Abstract
Novel and alternative options are being adopted to combat the initiation and progression of human cancers. One of the approaches is the use of molecules isolated from traditional medicinal herbs, edible dietary plants and seeds that play a pivotal role in the prevention/treatment of cancer, either alone or in combination with existing chemotherapeutic agents. Compounds that modulate these oncogenic processes are potential candidates for cancer therapy and may eventually make it to clinical applications. Diosgenin is a naturally occurring steroidal sapogenin and is one of the major bioactive compounds found in dietary fenugreek (Trigonella foenum-graecum) seeds. In addition to being a lactation aid, diosgenin has been shown to be hypocholesterolemic, gastro- and hepato-protective, anti-oxidant, anti-inflammatory, anti-diabetic, and anti-cancer. Diosgenin has a unique structural similarity to estrogen. Several preclinical studies have reported on the pro-apoptotic and anti-cancer properties of diosgenin against a variety of cancers, both in in vitro and in vivo. Diosgenin has also been reported to reverse multi-drug resistance in cancer cells and sensitize cancer cells to standard chemotherapy. Remarkably, diosgenin has also been reported to be used by pharmaceutical companies to synthesize steroidal drugs. Several novel diosgenin analogs and nano-formulations have been synthesized with improved anti-cancer efficacy and pharmacokinetic profile. In this review we discuss in detail the multifaceted anti-cancer properties of diosgenin that have found application in pharmaceutical, functional food, and cosmetic industries; and the various intracellular molecular targets modulated by diosgenin that abrogate the oncogenic process.
Keywords: diosgenin, steroidal sapogenins, anti-cancer, apoptosis, oncogenic, metastasis
1. Introduction
Cancer is a complex and heterogeneous disease that afflicts men and women worldwide; it is expected to increase due to human lifestyle changes and a rapidly aging population [1]. Hanahan and Weinberg’s proposed hallmarks of cancer are widely accepted towards understanding the biology of cancer cells, through a multi-stage and progressive process [2,3,4,5,6]. These hallmarks include sustained proliferation of cells, constitutive activation of pro-survival transcription factors, deregulated cellular functions, evading cell death signals and growth suppressors, an increased pro-inflammatory tumor microenvironment, camouflaging against immune cell destruction, promoting angiogenesis, activating cell movement from the primary site and metastasis, enabling replicative immortality, and finally, severe genome instability [2,3,5,6,7,8,9,10]. In addition, several deregulated cellular signaling networks underlying the above hallmarks have been extensively investigated in pre-clinical and clinical drug development [3,4,11,12,13,14,15,16,17,18]. In spite of detailed information of these semi-synthetic and synthetic anti-cancer agents, they only provide limited therapeutic advantages to patients due to highly toxic unwanted side effects and the development of chemoresistance [19,20,21]. Emerging approaches should include the identification of novel drug targets that are very effective in inhibiting the growth of cancer cells, while exhibiting fewer adverse effects. Natural products are a good source of compounds with novel chemical structures that are effective and less toxic [18,22,23,24,25,26,27]. High throughput technologies should be exploited for the screening of a large library of compounds for their anti-cancer activities [28,29,30]. The mainstream of United States Food and Drug Administration (US FDA)-approved compounds divulge that natural products and their derivatives occupy one-third of all novel drugs [22,23,24,31]. These natural compounds, in general, show multi-targeted effects and can modulate several oncogenic transcription factors that block the tumor microenvironment targets that usually sustain tumor growth [22,27,32]. These compounds can also be classified as cytotoxic or cytostatic compounds [30]. Therefore, many secondary metabolites and pure molecules isolated from herbs, spices, dietary fruits and vegetables, and even marine sources have been explored [33,34,35].
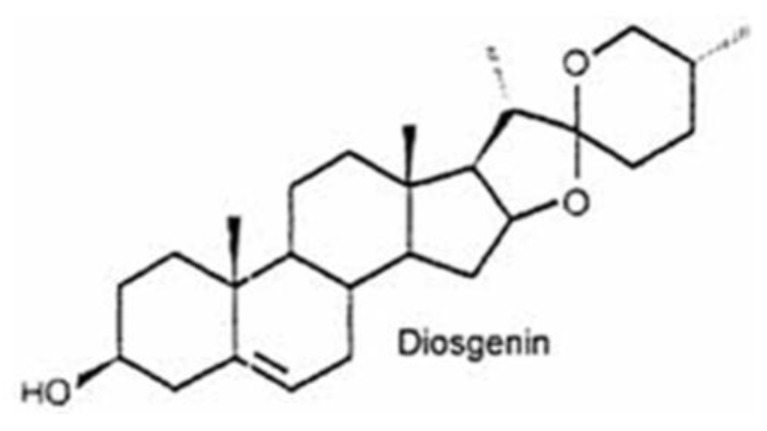
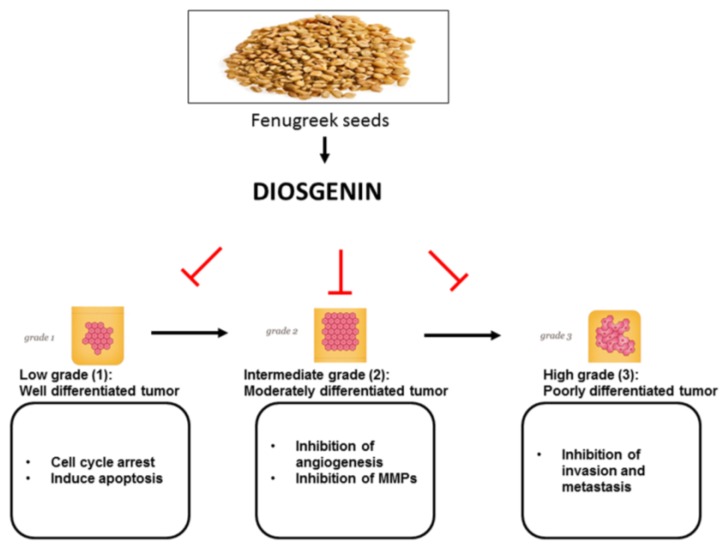
Several novel bioactive molecules have been found in various edible cereals, pulses, roots, and in several parts of medicinal plants. Fenugreek (Trigonella foenum-graecum) belongs to the family Leguminosae and is considered a traditional medicinal herb that is commonly used in India, China, Thailand, and South-East Asian countries; it is also cultivated in the Mediterranean region and Northern Africa [36,37,38,39,40,41]. Fenugreek seeds, shoots and leaves are used in Indian curry preparation as a condiment. Bibliometric data indicates that fenugreek extract has several pharmacological properties, such as being hypocholesterolemic, a lactation aid, antibacterial, a gastric stimulant, anti-anorexic, antidiabetic, galactogogic, hepatoprotective, and anti-cancer both in in vitro and in vivo studies [40,41,42,43]. Fenugreek seed extract contains several bioactive molecules in various classes of compounds, such as saponins, flavonoids, coumarins, and alkaloids that target several molecules involved in inflammation and cancer cell proliferation, invasion, migration, angiogenesis, and metastasis [40]. Sapogenins are a class of compounds that widely occur in natural products in their glycoside form and promote general healthy living. Among these compounds, steroidal sapogenins (otherwise known as spirostans) are the most potent bioactive compounds isolated from natural product sources [44,45]. Steroidal sapogenins exhibit ubiquitous pharmacological properties and the majority of them demonstrate anti-cancer activity in vitro and in pre-clinical animal models. Several clinical trials have been conducted with fenugreek seed extract, they are either completed or ongoing; however, diosgenin anti-cancer trials are yet to start [36,41]. Diosgenin is the most abundant steroidal sapogenin in fenugreek seeds. Fujii and Matsukawa isolated and identified diosgenin from Dioscorea tokoro Makino in 1935 [41,46] (Figure 1). Diosgenin is a phytosteroidal saponin and a major bioactive compound found in the seeds of T. foenum-graecum, commonly known as fenugreek, and in the roots of wild yam (Dioscorea villosa) [36,40,41,47,48]. Interestingly, diosgenin is also found in high levels in numerous plant species including Costus speciosus, Smilax menispermoidea, species of Paris, Aletris and Trillum, and in species of Dioscorea [41,49,50]. The steroidal saponin, diosgenin, is biosynthesized from cholesterol. Cholesterol is formed from lanosterol and catalysed by the cytochrome P450 system. Several other routes of synthesis have been identified, such as from squalene-2,3-oxide in two ways: From cycloartenol through the formation of sitosterol [51] and from lanosterol via cholesterol [52]. Several studies have demonstrated the diverse biological activities of diosgenin, such as hypolipidemic, anti-inflammatory, anti-proliferative, hypoglycemic activity, and as a potent anti-oxidant [44]. In addition, diosgenin inhibited cancer cell proliferation and induced apoptosis in a variety of cancer cell lines including colorectal, hepatocellular, breast, osteosarcoma, and leukemia [53,54,55]. The primary mechanism of action of diosgenin is through the modulation of multiple cell signaling pathways that play prominent roles in cell-cycle regulation, differentiation, and apoptosis [56]. Remarkably, pharmaceutical companies use diosgenin as a principal precursor compound for the manufacturing of several steroidal drugs [48]. Diosgenin is also an attractive molecule with multifaceted properties that has found application in pharmaceutical, functional food, and cosmetic industries. In this review we provide an in-depth evaluation of literature on diosgenin and its pharmacodynamics and pharmacokinetics, and discuss several of its novel derivatives and nanoformulations that increase its bioavailability and therapeutic efficacy. Diosgenin, over the years, has provided abundant data on the prevention and treatment of various inflammation-driven diseases, including cancers [36] (Figure 2).
Figure 1.
Chemical structure of diosgenin.
Figure 2.
Tumor stage-specific inhibition of molecular targets by diosgenin.
2. Fenugreek Seed Bioactive Compounds
Fenugreek contains several chemical constituents, such as alkaloids, steroidal sapogenins, saponins, flavonoids, lipids, amino acids, and carbohydrates. Diosgenin is a major bioactive steroidal sapogenin in the fenugreek seed, which is reported to have chemopreventive and therapeutic effects against inflammation and chronic inflammation-driven cancers in preclinical in vitro and in vivo models of cancer [36,57]. Table 1 illustrates the major constituents of fenugreek seeds and leaves.
Table 1.
Main phytochemical constituents of fenugreek (T. foenum-graecum).
| Class of Compounds | Phytochemical Constituents | Reference |
|---|---|---|
| Steroidal sapogenins | Diosgenin, Yamogenin, Smilagenin, Sarsasapogenin, Tigogenin, Neotigogenin, Gitogenin, Yuccagenin, Saponaretin | [40] |
| Flavonoids | Quercetin, Rutin, Vitexin, Isovitexin | [40] |
| Saponins | Graecunins, Fenugrin B, Fenugreekine, Trigofoenosides A–G | [40] |
| Alkaloids | Trimethylamine, Neurin, Trigonelline, Choline, Gentianine, Carpaine, and Betain | [40] |
| Fibers | Gum, Neutral detergent fiber | [40] |
| Lipids | Lipids, Triacylglycerols, Diacylglycerols, Monoacylglycerols, Phosphatidylcholine, Phosphatidylethanolamine, Phosphatidylinositol, Free fatty acids | [40] |
| Others | Coumarin, Amino acids, Vitamins, Minerals. 28% Mucilage; 22% Proteins; 5% of a stronger swelling, Bitter fixed oil | [40] |
3. In Vitro Anti-Cancer Effects of Diosgenin
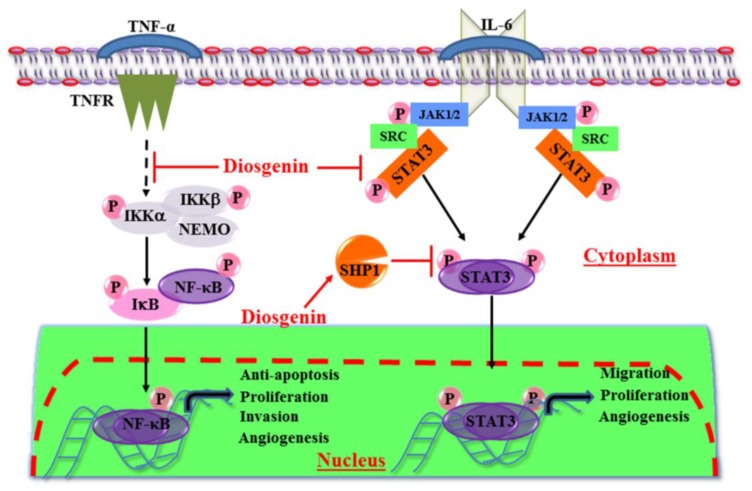
Diosgenin, the major steroidal sapogenin in the fenugreek seed, has been shown to potently suppress constitutively-activated pro-inflammatory and pro-survival signaling pathways in a variety of cancer cells, and induced apoptosis [58]. Some of the earlier studies by Shishodia and Aggarwal [58] reported that diosgenin abrogated TNF-α-induced NF-κB activation and suppressed osteoclastogenesis in RAW 264.7 macrophage cells [58]. In Her-2 positive breast cancer cells, diosgenin inhibited the expression of AKT, mTOR, JNK and their associated pro-survival signaling pathways, and induced apoptosis in these cells [53]. In another study by Li et al., they reported that diosgenin could inactivate the STAT3 signaling pathway in hepatocellular carcinoma (HCC) cells, by inhibiting intracellular signaling molecules such as c-SRC, JAK1, and JAK2 (Figure 3). Diosgenin also suppressed STAT3 transcriptional activity and the expression of its downstream gene products involved in proliferation, invasion and metastasis. In addition, diosgenin sensitized HCC cells to doxorubicin and paclitaxel, and synergistically augmented apoptosis, thereby suggesting that diosgenin is a potential bioactive compound for the treatment of HCC and other cancers [54]. Diosgenin inhibited proliferation, AKT and JNK in a dose- and time-dependent manner and induced caspase-dependent apoptosis in A431 and Hep2 skin squamous cell carcinoma cells [59]. HT-29 colon cancer cells have been reported to be resistant to TRAIL-induced apoptosis. Diosgenin was shown to sensitize the HT-29 colon cancer cell to TRAIL. In addition, it potently suppressed cell proliferation and induced apoptosis by suppressing the p38/MAPK signaling pathway and the overexpression of DR5 [60]. Furthermore, Romero-Hernandez et al. [61] demonstrated that diosgenin-derived thio(seleno)ureas and glycomimetics, bearing a 1,2,3-triazolyl tether on C-3, showed more potent anti-cancer activity against MDA-MB-231 and MCF-7 breast cancer cells, HepG2 hepatocellular carcinoma cells, and induced apoptosis, compared to its parent compound diosgenin [61]. In another study, diosgenin conjugated to methotrexate was found to be more potent in inhibiting the growth of transport-resistant breast cancer cells and dihydrofolate reductase (enzyme involved in DNA synthesis), compared to the parent diosgenin [62]. In chronic myeloid leukemia cells, diosgenin-induced autophagy inhibited the mTOR signaling pathway and induced apoptotic cell death [63]. Diosgenin induced cytotoxicity and significantly inhibited the growth and proliferation of MCF-7 breast cancer cells in a dose- and time-dependent manner. Diosgenin was also shown to inhibit N-nitroso-N-methylurea-induced breast cancer in rats [64].
Figure 3.
Role of diosgenin in NF-κB and STAT3 signaling pathways. Diosgenin abrogates TNF-α -induced activation of NF-κB and IL6-induced STAT3 signaling pathways in tumor cells. Diosgenin can hence prevent proliferation, invasion and angiogenesis; and induce apoptosis, a characteristic vastly looked for in cancer therapy.
Of several factors that contribute to the sustained growth and proliferation of tumors, one important factor is the abundant neovascularization or formation of new micro blood vessels at the tumor site, or within the tumor. This process is also known as tumor angiogenesis. Thus, the formation of new blood vessels in tumors actively supplies the essential nutrients and growth factors that allow tumors to acquire the ability to reject chemotherapeutic drugs and develop chemoresistance [27,65,66,67,68]. Diosgenin is also a potent inhibitor of cancer cell invasion, migration, and tumor-associated angiogenesis [27,69]. He et al., reported that diosgenin inhibited the invasion and migration of triple-negative breast cancer cells and was associated with the concomitant suppression of actin polymerization, phosphorylation of Vav2, and activation of Cdc42 oncoprotein expression. These proteins have been shown to be involved in the initiation of cancer cells’ invasive and migratory potential [70]. Similarly, diosgenin was found to inhibit PC3 androgen-independent prostate cancer cell invasion and migration. The inhibitory effect was mediated by the downregulation of matrix metalloproteinase (MMP)-2 and MMP-9, the key enzymes in matrix degradation and stroma invasion. Furthermore, diosgenin also downregulated the tissue inhibitors of metalloproteinase (TIMP)-2, vascular endothelial growth factor (VEGF), extracellular regulated kinase (ERK), Janus kinase (JNK), phosphotidyl-inositol-3 kinase/protein kinase B (PI3K/AKT), and NF-κB transcriptional activity [56]. In addition, diosgenin was reported to inhibit the expression of E-cadherin, integrin 5a and 6b, invasion, migration, and angiogenesis in hypoxia-sensitive BGC-823 gastric cancer cells [71]. Diosgenin was reported to inhibit proliferation of ER-positive MCF-7 breast cancer cells by the upregulation of the p53 tumor suppressor gene and activation of caspase 3, while it downregulated BCL2 in ER negative MDA-MB-231 triple-negative breast cancer cells [72]. Diosgenin, either alone, or in combination with thymoquinone, inhibited A431 and Hep2 squamous cell carcinoma cell proliferation, increased the Bax/Bcl2 ratio, and induced caspase 3-mediated apoptosis [59] (Table 2). The potential effect of diosgenin on NF-κB and STAT3 signaling pathways in tumor cells, is shown in Figure 3.
Table 2.
In vitro anti-cancer effects of diosgenin.
| Cancer model | Cell Lines | Diosgenin Dose | Molecular Target | References |
|---|---|---|---|---|
| Breast carcinoma | Estrogen receptor positive and estrogen receptor negative human breast cancer MCF-7 and MDA 231 cells | 20 μM and 30 μM | Inhibition of cell proliferation Induces apoptosis |
[72] |
| MDA-MB-231 breast cancer cells | 20 μM, 40 μM, and 60 μM | Downregulation of Bcl2 | [57] | |
| Her2 over-expressing breast cancer cells | 5–20 μM | Modulation of Akt, mTOR, and JNK phosphorylation | [53] | |
| MCF-7 breast cancer cells | 20 μM and 40 μM | Upregulation of p53 tumor suppressor gene | [73] | |
| Hepatocellular carcinoma | C3A, HUH-7, and HepG2 cells | 50 μM and 100 μM | Downregulation of STAT3 signaling pathway Upregulates SH-PTP2 expression Induces apoptosis Potentiates the apoptotic effects of doxorubicin and paclitaxel |
[54] |
| Prostate carcinoma | PC3 cells | 5 μM, 10 μM, and 20 μM | Downregulates NF-κB signaling pathway Inhibits matrix metalloproteinases Inhibits invasion and migration of cells |
[56] |
| Osteosarcoma | 1547 cells | 40 μM, 80 μM, and 100 μM | Inhibits cell proliferation Induces apoptosis |
[55] |
| 1547 cells | 40 μM | Inhibits cell proliferation Induces apoptosis Upregulation of p53 tumor suppressor gene |
[74] | |
| Human erythroleukemia | HEL cells, K562 cells | 40 μM | Inhibits NF-κB signaling pathway | [75] |
| HEL cells | 40 μM | Inhibits proliferation Induces apoptosis Upregulation of p21 |
[76] | |
| Human Laryngocarcinoma Human Melanoma | HEp-2 cells M4Beu cells |
40 μM | Inhibits cell proliferation Induces caspase-3 dependent apoptosis Upregulates p53 tumor suppressor gene |
[77] |
| Human cancer cells | Human epithelial carcinoma cell line (A431), human NSCLC cell line (A549), human ovarian cancer cell line (A2780), Human erythroleukemia (K562) and Dukes’ type C, colorectal adenocarcinoma (HCT-15) | 10 mmol/L | Induces apoptosis via mitochondrial dependent pathway | [78] |
| Multiple myeloma (U266), leukemia (U937), and breast cancer (MCF-7) | 50 μM and 100 μM | Inhibits NF-κB signaling pathway | [58] |
4. In Vivo Anti-Cancer Effects of Diosgenin
In addition to in vitro inhibition of cancer cell proliferation by dietary fenugreek seeds and its bioactive constituent diosgenin, several studies have provided evidence that diosgenin is a potent inhibitor of tumor growth in vivo in rodent models of cancer. In a rat colorectal tumor model, administration of diosgenin, given during the promotional stage, reduced azoxymethane (AOM)-induced colonic aberrant crypt foci formation [79]. Similarly, Malisetty et al., showed that diosgenin, at a dose of 15 mg/kg, significantly suppressed both the incidence and invasive potential of AOM-induced rat colon adenocarcinoma mass by 60% and colon tumor multiplicity (adenocarcinomas/rat) by 68% [80]. However, in the murine model of AOM/dextran sodium sulfate-induced colon aberrant crypt foci, diosgenin at doses of 20, 100 and 200 mg/kg b.w. in the diet did not reduce adenocarcinoma mass; nonetheless, a significant reduction in tumor multiplicity was observed with all three doses tested [81]. In another study, diosgenin (at a dose of 10 mg/kg b.w. administered intra-tumorally) significantly inhibited the growth of MCF-7 and MDA-MB-231 human breast cancer xenografts in mice [72]. In another study using inbred T739 mice, diosgenin was shown to significantly inhibit the growth of mouse LA795 lung adenocarcinoma tumors by 33.94% [82]. Diosgenin, at a dose of 80 mg/kg administered by oral gavage, was reported to inhibit the growth of oral tumors in a DMBA-induced hamster buccal pouch model [64]. Diosgenin, in combination with thymoquinone, exhibited significant tumor growth inhibition in a mice xenograft model [59]. Therefore, diosgenin modulates multiple targets and suppresses tumor growth in preclinical models of cancer. However, diosgenin’s poor solubility in organic solvents and its lack of bioavailability greatly hinder its translational process as a therapeutic compound. Further clinical trials are required to evaluate its potential either as a preventive or therapeutic anti-cancer agent.
5. Semisynthetic Derivatives of Diosgenin That Exhibit Anti-Cancer Activity
Diosgenin is used in the pharmaceutical industry as the main precursor in the synthesis of steroids [83]. It has the ability to penetrate cell membranes and bind to specific receptors [84]. Steroidal sapogenins are bioactive molecules that have shown exceptional antiproliferative activity against several human cancer cells. By making specific changes in the steroidal structure of diosgenin, it can affect its biological activity. In a recent report, using diosgenin as the parent molecule, the authors synthesized two novel steroidal oxime compounds that showed significant antiproliferative activity on cervical cancer cells and human lymphocytes. These compounds induced apoptosis and activated caspase 3 [85]. In another study Mohammad et al., reported on the anti-proliferative activity of diosgenin and its semi-synthetic derivatives against breast (HBL-100), colon (HCT-116 and HT-19) and lung (A549) cancer cells. A structure-activity relationship study revealed that the potent anti-proliferative activity was mainly attributed to the analogs with the simple phenyl R moiety or electron-withdrawing ortho-substituted R moieties attached to the parent diosgenin [86]. In another study, diosgenin was used as a parent compound to synthesize 1α-hydroxysolasodine; it showed significant anti-cancer activity against prostate cancer (PC3), cervical carcinoma (HeLa), and hepatocellular carcinoma (HepG2) cells [87]. Twelve different analogs of diosgenin containing a long chain fatty acid/ester of diosgenin-7-ketoxime exhibited anti-cancer activity when tested against a panel of cancer cell lines. Compound 16 in this series exhibited potent anti-proliferative activity against DU145 prostate cancer cells, which was associated to the suppression of lipopolysaccharide-induced activation of TNF-α and IL6. The compound was also identified as safe, with a maximum tolerated dose of 300 mg/kg in Swiss albino mice [88]. In a recent article by Ghosh et al., they reported the synthesis of diosgenin functionalized iron oxide nanoparticles that exhibited anti-breast cancer activity by inhibiting proliferation and migration, and by inducing apoptosis [89].
6. Conclusions and Future Perspectives
Compounds derived either from medicinal or dietary plant sources embrace distinct advantages, such as novel bioactive structures, low toxicity, and being multi-targeted in abrogating oncogenic processes; thereby, they may form the source of improved therapeutic options. A vast body of pre-clinical experimental evidence suggests that diosgenin has great potential as an anti-cancer agent. In this review we have compiled and analyzed the role of diosgenin in modulating various oncogenic transcription factors and intracellular molecular targets that drive tumor initiation, progression and metastasis. It is well known that the majority of cancers are a consequence of chronic inflammation, infection, dysfunctional cell death mechanisms, and deregulation of cell cycle molecules. The ability of diosgenin to prevent carcinogenesis by acting as an anti-oxidant and anti-inflammatory agent, and its ability to induce apoptosis of cancer cells, suggests that it can be useful as an anti-carcinogenic agent. Due to the complexities in the cellular processes involved, several new studies need to be conducted to decipher the exact molecular targets that can be exploited to prevent cancer progression. Interestingly, there are 12 reported clinical trials on fenugreek seed extract on a variety of human ailments, as reported in www.clinicaltrials.gov. However, to date, there are no cancer-related clinical trials reported either on diosgenin or on fenugreek seed extract. Several novel synthetic diosgenin derivatives have been shown to improve its anti-cancer efficacy. Several nano-formulations and delivery systems of diosgenin are also shown to improve its bioavailability. In conclusion, several challenges such as developing novel delivery systems, pharmaceutical formulations, and semi-synthetic derivatives that are water soluble, need to be overcome to uncover diosgenin’s benefits either as a chemopreventive or therapeutic agent.
Acknowledgments
A.P.K. was supported by grants from National Medical Research Council of Singapore, Medical Science Cluster, Yong Loo Lin School of Medicine, National University of Singapore and by the National Research Foundation Singapore and the Singapore Ministry of Education under its Research Centers of Excellence initiative to Cancer Science Institute of Singapore, National University of Singapore.
Author Contributions
G.S. and M.K.S. wrote the paper, F.A., A.P.K., S.W., M.M. and A.B. critically analyzed and revised the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Siegel R.L., Miller K.D., Jemal A. Cancer statistics, 2017. CA Cancer J. Clin. 2017;67:7–30. doi: 10.3322/caac.21387. [DOI] [PubMed] [Google Scholar]
- 2.Hanahan D., Weinberg R.A. The hallmarks of cancer. Cell. 2000;100:57–70. doi: 10.1016/S0092-8674(00)81683-9. [DOI] [PubMed] [Google Scholar]
- 3.Hanahan D., Weinberg R.A. Hallmarks of cancer: The next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 4.Hanahan D. Rethinking the war on cancer. Lancet. 2014;383:558–563. doi: 10.1016/S0140-6736(13)62226-6. [DOI] [PubMed] [Google Scholar]
- 5.Floor S.L., Dumont J.E., Maenhaut C., Raspe E. Hallmarks of cancer: Of all cancer cells, all the time? Trends Mol. Med. 2012;18:509–515. doi: 10.1016/j.molmed.2012.06.005. [DOI] [PubMed] [Google Scholar]
- 6.Pietras K., Ostman A. Hallmarks of cancer: Interactions with the tumor stroma. Exp. Cell Res. 2010;316:1324–1331. doi: 10.1016/j.yexcr.2010.02.045. [DOI] [PubMed] [Google Scholar]
- 7.Massague J., Obenauf A.C. Metastatic colonization by circulating tumour cells. Nature. 2016;529:298–306. doi: 10.1038/nature17038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chaffer C.L., Weinberg R.A. How does multistep tumorigenesis really proceed? Cancer Discov. 2015;5:22–24. doi: 10.1158/2159-8290.CD-14-0788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Iwatsuki M., Mimori K., Yokobori T., Ishi H., Beppu T., Nakamori S., Baba H., Mori M. Epithelial-mesenchymal transition in cancer development and its clinical significance. Cancer Sci. 2010;101:293–299. doi: 10.1111/j.1349-7006.2009.01419.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Almendro V., Marusyk A., Polyak K. Cellular heterogeneity and molecular evolution in cancer. Annu. Rev. Pathol. 2013;8:277–302. doi: 10.1146/annurev-pathol-020712-163923. [DOI] [PubMed] [Google Scholar]
- 11.Steeg P.S. Targeting metastasis. Nat. Rev. Cancer. 2016;16:201–218. doi: 10.1038/nrc.2016.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang Z., Dabrosin C., Yin X., Fuster M.M., Arreola A., Rathmell W.K., Generali D., Nagaraju G.P., El-Rayes B., Ribatti D., et al. Broad targeting of angiogenesis for cancer prevention and therapy. Semin. Cancer Biol. 2015;35:S224–S243. doi: 10.1016/j.semcancer.2015.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sethi G., Ahn K.S., Aggarwal B.B. Targeting nuclear factor-kappa B activation pathway by thymoquinone: Role in suppression of antiapoptotic gene products and enhancement of apoptosis. Mol. Cancer Res. 2008;6:1059–1070. doi: 10.1158/1541-7786.MCR-07-2088. [DOI] [PubMed] [Google Scholar]
- 14.Tang C.H., Sethi G., Kuo P.L. Novel medicines and strategies in cancer treatment and prevention. BioMed Res. Int. 2014;2014:474078. doi: 10.1155/2014/474078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ahn K.S., Sethi G., Aggarwal B.B. Nuclear factor-kappa B: From clone to clinic. Curr. Mol. Med. 2007;7:619–637. doi: 10.2174/156652407782564363. [DOI] [PubMed] [Google Scholar]
- 16.Chai E.Z., Shanmugam M.K., Arfuso F., Dharmarajan A., Wang C., Kumar A.P., Samy R.P., Lim L.H., Wang L., Goh B.C., et al. Targeting transcription factor stat3 for cancer prevention and therapy. Pharmacol. Ther. 2016;162:86–97. doi: 10.1016/j.pharmthera.2015.10.004. [DOI] [PubMed] [Google Scholar]
- 17.Li F., Zhang J., Arfuso F., Chinnathambi A., Zayed M.E., Alharbi S.A., Kumar A.P., Ahn K.S., Sethi G. NF-kappaB in cancer therapy. Arch. Toxicol. 2015;89:711–731. doi: 10.1007/s00204-015-1470-4. [DOI] [PubMed] [Google Scholar]
- 18.Shanmugam M.K., Kannaiyan R., Sethi G. Targeting cell signaling and apoptotic pathways by dietary agents: Role in the prevention and treatment of cancer. Nutr. Cancer. 2011;63:161–173. doi: 10.1080/01635581.2011.523502. [DOI] [PubMed] [Google Scholar]
- 19.Deorukhkar A., Krishnan S., Sethi G., Aggarwal B.B. Back to basics: How natural products can provide the basis for new therapeutics. Expert Opin. Investig. Drugs. 2007;16:1753–1773. doi: 10.1517/13543784.16.11.1753. [DOI] [PubMed] [Google Scholar]
- 20.Kneller R. The importance of new companies for drug discovery: Origins of a decade of new drugs. Nat. Rev. Drug Discov. 2010;9:867–882. doi: 10.1038/nrd3251. [DOI] [PubMed] [Google Scholar]
- 21.Bishayee A., Sethi G. Bioactive natural products in cancer prevention and therapy: Progress and promise. Semin. Cancer Biol. 2016;40–41:1–3. doi: 10.1016/j.semcancer.2016.08.006. [DOI] [PubMed] [Google Scholar]
- 22.Newman D.J., Cragg G.M. Natural products as sources of new drugs from 1981 to 2014. J. Nat. Prod. 2016;79:629–661. doi: 10.1021/acs.jnatprod.5b01055. [DOI] [PubMed] [Google Scholar]
- 23.Newman D.J. Developing natural product drugs: Supply problems and how they have been overcome. Pharmacol. Ther. 2016;162:1–9. doi: 10.1016/j.pharmthera.2015.12.002. [DOI] [PubMed] [Google Scholar]
- 24.Newman D.J. Bioactive cyclic molecules and drug design. Expert Opin. Drug Discov. 2018;13:379–385. doi: 10.1080/17460441.2018.1445085. [DOI] [PubMed] [Google Scholar]
- 25.Shanmugam M.K., Lee J.H., Chai E.Z., Kanchi M.M., Kar S., Arfuso F., Dharmarajan A., Kumar A.P., Ramar P.S., Looi C.Y., et al. Cancer prevention and therapy through the modulation of transcription factors by bioactive natural compounds. Semin. Cancer Biol. 2016;40–41:35–47. doi: 10.1016/j.semcancer.2016.03.005. [DOI] [PubMed] [Google Scholar]
- 26.Shanmugam M.K., Nguyen A.H., Kumar A.P., Tan B.K., Sethi G. Targeted inhibition of tumor proliferation, survival, and metastasis by pentacyclic triterpenoids: Potential role in prevention and therapy of cancer. Cancer Lett. 2012;320:158–170. doi: 10.1016/j.canlet.2012.02.037. [DOI] [PubMed] [Google Scholar]
- 27.Shanmugam M.K., Warrier S., Kumar A.P., Sethi G., Arfuso F. Potential role of natural compounds as anti-angiogenic agents in cancer. Curr. Vasc. Pharmacol. 2017;15:503–519. doi: 10.2174/1570161115666170713094319. [DOI] [PubMed] [Google Scholar]
- 28.Mishra K.P., Ganju L., Sairam M., Banerjee P.K., Sawhney R.C. A review of high throughput technology for the screening of natural products. Biomed. Pharmacother. 2008;62:94–98. doi: 10.1016/j.biopha.2007.06.012. [DOI] [PubMed] [Google Scholar]
- 29.Cragg G.M., Grothaus P.G., Newman D.J. Impact of natural products on developing new anti-cancer agents. Chem. Rev. 2009;109:3012–3043. doi: 10.1021/cr900019j. [DOI] [PubMed] [Google Scholar]
- 30.Harvey A.L., Edrada-Ebel R., Quinn R.J. The re-emergence of natural products for drug discovery in the genomics era. Nat. Rev. Drug Discov. 2015;14:111–129. doi: 10.1038/nrd4510. [DOI] [PubMed] [Google Scholar]
- 31.Newman D.J. Natural products as leads to potential drugs: An old process or the new hope for drug discovery? J. Med. Chem. 2008;51:2589–2599. doi: 10.1021/jm0704090. [DOI] [PubMed] [Google Scholar]
- 32.Chai E.Z., Siveen K.S., Shanmugam M.K., Arfuso F., Sethi G. Analysis of the intricate relationship between chronic inflammation and cancer. Biochem. J. 2015;468:1–15. doi: 10.1042/BJ20141337. [DOI] [PubMed] [Google Scholar]
- 33.Sung B., Prasad S., Yadav V.R., Aggarwal B.B. Cancer cell signaling pathways targeted by spice-derived nutraceuticals. Nutr. Cancer. 2012;64:173–197. doi: 10.1080/01635581.2012.630551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Newman D.J., Cragg G.M. Current status of marine-derived compounds as warheads in anti-tumor drug candidates. Mar. Drugs. 2017;15:99. doi: 10.3390/md15040099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Newman D.J., Cragg G.M. Drugs and drug candidates from marine sources: An assessment of the current “state of play”. Planta Med. 2016;82:775–789. doi: 10.1055/s-0042-101353. [DOI] [PubMed] [Google Scholar]
- 36.El Bairi K., Ouzir M., Agnieszka N., Khalki L. Anticancer potential of trigonella foenum graecum: Cellular and molecular targets. Biomed. Pharmacother. 2017;90:479–491. doi: 10.1016/j.biopha.2017.03.071. [DOI] [PubMed] [Google Scholar]
- 37.Shabbeer S., Sobolewski M., Anchoori R.K., Kachhap S., Hidalgo M., Jimeno A., Davidson N., Carducci M.A., Khan S.R. Fenugreek: A naturally occurring edible spice as an anticancer agent. Cancer Biol. Ther. 2009;8:272–278. doi: 10.4161/cbt.8.3.7443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Aggarwal B.B., Kunnumakkara A.B., Harikumar K.B., Tharakan S.T., Sung B., Anand P. Potential of spice-derived phytochemicals for cancer prevention. Planta Med. 2008;74:1560–1569. doi: 10.1055/s-2008-1074578. [DOI] [PubMed] [Google Scholar]
- 39.Kenny O., Smyth T.J., Hewage C.M., Brunton N.P. Antioxidant properties and quantitative UPLC-MS analysis of phenolic compounds from extracts of Fenugreek (Trigonella foenum-graecum) seeds and bitter melon (Momordica charantia) fruit. Food Chem. 2013;141:4295–4302. doi: 10.1016/j.foodchem.2013.07.016. [DOI] [PubMed] [Google Scholar]
- 40.Wani S.A., Kumar P. Fenugreek: A review on its nutraceutical properties and utilization in various food products. J. Saudi Soc. Agric. Sci. 2018;17:97–106. doi: 10.1016/j.jssas.2016.01.007. [DOI] [Google Scholar]
- 41.Kim J.K., Park S.U. An update on the biological and pharmacological activities of diosgenin. EXCLI J. 2018;17:24–28. doi: 10.17179/excli2017-894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bahmani M., Shirzad H., Mirhosseini M., Mesripour A., Rafieian-Kopaei M. A review on ethnobotanical and therapeutic uses of Fenugreek (Trigonella foenum-graceum L.) J. Evid. Based Complement. Altern. Med. 2016;21:53–62. doi: 10.1177/2156587215583405. [DOI] [PubMed] [Google Scholar]
- 43.Srinivasan K. Fenugreek (Trigonella foenum-graecum): A review of health beneficial physiological effects. Food Rev. Int. 2006;22:203–224. doi: 10.1080/87559120600586315. [DOI] [Google Scholar]
- 44.Jesus M., Martins A.P., Gallardo E., Silvestre S. Diosgenin: Recent highlights on pharmacology and analytical methodology. J. Anal. Methods Chem. 2016;2016:4156293. doi: 10.1155/2016/4156293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Yang S.F., Weng C.J., Sethi G., Hu D.N. Natural bioactives and phytochemicals serve in cancer treatment and prevention. Evid. Based Complement. Altern. Med. 2013;2013:698190. doi: 10.1155/2013/698190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Djerassi C., Rosenkranz G., Pataki J., Kaufmann S. Steroids, xxvii. Synthesis of allopregnane-3beta, 11beta, 17alpha-, 20beta, 21-pentol from cortisone and diosgenin. J. Biol. Chem. 1952;194:115–118. [PubMed] [Google Scholar]
- 47.Patel K., Gadewar M., Tahilyani V., Patel D.K. A review on pharmacological and analytical aspects of diosmetin: A concise report. Chin. J. Integr. Med. 2013;19:792–800. doi: 10.1007/s11655-013-1595-3. [DOI] [PubMed] [Google Scholar]
- 48.Chen Y., Tang Y.M., Yu S.L., Han Y.W., Kou J.P., Liu B.L., Yu B.Y. Advances in the pharmacological activities and mechanisms of diosgenin. Chin. J. Nat. Med. 2015;13:578–587. doi: 10.1016/S1875-5364(15)30053-4. [DOI] [PubMed] [Google Scholar]
- 49.Tang Y., Yi T., Chen H., Zhao Z., Liang Z., Chen H. Quantitative comparison of multiple components in dioscorea nipponica and d. Panthaica by ultra-high performance liquid chromatography coupled with quadrupole time-of-flight mass spectrometry. Phytochem. Anal. 2013;24:413–422. doi: 10.1002/pca.2428. [DOI] [PubMed] [Google Scholar]
- 50.Tang Y.N., Pang Y.X., He X.C., Zhang Y.Z., Zhang J.Y., Zhao Z.Z., Yi T., Chen H.B. UPLC-QTOF-MS identification of metabolites in rat biosamples after oral administration of dioscorea saponins: A comparative study. J. Ethnopharmacol. 2015;165:127–140. doi: 10.1016/j.jep.2015.02.017. [DOI] [PubMed] [Google Scholar]
- 51.Ciura J., Szeliga M., Grzesik M., Tyrka M. Next-generation sequencing of representational difference analysis products for identification of genes involved in diosgenin biosynthesis in Fenugreek (Trigonella foenum-graecum) Planta. 2017;245:977–991. doi: 10.1007/s00425-017-2657-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Vaidya K., Ghosh A., Kumar V., Chaudhary S., Srivastava N., Katudia K., Tiwari T., Chikara S.K. De novo transcriptome sequencing in Trigonella foenum-graecum L. to identify genes involved in the biosynthesis of diosgenin. Plant. Genome. 2013;6:1–11. doi: 10.3835/plantgenome2012.08.0021. [DOI] [Google Scholar]
- 53.Chiang C.T., Way T.D., Tsai S.J., Lin J.K. Diosgenin, a naturally occurring steroid, suppresses fatty acid synthase expression in HER2-overexpressing breast cancer cells through modulating Akt, mTOR and JNK phosphorylation. FEBS Lett. 2007;581:5735–5742. doi: 10.1016/j.febslet.2007.11.021. [DOI] [PubMed] [Google Scholar]
- 54.Li F., Fernandez P.P., Rajendran P., Hui K.M., Sethi G. Diosgenin, a steroidal saponin, inhibits STAT3 signaling pathway leading to suppression of proliferation and chemosensitization of human hepatocellular carcinoma cells. Cancer Lett. 2010;292:197–207. doi: 10.1016/j.canlet.2009.12.003. [DOI] [PubMed] [Google Scholar]
- 55.Moalic S., Liagre B., Corbiere C., Bianchi A., Dauca M., Bordji K., Beneytout J.L. A plant steroid, diosgenin, induces apoptosis, cell cycle arrest and cox activity in osteosarcoma cells. FEBS Lett. 2001;506:225–230. doi: 10.1016/S0014-5793(01)02924-6. [DOI] [PubMed] [Google Scholar]
- 56.Chen P.S., Shih Y.W., Huang H.C., Cheng H.W. Diosgenin, a steroidal saponin, inhibits migration and invasion of human prostate cancer pc-3 cells by reducing matrix metalloproteinases expression. PLoS ONE. 2011;6:e20164. doi: 10.1371/journal.pone.0020164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Raju J., Rao C.V. Diosgenin, a steroid saponin constituent of yams and fenugreek: Emerging evidence for applications. In: Rasooli I., editor. Bioactive Compounds Medicine in Phytomedicine. InTech.; London, UK: 2012. pp. 125–142. [Google Scholar]
- 58.Shishodia S., Aggarwal B.B. Diosgenin inhibits osteoclastogenesis, invasion, and proliferation through the downregulation of Akt, I kappa B kinase activation and NF-kappa B-regulated gene expression. Oncogene. 2006;25:1463–1473. doi: 10.1038/sj.onc.1209194. [DOI] [PubMed] [Google Scholar]
- 59.Das S., Dey K.K., Dey G., Pal I., Majumder A., MaitiChoudhury S., kundu S.C., Mandal M. Antineoplastic and apoptotic potential of traditional medicines thymoquinone and diosgenin in squamous cell carcinoma. PLoS ONE. 2012;7:e46641. doi: 10.1371/journal.pone.0046641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Lepage C., Leger D.Y., Bertrand J., Martin F., Beneytout J.L., Liagre B. Diosgenin induces death receptor-5 through activation of p38 pathway and promotes trail-induced apoptosis in colon cancer cells. Cancer Lett. 2011;301:193–202. doi: 10.1016/j.canlet.2010.12.003. [DOI] [PubMed] [Google Scholar]
- 61.Romero-Hernandez L.L., Merino-Montiel P., Montiel-Smith S., Meza-Reyes S., Vega-Baez J.L., Abasolo I., Schwartz S., Jr., Lopez O., Fernandez-Bolanos J.G. Diosgenin-based thio(seleno)ureas and triazolyl glycoconjugates as hybrid drugs. Antioxidant and antiproliferative profile. Eur. J. Med. Chem. 2015;99:67–81. doi: 10.1016/j.ejmech.2015.05.018. [DOI] [PubMed] [Google Scholar]
- 62.Cai B., Liao A., Lee K.K., Ban J.S., Yang H.S., Im Y.J., Chun C. Design, synthesis of methotrexate-diosgenin conjugates and biological evaluation of their effect on methotrexate transport-resistant cells. Steroids. 2016;116:45–51. doi: 10.1016/j.steroids.2016.10.006. [DOI] [PubMed] [Google Scholar]
- 63.Jiang S., Fan J., Wang Q., Ju D., Feng M., Li J., Guan Z.B., An D., Wang X., Ye L. Diosgenin induces ros-dependent autophagy and cytotoxicity via mtor signaling pathway in chronic myeloid leukemia cells. Phytomedicine. 2016;23:243–252. doi: 10.1016/j.phymed.2016.01.010. [DOI] [PubMed] [Google Scholar]
- 64.Jagadeesan J., Langeswaran K., Gowthamkumar S., Balasubramanian M.P. Diosgenin exhibits beneficial efficiency on human mammary carcinoma cell line MCF-7 and against N-nitroso-N-methylurea (NMU) induced experimental mammary carcinoma. Biomed. Prev. Nutr. 2013;3:381–388. doi: 10.1016/j.bionut.2013.06.009. [DOI] [Google Scholar]
- 65.Albini A., Tosetti F., Li V.W., Noonan D.M., Li W.W. Cancer prevention by targeting angiogenesis. Nat. Rev. Clin. Oncol. 2012;9:498–509. doi: 10.1038/nrclinonc.2012.120. [DOI] [PubMed] [Google Scholar]
- 66.Folkman J. Tumor angiogenesis: Therapeutic implications. N. Engl. J. Med. 1971;285:1182–1186. doi: 10.1056/NEJM197111182852108. [DOI] [PubMed] [Google Scholar]
- 67.Folkman J. Role of angiogenesis in tumor growth and metastasis. Semin. Oncol. 2002;29:15–18. doi: 10.1053/sonc.2002.37263. [DOI] [PubMed] [Google Scholar]
- 68.Folkman J. Angiogenesis: An organizing principle for drug discovery? Nat. Rev. Drug Discov. 2007;6:273–286. doi: 10.1038/nrd2115. [DOI] [PubMed] [Google Scholar]
- 69.Selim S., Al Jaouni S. Anti-inflammatory, antioxidant and antiangiogenic activities of diosgenin isolated from traditional medicinal plant, costus speciosus (Koen ex.Retz.) Sm. Nat. Prod. Res. 2016;30:1830–1833. doi: 10.1080/14786419.2015.1065493. [DOI] [PubMed] [Google Scholar]
- 70.He Z., Chen H., Li G., Zhu H., Gao Y., Zhang L., Sun J. Diosgenin inhibits the migration of human breast cancer MDA-MB-231 cells by suppressing VAV2 activity. Phytomedicine. 2014;21:871–876. doi: 10.1016/j.phymed.2014.02.002. [DOI] [PubMed] [Google Scholar]
- 71.Mao Z.J., Tang Q.J., Zhang C.A., Qin Z.F., Pang B., Wei P.K., Liu B., Chou Y.N. Anti-proliferation and anti-invasion effects of diosgenin on gastric cancer BGC-823 cells with HIF-1alpha shrnas. Int. J. Mol. Sci. 2012;13:6521–6533. doi: 10.3390/ijms13056521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Srinivasan S., Koduru S., Kumar R., Venguswamy G., Kyprianou N., Damodaran C. Diosgenin targets AKT-mediated prosurvival signaling in human breast cancer cells. Int. J. Cancer. 2009;125:961–967. doi: 10.1002/ijc.24419. [DOI] [PubMed] [Google Scholar]
- 73.Sowmyalakshmi S., Ranga R., Gairola C.G., Chendil D. Effect of diosgenin (Fenugreek) on breast cancer cells. Proc. Am. Assoc. Cancer Res. 2005;46:1382. [Google Scholar]
- 74.Corbiere C., Liagre B., Bianchi A., Bordji K., Dauca M., Netter P., Beneytout J.L. Different contribution of apoptosis to the antiproliferative effects of diosgenin and other plant steroids, hecogenin and tigogenin, on human 1547 osteosarcoma cells. Int. J. Oncol. 2003;22:899–905. doi: 10.3892/ijo.22.4.899. [DOI] [PubMed] [Google Scholar]
- 75.Liagre B., Bertrand J., Leger D.Y., Beneytout J.L. Diosgenin, a plant steroid, induces apoptosis in COX-2 deficient K562 cells with activation of the p38 map kinase signalling and inhibition of NF-kappaB binding. Int. J. Mol. Med. 2005;16:1095–1101. doi: 10.3892/ijmm.16.6.1095. [DOI] [PubMed] [Google Scholar]
- 76.Leger D.Y., Liagre B., Corbiere C., Cook-Moreau J., Beneytout J.L. Diosgenin induces cell cycle arrest and apoptosis in hel cells with increase in intracellular calcium level, activation of CPLA2 and COX-2 overexpression. Int. J. Oncol. 2004;25:555–562. doi: 10.3892/ijo.25.3.555. [DOI] [PubMed] [Google Scholar]
- 77.Corbiere C., Liagre B., Terro F., Beneytout J.L. Induction of antiproliferative effect by diosgenin through activation of p53, release of apoptosis-inducing factor (AIF) and modulation of caspase-3 activity in different human cancer cells. Cell. Res. 2004;14:188–196. doi: 10.1038/sj.cr.7290219. [DOI] [PubMed] [Google Scholar]
- 78.Wang S.L., Cai B., Cui C.B., Liu H.W., Wu C.F., Yao X.S. Diosgenin-3-O-alpha-l-rhamnopyranosyl-(1→4)-beta-d-glucopyranoside obtained as a new anticancer agent from dioscorea futschauensis induces apoptosis on human colon carcinoma HCT-15 cells via mitochondria-controlled apoptotic pathway. J. Asian Nat. Prod. Res. 2004;6:115–125. doi: 10.1080/1028602031000147357. [DOI] [PubMed] [Google Scholar]
- 79.Raju J., Patlolla J.M., Swamy M.V., Rao C.V. Diosgenin, a steroid saponin of Trigonella foenum graecum (Fenugreek), inhibits azoxymethane-induced aberrant crypt foci formation in F344 rats and induces apoptosis in HT-29 human colon cancer cells. Cancer Epidemiol. Biomarkers Prev. 2004;13:1392–1398. [PubMed] [Google Scholar]
- 80.Malisetty V.S., Patlolla J.M.R., Raju J., Marcus L.A., Choi C.L., Rao C.V. Chemoprevention of colon cancer by diosgenin, a steroidal saponin constituent of fenugreek. Proc. Am. Assoc. Cancer Res. 2005;46:2473. [Google Scholar]
- 81.Miyoshi N., Nagasawa T., Mabuchi R., Yasui Y., Wakabayashi K., Tanaka T., Ohshima H. Chemoprevention of azoxymethane/dextran sodium sulfate-induced mouse colon carcinogenesis by freeze-dried yam sanyaku and its constituent diosgenin. Cancer Prev. Res. (Phila.) 2011;4:924–934. doi: 10.1158/1940-6207.CAPR-10-0279. [DOI] [PubMed] [Google Scholar]
- 82.Yan L.L., Zhang Y.J., Gao W.Y., Man S.L., Wang Y. In vitro and in vivo anticancer activity of steroid saponins of paris polyphylla var. Yunnanensis. Exp. Oncol. 2009;31:27–32. [PubMed] [Google Scholar]
- 83.Marker R.E., Sterols C.V. The preparation of testosterone and related compounds from sarsasapogenin and diosgenin. J. Am. Chem. Soc. 1940;62:2543–2547. doi: 10.1021/ja01866a077. [DOI] [Google Scholar]
- 84.Tietze L.F., Bell H.P., Chandrasekhar S. Natural product hybrids as new leads for drug discovery. Angew. Chem. Int. Ed. 2003;42:3996–4028. doi: 10.1002/anie.200200553. [DOI] [PubMed] [Google Scholar]
- 85.Sanchez-Sanchez L., Hernandez-Linares M.G., Escobar M.L., Lopez-Munoz H., Zenteno E., Fernandez-Herrera M.A., Guerrero-Luna G., Carrasco-Carballo A., Sandoval-Ramirez J. Antiproliferative, cytotoxic, and apoptotic activity of steroidal oximes in cervicouterine cell lines. Molecules. 2016;21:1533. doi: 10.3390/molecules21111533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Masood Ur R., Mohammad Y., Fazili K.M., Bhat K.A., Ara T. Synthesis and biological evaluation of novel 3-O-tethered triazoles of diosgenin as potent antiproliferative agents. Steroids. 2017;118:1–8. doi: 10.1016/j.steroids.2016.11.003. [DOI] [PubMed] [Google Scholar]
- 87.Liu C., Xie F., Zhao G.D., Wang D.F., Lou H.X., Liu Z.P. Synthetic studies towards 1alpha-hydroxysolasodine from diosgenin and the unexpected tetrahydrofuran ring opening in the birch reduction process. Steroids. 2015;104:214–219. doi: 10.1016/j.steroids.2015.10.006. [DOI] [PubMed] [Google Scholar]
- 88.Hamid A.A., Kaushal T., Ashraf R., Singh A., Chand Gupta A., Prakash O., Sarkar J., Chanda D., Bawankule D.U., Khan F., et al. (22beta,25R)-3beta-hydroxy-spirost-5-en-7-iminoxy-heptanoic acid exhibits anti-prostate cancer activity through caspase pathway. Steroids. 2017;119:43–52. doi: 10.1016/j.steroids.2017.01.001. [DOI] [PubMed] [Google Scholar]
- 89.Ghosh S., More P., Derle A., Kitture R., Kale T., Gorain M., Avasthi A., Markad P., Kundu G.C., Kale S., et al. Diosgenin functionalized iron oxide nanoparticles as novel nanomaterial against breast cancer. J. Nanosci. Nanotechnol. 2015;15:9464–9472. doi: 10.1166/jnn.2015.11704. [DOI] [PubMed] [Google Scholar]