Abstract
Introduction:
Clostridium difficile (CD) is a serious and increasingly prevalent healthcare-associated infection. The pathogenesis of CD infection (CDI) involves the acquisition of CD with a concurrent disruption of the native gut flora. Antibiotics are a major risk although other contributing factors have also been identified. Clinical management combines discontinuation of the offending antibiotic, initiation of CD-specific antibiotic therapy, probiotic agent use, fecal microbiota transplantation (FMT), and surgery as the “last resort” option. The aim of this study is to review short-term clinical results following the implementation of FMT protocol (FMTP) at our community-based university hospital.
Methods:
After obtaining Institutional Review Board and Infection Control Committee approvals, we implemented an institution-wide FMTP for patients diagnosed with CDI. Prospective tracking of all patients receiving FMT between July 1, 2015, and February 1, 2017, was conducted using REDCap™ electronic data capture system. According to the FMTP, indications for FMT included (a) three or more CDI recurrences, (b) two or more hospital admissions with severe CDI, or (c) first episode of complicated CDI (CCDI). Risk factors for initial infection and for treatment failure were assessed. Patients were followed for at least 3 months to monitor for cure/failure, relapse, and side effects. Frozen 250 mL FMT samples were acquired from OpenBiome (Somerville, MA, USA). After 4 h of thawing, the liquid suspension was applied using colonoscopy, beginning with terminal ileum and proceeding distally toward mid-transverse colon. Monitored clinical parameters included disease severity (Hines VA CDI Severity Score or HVCSS), concomitant medications, number of FMT treatments, non-FMT therapies, cure rates, and mortality. Descriptive statistics were utilized to outline the study results.
Results:
A total of 35 patients (mean age 58.5 years, 69% female) were analyzed, with FMT-attributable primary cure achieved in 30/35 (86%) cases. Within this subgroup, 2/30 (6.7%) patients recurred and were subsequently cured with long-term oral vancomycin. Among five primary FMT failures (14% total sample), 3 (60%) achieved medical cure with long-term oral vancomycin therapy and 2 (40%) required colectomy. For the seven patients who either failed FMT or recurred, long-term vancomycin therapy was curative in all but two cases. For patients with severe CDI (HVCSS ≥3), primary and overall cure rates were 6/10 (60%) and 8/10 (80%), respectively. Patients with CCDI (n = 4) had higher HVCSS (4 vs. 3) and a mortality of 25%. Characteristics of patients who failed initial FMT included older age (70 vs. 57 years), female sex (80% vs. 67%), severe CDI (80% vs. 13%), and active opioid use during the initial infection (60% vs. 37%) and at the time of FMT (60% vs. 27%). The most commonly reported side effect of FMT was loose stools.
Conclusions:
This pilot study supports the efficacy and safety of FMT administration for CDI in the setting of a community-based university hospital. Following FMTP implementation, primary (86%) and overall (94%) nonsurgical cure rates were similar to those reported in other studies. The potential role of opioids as a modulator of CDI warrants further clinical investigation.
Keywords: Clinical protocol implementation, Clostridium difficile, Clostridium difficile infection, fecal microbiota transplantation, infectious colitis
INTRODUCTION
Clostridium difficile (CD) is an anaerobic, Gram-positive, spore-forming, toxin-producing bacillus.[1] The bacterium was first isolated in the 1930s and found to be the cause of antibiotic-associated pseudomembranous colitis (PMC) in the late 1970s.[2,3,4] Approximately 1%–4% of the general population and 7%–26% of hospitalized patients are colonized with CD,[5,6,7,8,9] thus contributing to the phenomenon of “asymptomatic carriage.”[9] Currently, there is no evidence to support active treatment of asymptomatic CD to either reduce symptomatic infections or disease transmission.[10,11] Transmission of CD occurs predominantly through the fecal–oral route.[12] Although lack of colonic microbial diversity plays a major role in the emergence of PMC, the exact mechanism that causes symptomatic infection is still unclear.[3,5,11,13]
Currently, CD is one of the most common healthcare-associated infections in US hospitals.[14] The acquisition of toxigenic CD involves horizontal transmission from the contaminated hands of healthcare personnel or other surfaces with a concurrent disruption of the gut microbiota. The principal virulence factors of CD are toxin A and B that contribute to cytoskeletal damage and disruption of cell–cell junctions, culminating in intestinal epithelial injury and the associated pseudomembranous lesions [Figure 1].[15,16] The prevalence of CD infection (CDI) has increased since the emergence of the BI/NAP/027 strain of CD in the 2000s.[17,18] It is also the most common cause of healthcare-associated infectious diarrhea and has become less responsive to treatment over the past two decades.[19,20] CDI-related mortality increased from 10/1,000,000 to 48/1,000,000 person-years between 1999 and 2007, with >80% of deaths occurring in patients ≥65 years old.[21,22] It is estimated that the annual incidence of CDI is 453,000, with approximately 29,300 deaths,[23] in addition to the attributable economic burden of US $6.3 billion and 2.4 million hospital days.[24]
Figure 1.
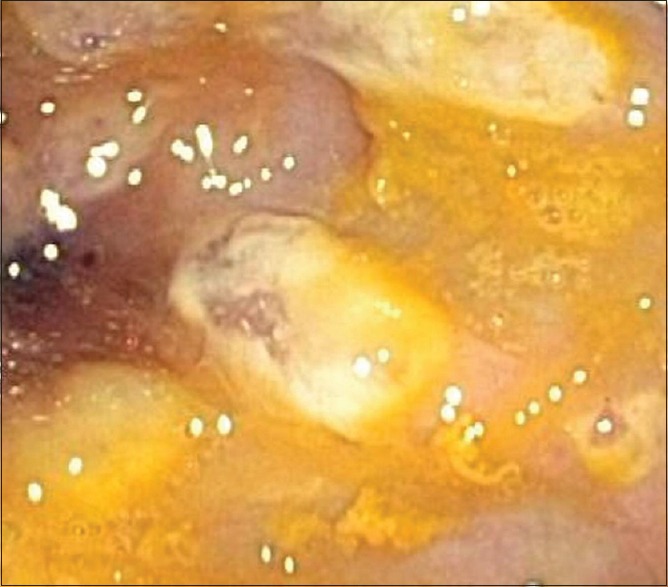
Colonoscopic image showing the presence of pseudomembranes characteristic of Clostridium difficile colitis
Antibiotics disrupt normal colonic microbiota diversity, with clindamycin and carbapenems carrying the highest risk for CDI.[25] Other factors associated with CDI include age >65 years, use of proton pump inhibitors (PPIs), and recent hospital admission.[26] The risk of CDI is greater if the prevalence of CDI at the hospital is high during an admission, a concept known as “colonization pressure.”[27] Inflammatory bowel disease, chemotherapy, ongoing malignancy, chronic kidney and liver disease, pregnancy, HIV infection, chronic corticosteroid treatment, and gastrointestinal (GI) surgery or manipulation (including feeding tubes) are additional risk factors.[6,11,12,28,29,30,31] The recent increase in the incidence of CDI may be due to demographic changes among admitted patients, including older age, multi-morbidity, polypharmacy, as well as the emergence of the BI/NAP/027 strain.[6,32] Recurrent CDI (RCDI) has been associated with continued use of antibiotics, severe initial CDI, older age, multiple comorbidities, and gastric acid suppression therapy.[11,33] About one-third of the recurrences are due to reinfection from a different strain.[34]
The diagnosis of CDI is mainly clinical (e.g., the emergence of abdominal pain and diarrhea) with laboratory confirmation.[7,11,12,35,36] Endoscopy may help estimate disease severity. Imaging studies such as computed tomography provide only limited diagnostic value but are useful in stratifying disease severity when pancolitis is observed [Figure 2].[7,36,37] The clinical spectrum of the disease ranges from mild diarrhea to potentially lethal colitis.[4,32] Leukocytosis >30,000/mL or 50% increase in baseline creatinine is associated with worse prognosis.[35,38]
Figure 2.
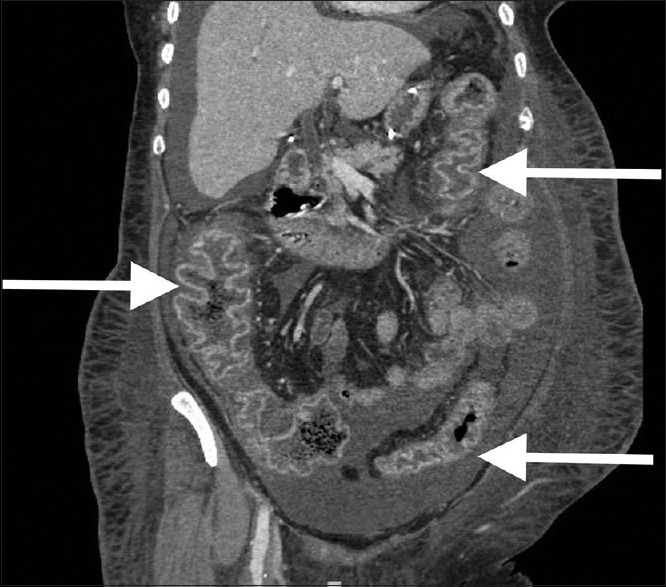
Appearance of pancolitis associated with Clostridium difficile infection on abdominal computed tomography scan. Note the presence of diffuse inflammation in ascending, descending and sigmoid colon (arrows)
Fecal microbiota transplantation
Descriptions of medical applications of fecal microbiota transplantation (FMT) date back to 4th century, with subsequent descriptions between 14th and 17th centuries.[29,39] In the 1950s, Eiseman described a series of four patients with PMC treated effectively with FMT.[40] However, it was not until 1983 that the first documented case of confirmed CDI was cured with FMT.[41] Although retention enema was the usual route for FMT delivery, the use of a nasogastric tube was also reported in early 1990s.[42] More recently, FMT application through self-administered enema or colonoscopy became the preferred approaches.[12,39,43] Over the past two decades, increasing amount of evidence accumulated in support of more routine use of FMT.[44] Major professional societies advocate the use of FMT in the setting of RCDI, with the major source of controversy being the number of recurrences before FMT is to be utilized (e.g., second vs. third).[12,13,35,45,46]
Rationale for the current study
The goal of the current study was to assess the feasibility and efficacy of FMT at our regional health network's community-based university hospital. We sought to describe in detail our early clinical outcomes and the steps involved in FMT protocol (FMTP) implementation, including any associated barriers.
METHODS
After approval by the Institutional Review Board (IRB), we performed a retrospective review of a prospective institutional FMT registry. The study examined FMT administration for the treatment of patients with RCDI, severe CDI, or complicated CDI (CCDI). Infection Control Committee approval was granted before FMT procedures were allowed to proceed. All study-related treatments took place between July 2015 and February 2017. Colonoscopic FMT procedures were performed at the same hospital, by six different physicians, and following the same standardized FMTP.
Before FMT procedure, all patients were screened for hepatitis A, B, C, syphilis, and HIV infections. Stool samples were also sent for routine culture and for ova and parasite assessments. Other considerations included prior history of malignancy and risk factors for immunosuppression. Patients were informed that although associated with high cure rates, FMT is not FDA approved. Disclosure was also made that our understanding of associated side effects is incomplete, including potential risk for transmission of unrecognized infectious disease(s), especially in immunosuppressed patients. After detailed discussion of risks and benefits, informed consent was obtained from each patient before the colonoscopic FMT procedure. Clinical follow-up with the treating physician was mandatory, and further follow-up included a nurse call after 3 months.
Definitions
Diarrhea was defined as three or more loose stools within any given 24-h period, corresponding to Bristol stool chart types 5–7.[35] CDI was defined as the presence of diarrhea plus identification of toxigenic CD by nucleic acid amplified test using polymerase chain reaction.[47]
Primary cure was considered when diarrhea resolved by the 6th day posttreatment or after a negative CDI test in those patients with persistent loose stools after treatment. Therapeutic failure was defined as persistent diarrhea and positive CDI test 6 days posttreatment. Testing for CDI in asymptomatic patients without diarrhea was not performed due to continued shedding of CD spores.[11] There is no standard definition for CDI recurrence, and it is often difficult to distinguish a relapse from reinfection with a different strain. Because most recurrences happen within 2–5 weeks of treatment, some authors consider an episode to represent a recurrence after 8 weeks of treatment.[12,35] We defined recurrence as the presence of diarrhea with a positive CDI test ≥21 days after initial cure.[48] Secondary cure was considered when CDI-associated diarrhea resolved after vancomycin retreatment, with or without repeat FMT.[49] Eradication of antibiotic-resistant CDI using FMT in the setting of recurrence was also considered secondary cure.[50] All patients in the current study were clinically followed for at least 3 months.
Disease severity assessment
To assess disease severity, we used the HVCSS due to its good correlation with severe CDI.[51] This score relies on clinical, laboratory, and radiological variables known to be associated with CDI severity, all obtained within 3 days of documented CDI.[52] In the current study, severe disease was defined as HVCSS ≥3. CCDI was defined as “ongoing clinical deterioration” with organ failure after 72 h of conventional treatment and/or severe colitis.
Fecal microbiota transplantation procedure
The technique used at our institution mirrors other previously published FMTPs using colonoscopy.[13,49,53,54,55] All patients took preprocedure oral vancomycin for CDI. The vancomycin was stopped after FMT, except in patients with CCDI. Although antibiotics and bowel lavage before FMT have been associated with post-FMT relapse,[56] preprocedure bowel preparation was done in all patients using polyethylene glycol. Fecal samples were acquired from OpenBiome (Somerville, Massachusetts, USA), a centralized nonprofit stool bank. Obtained from prescreened unrelated and anonymous donors, specimens consist of 250 mL of frozen stool. Each sample was then thawed for 4 h until the consistency of the suspension permitted aspiration with a syringe. At that time, colonoscopy was performed, and after visualizing the terminal ileum, systematic application of FMT was performed beginning in the terminal ileum and cecum and then continued distally until mid-transverse colon was reached.
Patient inclusion criteria
All study patients had active CDI, were at least 18 years of age, and had a life expectancy of at least 3 months. Patients were offered FMT after presenting with three or more CDI recurrences, having two or more hospital admissions with severe CDI, or experiencing their first episode of CCDI. As outlined above, IRB approval was obtained and each patient signed a separate informed consent before FMT.
Data collection
Centralized data collection was performed using the REDCap Electronic Data Capture system (REDCap Project, Vanderbilt University, Nashville, TN, USA). Variables collected included patient demographics (age and gender), severity of infection (HVCSS), number of CDI recurrences for each patient, history of antibiotic administration, use of PPIs, opioid utilization, history of previous cholecystectomy or colonic surgery, and immunosuppression. In terms of outcomes, each patient's response to treatment was recorded, including morbidity, mortality, clinical FMT efficacy and any treatment-associated side effects.
Statistical analysis
We used descriptive statistics, including frequencies and measures of central tendencies to report study results. Due to lack of comparison groups, no statistical testing was performed.
RESULTS
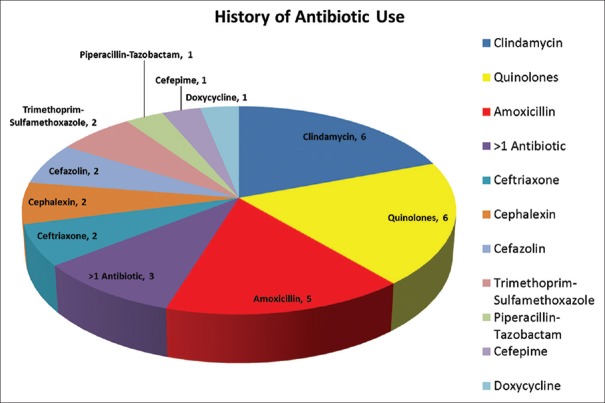
Thirty-five patients underwent FMT for clinical indications approved by our FMTP (e.g., RCDI, severe CDI, or CCDI). Colonoscopic FMT instillation was used in all 35 cases. Mean patient age was 58.6 ± 18.3 years, with females constituting 69% of the study sample (24/35). Overall, 31/35 patients (88.6%) had documented exposure to antibiotics before the CDI diagnosis. The most commonly reported antibiotic types included clindamycin (n = 6), cephalosporins (n = 7) and quinolones (n = 6) [Figure 3]. Five patients (14%) were admitted to the Intensive Care Unit for severe CDI.
Figure 3.
Breakdown of pre-Clostridium difficile infection antibiotic use among study patients
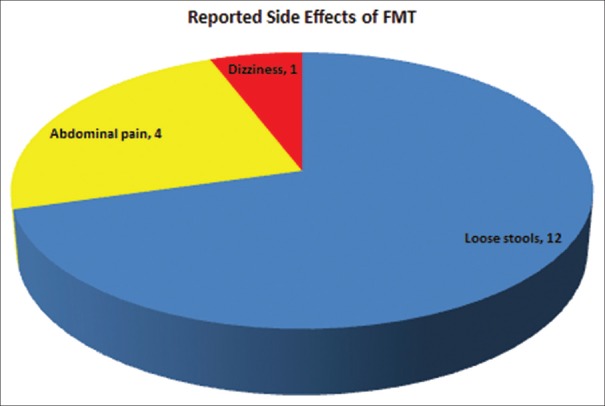
The majority of patients (21/35 or 60%) had ≥3 (mean, 2.7) CDI recurrences before undergoing FMT. Overall, FMT was successful in 33 patients (94%). Primary cure was achieved in 30 cases (86%). Two patients with initial primary cure experienced recurrence and were subsequently cured after prolonged (6 weeks) oral vancomycin treatment. Global study outcome summary is shown in Figure 4. Thirteen patients (37%) reported at least one FMT-related complaint, with four reporting more than one side effect [Figure 5].
Figure 4.
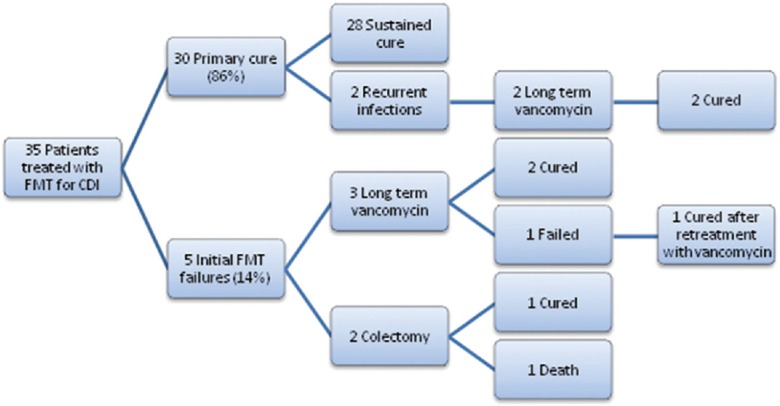
Each treatment stage for the study cohort, including primary modalities employed and the associated cure/failure incidences. CDI = Clostridium difficile infection; FMT = Fecal microbiota transplant
Figure 5.
Categorical breakdown of reported side effects among patients treated with fecal microbiota transplantation. Most common complaints included loose stools and abdominal pain
Patients with three or more recurrences
Three or more recurrences were recorded in 21/35 patients (60%) prior to undergoing FMT therapy. The overall cure rate in this group was 100%, with 90% (19/21) primary cures. Of note, 6/12 (29%) of patients in this group were classified as having severe CDI at the time of FMT administration.
Patients with severe Clostridium difficile infection
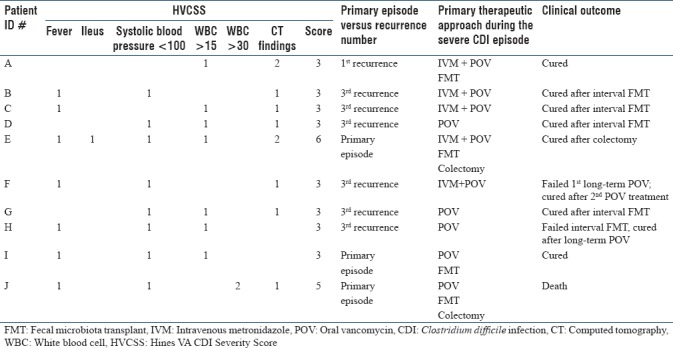
Overall, 10/35 (29%) patients had severe disease before undergoing FMT. The median HVCSS in this group was 3 (range 3–6). Within this subset, FMT was successful in 80% (n = 8), with primary cure achieved in 60% (n = 6) of cases. All patients were treated with oral vancomycin, and intravenous metronidazole was added in four patients. All patients with severe CDI received at least 7 days of antibiotics. Further details regarding outcomes in this group of patients are outlined in Table 1, including detailed listing of HVCSS subcomponents.
Table 1.
Summary of study patients with severe Clostridium difficile infection
Patients with complicated Clostridium difficile infection
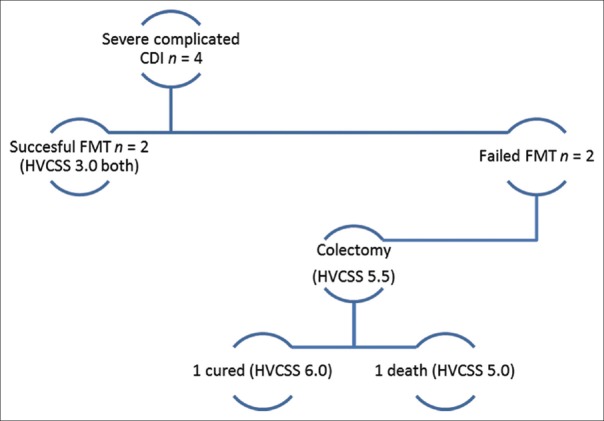
CCDI was present in four cases, with three CCDI cases occurring during the initial episode/presentation [Figure 6]. In this group, FMT was utilized during active inpatient treatment, with affected patients experiencing clinical deterioration despite 72 h of conventional management. Fifty percent of patients (2/4) in this group were cured after FMT and two progressively worsened, requiring colectomy for refractory CCDI (with 1 cure and 1 death in this subgroup). Of note, patients who required colectomy had higher HVCSS (5.5 vs. 3.0) compared to patients with CCDI who did not require surgery.
Figure 6.
The distribution of severe complicated Clostridium difficile infection cases. The Hines VA Clostridium difficile infection Severity Score was higher for cases requiring colectomy (5.5) than for cases where complicated Clostridium difficile infection resolved after fecal microbiota transplantation-based management
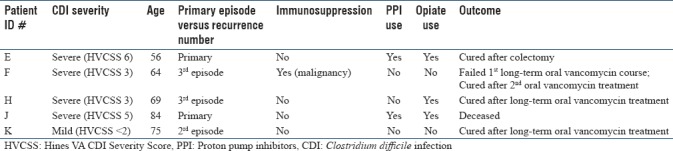
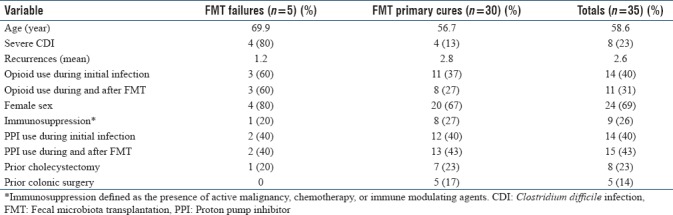
Initial fecal microbiota transplantation failures
Initial FMT failure to cure was noted in 5/35 (14.3%) of CDI cases [Table 2]. These patients were subsequently treated with either long-term vancomycin or colectomy. A 6-week course of vancomycin was used in three cases, with two secondary cures after single treatment. Another patient with post-chemotherapy neutropenia required two courses of vancomycin before achieving cure. Two patients had CCDI and required colectomy as outlined in Figures 4 and 6.
Table 2.
Summary of cases involving initial fecal microbial transplantation failure (n=5)
When compared to cases where primary FMT cure was achieved, FMT failures were associated with greater age (69.9 vs. 56.7), severe CDI (80% vs. 13%), female gender (80% vs. 67%), as well as active opioid use at the time of both the initial infection (60% vs. 37%) and FMT (60% vs. 27%). There were no differences between primary FMT failures and cures in terms of PPI use (40% vs. 40%), immunosuppression (20% vs. 27%), or previous cholecystectomy (20% vs. 23%). Of note, prior colonic surgery was less common in the FMT failure group (0% vs. 17%). Table 3 depicts full comparison of primary FMT cures and failures.
Table 3.
Comparison of “primary failure” versus “primary cure” fecal microbiota transplantation groups
DISCUSSION
Human stool is composed mostly of water, with approximately 6%–14% of the total content being microbial cells.[43,57,58] Of those, about half are live bacteria from over 1000 species and the remainder is attributed to dead microbial matter.[43,57,58] Bacterial presence in the GI tract helps protect the host against pathogenic organisms, plays a role in protein metabolism and vitamin synthesis, and is critical during the proliferation and differentiation of colonic epithelial cells.[59,60] The loss of the fragile intestinal microbiota balance (e.g., “dysbiosis”) can be associated with severe consequences, including life-threatening infections.[4,43] It is well known that antibiotics disrupt the indigenous microbiota that “resist” the colonization of the gut by pathogenic bacteria. Not all antibiotics carry the same CDI risk profile. Clindamycin and carbapenems carry the highest risk which is further increased with the use of multiple antibiotics and prolonged therapeutic exposures.[4,25,61] In this series, CDI was most frequently associated with exposure to clindamycin, cephalosporins, and quinolones.
The traditional CDI management approach consists of an attempt to eradicate CD using vancomycin and/or metronidazole for a pre-defined time course, depending on disease severity and initial versus recurrent episode status.[12,62] FMT became increasingly popular as an option for the management of recurrent, difficult-to-treat CDI.[44,63] In general, FMT is thought to prevent CDI recurrences through the introduction of microbiota that more effectively compete with CD for nutrients and thus reduce colonization risk. More specifically, healthy patients have much higher colonic content of Bacteroides spp., while patients with CDI have proportionately more Proteobacteria.[64] Application of FMT reestablishes normal bowel homeostasis, function, and metabolic balance by restoring the intestinal microbiota and facilitating “colonization resistance” through promoting the growth of Bacteroides and Firmicutes while reducing Proteobacteria.[65,66,67] Studies show that microbiota of FMT recipients are similar to those of the donor at 2 weeks post-FMT, including the restoration of Bacteroides spp. [39,58] Importantly, post-FMT microbiota diversity may be lower in elderly patients.[68] Roseburia and Lachnospiraceae are butyrate-producing bacteria implicated in host defense, maintaining epithelial integrity and reducing intestinal permeability. They are critical to stopping the perpetuation and worsening of CDI.[29,65,68] Other endogenous bacteria either directly or indirectly inhibit CD via diverse mechanisms.[5,69,70] End result of FMT is the restoration of physiologic and immune homeostasis within previously disrupted colonic mucosa, limiting the growth of pathogenic CD.[4,5,61]
The current study demonstrates high efficacy of FMT for RCDI, with the primary FMT-attributable cure of 86%. It is important to note that FMT-attributable cures can vary from 81% for “primary cure”[71,72] to 100% for “overall cure,”[73] with the most commonly reported “cure range” between 87% and 94%.[11,12,30,43,45,46,56,73,74,75,76,77] The literature supports our findings, with approximately 80% reported cure rate for FMT, compared to 23%–31% cure rates with bowel lavage plus vancomycin or vancomycin alone.[44,71] Of note, overall FMT cures were previously reported at 94%, in line with the findings of the current series.[71] Our patients underwent treatment with donated stool samples. This approach was chosen a priori due to published evidence that banked stool may have greater efficacy than autologous samples.[78] Although more expensive, colonoscopic FMT instillation has been associated with better treatment response.[66,74] The use of frozen stool samples appears noninferior to fresh stool utilization.[56,79,80]
The cure rate also appears to correlate with patient age, with the cutoff of 65 years being associated with an approximate 10%–15% reduction in reported success rates.[76] This is corroborated by our study's findings, with therapeutic FMT failure group being approximately 14 years older than the FMT cure group. Of importance, it was previously noted that the absence of BI/NAP/027 strain is associated with near-universal cure while its presence decreases the cure rate to <90%.[73]
The current series supports the notion that severe CDI or CCDI is significantly more likely to be associated with FMT failure. In severe CDI, our primary and overall cures were 60% and 80%, respectively. The primary cure dropped further to 50% in CCDI cases, with ongoing clinical deterioration necessitating colectomies in the affected subgroup. Literature corroborates these findings, with substantially lower primary cure rates seen in both severe CDI (<80%) and CCDI (<50%).[81,82] In addition, FMT failure has been associated with the number of CDI-related hospitalizations, where each consecutive episode increased the probability of failure by 1.4-fold.[72] It is important to note that increasing HVCSS strongly correlates with the severity of CDI.[51] In the current series, surgery was needed in two patients (6%) and was associated with mortality of 50%. Although the use of surgery for CDI has decreased following the introduction of FMT,[30] mortality associated with treatment failures requiring surgery can be as high as 75%.[61]
The FMT procedure is very well tolerated by patients, with up to 97% of recipients being willing to repeat treatment if needed.[29,83] In our experience, adverse events associated with FMT are usually mild and self-limited. Abdominal pain, loose stools, and dizziness constituted the most common complaints, largely consistent with the previously published data.[84] Even in cases involving clinical cure, initial post-FMT stool consistency can still be loose and it may take weeks for the bowel pattern to normalize.[11] FMT application is not risk-free, however. Nasogastric or nasojejunal administration routes have been associated with bleeding, enteritis, peritonitis, aspiration pneumonia, and very rarely mortality.[84,85] Colonoscopic FMT instillation has been associated with colonic perforations.[29,73] Although causative relationship is difficult to prove, there are also reports of a febrile response, headache, lymphadenopathy, transiently elevated C-reactive protein, Gram-negative bacteremia, urinary tract infection, constipation, renal failure, as well as opportunistic viral infections.[69,84,86]
Reporting on long-term adverse effects of FMT is still limited. However, patients should be counseled about the possibility of as-yet-unknown risks associated with FMT given the complex interplay between transplanted microbiota and the recipient bowel.[67,69,87] Experience is also limited regarding FMT safety in immunosuppressed patients, although FMTs were performed in this setting without apparent adverse events.[22,39,45] Approximately one in four patients in the current study had evidence of some degree of immunosuppression, including history of malignancy, recent chemotherapy, and use of immunomodulating agents. No severe adverse effects were reported in this group. Because FMT is categorized as human tissue, the FDA exercises enforcement discretion in the use of FMT for the treatment of CDI refractory to standard therapy and stresses the importance of informing patients about the investigational nature of FMT while obtaining informed consent.[13,22,29]
Clinical management guidelines for CDI discourage the use of antimotility agents,[31,35] primarily due to the association between antiperistaltic regimens and the onset of complicated disease featuring acute pathologic colonic distention and increased mortality.[6,12,36] These guidelines do not specifically address opioids, despite similar antimotility properties and greater incidence of CDI with moderate-to-high opiate use.[88] In most cases of high acuity illness, the use of opioids may be unavoidable due to the presence of severe pain. Physiologic effects of opioids on the bowel include the appearance of nonperistaltic contractions and increased absorption of fluids, resulting in harder stools.[89,90,91,92] Therefore, opioids may help prolong bacterial spore contact with the colonic mucosa and thus promote CD colonization.[88] Consequently, it is not surprising that opioid use has been associated with 3-fold increase in CDI risk [27] as well as 2.5 times the odds of severe and CCDI.[93,94] In the current series, substantial proportion of patients utilized opioids before and during their active infection, with many continuing opioid use beyond the acute CDI episode. It is not clear whether opioids contributed to recurrence in our study; however, the proportion of opioid users was much higher among patients who failed FMT when compared to those who experienced primary cure. Further investigation of the relationship between opioid use and CDI recurrence is warranted. It is unknown if laxatives or opioid antagonists could counteract the effect of opioids on the overall CDI risk profile.
Challenges in implementing institutional fecal microbiota transplantation protocol
Despite the potential for complications being very low, institutional implementation of FMTP requires buy-in from both administrative and clinical leadership. Initial deployment may require approvals from institutional Infection Control Committee, Ethics Committee/IRB, and other local regulatory bodies. Team members responsible for FMTP introduction should have good working knowledge of any pertinent institutional, local, regional, and national laws and regulations. A group of FMT experts should be created to guide this process and to ensure harmonious FMTP implementation with focus on staff education and patient safety.
After institutional approvals were granted for FMTP deployment at our institution, significant amount of effort was devoted to educating providers and staff about the process, ensuring that the protocol can be implemented without creating confusion or workflow disruptions. Close oversight by pertinent institutional leadership was provided during the initial FMT applications. There was great attention to procedural harmonization, especially since every FMT was performed via colonoscopy, following the same steps regardless of the administering provider. It is critical to maintain this harmonization, regardless of whether surgical or gastroenterology services perform the procedure.
We also feel strongly that the best way to streamline the FMT process would be to have on-demand access to banked frozen stool, thus facilitating the conduct of emergency procedures. At our institution, FMT stool specimens are obtained from a centralized stool bank per request, with two samples made available for each instance/application. A dedicated fridge assigned exclusively to FMT storage is mandated, with frozen stool specimens requiring stable temperature of −80°C. Properly stored specimens are considered viable for 6 months. Because FMT is classified by the FDA as a biological agent, regulatory considerations apply. Currently, the FDA strongly encourages local IRB approval for FMT under an investigational new drug pathway.[95] Finally, FMT may not be universally covered by health insurance plans; therefore, the patient may be required to pay for the procedure out of pocket.
CONCLUSIONS
This report of single-institution experience supports the use of FMT as a safe and effective therapy for RCDI or severe CDI. Given its retrospective design, the current study is inherently subject to biases. The purely descriptive nature of this report, combined with the potential presence of confounding variables, limits the applicability of our results to other institutions or settings. Despite promising short-term results, questions remain about long-term safety of FMT in both immunocompetent and immunosuppressed patients. Clinical indications for FMT will likely continue to evolve, including the number of CDI recurrences before FMT is triggered. It is also not clear how many FMT application procedures are required to optimize treatment efficacy. In the current study, opioid use was more prevalent among patients who failed FMT, both before and during active treatment. Consequently, the role of opioids as modulators of CDI severity should be further investigated.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Chapin KC, Dickenson RA, Wu F, Andrea SB. Comparison of five assays for detection of Clostridium difficile toxin. J Mol Diagn. 2011;13:395–400. doi: 10.1016/j.jmoldx.2011.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bartlett JG, Chang TW, Gurwith M, Gorbach SL, Onderdonk AB. Antibiotic-associated pseudomembranous colitis due to toxin-producing clostridia. N Engl J Med. 1978;298:531–4. doi: 10.1056/NEJM197803092981003. [DOI] [PubMed] [Google Scholar]
- 3.McFarland LV, Mulligan ME, Kwok RY, Stamm WE. Nosocomial acquisition of Clostridium difficile infection. N Engl J Med. 1989;320:204–10. doi: 10.1056/NEJM198901263200402. [DOI] [PubMed] [Google Scholar]
- 4.Borgia G, Maraolo AE, Foggia M, Buonomo AR, Gentile I. Fecal microbiota transplantation for Clostridium difficile infection: Back to the future. Expert Opin Biol Ther. 2015;15:1001–14. doi: 10.1517/14712598.2015.1045872. [DOI] [PubMed] [Google Scholar]
- 5.Keller JJ, Kuijper EJ. Treatment of recurrent and severe Clostridium difficile infection. Annu Rev Med. 2015;66:373–86. doi: 10.1146/annurev-med-070813-114317. [DOI] [PubMed] [Google Scholar]
- 6.Khanna S, Pardi DS. Clostridium difficile infection: New insights into management. Mayo Clin Proc. 2012;87:1106–17. doi: 10.1016/j.mayocp.2012.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cohen SH, Gerding DN, Johnson S, Kelly CP, Loo VG, McDonald LC, et al. Clinical practice guidelines for Clostridium difficile infection in adults: 2010 update by the Society for healthcare epidemiology of America (SHEA) and the infectious diseases society of America (IDSA) Infect Control Hosp Epidemiol. 2010;31:431–55. doi: 10.1086/651706. [DOI] [PubMed] [Google Scholar]
- 8.Penders J, Thijs C, Vink C, Stelma FF, Snijders B, Kummeling I, et al. Factors influencing the composition of the intestinal microbiota in early infancy. Pediatrics. 2006;118:511–21. doi: 10.1542/peds.2005-2824. [DOI] [PubMed] [Google Scholar]
- 9.SHEA Long-Term-Care Committee. Simor AE, Bradley SF, Strausbaugh LJ, Crossley K, Nicolle LE. Clostridium difficile in long-term-care facilities for the elderly. Infect Control Hosp Epidemiol. 2002;23:696–703. doi: 10.1086/501997. [DOI] [PubMed] [Google Scholar]
- 10.Nelson RL, Suda KJ, Evans CT. Antibiotic treatment for Clostridium difficile-associated diarrhoea in adults. Cochrane Database Syst Rev. 2017;3:CD004610. doi: 10.1002/14651858.CD004610.pub5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bagdasarian N, Rao K, Malani PN. Diagnosis and treatment of Clostridium difficile in adults: A systematic review. JAMA. 2015;313:398–408. doi: 10.1001/jama.2014.17103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Surawicz CM, Brandt LJ, Binion DG, Ananthakrishnan AN, Curry SR, Gilligan PH, et al. Guidelines for diagnosis, treatment, and prevention of Clostridium difficile infections. Am J Gastroenterol. 2013;108:478–98. doi: 10.1038/ajg.2013.4. [DOI] [PubMed] [Google Scholar]
- 13.Kassam Z, Lee CH, Hunt RH. Review of the emerging treatment of Clostridium difficile infection with fecal microbiota transplantation and insights into future challenges. Clin Lab Med. 2014;34:787–98. doi: 10.1016/j.cll.2014.08.007. [DOI] [PubMed] [Google Scholar]
- 14.Miller BA, Chen LF, Sexton DJ, Anderson DJ. Comparison of the burdens of hospital-onset, healthcare facility-associated Clostridium difficile infection and of healthcare-associated infection due to methicillin-resistant Staphylococcus aureus in community hospitals. Infect Control Hosp Epidemiol. 2011;32:387–90. doi: 10.1086/659156. [DOI] [PubMed] [Google Scholar]
- 15.Vyas D, Aekka A, Vyas A. Fecal transplant policy and legislation. World J Gastroenterol. 2015;21:6–11. doi: 10.3748/wjg.v21.i1.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ünal CM, Steinert M. Novel therapeutic strategies for Clostridium difficile infections. Expert Opin Ther Targets. 2016;20:269–85. doi: 10.1517/14728222.2016.1090428. [DOI] [PubMed] [Google Scholar]
- 17.Lavallée C, Laufer B, Pépin J, Mitchell A, Dubé S, Labbé AC, et al. Fatal Clostridium difficile enteritis caused by the BI/NAP1/027 strain: A case series of ileal C. difficile infections. Clin Microbiol Infect. 2009;15:1093–9. doi: 10.1111/j.1469-0691.2009.03004.x. [DOI] [PubMed] [Google Scholar]
- 18.Petrella LA, Sambol SP, Cheknis A, Nagaro K, Kean Y, Sears PS, et al. Decreased cure and increased recurrence rates for Clostridium difficile infection caused by the epidemic C. difficile BI strain. Clin Infect Dis. 2012;55:351–7. doi: 10.1093/cid/cis430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Issa M, Vijayapal A, Graham MB, Beaulieu DB, Otterson MF, Lundeen S, et al. Impact of Clostridium difficile on inflammatory bowel disease. Clin Gastroenterol Hepatol. 2007;5:345–51. doi: 10.1016/j.cgh.2006.12.028. [DOI] [PubMed] [Google Scholar]
- 20.Bakken JS, Borody T, Brandt LJ, Brill JV, Demarco DC, Franzos MA, et al. Treating Clostridium difficile infection with fecal microbiota transplantation. Clin Gastroenterol Hepatol. 2011;9:1044–9. doi: 10.1016/j.cgh.2011.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hall AJ, Curns AT, McDonald LC, Parashar UD, Lopman BA. The roles of Clostridium difficile and norovirus among gastroenteritis-associated deaths in the United States, 1999-2007. Clin Infect Dis. 2012;55:216–23. doi: 10.1093/cid/cis386. [DOI] [PubMed] [Google Scholar]
- 22.Borody T, Fischer M, Mitchell S, Campbell J. Fecal microbiota transplantation in gastrointestinal disease: 2015 update and the road ahead. Expert Rev Gastroenterol Hepatol. 2015;9:1379–91. doi: 10.1586/17474124.2015.1086267. [DOI] [PubMed] [Google Scholar]
- 23.Lessa FC, Winston LG, McDonald LC. Emerging Infections Program C. difficile Surveillance Team. Burden of Clostridium difficile infection in the United States. N Engl J Med. 2015;372:2369–70. doi: 10.1056/NEJMc1505190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhang S, Palazuelos-Munoz S, Balsells EM, Nair H, Chit A, Kyaw MH, et al. Cost of hospital management of Clostridium difficile infection in United States – A meta-analysis and modelling study. BMC Infect Dis. 2016;16:447. doi: 10.1186/s12879-016-1786-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Vardakas KZ, Trigkidis KK, Boukouvala E, Falagas ME. Clostridium difficile infection following systemic antibiotic administration in randomised controlled trials: A systematic review and meta-analysis. Int J Antimicrob Agents. 2016;48:1–0. doi: 10.1016/j.ijantimicag.2016.03.008. [DOI] [PubMed] [Google Scholar]
- 26.Yearsley KA, Gilby LJ, Ramadas AV, Kubiak EM, Fone DL, Allison MC, et al. Proton pump inhibitor therapy is a risk factor for Clostridium difficile-associated diarrhoea. Aliment Pharmacol Ther. 2006;24:613–9. doi: 10.1111/j.1365-2036.2006.03015.x. [DOI] [PubMed] [Google Scholar]
- 27.Dubberke ER, Reske KA, Olsen MA, McMullen KM, Mayfield JL, McDonald LC, et al. Evaluation of Clostridium difficile-associated disease pressure as a risk factor for C difficile-associated disease. Arch Intern Med. 2007;167:1092–7. doi: 10.1001/archinte.167.10.1092. [DOI] [PubMed] [Google Scholar]
- 28.Tauxe WM, Haydek JP, Rebolledo PA, Neish E, Newman KL, Ward A, et al. Fecal microbiota transplant for Clostridium difficile infection in older adults. Therap Adv Gastroenterol. 2016;9:273–81. doi: 10.1177/1756283X15622600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cohen NA, Ben Ami R, Guzner-Gur H, Santo ME, Halpern Z, Maharshak N, et al. Fecal microbiota transplantation for Clostridium difficile-associated diarrhea. Isr Med Assoc J. 2015;17:510–4. [PubMed] [Google Scholar]
- 30.Cammarota G, Ianiro G, Gasbarrini A. Fecal microbiota transplantation for the treatment of Clostridium difficile infection: A systematic review. J Clin Gastroenterol. 2014;48:693–702. doi: 10.1097/MCG.0000000000000046. [DOI] [PubMed] [Google Scholar]
- 31.Khanna S, Pardi DS. Clostridium difficile infection: Management strategies for a difficult disease. Therap Adv Gastroenterol. 2014;7:72–86. doi: 10.1177/1756283X13508519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.ESCMID Study Group for Clostridium difficile; EU Member States. Kuijper EJ, Coignard B, Tüll P. European Centre for Disease Prevention and Control. Emergence of Clostridium difficile-associated disease in North America and Europe. Clin Microbiol Infect. 2006;12(Suppl 6):2–18. doi: 10.1111/j.1469-0691.2006.01580.x. [DOI] [PubMed] [Google Scholar]
- 33.Dalton BR, Lye-Maccannell T, Henderson EA, Maccannell DR, Louie TJ. Proton pump inhibitors increase significantly the risk of Clostridium difficile infection in a low-endemicity, non-outbreak hospital setting. Aliment Pharmacol Ther. 2009;29:626–34. doi: 10.1111/j.1365-2036.2008.03924.x. [DOI] [PubMed] [Google Scholar]
- 34.Tang-Feldman Y, Mayo S, Silva J, Jr, Cohen SH. Molecular analysis of Clostridium difficile strains isolated from 18 cases of recurrent Clostridium difficile-associated diarrhea. J Clin Microbiol. 2003;41:3413–4. doi: 10.1128/JCM.41.7.3413-3414.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Debast SB, Bauer MP, Kuijper EJ. European Society of Clinical Microbiology and Infectious Diseases. European society of clinical microbiology and infectious diseases: Update of the treatment guidance document for Clostridium difficile infection. Clin Microbiol Infect. 2014;20(Suppl 2):1–26. doi: 10.1111/1469-0691.12418. [DOI] [PubMed] [Google Scholar]
- 36.Sartelli M, Malangoni MA, Abu-Zidan FM, Griffiths EA, Di Bella S, McFarland LV, et al. WSES guidelines for management of Clostridium difficile infection in surgical patients. World J Emerg Surg. 2015;10:38. doi: 10.1186/s13017-015-0033-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bartlett JG, Gerding DN. Clinical recognition and diagnosis of Clostridium difficile infection. Clin Infect Dis. 2008;46(Suppl 1):S12–8. doi: 10.1086/521863. [DOI] [PubMed] [Google Scholar]
- 38.Gujja D, Friedenberg FK. Predictors of serious complications due to Clostridium difficile infection. Aliment Pharmacol Ther. 2009;29:635–42. doi: 10.1111/j.1365-2036.2008.03914.x. [DOI] [PubMed] [Google Scholar]
- 39.Bowman KA, Broussard EK, Surawicz CM. Fecal microbiota transplantation: Current clinical efficacy and future prospects. Clin Exp Gastroenterol. 2015;8:285–91. doi: 10.2147/CEG.S61305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Eiseman B, Silen W, Bascom GS, Kauvar AJ. Fecal enema as an adjunct in the treatment of pseudomembranous enterocolitis. Surgery. 1958;44:854–9. [PubMed] [Google Scholar]
- 41.Schwan A, Sjölin S, Trottestam U, Aronsson B. Relapsing Clostridium difficile enterocolitis cured by rectal infusion of normal faeces. Scand J Infect Dis. 1984;16:211–5. doi: 10.3109/00365548409087145. [DOI] [PubMed] [Google Scholar]
- 42.Aas J, Gessert CE, Bakken JS. Recurrent Clostridium difficile colitis: Case series involving 18 patients treated with donor stool administered via a nasogastric tube. Clin Infect Dis. 2003;36:580–5. doi: 10.1086/367657. [DOI] [PubMed] [Google Scholar]
- 43.Gupta S, Allen-Vercoe E, Petrof EO. Fecal microbiota transplantation: In perspective. Therap Adv Gastroenterol. 2016;9:229–39. doi: 10.1177/1756283X15607414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Knight P, Anagnostakos JP, Lin MJ, Geme B, Stawicki SP. Fecal microbiota transplantation (FMT) for Clostridium difficile colitis. OPUS 12 Sci. 2015;9:1–8. [Google Scholar]
- 45.Moayyedi P, Marshall JK, Yuan Y, Hunt R. Canadian association of gastroenterology position statement: Fecal microbiota transplant therapy. Can J Gastroenterol Hepatol. 2014;28:66–8. doi: 10.1155/2014/346590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Drekonja D, Reich J, Gezahegn S, Greer N, Shaukat A, MacDonald R, et al. Fecal microbiota transplantation for Clostridium difficile infection: A systematic review. Ann Intern Med. 2015;162:630–8. doi: 10.7326/M14-2693. [DOI] [PubMed] [Google Scholar]
- 47.de Jong E, de Jong AS, Bartels CJ, van der Rijt-van den Biggelaar C, Melchers WJ, Sturm PD, et al. Clinical and laboratory evaluation of a real-time PCR for Clostridium difficile toxin A and B genes. Eur J Clin Microbiol Infect Dis. 2012;31:2219–25. doi: 10.1007/s10096-012-1558-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zar FA, Bakkanagari SR, Moorthi KM, Davis MB. A comparison of vancomycin and metronidazole for the treatment of Clostridium difficile-associated diarrhea, stratified by disease severity. Clin Infect Dis. 2007;45:302–7. doi: 10.1086/519265. [DOI] [PubMed] [Google Scholar]
- 49.Brandt LJ, Aroniadis OC, Mellow M, Kanatzar A, Kelly C, Park T, et al. Long-term follow-up of colonoscopic fecal microbiota transplant for recurrent Clostridium difficile infection. Am J Gastroenterol. 2012;107:1079–87. doi: 10.1038/ajg.2012.60. [DOI] [PubMed] [Google Scholar]
- 50.Millan B, Park H, Hotte N, Mathieu O, Burguiere P, Tompkins TA, et al. Fecal microbial transplants reduce antibiotic-resistant genes in patients with recurrent Clostridium difficile infection. Clin Infect Dis. 2016;62:1479–86. doi: 10.1093/cid/ciw185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fujitani S, George WL, Murthy AR. Comparison of clinical severity score indices for Clostridium difficile infection. Infect Control Hosp Epidemiol. 2011;32:220–8. doi: 10.1086/658336. [DOI] [PubMed] [Google Scholar]
- 52.Belmares J, Gerding DN, Parada JP, Miskevics S, Weaver F, Johnson S, et al. Outcome of metronidazole therapy for Clostridium difficile disease and correlation with a scoring system. J Infect. 2007;55:495–501. doi: 10.1016/j.jinf.2007.09.015. [DOI] [PubMed] [Google Scholar]
- 53.Allegretti JR, Korzenik JR, Hamilton MJ. Fecal microbiota transplantation via colonoscopy for recurrent C. difficile infection. J Vis Exp. 2014;94:52154. doi: 10.3791/52154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Costello SP, Tucker EC, La Brooy J, Schoeman MN, Andrews JM. Establishing a fecal microbiota transplant service for the treatment of Clostridium difficile infection. Clin Infect Dis. 2016;62:908–14. doi: 10.1093/cid/civ994. [DOI] [PubMed] [Google Scholar]
- 55.Tauxe WM, Dhere T, Ward A, Racsa LD, Varkey JB, Kraft CS, et al. Fecal microbiota transplant protocol for Clostridium difficile infection. Lab Med. 2015;46:e19–23. doi: 10.1309/LMCI95M0TWPDZKOD. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Gough E, Shaikh H, Manges AR. Systematic review of intestinal microbiota transplantation (fecal bacteriotherapy) for recurrent Clostridium difficile infection. Clin Infect Dis. 2011;53:994–1002. doi: 10.1093/cid/cir632. [DOI] [PubMed] [Google Scholar]
- 57.Bojanova DP, Bordenstein SR. Fecal transplants: What is being transferred? PLoS Biol. 2016;14:e1002503. doi: 10.1371/journal.pbio.1002503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Pang X, Hua X, Yang Q, Ding D, Che C, Cui L, et al. Inter-species transplantation of gut microbiota from human to pigs. ISME J. 2007;1:156–62. doi: 10.1038/ismej.2007.23. [DOI] [PubMed] [Google Scholar]
- 59.Nicholson JK, Holmes E, Kinross J, Burcelin R, Gibson G, Jia W, et al. Host-gut microbiota metabolic interactions. Science. 2012;336:1262–7. doi: 10.1126/science.1223813. [DOI] [PubMed] [Google Scholar]
- 60.Sommer F, Bäckhed F. The gut microbiota-masters of host development and physiology. Nat Rev Microbiol. 2013;11:227–38. doi: 10.1038/nrmicro2974. [DOI] [PubMed] [Google Scholar]
- 61.Han S, Shannahan S, Pellish R. Fecal microbiota transplant: Treatment options for Clostridium difficile infection in the Intensive Care Unit. J Intensive Care Med. 2016;31:577–86. doi: 10.1177/0885066615594344. [DOI] [PubMed] [Google Scholar]
- 62.Surawicz CM, McFarland LV, Greenberg RN, Rubin M, Fekety R, Mulligan ME, et al. The search for a better treatment for recurrent Clostridium difficile disease: Use of high-dose vancomycin combined with Saccharomyces boulardii. Clin Infect Dis. 2000;31:1012–7. doi: 10.1086/318130. [DOI] [PubMed] [Google Scholar]
- 63.McFarland LV, Elmer GW, Surawicz CM. Breaking the cycle: Treatment strategies for 163 cases of recurrent Clostridium difficile disease. Am J Gastroenterol. 2002;97:1769–75. doi: 10.1111/j.1572-0241.2002.05839.x. [DOI] [PubMed] [Google Scholar]
- 64.Aroniadis OC, Brandt LJ. Intestinal microbiota and the efficacy of fecal microbiota transplantation in gastrointestinal disease. Gastroenterol Hepatol (N Y) 2014;10:230–7. [PMC free article] [PubMed] [Google Scholar]
- 65.Shahinas D, Silverman M, Sittler T, Chiu C, Kim P, Allen-Vercoe E, et al. Toward an understanding of changes in diversity associated with fecal microbiome transplantation based on 16S rRNA gene deep sequencing. MBio. 2012:e00338–12. doi: 10.1128/mBio.00338-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Sofi AA, Silverman AL, Khuder S, Garborg K, Westerink JM, Nawras A, et al. Relationship of symptom duration and fecal bacteriotherapy in Clostridium difficile infection-pooled data analysis and a systematic review. Scand J Gastroenterol. 2013;48:266–73. doi: 10.3109/00365521.2012.743585. [DOI] [PubMed] [Google Scholar]
- 67.Rao K, Young VB. Fecal microbiota transplantation for the management of Clostridium difficile infection. Infect Dis Clin North Am. 2015;29:109–22. doi: 10.1016/j.idc.2014.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Girotra M, Garg S, Anand R, Song Y, Dutta SK. Fecal microbiota transplantation for recurrent Clostridium difficile infection in the elderly: Long-term outcomes and microbiota changes. Dig Dis Sci. 2016;61:3007–15. doi: 10.1007/s10620-016-4229-8. [DOI] [PubMed] [Google Scholar]
- 69.Kelly CR, Kahn S, Kashyap P, Laine L, Rubin D, Atreja A, et al. Update on fecal microbiota transplantation 2015: Indications, methodologies, mechanisms, and outlook. Gastroenterology. 2015;149:223–37. doi: 10.1053/j.gastro.2015.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Weingarden AR, Dosa PI, DeWinter E, Steer CJ, Shaughnessy MK, Johnson JR, et al. Changes in colonic bile acid composition following fecal microbiota transplantation are sufficient to control Clostridium difficile germination and growth. PLoS One. 2016;11:e0147210. doi: 10.1371/journal.pone.0147210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.van Nood E, Vrieze A, Nieuwdorp M, Fuentes S, Zoetendal EG, de Vos WM, et al. Duodenal infusion of donor feces for recurrent Clostridium difficile. N Engl J Med. 2013;368:407–15. doi: 10.1056/NEJMoa1205037. [DOI] [PubMed] [Google Scholar]
- 72.Fischer M, Kao D, Mehta SR, Martin T, Dimitry J, Keshteli AH, et al. Predictors of early failure after fecal microbiota transplantation for the therapy of Clostridium difficile infection: A multicenter study. Am J Gastroenterol. 2016;111:1024–31. doi: 10.1038/ajg.2016.180. [DOI] [PubMed] [Google Scholar]
- 73.Kassam Z, Lee CH, Yuan Y, Hunt RH. Fecal microbiota transplantation for Clostridium difficile infection: Systematic review and meta-analysis. Am J Gastroenterol. 2013;108:500–8. doi: 10.1038/ajg.2013.59. [DOI] [PubMed] [Google Scholar]
- 74.Postigo R, Kim JH. Colonoscopic versus nasogastric fecal transplantation for the treatment of Clostridium difficile infection: A review and pooled analysis. Infection. 2012;40:643–8. doi: 10.1007/s15010-012-0307-9. [DOI] [PubMed] [Google Scholar]
- 75.Rossen NG, MacDonald JK, de Vries EM, D'Haens GR, de Vos WM, Zoetendal EG, et al. Fecal microbiota transplantation as novel therapy in gastroenterology: A systematic review. World J Gastroenterol. 2015;21:5359–71. doi: 10.3748/wjg.v21.i17.5359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Li YT, Cai HF, Wang ZH, Xu J, Fang JY. Systematic review with meta-analysis: Long-term outcomes of faecal microbiota transplantation for Clostridium difficile infection. Aliment Pharmacol Ther. 2016;43:445–57. doi: 10.1111/apt.13492. [DOI] [PubMed] [Google Scholar]
- 77.Hagel S, Fischer A, Ehlermann P, Frank T, Tueffers K, Sturm A, et al. Fecal microbiota transplant in patients with recurrent Clostridium difficile infection. Dtsch Arztebl Int. 2016;113:583–9. doi: 10.3238/arztebl.2016.0583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Kelly CR, Khoruts A, Staley C, Sadowsky MJ, Abd M, Alani M, et al. Effect of fecal microbiota transplantation on recurrence in multiply recurrent Clostridium difficile infection: A Randomized trial. Ann Intern Med. 2016;165:609–16. doi: 10.7326/M16-0271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Malani PN, Rao K. Expanded evidence for frozen fecal microbiota transplantation for Clostridium difficile infection: A Fresh take. JAMA. 2016;315:137–8. doi: 10.1001/jama.2015.18100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lee CH, Steiner T, Petrof EO, Smieja M, Roscoe D, Nematallah A, et al. Frozen vs. fresh fecal microbiota transplantation and clinical resolution of diarrhea in patients with recurrent Clostridium difficile infection: A Randomized clinical trial. JAMA. 2016;315:142–9. doi: 10.1001/jama.2015.18098. [DOI] [PubMed] [Google Scholar]
- 81.Fischer M, Sipe BW, Rogers NA, Cook GK, Robb BW, Vuppalanchi R, et al. Faecal microbiota transplantation plus selected use of vancomycin for severe-complicated Clostridium difficile infection: Description of a protocol with high success rate. Aliment Pharmacol Ther. 2015;42:470–6. doi: 10.1111/apt.13290. [DOI] [PubMed] [Google Scholar]
- 82.Zainah H, Hassan M, Shiekh-Sroujieh L, Hassan S, Alangaden G, Ramesh M, et al. Intestinal microbiota transplantation, a simple and effective treatment for severe and refractory Clostridium difficile infection. Dig Dis Sci. 2015;60:181–5. doi: 10.1007/s10620-014-3296-y. [DOI] [PubMed] [Google Scholar]
- 83.Zellmer C, De Wolfe TJ, Van Hoof S, Blakney R, Safdar N. Patient perspectives on fecal microbiota transplantation for Clostridium difficile infection. Infect Dis Ther. 2016;5:155–64. doi: 10.1007/s40121-016-0106-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Baxter M, Colville A. Adverse events in faecal microbiota transplant: A review of the literature. J Hosp Infect. 2016;92:117–27. doi: 10.1016/j.jhin.2015.10.024. [DOI] [PubMed] [Google Scholar]
- 85.Baxter M, Ahmad T, Colville A, Sheridan R. Fatal aspiration pneumonia as a complication of fecal microbiota transplant. Clin Infect Dis. 2015;61:136–7. doi: 10.1093/cid/civ247. [DOI] [PubMed] [Google Scholar]
- 86.Wang S, Xu M, Wang W, Cao X, Piao M, Khan S, et al. Systematic review: Adverse events of fecal microbiota transplantation. PLoS One. 2016;11:e0161174. doi: 10.1371/journal.pone.0161174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Vehreschild MJ, Cornely OA. Fecal microbiota transfer 2.0. J Infect Dis. 2016;214:169–70. doi: 10.1093/infdis/jiv768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Mora AL, Salazar M, Pablo-Caeiro J, Frost CP, Yadav Y, DuPont HL, et al. Moderate to high use of opioid analgesics are associated with an increased risk of Clostridium difficile infection. Am J Med Sci. 2012;343:277–80. doi: 10.1097/MAJ.0b013e31822f42eb. [DOI] [PubMed] [Google Scholar]
- 89.Dorn S, Lembo A, Cremonini F. Opioid-induced bowel dysfunction: Epidemiology, pathophysiology, diagnosis, and initial therapeutic approach. Am J Gastroenterol Suppl. 2014;2:31–7. doi: 10.1038/ajgsup.2014.7. [DOI] [PubMed] [Google Scholar]
- 90.Lee AA, Hasler WL. Opioids and GI motility-friend or foe? Curr Treat Options Gastroenterol. 2016;14:478–94. doi: 10.1007/s11938-016-0112-0. [DOI] [PubMed] [Google Scholar]
- 91.Wood JD, Galligan JJ. Function of opioids in the enteric nervous system. Neurogastroenterol Motil. 2004;16(Suppl 2):17–28. doi: 10.1111/j.1743-3150.2004.00554.x. [DOI] [PubMed] [Google Scholar]
- 92.Galligan JJ, Akbarali HI. Molecular physiology of enteric opioid receptors. Am J Gastroenterol Suppl. 2014;2:17–21. doi: 10.1038/ajgsup.2014.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Shivashankar R, Khanna S, Kammer PP, Harmsen WS, Zinsmeister AR, Baddour LM, et al. Clinical factors associated with development of severe-complicated Clostridium difficile infection. Clin Gastroenterol Hepatol. 2013;11:1466–71. doi: 10.1016/j.cgh.2013.04.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Lichtenstein GR, Feagan BG, Cohen RD, Salzberg BA, Diamond RH, Chen DM, et al. Serious infections and mortality in association with therapies for Crohn's disease: TREAT registry. Clin Gastroenterol Hepatol. 2006;4:621–30. doi: 10.1016/j.cgh.2006.03.002. [DOI] [PubMed] [Google Scholar]
- 95.IDSA. Fecal Microbiota Transplantation: Investigational New Drug Protocol. [Last accessed on 2017 Dec 04]. Available from: http://www.idsociety.org/FMT/.