Summary
Neoadjuvant chemoradiation (CRT) followed by surgical resection is the standard of care for resectable, locally advanced esophageal cancer. There are promising results using 41.4 Gy relative to historical controls using higher doses, but the utilization and efficacy of lower neoadjuvant radiation dosing is unclear. This study uses the National Cancer Database (NCDB) to explore patterns of care for neoadjuvant CRT dose levels and outcomes. The NCDB was queried for localized invasive esophageal adenocarcinoma (AC) or squamous cell carcinoma (SCC) receiving neoadjuvant CRT with doses from 40 to 54 Gy followed by surgical resection. Patients were divided into radiation levels: 40–41.4, 45, 50.4, and 54 Gy, respectively. Factors predicting use of 40–41.4 Gy vs. all other dose levels were compared using multivariable logistic regression. Factors affecting overall survival (OS) were compared using univariate and multivariate modeling. A total of 6,274 patients with AC (n = 5,176) or SCC (n = 1,098) receiving neoadjuvant CRT and definitive resection were identified. Hispanic race (OR 2.67 [95% CI 1.22–5.81]) and treatment at an academic center (OR 2.72 [95% CI 1.15–6.41]) predicted for use of low-dose CRT. Lower dose CRT increased from 3.9% in 2004 to 7.2% in 2013. There was no difference in OS when stratified according to radiation dose level (P = 0.48). Multivariable analysis found private/government insurance, higher education, higher median income, and treatment at an academic center were associated with improved OS. Age, male gender, Charlson–Deyo comorbidity score, stage, tumor grade, and treatment in the South were associated with worse OS. Use of lower neoadjuvant CRT dose is more common at academic centers and shows possible increasing usage. Neoadjuvant radiation dose for esophageal cancer is not associated with differences in OS in this large database.
Keywords: chemoradiation, esophageal cancer, radiation dose, surgery
INTRODUCTION
The benefit of neoadjuvant chemoradiation (CRT) followed by surgical resection in the management of locally advanced esophageal cancer has been explored in multiple randomized trials.1–3 While there was some discrepancy or deficiencies in results from earlier trials with regard to the benefit of neoadjuvant CRT, the Chemoradiotherapy for Oesophageal Cancer Followed by Surgery Study (CROSS) trial established a clear overall survival benefit over surgery alone with this approach.4 The benefit was seen in both adenocarcinoma (AC) and squamous cell carcinoma (SCC). Interestingly, other parameters were also shown to improve using this neoadjuvant regimen, including local recurrence, nodal recurrence, and even peritoneal recurrence rates.5
In addition to showing an improvement in outcomes, the CROSS trial raised new clinical questions based the type and dose of chemoradiation used. To start, carboplatin and paclitaxel were used in contrast to the 5-fluorouracil (5FU) and cisplatin used in previous randomized trials. The radiation dose used was also 41.4 Gy delivered in 23 fractions, significantly reduced from the standard dose used in most clinical trials of 50.4 Gy or higher. The radiation fields were also slightly smaller than used in the Cancer and Leukemia Group B (CALGB) 9781 study.3
With such promising results from the CROSS trial in terms of pathological complete response (pCR), local control, and overall survival (OS), a major question remains whether there is any benefit to using higher neoadjuvant doses in operable patients. In theory, lower neoadjuvant radiation dose reduces acute toxicity, shortens time before surgery, may decrease postoperative complications, and reduce late radiation toxicity. The late effects of radiation in esophageal cancer are becoming increasingly scrutinized as more data emerges regarding late cardiopulmonary toxicity from radiation for esophageal cancer.6,7 Conversely, one might argue that higher doses would produce even superior outcomes than CROSS with regard to pCR, local control, and survival. While no benefit was found to dose escalation in Intergroup Trial 0122, this was tested in the definitive setting and at higher doses than reflect the current standard of care.8 Another concern is that 41.4Gy would be an inadequate definitive dose in the event that patients are ultimately unable to undergo surgery after neoadjuvant treatment.
This analysis uses the National Cancer Database (NCDB) to explore the effect of radiation dose level in the neoadjuvant setting for curative treatment of esophageal cancer with regard to overall survival. Patterns of care and predictors for the use of lower dose neoadjuvant CRT are also explored as well as factors predicting mortality.
METHODS
Data source
The NCDB is a hospital-based retrospective dataset of oncology patients managed as a joint project from the American Cancer Society and the American College of Surgeons’ Commission on Cancer (CoC). This registry captures data from over 1,500 hospitals nationwide and represents approximately 70% of cancers diagnosed in the United States. The data is deidentified and available upon request for analysis by CoC participating locations. After our request, we were provided with all data on cases of esophageal cancer diagnosed from 2004 to 2013. This study was determined to be exempt from Institutional Review Board given deidentified data.
Study patients
From the initial database, 114,758 patients with invasive esophageal cancer were identified. Figure 1 includes a CONSORT diagram that details the type and extent of patient exclusions. We excluded patients that were metastatic (clinically or pathologically), had unknown staging, or had histology other than SCC or AC. We further excluded patients that did not undergo surgery, had unknown surgery, or had nononcologic surgeries such as local ablation. Patients were excluded if they had no radiation (RT), did not receive neoadjuvant radiation as coded in NCDB, doses outside standard of care (40–54 Gy), or did not receive concurrent chemotherapy. While not directly coded in the database, concurrent chemoradiation was defined as starting chemotherapy and radiation within 30 days of the other. The final analysis cohort included 6,274 total patients.
Fig. 1.
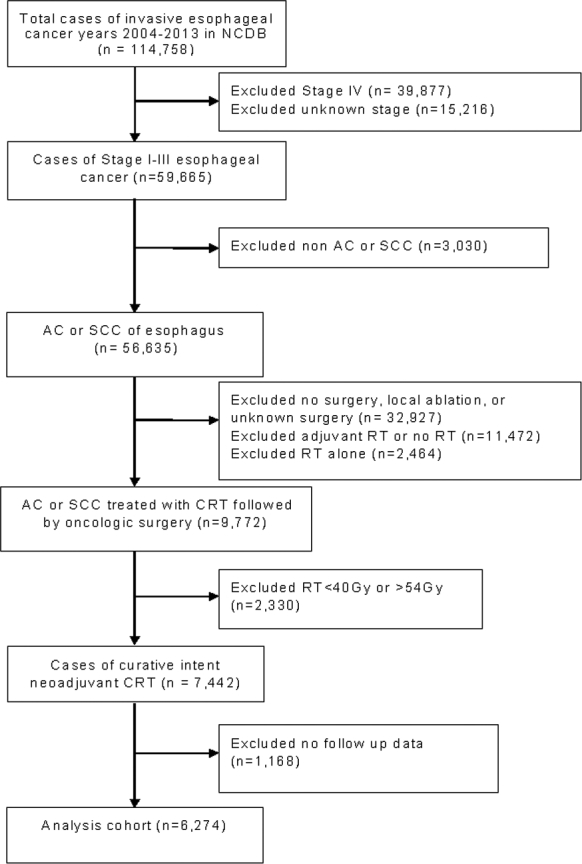
CONSORT diagram.
Prognostic variables
Potentially relevant demographic and clinicopathologic variables were included. Demographic variables included age, gender, race, Hispanic origin, insurance status, median household income, education, distance from medical facility, facility type, geographical region, rural/urban status, and year of diagnosis. Age was categorized as ≥60 or <60 years. Race was categorized as white, black, Asian, and other. Insurance status included uninsured, private, and government, with the latter including both Medicare and Medicaid. Education was assessed by the percentage of adults without high school diploma in 2012 U.S. Census data: <7%, 7%–12.9%, 13%–20.9, and ≥21%. Median household income was assessed by linking a patient's zip code to 2012 U.S. Census data and grouped as <$30,000, $30,000–$35,999, $36,000–$45,999, $46,000+. Facility type was categorized into community, comprehensive, academic, and other. Distance from facility was estimated as <12.5, 12.5–50, and ≥50 miles. Location of facility was divided into Northeast, Central, South, and West. Rurality and urban influence was coded as Rural, Urban, and Metro. Clinicopathologic variables included a Charlson–Deyo comorbidity score (CDCC), histology, stage, and grade. Tumor grade was categorized as well, moderately, poorly differentiated and undifferentiated. Histologies were grouped as squamous cell carcinoma or adenocarcinoma.
Missing data
Missing data for prognostic variables outlined above were handled by multiple imputation using chained equations.9 Among these variables, Hispanic origin had the highest frequencies of missing data at 6.8%, followed by rural/urban status (3.6%), median income (2.0%), education (2.0%), facility type, and geographical region (1.6%), insurance status (1.3%), race (0.7%) and distance from residence to facility (0.03%). This sequential regression imputation method was implemented using the IVEware software system with 10 repetitions to generate 15 imputed datasets.
Statistical analysis
The primary objective of this study is to compare overall survival (OS), defined as the months from diagnosis to last contact or death, among four radiation dose groups (40–41.4, 45, 50.4, and 54 Gy). Secondary objectives included identifying trends in utilization of the lowest dose group (40–41.4Gy) over the period of 2004–2013. An additional objective was to explore predictors for the use of lower dose radiation compared to higher neoadjuvant doses.
Demographic and clinicopathologic variables outlined above were compared among dose groups using a Chi-square test. Continuous variables were reported as mean (SD) and nominal variables were reported as n (%). A multivariable logistic regression model was used to estimate adjusted odds ratios (OR) and 95% confidence intervals (95% CI) to evaluate the association between each variable and use of the lowest dose group (40–41.4 Gy) while adjusting for variables outlined above.
OS curves were computed by the Kaplan–Meier method and compared among dose groups with the log-rank test. The Cox proportional hazards (CPH) model was used to estimate univariable and multivariable hazard ratios (MHRs) for OS and corresponding 95% CIs. All prognostic variables identified above were included in the multivariable CPH model. Pairwise MHRs were also computed to compare the hazard rates in lower dose with higher dose groups. To account for the multiple imputations of missing data, CPH regression was performed on each of the 15 imputed datasets, and estimates of corresponding HRs and 95% CIs were then appropriately combined using the MIANALYZE procedure in ref.10. All statistical analyses described above were performed using SAS Version 9.4 (SAS Institute, Cary, NC). Hypothesis testing was two-sided and conducted at the 5% level of significance.
The Joinpoint Regression Program (Version 4.3.1.0) developed by the US National Cancer Institute was used to assess temporal trends in annual utilization rates of the lowest dose group. The joinpoint software fits the simplest model to describe the utilization rate trend data, starting with a straight line (0 joinpoint) and then adding more joinpoints to determine whether multiple connecting lines better describe the data points. The software identifies the year(s) when the annual percentage change (APC) trends appear to shift upward or downward and whether these trends are statistically significant.
RESULTS
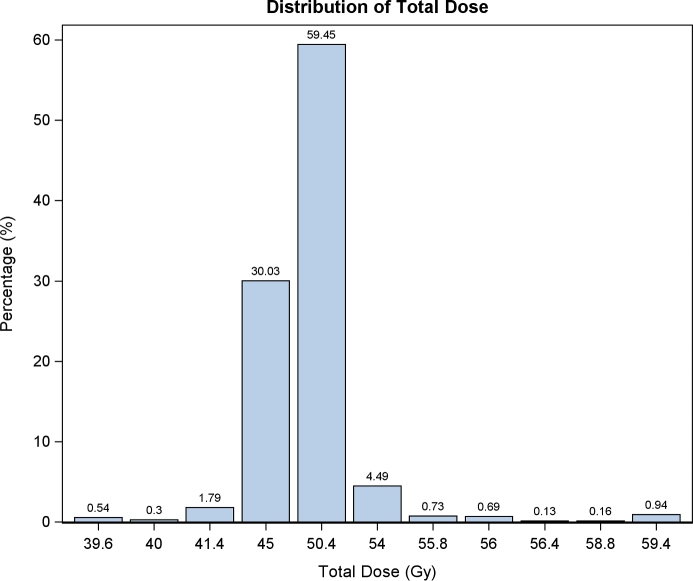
In total, 6,274 patients with localized esophageal AC (n = 5,176) or SCC (1,098) treated with neoadjuvant CRT followed by definitive resection were identified from the years 2004–2013. Definitive radiation doses were defined between 40 and 54 Gy, and all patients received concurrent chemotherapy. A histogram of neoadjuvant doses used in the total cohort is shown in Figure 2. Multiagent chemotherapy was used in 88.6% (n = 5,559), single agent was used in 5.9% (n = 371), and unspecified agent number was used in 5.5% (n = 344). Radiation dose levels were further defined into four groups based on either clinical definitions or natural clustering: 40–41.4 Gy 9 (n = 177), 45 Gy (n = 1884), 50.4 Gy (n = 3730), or 54 Gy (n = 483). Patient characteristics with regard to dose levels are summarized in Table 1. Overall, the groups were well matched in terms in clinical features except for ethnicity (Hispanic vs. non-Hispanic), although statistically significant differences without obvious trends were seen in some parameters. A notable exception is a disproportionate use of lower dose radiation in academic centers (56%) vs. community (3%) or comprehensive cancer centers (36%) (P < 0.001). There also appeared to be less use of lower dose radiation for AC than for SCC. There was no difference between the groups with respect to stage, age, race, gender, insurance status, and Charlson-Deyo comorbidity score.
Fig. 2.
Histogram of neoadjuvant radiation doses used for at least 0.1% of the population.
Table 1.
Baseline patient, facility, and disease characteristics by dose level N = 6,274
| Dose level (Gy) | Multivariable OR 45, 50.4, 54 vs 40–41.4 Gy | ||||||
|---|---|---|---|---|---|---|---|
| 40–41.4 | 45 | 50.4 | 54 | ||||
| N | 177 | 1,884 | 3,730 | 483 | P-value | OR (95% CI) | P-value |
| Age | |||||||
| <60 | 76 (43%) | 813 (43%) | 1634 (44%) | 212 (44%) | 0.96 | ||
| >= 60 | 101 (57%) | 1071 (57%) | 2096 (56%) | 271 (56%) | |||
| Mean (SD) | 62.36 (9.81) | 61.34 9.30 | 61.49 9.38 | 61.56 9.16 | 1.01 (0.99, 1.03) | 0.35 | |
| Median (Range) | 62 [38,86] | 62 [22,89] | 62 [23,88] | 62 [30,83] | |||
| Sex | |||||||
| Female | 37 (21%) | 281 (15%) | 543 (15%) | 65 (13%) | 0.11 | Ref | |
| Male | 140 (79%) | 1603 (85%) | 3187 (85%) | 418 (87%) | 0.75 (0.51, 1.12) | 0.16 | |
| Hispanic | |||||||
| Non-Hispanic | 169 (95%) | 1850 (98%) | 3657 (98%) | 468 (97%) | 0.03 | Ref | 0.014 |
| Hispanic | 8 (5%) | 34 (2%) | 73 (2%) | 15 (3%) | 2.67 (1.22, 5.81) | ||
| Race | |||||||
| White | 166 (94%) | 1768 (94%) | 3509 (94%) | 451 (93%) | 0.80 | Ref | |
| Black | 8 (5%) | 84 (4%) | 166 (4%) | 21 (4%) | 0.91 (0.41, 2.00) | 0.81 | |
| Asian | 3 (2%) | 21 (1%) | 44 (1%) | 8 (2%) | 1.00 (0.30, 3.32) | 0.99 | |
| Other | 0 (0%) | 11 (1%) | 11 (0%) | 3 (1%) | Not estimable | 0.97 | |
| Insurance type | |||||||
| Not insured | 4 (2%) | 43 (2%) | 73 (2%) | 15 (3%) | 0.53 | Ref | |
| Private | 89 (50%) | 1014 (54%) | 1962 (53%) | 244 (51%) | 0.99 (0.35, 2.80) | 0.97 | |
| Government | 84 (47%) | 827 (44%) | 1695 (45%) | 224 (46%) | 0.96 (0.33, 2.77) | 0.93 | |
| % without HS education | |||||||
| > = 21% | 20 (11%) | 229 (12%) | 418 (11%) | 72 (15%) | 0.25 | Ref | |
| 13%–20.9% | 50 (28%) | 488 (26%) | 967 (26%) | 134 (28%) | 1.22 (0.68, 2.18) | 0.50 | |
| 7%–12.9% | 64 (36%) | 733 (39%) | 1404 (38%) | 170 (35%) | 1.00 (0.54, 1.89) | 0.99 | |
| <7% | 43 (24%) | 434 (23%) | 941 (25%) | 107 (22%) | 0.82 (0.40, 1.69) | 0.59 | |
| Median Income Quartiles | |||||||
| <$30,000 | 29 (16%) | 326 (17%) | 534 (14%) | 96 (20%) | 0.004 | Ref | |
| $30,000– $35,999 | 45 (25%) | 490 (26%) | 944 (25%) | 127 (26%) | 0.98 (0.58, 1.64) | 0.9374 | |
| $36,000– $45,999 | 42 (24%) | 499 (26%) | 1119 (30%) | 133 (28%) | 0.89 (0.50, 1.58) | 0.6856 | |
| $46,000+ | 61 (34%) | 569 (30%) | 1133 (30%) | 127 (26%) | 1.64 (0.87, 3.09) | 0.1269 | |
| Charlson–Deyo Score | |||||||
| 0 | 136 (77%) | 1435 (76%) | 2828 (76%) | 368 (76%) | 0.84 | Ref | |
| 1 | 37 (21%) | 369 (20%) | 753 (20%) | 92 (19%) | 1.05 (0.72, 1.53) | 0.80 | |
| 2 | 4 (2%) | 80 (4%) | 149 (4%) | 23 (5%) | 0.53 (0.19, 1.46) | 0.21 | |
| Histology | |||||||
| SCC | 45 (25%) | 307 (16%) | 638 (17%) | 108 (22%) | <0.001 | Ref | 0.02 |
| Adeno | 132 (75%) | 1577 (84%) | 3092 (83%) | 375 (78%) | 0.62 (0.42, 0.91) | ||
| Stage | |||||||
| Stage I | 41 (23%) | 337 (18%) | 665 (18%) | 92 (19%) | 0.07 | Ref | |
| Stage II | 65 (37%) | 879 (47%) | 1629 (44%) | 213 (44%) | 0.68 (0.45, 1.03) | 0.06 | |
| Stage III | 71 (40%) | 668 (35%) | 1436 (38%) | 178 (37%) | 0.89 (0.60, 1.33) | 0.57 | |
| Grade | |||||||
| Well differentiated | 7 (4%) | 116 (6%) | 188 (5%) | 27 (6%) | 0.01 | Ref | |
| Moderately differentiated | 74 (42%) | 819 (43%) | 1595 (43%) | 210 (43%) | 1.20 (0.52, 2.76) | 0.66 | |
| Poorly differentiated | 90 (51%) | 928 (49%) | 1857 (50%) | 243 (50%) | 1.25 (0.57, 2.78) | 0.57 | |
| Undifferentiated | 6 (3%) | 21 (1%) | 90 (2%) | 3 (1%) | 2.14 (0.68, 6.69) | 0.19 | |
| Great circle distance | |||||||
| <12.5 | 82 (46%) | 810 (43%) | 1739 (47%) | 201 (42%) | 0.003 | Ref | |
| 12.5–50 | 45 (25%) | 685 (36%) | 1251 (34%) | 169 (35%) | 0.61 (0.40, 0.91) | 0.02 | |
| > = 50 | 50 (28%) | 389 (21%) | 740 (20%) | 113 (23%) | 0.95 (0.59, 1.53) | 0.8267 | |
| Facility type | |||||||
| Community | 6 (3%) | 125 (7%) | 263 (7%) | 37 (8%) | <0.001 | Ref | |
| Comprehensive | 64 (36%) | 839 (45%) | 1388 (37%) | 185 (38%) | 2.09 (0.89, 4.92) | 0.08 | |
| Academic | 100 (56%) | 758 (40%) | 1851 (50%) | 224 (46%) | 2.72 (1.15, 6.41) | 0.02 | |
| Other | 7 (4%) | 162 (9%) | 228 (6%) | 37 (8%) | 1.33 (0.44, 4.02) | 0.61 | |
| Facility location | |||||||
| Northeast | 41 (23%) | 316 (17%) | 939 (25%) | 104 (22%) | <0.001 | Ref | |
| Central | 64 (36%) | 694 (37%) | 1291 (35%) | 123 (25%) | 1.05 (0.69, 1.60) | 0.82 | |
| South | 48 (27%) | 347 (34%) | 1063 (28%) | 186 (39%) | 0.91 (0.58, 1.42) | 0.67 | |
| West | 24 (14%) | 227 (12%) | 437 (12%) | 70 (14%) | 1.10 (0.64, 1.89) | 0.73 | |
| Urban/Rural | |||||||
| Rural | 3 (2%) | 48 (3%) | 121 (3%) | 13 (3%) | 0.26 | Ref | |
| Urban | 49 (28%) | 393 (21%) | 781 (21%) | 105 (22%) | 2.19 (0.66, 7.28) | 0.19 | |
| Metro | 125 (71%) | 1443 (77%) | 2828 (76%) | 365 (76%) | 1.24 (0.37, 4.21) | 0.72 | |
| Year of diagnosis | |||||||
| 2004 | 18 (10%) | 213 (11%) | 179 (5%) | 44 (9%) | <0.001 | Ref | |
| 2005 | 21 (12%) | 234 (12%) | 223 (6%) | 45 (9%) | 0.98 (0.51, 1.89) | 0.96 | |
| 2006 | 5 (3%) | 218 (12%) | 330 (9%) | 39 (8%) | 0.20 (0.08, 0.56) | 0.002 | |
| 2007 | 18 (10%) | 226 (12%) | 325 (9%) | 41 (8%) | 0.73 (0.37, 1.43) | 0.35 | |
| 2008 | 8 (5%) | 231 (12%) | 385 (10%) | 48 (10%) | 0.30 (0.13, 0.69) | 0.005 | |
| 2009 | 16 (9%) | 194 (10%) | 463 (12%) | 50 (10%) | 0.54 (0.27, 1.07) | 0.08 | |
| 2010 | 14 (8%) | 177 (9%) | 507 (14%) | 56 (12%) | 0.41 (0.20, 0.84) | 0.01 | |
| 2011 | 26 (15%) | 213 (11%) | 605 (16%) | 72 (15%) | 0.68 (0.36, 1.26) | 0.21 | |
| 2012 | 51 (29%) | 178 (9%) | 713 (19%) | 88 (18%) | 1.19 (0.68, 2.09) | 0.54 | |
Predictors for the use of low-dose radiation were explored by comparing use of 40–41.4 Gy vs. all other dose levels in a multivariable model. Hispanic race (OR 2.67 [95% CI 1.22–5.81]); P = 0.01) and treatment at an academic center (OR 2.72 [95% CI 1.15–6.41]; P = 0.02) predicted for higher use of low- dose neoadjuvant CRT. Conversely, AC histology and distance 12.5–50 miles from a treatment facility (but not greater than 50 miles) were less likely to receive lower dose radiation. Lower dose radiation represented a relatively small percentage of the overall cohort but increased from 3.9% in 2004 to 7.2% in 2013. There was a 12.6%, nonsignificant, annual percent change (APC) decrease in low-dose radiation from 2004 to 2010. There was a borderline significant 63.6% increase in the use of lower dose neoadjuvant chemoradiation from 2010 to 2013 (P = 0.07; 95% CI −5.73, 183.79).
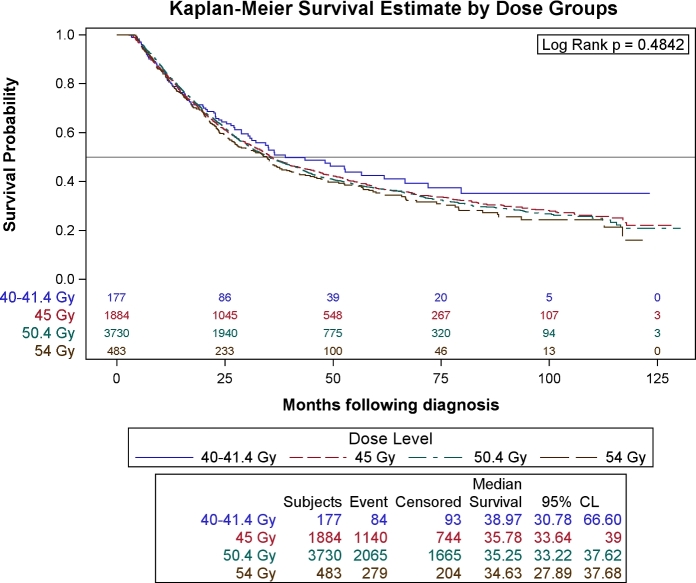
There was no difference in overall survival of patients when stratified according radiation dose level (P = 0.48), (Fig. 3). Pairwise multivariable hazard ratios were generated for low-dose radiation vs. higher dose radiation and no significant differences were found. Univariable and multivariable analysis was performed to look for other predictors for OS in this cohort (Table 2). Private or government insurance (vs. uninsured), higher educational level, higher median income, treatment at an academic center were all associated with improved OS. Age, male gender, higher Charlson–Deyo comorbidity score, higher stage, higher tumor grade, and facility location in the South were all associated significantly worse OS.
Fig. 3.
Kaplan–Meier overall survival estimates of the entire cohort stratified for neoadjuvant radiation dose level.
Table 2.
Baseline patient, facility, and disease characteristics affecting overall survival (N = 6,274)
| Univariable hazard | P-value testing | Multivariable† | P-value testing | |
|---|---|---|---|---|
| ratio [95% CI] | HR = 1 | hazard ratio [95% CI] | HR = 1 | |
| Age | 1.02 [1.01–1.02] | <0.001 | 1.01 [1.01, 1.02] | <0.001 |
| Gender | ||||
| Female | Ref | Ref | ||
| Male | 1.12 [1.02, 1.23] | 0.02 | 1.10 [1.00, 1.22] | 0.05 |
| Hispanic | ||||
| No | Ref | Ref | ||
| Yes | 0.89 [0.68, 1.15] | 0.35 | 0.88 [0.68, 1.15] | 0.36 |
| Race | ||||
| White | Ref | Ref | ||
| Black | 1.05[0.90, 1.23] | 0.51 | 1.06 [0.90, 1.26] | 0.48 |
| Asian | 1.17 [0.86, 1.58] | 0.32 | 1.26 [0.92, 1.72] | 0.14 |
| Other | 0.81[0.45, 1.43] | 0.46 | 0.94 [0.53, 1.68] | 0.83 |
| Insurance | ||||
| Uninsured | Ref | Ref | ||
| Private | 0.61 [0.49, 0.75] | <0.001 | 0.62 [0.50, 0.77] | <0.001 |
| Government | 0.80 [0.64, 0.99] | 0.04 | 0.73 [0.58, 0.91] | 0.005 |
| % without HS education | ||||
| >21% | Ref | Ref | ||
| 13–20.9% | 0.87 [0.78, 0.97] | 0.01 | 0.86 [0.77, 0.97] | 0.02 |
| 7–12.9% | 0.81 [0.73, 0.90] | <0.001 | 0.83 [0.73, 0.94] | 0.004 |
| <7% | 0.78 [0.70, 0.87] | <0.001 | 0.82 [0.70, 0.95] | 0.008 |
| Median Income | ||||
| <$30000 | Ref | Ref | ||
| $30000–$35999 | 0.92 [0.83, 1.03] | 0.14 | 1.00 [0.90, 1.13] | 0.94 |
| $36000–$45999 | 0.90 [0.81, 1.00] | 0.05 | 1.02 [0.90, 1.15] | 0.75 |
| >$46000 | 0.84 [0.76, 0.93] | <0.001 | 0.99 [0.86, 1.13] | 0.83 |
| Deyo/Charlson Comorbidity Score | ||||
| 0 | Ref | Ref | ||
| 1 | 1.20 [1.11, 1.30] | <0.001 | 1.18 [1.08, 1.27] | <0.001 |
| ≥2 | 1.24 [1.05, 1.45] | 0.010 | 1.21 [1.03, 1.43] | 0.02 |
| Histology | ||||
| SCC | Ref | 0.20 | Ref | 0.15 |
| Adeno | 1.06 [0.97, 1.16] | 0.93 [0.84, 1.03] | ||
| Stage | ||||
| 1 | Ref | Ref | ||
| 2 | 1.30 [1.18, 1.44] | <0.001 | 1.30 [1.18, 1.45] | <0.001 |
| 3 | 1.66 [1.50, 1.83] | <0.001 | 1.71 [1.54, 1.89] | <0.001 |
| Grade | ||||
| Well differentiated | Ref | Ref | ||
| Moderately differentiated | 1.27 [1.07, 1.50] | 0.007 | 1.21 [1.02, 1.44] | 0.03 |
| Poorly differentiated | 1.49 [1.26, 1.77] | <0.001 | 1.43 [1.20, 1.70] | <0.001 |
| Undifferentiated | 1.25 [0.91, 1.72] | 0.15 | 1.24 [0.90, 1.70] | 0.18 |
| Distance to facility (miles) | ||||
| <12.5 | Ref | Ref | ||
| 12.5–50 | 1.04 [0.97, 1.12] | 0.29 | 1.02 [0.94, 1.10] | 0.67 |
| ≥50 | 0.96 [0.88, 1.05] | 0.36 | 0.93 [0.83, 1.04] | 0.19 |
| Facility type | ||||
| Community Cancer Program | Ref | Ref | ||
| Comprehensive Comm Cancer | 0.93 [0.80, 1.07] | 0.29 | 0.94 [0.82, 1.08] | 0.38 |
| Academic/Research Program | 0.81 [0.70, 0.93] | 0.004 | 0.83 [0.73, 0.96] | 0.01 |
| Other | 0.89 [0.72, 1.09] | 0.2478 | 0.92 [0.77, 1.10] | 0.34 |
| Facility location | ||||
| Central | Ref | Ref | ||
| Northeast | 1.04 [0.95, 1.14] | 0.35 | 1.05 [0.96, 1.16] | 0.27 |
| South | 1.18 [1.08, 1.29] | <0.001 | 1.14 [1.04, 1.25] | 0.008 |
| West | 0.99 [0.88, 1.12] | 0.92 | 0.94 [0.83, 1.07] | 0.35 |
| Facility setting | ||||
| Urban | Ref | Ref | ||
| Rural | 0.99 [0.80, 1.21] | 0.88 | 1.01 [0.82, 1.25] | 0.90 |
| Metro | 0.89 [0.73, 1.08] | 0.23 | 0.93 [0.75, 1.15] | 0.49 |
| Year of diagnosis | ||||
| 2004 | Ref | Ref | ||
| 2005 | 1.01 [0.87, 1.17] | 0.89 | 1.05 [0.91, 1.22] | 0.52 |
| 2006 | 0.89 [0.76, 1.03] | 0.10 | 0.86 [0.74, 1.00] | 0.05 |
| 2007 | 0.95 [0.82, 1.11] | 0.53 | 0.91 [0.79, 1.06] | 0.23 |
| 2008 | 0.93 [0.80, 1.07] | 0.29 | 0.92 [0.79, 1.06] | 0.23 |
| 2009 | 0.94 [0.81, 1.09] | 0.39 | 0.93 [0.80, 1.07] | 0.30 |
| 2010 | 0.98 [0.84, 1.13] | 0.75 | 0.97 [0.83, 1.12] | 0.65 |
| 2011 | 0.95 [0.82, 1.10] | 0.52 | 0.95 [0.82, 1.10] | 0.53 |
| 2012 | 0.90 [0.77, 1.05] | 0.18 | 0.90 [0.77, 1.05] | 0.18 |
P-value < 0.05 bolded.
Multivariable HR was estimated from the full Cox proportional hazards model including all covariates presented in this table.
CI, confidence interval; HR, hazard ratio.
DISCUSSION
With a well-established benefit in OS using neoadjuvant CRT for the treatment of esophageal cancer over surgery alone, clinical questions are now focusing more on optimization of therapeutic ratio and appropriate patient selection. For instance, Alliance for Clinical Trials in Oncology recently completed a trial using positron emission tomography (PET) to tailor induction chemotherapy prior to neoadjuvant chemoradiation and surgery. Other trials are exploring novel chemotherapeutic such as Cetuximab (Radiation Therapy Oncology Group 0436) or Trastuzumab in addition to standard chemotherapy regimens. Some of the important questions raised by the CROSS trial, namely the relative efficacy of 5FU/cisplatin vs. carboplatin/paclitaxel and 41.4 Gy vs. 50.4 Gy, will likely never be addressed in the randomized setting. This would involve very large and costly noninferiority designs, which might not be an optimal allocation of increasingly limited resources. In addition, the relative ease of delivering Carboplatin/Paclitaxel has already won over many medical oncologists, and it might be difficult to randomize patients back to 5FU/Cisplatin.
This study, using a large national database with excellent radiation records with regard to dose, represents perhaps the best way to address the radiation dose question short of a randomized trial. A similar analysis recently used the NCDB to look at dose escalation for definitive radiation for esophageal cancer.11 Smaller, single institution analyses have been done to address the chemotherapy questions, but they lack the power needed to make meaningful conclusions.12 With the statistical power of over 6000 patients, we did not find any difference in OS based on neoadjuvant dose levels regardless of histology after controlling for available confounding variables. As expected, variables such as stage, AC, and advanced age were associated with worse OS. Interestingly, patients in academic centers appeared to fare better with regard to OS, perhaps suggesting better outcomes with higher volume centers, which has been shown in other studies for esophageal cancer.13,14
There was a trend to increasing use of low-dose radiation over the course of this study, and this was especially prominent around year 2013. Because the numbers are small, there is limited utility to performing APC analysis, and the data should be interpreted with regard to absolute numbers as well. This increase possibly correlates to presentation and publication of the CROSS trial in 2011–2012, and we hypothesize that the trend will continue to increase. As more data are released by the NCDB, and there will likely be a more balanced comparison by number. This study should therefore be updated in 3–5 years to verify its findings. Not surprisingly, academic centers appear to be embracing this clinical change ahead of other institutions.
There are several limitations of this study we want to address. Despite large patient numbers, this study is affected the same deficiencies as any retrospective analysis with respect to bias. We attempted to refine our study population to patients with known staging, treatment (including radiation dose), and follow-up to address general criticisms of large databases. In addition, there is no recorded information on local control, disease-specific survival, or treatment toxicity, which are important determinants of therapeutic ratio. Pathological complete response rate is also of clinical interest, but this variable is incompletely recorded in the database such that it is not easily evaluated. The database does not include specifics of radiation field design or chemotherapy regimen, cycles, or doses. It is possible that unknown imbalances in chemotherapy regimens in our groups could affect the outcome. There is a relatively low number of low-dose radiation used, discussed above as likely related to recent publication of CROSS trial.
As more data are released, it is likely that the number of low-dose neoadjuvant CRT cases will increase, possibly affecting conclusions.
In summary, we found no OS benefit to using doses higher than 41.4 Gy in neoadjuvant CRT for surgically resected esophageal cancer. With prospective evaluation unlikely, this represents the largest study of the impact of radiation dose in neoadjuvant CRT for esophageal cancer. These data might assist a radiation oncologist in choosing neoadjuvant CRT dosing in the future.
Notes
Conflicts of Interest and Disclosures: None declare.
References
- 1. Walsh T N, Noonan N, Hollywood D, Kelly A, Keeling N, Hennessy T P. A comparison of multimodal therapy and surgery for esophageal adenocarcinoma. N Engl J Med 1996; 335: 462–7. [DOI] [PubMed] [Google Scholar]
- 2. Bosset J F, Gignoux M, Triboulet J P et al. Chemoradiotherapy followed by surgery compared with surgery alone in squamous-cell cancer of the esophagus. N Engl J Med 1997; 337: 161–7. [DOI] [PubMed] [Google Scholar]
- 3. Tepper J, Krasna M J, Niedzwiecki D et al. Phase III trial of trimodality therapy with cisplatin, fluorouracil, radiotherapy, and surgery compared with surgery alone for esophageal cancer: CALGB 9781. J Clin Oncol 2008; 26: 1086–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. van Hagen P, Hulshof M C, van Lanschot J J et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med 2012; 366: 2074–84. [DOI] [PubMed] [Google Scholar]
- 5. Oppedijk V, van der Gaast A, van Lanschot J J et al. Patterns of recurrence after surgery alone versus preoperative chemoradiotherapy and surgery in the CROSS trials. J Clin Oncol 2014; 32: 385–91. [DOI] [PubMed] [Google Scholar]
- 6. Lin S H, Wang L, Myles B et al. Propensity score-based comparison of long-term outcomes with 3-dimensional conformal radiotherapy vs intensity-modulated radiotherapy for esophageal cancer. Int J Radiat Oncol Biol Phys 2012; 84: 1078–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Lin S H, Zhang N, Godby J et al. Radiation modality use and cardiopulmonary mortality risk in elderly patients with esophageal cancer. Cancer 2016; 122: 917–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Minsky B D, Neuberg D, Kelsen D P, Pisansky T M, Ginsberg R, Benson A 3rd. Neoadjuvant chemotherapy plus concurrent chemotherapy and high-dose radiation for squamous cell carcinoma of the esophagus: a preliminary analysis of the phase II intergroup trial 0122. J Clin Oncol 1996; 14: 149–55. [DOI] [PubMed] [Google Scholar]
- 9. White I R, Royston P, Wood A M. Multiple imputation using chained equations: issues and guidance for practice. Stat Med 2011; 30: 377–99. [DOI] [PubMed] [Google Scholar]
- 10. Yuan Y. Multiple imputation using SAS software. J Stat Softw 2011; 45: 1–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Brower J V, Chen S, Bassetti M F et al. Radiation dose escalation in esophageal cancer revisited: a contemporary analysis of the national cancer data base, 2004 to 2012. Int J Radiat Oncol Biol Phys 2016; 96: 985–93. [DOI] [PubMed] [Google Scholar]
- 12. Nabavizadeh N, Shukla R, Elliott D A et al. Preoperative carboplatin and paclitaxel-based chemoradiotherapy for esophageal carcinoma: results of a modified CROSS regimen utilizing radiation doses greater than 41.4 Gy. Dis Esophagus 2016; 29: 614–20. [DOI] [PubMed] [Google Scholar]
- 13. Speicher P J, Englum B R, Ganapathi A M et al. Traveling to a high-volume center is associated with improved survival for patients with esophageal cancer. Ann Surg 2017; 265: 743–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Fuchs H F, Harnsberger C R, Broderick R C et al. Mortality after esophagectomy is heavily impacted by center volume: retrospective analysis of the Nationwide Inpatient Sample. Surg Endosc 2017; 31: 2491–97. [DOI] [PubMed] [Google Scholar]