Abstract
Osteoporosis is a chronic disease affecting millions of people worldwide caused by an imbalance between bone-forming osteoblasts and bone-resorbing osteoclasts. Despite recent developments in pharmacological agents to prevent osteoporotic-related fractures, much less attention has been placed on the repair of bone defects following fracture. Critical to this process is the recruitment of mesenchymal stem cells (MSCs) to defect sites by growth factors. One method which has been effective for the sustained release of growth factors is that of gene therapy. The aim of the present study was to investigate newly developed mesoporous bioglass/silk fibrin scaffolds containing adPDGF-b and adBMP-7 into osteoporotic critical-sized femur defects in ovariectomised rats following treatment periods of 2 and 4 weeks. In vivo osteogenetic efficiency evaluated by μ-CT analysis, hematoxylin and eosin staining, and immunohistochemical (type I collagen, osteopontin and BSP) revealed significantly new bone formation in defects containing adenovirus for both PDGF-b and BMP-7 when compared to scaffolds alone and scaffolds containing BMP-7. TRAP-positive staining also demonstrated the ability for these scaffolds to be degraded over time and initiate bone turnover/remodeling. Although the use of gene therapy for clinical applications is still in its infancy, results from the present study demonstrate their potent ability to recruit mesenchymal progenitor cells through sustained release of PDGF-b and BMP-7 which may be beneficial for patients suffering from osteoporotic-related fractures.
Keywords: Gene therapy, Osteoporosis, Scaffold, PDGF, BMP, Adenovirus
1. Introduction
Osteoporosis is a worldwide chronic disease characterized by low bone mass, poor bone strength and microarchitectural deterioration of bone tissue leading to increased bone fragility and fracture risk [1]. It is an age-related disease caused by the imbalance between bone-forming osteoblasts and bone-resorbing osteoclasts commonly resulting from postmenopausal estrogen deficiency [2,3]. An estimated 200 million people are affected worldwide, with 80% being women [4]. Osteoporotic fractures are a major cause of morbidity in older women where 50% of 65-year-old post-menopausal white or Asian women will experience at least one osteoporotic fracture [5]. A recent systematic review found that the incidence of death for patients following hip fracture is at least doubled for an age-matched control without such fractures, and is continuously rising [6]. While the average life expectancy worldwide continues to increase, the annual cost of osteoporotic fractures and related issues is projected to account for more than $20 billion by 2015 in the United States alone [7].
For decades, studies have demonstrated that bone healing in postmenopausal osteoporotic women are delayed, mainly due to estrogen deficiencies which leads to a rise in osteoclast number [3,8–10]. At present, the two major pharmacological approaches for the treatment of osteoporosis are anabolic agents such as parathyroid hormone (PTH) or its analog act by stimulating the bone formation process, and anti-resorptive agents including bisphosphonates, calcitonin, raloxifene, and estrogen which act by inhibiting osteoclastic bone resorption [11]. Although much emphasis has been placed on the treatment of osteoporosis by the above-mentioned agents, less emphasis has been placed on the repair of osseous defects following fracture.
Critical to the healing process of bone defects is the homing of stem cells to sites of injury [12]. These processes are mediated through local concentration gradients of cytokines and growth factors. Two growth factors approved by the FDA that have received enormous attention for tissue regeneration are platelet derived growth factor (PDGF) and bone morphogenetic protein (BMP) [13–17]. PDGF was initially discovered as a mitogen for fibroblasts contained in human serum and localized in the alpha granules of platelets [18]. It is a potent cytokine capable of rapid mesenchymal recruitment to sites of injury with wound healing capabilities [19]. BMPs were developed for the regeneration of osseous defects and have since been shown to affect cell recruitment, proliferation and differentiation of mesenchymal cells to the osteoblast lineage [20].
Despite the clinical advantages of recombinant proteins, concerns have been raised regarding their transient action and high degradation rate in vivo. More recently the development of an alternative approach for delivery of growth factors is that of gene therapy which has been an effective alternative for sustained bioactivity and controlled release of local growth factors. Adeno-virus vectors (Ad) exhibit a high in vivo transduction efficiency with a relatively short expression period [21] making them well suitable for tissue regeneration without eliciting long-term health concerns [22,23].
Equally as important for bone tissue engineering is the fabrication of an osteoconductive scaffold. Various tissue engineering strategies have been employed to achieve this goal [24–28]. Recently we have fabricated an MBG/silk scaffold with a well-ordered nanochannel structure which demonstrates better physiochemical properties with superior mechanical strength, in vitro apatite mineralization, Si ion release and pH stability [29]. This study sought to investigate the combination of this newly developed scaffold with the advantages of gene therapy delivery system for the treatment of osteoporosis. Since osteoporosis-related fractures are commonly occurring in long bone sites under loading environment, the aim of the present study was to test these new scaffolds into osteoporotic critical-sized femur defects in ovariectomised rats and to study the involved bone remodeling process following treatment periods of 2 and 4 weeks.
2. Materials and methods
2.1. Fabrication of porous mesoporous-glass/silk scaffolds containing adenoviruses
Mesoporous-glass/silk scaffolds containing adenovirus were fabricated according to the method as previously described [29]. Porous MBG/silk scaffolds with 10 wt.% MBG were fabricated using a freeze-drying method: 0.1 g of MBG powders were added to 20 ml of 5% (w/v) silk water solutions under stirring for 2 h and ultrasonic dispersing for 10 min to form a uniform mixture, 2 ml of which was quickly transferred into the wells of a 24-well cell culture plate. The plate was then placed in a freezer at −20 °C overnight to solidify the mixture and induce solid–liquid phase separation. The solidified mixture was freeze-dried in a freeze-drying machine (OHRIST BETA 1–15, Germany) for 48 h to obtain porous MBG/silk scaffolds. The scaffolds were soaked in ethanol (99%) for 10 min and immersed in 75% ethanol solution for 12 h for sterilization, and then lyophilized again to get neutral, asepsis scaffolds. Then 2 ml adenovirus solution was dropped onto 1 mg dried MBG/silk scaffolds, and kept at 4 °C overnight for full incorporation of MBG/silk scaffolds with viruses. These complexes were then frozen by immersion into −80 °C for 2 h and lyophilized for next use. The pore morphology and surface microstructure of the scaffolds were characterized by scanning electron microscopy (SEM; Joel JSM 6510).
2.2. Animals and surgical protocols
Animal handling and surgical procedures were conducted according to the guidelines for animal care and use committee of Wuhan University, People’s Republic of China, and approved by the Ethics Committee at the School of Dentistry, prior to the start of this experiment. All animals were kept at 20–25 °C under a 12-h light/dark cycle and allowed food and water ad libitum. All operations were carried out under sterile conditions with a gentle surgical technique. The surgeon was blinded to the treatment. A single intramuscular dose of penicillin 40,000 IU/ml was then administered postoperatively. No significant perioperation or postoperation fractures were produced.
2.2.1. Ovariectoporosis model
5-month-old mature female Wistar rats were used for this experiment. In order to induce an osteoporosis model, the rats were subjected to bilateral ovariectomy (OVX) or sham operation (Sham) at 3 months of age. After anesthetization by intraperitoneal injection of sodium pentobarbital (40 mg/kg body weight), a lumbar lateral incision was made around the midpoint between the lower margin of free ribs and iliac crest, where the ovary was located. A suture was placed around the ovarian artery and vein prior to the removal of ovary. To tightly close the incision, the muscles were repositioned in layers and sutured with resorbable suture, and the skin was closed with nylon 4-0 suture.
2.2.2. Femur defect model
After 2-month-induction, femur defect drilling was performed under general anaesthesia by intraperitoneal injection of sodium pentobarbital (40 mg/kg body weight). A linear skin incision of approximately 1 cm in the distal femoral epiphysis was made bilaterally and blunt dissection of the muscles was performed to expose the femoral condyle. Then, a 2-mm-diameter anteroposterior bicortical channel was created perpendicular to the shaft axis to destroy cancellous trabecular bone, by using a trephine bur at a slow speed irrigated under saline solution to avoid thermal necrosis. The grilled holes were rinsed by injection with saline solution in order to remove bone fragments from the cavity. Implant scaffolds were then gently placed to fill the grilled defects according to group allocation. Subsequently, the incision was closed as mentioned above.
Thereafter, the rats were divided into groups including Sham (n = 3), OVX (n = 3), OVX (blank control; n = 6), OVX + drilled defect (drill control; n = 6), OVX + scaffold alone (n = 6), OVX + adBMP-7 scaffold (n = 6) and OVX + adPDGF-b + adBMP-7 scaffold (n = 6) respectively. The rats from Sham and OVX were considered as confirmation of osteoporosis model, and were sacrificed after two-month-induction by cervical dislocation. At each time point, 14 and 28 days after femur surgery, rats in the remaining three groups were sacrificed, accordingly. All femurs were removed and assigned to micro-computerized tomography (μ-CT) analysis and histological studies.
2.3. μ-CT analysis
The samples were fixed in 4% paraformaldehyde for 24 h at room temperature. A μ-CT imaging system (μ-CT50, Scanco Medical, Basersdorf, Switzerland) was used: i) to identify the establishment of osteoporosis model with microarchitectural changes and ii) to evaluate new bone formation within the defect region. All samples were placed in a custom-made holder to ensure that the long axis of the drilled channel was oriented perpendicular to the axis of X-ray beam. Scanning was performed at 55 kV and 114 μA with a thickness of 0.048 mm per slice in medium-resolution mode, 1024 reconstruction matrix, and 200 ms integration time. These images and parameters were compared between two groups to confirm the osteoporosis model induced in rats. A Gaussian filter (sigma = 0.8 and support = 1) was used to remove noise. The mineralized bone tissue was differentially segmented to exclude the non-mineralized tissue with a fixed threshold (value = 190).
For conformation of established osteoporosis model, a series of slices starting at a distance of 1 mm proximal from the end of the growth plate with a length of 2 mm were chosen for the evaluation. For evaluation of bone regeneration within the defect, the central 1.5-mm-diameter region of the 2-mm-diameter defect was defined by drawing circular contour as area of measurement per slice, thus to obtain a consistent volume of interest (VOI) and to avoid including the native bone margins. After 3D reconstruction, the bone volume faction (BV/TV), trabecular number (Tb.N), trabecular thickness (Tb.Th) and trabecular separation (Tb.Sp) were automatically determined for identification of osteoporosis model while bone volume fraction (BV/TV) in defect regions were used to evaluate new bone formation, using a protocol provided by the manufacturer of the micro-CT scanner. The porosity (P) of bone was calculated as [30]:
All digitalized data and 3D images were generated by the built-in software of the μ-CT.
2.4. Histological preparation and immunohistochemical studies
After μ-CT imaging, femoral condyles were decalcified in 10% ethylene diaminetetraacetic acid (EDTA) for 2 weeks, changed twice per week, and then dehydrated in a series of graded concentration of ethanol from 70% to 100%. To get a distinct view of the defect, the orientation and alignment of femurs were carefully considered during paraffin embedding. Longitudinal serial sections, 4 mm thick, were cut and mounted on polylysine-coated microscope slides. For general histological studies, hematoxylin and eosin (H&E) staining, Safranin O staining (Sigma #S2255; Sigma–Aldrich, St. Louis, USA.) and tartrate-resistant acid phosphatase (TRAP) staining (Sigma #387A; Sigma–Aldrich, St. Louis, USA.) were performed according to manufacturer’s protocol.
For immunohistochemical assessment, the expression of type I collagen (COL I) and osteopontin (OPN) were detected according to the following procedure. Deparaffinised sections were washed with PBS, incubated with 0.3% hydrogen perioxide for 20 min to block endogenous peroxidase activity, followed by incubation with 5% bovine serum albumin (BSA). Then, sections were incubated with optimal concentrations of primary antibody for COL I (1:100; Sigma–Aldrich, St. Louis, USA.), OPN (1:200; Biomedical Technologies, Stoughton, MA) overnight at 4°C. After three times washing with PBS, sections were incubated with biotinylated secondary antibody (Zhongshan Biotechnology Co., Ltd, China) for 20 min before incubation with horseradish peroxidase-conjugated avidin–biotin complex (ABC) (Zhongshan Biotechnology Co., Ltd, China) for another 20 min. Immunostaining was visualized after the addition of a buffered 3,3-diaminobenzidiinetetrahydrochloride (DAB) (Zhongshan Biotechnology Co., Ltd, China) substrate. Sections were then counterstained with Harris’s hematoxylin for 40 s each, in between 3 min rinses with running water. Subsequently, sections were dehydrated with ascending concentrations of ethanol solutions, cleared with xylene, and mounted with coverslips. Specimens were examined under microscopic light by using Olympus DP71 microscope (Olympus Co., Japan). To validate the results, each experiment was repeated at least three times.
Bone regeneration of these histological sections was scored on a semi-quantitative scale by an individual blinded observer, using a modified scoring method similar to that described previously [31] (Table 1). By using ImageJ 1.44 software from the National Institute of Health (NIH, Bethesda, Maryland, USA), the fraction of scaffold remnants calculated as previously described [32,33]. According to previous report, the number of osteoclasts was counted under a light microscope (Olympus DP71; Olympus Co., Japan) [34]. Cells positively stained for TRAP containing more than three nuclei were defined as osteoclasts [35]. The bone histomorphometry and TRAP-positive multinuclear osteoclast measurements were performed on three consecutive sections of each specimen. From each section, three representative fields (1024 × 1536 pixels) were identified (original magnification ×10) and averaged.
Table 1.
Semiquantitative scale for evaluation of bone regeneration.
| Score | Extent of new bone in defect |
|---|---|
| 0 | No bone formation |
| 1 | Minimal bone formation (only very small portion in the defect) |
| 2 | Low bone formation (less than one-fourth of the defect) |
| 3 | Moderate bone formation (less than one-half and more than one-fourth of the defect) |
| 4 | Abundant bone formation (less than three-quarter and more than one-half of the defect) |
| 5 | Extensive bone formation (more than three-quarter of the defect) |
2.5. Statistical analysis
All statistical analysis was performed by using SPSS 17.0 software (SPSS, Chicago, IL). Data were expressed as mean ± standard deviation (SD) and were analyzed using one-way ANOVA and t-test. For the bone regeneration score, the Kruskal–Wallis H-test was used followed by Mann–Whitney U-tests if statistically significant. A 5% (P < 0.05) level of significance was adopted.
3. Results
3.1. Characterization of the prepared porous scaffolds
The MBG/silk and MBG/silk + BMP-7 scaffolds were highly porous (Fig. 1). It was noted that the pore size of the MBG/silk scaffolds is larger than that of MBG/silk + BMP-7 scaffolds.
Fig. 1.
SEM micrographs of scaffolds (A), lower magnification of pure MBG/silk scaffolds (B), higher magnification of pure MBG/silk scaffolds (C), lower magnification of pure MBG/silk + BMP-7 scaffolds (D) higher magnification of pure MBG/silk + BMP-7 scaffolds.
3.2. Establishment of rat osteoporotic model
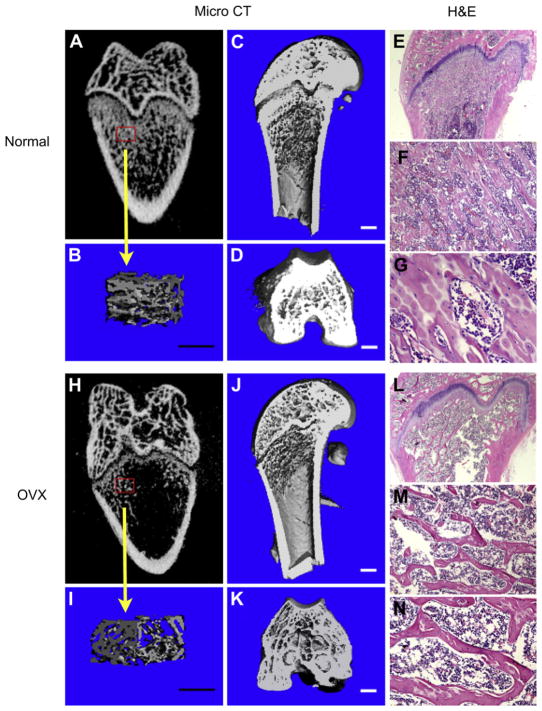
Both 2D representation and 3D μ-CT images of ovariectomized rats showed a striking decrease in the subchondral trabecular bone volume, thickness and density, and significant increase in trabecular separation, discontinued microarchitecture, reduced cortical thickness and enlarged marrow cavities, as compared with sham-operated rats (Fig. 2). After 3D reconstruction (Fig. 3), parameters such as the BV/TV, Tb.N, Tb.Th, Conn.D in the distal femur were significantly lower in OVX rats than in Sham controls, whereas the Tb.Sp was significantly higher in OVX rats (Fig. 5). H&E staining also supported the noticeable changes, such as plate-like trabecular pattern with healthy marrow-like tissue in Sham controls and rodlike deterioration of trabecular patterns with fat-rich bone marrow-like tissue in ovariectomized rats.
Fig. 2.
Establishment of rat osteoporotic model in femur head. 2D representation of normal bone (A) and osteoporotic bone (H), 3D visualization for subchondral trabecular regions of the normal bone (B) in (A) and the osteoporotic bone (I) in (H). 3D μ-CT images of normal bone (C, D) and osteoporotic bone (J, K) from axial section and cross section (bar = 1 mm). Representative H&E staining for the normal (E, F, G) and osteoporotic (L, M, N) femur in magnification of 2×, 10× and 40×, respectively.
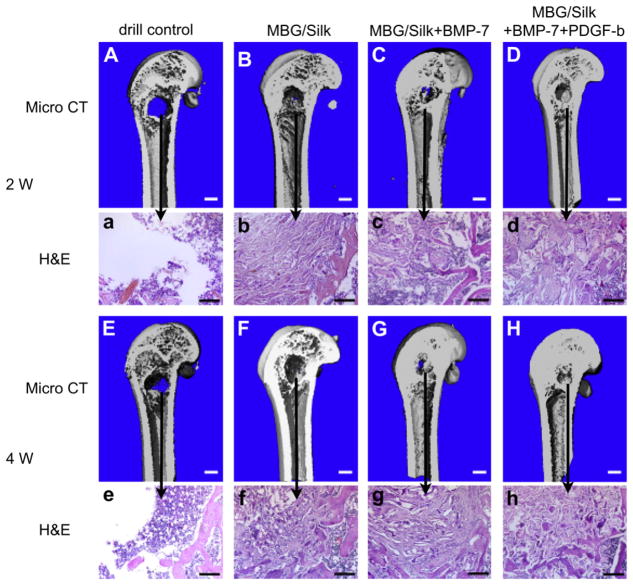
Fig. 3.
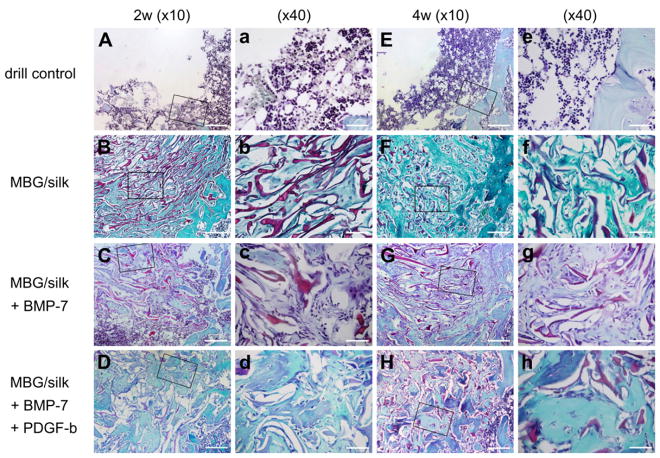
3D μ-CT images of mineralized bone formation in distal femur defects left alone (A, E) and filled with MBG/silk scaffold (B, F), MBG/silk + BMP-7 scaffold (C, G) and MBG/silk + BMP-7 + PDGF-b scaffold (D, H) after 2 and 4 weeks (bar = 1 mm). Representative H&E staining demonstrating new bone matrix deposition within defects left alone (a, e) and implanted with MBG/silk scaffold (b, f), MBG/silk + BMP-7 scaffold (c, g) and MBG/silk + BMP-7 + PDGF-b scaffold (d, h), respectively (bar = 200 μm).
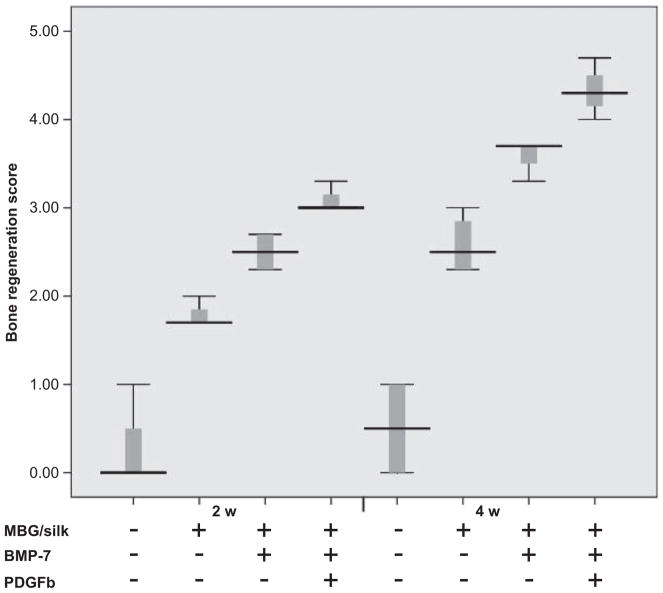
Fig. 5.
Semiquantitative scores of bone regeneration within femur defects are present as box plots. The box represents the 25 and 75 percentiles and the horizontal line in the box represents median value. Lines outside the box represent the spread of values.
3.3. μ-CT analysis of osteogenesis
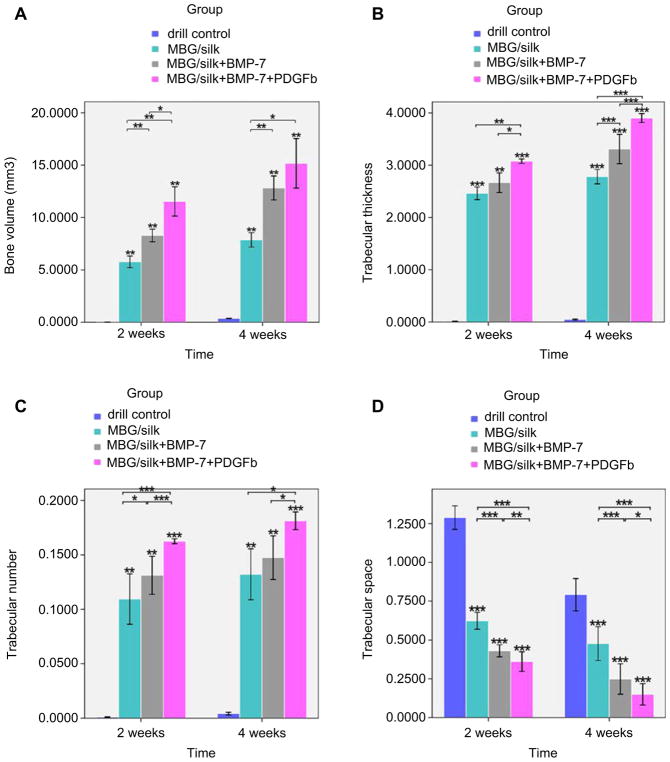
After 3D reconstruction, μ-CT images and microarchitectural parameters were used as quantitative analysis of the morphology and mineralization of regenerative new bone formation within defects. The 3D images clearly depicted the differences among four groups at both time points. Over the study duration, the drill control group remained primarily empty, indicating the critical size (non-self-healing ability) of osteoporotic femur defects (Fig. 3). At 2 weeks, small amounts of mineralized tissue predominantly located at the cortical margins and the defect periphery in the MBG/silk scaffold group were observed, and the defect area gradually decreased as the border shrinked centripetally 4 weeks post-surgery (Fig. 3). Some mineralized tissue bridging can be seen in MBG/silk + BMP-7 scaffold group at 2 weeks post-surgery, and the newly formed bone expanded especially from the center of the defect region (Fig. 3). As expected, significantly more bone volume can be observed in MBG/silk scaffold in combination with BMP-7 + PDGF-b compared with those in MBG/silk alone or MBG/silk + BMP-7 scaffold following a 4-week healing period. Noticeably, defects filled with MBG/silk + BMP-7 + PDGF-b scaffolds were almost fully healed with new plate-like-pattern bone appearing both peripherally and centrally by 4 weeks (Fig. 3). In correlation with the image observation, 3D microarchitectural parameters of VOI were assessed as quantitative indicators of new bone formation. The control group had the lowest values in BV, Tb.Th, Tb.N and the highest value in Tb.Sp (Fig. 5) among all other groups. Compared with MBG/silk scaffold transplanted alone, a significant increase of BV, Tb.Th, Tb.N and constant decrease of Tb.Sp were revealed for the incorporation of BMP-7 and BMP-7 + PDGF-b scaffolds (Fig. 5).
3.4. Histological observation and immunohistochemical assessment
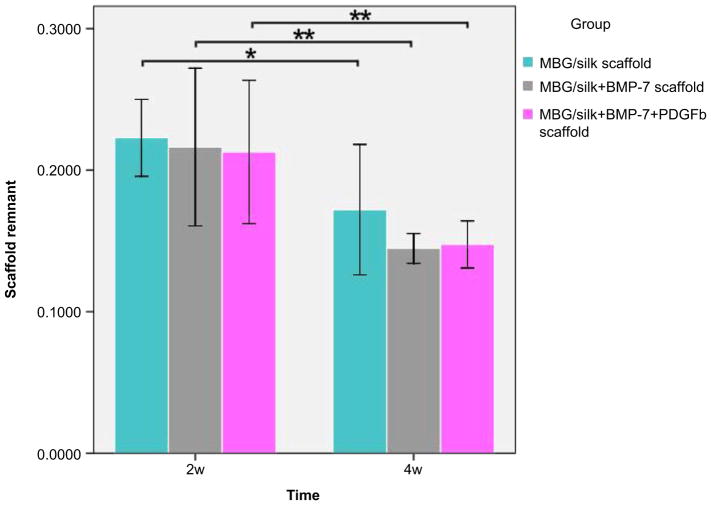
No inflammation or necrosis was present for all animals. Representative histological findings of bone formation further proved the results from μ-CT analysis. Abundant bone formation in BMP-7 + PDGF-b loaded MBG/silk scaffold, moderate bone matrix deposition in BMP-7 loaded MBG/silk scaffold, less organized bone formation in MBG/silk and no obvious bone formation was found in drill controls. During the study period, defects in the drill controls were sparsely populated with fibrous tissue but no appreciable bone formation, along with a clear boundary of native bone (Figs. 3 and 4) and abundant osteoclasts typically lining around the osteoporotic trabecular surface. Instead of self-healing bone formation, fat-rich osteoporotic bone marrow-like tissue gradually filled the control defect over the study duration. Sections from BMP-7 + PDGF-b loaded MBG/silk scaffolds displayed a large scale of well-arranged bone tissue and mineralized matrix deposition at both time points post-surgery along with plenty of blood vessels randomly dispersed within the defect area, leaving a disappearing boundary between native bone and regenerated bone tissue (Fig. 6). As compared with MBG/silk scaffolds alone (Fig. 6), substantial new bone tissue could be observed within defects from BMP-7 loaded MBG/silk scaffolds both at the periphery and in the center of defect sites, as well as an increased number of osteoblastic cells and matrix (Fig. 6). Interestingly, the regenerated woven bone was organized similar to healthy bone, unlike the microstructural deteriorated host trabeculae. The scaffolds failed to maintain their structure integrity and markedly degraded into small fragments, making way for the increasing amount of osteogenic cells and osteoid by 4 weeks, accompanied by traces of cartilage matrix (Figs. 6 and 7).
Fig. 4.
The 3D microarchitectural parameters analyzed by μ-CT. The regenerative potential of bone healing were compared following local implantation of MBG/silk scaffold alone or in combination with BMP-7 or BMP-7 + PDGF-b in femur defects. Data are presented as mean ± SD and analyzed by one-way ANOVA and SNK test. *P < 0.05, **P < 0.01, ***P < 0.001.
Fig. 6.
Representative Saffarin O staining of subchondral bone formation within defects 2 and 4 weeks post-surgery at low (×10; bar = 200 μm) and high (×40; bar = 50 μm) magnification. Fat-rich bone marrow-like tissue gradually filled the defect in the drill control group (A, a, E, e). The degradation of remaining scaffolds (black arrow head) were accompanied with increased bony ingrowth (black star). A large quantity of stranded connective tissue surrounded by osteoblastic cells can be observed in MBG/silk scaffold group (B, b, F, f), while more mature bone matrix was detected in MBG/silk + BMP-7 scaffold group (C, c, G, g). Plate-like trabeculae with osteocytes deposition was observed in MBG/silk + BMP-7 + PDGF-b scaffold group (D, d, H, h). Dotted lines represent defect margin.
Fig. 7.
Quantitative comparison of scaffold remnant fraction in MBG/silk scaffold, MBG/silk + BMP-7 scaffold and MBG/silk + BMP-7 + PDGF-b scaffold groups at two time points (Scaffold remnant = area of silk scaffolds/total area). Data are presented as mean ± SD and analyzed by one-way ANOVA and SNK test. *P < 0.05, **P < 0.01.
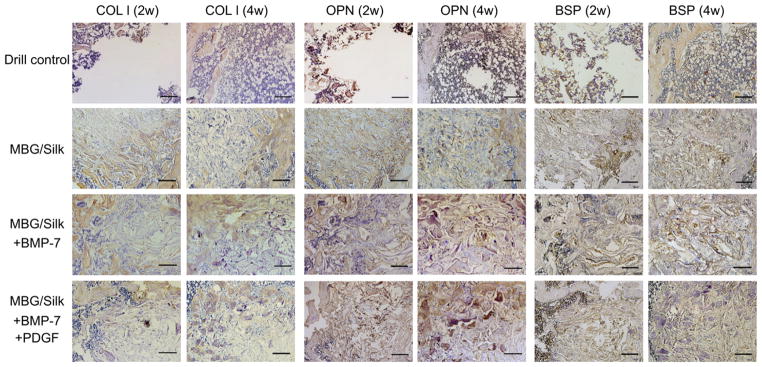
Immunohistochemical markers including COL I, OPN and BSP were qualitatively detected from sections of the defects in all groups, indicating an increase in bone matrix deposition and maturation of active bone-forming sites. Although COL I was positively expressed within all scaffold implanted groups at both 2 and 4 weeks, the staining had significantly greater intensity and area at both time points within the BMP-7 + PDGF-b loaded scaffold (Fig. 8). Prevalently stronger expression of OPN and BSP could be seen peripheral to the newly formed border bone rather than that located centrally in the MBG/silk + BMP-7 + PDGF-b scaffold group (Fig. 8).
Fig. 8.
Representative immunohistological detection of COL I, OPN and BSP in distal femur defects left alone and implanted with MBG/silk scaffold, MBG/silk + BMP-7 scaffold and MBG/silk + BMP-7 + PDGF-b scaffold. MBG/silk + BMP-7 + PDGF-b scaffold stimulates the positively strongest expression with the largest area of COL I, OPN and BSP within defects both at 2 and 4 weeks (bar = 200 μm). Dotted lines represent defect margin.
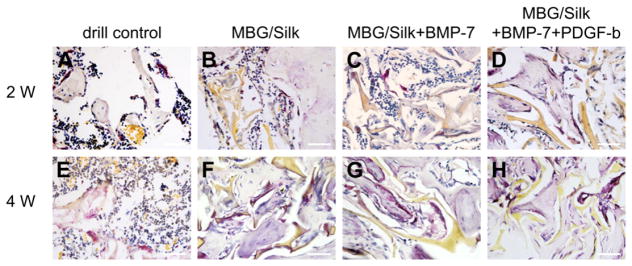
To represent the capability of osteoclastic resorption among the four groups during bone remodeling, TRAP staining was performed at both 2 and 4 weeks. In the drill control group, osteoclasts in shuttle shape were found lining the osteoporotic trabecular surface at 2 weeks, and abundant mononuclear macrophages gradually fused and differentiated into mature osteoclasts (nuclei of TRAP-positive staining cells ≥3) by 4 weeks, demonstrating an activated bone remodeling process (Fig. 9). An increasing amount of mono-nuclear macrophages co-existed with osteogenic cells could be seen in all defect sites at 4 weeks, especially those forming a cluster of osteoclasts cleaved to remnant scaffolds in MBG/silk + BMP-7 and MBG/silk + BMP-7 + PDGF-b implanted groups (Fig. 9). The smallest quantity of TRAP-positive cells with mature morphology were observed scattered within MBG/silk scaffolds (Fig. 9).
Fig. 9.
Representative TRAP staining of osteoclasts (white arrow) in shuttle shape were lining around the osteoporotic trabecular surface in the drill control group at 2 weeks (A), abundant mononuclear macrophages gradually fused and differentiated into mature osteoclasts by 4 weeks (E); more osteoclasts co-existed with osteogenic cells was found especially surrounding remaining scaffolds after 4 weeks (F,G,H) compared with the corresponding scaffold groups at 2-week time point (B,C,D). bar = 50 μm.
4. Discussion
Osteoporosis is one of the most common diseases of the elderly characterized by disequilibrium of bone formation and bone resorption leading to the deterioration of bone microarchitecture, low bone density, and poor bone strength [36]. Currently the majority of therapeutic options mainly focus on treatment via medication by inhibiting bone resorption (anticatabolic compounds) or improving bone formation (PTH). Despite recent developments in pharmacological agents to prevent osteoporotic-related fractures, much less attention has been placed on the study of the healing of bone defects following fracture [5]. With a continuously aging population, it is anticipated that osteoporosis-related issues will pose a significant demand on healthcare necessitating viable treatment options. Previously many investigators have investigated the local release of therapeutic agents in osteoporotic bone [37–43]. While many of these therapies have focused on the ability to prevent osteoclast formation, in this study we focused primarily on the ability to regenerate host tissues with insufficient bone.
To date, a plethora of research has focused on gene therapy for the delivery of growth factors to their local environment [44]. However, most of these studies using adenovirus first transfect cells ex-vivo followed by re-implantation in vivo [45–48]. The in vitro infection prior to implantation negatively influences cell behavior by decreasing the time for total protein secretion by transfected cells in vivo. The biomimetic method proposed in this study is technically easier and more effective than traditional approaches by allowing a more sustained release of growth factors over a longer period of time.
In order to reduce complications and ease delivery, investigators have recently focused on the direct injection of adenovirus carrying BMPs into host bone defects. This strategy has been demonstrated to heal critical-sized segmental defects in rabbits and rat [49,50], accelerate healing of surgically created large osteochondral defects in a weight-bearing pony femoral condyle [51] and accelerate healing of tibial fractures in rabbits [52]. One study has also focused on the local injection of adBMP-2 in an osteroporotic sheep model [53]. Osteotomies created surgically in the tibias of adult female sheep demonstrated greater in vivo bending stiffness as well as larger and stiffer cross-sectional callus area density after treatment with adBMP-2 [53]. In an attempt to optimize the direct injection of viral vectors, Bertz et al. studied the effects of timing on administration of adBMP-2 for the repair of osseous defects [54]. They concluded that a delayed injection of adBMP-2 until 5 or 10 days post-surgery enabled a greater percentage of critical-sized, segmental defects to achieve radiological union, producing a repair tissue with enhanced mineralization and greater mechanical strength. While varying the injection date of adBMP-2 demonstrated positive outcomes, other investigators have suggested that alternatives such as use of adeno-associated viral vectors, use of cells or scaffolds as delivery vehicles, and various targeting strategies may enhance their biologic response to BMPs [51].
The aim of the present study was to investigate MGB/silk fibrin scaffolds combined with the advantages of gene delivery for BMP-7 and PDGF-b following local implantation into critical-sized osteoporotic metaphyseal defects created in a rat distal femur model. Essential to the repair of bone defects is the availability and properties of progenitor cells in the local biologic environment. It has previously been demonstrated that MSCs isolated from osteoporotic bone exhibit lower proliferation rates and favour their differentiation to adipose-tissues as opposed to osteoblasts [55]. This in turn can have significant ramifications on the healing of osseous defects as a result of an insufficient number of fully functional osteoblasts [56].
In the present study, the combination of a scaffold with a gene delivery system was employed to enhance the recruitment and differentiation of mesenchymal progenitor cells to osteoblasts. BMPs are potent osteoinductive growth factors for the differentiation of mesenchymal cells to osteoblasts [20]. It has been suggested that BMP concentration decreases with age and that greater osteogenic stimuli are required to overcome these age-related declines [57]. While adBMP-7 did improve new bone formation in osteoporotic defects when compared to scaffolds alone, the combination of adBMP-7 with adPDGF-b significantly enhanced bone regeneration. Previously it has been demonstrated that primary human MSCs exposed to BMP-2, BMP-4 and PDGF displayed a 3.5-, 3.6- and 22-fold increase in recruitment displaying the potent ability of PDGF when compared to BMPs [58]. The ability of combining a strong chemotactant growth factor (PDGF) for the recruitment of MSCs with an osteoinductive growth factor (BMP-7) for their differentiation to osteoblasts demonstrated a synergistic enhancement for the repair of osseous defects.
It was also observed that scaffolds containing adenovirus vectors displayed a higher number of TRAP-positive cells. Furthermore, it was observed that these scaffolds also degraded more quickly while still being able to increase new bone formation suggesting a more rapid rate of bone turnover/remodelling. The results from the present experiments suggest that as the scaffolds become degraded, subsequent release of viral vectors to the surrounding area initiate future recruitment of mesenchymal progenitor cells. As it has previously been demonstrated that osteoporotic-cells are less able to proliferate and differentiate in response to a lower concentration of growth factors, the need for delivery systems with an increased and sustained release of these factors might be advantageous for the treatment of osteoporotic-related fractures.
5. Conclusion
Our results demonstrate that these MBG/silk fibrin scaffolds are able to recruit and differentiate mesenchymal cells through the release of adPDGF-b and adBMP-7 allowing significantly new bone formation in critical-sized defects in ovariectomised rat femurs. Although the clinical use of gene therapy is still in its infancy, this study illustrates the successful delivery of low-cost, effective growth factor delivery for the treatment of osseous defects.
Acknowledgments
Funding for this study was supported by Program for New Century Excellent Talents in University (NCET-11-0414) and Excellent Youth Foundation of Hubei.
References
- 1.Genant HK, Cooper C, Poor G, Reid I, Ehrlich G, Kanis J, et al. Interim report and recommendations of the World Health Organization Task-Force for osteoporosis. Osteoporos Int. 1999;10(4):259–64. doi: 10.1007/s001980050224. [DOI] [PubMed] [Google Scholar]
- 2.Tontonoz P, Pei LM. Fat’s loss is bone’s gain. J Clin Invest. 2004;113:805–6. doi: 10.1172/JCI21311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rodan GA, Martin TJ. Therapeutic approaches to bone diseases. Science. 2000;289:1508–14. doi: 10.1126/science.289.5484.1508. [DOI] [PubMed] [Google Scholar]
- 4.Roush K. Prevention and treatment of osteoporosis in postmenopausal women: a review. Am J Nurs. 2011;111:26–35. doi: 10.1097/01.NAJ.0000403358.44058.f7. [DOI] [PubMed] [Google Scholar]
- 5.Adachi JD, Adami S, Gehlbach S, Anderson FA, Jr, Boonen S, Chapurlat RD, et al. Impact of prevalent fractures on quality of life: baseline results from the global longitudinal study of osteoporosis in women. Mayo Clin Proc. 2010;85:806–13. doi: 10.4065/mcp.2010.0082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Abrahamsen B, van Staa T, Ariely R, Olson M, Cooper C. Excess mortality following hip fracture: a systematic epidemiological review. Osteoporos Int. 2009;20:1633–50. doi: 10.1007/s00198-009-0920-3. [DOI] [PubMed] [Google Scholar]
- 7.Burge R, Dawson-Hughes B, Solomon DH, Wong JB, King A, Tosteson A. Incidence and economic burden of osteoporosis-related fractures in the United States, 2005–2025. J Bone Miner Res. 2007;22:465–75. doi: 10.1359/jbmr.061113. [DOI] [PubMed] [Google Scholar]
- 8.Moazzaz P, Gupta MC, Gilotra MM, Gilotra MN, Maitra S, Theerajunyaporn T, et al. Estrogen-dependent actions of bone morphogenetic protein-7 on spine fusion in rats. Spine (Phila Pa 1976) 2005;30:1706–11. doi: 10.1097/01.brs.0000172230.01655.55. [DOI] [PubMed] [Google Scholar]
- 9.Hao YJ, Zhang G, Wang YS, Qin L, Hung WY, Leung K, et al. Changes of microstructure and mineralized tissue in the middle and late phase of osteoporotic fracture healing in rats. Bone. 2007;41:631–8. doi: 10.1016/j.bone.2007.06.006. [DOI] [PubMed] [Google Scholar]
- 10.Namkung-Matthai H, Appleyard R, Jansen J, Hao Lin J, Maastricht S, Swain M, et al. Osteoporosis influences the early period of fracture healing in a rat osteoporotic model. Bone. 2001;28:80–6. doi: 10.1016/s8756-3282(00)00414-2. [DOI] [PubMed] [Google Scholar]
- 11.Silva BC, Bilezikian JP. New approaches to the treatment of osteoporosis. Annu Rev Med. 2011;62:307–22. doi: 10.1146/annurev-med-061709-145401. [DOI] [PubMed] [Google Scholar]
- 12.Chen FM, Wu LA, Zhang M, Zhang R, Sun HH. Homing of endogenous stem/progenitor cells for in situ tissue regeneration: promises, strategies, and translational perspectives. Biomaterials. 2011;32:3189–209. doi: 10.1016/j.biomaterials.2010.12.032. [DOI] [PubMed] [Google Scholar]
- 13.White AP, Vaccaro AR, Hall JA, Whang PG, Friel BC, McKee MD. Clinical applications of BMP-7/OP-1 in fractures, nonunions and spinal fusion. Int Orthop. 2007;31:735–41. doi: 10.1007/s00264-007-0422-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Govender S, Csimma C, Genant HK, Valentin-Opran A, Amit Y, Arbel R, et al. Recombinant human bone morphogenetic protein-2 for treatment of open tibial fractures: a prospective, controlled, randomized study of four hundred and fifty patients. J Bone Joint Surg Am. 2002;84-A:2123–34. doi: 10.2106/00004623-200212000-00001. [DOI] [PubMed] [Google Scholar]
- 15.Steed DL, Donohoe D, Webster MW, Lindsley L. Effect of extensive debridement and treatment on the healing of diabetic foot ulcers. Diabetic Ulcer Study Group. J Am Coll Surg. 1996;183:61–4. [PubMed] [Google Scholar]
- 16.Wieman TJ, Smiell JM, Su Y. Efficacy and safety of a topical gel formulation of recombinant human platelet-derived growth factor-BB (becaplermin) in patients with chronic neuropathic diabetic ulcers. A phase III randomized placebo-controlled double-blind study. Diabetes Care. 1998;21:822–7. doi: 10.2337/diacare.21.5.822. [DOI] [PubMed] [Google Scholar]
- 17.Miron RJ, Zhang YF. Osteoinduction: a review of old concepts with new standards. J Dent Res. 2012 doi: 10.1177/0022034511435260. http://dx.doi.org/10.1177/0022034511435260. [DOI] [PubMed]
- 18.Kaplan DR, Chao FC, Stiles CD, Antoniades HN, Scher CD. Platelet alpha granules contain a growth factor for fibroblasts. Blood. 1979;53:1043–52. [PubMed] [Google Scholar]
- 19.Habisch HJ, Fiedler J, Ludolph AC, Storch A, Brenner RE. Altered migration and adhesion potential of proneurally converted human bone marrow stromal cells. Cytotherapy. 2008;10:824–33. doi: 10.1080/14653240802474331. [DOI] [PubMed] [Google Scholar]
- 20.Lissenberg-Thunnissen SN, de Gorter DJ, Sier CF, Schipper IB. Use and efficacy of bone morphogenetic proteins in fracture healing. Int Orthop. 2011;35:1271–80. doi: 10.1007/s00264-011-1301-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ghosh SS, Gopinath P, Ramesh A. Adenoviral vectors: a promising tool for gene therapy. Appl Biochem Biotechnol. 2006;133:9–29. doi: 10.1385/abab:133:1:9. [DOI] [PubMed] [Google Scholar]
- 22.Chang PC, Cirelli JA, Jin Q, Seol YJ, Sugai JV, D’Silva NJ, et al. Adenovirus encoding human platelet-derived growth factor-B delivered to alveolar bone defects exhibits safety and biodistribution profiles favorable for clinical use. Hum Gene Ther. 2009;20:486–96. doi: 10.1089/hum.2008.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gu DL, Nguyen T, Gonzalez AM, Printz MA, Pierce GF, Sosnowski BA, et al. Adenovirus encoding human platelet-derived growth factor-B delivered in collagen exhibits safety, biodistribution, and immunogenicity profiles favorable for clinical use. Mol Ther. 2004;9:699–711. doi: 10.1016/j.ymthe.2004.02.018. [DOI] [PubMed] [Google Scholar]
- 24.Wei G, Ma PX. Partially nanofibrous architecture of 3D tissue engineering scaffolds. Biomaterials. 2009;30:6426–34. doi: 10.1016/j.biomaterials.2009.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dorozhkin SV. Bioceramics of calcium orthophosphates. Biomaterials. 2010;31:1465–85. doi: 10.1016/j.biomaterials.2009.11.050. [DOI] [PubMed] [Google Scholar]
- 26.Zhang ZY, Teoh SH, Hui JH, Fisk NM, Choolani M, Chan JK. The potential of human fetal mesenchymal stem cells for off-the-shelf bone tissue engineering application. Biomaterials. 2012;33:2656–72. doi: 10.1016/j.biomaterials.2011.12.025. [DOI] [PubMed] [Google Scholar]
- 27.Karageorgiou V, Kaplan D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials. 2005;26:5474–91. doi: 10.1016/j.biomaterials.2005.02.002. [DOI] [PubMed] [Google Scholar]
- 28.Lu Q, Wang X, Lu S, Li M, Kaplan DL, Zhu H. Nanofibrous architecture of silk fibroin scaffolds prepared with a mild self-assembly process. Biomaterials. 2011;32:1059–67. doi: 10.1016/j.biomaterials.2010.09.072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wu C, Zhang Y, Zhou Y, Fan W, Xiao Y. A comparative study of mesoporous glass/silk and non-mesoporous glass/silk scaffolds: physiochemistry and in vivo osteogenesis. Acta Biomater. 2011;7:2229–36. doi: 10.1016/j.actbio.2010.12.019. [DOI] [PubMed] [Google Scholar]
- 30.Lima I, Farias MLF, Percegoni N, Rosenthal D, de Assis JT, Anjos MJ, et al. Micro imaging analysis for osteoporosis assessment. Spectrochim Acta B. 2010;65:253–7. [Google Scholar]
- 31.Mankani MH, Kuznetsov SA, Avila NA, Kingman A, Robey PG. Bone formation in transplants of human bone marrow stromal cells and hydroxyapatite-tricalcium phosphate: prediction with quantitative CT in mice. Radiology. 2004;230:369–76. doi: 10.1148/radiol.2302011529. [DOI] [PubMed] [Google Scholar]
- 32.Hutmacher DW, Sawyer AA, Song SJ, Susanto E, Chuan P, Lam CXF, et al. The stimulation of healing within a rat calvarial defect by mPCL-TCP/collagen scaffolds loaded with rhBMP-2. Biomaterials. 2009;30:2479–88. doi: 10.1016/j.biomaterials.2008.12.055. [DOI] [PubMed] [Google Scholar]
- 33.Ebneter A, Casson RJ, Wood JP, Chidlow G. Microglial activation in the visual pathway in experimental glaucoma: spatiotemporal characterization and correlation with axonal injury. Invest Ophthalmol Vis Sci. 2010;51:6448–60. doi: 10.1167/iovs.10-5284. [DOI] [PubMed] [Google Scholar]
- 34.Fernandes JC, Wang H, Jreyssaty C, Benderdour M, Lavigne P, Qiu X, et al. Bone-protective effects of nonviral gene therapy with folate-chitosan DNA nanoparticle containing interleukin-1 receptor antagonist gene in rats with adjuvant-induced arthritis. Mol Ther. 2008;16:1243–51. doi: 10.1038/mt.2008.99. [DOI] [PubMed] [Google Scholar]
- 35.Kawaguchi H, Akune T, Ohba S, Kamekura S, Yamaguchi M, Chung UI, et al. PPAR gamma insufficiency enhances osteogenesis through osteoblast formation from bone marrow progenitors. J Clin Invest. 2004;113:846–55. doi: 10.1172/JCI19900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kanakaris NK, Petsatodis G, Tagil M, Giannoudis PV. Is there a role for bone morphogenetic proteins in osteoporotic fractures? Injury. 2009;40(Suppl 3):S21–6. doi: 10.1016/S0020-1383(09)70007-5. [DOI] [PubMed] [Google Scholar]
- 37.Peter B, Gauthier O, Laib S, Bujoli B, Guicheux J, Janvier P, et al. Local delivery of bisphosphonate from coated orthopedic implants increases implants mechanical stability in osteoporotic rats. J Biomed Mater Res A. 2006;76:133–43. doi: 10.1002/jbm.a.30456. [DOI] [PubMed] [Google Scholar]
- 38.Peter B, Pioletti DP, Laib S, Bujoli B, Pilet P, Janvier P, et al. Calcium phosphate drug delivery system: influence of local zoledronate release on bone implant osteointegration. Bone. 2005;36:52–60. doi: 10.1016/j.bone.2004.10.004. [DOI] [PubMed] [Google Scholar]
- 39.Cesareo R, Iozzino M, Alva D, Napolitano C, De Rosa B, Contini S, et al. Evidence based medicine and effective interventions of pharmacological therapy for the prevention of osteoporotic fractures. Minerva Endocrinol. 2007;32:275–95. [PubMed] [Google Scholar]
- 40.Verron E, Gauthier O, Janvier P, Pilet P, Lesoeur J, Bujoli B, et al. In vivo bone augmentation in an osteoporotic environment using bisphosphonate-loaded calcium deficient apatite. Biomaterials. 2010;31:7776–84. doi: 10.1016/j.biomaterials.2010.06.047. [DOI] [PubMed] [Google Scholar]
- 41.Tang BM, Eslick GD, Nowson C, Smith C, Bensoussan A. Use of calcium or calcium in combination with vitamin D supplementation to prevent fractures and bone loss in people aged 50 years and older: a meta-analysis. Lancet. 2007;370:657–66. doi: 10.1016/S0140-6736(07)61342-7. [DOI] [PubMed] [Google Scholar]
- 42.Liu X, Pettway GJ, McCauley LK, Ma PX. Pulsatile release of parathyroid hormone from an implantable delivery system. Biomaterials. 2007;28:4124–31. doi: 10.1016/j.biomaterials.2007.05.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Faucheux C, Verron E, Soueidan A, Josse S, Arshad MD, Janvier P, et al. Controlled release of bisphosphonate from a calcium phosphate biomaterial inhibits osteoclastic resorption in vitro. J Biomed Mater Res A. 2009;89:46–56. doi: 10.1002/jbm.a.31989. [DOI] [PubMed] [Google Scholar]
- 44.Evans C. Gene therapy for the regeneration of bone. Injury. 2011;42:599–604. doi: 10.1016/j.injury.2011.03.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Phillips JE, Gersbach CA, Garcia AJ. Virus-based gene therapy strategies for bone regeneration. Biomaterials. 2007;28:211–29. doi: 10.1016/j.biomaterials.2006.07.032. [DOI] [PubMed] [Google Scholar]
- 46.Chen FM, Zhang M, Wu ZF. Toward delivery of multiple growth factors in tissue engineering. Biomaterials. 2010;31:6279–308. doi: 10.1016/j.biomaterials.2010.04.053. [DOI] [PubMed] [Google Scholar]
- 47.Jiang X, Zhao J, Wang S, Sun X, Zhang X, Chen J, et al. Mandibular repair in rats with premineralized silk scaffolds and BMP-2-modified bMSCs. Biomaterials. 2009;30:4522–32. doi: 10.1016/j.biomaterials.2009.05.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gugala Z, Davis AR, Fouletier-Dilling CM, Gannon FH, Lindsey RW, Olmsted-Davis EA. Adenovirus BMP2-induced osteogenesis in combination with collagen carriers. Biomaterials. 2007;28:4469–79. doi: 10.1016/j.biomaterials.2007.07.007. [DOI] [PubMed] [Google Scholar]
- 49.Betz OB, Betz VM, Nazarian A, Pilapil CG, Vrahas MS, Bouxsein ML, et al. Direct percutaneous gene delivery to enhance healing of segmental bone defects. J Bone Joint Surg Am. 2006;88:355–65. doi: 10.2106/JBJS.E.00464. [DOI] [PubMed] [Google Scholar]
- 50.Baltzer AW, Lattermann C, Whalen JD, Wooley P, Weiss K, Grimm M, et al. Genetic enhancement of fracture repair: healing of an experimental segmental defect by adenoviral transfer of the BMP-2 gene. Gene Ther. 2000;7:734–9. doi: 10.1038/sj.gt.3301166. [DOI] [PubMed] [Google Scholar]
- 51.Menendez MI, Clark DJ, Carlton M, Flanigan DC, Jia G, Sammet S, et al. Direct delayed human adenoviral BMP-2 or BMP-6 gene therapy for bone and cartilage regeneration in a pony osteochondral model. Osteoarthr Cartil. 2011 Aug;19:1066–75. doi: 10.1016/j.joca.2011.05.007. [DOI] [PubMed] [Google Scholar]
- 52.Bertone AL, Pittman DD, Bouxsein ML, Li J, Clancy B, Seeherman HJ. Adenoviral-mediated transfer of human BMP-6 gene accelerates healing in a rabbit ulnar osteotomy model. J Orthop Res. 2004;22:1261–70. doi: 10.1016/j.orthres.2004.03.014. [DOI] [PubMed] [Google Scholar]
- 53.Egermann M, Baltzer AW, Adamaszek S, Evans C, Robbins P, Schneider E, et al. Direct adenoviral transfer of bone morphogenetic protein-2 cDNA enhances fracture healing in osteoporotic sheep. Hum Gene Ther. 2006;17:507–17. doi: 10.1089/hum.2006.17.507. [DOI] [PubMed] [Google Scholar]
- 54.Betz OB, Betz VM, Nazarian A, Egermann M, Gerstenfeld LC, Einhorn TA, et al. Delayed administration of adenoviral BMP-2 vector improves the formation of bone in osseous defects. Gene Ther. 2007;14:1039–44. doi: 10.1038/sj.gt.3302956. [DOI] [PubMed] [Google Scholar]
- 55.Giannoudis P, Tzioupis C, Almalki T, Buckley R. Fracture healing in osteoporotic fractures: is it really different? A basic science perspective. Injury. 2007;38(Suppl 1):S90–9. doi: 10.1016/j.injury.2007.02.014. [DOI] [PubMed] [Google Scholar]
- 56.Rodriguez JP, Montecinos L, Rios S, Reyes P, Martinez J. Mesenchymal stem cells from osteoporotic patients produce a type I collagen-deficient extracellular matrix favoring adipogenic differentiation. J Cell Biochem. 2000;79:557–65. doi: 10.1002/1097-4644(20001215)79:4<557::aid-jcb40>3.0.co;2-h. [DOI] [PubMed] [Google Scholar]
- 57.Bessho K, Iizuka T. Changes in bone inducing activity of bone morphogenetic protein with aging. Ann Chir Gynaecol Suppl. 1993;207:49–53. [PubMed] [Google Scholar]
- 58.Fiedler J, Roderer G, Gunther KP, Brenner RE. BMP-2, BMP-4, and PDGF-bb stimulate chemotactic migration of primary human mesenchymal progenitor cells. J Cell Biochem. 2002;87:305–12. doi: 10.1002/jcb.10309. [DOI] [PubMed] [Google Scholar]