Abstract
Background and objectives:
Laminin is a significant basement membrane (BM) glycoprotein, the expression of which reflects BM integrity more precisely than do other ECM proteins. The present study aimed to evaluate laminin expression in oral squamous cell carcinomas OSCC and to determine any associations with clinico-pathological parameters (surgical margin status, lymph node involvement, survival and recurrence).
Methods:
Laminin expression was evaluated in 31 cases of biopsy-proven OSCC by immunohistochemical staining and its association with prognosticators and the Brynes grading system was determined by appropriate statistical analysis.
Results:
We observed a significant increase in linear staining pattern (p<0.001) at the tumour-host interface in well-differentiated OSCC cases, in contrast to poorly differentiated lesions which exhibited intense cytoplasmic expression within tumour cells. Higher cytoplasmic laminin expression was seen in 33.3% of cases with involved surgical margins and 69.2% of cases with lymph node metastasis (along with weak/absent staining of laminin around the tumour-host interface – Basement membrane around tumour islands). Similarly, in 60% of the cases who died and in 81.8% of cases with tumour recurrence, moderately intense cytoplasmic laminin expression was seen within tumour cells. On comparing variables of the Brynes grading system, significant cytoplasmic expression of laminin was linked with mild inflammation (p<0.0016) and increased mitotic activity (p<0.008).
Conclusion:
Based on these observations, immunohistochemical expression of laminin might be useful to evaluate histological differentiation and aggressiveness of OSCCs.
Keywords: Laminin, basement membrane, OSCC, prognosis
Introduction
Basement membrane (BM) is a thin extracellular structure that separates epithelium from underlying stroma. It is composed of numerous glycoproteins that form an organized scaffold to support the tissues. It modulates cellular function while also serving as a robust structural barrier against tumour invasion. Laminin (a 900kDa mosaic BM glycoprotein) reflects the integrity of BM more ably in comparison to other extracellular matrix (ECM) proteins and thus, may be used as a marker suggestive of the status of intact or degraded basement membrane during tumorigenesis (Souza et al., 2007).
Some carcinomas exhibit characteristic high-affinity sites on the cell surface for binding with laminin and may act as ligand/ laminin receptors for invading tumours. Upon their adherence to laminin, they get stabilized and subsequently stimulated to produce metalloproteinases, which begin fragmentation and degradation of the BM (Sharma et al., 2013). The process of tumour invasion necessitates the neoplastic epithelial cells to traverse in different tissue compartments. Therefore, an assessment of factors that influence these processes is important in understanding tumour behavior (Koshy et al., 2014). As oral cancer is one of the most invasive human tumours with high mortality and morbidity, unravelling the effects of laminin on tumour progression is essential for the development of new prognostic indicators and treatment strategies for the same.
Keeping this in mind, the present study aimed to assess the expression of laminin in oral squamous cell carcinoma (OSCC) and to correlate the same with the clinico-pathological parameters (Recurrence, survival, lymph node metastasis and status of resected margins).
Materials and Methods
Formalin-fixed, paraffin-embedded tissue blocks of 31 cases of biopsy-proven OSCC were retrieved from departmental archives. Two serial sections of 4µm thickness each were taken. One section was stained using routine Haematoxylin and Eosin, to perform histological grading for differentiation (Bryne’s grading system for OSCC at invasive tumour front) and the corresponding sections for the margins and the lymphnode status of the selected cases were evaluated for presence of pathological nodal involvement and margin status
The other section was subjected to immunohistochemical staining as per manufacturer’s instructions using anti-Laminin polyclonal antibody [(6.0 ml) BioGenexTM product code AR078-5R. BioGenex Laboratories, Inc. 49026 Milmont Drive, Fremont, CA 94538, USA] with appropriate controls in each batch for IHC staining. Sections from normal buccal mucosa were used as staining controls and normal blood vessels in the tissue sections provided the internal positive control for IHC staining with laminin. For the negative controls, the aforementioned protocol was followed with omission of the primary antibody.
Scoring Methodology for Laminin Expression
Ten consecutive representative fields at ITF were examined in both 10x and 40x in each case of OSCC and the intensity of the staining was visualized and compared with the internal control and batch control and scored.
Using the modified method proposed by Ono et al., (1999) the cytoplasmic staining of laminin in the tumour cells was graded as 3 for intensity similar to that of control, 2 for lesser intensity as the control but definitely discernable cytoplasmic staining, 1 for mild staining and 0 for no stain.
Further, the pattern of laminin staining around the islands (at the tumour host interface) was graded using the modified method proposed by Arduino et al., (2010) which were graded as 3 for continuous linear staining with definite colour, 2 for linear staining with moderate colour,1 for weak staining, 0 for absent or very weak staining.
Statistical Analysis
The data obtained were statistically analyzed using SPSS statistical software package version 20.0. Laminin expression was compared using chi square test (Fishers exact test).
The expression of laminin in the cytoplasm of tumour cells as well as its linear expression at the basement membrane was compared within the different grades of OSCC. Laminin expression was also comparatively evaluated in regard to parameters of recurrence, survival and involvement of lymphnodes and surgical margins of the involved cases of OSCC.
Results
In the present study, the mean age of the patients with OSCC was 56.23± 11.13 years. Of the 31 OSCC patients, 21(67.7%) were males and 10(32.3%) were females. The most frequent clinical stage was stage I (41.9%) and the predominant histological grade was the well-differentiated grade of OSCC (WDSCC-38.70%). Surgery was the primary treatment modality given to the patients. The average 5-year survival rate in patients was found to be 76.19%. Assessment of the pathological lymphnode status revealed that 18 (58.06%) of the cases were free of lymphnode metastasis in contrast to the surgical resection margins which were found to be involved in 15 (53.57%) cases. Most of the patients (n=8; 47.06%) in our study had a habit of tobacco chewing when compared to the habit of smoking alone or combination of smoking as well as chewing, and few of them reported no history of any habit. Of all the OSCC cases, the buccal mucosa and tongue were equally leading sites of occurrence (32.3% each) followed by mandibular alveolar complex (19.4%), maxilla (12.9%) and lip (3.2%).
Laminin expression in different grades of OSCC (Table 1)
Table 1.
Comparison of Laminin in Different Grades of OSCC
| Histological grades of OSCC | Chi Square Value | P Value | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Well- Differentiated | Moderately- Differentiated | Poorly- Differentiated | |||||||
| Parameter | Scoring | Count | Percent | Count | Percent | Count | Percent | ||
| Cytoplasmic expression of Laminin within tumour cells | Mild or absent | 11 | 91.70% | 1 | 9.10% | 1 | 12.50% | 32.644 | <0.001 |
| Moderate | 1 | 8.30% | 10 | 90.90% | 1 | 12.50% | |||
| Intense | 0 | 0.00% | 0 | 0.00% | 6 | 75.00% | |||
| Pattern of laminin expression at tumour stroma interface | No or very weak staining | 7 | 58.30% | 11 | 100.00% | 8 | 100.00% | 7.308 | 0.108 |
| Weak staining | 1 | 8.30% | 0 | 0.00% | 0 | 0.00% | |||
| Linear staining, moderately-coloured | 2 | 16.70% | 0 | 0.00% | 0 | 0.00% | |||
| Continuous linear staining, definitely-coloured | 2 | 16.70% | 0 | 0.00% | 0 | 0.00% | |||
An overall comparison of laminin expression revealed that 75% of the poorly-differentiated cases of OSCC (PDSCC) expressed intense cytoplasmic laminin in the tumour islands and nests (p<0.001). (Figure 1a) In contrast, 91.7% WDSCC exhibited nil to mild expression of laminin and 90.9% of moderately-differentiated (MDSCC) cases exhibited moderate laminin expression. (Figure 1b, Figure 1c) Evaluation of laminin at the tumour-stroma interface revealed no staining for laminin in all cases of moderately- and poorly-differentiated carcinomas (100%) whereas linear staining was seen in 33.4% of WDSCC. (Figure 1d) However, this finding was statistically not significant (p=0.108).
Figure 1.
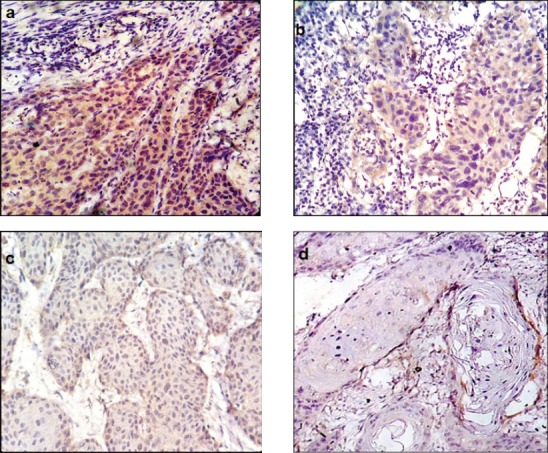
a, Intense cytoplasmic immunohistochemical positivity seen in PDSCC (40x); b, Cytoplasmic immunohistochemical positivity seen in MDSCC (40x); c, Cytoplasmic immunohistochemical positivity seen in WDSCC (40x); d, Linear immunohistochemical positivity seen at tumour host interface in WDSCC (40x).
We observed that 33.30% of OSCC cases in which margins were involved showed intense cytoplasmic expression of laminin within the tumour cells while only 7.70% of cases exhibited laminin expression when margins were free of tumour. The laminin expression at the tumour-host interface was weak or absent in 93.30% of margin-involved cases, while 23% of cases exhibited linear staining when margins were free. However, the findings were not statistically significant.
We observed that 69.2% of cases with lymph nodes involved by tumour showed moderate to intense cytoplasmic expression of laminin within tumour cells as compared to only 50% of un-involved cases but the difference was not statistically significant. Continuous linear staining was seen at tumour-host interface in 11.1% of uninvolved cases while none of lymph node involved cases showed any staining (significant association at 90% CI with p value of 0.07). (Table 2)
Table 2.
Comparison of Laminin Expression and Lymphnode Status
| Lymphnode status | |||||||
|---|---|---|---|---|---|---|---|
| Free | Involved | Chi Square | P Value | ||||
| Count | Column N % | Count | Column N % | ||||
| Cytoplasmic expression of Laminin within tumour cells | Mild or absent | 9 | 50.00% | 4 | 30.80% | 2.12 | 0.42 |
| Moderate | 5 | 27.80% | 7 | 53.80% | |||
| Intense | 4 | 22.20% | 2 | 15.40% | |||
| Pattern of laminin expression at tumour stroma interface | No or very weak staining | 16 | 88.90% | 10 | 76.90% | 4.83 | 0.07 |
| Weak staining | 0 | 0.00% | 1 | 7.70% | |||
| Linear staining, moderately coloured | 0 | 0.00% | 2 | 15.40% | |||
| Continuous linear staining, definitely coloured | 2 | 11.10% | 0 | 0.00% | |||
Survival data was available for only 21 of the 31 cases of OSCC. All the cases that survived showed variable cytoplasmic laminin with 9 out of 16 survivors exhibiting mild or absent staining in the tumour cells. Moderate expression of cytoplasmic laminin was seen in only 25% (n=4) of survived cases and in 60% (n=3) of patients who succumbed to disease. The linear staining pattern of laminin did not show significant association with survival as it was completely absent or weak in all cases that died and showed similar findings with the survivors (68.8%) most of the survivors (68.8%) too showed similar findings.
Majority of the cases that recurred (81.8%) showed moderate cytoplasmic expression of laminin within tumour cells and weak/absent linear staining of laminin around the tumour-host interface. In the absence of sufficient number of cases with no recurrence, the correlation between recurrence and laminin expression was deemed insignificant.
Amongst the variables in Brynes grading system (keratinization, mitotic activity, nuclear pleomorphism, pattern of invasion and lymphocytic-host response), increased mitosis and mild inflammation were significantly associated (p value <0.008 and 0.016 respectively) with an intense expression of laminin within the tumour cells and weak/ absence of laminin expression at tumour host interface (Table 3).
Table 3.
Comparison of Laminin Expression and Individual Parameters in Brynes Grading Criteria
| Brynes parameters | Criteria | Cytoplasmic laminin expression in tumour cells | Pattern of laminin expression at tumour stroma interface | |||||
|---|---|---|---|---|---|---|---|---|
| Mild/ absent | Moderate | Intense | No/ weak | Weak | Linear moderately colour | Continous definite colour | ||
| Keratinization | High >75% | 0.00% | 8 (88.9%) | 1 (11.10%) | 6 (66.70%) | 0.00% | 2 (22.00%) | 1 (11.10%) |
| Moderate 75%-25% | 0.00% | 11 (78.60) | 3 (21.40%) | 13 (92.90%) | 1 (7.10%) | 0.00% | 0.00% | |
| Mininmum<25% | 2 (25.00%) | 4 (50.00%) | 2 (25.00%) | 7 (87.50%) | 0.00% | 0.00% | 1 (12.50%) | |
| χ2=7.158 P=0.218 | χ2=8.228 P=0.222 | |||||||
| Nuclear Pleomorphism | Mild | 7 (77.80)% | 2 (22.20%) | 0.00% | 7 (77.80)% | 0.00% | 1 (11.10%) | 1 (11.10%) |
| Moderate | 5 (35.70)% | 6 (42.90%) | 3 (21.40%) | 11 (78.60%) | 1 (7.10%) | 1 (7.10%) | 1 (7.10%) | |
| Extreme | 1 (12.50%) | 4 (50.00%) | 3 (37.50%) | 8 (100.00%) | 0.00% | 0.00% | 0.00% | |
| χ2=8.051 P=0.075 | χ2=3.668 P=1.0 | |||||||
| Mitosis (in 10HPF) | 0-1 | 3 (75.00%) | 1 (25.00%) | 0.00% | 2 (50.00)% | 1 (25.00%) | 0.00% | 1 (25.00%) |
| 2-3 | 10 (62.50)% | 4 (25.00%) | 2 (12.50%) | 13 (81.20%) | 0.00% | 2 (12.50%) | 1 (6.20%) | |
| 4-5 | 0.00% | 6 (66.70%) | 3 (33.30%) | 9 (100.00%) | 0.00% | 0.00% | 0.00% | |
| >5 | 0.00% | 1 (50.00%) | 1 (50.00%) | 2 (100.00%) | 0.00% | 0.00% | 0.00% | |
| χ2=13.66 P<0.008 | χ2=10.954 P=0.263 | |||||||
| Pattern of invasion | Sheets | 3 (75.00%) | 1 (25.00%) | 0.00% | 3 (75.00%) | 0.00% | 0.00% | 1 (25.00%) |
| Islands | 9 (47.40)% | 8 (42.10%) | 2 (10.50%) | 16 (84.20%) | 1 (5.30%) | 1 (5.30%) | 1 (5.30%) | |
| Nests and cords | 1 (25.00%) | 2 (50.00)% | 1 (25.00%) | 3 (75.00%) | 0.00% | 1 (25.00%) | 0.00% | |
| Strands and individual cells | 0.00% | 1 (25.00%) | 3 (75.00%) | 4 (100.00%) | 0.00% | 0.00% | 0.00% | |
| χ 2=8.967 P=0.102 | χ 2=7.733 P=0.713 | |||||||
| Inflammation (Density) | Extreme | 1 (25.00%) | 3 (75.00%) | 0.00% | 3 (75.00%) | 0.00% | 1 (25.00%) | 0.00% |
| Moderate | 1 (8.30%) | 11 (91.70%) | 0.00% | 9 (75.00%) | 1 (8.30%) | 1 (8.30%) | 1 (8.30%) | |
| Mild | 0.00% | 9 (60.00)% | 6 (40.00%) | 14 (93.30%) | 0.00% | 0.00% | 1 (6.70%) | |
| χ2=9.37 P<0.016 | χ2=6.049 P=0.405 | |||||||
Discussion
Laminin, a multifunctional extracellular matrix (ECM) glycoprotein expressed predominantly in the BM structure is associated with an aggressive cancer phenotype by being a potent regulator of cell adhesion and cell migration. Laminin is produced by the cell and is transported extracellularly to be deposited in the basement membrane zone.
In our study 75% of the PDSCC cases expressed intense cytoplasmic staining of laminin within the tumour cells where as WDSCC and MDSCC cases exhibited mild to moderate expression of laminin in the cytoplasm. Ono et al found high invasive potential of cancer cells correlating with poor prognosis when cytoplasmic expression of Laminin-5 γ2 chain was seen in cases of tongue carcinoma (Ono et al., 1999).
Thus, poorly differentiated tumour cells which exhibit active migration and invasive potential, correlate with the increased expression of cytoplasmic laminin receptor antigen. Also, Wewer et al suggested that tumor cells process the laminin receptor antigen in an intrinsically different manner compared to non-neoplastic cells. The higher cytoplasmic distribution reflects an internalization of the receptor as a part of the sequence of attachment-detachment cycle that is exploited by tumour cells to migrate. Thus, they speculated that an actively invading carcinoma cell may coat its own surface with endogenous laminin and then internalize the receptor and the ligand (Wewer et al., 1987).
Patel et al., (2002) observed that laminin which was over-expressed in tumor tissues in head and neck SCC appeared to be confined to the intracellular (cytoplasmic) area with no evidence of its extracellular export. They suggested that only the γ subunit of laminin gets synthesized and remains unassembled in the cytoplasm, thereby precluding its extracellular transport. Thus laminin polypeptides are not incorporated into the basement membrane and accumulate within the neoplastic cells. The higher cytoplasmic laminin may also be due to preferential production of laminin γ2 monomer during carcinogenesis. This may hinder the extracellular export of other laminin chains adding to the cytoplasmic accumulation of laminin (Koshikawa et al., 1999).
We also found linear staining of laminin at the tumour connective tissue interface in 33.4% cases of WDSCC while no laminin was expressed in MDSCC and PDSCC. Souza et al., (2007) also observed a continuous laminin staining around the basement membrane of WDSCC cases suggesting that well-differentiated malignant cells often retain some ability to produce BM components (Shruthy et al., 2013). However, with the progression of disease, cells in MDSCC and PDSCC also release metalloproteinases causing fragmentation and degradation of the membrane. Thus, laminin, even if secreted, gets destroyed along with the basement membrane and is perceived as lack of BM staining. This apparent destruction of secreted laminin appears to be inherent to the malignant cell phenotype (Tani et al.,1997; Lazaris et al., 2003) and may explain the absence of laminin staining at tumour-stroma interface in MDSCC and PDSCC cases in our study too.
However, there are several reports which show both intracytoplasmic as well as basement membrane staining of laminin (Hao et al., 1996; Tani et al., 1997; Henning, 1999).
Intense cytoplasmic expression of laminin was seen in tumour cells in 33.3% of cases in which margins were involved while only 7.7% of cases exhibited laminin expression when margins were free of tumour. At the tumour-host interface the laminin expression was weak or absent in 93.3% of margin-involved cases, while 23% of cases exhibited linear staining when margins were free. However, the findings were not statistically significant.
Moderate to intense cytoplasmic expression of laminin within tumour islands/nests was observed in 69.2% of cases with lymph nodes involved by tumour compared to only 50% of un-involved cases (not statistically significant). At the tumour-host interface continuous linear staining was seen in 11.1% of uninvolved cases while none of LN-involved cases showed any staining. (Table 2)
Though statistically insignificant, laminin seemed to be clinically associated with various clinico-pathologic parameters as indicated by invasion and metastasis. Lenander et al.,2001 also observed an association of laminin-5 γ2 chain expression with local invasiveness of colon carcinomas. These findings may reflect various hypotheses described herein:
Corbalan-Velez R et al postulated a pathway for metastasis and suggested that Laminin-5 interacts with integrin α6β4 and epidermal growth factor receptor (EGFR), thereby activating phosphatidylinositol-3-kinase (pI3K) a key regulator of several cell processes such as proliferation, growth and apoptosis. Through the activation of pI3K, laminin-5 promotes tumor invasion in SCC and is associated with more advanced TNM stages and worse prognosis with involvement of lymph nodes at times (Corbalan-Velez et al., 2012).
Fitsialos et al., (2008) proposed the role of hypoxia in regulating laminin expression. Increased expression of HIF1α results in the upregulation of the HIF1 target genes including LAM A3 (that encodes the α3 subunit of laminin-5). Upregulation of LAM A3 in turn upregulates the synthesis of laminin-5. The resultant increased deposition of laminin-5 in the ECM strongly influences keratinocyte adhesion and their ability to migrate. Presuming carcinomas to be non-healing wounds and with the understanding that necrosis stimulates HIF-1, it is highly possible that this mechanism plays a role in regulating laminin expression in OSCC.
Various studies in invasive carcinomas have reported over-expression of laminin γ2 chain and lowered or impaired expression of other laminin chains. Even though laminin γ2 chains are increased, their lack of integrin-binding sites prevents their facilitating integrin-mediated cell adhesion. Thus, the laminin γ2 chain monomer may contribute to loss of BM structures by its non-integration with other laminin chains thus enhancing tumor invasion (Miyazaki, 2006).
Recent studies in colorectal carcinoma have also shown that the laminin epidermal growth factor-like domain (LE) of the laminin γ2 chain can be released from the proteolytic cleavage caused by MMP2 and membrane-type 1 matrix metalloproteinase (MT1-MMP) released by circulating tumour cells. The γ2 chain of laminin may stimulate cell migration by binding to the epidermal growth factor receptor. This coordinated action between γ2 chain of laminin-5 and matrix proteinases is reported to be brought by the β-WNT signaling pathway. Iwai et al reported evidence of β-WNT signaling in the invasion and migration of OSCC. Thus, the interaction between β-WNT signaling, laminin and MMP’s may be important for enhanced cell migration in tumor tissues and metastasis in OSCC (Miyazaki 2006; Iwai et al., 2010).
Navdaev A and Eble JA have speculated that laminin stimulates carcinoma cells to form lamellipodia which in turn enhances cell migration and invasion (Navdaev and Eble, 2011).
The expression of laminin-5 is enhanced by the induction of urokinase-type plasminogen activator (u-PA) and loss of α6β4 integrin. Several factors including hepatocyte growth factor (HGF), transforming growth factor (TGF)-α and TGF-β are also known to induce expression of laminin-5 with over-expressed laminin being retained in the cytoplasm of the carcinoma cells. Thus, several molecules may work in concert with laminin-5 to facilitate cancer cell invasion (Moriya et al., 2001) (Figure 2).
Figure 2.
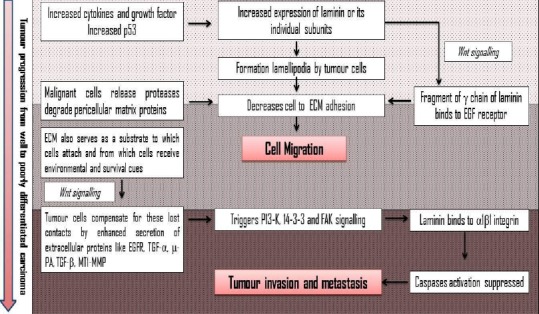
Comprehensive Compilation of Role of Laminin in Tumour Invasion (Inputs from References Corbalan-Velez et al., 2012; Navdaev and Eble, 2011; Moriya et al., 2001; Marinkovich, 2007; Spenle et al., 2014)
More than half the cases with good 5-year survival (9 out of 16 survivors) showed mild/absent cytoplasmic expression of laminin in the tumour cells. The linear staining of laminin around the tumour-host interface was seen in 31.20% of survived cases in contrast to absent/very weak linear staining of laminin around the tumour islands in the cases that did not survive. These findings are in accordance with a study by Ono et al in which they observed significantly reduced survival with increased expression of laminin-5 γ2 chain in SCC of the tongue (Ono et al.,1999). However, Lenander et al., (2001) used multivariate analyses on 93 cases of primary colon carcinomas to suggest that laminin-5 expression was not an independent prognostic factor for survival.
Of the 31 cases in our cohort, the presence/absence of recurrence was recorded in 13 cases of which only 11 showed recurrence. It is reasonable to presume that in the absence of patient compliance for follow-up only a recurrent lesion may cause the patient to come to the hospital where the record is retrievable. In the cases that recurred, 9 (81.8%) cases showed moderate cytoplasmic expression of laminin within tumour islands/nests and weak/absent linear staining of laminin around the tumour-host interface. Regardless, the expression of laminin did not show significant association with recurrence of OSCC in our study, which could be attributed to the absence of sufficient patient data with no recurrence whereas, Yamamoto et al., (2001) observed laminin-5 to be significantly correlated with recurrence in their study on esophageal SCC.
Marques et al opined that laminin expression is efficient in determining the risk of recurrence rather than survival/death and found that the prognostic importance of laminin was considerable only when lymph node status was accounted for assessment (Marques et al.,1990).
From amongst the variables in Brynes grading system (keratinization, mitotic activity, nuclear pleomorphism, pattern of invasion and lymphocytic-host response), only increased mitosis and mild inflammation were significantly associated with an intense expression of laminin within the tumour cells.
Laminin γ2 chains interact with EGFR, P13k and integrins which initiate tumour proliferation that reflected as increased mitotic activity as seen in our cases (Marinkovich, 2007).
In inflammatory bowel disease, laminin is over-expressed at BM as a protective mechanism to prevent epithelial damage (Spenle et al., 2014). Similarly in OSCC, mild inflammation may induce cytoplasmic over-production of laminin as a protective mechanism. But increased inflammation will increase cytokines and growth factors that enhance laminin expression near the tumour-host interface probably as a part of defense mechanism. Also, worsening grades of tumour cause expression of mutated p53 which displaces laminin from cytoplasm towards tumour-host interface resulting in an overall increase in laminin at the interface. Increased laminin expression at tumour-host interface in turn decreases inflammation creating a pro-tumorigenic niche and also acts in concert with proteases like MMP’s thus destroying the formed basement membrane and favouring invasion and metastasis (Spenle et al.,2014; Bouatrouss et al., 2000).
In conclusion, evaluation of the Laminin expression in OSCC showed that the WDSCC cases exhibited predominantly linear staining pattern at the interface, whereas cases of PDSCC exhibited intense cytoplasmic expression within tumour cells. A trend of higher cytoplasmic laminin expression was observed in cases of involved surgical margins and lymphnode metastasis along with weak/absent linear staining of laminin around the tumour-host interface. Likewise, in most of the cases of death and recurrence of tumour, moderately intense cytoplasmic laminin expression was observed within the tumour cells along with weak/absent linear staining of laminin around the tumour-host interface. Thus, this could be suggestive of the role of Laminin in inducing cell migration and metastasis by regulating the formation of lamellipodia as well as its interactions with integrins, proteases, EGFR and various signaling pathways.
Thus, laminin can be adopted as a useful marker in evaluating the histological differentiation and aggressiveness of oral carcinoma.
References
- Arduino PG, Carrozzo M, Pagano M, et al. Immunohistochemical expression of basement membrane proteins of verrucous carcinoma of the oral mucosa. Clin Oral Invest. 2010;14:297–302. doi: 10.1007/s00784-009-0296-y. [DOI] [PubMed] [Google Scholar]
- Bouatrouss Y, Herring-Gillam FE, Gosselin J, Poisson J, Beaulieu JF. Altered expression of laminins in Crohn's disease small intestinal mucosa. Am J Pathol. 2000;156:45–50. doi: 10.1016/S0002-9440(10)64704-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corbalan-Velez R, Martínez-Barba E, Lopez-Poveda MJ, et al. The value of laminin 322 staining in distinguishing between keratoacanthoma, keratoacanthoma with areas of squamous cell carcinoma, and crateriform squamous cell carcinoma. Actas Dermosifiliogr. 2012;103:308–16. doi: 10.1016/j.ad.2011.09.004. [DOI] [PubMed] [Google Scholar]
- Fitsialos G, Bourget I, Augier S, et al. HIF1 transcription factor regulates laminin-332 expression and keratinocyte migration. J Cell Sci. 2008;121:2992–3001. doi: 10.1242/jcs.029256. [DOI] [PubMed] [Google Scholar]
- Hao J, Yang Y, Mc Daniel KM, et al. Differential expression of laminin 5 (alpha 3 beta 3 gamma 2) by human malignant and normal prostate. Am J Pathol. 1996;149:1341–9. [PMC free article] [PubMed] [Google Scholar]
- Henning K, Berndt A, Katenkamp D, Kosmehl H. Loss of laminin-5 in the epithelium-stroma interface:an immunohistochemical marker of malignancy in epithelial lesions of the breast. Histopathology. 1999;34:305–9. doi: 10.1046/j.1365-2559.1999.00634.x. [DOI] [PubMed] [Google Scholar]
- Iwai S, Yonekawa A, Harada C, et al. Involvement of the Wnt-ß-Catenin pathway in invasion and migration of oral squamous carcinoma cells. Int J Oncol. 2010;37:1095–103. doi: 10.3892/ijo_00000761. [DOI] [PubMed] [Google Scholar]
- Koshikawa N, Moriyama K, Takamura H, et al. Overexpression of laminin γ2chain monomer in invading gastric carcinoma cells. Cancer Res. 1999;59:5596–601. [PubMed] [Google Scholar]
- Koshy AV, Rao NN, Kamat SS, et al. Expression of extracellular matrix- laminin in oral squamous cell carcinoma:an immunohistochemical study. J Contemp Dent Pract. 2012;13:194–200. doi: 10.5005/jp-journals-10024-1120. [DOI] [PubMed] [Google Scholar]
- Lazaris ACH, Tzoumani AN, Thimara I, et al. Immunohistochemical assessment of basement membrane components in colorectal cancer:prognostic implications. J Exp Clin Cancer Res. 2003;22:599–606. [PubMed] [Google Scholar]
- Lenander C, Habermann JK, Ost A, et al. Laminin-5 gamma chain expression correlates with unfavorable prognosis in colon carcinomas. Anal Cell Pathol. 2001;22:201–9. doi: 10.1155/2001/137404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marinkovich MP. Tumour microenvironment:Laminin 332 in squamous-cell carcinoma. Nat Rev Cancer. 2007;7:370–80. doi: 10.1038/nrc2089. [DOI] [PubMed] [Google Scholar]
- Marques LA, Franco EL, Torloni H, et al. Independent prognostic value of laminin receptor expression in breast cancer survival. Cancer Res. 1990;50:1479–83. [PubMed] [Google Scholar]
- Miyazaki K. Laminin-5 (laminin-332):Unique biological activity and role in tumor growth and invasion. Cancer Sci. 2006;97:91–8. doi: 10.1111/j.1349-7006.2006.00150.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moriya Y, Niki T, Yamada T, et al. Increased expression of laminin 5 and its prognostic significance in lung adenocarcinomas of small size. An immunohistochemical analysis of 102 cases. Cancer. 2001;91:1129–41. doi: 10.1002/1097-0142(20010315)91:6<1129::aid-cncr1109>3.0.co;2-c. [DOI] [PubMed] [Google Scholar]
- Navdaev A, Eble JA. Components of cell-matrix linkage as potential new markers for prostate. Cancer. 2011;3:883–96. doi: 10.3390/cancers3010883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ono Y, Nakanishi Y, Ino Y, et al. Clinoco-pathologic significance of laminin-5gamma2 chain expression in squamous cell carcinoma of the tongue:immunohistochemical analysis of 67 lesions. Cancer. 1999;85:2315–21. [PubMed] [Google Scholar]
- Patel V, Aldridge K, Ensley JF, et al. Laminin- γ2overexpression in head-and-neck squamous cell carcinoma. Int J Cancer. 2002;99:583–88. doi: 10.1002/ijc.10403. [DOI] [PubMed] [Google Scholar]
- Sharma M, Sah P, Sharma SS, Radhakrishnan R. Molecular changes in invasive front of oral cancer. J Oral Maxillofac Pathol. 2013;17:240–7. doi: 10.4103/0973-029X.119740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shruthy R, Sharada P, Swaminathan U, Nagamalini BR. Immunohistochemical expression of basement membrane laminin in histological grades of oral squamous cell carcinoma:A semiquantitative analysis. J Oral Maxillofac Pathol. 2013;17:185–9. doi: 10.4103/0973-029X.119755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Souza LF, Souza VF, Silva LD, Santos JN, Reis SR. Expression of basement membrane laminin in oral squamous cell carcinomas. Braz J Otolaryngol. 2007;73:768–74. doi: 10.1016/S1808-8694(15)31173-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spenle C, Lefebvre O, Lacroute J, et al. The Laminin response in inflammatory bowel disease:protection or malignancy? PLoS One. 2014;9:e111336. doi: 10.1371/journal.pone.0111336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tani T, Lumme A, Linnala A, et al. Pancreatic carcinomas deposit laminin-5, preferably adhere to laminin-5, and migrate on the newly deposited basement membrane. Am J Pathol. 1997;151:1289–302. [PMC free article] [PubMed] [Google Scholar]
- Wewer UM, Taraboletti G, Sobel ME, Albrechtsen R, Liotta LA. Role of laminin receptor in tumor cell migration. Cancer Res. 1987;47:5691–8. [PubMed] [Google Scholar]
- Yamamoto H, Itoh F, Iku S, Hosokawa M, Imai K. Expression of the gamma (2) chain of laminin-5 at the invasive front is associated with recurrence and poor prognosis in human esophageal squamous cell carcinoma. Clin Cancer Res. 2001;7:896–900. [PubMed] [Google Scholar]


