Abstract
Objectives
To compare the effect of femoral bone tunnel configuration on tendon-bone healing in an anterior cruciate ligament (ACL) reconstruction animal model.
Methods
Anterior cruciate ligament reconstruction using the plantaris tendon as graft material was performed on both knees of 24 rabbits (48 knees) to mimic ACL reconstruction by two different suspensory fixation devices for graft fixation. For the adjustable fixation device model (Socket group; group S), a 5 mm deep socket was created in the lateral femoral condyle (LFC) of the right knee. For the fixed-loop model (Tunnel group; group T), a femoral tunnel penetrating the LFC was created in the left knee. Animals were sacrificed at four and eight weeks after surgery for histological evaluation and biomechanical testing.
Results
Histologically, both groups showed a mixture of direct and indirect healing patterns at four weeks, whereas only indirect healing patterns were observed in both groups at eight weeks. No significant histological differences were seen between the two groups at four and eight weeks in the roof zone (four weeks, S: mean 4.8 sd 1.7, T: mean 4.5 sd 0.5, p = 0.14; eight weeks, S: mean 5.8 sd 0.8, T: mean 4.8 sd 1.8, p = 0.88, Mann-Whitney U test) or side zone (four weeks, S: mean 5.0 sd 1.2, T: mean 4.8 sd 0.4, p = 0.43; eight weeks, S: mean 5.3 sd 0.8,T: mean 5.5 sd 0.8, p = 0.61, Mann-Whitney U test) . Similarly, no significant difference was seen in the maximum failure load between group S and group T at four (15.6 sd 9.0N and 13.1 sd 5.6N) or eight weeks (12.6 sd 3.6N and 17.1 sd 6.4N, respectively).
Conclusion
Regardless of bone tunnel configuration, tendon-bone healing after ACL reconstruction primarily occurred through indirect healing. No significant histological or mechanical differences were observed between adjustable and fixed-loop femoral cortical suspension methods.
Cite this article: Y. Sato, R. Akagi, Y. Akatsu, Y. Matsuura, S. Takahashi, S. Yamaguchi, T. Enomoto, R. Nakagawa, H. Hoshi, T. Sasaki, S. Kimura, Y. Ogawa, A. Sadamasu, S. Ohtori, T. Sasho. The effect of femoral bone tunnel configuration on tendon-bone healing in an anterior cruciate ligament reconstruction: An animal study. Bone Joint Res 2018;7:327–335. DOI: 10.1302/2046-3758.75.BJR-2017-0238.R2.
Keywords: Tendon-bone healing, Sharpey-like fibres, Rabbits, Anterior cruciate ligament reconstruction, Suspensory fixation
Article focus
It is currently unclear whether fixed-loop or adjustable loop devices induce differences in tendon-bone healing after ACL reconstruction surgery.
Key messages
Regardless of whether the graft was compressed against the roof of the socket or not, tendon-bone healing after ACL reconstruction primarily occurred through indirect healing, and no significant histological or mechanical differences were observed between adjustable and fixed-loop femoral cortical suspension methods.
Strengths and limitations
Histological and biomechanical assessments were conducted.
Limitations were: the use of a small animal; the short study period; only two surgeries on each animal; the subjective nature of histological scoring; actual suspensory devices were not used; finally, a sample size calculation was not performed, therefore differences may actually exist, but were not detected.
Introduction
Anterior cruciate ligament (ACL) reconstruction is one of the most widely performed orthopaedic procedures.1,2 Hamstring tendon autografts are one of the most popular graft types,3,4 and cortical suspension devices are the most popular fixation method used.5,6
There are two major types of cortical suspension devices with a loop design. The EndoButton CL (Smith & Nephew plc, London, United Kingdom) is a fixed-loop type in which the loop length cannot be changed. This method leaves dead space between the graft end and the roof of the bone socket due to ‘flipping space’. The TightRope (Arthrex Inc., Naples, Florida) is an adjustable loop cortical suspension device in which the loop length can be shortened.7 By shortening the loop, the graft can be put into the femoral tunnel until the graft touches the roof of the socket. This leaves no gap between the graft tendon and bone socket, such that the graft is compressed against the roof. Although it is currently unclear whether fixed-loop or adjustable loop devices induce differences in tendon-bone healing. The differences in tendon-bone healing in animal models have not been reported in the literature. We hypothesized that the adjustable device creates a larger contact area between the graft tendon and bone socket and more rigid fixation, which in turn accelerates tendon-bone healing and results in a biomechanically stronger reconstructed ACL. Here, we evaluated the tendon-bone healing process and biomechanical properties of reconstructed ACLs in a rabbit model by comparing two conditions that mimic adjustable and fixed femoral cortical suspension devices.
Materials and Methods
The research protocol was reviewed and approved by the Animal Research and Care Committees of the authors’ institutions. All rabbits were supplied by Japan SLC Incorporated (Shizuoka, Japan). Skeletally mature 28-week-old New Zealand White rabbits (average body weight 3.5 kg ± 0.2) were used for the experimental model. Anterior cruciate ligament reconstruction using plantaris tendon as graft material was performed on both knees of 24 rabbits (48 knees) in order to mimic reconstruction with two different suspensory fixation devices by creating two different femoral bone tunnel configurations with the same tibial bone tunnel configuration.
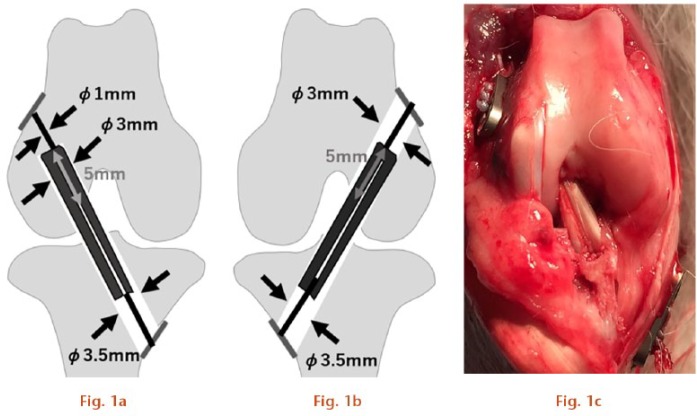
All surgical procedures were performed in the operation room attached to the institutional animal research facility. Surgery was performed under sterilization and under anaesthesia by intramuscular injection of ketamine 30 mg/kg and xylazine 2 mg/kg. The plantaris tendon was harvested from the ipsilateral extremity as a graft with a length of 35 mm. Following arthrotomy, two types of bone tunnel were created with cannulated drills using a 1.0 mm Kirschner wire as a guide, i.e. a 3.0 mm diameter, 5 mm deep socket in the lateral femoral condyle (LFC) in the right knee for the adjustable fixation device model (Socket group: group S) in which the graft end would be compressed against the socket roof, or a 3.0 mm diameter through tunnel penetrating the LFC of the left knee for the fixed-loop model (Tunnel group: group T) in which the graft end would not touch the roof. A tunnel was created in the tibia in both models using a 3.5 mm diameter drill by direct visualization to penetrate the anteromedial cortex. The harvested plantaris tendon was looped over a 2-0 silk braid and the distal end was sutured with 2-0 FiberWire (Arthrex Inc.) using the Krackow stitch. The other 2-0 FiberWire was passed into the proximal loop end of the graft. The prepared graft was introduced into the femur in an inside-out manner and pulled up so that it could be compressed against the roof of the socket in group S, or inserted only 5 mm to leave a dead space above the graft in group T. In both models, the proximal 2-0 FiberWire was connected with a suture button, the 3.5 mm AR-8920 (Arthrex Inc.), placed on the lateral femoral cortex, which for group S was tightened with maximum tension to ensure that the graft was compressed against the roof of the socket. After femoral fixation, the graft was pulled down and the distal FiberWire was sutured to the suture button placed on the anteromedial tibial cortex with sufficient tension (Fig. 1). The animals were housed individually in cages (cage area, 0.5 m2) and allowed unrestricted activity.
a) For the Socket group (group S), anterior cruciate ligament (ACL) surgeries were conducted in the right knee to mimic adjustable fixation devices; b) for the Tunnel group (group T), ACL surgeries were conducted in the left knee to mimic fixed fixation devices; c) after final fixation (Right knee).
Histological assessment
Five rabbits were sacrificed at four weeks and five at eight weeks. After macroscopic observation of the entire knee joint, resected knees were fixed in paraformaldehyde, decalcified in 20% ethylenediaminetetraacetic acid (EDTA) for four weeks, and embedded in paraffin blocks. Sections of 7 μm thickness were cut parallel to the femoral tunnel. Sections were stained with haematoxylin and eosin (H&E) to assess graft tendon integrity and fusion to the femoral bone, and Safranin O and Fast Green to assess fibrocartilage integration. Immunohistochemical (IHC) staining for collagen types II and III was performed to assess tendon remodelling. Collagen type II was labelled with anti-human collagen type II mouse monoclonal antibody clone, II-4c11 primary antibodies (1:500 dilution; Kyowa Pharma Chemical Co. Ltd, Toyama, Japan), followed by peroxidase-labelled anti-rabbit immunoglobulin G (IgG) polyclonal secondary antibody (Nichirei Biosciences Inc., Tokyo, Japan). Collagen type III was labelled with anti-human collagen type III goat polyclonal primary antibody (1:400 dilution; Southern Biotechnology Associates Inc., Birmingham, Alabama) and biotin-labelled anti-goat immunoglobulin rabbit secondary antibody (E0466; Dako Sweden AB, Stockholm, Sweden). A 50 mM phosphate buffer (pH 7.6) supplemented with bovine serum albumin (BSA) was used for all antibody diluents (diluting solution) to dilute the final concentration of 1%. Fibrocartilage and new bone formation on the margins of the graft tendon, and bonding of the graft to adjacent tissue, were assessed based on the histological scoring system developed by Yeh and Lin,8 in which the three items were scored on a four-point scale (Table I). Scoring was performed at two areas of the graft tendon: the ‘roof zone’, defined as the area spanning a length of 1.5 mm from the end of the graft loop to the roof of the socket; and the ‘side zone’, defined as the area 2 mm distal to the end of the roof zone on each side (Fig. 2). Scoring was conducted independently by three trained observers who were blinded to group and time of sacrifice. Reliability of the histological scoring was assessed by calculating the intraclass correlation coefficient (ICC) using the SPSS software (version 23.0; SPSS IBM, Hakozaki, Japan).
Table I.
Scoring system for histological examination8
| Characteristic points |
|---|
| Fibrocartilage formation |
| Abundant 3 |
| Moderate 2 |
| Slight 1 |
| None 0 |
| New bone formation |
| Abundant 3 |
| Moderate 2 |
| Slight 1 |
| None 0 |
| Tendon graft bonding to adjacent tissue |
| 75% to 100% 3 |
| 50% to 75% 2 |
| 25% to 50% 1 |
| 0% to 25% 0 |
| Highest total score 9 |
Fig. 2.
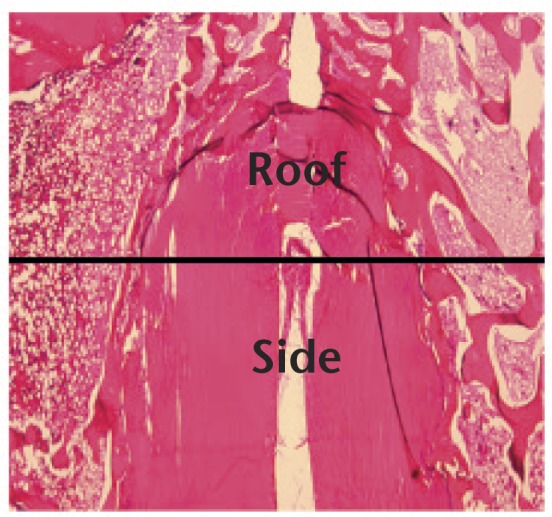
The roof zone was defined as the area spanning a length of 1.5 mm from the end of the graft loop to the roof of the socket. The side zone was defined as the area 2 mm distal to the end of the roof zone on each side (scale bar, 1.0 mm).
Biomechanical assessment
Seven rabbits were sacrificed for biomechanical testing at four weeks and another seven sacrified at eight weeks. Knee joints were disarticulated at the hip joint, and the femoral suture button and silk braid were removed before knee joints were immediately frozen and stored at -30°C. Samples were thawed and warmed to room temperature (24°C) 18 hours prior to biomechanical testing. All soft tissues were removed, leaving the ACL graft as the only structure connecting the femur and tibia. The biomechanical properties of the graft and graft-bone attachment sites were tested by applying traction to the knee in the direction of the graft using a biomechanical tester (Autograph AG-Xplus; Shimadzu Corp., Kyoto, Japan). The femur was fixed to a custom-made clamp while the tibial graft complex was pulled with a newly added 2-0 FiberWire threaded at the suture button. The reference position was the position at which a preload of 1 N was applied for five minutes at 24°C, and was defined as 0 mm. The specimen was stretched to failure at a rate of 10 mm/minute for uniaxial tensile testing, and final failure load and elongation were recorded on a load-displacement curve. The results were analyzed using the material test operation software TRAPEZIUM X (Shimadzu Corp).
Statistical analysis
All data are presented as mean and sd. The Mann-Whitney U test was used to analyze between-group differences for biomechanical and histological assessments at each timepoint. Reliability of the histological scoring was assessed by calculating the intraclass correlation coefficient (ICC). All analyses were performed using SPSS software (version 23.0; SPSS IBM, Hakozaki, Japan). Differences were considered statistically significant at p < 0.05. A sample size calculation was not performed.
Results
Gross findings
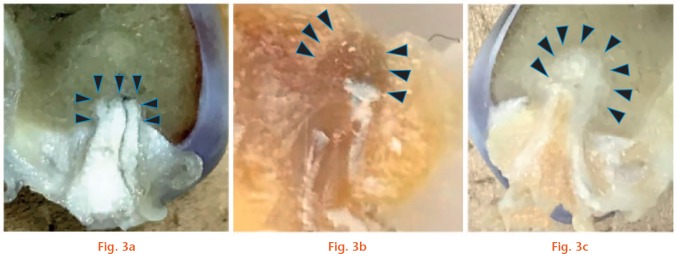
In all knees from group T, graft markings were maintained in the aperture of the femoral tunnel. At the time of insertion into the tunnel, graft ends in group T were loop-shaped (semi-elliptical), observed when the specimen was cut at the appropriate position. At four weeks, there was new bone formation in the tunnel above the graft end, however, the boundary between the surrounding cancellous bone was still distinguishable. At eight weeks, the newly formed bone was replaced with cancellous bone-like tissue that was integrated into the surrounding cancellous bone. In knees from group S, grafts were in close contact with the socket at both four and eight weeks. The graft end was quadrangular, fitting into the end of the socket. No gap remained between the graft end and the roof of the socket in any of the specimens (Fig. 3).
Sections along the tunnel axis after fixation in paraformaldehyde are shown (cut at the centre of the cross section of the graft): a) in the knees from the Socket group (right side), grafts were in close contact with the socket at both four and eight weeks. The graft end was quadrangular, fitting into the end of the socket; b) at four weeks, new bone formation in the tunnel above the graft end was observed, however, the boundary between the surrounding cancellous bone was clearly distinguishable; c) at eight weeks, the graft end in the Tunnel group was loop-shaped (semi-elliptical) at the time of insertion into the tunnel, and the newly formed bone was replaced with cancellous bone-like tissue which had integrated into the surrounding cancellous bone when the specimen was cut at the appropriate position. Arrowheads in (a), (b) and (c) indicate the contour of the tendon/bone interface.
Histological assessment of group T
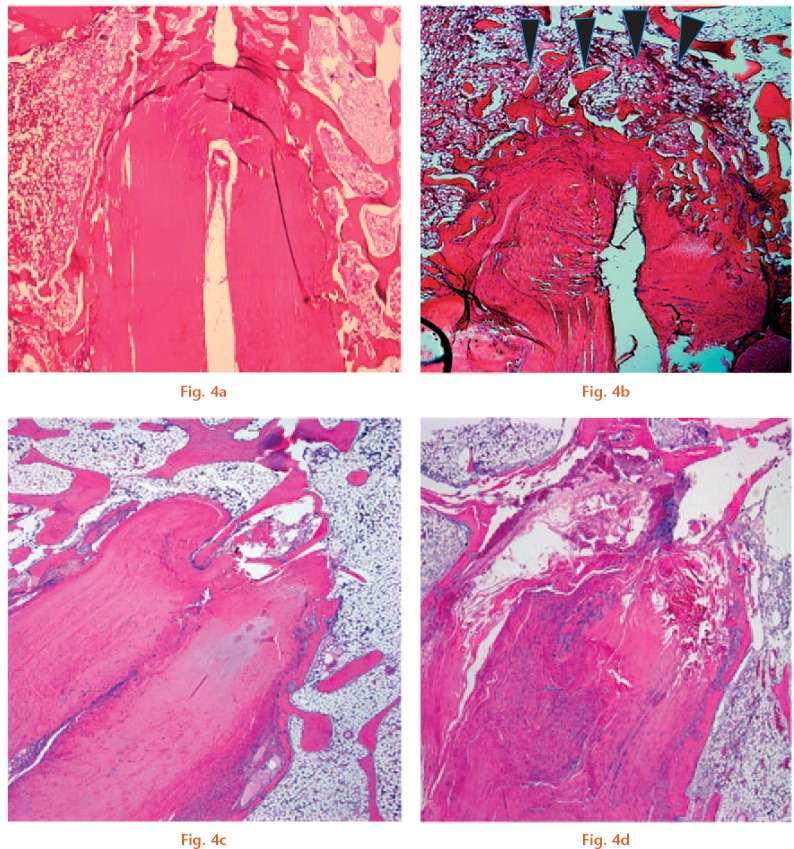
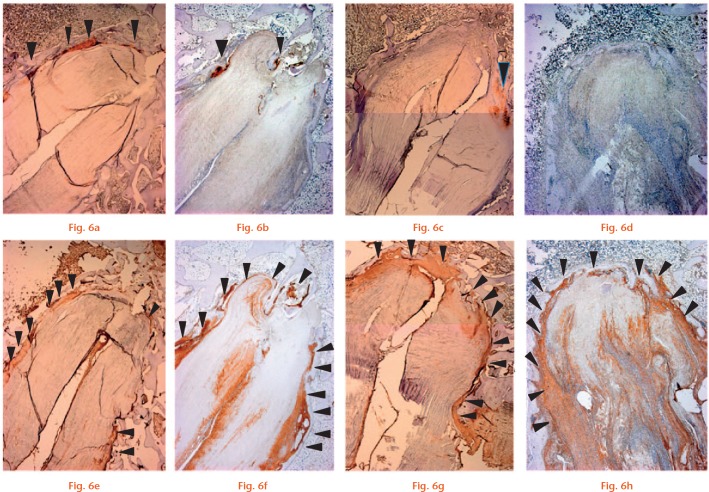
At four weeks, newly formed trabecular bone filled the gap in the tunnel above the graft end (Fig. 4b). Some Safranin O and type II collagen staining was observed in the roof zone, showing signs of direct healing (Figs 5b and 6c). Type III collagen was observed at the interface between the graft and new trabecular bone, showing signs of initial indirect healing. Osteointegration of the graft had begun at the interface between the graft and sidewall of the host bone, and type III collagen staining was more abundant in the side zone compared with the roof zone (Fig. 6f).
Representative photomicrographs of the tendon/bone interface with haematoxylin and eosin (H&E) staining (magnification × 25): a) specimen from the Socket group at four weeks; b) specimen from the Tunnel group at four weeks shows that new trabecular bone (arrowheads) has already covered the roof zone; c) specimen from the Socket group; and d) Tunnel group at eight weeks (scale bars, 1.0 mm).
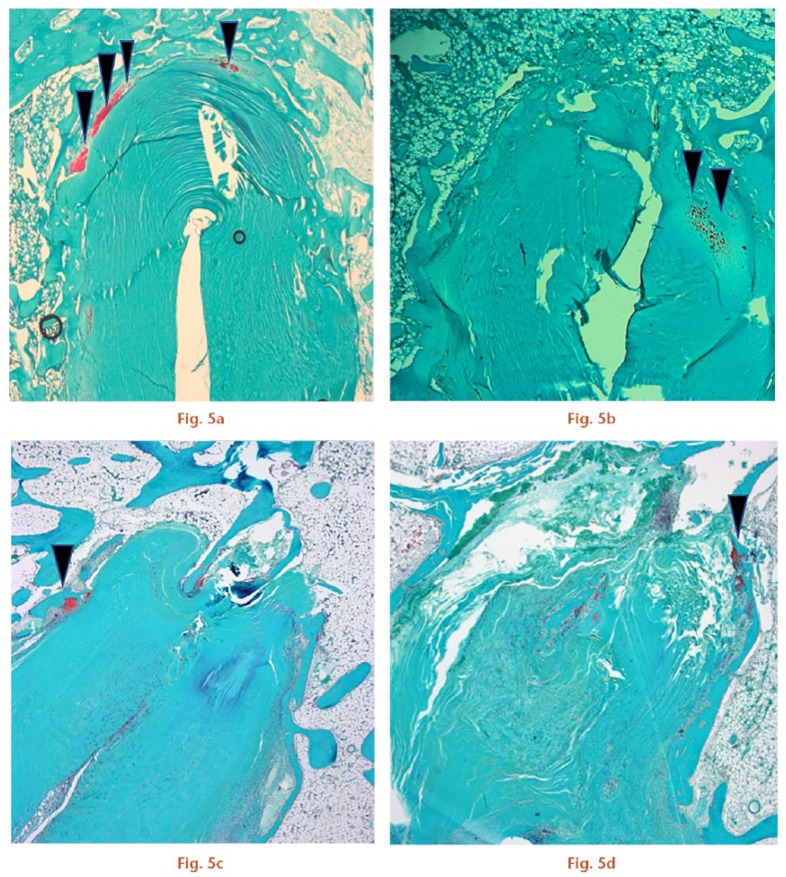
Photomicrographs of the tendon/bone interface with Safranin O staining (magnification × 25). Cartilage tissue is indicated by the arrowheads. a) The Socket group had more cartilage tissue than b) the Tunnel group at four weeks. Cartilage tissue was reduced at eight weeks in both c) the Socket group and d) Tunnel group (scale bars, 1.0 mm).
Photomicrographs of the tendon/bone interface with immunohistochemical staining for cartilage (type II collagen) (a-d) (magnification × 25). Cartilage tissue (arrowheads) was observed at four weeks in a) the Socket group and b) Tunnel group. At eight weeks, staining for cartilage was reduced in both the c) Socket group and d) Tunnel group. e-h) Photomicrographs of the tendon/bone interface with immunohistochemical staining for type III collagen (magnification × 25). Type III collagen (equivalent to Sharpey’s fibres; arrowheads) was primarily expressed at the tendon/bone interface at four weeks in the e) Socket and f) Tunnel groups. Staining was similar at eight weeks in the g) Socket and h) Tunnel groups (scale bars, 1.0 mm).
At eight weeks, trabecular bone within the roof zone had completely integrated into the surrounding cancellous bone (Fig. 4d). Cartilage tissue in the roof zone had decreased compared with that observed at four weeks (Fig. 5d). The roof and side zones were rich in type III collagen (Fig. 6h).
Histological assessment of group S
At four weeks, osteointegration of the graft had begun in the roof and side zones. Four rabbits showed staining for Safranin O and type II collagen at the interface of the roof and side zones, showing signs of direct healing (Figs 5a and 6a), while one rabbit showed no fibrocartilage staining. More abundant staining for fibrocartilage was observed in group S than in group T. Type III collagen was expressed in the roof and side zones, showing signs of indirect healing (Fig. 6f).
At eight weeks, only two specimens showed scant staining for cartilage tissue in both zones (Fig. 5d), while staining for type III (Sharpey-like) collagen fibres was increased around the graft, as was observed in group T (Fig. 6g).
Quantitative assessment of tendon-bone healing
The ICC (Intraclass correlation coefficient) was greater than 0.9 for all histological scorings (Table II). There were no significant differences between the two groups at four weeks in the roof zone (S: mean 4.8 sd 1.7, T: mean 4.5 sd 0.5, p = 0.14, Mann-Whitney U test) or side zone (S: mean 5.0 sd 1.2, T: mean 4.8 sd 0.4, p = 0.43, Mann-Whitney U test). Similarly, there were no significant differences between the two groups at eight weeks in the roof zone (S: mean 5.8 sd 0.8, T: mean 4.8 sd 1.8, p = 0.88, Mann-Whitney U test) or side zone (S: mean 5.3 sd 0.8,T: mean 5.5 sd 0.8, p = 0.61, Mann-Whitney U test) (Table II).
Table II.
Histological score
| Weeks from anterior cruciate ligament operation | Socket group | Tunnel group | p-value |
|---|---|---|---|
| Roof at 4 weeks | 4.8 (1.7) | 4.5 (0.5) | 0.14 |
| Side at 4 weeks | 5.0 (1.2) | 4.8 (0.4) | 0.88 |
| Roof at 8 weeks | 5.8 (0.8) | 4.8 (1.8) | 0.43 |
| Side at 8 weeks | 5.3 (0.8) | 5.5 (0.8) | 0.61 |
All data indicate mean (sd). All comparisons were conducted using a Mann-Whitney U test. Intraclass correlation coefficient (ICC) for the ‘roof zone’: 0.900 (95 % CI 0.797 to 0.955), ICC for the ‘side zone’: 0.921 (95% CI 0.839 to 0.965).
Biomechanical assessment
In all cases, grafts were stretched prior to pulling out from the bone tunnel. There was no significant difference in the maximum failure load between group S and group T at four weeks (15.6 N, sd 9.0 and 13.6 N, sd 5.6, respectively; p = 0.753) or at eight weeks (12.6 N, sd 3.6 and 17.1 N, sd 6.4, respectively; p = 0.237) In addition, the maximum failure load did not differ between four and eight weeks in either the S or T groups (Table III).
Table III.
Maximum failure load (N)
| Time from anterior cruciate ligament operation | Socket group | Tunnel group | p-value |
|---|---|---|---|
| Four weeks | 15.6 (9.0) | 13.6 (5.6) | 0.75* |
| Eight weeks | 12.6 (3.6) | 17.1 (6.4) | 0.24* |
| p-value | 0.88† | 0.57† |
Analysis between two groups.
Analysis between two timepoints.
All data indicate mean (sd). All comparisons were conducted using the Mann-Whitney U test.
Discussion
Fibrous (Sharpey’s fibre) and fibrocartilaginous entheses have been referred to as ‘indirect’ and ‘direct’, respectively.9 While a mixture of direct and indirect healing was observed in both groups at four weeks, indirect healing was primarily observed in both groups at eight weeks. Additionally, there was no significant difference in mechanical strength between the two groups at four and eight weeks. At the time of animal surgery, a cavity was created between the graft end roof of the femoral tunnel in group T to mimic fixed-loop ACL procedures in humans. In contrast, the graft adhered to the top of the femoral tunnel leaving no cavity in the adjustable fixation device model (group S). At four weeks, group S showed signs of direct healing and group T exhibited the initial stages of trabecular bone tissue formation at the graft end. At eight weeks, the healing features around the graft were similar between the two groups. Therefore, both groups exhibited a similar healing process, primarily through an indirect healing pattern. There was no significant difference in the amount of cartilage tissue between the two groups at eight weeks. Type III collagen, which appears to correspond to Sharpey’s fibres, was mainly observed in the tendon/bone interface in both groups at eight weeks.
Recently, Smith et al10 conducted a similar animal experiment with an adjustable fixation device using TightRope in a dog model. They reported that fixation using TightRope resulted in direct healing due to a reduction in bungee-cord and windshield-wiper effects. Their model was a hybrid double-bundle reconstruction, which left the posterolateral bundles of the ACL intact. Therefore, the load on the reconstructed grafts (i.e. anteromedial bundles) was small, potentially leading to further reductions in the bungee-cord effect, facilitating direct healing. This suggests that, if the ideal environment is present, direct healing would occur without adding exogenous growth factors. In the present study, we found no significant differences between the two groups at eight weeks. However, slightly wider cartilage areas were observed in the S group at four weeks, which we speculated was attributable to less graft movement due to the existence of the socket roof. However, this decrease might still be insufficient in comparison with Smith’s hybrid dog model. Therefore, it remains controversial whether adjustable fixation devices can better facilitate direct healing than can fixed-loop devices.
In general, tendon-bone healing after ACL reconstruction has been reported to occur via indirect healing. Tabuchi et al11 reported that, in rabbits, type III collagen (Sharpey's fibres) was expressed at the tendon/bone interface at around four weeks post-ACL reconstruction, and gradually increased between six and 12 weeks, after which they were replaced by type I collagen which formed strong bonds with bone. On the other hand, a variety of studies have attempted to induce a direct healing pattern to form the same structure as that of normal ACL enthesis by employing the addition of cytokines or cells. Some reports have shown that mesenchymal stem cells (MSCs) can stimulate a direct healing pattern.12,13 Pan et al14 and Cheng et al15 reported that growth factors, such as bone morphogenetic protein 2 (BMP-2), are effective for initiating a direct healing pattern. Zhang et al16 reported that the combined use of kartogenin and platelet-rich plasma (PRP) promotes fibrocartilage formation in the wounded rat Achilles tendon. However, unless the tendon/bone interface is incubated with growth factors, indirect healing is the most common mechanism,12-15 regardless of surgical procedure or bone tunnel configuration. In our study, cartilage tissue was observed at the tendon-bone boundary at four weeks in both groups, some of which showed direct healing patterns. At eight weeks, however, most showed indirect healing patterns. Therefore, our results were similar to those of these previous studies, with groups S and T showing indirect healing and little difference in the healing process despite differences in bone tunnel configuration. In our study, no significant difference was seen in mechanical strength between the two groups at four and eight weeks, since indirect healing was predominantly observed in both groups. All graft failures occurred in such a way that grafts pulled out of the bone tunnel following graft elongation, which suggests that tendon bone healing is not sufficient at four weeks and eight weeks after ACL reconstruction in rabbits.11
This study had several limitations. First, we used rabbits to generate our experimental model. Since these were too small to allow use of the actual suspension devices, we could only mimic the situation of the two types of suspensory fixations by creating different types of bone tunnels. Kondo et al17 performed double-bundle reconstruction using semitendinosus tendon with EndoButton fixation in goats. Zantop et al18 reported differences in mechanical strength due to tendon graft insertion length in goats. Using large animals would be better in this type of study but rabbits were large enough to serve as a reproducible surgical model. In addition, the lack of a significant difference in healing between group T and group S may be due to the small diameter of the bone tunnels in rabbits (3 mm). However, a similar process of new bone-occupied tunnels has also been reported in humans.19 Thus, we speculate that our model was not too dissimilar to actual clinical conditions. Second, our study lacked a sample size calculation due to the lack of a pilot study. We determined the sample sizes simply on the basis of similar reports8,20-23 in which three to seven rabbits were used per group. Third, the short study period might be another limitation. Histological differences may be seen in a longer observation. Fourth, two surgeries were performed on a rabbit to minimize the number of animals used. Fifth, the subjective nature of histological assessment is an inevitable limitation of this type of this study.
The results of the present study suggest that surgeons may choose from among various types of suspensory fixation devices in terms of tendon-bone healing of the femur. Regardless of whether the graft was separated from, or compressed against, the roof of the socket, tendon-bone healing after ACL reconstruction primarily occurred through indirect healing. No significant histological or mechanical differences were observed between the socket-type bone tunnel and through-type tunnel in the sample size and time frame studied.
Acknowledgments
The authors acknowledge the invaluable contributions of Prof Takahisa Sasho and the Department of Orthopaedic Surgery, Graduate School of Medicine, Chiba University.
Footnotes
Author Contributions: Y. Sato, Performed experiment and drafted the manuscript.
R. Akagi, Discussed the results and implications and commented on the manuscript.
Y. Akatsu, Technical support and conceptual advice.
Y. Matsuura, Technical support and conceptual advice.
S. Takahashi, Helped to collect the data and perform the analysis.
S. Yamaguchi, Discussed the results and implications and commented on the manuscript.
T. Enomoto, Helped to collect the data and perform the analysis.
R. Nakagawa, Helped to collect the data and perform the analysis.
H. Hoshi, Technical support and conceptual advice.
T. Sasaki, Technical support and conceptual advice.
S. Kimura, Sacrifice of animals and macroscopic evaluation.
Y. Ogawa, Sacrifice of animals and macroscopic evaluation.
A. Sadamasu, Sacrifice of animals and macroscopic evaluation.
S. Ohtori, Supervised the project.
T. Sasho, Designed the project and drafted the manuscript.
Conflict of Interest Statement: None declared
Follow us @BoneJointRes
Funding Statement
None declared
References
- 1. S’migielski R, Zdanowicz U, Drwie˛ga M, Ciszek B, Williams A. The anatomy of the anterior cruciate ligament and its relevance to the technique of reconstruction. Bone Joint J 2016;98-B:1020-1026. [DOI] [PubMed] [Google Scholar]
- 2. Ahmed I, Salmon L, Roe J, Pinczewski L. The long-term clinical and radiological outcomes in patients who suffer recurrent injuries to the anterior cruciate ligament after reconstruction. Bone Joint J 2017;99-B:337-343. [DOI] [PubMed] [Google Scholar]
- 3. Chechik O, Amar E, Khashan M, et al. An international survey on anterior cruciate ligament reconstruction practices. Int Orthop 2013;37:201-206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Grassi A, Nitri M, Moulton SG, et al. Does the type of graft affect the outcome of revision anterior cruciate ligament reconstruction? a meta-analysis of 32 studies. Bone Joint J 2017;99-B:714-723. [DOI] [PubMed] [Google Scholar]
- 5. Petre BM, Smith SD, Jansson KS, et al. Femoral cortical suspension devices for soft tissue anterior cruciate ligament reconstruction: a comparative biomechanical study. Am J Sports Med 2013;41:416-422. [DOI] [PubMed] [Google Scholar]
- 6. Longo UG, Ciuffreda M, Casciaro C, et al. Anterior cruciate ligament reconstruction in skeletally immature patients: a systematic review. Bone Joint J 2017;99-B:1053-1060. [DOI] [PubMed] [Google Scholar]
- 7. Andrew J, Michael J. All-inside anterior cruciate ligament reconstruction. J Knee Surg 2014;127:347-352. [DOI] [PubMed] [Google Scholar]
- 8. Yeh W-L, Lin S. Effects of hyperbaric oxygen treatment on tendon graft and tendon-bone integration in bone tunnel: biochemical and histological analysis in rabbits. J Orthop Res 2007;25:636-645. [DOI] [PubMed] [Google Scholar]
- 9. Apostolakos J, Durant TJS, Dwyer CR, et al. The enthesis: a review of the tendon-to-bone insertion. Muscles Ligaments Tendons J 2014;4:333-342. [PMC free article] [PubMed] [Google Scholar]
- 10. Smith PA, Stannard JP, Pfeiffer FM, et al. Suspensory versus interference screw fixation for arthroscopic anterior cruciate ligament reconstruction in a translational large-animal model. Arthroscopy 2016;32:1086-1097. [DOI] [PubMed] [Google Scholar]
- 11. Tabuchi K, Soejima T, Kanazawa T, Noguchi K, Nagata K. Chronological changes in the collagen-type composition at tendon-bone interface in rabbits. Bone Joint Res 2012;1:218-224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Dong Y, Zhang Q, Li Y, Jiang J, Chen S. Enhancement of tendon-bone healing for anterior cruciate ligament (ACL) reconstruction using bone marrow-derived mesenchymal stem cells infected with BMP-2. Int J Mol Sci 2012;13:13605-13620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Jang K-M, Lim HC, Jung WY, Moon SW, Wang JH. Efficacy and safety of human umbilical cord blood-derived mesenchymal stem cells in anterior cruciate ligament reconstruction of a rabbit model: new strategy to enhance tendon graft healing. Arthroscopy 2015;31:1530-1539. [DOI] [PubMed] [Google Scholar]
- 14. Pan W, Wei Y, Zhou L, Li D. Comparative in vivo study of injectable biomaterials combined with BMP for enhancing tendon graft osteointegration for anterior cruciate ligament reconstruction. J Orthop Res 2011;29:1015-1021. [DOI] [PubMed] [Google Scholar]
- 15. Cheng P, Han P, Zhao C, et al. High-purity magnesium interference screws promote fibrocartilaginous entheses regeneration in the anterior cruciate ligament reconstruction rabbit model via accumulation of BMP-2 and VEGF. Biomaterials 2016;81:14-26. [DOI] [PubMed] [Google Scholar]
- 16. Zhang J, Yuan T, Zheng N, et al. The combined use of kartogenin and platelet-rich plasma promotes fibrocartilage formation in the wounded rat Achilles tendon entheses. Bone Joint Res 2017;6:231-244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Kondo E, Yasuda K, Katsura T, et al. Biomechanical and histological evaluations of the doubled semitendinosus tendon autograft after anterior cruciate ligament reconstruction in sheep. Am J Sports Med 2012;40:315-324. [DOI] [PubMed] [Google Scholar]
- 18. Zantop T, Ferretti M, Bell KM, et al. Effect of tunnel-graft length on the biomechanics of anterior cruciate ligament-reconstructed knees: intra-articular study in a goat model. Am J Sports Med 2008;36:2158-2166. [DOI] [PubMed] [Google Scholar]
- 19. Weber AE, Delos D, Oltean HN, et al. Tibial and femoral tunnel changes after ACL reconstruction: a prospective 2-year longitudinal MRI study. Am J Sports Med 2015;43:1147-1156. [DOI] [PubMed] [Google Scholar]
- 20. Kuang GM, Yau WP, Lu WW, Chiu KY. Use of a strontium-enriched calcium phosphate cement in accelerating the healing of soft-tissue tendon graft within the bone tunnel in a rabbit model of anterior cruciate ligament reconstruction. Bone Joint J 2013;95-B:923-928. [DOI] [PubMed] [Google Scholar]
- 21. Kuang GM, Yau WP, Lu WW, Chiu KY. Local application of strontium in a calcium phosphate cement system accelerates healing of soft tissue tendon grafts in anterior cruciate ligament reconstruction: experiment using a rabbit model. Am J Sports Med 2014;42:2996-3002. [DOI] [PubMed] [Google Scholar]
- 22. Soon MYH, Hassan A, Hui JH, Goh JC, Lee EH. An analysis of soft tissue allograft anterior cruciate ligament reconstruction in a rabbit model: a short-term study of the use of mesenchymal stem cells to enhance tendon osteointegration. Am J Sports Med 2007;35:962-971. [DOI] [PubMed] [Google Scholar]
- 23. Martinek V, Latterman C, Usas A, et al. Enhancement of tendon-bone integration of anterior cruciate ligament grafts with bone morphogenetic protein-2 gene transfer: a histological and biomechanical study. J Bone Joint Surg [Am] 2002;84-A:1123-1131. [DOI] [PubMed] [Google Scholar]