.
Introduction
Diarrhea caused by infectious diseases is a common problem in feline medicine, particularly in young animals.1 Many studies have identified an important association between diarrhea and trichomonad infection in domestic cats.2–4
Two intestinal trichomonad species have been described in cats and have received attention from the scientific community.3 Tritrichomonas foetus is a flagellate protozoan recognized as an important agent of large bowel diarrhea in domestic cats. The parasite colonizes portions of the large intestine, and the infection manifests as chronic and recurrent diarrhea with mucus and fresh blood.5
Pentatrichomonas hominis is usually considered commensal and opportunistic, and thus its pathogenic importance has been questioned in several cases,3 although some authors describe that marked growth can lead to diarrhea.6 This flagellate inhabits the gastrointestinal tract of many vertebrates, such as humans, dogs, monkeys, guinea pigs and cats.7,8
In people, P hominis is described as the agent of gastrointestinal disorders, with clinical manifestations similar to those of irritable bowel syndrome.9 In addition, the parasite has already been isolated from the pleural effusion fluid of a woman with lupus,8 and has also been identified in the fecal samples of a man with diarrhea, fever and arthralgia.10
The transmission of trichomonads occurs directly between hosts, likely through the fecal–oral route via the ingestion of trophozoites.11
In some studies, based on light microscopic examination, P hominis was erroneously identified as the cause of chronic diarrhea in cats.2–4 After a molecular assay was used to analyze the 18S rRNA gene present in trichomonad isolates, the agent was identified as T foetus.12
The diagnosis of trichomonad infection can be made by the examination of fresh fecal smears, isolation after using a specific culture system or the detection of DNA using PCR and sequencing.13
The most employed techniques for diagnosis in routine feline medicine are direct examination and fecal culture, but they require living organisms in order to prevent false-negative results.14 In addition, these methods do not differentiate between T foetus and P hominis. Both species have the same type of motility and similar forms, and are able to grow in the same culture medium.15 Detection by PCR using specific primers is the more specific and sensitive method to correctly identify the trichomonad involved.16
It is crucial to correctly identify these two parasites, especially as there are different therapeutic approaches for each case.15,17 Feline trichomoniasis caused by T foetus infection is treated using ronidazole, a potentially neurotoxic drug.18 In contrast, the treatment of P hominis infection is described as the use of drugs such as metronidazole,18 which is proven ineffective against T foetus.17
In this report, we describe two cases of young domestic cats with chronic diarrhea and feline leukemia virus (FeLV) infection that presented with P hominis in their feces and received treatment with ronidazole.
Case series description
A 1-year-old, neutered, mixed-breed female was presented to our research group with a history of persistent chronic diarrhea with mucus and blood and fecal incontinence for more than 2 months. The animal had been rescued from a park at 3 months of age and at the time of presentation lived confined in an apartment with other dogs and cats, and was infected with FeLV.
The second cat lived in the same place as the first cat. It was a 2-year-old, neutered, mixed-breed male cat presenting with a >2 month history of chronic diarrhea with mucus and malodor. The animal was adopted at 6 months of age and was also infected with FeLV.
The two cats had been treated for diarrhea with mebendazole (22 mg/kg PO q24h for 3 days), febendazole (50 mg/kg PO q24h for 5 days), sulfonamides (15 mg/kg PO q12h for 7 days) and metronidazole (12.5 mg/kg PO q12h for 7 days) in association with a change in diet. The owner was not able to give the correct doses of the medications used, and none of the therapies were successful in eliminating the diarrhea.
On physical examination, the first, female, cat was in bad body condition (score 1/5) and displayed prostration and apathy. The second, male, cat was more alert and in a better body condition (score 2/5). According to the owner, they were fed commercial intestinal food and given filtered water.
Fresh fecal samples were collected after spontaneous defecation and added to a saline solution for a maximum of 6 h until they were processed in the laboratory. First, the fecal samples were examined directly using a microscope set to × 200 and × 400 magnification. Motile trophozoites with the morphology and movement characteristic of trichomonads were identified. An aliquot of each fecal sample was then inoculated in modified Diamond’s medium, incubated at 37°C and examined under an optic microscope daily. The results from the culture system also revealed trichomonad trophozoites. Aliquots of the fresh fecal samples and of the cultures were stored at −20°C until PCR processing began. Another aliquot of each fecal sample was stored in Raillet and Henry’s solution (formalin/acetic acid) and examined by two parasitological techniques – spontaneous sedimentation and flotation using zinc sulfate solution – to search for other gastrointestinal parasites. No parasites were found during these examinations. The two fecal samples were also tested for Giardia species, using PCR, and the results were negative.
As the cats had presented with chronic diarrhea and were in a fragile state of health it was crucial to start treatment as soon as possible without waiting for PCR results. Based on the results of direct examination and fecal culture, and with the assumption that the infection was caused by T foetus, treatment with ronidazole was started.
Therapy was initiated with ronidazole (30 mg/kg PO q24h for 14 days). The female cat had an isolated episode of vomiting, which was controlled with antiemetic support therapy. The other cat did not show any adverse event during the therapy. One week after the start of treatment, the fecal consistency of the two cats had improved, presenting no mucus or blood.
Once the treatment with ronidazole was finished, new fecal samples were collected and did not test positive during any of the parasitological examinations. Three months later, the female cat died, likely owing to complications of FeLV infection, which is associated with respiratory disorders. The owner did not authorize a necropsy of the animal. The second cat, the male, remained asymptomatic.
The molecular assay of the fecal samples of the two cats needed to confirm the real agent could only be performed later. DNA extraction and PCR were performed at the Laboratory of Parasites` Molecular Biology, at Universidade Federal Fluminense. DNA was extracted directly from the positive fecal cultures by thermal shock via heating and freezing.
The parasites T foetus and P hominis were tested for using the primers previously described by Levy et al and Gookin et al.12,19
For T foetus, in the primary PCR, the primers used were TRF3 and TRF4, in the following conditions: buffer (1×); Mg (2.5 mM); dNTP mix (0.2 mM); primer (200 ng); Taq Platinum 2.5 U (Invitrogen); DNA 3 µl. Cycling was carried out at as follows: 45 cycles at 94ºC for 5 mins (94ºC for 30 s; 67ºC for 30 s; 72ºC for 30 s), followed by 7 mins at 72ºC. For the nested PCR, the primers were TFITS-F and TFITS-R and the conditions were as follows: Buffer (1×); Mg (5 mM); dNTP mix (0.2 mM); primer (200 ng); Taq Platinum 2.5 U (Invitrogen); DNA 3 µl. Cycling conditions were as follows: 35 cycles at 95ºC for 5 mins (95ºC for 30 s; 57ºC for 30 s; 72ºC for 30 s) followed by 7 mins at 72ºC.
For P hominis, we used the primers Th3 and Th5, which amplify a 339 bp sequence of the 18S rRNA gene. The PCR conditions were as follows: buffer (1×); Mg (3 mM); dNTP (0.2 mM); primer (200 ng), Taq Platinum 2.5 U (Invitrogen); 5 µl DNA. Cycling conditions were as follows: 45 cycles at 95ºC for 5 mins (95ºC for 1 min, 64ºC for 1 min, 72ºC for 2 mins) followed by 5 mins at 72ºC.
There was only amplification of the samples in the test for P hominis (Figure 1), and the corresponding amplicon were purified with Kit Wizard SV Gel and PCR Clean-Up System (Promega), following the manufacturer’s protocols. Obtained products were sequenced directly in both strips in an automatic sequencer (Applied Biosystems) by Genomic Platform – DNA Sequencing-RPT01A (Rede de Plataformas Tecnológicas Fiocruz).
Figure 1.
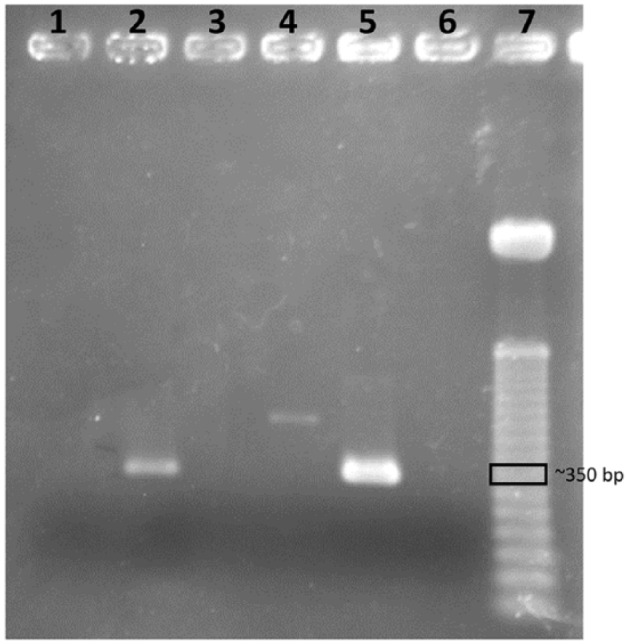
Agarose gel electrophoresis (2.2%) of 18S gene PCR amplification from cultures of Pentatrichomonas hominis. Lanes 1, 3 and 4: suspected negative samples (from others cats studied by our group, and not described in this paper). Lane 2: positive sample from the male cat. Lane 5: positive sample from the female cat. Lane 6: negative control. Lane 7: 50 bp DNA ladder (Promega)
Chromas Lite 2.1 and BioEdit 7.2.5 were used to edit, analyze and align sequences, which were compared with the Genbank data base. The nucleotide sequence determined was submitted to GenBank under accession number MG015711.
Discussion
Traditionally, P hominis has been considered commensal, being mentioned in cases of asymptomatic infections in cats,20 and clinical signs such as diarrhea would be caused by an opportunistic overgrowth of endogenous fauna, favoring multiplication of the parasite.21–23
Some authors have reported that P hominis was only detected in fecal samples that were also positive for T foetus infection.19 However, researchers now suggest that there is evidence that P hominis may cause gastrointestinal disorders and that the occurrence of this infection is probably underestimated because it is misidentified as T foetus.24
In our study, the two cats presenting with chronic diarrhea were diagnosed with P hominis, and not T foetus, as was suspected. We believe that the retroviral status of the cats was a possible explanation for the clinical manifestations of these animals that were parasitized by P hominis, as FeLV is known to be an immunosuppressive disease.
In this sense, P hominis infection should be considered as a differential diagnosis in immunocompromised cats with chronic diarrhea, and correct identification of the trichomonad involved is essential.
Diagnostic techniques such as direct examination and fecal culture do not differentiate between P hominis and T foetus, and thus the diagnosis should be given as a trichomonad infection. Characteristics such as the numbers of anterior flagella and axostyle morphology are not clearly detected in living, motile specimens.5,20
Therefore, the adoption of molecular tools, such as PCR and DNA sequencing, is indispensable to confirm the species of trichomonad. Additionally, the combination of the three techniques allowed an accurate diagnosis of P hominis in these two cats with diarrhea.
Before the diagnosis of trichomonad infection, the cats had been unsuccessfully treated several times with metronidazole, which is considered the drug of choice for the treatment of trichomoniasis caused by P hominis; this suggests that the parasite may have developed resistance to this compound or that the owner did not administer the medication properly. Further studies are required to prove P hominis pathogenicity and its resistance to metronidazole.
Conclusions
Our findings support the idea that, although it is considered a commensal parasite, P hominis infection should be evaluated as a differential diagnosis in immunocompromised cats with chronic diarrhea. More detailed epidemiological studies of this infection should be conducted, which would clarify its pathogenicity and treatment.
Acknowledgments
The authors are grateful to the Genomic Platform - DNA Sequencing - RPT01A (Rede de Plataformas Tecnológicas Fiocruz). We also thank Professor Dr Vera Lucia de Jesus and Caroline Cunha Carneiro, from Universidade Federal Rural do Rio de Janeiro (UFRRJ) for their attention and assistance in preparing the culture medium.
Footnotes
Accepted: 7 April 2018
Conflict of interest: The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The authors received support from Fundação Capes, Fundação Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Proppi of Universidade Federal Fluminense.
References
- 1. Cook AK. Feline infectious diarrhea. Top Comp Anim Med 2008; 23: 169–176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Romatowski J. An uncommon protozoan parasite (Pentatrichomonas hominis) associated with colitis in three cats. Feline Pract 1996, 24: 10–14. [Google Scholar]
- 3. Gookin JL, Levy MG, Gager RB, et al. Diarrhea associated with trichomonosis in cats. J Am Vet Med Assoc 1999; 215: 1450–1454. [PubMed] [Google Scholar]
- 4. Romatowski J. Pentatrichomonas hominis infection in four kittens. J Am Vet Med Assoc 2000; 216: 1270–1272. [DOI] [PubMed] [Google Scholar]
- 5. Gookin JL, Levy MG, Law JM, et al. Experimental infection of cats with Tritrichomonas foetus. Am J Vet Res 2001; 62: 1690–1697. [DOI] [PubMed] [Google Scholar]
- 6. Kim Y, Kim H, Lee S. PCR detection and molecular characterization of Pentatrichomonas hominis from feces of dogs with diarrhea in the Republic of Korea. Korean J Parasitol 2010; 48: 9–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Wenrich DH, Emmerson MA. Studies on the morphology of Tritrichomonas foetus (Riedmüller) (Protozoa, Flagellata) from American cows. J Morphol 1944; 55: 193–205. [Google Scholar]
- 8. Jongwutiwes S, Silachamroon U, Putaporntip C. Pentatrichomonas hominis in empyema thoracis. T Roy Soc Trop Med H 2000; 94: 185–186. [DOI] [PubMed] [Google Scholar]
- 9. Meloni D, Mantini C, Goustille J, et al. Molecular identification of Pentatrichomonas hominis in two patients with gastrointestinal symptoms. J Clin Pathol 2011; 64: 933–935. [DOI] [PubMed] [Google Scholar]
- 10. Comparoé C, Lekpa FK, Nebie L, et al. Pentatrichomonas hominis infection in rheumatoid arthritis treated with adalimumab. Rheumatol 2013; 52: 1534–1535. [DOI] [PubMed] [Google Scholar]
- 11. Farmer MA. Ultrastructure of Ditrichomonas honigbergii ng, n sp (Parabasalia) and its relationships to amitochondrial protists. J Eukaryot Microbiol 1993; 40: 619–626. [Google Scholar]
- 12. Levy MG, Gookin JL, Poore M, et al. Tritrichomonas foetus and not Pentatrichomonas hominis is the etiologic agent of feline trichomonal diarrhea. J Parasitol 2003; 89: 99–104. [DOI] [PubMed] [Google Scholar]
- 13. Gookin JL, Birkenheuer AJ, Breitschwerdt EB, et al. Single-tube nested PCR for diagnosis of Tritrichomonas foetus in feline feces. J Vet Clin Microbiol 2002; 40: 4126–4130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Gookin JL, Foster DM, Poore MF, et al. Use of a commercially available culture system for diagnosis of Tritrichomonas foetus infection in cats. J Am Vet Med Assoc 2003; 222: 1–4. [DOI] [PubMed] [Google Scholar]
- 15. Ceplecha V, Svoboda M, Cepicka I, et al. InPouch TF-Feline medium is not specific for Tritrichomonas foetus. Vet Parasitol 2013; 196: 503–505. [DOI] [PubMed] [Google Scholar]
- 16. Gookin JL, Stebbins ME, Hunt E. Prevalence of and risk factors for feline Tritrichomonas foetus and Giardia infection. J Clin Microbiol 2004; 42: 2707–2710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Gookin JL, Copple CN, Papich MG. Efficacy of ronidazole for treatment of feline Tritrichomonas foetus infection. J Vet Intern Med 2006; 20: 536–543. [DOI] [PubMed] [Google Scholar]
- 18. Gookin JL, Birkenheuer AJ, St John V, et al. Molecular characterization of trichomonads from feces of dogs with diarrhoea. J Parasitol 2005; 91: 939–943. [DOI] [PubMed] [Google Scholar]
- 19. Gookin JL, Stauffer SH, Levy MG. Identification of Pentatrichomonas hominis in feline fecal samples by polymerase chain reaction assay. Vet Parasitol 2007; 145: 11–15. [DOI] [PubMed] [Google Scholar]
- 20. Santos CS, Jesus VL, McIntosh D, et al. Co-infection by Tritrichomonas foetus and Pentatrichomonas hominis in asymptomatic cats. Pesq Vet Bras 2015; 35: 980–988. [Google Scholar]
- 21. Dimski DS. Helminth and noncoccidial protozoan parasites of the gastrointestinal tract. In: Sherding RG. (ed). The cat: diseases and clinical management. 2nd ed. New York: Churchill Livingstone, 1989, pp 459–477. [Google Scholar]
- 22. Burrows CF, Batt RM, Sherding RG. Diseases of the small intestine. In: Ettinger SJ, Feldman EC. (eds). Textbook of veterinary internal medicine. 4th ed. Philadelphia, PA: WB Saunders, 1995, pp 1169–1232. [Google Scholar]
- 23. Barr SC. Enteric protozoal infections. In: Greene CE. (ed). Infectious diseases of the dog and cat. 2nd ed. Philadelphia, PA: WB Saunders, 1998, pp 482–491. [Google Scholar]
- 24. Santos CS, McIntosh D, Berto BP, et al. Diagnosis of Pentatrichomonas hominis from domestic cats in Southeastern Brazil. Pesq Bras Med Vet 2015; 37: 25–31. [Google Scholar]


