Abstract
Gynecological tumors, including endometrial, cervical and ovarian cancer, have increased in incidence over time. The widespread introduction of screening programs and advances in diagnostic imaging methods has lead to a progressive increase in gynecological cancer detection. Accurate diagnosis and proper monitoring of disease remain the primary target for a successful treatment. In the last years, knowledge about cancer biomarkers has considerably increased providing great opportunities for improving cancer detection and treatment. In addition, in the last few years there has been an important development of imaging techniques. Nowadays, a multimodal approach including the evaluation of serum tumor biomarkers combined with imaging techniques, seems to be the best strategy for assessing tumor presence, spread, recurrence, and/or the response to treatment in female cancer patients In this review we provide an overview of the application of biomarkers combined with novel imaging methods and highlight their roles in female cancer diagnosis and follow-up.
Keywords: Tumor markers, Imaging, Endometrial cancer, Ovarian cancer, Cervical cancer
Introduction
Gynecological tumors, including endometrial, cervical and ovarian cancer, have increased in incidence over time. Accurate diagnosis and proper monitoring of disease still represent the primary target for a successful treatment in this group of patients. Currently a multimodal approach, combining the evaluation of serum tumor biomarkers with imaging techniques, seems the best strategy for assessing tumor presence, recurrence, and/or the response to treatment in female cancer patients (Ferlay et al., 2015). Currently Oncology research is directed to the development of noninvasive methods to detect and monitor tumors and, in the last years, knowledge about cancer biomarkers has considerably increased (Wagner et al., 2012). Great attention has been paid to identify biological markers to be used as indicators of disease activity but also as prognostic and predictive factors of survival, recurrence, and treatment response in female patients (Binder et al., 2015). In addition, in the last few years there has been an important development of imaging techniques which has led to a significant improvement in tumor staging, monitoring and early detection of disease relapses.
The final goal is to achieve a more personalized management based not only on tumor stage but also on the molecular tumor features, to improve patients care combining information provided by imaging and tumor markers.
The association of tumor markers assessment with different imaging techniques can lead to several advantage allowing to localize the sites of the primary or secondary lesions, while tumor markers alone provide only a general indication of disease presence and in addition to confirm disease relapses, possibly before the related clinical symptoms.
In this review we provide an overview of the application of biomarkers combined with novel imaging methods and highlight their roles in female cancer diagnosis and follow-up.
Endometrial Cancer
Endometrial cancer is the most common gynecologic malignancy in Western countries, with an estimated annually incidence of 25.1 new cases per 100. 000 women (Siegel et al., 2015).
Endometrial cancer is more frequent in postmenopausal women, and is often associated with obesity, nulliparity, anovulation, diabetes and hypertension (Soliman et al., 2005).
Thanks to the early symptoms (about 90% of patients experience abnormal uterine bleeding and vaginal discharge), diagnosis is often made at early stage (FIGO stage I or II), with tumor still confined to the uterine corpus in around 75 % of cases. The 5-year overall survival is estimated around 80 % but it varies for different histological subtypes and tumor stages (Creasman et al., 2006).
However, after primary surgery, around 15–20 % of these tumors recur in the vagina/pelvis or at distant sites . In the metastatic disease, the prognosis is poor with reported median survival of only 7–12 month (Oza et al., 2011).
Materials and Methods
Endometrial cancer imaging
Modern imaging tools play an important role in the preoperative assessment of disease extent allowing the proper therapeutic approach for each patient, but also in the post-operative setting, for the early detection of tumor relapses. The clinical challenge is to select patients with high risk of relapses, who undergo to more extensive surgery, avoiding overtreatment in low-risk patients.
Ultrasound examination (US) with endovaginal approach (TVU) is the first tool of investigation to evaluate the endometrial thickening and to select the patients undergoing to biopsy. Ortoft et al. found that the combination of the preoperative biopsy with ultrasound achieved an accuracy of about 72–83 % in the diagnosis of high-risk endometrial cancer (Ortoft et al., 2013).
Several Authors validated the high diagnostic performance of TVU for the local staging and particularly to assess the degree of myometrial infiltration (reported Sensitivity 71–85 %, Specificity 72–84 %, Accuracy 92–94 %), to evaluate the extension to cervical region (Sensitivity 72–90 %, Specificitiy 29–93 % and Accuracy 78–92 %) and in assessing invasion of the serosa (Sensitivitity 67%, Specificity 95% and Accuracy 95%) (Alcazar et al., 2015; Fischerova et al., 2014).
More recently the employment of three-dimensional (3D) ultrasound technology has been introduced in the clinical practice allowing to acquire images of an entire volume from an organ or tissue of interest that may be reconstructed in different planes (Ong et al., 2016).
Alcazar et al., (2009) in their series of 96 patients reported a good diagnostic values of 3DUS (sensitivity 93 %, specificity 82% and accuracy 85 %) in detecting myometrial invasion.
Magnetic Resonance Imaging (MRI) is considered the best method to investigate pelvic female thanks to the multi-parametric capabilities of the method.
In the preoperative staging of endometrial cancer, particularly for the assessment of loco-regional disease spread, MRI allows to detect deep myometrial invasion (Se 33–100 %, Spe 44–100 %, Accuracy 58–100 %) cervical stroma invasion (Se 33–69 %, Spe 82–96 %, Accuracy 46–89 %) and metastatic lymph nodes (Se 17–80 % Spec 88–100 %, Accuracy 83–93 %.) (Haldorsen et al., 2012a; Haldorsen et al., 2012b; Rechichi et al., 2010).
Contrast-Enhancement Multi Detector Computed Tomography (CE-MDCT) is widely employed preoperatively for the detection of lymph node metastases and distant spread in endometrial cancer (Pecorelli et al., 2009).
An important challenge is represented by the lymph nodes involvement. The ability to correctly identify metastatic lymph nodes depends on lymph node size; in fact sensitivity varies with nodes diameter (100, 67, and 13 % in metastatic nodes ≥10, 5–9, and ≤4 mm, respectively).
Positron Emission Tomography-CT (PET-CT) is a valid diagnostic tool in this field combining two imaging methods and visualizing both morphologic and metabolic tumor characteristics at the same time and showing high values of sensitivity, specificity, PPV, NPV, and accuracy in predicting lymph node metastases (74%, 93%, 59%, 96%, and 91% respectively) (Antonsen et al., 2013; Kitajima et al., 2015).
The imaging follow-up of patients treated for endometrial cancer is still controversial. Most relapses tend to occur in high risk patients within 3 years, the commonest sites being lymph nodes and vaginal vault (Sohaib et al., 2007).
The most appropriate follow-up strategy is likely one based upon the risk of recurrence and the natural history of the disease.
US and MRI are usually employed to local tumor relapse evaluation while MDCT is essential for the assessment of lymph-node status and distance metastasis with an overall accuracy of 92%
PET with contrast-enhanced CT is an accurate imaging modality for the assessment of uterine cancer recurrence and can also change the treatment planning by localizing the recurrent lesions.
A study showed that in the detection of recurrence FDG-PET, implemented by CT and/or MRI, performed better (sensitivity 100%, specificity 88.2%, and accuracy 93.3%) than CT and/or MRI alone (sensitivity 84.6%, specificity 85.7%, and accuracy 85%) (Kitajima et al., 2010; Park et al., 2008).
Results
Endometrial cancer biomarkers
The use of tumor markers in the diagnosis of endometrial cancer is limited by their low specificity. In the last years, many research group performed various studies aiming to evaluate the diagnostic performance of serum tumor biomarkers and to find novel neoplastic indicators for the management of endometrial cancer .
CA125 is routinely evaluated at the diagnosis and follow-up of endometrial cancer. Elevated serum levels of CA125 are associated with some clinicopathologic characteristics (local tumor spread, enlarged lymph-nodes, carcinomatosis implants and metastases) and poor prognosis for this malignancy (Chung et al., 2006). However, in the early or very early stages of the disease the sensitivity of CA125 is poor, therefore the measurement of CA125 for endometrial cancer diagnosis is recommended in advanced disease (Santala et al., 2003).
Ca 19-9 tumor marker can be found elevated is elevated in up to 35% of patients with endometrial cancer.
A previous study of Cerchi et al. demonstrated that the combination of CA 125 and CA 19.9 offered interesting results for post-treatment surveillance showing a high sensitivity (83.3%) for the detection of recurrence, with only 12.8% of false positive cases (Cherchi et al., 1999).
In addition Ca 19-9 was found to correlate with shortened survival in univariate analyses, but no significance could be established in multivariate analyses (Lo et al., 1997).
However several studies demonstrated that CA19-9 has limited prognostic value and is not usually part of routine examinations.
Loco-regional tumor spread is a negative prognostic factor for all endometrial cancer types in terms of disease free survival and mortality. Nevertheless Chen et al., (2011) suggested that elevated preoperative CA 125 level of endometrial cancer patients correlated with the presence of lympho-vascular invasion suggesting that pre-operative CA125 levels may be considered a useful prognostic tool in endometrial cancer management.
Recently an increasing interest has been reported in literature on human epididymis protein 4 (HE4) demonstrating to be a useful clinical marker with high sensitivity and specificity even at early stage of endometrial cancer .
Moore et al., (2012) first reported elevated serum HE4 levels in all stages of disease, with a better sensitivity compared to CA125 also in the early stages . In addition HE4 levels seems to be related to the disease stage (Steffensen et al., 2011; Huhtinen et al., 2009).
In contrast, Ylmaz et al., (2016) contradicted the previous results of Moore’s study, founding no significant relation between HE-4 level and the tumor stage and suggesting a better sensitivity and specificity in terms of diagnosis with the combined use of HE4 and CA125. In addition in this paper no statistical relation between preoperative CA 125 and HE4 levels and the depth of the myometrial invasion detected at MR imaging was reported.
However, Saarelainen et al., (2013) described a statistically significant correlation between HE4 levels and metastases (p = 0.001), deep myometrial invasion (p < 0.001), and histologic grade (p = 0.012).
According to international guidelines, in presence of deep MI, lymphadenectomy must be performed for the increased risk of lymph node metastasis.
Despite the correlation between HE4 and lymph node involvement remains controversial, its measurement could afford additional tool for the choice of lymphadenectomy, improving overall patient management (Zanotti et al., 2012; Angioli et al., 2013) (Figure 1).
Figure 1.

Endometrial Cancer Checklist, a Proposal for a Correct Patients Diagnosis, Staging and Follow-up. TVU, Transvaginal Ultrasound; MRI, Magnetic Resonance Imaging; CECT, Constrast Enhanced Computed Tomography; PET/CT, Positron Emission Tomography - Computed Tomography.
Ovarian Cancer
Though rare, accounting for more than 3% of all cancers in women, epithelial ovarian cancer (EOC) is the leading cause of gynecological cancer related death (Siegel et al., 2016).
More than 70 % of patients present advanced tumor stage at diagnosis with widespread intra-peritoneal or distant metastases due to the lack of early symptoms to guide timely image-driven investigations (Modugno et al., 2012; Marcus et al., 2014). The proper choice of treatment for ovarian cancer depends on accurate staging, in order to distinguish patients for whom primary surgery is unlikely to be successful.
Standard treatment includes aggressive surgery followed by platinum- and taxane-based chemotherapy. Patients with not completely resectable disease undergo to neoadjuvant chemotherapy followed by interval surgery.
Ovarian cancer imaging
Characterize the nature of the ovarian masses is the first goal of diagnostic imaging in the clinical setting of EOC.
TVU is the primary imaging technique in ovarian cancer especially if carried out by an experienced sonographer allowing an accurate characterization of about 90% of adnexal masses.
Morphology on TVU combined with Doppler waveform analysis are the most important features to be considered for an accurate risk assessment of adnexal lesions (Cramer et al., 2011).
In addition, TVU is considered the best tool for ovarian cancer screening in patients at increased risk for ovarian carcinoma based either on their genetic profile or on serum markers (Manganaro et al., 2012a; Manganaro et al., 2012b).
The IOTA group proposed a mathematical model, the so-called ADNEX (the assessment of different neoplasia in the adnexa), to predict the risk that an ovarian mass is benign, borderline, stage I, stages II-IV or metastatic (Spencer et al., 2010).
The reported diagnostic performance of ADNEX in differentiating between benign and malignant masses was 0.937 (95% CI: 0.915-0.954) when CA125 was included, and 0.925 (95% CI: 0.902-0.943) when CA125 was excluded (Freydanck et al., 2012) .
According to the literature about 8% of adnexal masses detected by sonography remains indeterminate at adnexal sonographic investigation.
In these cases Magnetic Resonance Imaging (MRI) has maximal diagnostic and therapeutic impact resolving most of these uncertainties.
The staging of EOC include the evaluation peritoneal dissemination, lymph-node involvement and parenchymal metastasis . Currently CT or MRI are used in this context .
Computed Tomography, is the imaging modality of choice for staging ovarian cancer but also for treatment follow-up, according to the European Society of Urogenital Radiology and the American College of Radiology guidelines for ovarian cancer staging and follow up, ensuring reproducibility of the results for future comparison in a short examination time with a reported accuracy of about 92%-97% (Alt, 2011; Hynninen et al., 2013).
In particular, the development of multidetector CT (MDCT) has improved the diagnostic ability of this imaging technique to depict s more clearly and directly intra-abdominal lesion.
MDCT has the advantages of having thinner sections and multiplanar reformations, providing more accurate preoperative staging and planning.
MRI, thanks to the higher soft-tissue contrast has some advantages for the evaluation of the local tumor spread the pelvis, and can be used in situations where CT is contraindicated.
In addition DCE-MRI is very accurate in detecting recurrent peritoneal ovarian cancer implants showing values of Sensitivity (90%) and specificity (100%). However, in case of small and diffuse implants of carcinomatosis (<0.5 cm) and lymph-nodes with largest diameter less than 1 cm, CT and MRI could underestimate disease extent.
PET/CT has improved the accuracy of staging ovarian carcinoma (Hynninen et al., 2013). However its role in EOC staging is still controversial while it is considered more accurate than CT alone in detecting tumor recurrence (sensitivity = 95–97%, specificity = 80–100%) (Addley et al., 2010). Furthermore, integrated FDG PET/CT is useful specifically in optimizing the treatment plan and it might play an important role in treatment stratification in the future (Lee et al., 2015).
Ovarian cancer biomarkers
The current reference biomarker for ovarian cancer detection is CA125 which it is considered also a prognostic tool for the detection of disease recurrence after surgery and adjuvant chemotherapy (Bilici et al., 2010; Engelen et al., 2006; Sturgeon et al., 2011; Anastasi et al., 2013).
However, CA125 antigen is not elevated in most primary ovarian mucinous neoplasms . In primary ovarian mucinous tumors, CA19-9 antigen is used as a marker instead (Cho et al., 2014).
Terzic et al., (2014) in their series including 358 consecutive patients found that the positive rate of Ca 125, Ca 19-9, Ca 15-3, and CEA in serous tumors was 57.9, 7.9, 7.9 and 15.8%, respectively while for mucinous tumors was 31.8, 40.9, 27.3 and 40.9 %.
In the last few years human epididymis protein 4 (HE4) has emerged as an important biomarker in differential diagnosis, early detection, prognosis and monitoring the response to chemotherapy of EOC. HE4 is commonly overexpressed both in the early stage and in the recurrence of disease and it has been demonstrated that HE4 serum levels are not affected by the menstrual cycle, oral contraceptive use or endometriosis (Granato et al., 2012; Anastasi et al., 2010a; Anastasi et al., 2010; Granato et al., 2015).
The suggestion that HE4 is a good indicator for the remission from the disease was recently reported by follow-up studies, in which it was shown that the values of HE4 correlated with the clinical response to treatment or remission from the disease, as documented by CT imaging (Midulla et al., 2012).
It is important to note that HE4 serum levels combined with MDCT may improve the monitoring management of women affected by ovarian cancer (Anastasi et al., 2017).
Recent investigations suggested also a potential role of Gal-3 in EOC carcinogenesis.
Interestingly, it has also been suggested a strong correlation between high levels of Gal-3 and MDCT imaging findings of disease progression in patients with recurrent EOC (Wu et al., 2014).
In the recent years, research has focused on new biomarkers such as mesothelin, inhibin, osteopontin and Ca72.4: the benefits of these markers remain unclear, and even if sensitivity is increased, specificity is sub-optimal (Walentowicz et al., 2014; Tilli et al., 2014).
The expression of mesothelin in ovarian tissue correlated to chemotherapy resistance and poor prognosis suggesting a role for mesothelin in diagnosis and disease staging (Tilli et al., 2014).
It has been validated that the alteration of the inhibin/activin pathway may contribute to the development of epithelial ovarian cancer and high levels of inibin A are associated to a poor prognosis and a low survival at 5 years (Tilli et al., 2011).
Osteopontin enhances proliferation, migration and invasion of ovarian cancer cells but the role of this glycoprotein to discriminate malignant and benign ovarian tumors has not been sufficiently elucidated (Tilli et al., 2014; Tilli et al., 2011).
Serum biomarker CA72.4 has also been shown to be elevated in EOC: it is slightly increased also in benign conditions but is not affected by pregnancy or the menstrual period (Lenhard et al., 2009).
However, according to the current guidelines measurement of serum CA125 antigen remains the gold standard in the follow-up EOC (Lenhard et al., 2009; Moore et al., 2012; Park et al., 2011) and some authors have demonstrated the important role of the biomarker CA72.4 combined with CA125 as a predictive factor of recurrence (Figure 2).
Figure 2.
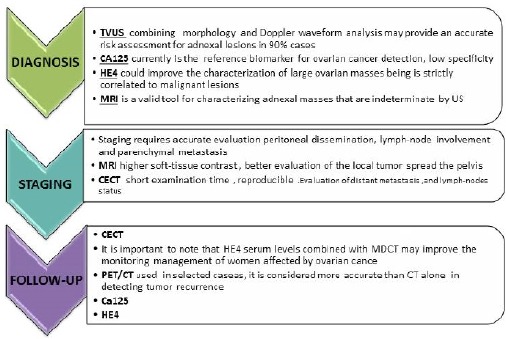
Ovarian Cancer Checklist, a Proposal for a Correct Patients Diagnosis, Staging and Follow-up. TVU, Transvaginal Ultrasound; MRI, Magnetic Resonance Imaging; CECT, Constrast Enhanced Computed Tomography; PET/CT, Positron Emission Tomography - Computed Tomography.
Discussion
Cervical Cancer
Cervical cancer is the third most common gynecologic malignancy and the fourth leading cause of death in women worldwide, with an estimated global incidence of 470,000 new cases per-year (Darlin et al., 2014). The major risk-factor for developing cervical cancer is infection with high-risk human papillomavirus that lead to over-expression of two oncogenes (E6 and E7) related to transformation of neoplastic cells (Cattani et al., 2009).
Although effective and prevention programs has been introduced about 30% of patients present an advanced stage at diagnosis and 30-50% of them relapse in the first 5-years after primary treatment (Wagner et al., 2013). Proper staging is essential because patients at an early stage of disease (stage IA, IB1, IIa1) can be treated surgically, while surgery remains controversial in patients with locally advanced cervical carcinoma (IIb and beyond), which are usually treated with neoadjuvant chemo-radiotherapy (Kato et al., 2015; Tangjitgamol et al., 2014).
Cervical cancer imaging
In 2009, the International Federation of Gynecology and Obstetrics undertook a review of the clinical staging of cervical cancer, which recommended the use of modern imaging methods in evaluating these significant prognostic factors (Kato et al., 2015). The American College of Radiology doesn’t recommend US for assessing the extent of cervical cancer (Siegel et al., 2012) However, recently, in Literature there is a crescent interest about the value of ultrasound examination for disease assessment (Chiappa et al., 2015) Fisherova et al., (2008) examinated 95 with proven cervical cancer and found an accuracy of US of 93.7% (95% CI 86.8 to 97.6), with a sensitivity of 93.4% (95% CI 85.3 to 97.8), and specificity of 94.7% (95% CI 73.9 to 99.8) in detecting tumor lesions.
Similar results were obtained by Testa et al (Accuracy 93%, 95% CI 84 to 97) who carried out TVU examination in 75 patients with early-stage disease and demonstrated that ultrasound was able to detect the presence of the tumor, deep stromal tumor invasion and infiltrated parametria (Testa et al., 2009).
However, US is not able to correctly assess lymph nodes involvement showing very low low accuract (arounding 38–43%.) if compared to other imaging methods(Pálsdóttir et al., 2015).
MRI is the most reliable imaging modality for staging, treatment planning and follow-up of cervical cancer with reported values of sensitivity up to 100% in evaluating the parametrial infiltration, the degree of longitudinal extension and the bladder and rectal infiltration with an overall diagnostic accuracy values of about 93% (Kraljević et al., 2013).
High resolution T2-weighted imaging is the mainstay for tumor detection. The high T2 signal of cervical lesions enables its differentiation from the normal cervical stroma that presents low T2 signal .
MRI is crucial for evaluating parametrial involvement (FIGO IB) that precludes surgical treatment, with reported accuracy values ranging from 88%-97%, significantly higher compared to clinical examination (Bourgioti et al., 2016).
More recently, DWI sequences has been implemented to MRI protocols for cervical cancer staging due to an excellent tissue contrast between neoplastic and non-neoplastic tissues . DWI are also able to distinguish residual tumor from fibrosis, especially in patients previous treated with chemo-radiotherapy (Kuang et al., 2015; Bollineni et al. 2015). Park et al., (2014)demonstrated that tumor ADC on MRI was an independent predictors of pathologic parametrial infiltration.
It has also been suggested that the ADC values may reflect the biologic heterogeneity of tumors thus providing information regarding its behavior, subtype characterization and cancer treatment response. In addition Nakamura et al., (2012) showed that ADC values were an independent prognostic factor for disease free survival after radical hysterectomy.
More recently, several studies emphasized the importance of MRI in predicting response to neoadjuvant chemotherapy. Indeed the reconstitution of the normal signal hypointensity of stromal ring, on T2-dependent images, combined with DWI features are the most important signs of macroscopic response to treatment with a negative predictive value of 97% (Addley et al., 2010).
PET-CT has been recently introduced into clinical practice and has been demonstrated to be useful in the staging of cervical cancer and superior to either CT or MRI in the detection of nodal disease. Particuarly, a recent meta-analysis based on 67 Studies showed that PET or PET/CT has the highest specificity, and DWI-MRI has the highest sensitivity in detecting the lymph nodes metastases in patients with cervical Cancer (AUC of DWI-MRI 0.92 and PET or PET/CT 0.90 compared with CT 0.83) (Liu et al., 2017). Surveillance of women previously treated for cervical cancer remains problematic. PET-CT is recommanded in selected cases as surveillance method after treatment for advanced-stage cervical cancer with reported value of sensitivity and specificity of 91% and 94%, respectively (Ding et al., 2014).
Cervical cancer biomarkers
Today there are not validated biomarkers for the diagnosis, prognosis and follow-up of patients affected by cervical cancer. Increased serum squamous cell carcinoma antigen (SCC-Ag) levels have been observed in the majority of cervical squamous cell carcinomas. The SCC-Ag is normally expressed by basal and parabasal layers of normal squamous epithelium, but is found to be overexpressed in epithelia of cancerous tissue including cervical cancer (Kato et al., 1996).
The clinical relevance of SCC-Ag in the management of cervical cancer is still controversial. According to some authors SCC evaluation during the follow-up do not improve the early detection of the recurrence . By contrary, some authors described that increased SCC-Ag levels have been shown to be related to the stage of the disease, size of the tumor, depth of the stromal, lymphoadenopaties (Gaarenstroom et al., 2000).
Some authors demonstrated a linear correlation between CEA and stage of the disease suggesting its prognostic value in cervical cancer (Borras et al., 1995).
Keratins, formerly known as cytokeratins, are the major epithelial-specific subgroup of intermediate filament proteins. CYFRA21-1, a fragment of cytokeratin- 19 is expressed in normal epithelium and in carcinomas of the uterine cervix. To date, a number of studies have investigated the diagnostic accuracy of CYFRA21-1 for cervix cancer, but the results have been controversial. Suzuki et al. have been demonstrated a correlation between high Cyfra 21-1 levels and stage or recurrence of the disease, suggesting the use of this biomarker for the monitoring the outcome of patients affected by cervical cancer (Suzuki et al., 2000).
By contrary, Gaarenstroom et al., (2000) reported that increased CYFRA21-1 levels were strongly related to tumor burden, but insufficiently reliable for identifying patients at risk of the presence of lymph node metastases or parametrial involvement. In summary SCC-Ag, CEA and Cyfra 21-1 cannot be considered good markers for prognosis and follow-up of cervical cancer, more accurate and reliable diagnostic methods/biomarkers should be discovered (Figure 3).
Figure 3.

Cervical Cancer Disease Progression and Diagnostic Workup. US, Ultrasound; MRI, Magnetic Resonance Imaging; CECT, Constrast Enhanced Computed Tomography; PET/CT, Positron Emission Tomography - Computed Tomography.
Vulvar Cancer
Vulvar cancer (VC) is a rare malignancy accounting for about 5% of cancers of the female genital tract and most often occurs in older women (Hiniker et al., 2013).
About 90% of vulvar carcinomas are squamous cell cancers and lesions are multifocal in about 5% of cases. The labia majora are involved in about 50% of cases followed by labia minor, mons pubis, clitoris, Bartholin glands, and perineum (Barton et al., 2003).
The overall incidence of vulvar cancer has risen over the last decade, probably because of an increase in human papilloma virus (HPV) infections (Viens et al., 2016)
Vulvar cancer imaging
The diagnosis of vulvar carcinoma is usually made by clinical examination. Imaging plays an important role in management of vulvar carcinoma, especially locally advanced disease.
US is often used to perform image-guided biopsy procedures.
MRI is crucial for locoregional staging allowing to assess the involvement of adjacent structures (e.g., the anal sphincter, urethra, or vaginal wall) and pelvic nodal involvement thus helping to accurately plan the extent of surgery. In addition MRI can help in evaluating the extent of the local recurrence occurring
In about 30–50% of patients within the first 2 years (Griffin et al., 2008).
Vulvar carcinoma spreads primarily by local extension via the lymphatic system while hematogenous spread is uncommon. The lymph node status has been identified as the most important prognostic factor in patients with vulvar cancer (Gonzalez et al., 2007).
CT provides information on the presence of pelvic lymphadenopathy and distant metastases with a sensitivity and specificity of 58% and 75%, respectively (Land et al., 2006).
Cohn et al., (2002) evaluating the role of PET/CT in detecting groin lymphadenopathy in locally advanced vulvar cancer, before nodal dissection and found a sensitivity of 80%, specificity of 90%, positive predictive value of 80%, and negative predictive value of 90%.
Vulvar cancer biomarkers
Few biological markers have demonstrated clinical value for the management of vulvar cancer. Previous studies demonstrated that HPV and the surrogate biomarker p16 are associated with a less aggressive behaviour of vulvar cancer., while p53 positivity seems to be related with poor prognosis and significantly increased recurrence (Hay et al., 2016).
In conclusion several markers are currently employed in oncology practice as indicators of particular tumor types; some of them being strictly tissue-specific. However, despite the high numbers of promising biomarkers proposed, none of these, used alone, achieve high diagnostic accuracy values. Recently imaging research has been focused on the possibility to determine functional tissue characteristics thus changing the diagnostic approach of radiologists which was previously based on morphologic and macroscopic tumor features thanks to different imaging methods such as DWI and PET. DWI has improved the diagnostic accuracy of conventional magnetic resonance imaging, providing useful information about tumor cellularity while PET has been introduced in the diagnostic setting for its ability to assess tumor metabolism.
In conclusion despite these advances in imaging techniques and the discovery of new biological markers have provided significant improvement in the care of female cancer patients, further advancements are needed to improve patient care., a multimodal approach including the evaluation of serum tumor biomarkers combined with imaging techniques, seems to be the best strategy for assessing tumor presence, spread, recurrence, and/or the response to treatment in female cancer patients.
References
- Addley HC, Vargas HA, Moyle PL, et al. Pelvic imaging following chemotherapy and radiation therapy for gynecologic malignancies. RadioGraphics. 2010;30:1843–56. doi: 10.1148/rg.307105063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alcazar JL, Pineda L, Martinez-Astorquiza Corral T, et al. Transvaginal/transrectal ultrasound for assessing myometrial invasion in endometrial cancer:a comparison of six different approaches. J Gynecol Oncol. 2015a;26:201–7. doi: 10.3802/jgo.2015.26.3.201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alcázar JL, Galván R, Albela S, et al. Assessing myometrial infiltration by endometrial cancer:uterine virtual navigation with three-dimensional US. Radiology. 2009;250:776–83. doi: 10.1148/radiol.2503080877. [DOI] [PubMed] [Google Scholar]
- Alt CD, Brocker KA, Eichbaum M, et al. Imaging of female pelvic malignancies regarding MRI, CT, and PET/CT:Part 2. Strahlenther Oncol. 2011;187:705–14. doi: 10.1007/s00066-011-4002-z. [DOI] [PubMed] [Google Scholar]
- Anastasi E, Marchei GG, Viggiani V, et al. HE4:A new potential early biomarker for the recurrence of ovarian cancer. Tumor Biol. 2010a;31:113–9. doi: 10.1007/s13277-009-0015-y. [DOI] [PubMed] [Google Scholar]
- Anastasi E, Granato T, Marchei GG, et al. Ovarian tumor marker HE4 is differently expressed during the phases of the menstrual cycle in healthy young women. Tumour Biol. 2010b;31:411–5. doi: 10.1007/s13277-010-0049-1. [DOI] [PubMed] [Google Scholar]
- Anastasi E, Manganaro L, Granato T, et al. Is CA72-4 a useful biomarker in differential diagnosis between ovarian endometrioma and epithelial ovarian cancer? Dis Markers. 2013;35:331–5. doi: 10.1155/2013/984641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anastasi D, Gigli S, Santulli M, et al. Role of galectin-3 combined with multi-detector contrast enhanced computed tomography in predicting disease recurrence in patients with ovarian cancer. Asian Pac J Cancer Prev. 2017;18:1277–82. doi: 10.22034/APJCP.2017.18.5.1277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Angioli R, Plotti F, Capriglione S, et al. The role of novel biomarker HE4 in endometrial cancer:a case control prospective study. Tumour Biol. 2013;34:571–6. doi: 10.1007/s13277-012-0583-0. [DOI] [PubMed] [Google Scholar]
- Antonsen SL, Jensen LN, Loft A, et al. MRI, PET/CT and ultrasound in the preoperative staging of endometrial cancer - a multicenter prospective comparative study. Gynecol Oncol. 2013;128:300–8. doi: 10.1016/j.ygyno.2012.11.025. [DOI] [PubMed] [Google Scholar]
- Barton DP. The prevention and management of treatment related morbidity in vulval cancer. Best Pract Res Clin Obstet Gynaecol. 2003;17:683–701. doi: 10.1016/s1521-6934(03)00045-2. [DOI] [PubMed] [Google Scholar]
- Bilici A, Ustaalioglu BBO, Seker M, et al. Clinical value of FDG PET/CT in the diagnosis of suspected recurrent ovarian cancer:is there an impact of FDG PET/CT on patient management? Eur J Nucl Med Mol Imaging. 2010;37:1259–69. doi: 10.1007/s00259-010-1416-2. [DOI] [PubMed] [Google Scholar]
- Binder PS, Prat J, Mutch DG. Molecular staging of gynecological cancer:What is the future? Best Pract Res Clin Obstet Gynaecol. 2015;29:776–89. doi: 10.1016/j.bpobgyn.2015.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bollineni VR, Kramer G, Liu Y, et al. A literature review of the association between diffusion-weighted MRI derived apparent diffusion coefficient and tumour aggressiveness in pelvic cancer. Cancer Treat Rev. 2015;41:496–502. doi: 10.1016/j.ctrv.2015.03.010. [DOI] [PubMed] [Google Scholar]
- Borras G, Molina R, Xercavins J, et al. Tumor antigens CA 19.9, CA 125, and CEA in carcinoma of the uterine cervix. Gynecol Oncol. 1995;57:205–11. doi: 10.1006/gyno.1995.1126. [DOI] [PubMed] [Google Scholar]
- Bourgioti C, Chatoupis K, Rodolakis A, et al. Incremental prognostic value of MRI in the staging of early cervical cancer:a prospective study and review of the literature. Clin Imaging. 2016;40:72–8. doi: 10.1016/j.clinimag.2015.09.012. [DOI] [PubMed] [Google Scholar]
- Cattani P, Zannoni GF, Ricci C, et al. Clinical performance of human papillomavirus E6 and E7 mRNA testing for high-grade lesions of the cervix. J Clin Microbiol. 2009;47:3895–901. doi: 10.1128/JCM.01275-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen YL, Huang C-Y, Chien T-Y, et al. Value of pre-operative serum CA125 level for prediction of prognosis in patients with endometrial cancer. Aust New Zeal J Obstet Gynaecol. 2011;51:397–402. doi: 10.1111/j.1479-828X.2011.01325.x. [DOI] [PubMed] [Google Scholar]
- Cherchi PL, Dessole S, Ruiu GA, et al. The value of serum CA 125 and association CA 125/CA 19-9 in endometrial carcinoma. Eur J Gynaecol Oncol. 1999;20:315–7. [PubMed] [Google Scholar]
- Chiappa V, Di Legge A, Valentini AL, et al. Agreement of two-dimensional and three-dimensional transvaginal ultrasound with magnetic resonance imaging in assessment of parametrial infiltration in cervical cancer. Ultrasound Obstet Gynecol. 2015;45:459–69. doi: 10.1002/uog.14637. [DOI] [PubMed] [Google Scholar]
- Cho HY, Kyung MS. Serum CA19-9 as a predictor of malignancy in primary ovarian mucinous tumors:a matched case-control study. Med Sci Monit. 2014;20:1334–9. doi: 10.12659/MSM.890954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung HH, Kim JW, Park N-H, et al. Use of preoperative serum CA-125 levels for prediction of lymph node metastasis and prognosis in endometrial cancer. Acta Obstet Gynecol Scand. 2006;85:1501–5. doi: 10.1080/00016340601022777. [DOI] [PubMed] [Google Scholar]
- Cramer DW, Bast RC, Berg CD, et al. Ovarian cancer biomarker performance in prostate, lung, colorectal, and ovarian cancer screening trial specimens. Cancer Prev Res. 2011;4:365–74. doi: 10.1158/1940-6207.CAPR-10-0195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohn DE, Dehdashti F, Gibb RK, et al. Prospective evaluation of positron emission tomography for the detection of groin node metastases from vulvar cancer. Gynecol Oncol. 2002;85:179–84. doi: 10.1006/gyno.2002.6605. [DOI] [PubMed] [Google Scholar]
- Creasman WT, Odicino F, Maisonneuve P, et al. Carcinoma of the corpus uteri. FIGO 26th annual report on the results of treatment in gynecological cancer. Int J Gynaecol Obstet. 2006;95:105–43. doi: 10.1016/S0020-7292(06)60031-3. [DOI] [PubMed] [Google Scholar]
- Darlin L, Borgfeldt C, Widén E, Kannisto P. Elderly women above screening age diagnosed with cervical cancer have a worse prognosis. Anticancer Res. 2014;34:5147–51. [PubMed] [Google Scholar]
- Ding X-P, Feng L, Ma L. Diagnosis of recurrent uterine cervical cancer:PET versus PET/CT:a systematic review and meta-analysis. Arch Gynecol Obstet. 2014;290:741–7. doi: 10.1007/s00404-014-3263-z. [DOI] [PubMed] [Google Scholar]
- Engelen MJA, Kos HE, Willemse PHB, et al. Surgery by consultant gynecologic oncologists improves survival in patients with ovarian carcinoma. Cancer. 2006;106:589–98. doi: 10.1002/cncr.21616. [DOI] [PubMed] [Google Scholar]
- Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide:Sources, methods and major patterns in Globocan 2012. Int J Cancer. 2015;136:359–86. doi: 10.1002/ijc.29210. [DOI] [PubMed] [Google Scholar]
- Fischerova D, Cibula D, Stenhova H, et al. Transrectal ultrasound and magnetic resonance imaging in staging of early cervical cancer. Int J Gynecol Cancer. 2008;18:766–72. doi: 10.1111/j.1525-1438.2007.01072.x. [DOI] [PubMed] [Google Scholar]
- Fischerova D, Frühauf F, Zikan M, et al. Factors affecting sonographic preoperative local staging of endometrial cancer. Ultrasound Obstet Gynecol. 2014;43:575–85. doi: 10.1002/uog.13248. [DOI] [PubMed] [Google Scholar]
- Freydanck MK, Laubender RP, Rack B, et al. Two-marker combinations for preoperative discrimination of benign and malignant ovarian masses. Anticancer Res. 2012;32:2003–8. [PubMed] [Google Scholar]
- Gaarenstroom KN, Kenter GG, Bonfrer JM, et al. Can initial serum cyfra 21-1, SCC antigen, and TPA levels in squamous cell cervical cancer predict lymph node metastases or prognosis? Gynecol Oncol. 2000;77:164–70. doi: 10.1006/gyno.2000.5732. [DOI] [PubMed] [Google Scholar]
- Gonzalez Bosquet J, Magrina JF, Magtibay PM, et al. Patterns of inguinal groin metastases in squamous cell carcinoma of the vulva. Gynecol Oncol. 2007;105:742–6. doi: 10.1016/j.ygyno.2007.02.014. [DOI] [PubMed] [Google Scholar]
- Granato T, Midulla C, Longo F, et al. Role of HE4, CA72.4, and CA125 in monitoring ovarian cancer. Tumor Biol. 2012;33:1335–9. doi: 10.1007/s13277-012-0381-8. [DOI] [PubMed] [Google Scholar]
- Granato T, Porpora MG, Longo F, et al. HE4 in the differential diagnosis of ovarian masses. Clin Chim Acta. 2015;446:147–55. doi: 10.1016/j.cca.2015.03.047. [DOI] [PubMed] [Google Scholar]
- Griffin N, Grant LA, Sala E. Magnetic resonance imaging of vaginal and vulval pathology. Eur Radiol. 2008;18:1269–80. doi: 10.1007/s00330-008-0865-5. [DOI] [PubMed] [Google Scholar]
- Haldorsen IS, Husby JA, Werner HMJ, et al. Standard 1.5-T MRI of endometrial carcinomas:modest agreement between radiologists. Eur Radiol. 2012a;22:1601–11. doi: 10.1007/s00330-012-2400-y. [DOI] [PubMed] [Google Scholar]
- Haldorsen IS, Salvesen HB. Staging of endometrial carcinomas with MRI using traditional and novel MRI techniques. Clin Radiol. 2012b;67:2–12. doi: 10.1016/j.crad.2011.02.018. [DOI] [PubMed] [Google Scholar]
- Hay CM, Lachance JA, Lucas FL, et al. Biomarkers p16, HPV, and p53 Predict Recurrence and Survival in Early Stage Squamous Cell Carcinoma of the Vulva. J Low Genit Tract Dis. 2016;20:252–6. doi: 10.1097/LGT.0000000000000182. [DOI] [PubMed] [Google Scholar]
- Hiniker SM, Roux A, Murphy JD, et al. Primary squamous cell carcinoma of the vagina:prognostic factors, treatment patterns and outcomes. Gynecol Oncol. 2013;131:380–5. doi: 10.1016/j.ygyno.2013.08.012. [DOI] [PubMed] [Google Scholar]
- Huhtinen K, Suvitie P, Hiissa J, et al. Serum HE4 concentration differentiates malignant ovarian tumours from ovarian endometriotic cysts. Br J Cancer. 2009;100:1315–9. doi: 10.1038/sj.bjc.6605011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hynninen J, Kemppainen J, Lavonius M, et al. A prospective comparison of integrated FDG-PET/contrast-enhanced CT and contrast-enhanced CT for pretreatment imaging of advanced epithelial ovarian cancer. Gynecol Oncol. 2013;131:389–94. doi: 10.1016/j.ygyno.2013.08.023. [DOI] [PubMed] [Google Scholar]
- Kato T, Takashima A, Kasamatsu T, et al. Clinical tumor diameter and prognosis of patients with FIGO stage IB1 cervical cancer (JCOG0806-A) Gynecol Oncol. 2015;137:34–9. doi: 10.1016/j.ygyno.2015.01.548. [DOI] [PubMed] [Google Scholar]
- Kato H. Expression and function of squamous cell carcinoma antigen. Anticancer Res. 1996;16:2149–53. [PubMed] [Google Scholar]
- Kitajima K, Suzuki K, Nakamoto Y, et al. Low-dose non-enhanced CT versus full-dose contrast-enhanced CT in integrated PET/CT studies for the diagnosis of uterine cancer recurrence. Eur J Nucl Med Mol Imaging. 2010;37:1490–8. doi: 10.1007/s00259-010-1440-2. [DOI] [PubMed] [Google Scholar]
- Kitajima K, Suenaga Y, Ueno Y, et al. Preoperative risk stratification using metabolic parameters of (18)F-FDG PET/CT in patients with endometrial cancer. Eur J Nucl Med Mol Imaging. 2015;42:1268–75. doi: 10.1007/s00259-015-3037-2. [DOI] [PubMed] [Google Scholar]
- KraljevićZ ViskovićK, Ledinsky M, et al. Primary uterine cervical cancer:correlation of preoperative magnetic resonance imaging and clinical staging (FIGO) with histopathology findings. Coll Antropol. 2013;37:561–8. [PubMed] [Google Scholar]
- Kuang F, Yan Z, Li H, Feng H. Diagnostic accuracy of diffusion-weighted MRI for differentiation of cervical cancer and benign cervical lesions at 3.0T:Comparison with routine MRI and dynamic contrast-enhanced MRI. J Magn Reson Imaging. 2015;42:1094–9. doi: 10.1002/jmri.24894. [DOI] [PubMed] [Google Scholar]
- Land R, Herod J, Moskovic E, et al. Routine computerized tomography scanning, groin ultrasound with or without fine needle aspiration cytology in the surgical management of primary squamous cell carcinoma of the vulva. Int J Gynecol Cancer. 2006;16:312–7. doi: 10.1111/j.1525-1438.2006.00340.x. [DOI] [PubMed] [Google Scholar]
- Lee SI, Catalano OA, Dehdashti F. Evaluation of gynecologic cancer with MR imaging, 18F-FDG PET/CT, and PET/MR imaging. J Nucl Med. 2015;56:436–43. doi: 10.2967/jnumed.114.145011. [DOI] [PubMed] [Google Scholar]
- Lenhard MS, Nehring S, Nagel D, et al. Predictive value of CA 125 and CA 72-4 in ovarian borderline tumors. Clin Chem Lab Med. 2009;47:537–42. doi: 10.1515/CCLM.2009.134. [DOI] [PubMed] [Google Scholar]
- Liu B, Gao S, Li S. A comprehensive comparison of CT, MRI, positron emission tomography or positron emission tomography/CT, and diffusion weighted imaging-MRI for detecting the lymph nodes metastases in patients with cervical cancer:A meta-analysis based on 67 studies. Gynecol Obstet Invest. 2017;82:209–22. doi: 10.1159/000456006. [DOI] [PubMed] [Google Scholar]
- Lo SS, Cheng DK, Ng TY, et al. Prognostic significance of tumour markers in endometrial cancer. Tumour Biol. 1997;18:241–9. doi: 10.1159/000218037. [DOI] [PubMed] [Google Scholar]
- Manganaro L, Vittori G, Vinci V, et al. Beyond laparoscopy:3-T magnetic resonance imaging in the evaluation of posterior cul-de-sac obliteration. Magn Reson Imaging. 2012a;30:1432–8. doi: 10.1016/j.mri.2012.05.006. [DOI] [PubMed] [Google Scholar]
- Manganaro L, Fierro F, Tomei A, et al. Feasibility of 3.0T pelvic MR imaging in the evaluation of endometriosis. Eur J Radiol. 2012b;81:1381–7. doi: 10.1016/j.ejrad.2011.03.049. [DOI] [PubMed] [Google Scholar]
- Marcus CS, Maxwell GL, Darcy KM, et al. Current approaches and challenges in managing and monitoring treatment response in ovarian cancer. J Cancer. 2014;5:25–30. doi: 10.7150/jca.7810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Midulla C, Manganaro L, Longo F, et al. HE4 combined with MDCT imaging is a good marker in the evaluation of disease extension in advanced epithelial ovarian carcinoma. Tumor Biol. 2012;33:1291–8. doi: 10.1007/s13277-012-0376-5. [DOI] [PubMed] [Google Scholar]
- Modugno F, Edwards RP. Ovarian cancer:prevention, detection, and treatment of the disease and its recurrence. Molecular mechanisms and personalized medicine meeting report. Int J Gynecol Cancer. 2012;22:45–57. doi: 10.1097/IGC.0b013e31826bd1f2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore RG, Miller MC, Eklund EE, et al. Serum levels of the ovarian cancer biomarker HE4 are decreased in pregnancy and increase with age. Am J Obstet Gynecol. 2012;206:349. doi: 10.1016/j.ajog.2011.12.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura K, Joja I, Nagasaka T, et al. The mean apparent diffusion coefficient value (ADCmean) on primary cervical cancer is a predictive marker for disease recurrence. Gynecol Oncol. 2012;127:478–83. doi: 10.1016/j.ygyno.2012.07.123. [DOI] [PubMed] [Google Scholar]
- Ong CL. The current status of three-dimensional ultrasonography in gynaecology. Ultrason (Seoul, Korea) 2016;35:13–24. doi: 10.14366/usg.15043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ørtoft G, Dueholm M, Mathiesen O, et al. Preoperative staging of endometrial cancer using TVS, MRI, and hysteroscopy. Acta Obstet Gynecol Scand. 2013;92:536–45. doi: 10.1111/aogs.12103. [DOI] [PubMed] [Google Scholar]
- Oza AM, Elit L, Tsao M-S, et al. Phase II study of temsirolimus in women with recurrent or metastatic endometrial cancer:a trial of the NCIC clinical trials group. J Clin Oncol. 2011;29:3278–85. doi: 10.1200/JCO.2010.34.1578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pálsdóttir K, Fischerova D, Franchi D, et al. Preoperative prediction of lymph node metastasis and deep stromal invasion in women with invasive cervical cancer:prospective multicenter study using 2D and 3D ultrasound. Ultrasound Obstet Gynecol. 2015;45:470–5. doi: 10.1002/uog.14643. [DOI] [PubMed] [Google Scholar]
- Park J-Y, Kim EN, Kim D-Y, et al. Clinical impact of positron emission tomography or positron emission tomography/computed tomography in the posttherapy surveillance of endometrial carcinoma:evaluation of 88 patients. Int J Gynecol Cancer. 2008;18:1332–8. doi: 10.1111/j.1525-1438.2008.01197.x. [DOI] [PubMed] [Google Scholar]
- Park jj CK, Park SY, Park KB. Value of diffusion-weighted imaging in predicting parametrial invasion in stage IA2-IIA cervical cancer. Eur Radiol. 2014;2014 doi: 10.1007/s00330-014-3109-x. [DOI] [PubMed] [Google Scholar]
- Park Y, Lee J-H, Hong DJ, Lee EY, et al. Diagnostic performances of HE4 and CA125 for the detection of ovarian cancer from patients with various gynecologic and non-gynecologic diseases. Clin Biochem. 2011;44:884–8. doi: 10.1016/j.clinbiochem.2011.04.011. [DOI] [PubMed] [Google Scholar]
- Pecorelli S. Revised FIGO staging for carcinoma of the vulva, cervix, and endometrium. Int J Gynaecol Obstet. 2009;105:103–4. doi: 10.1016/j.ijgo.2009.02.012. [DOI] [PubMed] [Google Scholar]
- Rechichi G, Galimberti S, Signorelli M, et al. Myometrial invasion in endometrial cancer:diagnostic performance of diffusion-weighted MR imaging at 1.5-T. Eur Radiol. 2010;20:754–62. doi: 10.1007/s00330-009-1597-x. [DOI] [PubMed] [Google Scholar]
- Saarelainen SK, Peltonen N, Lehtimäki T, et al. Predictive value of serum human epididymis protein 4 and cancer antigen 125 concentrations in endometrial carcinoma. Am J Obstet Gynecol. 2013;209:142–6. doi: 10.1016/j.ajog.2013.04.014. [DOI] [PubMed] [Google Scholar]
- Santala M, Talvensaari-Mattila A, Kauppila A. Peritoneal cytology and preoperative serum CA 125 level are important prognostic indicators of overall survival in advanced endometrial cancer. Anticancer Res. 2003;23:3097–103. [PubMed] [Google Scholar]
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015;65:5–29. doi: 10.3322/caac.21254. [DOI] [PubMed] [Google Scholar]
- Siegel CL, Andreotti RF, Cardenes HR, et al. ACR Appropriateness criteria®pretreatment planning of invasive cancer of the cervix. J Am Coll Radiol. 2012;9:395–402. doi: 10.1016/j.jacr.2012.02.021. [DOI] [PubMed] [Google Scholar]
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66 doi: 10.3322/caac.21332. [DOI] [PubMed] [Google Scholar]
- Sohaib SA, Houghton SL, Meroni R, et al. Recurrent endometrial cancer:patterns of recurrent disease and assessment of prognosis. Clin Radiol. 2007;62:28–34. doi: 10.1016/j.crad.2006.06.015. [DOI] [PubMed] [Google Scholar]
- Soliman PT, Oh JC, Schmeler KM, et al. Risk factors for young premenopausal women with endometrial cancer. Obstet Gynecol. 2005;105:575–80. doi: 10.1097/01.AOG.0000154151.14516.f7. [DOI] [PubMed] [Google Scholar]
- Spencer JA, Ghattamaneni S. MR imaging of the sonographically indeterminate adnexal mass. Radiology. 2010;256:677–94. doi: 10.1148/radiol.10090397. [DOI] [PubMed] [Google Scholar]
- Steffensen KD, Waldstrøm M, Brandslund I, Jakobsen A. Prognostic impact of prechemotherapy serum levels of HER2, CA125, and HE4 in ovarian cancer patients. Int J Gynecol Cancer. 2011;21:1040–7. doi: 10.1097/IGC.0b013e31821e052e. [DOI] [PubMed] [Google Scholar]
- Sturgeon CM, Duffy MJ, Walker G. The national institute for health and clinical excellence (NICE) guidelines for early detection of ovarian cancer:the pilotal role of the clinical laboratory. Ann Clin Biochem. 2011;48:295–9. doi: 10.1258/acb.2011.011117. [DOI] [PubMed] [Google Scholar]
- Suzuki Y, Nakano T, Ohno T, et al. Serum CYFRA 21-1 in cervical cancer patients treated with radiation therapy. J Cancer Res Clin Oncol. 2000;126:332–6. doi: 10.1007/s004320050352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tangjitgamol S, Katanyoo K, Laopaiboon M, et al. Adjuvant chemotherapy after concurrent chemoradiation for locally advanced cervical cancer. Cochrane Database Syst Rev. 2014;12:CD010401. doi: 10.1002/14651858.CD010401.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terzic M, Dotlic J, Likic1 I, et al. Diagnostic value of serum tumor markers for adnexal masses. Cent Eur J Med. 2014;9:417–23. [Google Scholar]
- Testa AC, Ludovisi M, Manfredi R, et al. Transvaginal ultrasonography and magnetic resonance imaging for assessment of presence, size and extent of invasive cervical cancer. Ultrasound Obstet Gynecol. 2009;34:335–44. doi: 10.1002/uog.7325. [DOI] [PubMed] [Google Scholar]
- Tilli TM, Franco VF, Robbs BK, et al. Osteopontin-c splicing isoform contributes to ovarian cancer progression. Mol Cancer Res. 2011;9:280–93. doi: 10.1158/1541-7786.MCR-10-0463. [DOI] [PubMed] [Google Scholar]
- Tilli TM, Bellahcène A, Castronovo V, et al. Changes in the transcriptional profile in response to overexpression of the osteopontin-c splice isoform in ovarian (OvCar-3) and prostate (PC-3) cancer cell lines. BMC Cancer. 2014;14:433. doi: 10.1186/1471-2407-14-433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Viens LJ, Henley SJ, Watson M, et al. Human papillomavirus-associated cancers –United States, 2008-2012. MMWR Morb Mortal Wkly Rep. 2016;65:661–6. doi: 10.15585/mmwr.mm6526a1. [DOI] [PubMed] [Google Scholar]
- Wagner PD, Srivastava S. New paradigms in translational science research in cancer biomarkers. Transl Res. 2012;159:343–53. doi: 10.1016/j.trsl.2012.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner AE, Pappas L, Ghia AJ, Gaffney DK. Impact of tumor size on survival in cancer of the cervix and validation of stage IIA1 and IIA2 subdivisions. Gynecol Oncol. 2013;129:517–21. doi: 10.1016/j.ygyno.2013.03.008. [DOI] [PubMed] [Google Scholar]
- Walentowicz P, Krintus M, Sadlecki P, et al. Serum inhibin A and inhibin B levels in epithelial ovarian cancer patients. PLoS One9. 2014;9:e90575. doi: 10.1371/journal.pone.0090575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu X, Li D, Liu L, et al. Serum soluble mesothelin-related peptide (SMRP):a potential diagnostic and monitoring marker for epithelial ovarian cancer. Arch Gynecol Obstet. 2014;289:1309–14. doi: 10.1007/s00404-013-3128-x. [DOI] [PubMed] [Google Scholar]
- Yılmaz EPT, Kumtepe Y. Endometrial and ovarian cancer with MR imaging importance of serum HE4 and CA 125 levels in the extent of disease at evaluation. Eurasian J Med. 2016;48:192–8. doi: 10.5152/eurasianjmed.2016.0259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zanotti L, Bignotti E, Calza S, et al. Human epididymis protein 4 as a serum marker for diagnosis of endometrial carcinoma and prediction of clinical outcome. Clin Chem Lab Med. 2012;50:2189–98. doi: 10.1515/cclm-2011-0757. [DOI] [PubMed] [Google Scholar]


