Abstract
The incidence of Klebsiella pneumoniae liver abscess (KPLA) has increased in a number of Asian countries over the past 30 years. Diabetes mellitus (DM) is a risk factor for KPLA. The prevalence and clinical features of KPLA in patients with and without DM have been well described; however, the underlying molecular mechanism responsible for the increased incidence of KPLA in patients with DM remains unclear. In the present study, a mouse model of DM was constructed and mice were infected with K. pneumoniae. Tissues were harvested for immunohistochemical and inflammatory factor expression analyses. The results revealed that the number of liver abscesses in mice with DM was greater than that observed in normal mice. The expression of interleukin (IL)-1β, IL-2, IL-6, macrophage inflammatory protein-1α and tumor necrosis factor-α in the liver tissues of mice with DM was significantly higher compared with normal mice. Western blotting results revealed that the expression of phosphorylated (p)-inhibitor of nuclear factor κB (NFκB) kinase subunit β, p-NFκB and p-inhibitor of NFκB was significantly increased in the liver tissue of mice with DM compared with that of normal mice. These results suggest that activation of the NFκB signaling pathways has a regulatory effect on the pathogenesis of K. pneumoniae bacteria liver abscesses and that high glucose conditions may promote the activation of NFκB signaling.
Keywords: Klebsiella pneumoniae, liver abscess, nuclear factor κB, diabetes, inflammatory
Introduction
The incidence of Klebsiella pneumoniae liver abscesses (KPLA) has increased in a number of Asian countries over the past 30 years, particularly in Taiwan and Korea (1,2). Shi et al (3)analyzed clinical data from 296 cases of bacterial liver abscess collected between 1994 and 2015. Their results revealed that KPLA accounted for 63.9% of cases, while 13.2% were attributable to Escherichia coli liver abscesses (3). K. pneumoniae is a Gram negative bacteria that is colonized as normal flora in the human gut but is prone to causing parenteral infection and even bacteremia in patients with poor immunity (4).
A number of factors predispose patients with diabetes mellitus (DM) to infections, including genetic susceptibility to infection, altered cellular and humoral immune defense mechanisms, local factors (e.g., poor blood supply and nerve damage) and DM-associated metabolic changes (5). Increasing evidence suggests that DM-induced hyperglycemia may cause vascular endothelial damage (6,7), which contributes to infections. K. pneumoniae is considered to be a common pathogen in patients with DM (8). A previous study reported that patients with DM were 3.6× more likely to develop bacterial liver abscesses compared with control subjects (9). Lin et al (10) reported that patients with poor blood glucose control (glycosylated hemoglobin HbA1c <7%) and had a higher prevalence of recessive liver abscesses, liver abscesses and gas transfer compared with patients with good blood glucose control (HbA1c=7%) (11). However, the molecular mechanism underlying the increased incidence of K. pneumoniae liver abscess in patients with DM remains unclear. It has been demonstrated that K. pneumoniae infection increases the inflammation responses (12).
In the present study, a streptozotocin (STZ)-induced DM mouse model was constructed and infected with K. pneumoniae to induce KPLA. Pathological changes in liver tissues and the expression of NFκB pathway components were assessed.
Materials and methods
Bacterial strains and animals
K. pneumoniae was isolated from a male 58 year old patient with a liver abscess confirmed by the Department of Interventional and Vascular Surgery, Tenth People's Hospital of Tongji University. This procedure included a fine-needle aspiration of the patients' fluctuant mass from which and 1 ml of purulent fluid was obtained and sent for culture under 37°C and with a Sheep Blood AGAR Plate (Shanghai Yihua Medical Technology Co., Ltd.) which yielded Klebsiella pneumoniae, all process was under anesthesia with 3% sevoflurane (anesthetized by inhalation). A total of 48 6–8-week-old male C57BL/6 mice (n=48) were purchased from Fudan University Animal Care Committee (Shanghai, China) and were individually housed at 22°C with 60% humidity, a 12 h light/dark cycle and free access to tap water and food. Animals were acclimated to the laboratory environment for ≥3 weeks prior to experiments.
Ethics statement
All experiments were approved by the Ethics Committee of the Tenth People's Hospital of Tongji University (Shanghai, China) and performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (National Institutes of Health Bethesda, MD, USA). All mice were anesthetized by intraperitoneal injection of sodium pentobarbital (30 mg/kg) prior to euthanasia.
Generation of the type 1 DM mouse model
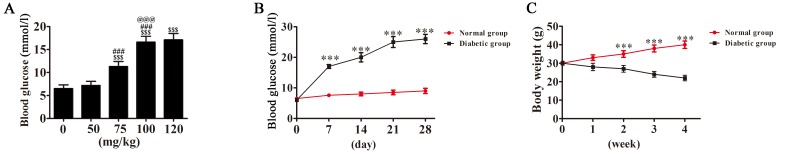
Mice were intraperitoneally injected with 50, 75, 100 or 120 mg/kg of streptozotocin (STZ; Sigma-Aldrich, Merck KGaA, Darmstadt, Germany) dissolved in 0.05 M citrate buffer (pH 4.1–4.5). Blood glucose levels and weight were measured at different times (including 0, 7, 14, 21 and 28 days). Based on the results (Fig. 1), treatment with 100 mg/kg, STZ resulted in a significant increase in blood glucose compared with lower dosages, however no significant difference in blood glucose levels was observed between the 100 and 120 mg/kg dosage groups. As such, 100 mg/kg STZ was selected as previously reported (3). Control mice were treated with the same amount of citrate buffer without STZ. Mice with blood sugar levels >20 mmol/l were considered to have DM. Following 7 days of STZ treatment, mice exhibited symptoms of DM, including polydipsia, polyphagia, polyuria and emaciation. Blood glucose levels were >20 mmol/l. At 1 month following injection, the blood glucose levels stabilized at 27–30 mmol/l.
Figure 1.
Blood glucose and weight changes. (A) The change of blood glucose following treatment with different concentrations of STZ. $$$P<0.001 vs. control. ###P<0.001 vs. 50 mg/kg STZ treatment group. @@@P<0.001 vs. 75 mg/kg STZ treatment group. (B) Blood sugar changes over time following treatment with 100 mg/kg of STZ. ***P<0.001 vs. normal group. (C) Mouse weight change following treatment with 100 mg/kg of STZ for different time. ***P<0.001 vs. normal group. STZ, streptozotocin.
Infection of mice with K. pneumoniae bacteria
The K. pneumoniae strain was cultured on sheep blood agar plates (Shanghai Yihua Medical Technology Co., Ltd.) overnight under 37°C and harvested using PBS to make a bacterial suspension of 3×107 colony-forming units (CFU) per 50 µl. The 50-µl bacterial suspensions were orally inoculated into the mice. Mice were divided into four groups: PBS control normal mouse group (n=6), PBS-control DM group (n=6), K. pneumoniae-infected DM group (n=18) and K. pneumoniae-infected normal mouse group (n=18).
Liver tissue pathological examinations
Liver tissues were dissected 7 days following inoculation with K. pneumoniae, fixed in 10% formalin for 24 h at room temperature, washed twice with PBS and maintained in PBS for 24 h prior to dehydration in a graded series of ethanol for 45 min. Finally, the samples were immersed in xylene for 20 min at room temperature and tissues were embedded in wax. Sections from the tissue block were cut on a microtome to a thickness of 5 µm and fixed on a glass slide. Paraffin was removed using xylene and then the slices were rinsed with a graded series of ethanol followed by distilled water. The tissue sections were stained with hematoxylin solution at room temperature for 3 min and rinsed with acidified water and ammonia with water for 30 sec. Tissue sections were rinsed in water for 1 h and then stored in distilled water, following which they were rinsed with 70 and 90% alcohol for 10 min each. Sections were stained with eosin for 2 min at room temperature. The stained sections were dehydrated using absolute alcohol and xylene and subsequently covered with sealing gum and glass coverslips.
The degree of liver inflammation was determined by a blinded investigator as previously described (13). Scoring was as follows: 1, <10 micro-abscesses on each liver section and no necrotic regions; 2, >10 and <20 micro-abscesses on each liver section and no necrotic regions; 3, >20 and <30 micro-abscesses on each liver section and that no necrotic regions; 4, >30 micro-abscesses on each liver section and no necrotic regions; 5, <5 necrotic regions; 6, >5 and <10 necrotic regions; 7, >10 and <15 necrotic regions; 8, >15 necrotic regions. A total of three different sections from the largest liver lobule of each mouse were examined. Histological assessments of intestines were performed using a scoring system as previously described (14).
ELISA
The expression of inflammatory factors and chemokines, including interleukin (IL)-1β (cat. no. EMC021), IL-2 (cat. no. EMC0623), IL-6 (cat. no. EMC0003), IL-10 (cat. no. EMC106), tumor necrosis factor (TNF)-α (cat. no. EMC323) and macrophage inflammatory protein (MIP)-1α (cat. no. EMC266) in the liver lysates (liver tissues were harvested in lysis buffer (BD Biosciences, Franklin Lakes, NJ, USA) and lysates were cleared using centrifugation at 12,000 × g for 10 min at 4°C) was measured by using standard ELISA kits (QuantiCyto, Shenzhen, China) according to the manufacturer's protocol and normalized to the levels of total proteins.
Western blot analysis
Livers from all experimental groups were homogenized with lysis buffer (8 M urea, 50 mM DTT, 2% CHAPS and complete protease and phosphatase inhibitors). The concentration of resulting suspensions (liver tissues were harvested in lysis buffer (BD Biosciences) and lysates were cleared by centrifugation at 12,000 × g for 10 min at 4°C) was quantified with the two-dimensional Quant kit (GE Healthcare Life Sciences, Little Chalfont, UK). A total of 30 µg protein/lane was separated by 8% SDS-PAGE and transferred onto a polyvinylidene difluoride membranes (EMD Millipore, Billerica, MA, USA). Membranes were blocked with 2% skim milk at room temperature for 1 h and probed with the following antibodies at 4°C overnight: anti-inhibitor of κB (IκB) kinase α (IKKα; cat. no. ab32041; 1:500; Abcam, Cambridge, UK), anti-IKKβ (cat. no. 2684; 1:500), anti-IKBα (cat. no. 9242; 1:500), anti-NFκB (cat. no. 4810; 1:500), anti-phosphorylated (p)-IKKα (cat. no. 11930; 1:1,000), anti-p-IKKβ (cat. no. 8943; 1:1,000), anti-p-IKBα (cat. no. 4814; 1:1,000) and anti-p-NFκB (cat. no. 8242; 1:1,000; all Cell Signaling Technology, Inc., Danvers, MA, USA). Following hybridization with horseradish peroxidase (HRP)-conjugated secondary antibodies (cat. no. 5836; 1:1,000; Cell Signaling Technology, Inc.) for 1 h at room temperature, the blots were developed with Immobilon™ Western Chemiluminescent HRP Substrate (EMD Millipore) and visualized with an ImageQuant™ LAS 4000 Mini Biomolecular Imager (GE Healthcare Life Sciences). The band intensities were quantified using a UN-SCAN-IT gel, version 6.0 software (Silk Scientific, Orem, UT, USA).
Statistical analysis
Statistical analyses were performed using SPSS version 2.0 (SPSS, Inc., Chicago, IL, USA). Statistical results are expressed as the mean ± standard deviation. Statistical analyses were performed using one-way analysis of variance and a post-hoc Tukey's test. P<0.05 was considered to indicate a statistically significant difference.
Results
DM mouse model construction
In order to construct a model of mice with stable DM, 0–120 mg/kg STZ was administered via intraperitoneal injection. Following 7 days, blood glucose levels were detected and the results revealed that 100 mg/kg STZ resulted in a stable increase in blood glucose, and so was selected for further experiments.
Pathological changes in mouse liver tissues
Liver tissue abscesses were observed under an optical microscope (magnification, ×200). The liver cells from normal mice and mice with DM were round, borders were clear, cell nuclei and cytoplasm were stained normally and there were no inflammatory cell aggregations in the portal areas. However, in normal mice and mice with DM infected with K. pneumoniae, foci of hepatic necrosis, diffuse infiltration of neutrophils, small amounts of lymphocytes in the necrotic areas and balloon-like lesions of the peripheral liver cells were observed. In 2/18 K. pneumoniae-infected normal mice the focal liver cells were enlarged, cell boundaries were unclear, cytoplasm was pale and loose, and a small amount of neutrophils and lymphocytes had collected in the portal areas. The liver tissues of 5/18 mice in this group exhibited no inflammatory responses. A total of 3/18 K. pneumoniae-infected normal mice presented with focal hepatic necrosis, diffuse infiltration of neutrophils, small numbers of lymphocytes in the necrotic areas and balloon-like lesions of the peripheral liver cells. Furthermore, 4/18 K. pneumoniae-infected mice with DM had enlarged focal liver cells, unclear cell boundaries, pale and loose cytoplasm and small numbers of neutrophils and lymphocytes were collected in the portal areas (Fig. 2A-F). To obtain further insight into the role of the diabetic microenvironment in KPLA, semi quantitative analyses were performed on liver sections prepared from K. pneumoniae treated mice. Significantly more liver damage (assessed by a higher pathology score) was observed in K. pneumoniae-infected mice with DM compared with K. pneumoniae-infected normal mice (P<0.001; Fig. 2G). Little to no liver damage was observed in normal mice and mice with DM.
Figure 2.
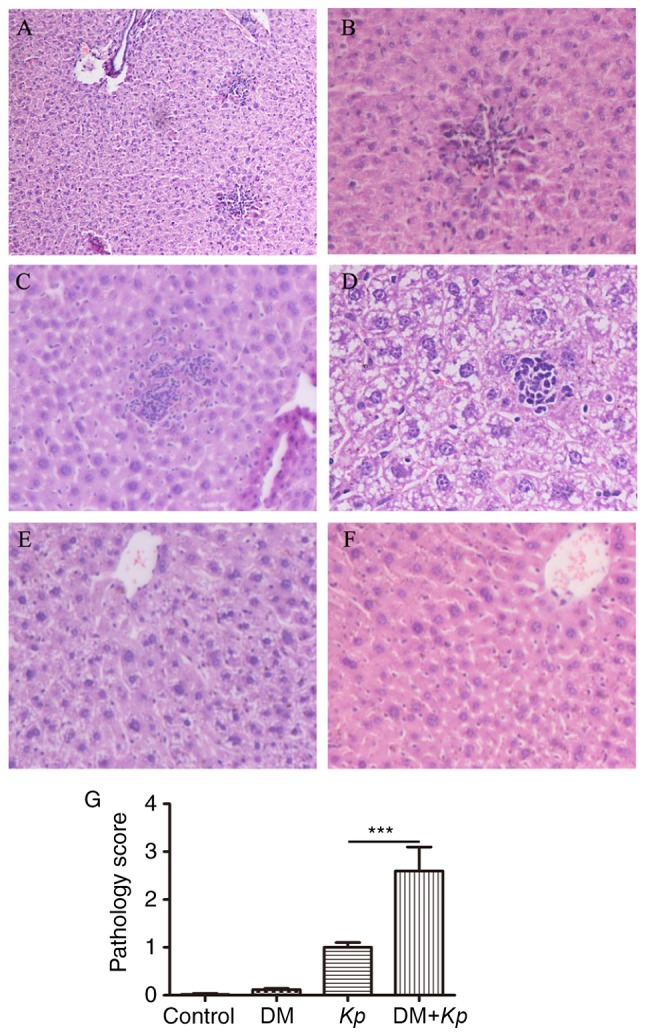
Histochemical analysis of liver tissues. (A-C) Kp-infected mice with DM. The magnifications utilized for (A-C) are ×100, ×200 and ×200, respectively. (D) Kp-infected normal mice (magnification, ×400). (E) Kp-infected normal mice (magnification, ×200). (F) Control mice (magnification, ×200). (G) Semi-quantitative analysis of histological observations (magnification, ×200). ***P<0.001. Kp, Klebsiella pneumoniae; DM, diabetes mellitus.
Expression of tissue inflammatory factors
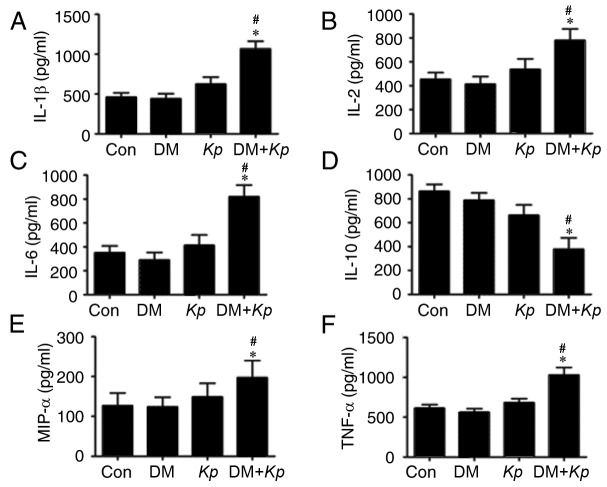
The results revealed that the expression of IL-1β, IL-2, IL-6, MIP-1α and TNFα was significantly increased in liver tissues from K. pneumoniae-infected mouse with DM compared with K. pneumoniae-infected normal mice (Fig. 3). The expression of IL-1β, IL-2, IL-6, MIP-1α and TNFα was increased in liver tissues from K. pneumoniae-infected mice with DM compared with PBS control mice with DM (P<0.05; Fig. 3). The expression of IL-1β, IL-2, IL-6, MIP-1α and TNFα were not significantly different in liver tissues from K. pneumoniae-infected normal mice compared with control mice (P<0.05; Fig. 3), suggesting that K. pneumoniae infection promotes inflammatory responses.
Figure 3.
ELISA assays for (A) IL-1β, (B) IL-2, (C) IL-6, (D) IL-10, (E) MIP-1α, (F) TNFα in mice with or without Kp and DM. *P<0.05 vs. Con; #P<0.05 vs. K. pneumoniae infected normal mice. IL, interleukin; MIP, macrophage inflammatory protein; TNF, tumor necrosis factor; Con, control; Kp, Klebsiella pneumoniae; DM, diabetes mellitus.
Western blot analysis
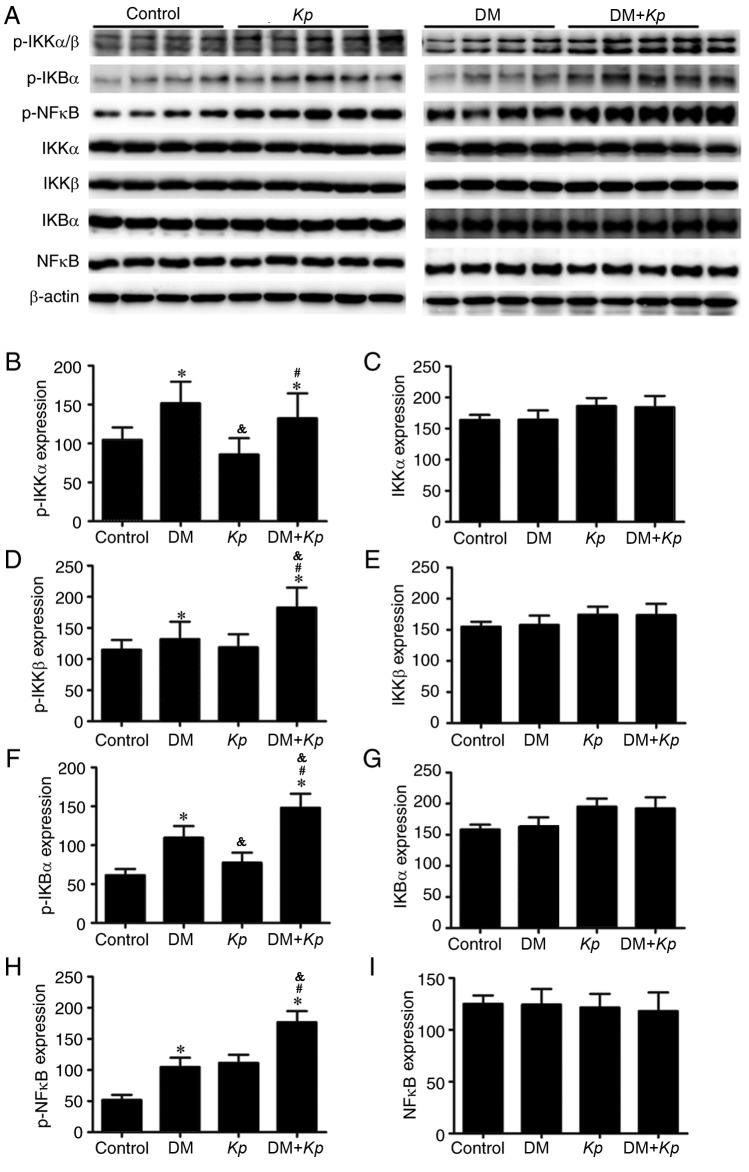
The results of western blotting revealed that the expression of p-IKKβ, p-IKBα and p-NFκB was significantly higher in liver tissues from K. pneumoniae-infected mice with DM compared with K. pneumoniae-infected normal mice (P<0.05; Fig. 4). But the expression of IKKα, IKKβ, IKBα and p-NFκB did not exhibit any significant differences. The expression of p-IKKα, p-IKKβ, p-IKBα and p-NFκB in the liver tissues of K. pneumoniae-infected mice with DM was significantly higher compared with normal mice with DM (P<0.05; Fig. 4). The expression of p-IKKα, p-IKKβ, p-IKBα and p-NFKB in liver tissues from K. pneumoniae-infected normal mice was significantly higher compared with control mice (P<0.05; Fig. 4).
Figure 4.
(A) Expression of NFκB signaling pathway-associated proteins was measured using western blotting. Relative expression of (B) p-IKKα, (C) IKKα, (D) p-IKKβ, (E) IKKβ, (F) p-IKBα, (G) IKBα, (H) p-NFκB and (I) NFκB. *P<0.05 vs. control. #P<0.05 vs. K. pneumoniae-infected normal mice. &P<0.05 vs. uninfected mice with DM. NF, nuclear factor; p, phosphorylated; IKK, inhibitor of κB kinase; Kp, Klebsiella pneumoniae; DM, diabetes mellitus.
Discussion
In the present study, DM was induced in mice via intraperitoneal injections of 100 mg/kg STZ. Each mouse was orally inoculated with K. pneumoniae to establish the liver abscess model and liver tissues were separated 7 days later. Histopathological examination revealed focal necrosis of liver cells, diffuse infiltration of neutrophils and lymphocytes, hepatocyte swelling, unclear cell boundaries, slightly stained cytoplasm, cytoplasmic osteoporosis and ballooning, all of which are indicative of liver abscess formation. In this study, the incidence of liver abscess formation in mice with DM was 50%, which is lower than previously reported (15). This may be because liver extraction was performed prior to liver cell edema and the infiltration of small amounts of inflammatory cells did not result in abscess formation. Alternatively, the K. pneumoniae strain may have been to the previous study and have different virulence. Lastly, when bacterial suspensions were orally inoculated, the operation procedure may have resulted in the bacterial suspension failing to enter the stomach of the mouse.
In the present study, the number of liver abscesses observed in the K. pneumoniae-infected mice with DM was higher compared with the K. pneumoniae-infected normal mice. This indicated that that high glucose induced the NFκB signaling pathways exerted a promotional effect on the pathogenesis of K. pneumoniae bacteria induced liver abscess. After microbial infection, the host immune system recognizes pathogen-associated molecular patterns and stimulates the production of inflammatory mediators, including ILs, complement, TNFα and neutrophils, as well as macrophage aggregation, bacterial phagocytosis and degradation, resulting in a protective immune response (16–18). In the present study, the expression of IL-1β, IL-2, IL-6, MIP-1α and TNFα in the K. pneumoniae-infected mice with DM was increased compared with the control mice with DM. Furthermore, the expression of these factors was also increased in K. pneumoniae-infected normal mice compared with control mice. Kupffer cells, capillary endothelial cells, monocytes and macrophages produce IL-1β, IL-2, IL-6, MIP-1α and TNFα to activate the inflammatory signaling pathways of NFκB, neutrophil aggregation, phagocytosis and degradation (19,20). In acute inflammation, IL-1β and TNFα are the main inflammatory mediators and may be quickly released to activate the inflammatory reaction cascade, or even induce endotoxin shock, with the synergistic effects of NFκB (21).
MIP-1α is a chemotactic factor secreted by macrophages that attracts neutrophils and macrophages to the site of infection (16). High IL-1β, TNFα and MIP-1α expression in mice with DM may increase the expression of NFκB signaling pathway proteins and the acute inflammatory reaction in the liver. Excessive inflammatory reactions in the body can cause uncontrollable tissue damage and liver inflammation, especially in hyperglycemic tissue microenvironments, resulting in necrosis of the liver cells and abscess formation (22).
NFκB is a nuclear transcription factor that exists as a dimer and is widely distributed in multicellular organisms. IKK, IKB, NFκB and other family members coordinate inflammatory responses, innate and adaptive immune responses and cell differentiation and proliferation (21,23–25). In inflammatory conditions, extracellular stimulation is able to activate the NFκB signaling pathway proteins, whose expression is crucial for the progression of the inflammatory response. IKK is a IKB protein kinas, comprising three subunits; IKKα, IKKβ and IKK (26). IKK is inactive in resting cells; however, extracellular K. pneumoniae, IL-1β and TNFα stimulate IKKα, IKKβ and NFκB activation via phosphorylation (24,25,27). In the present study, the expression of p-IKKα, p-IKKβ, p-IKBα and p-NFκB in the K. pneumoniae-infected mice with DM was increased compared with normal mice with DM. Furthermore, levels of these proteins were also increased in K. pneumoniae-infected normal mice compared with the control group. Following K. pneumoniae infection of liver tissues, macrophages and Kupffer cells secrete IL-1β, TNFα, MIP-1α and other cytokines, causing hepatocyte IKKα and IKKβ phosphorylation, as well as the activation and further phosphorylation of IKBα and NFκB (26). The activation of these proteins, in turn, promotes the transcription of inflammatory cytokines and the cascade of inflammatory processes, leading to liver cell damage, necrosis and liver abscess formation (27). In the present study, the expression of p-IKKβ, p-IKBα and p-NFκB in K. pneumoniae-infected mice with DM was significantly higher compared with K. pneumoniae-infected normal mice. Increased p-IKKβ, p-IKBα and p-NFκB expression in DM mouse liver cells stimulates monocytes, macrophages and liver Kupffer cells to secrete larger amounts of IL-1β, IL-2, IL-6, MIP-1α, TNFα and other cytokines, attracting inflammatory cells to the infected foci and causing hepatic edema, lysis, death and even abscess formation (28).
In summary, the results of the present study suggest that mice with DM have a higher incidence of KPLA due to the excessive activation of NFκB cell signaling pathway proteins, increased inflammation and increases liver cell necrosis. However, further investigation is required to validate these results and investigate additional factors that may contribute to KPLA.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Authors' contributions
XL, LP, MZ and ML generated and analyzed the data. DX, CC, RS, JL and SH designed the experiments and drafted the manuscript. All authors approved the final version of the manuscript.
Ethics approval and consent to participate
All experiments were approved by the Ethics Committee of the Tenth People's Hospital of Tongji University (Shanghai, China) and performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (National Institutes of Health Bethesda, MD, USA). All mice were anesthetized by intraperitoneal injection of sodium pentobarbital (30 mg/kg) prior to euthanasia.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Chung DR, Lee SS, Lee HR, Kim HB, Choi HJ, Eom JS, Kim JS, Choi YH, Lee JS, Chung MH, et al. Emerging invasive liver abscess caused by K1 serotype Klebsiella pneumoniae in Korea. J Infect. 2007;54:578–583. doi: 10.1016/j.jinf.2006.11.008. [DOI] [PubMed] [Google Scholar]
- 2.Yeh KM, Kurup A, Siu LK, Koh YL, Fung CP, Lin JC, Chen TL, Chang FY, Koh TH. Capsular serotype K1 or K2, rather than magA and rmpA, is a major virulence determinant for Klebsiella pneumoniae liver abscess in Singapore and Taiwan. J Clin Microbiol. 2007;45:466–471. doi: 10.1128/JCM.01150-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Shi R, Jin Y, Cao C, Han S, Shao X, Meng L, Cheng J, Zhang M, Zheng J, Xu J, Li M. Localization of human adipose-derived stem cells and their effect in repair of diabetic foot ulcers in rats. Stem Cell Res Ther. 2016;7:155. doi: 10.1186/s13287-016-0412-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wu JH, Tsai CG. Infectivity of hepatic strain Klebsiella pneumoniae in diabetic mice. Exp Biol Med (Maywood) 2005;230:757–761. doi: 10.1177/153537020523001009. [DOI] [PubMed] [Google Scholar]
- 5.Pozzilli P, Leslie RD. Infections and diabetes: Mechanisms and prospects for prevention. Diabet Med. 1994;11:935–941. doi: 10.1111/j.1464-5491.1994.tb00250.x. [DOI] [PubMed] [Google Scholar]
- 6.Chao CL, Chuang CP, Cheng YF, Lee KR, Chang Y, Cheng SP, Chan WK, Ho FM. The protective role of autophagy in matrix metalloproteinase-mediated cell transmigration and cell death in high-glucose-treated endothelial cells. Inflammation. 2016;39:830–838. doi: 10.1007/s10753-016-0313-7. [DOI] [PubMed] [Google Scholar]
- 7.Weikel KA, Cacicedo JM, Ruderman NB, Ido Y. Knockdown of GSK3β increases basal autophagy and AMPK signalling in nutrient-laden human aortic endothelial cells. Biosci Rep. 2016;36:e00382. doi: 10.1042/BSR20160174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Huang YT, Jiang JY, Hsu MS, Hsu HS, Liao CH, Hsueh PR. The prevalence of rectal carriage of Klebsiella pneumoniae amongst diabetic patients and their clinical relevance in Taiwan: A five-year prospective study. J Microbiol Immunol Infect: S1684-1182(17)30119-6. 2017 doi: 10.1016/j.jmii.2017.05.009. [DOI] [PubMed] [Google Scholar]
- 9.Keynan Y, Rubinstein E. Diabetes mellitus and pyogenic liver abscess: Risk and prognosis. Clin Infect Dis. 2007;45:801. doi: 10.1086/521170. [DOI] [PubMed] [Google Scholar]
- 10.Lin YT, Wang FD, Wu PF, Fung CP. Klebsiella pneumoniae liver abscess in diabetic patients: Association of glycemic control with the clinical characteristics. BMC Infect Dis. 2013;13:56. doi: 10.1186/1471-2334-13-56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fung CP, Chang FY, Lin JC, Ho DM, Chen CT, Chen JH, Yeh KM, Chen TL, Lin YT, Siu LK. Immune response and pathophysiological features of Klebsiella pneumoniae liver abscesses in an animal model. Lab Invest. 2011;91:1029–1039. doi: 10.1038/labinvest.2011.52. [DOI] [PubMed] [Google Scholar]
- 12.Cohen J. The immunopathogenesis of sepsis. Nature. 2002;420:885–891. doi: 10.1038/nature01326. [DOI] [PubMed] [Google Scholar]
- 13.Lin YT, Liu CJ, Yeh YC, Chen TJ, Fung CP. Ampicillin and amoxicillin use and the risk of Klebsiella pneumoniae liver abscess in Taiwan. J Infect Dis. 2013;208:211–217. doi: 10.1093/infdis/jit157. [DOI] [PubMed] [Google Scholar]
- 14.Hasegawa M, Yamazaki T, Kamada N, Tawaratsumida K, Kim YG, Núñez G, Inohara N. Nucleotide-binding oligomerization domain 1 mediates recognition of Clostridium difficile and induces neutrophil recruitment and protection against the pathogen. J Immunol. 2011;186:4872–4880. doi: 10.4049/jimmunol.1003761. [DOI] [PubMed] [Google Scholar]
- 15.Wu MF, Yang CY, Lin TL, Wang JT, Yang FL, Wu SH, Hu BS, Chou TY, Tsai MD, Lin CH, Hsieh SL. Humoral immunity against capsule polysaccharide protects the host from magA+ Klebsiella pneumoniae-induced lethal disease by evading Toll-like receptor 4 signaling. Infect Immun. 2009;77:615–621. doi: 10.1128/IAI.00931-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Guo N, Xu Y, Cao Z. Absinthin attenuates LPS-induced ALI through MIP-1α-mediated inflammatory cell infiltration. Exp Lung Res. 2015;41:514–524. doi: 10.3109/01902148.2015.1093566. [DOI] [PubMed] [Google Scholar]
- 17.Gerondakis S, Grumont R, Gugasyan R, Wong L, Isomura I, Ho W, Banerjee A. Unravelling the complexities of the NF-kappaB signalling pathway using mouse knockout and transgenic models. Oncogene. 2006;25:6781–6799. doi: 10.1038/sj.onc.1209944. [DOI] [PubMed] [Google Scholar]
- 18.Vallabhapurapu S, Karin M. Regulation and function of NF-kappaB transcription factors in the immune system. Annu Rev Immunol. 2009;27:693–733. doi: 10.1146/annurev.immunol.021908.132641. [DOI] [PubMed] [Google Scholar]
- 19.Cho YC, Kim BR, Le HTT, Cho S. Antiinflammatory effects on murine macrophages of ethanol extracts of Lygodium japonicum spores via inhibition of NFκB and p38. Mol Med Rep. 2017;16:4362–4370. doi: 10.3892/mmr.2017.7070. [DOI] [PubMed] [Google Scholar]
- 20.Yadav N, Chandra H. Suppression of inflammatory and infection responses in lung macrophages by eucalyptus oil and its constituent 1,8-cineole: Role of pattern recognition receptors TREM-1 and NLRP3, the MAP kinase regulator MKP-1, and NFκB. PLoS One. 2017;12:e0188232. doi: 10.1371/journal.pone.0188232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hayden MS, Ghosh S. NF-κB, the first quarter-century: Remarkable progress and outstanding questions. Genes Dev. 2012;26:203–234. doi: 10.1101/gad.183434.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Giménez-Scherer JA, Cárdenas G, López-Osuna M, Velázquez JR, Rico G, Isibasi A, Mdel Maldonado C, Morales ME, Fernández-Diez J, Kretschmer RR. Immunization with a tetramer derivative of an anti-inflammatory pentapeptide produced by Entamoeba histolytica protects gerbils (Meriones unguiculatus) against experimental amoebic abscess of the liver. Parasite Immunol. 2004;26:343–349. doi: 10.1111/j.0141-9838.2004.00718.x. [DOI] [PubMed] [Google Scholar]
- 23.Hoffmann A, Baltimore D. Circuitry of nuclear factor kappaB signaling. Immunol Rev. 2006;210:171–186. doi: 10.1111/j.0105-2896.2006.00375.x. [DOI] [PubMed] [Google Scholar]
- 24.Liu F, Xia Y, Parker AS, Verma IM. IKK biology. Immunol Rev. 2012;246:239–253. doi: 10.1111/j.1600-065X.2012.01107.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hoffmann A, Levchenko A, Scott ML, Baltimore D. The IkappaB-NF-kappaB signaling module: Temporal control and selective gene activation. Science. 2002;298:1241–1245. doi: 10.1126/science.1071914. [DOI] [PubMed] [Google Scholar]
- 26.Lin CH, Cheng HW, Ma HP, Wu CH, Hong CY, Chen BC. Thrombin induces NF-kappaB activation and IL-8/CXCL8 expression in lung epithelial cells by a Rac1-dependent PI3K/Akt pathway. J Biol Chem. 2011;286:10483–10494. doi: 10.1074/jbc.M110.112433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Regueiro V, Moranta D, Campos MA, Margareto J, Garmendia J, Bengoechea JA. Klebsiella pneumoniae increases the levels of Toll-like receptors 2 and 4 in human airway epithelial cells. Infect Immun. 2009;77:714–724. doi: 10.1128/IAI.00852-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Duseja A, Singh SP, Saraswat VA, Acharya SK, Chawla YK, Chowdhury S, Dhiman RK, Jayakumar RV, Madan K, Misra SP, et al. Non-alcoholic fatty liver disease and metabolic syndrome-position paper of the indian national association for the study of the liver, endocrine society of India, Indian college of cardiology and indian society of gastroenterology. J Clin Exp Hepatol. 2015;5:51–68. doi: 10.1016/j.jceh.2015.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.