Abstract
Objective
The aim of this study is to evaluate the effects of admission glycemic variability (AGV) on in-hospital outcomes in diabetic patients with non-ST segment elevation acute coronary syndrome (NSTE-ACS) undergoing percutaneous coronary intervention (PCI).
Methods
We studied 759 diabetic patients with NSTE-ACS undergoing PCI. AGV was accessed based on the mean amplitude of glycemic excursions (MAGEs) in the first 24 hours after admission. Primary outcome was a composite of in-hospital events, all-cause mortality, new-onset myocardial infarction, acute heart failure, and stroke. Secondary outcomes were each of these considered separately. Predictive effects of AGV on the in-hospital outcomes in patients were analyzed.
Results
Patients with high MAGE levels had significantly higher incidence of total outcomes (9.9% vs. 4.8%, p=0.009) and all-cause mortality (2.3% vs. 0.4%, p=0.023) than those with low MAGE levels during hospitalization. Multivariable analysis revealed that AGV was significantly associated with incidence of in-hospital outcomes (Odds ratio=2.024, 95% CI 1.105-3.704, p=0.022) but hemoglobin A1c (HbA1c) was not. In the receiver-operating characteristic curve analysis for MAGE and HbA1c in predicting in-hospital outcomes, the area under the curve for MAGE (0.608, p=0.012) was superior to that for HbA1c (0.556, p=0.193).
Conclusion
High AGV levels may be closely correlated with increased in-hospital poor outcomes in diabetic patients with NSTE-ACS following PCI.
Keywords: glycemic variability, diabetes, acute coronary syndrome, risk factor
Introduction
Glycometabolic disturbances have been associated with increased morbidity and mortality in patients with acute coronary syndrome (ACS) (1). Some studies show that admission glycometabolic disturbances are of independent prognostic value with regard to future cardiovascular complications in patients with ACS, irrespective of their diabetes status (2, 3). Glycemic variability (GV) is also one component of dysglycemia, which refers to multiple fluctuations of glycemia that occur throughout the day or for even over longer periods of time. Several lines of evidence show that increased GV carries a significant risk of short-term and long-term adverse outcomes (4, 5). Higher GV is associated with longer length of hospital stay, infections, and in-hospital mortality (6, 7). Increasing GV is associated with retinopathy, nephropathy, cardiovascular events, and possibly mortality (8-11) and may be an independent risk predictor when compared with hemoglobin A1c (HbA1c) levels alone (5, 10). However, the predictive value of admission glycemic variability (AGV) for in-hospital outcomes in diabetic patients with ACS remains unclear. In this study, we investigated the prognostic value of AGV for in-hospital major adverse cardiac events (MACE) in diabetic patients with non-ST segment elevation acute coronary syndrome (NSTE-ACS) undergoing percutaneous coronary intervention (PCI).
Methods
Study population
Overall, 759 type 2 diabetes mellitus (T2DM) patients with NSTE-ACS who underwent elective PCI were enrolled in the study from January 2015 to December 2016; the baseline clinical data, including the admission mean amplitude of glycemic excursion (MAGE), blood glucose, and HbA1c levels, were recorded. The inclusion criteria included 1) confirmed admission diagnosis of NSTE-ACS and T2DM, 2) admission glucose level <16.7 mmol/L, and 3) no diabetic ketosis or nonketotic hyperosmolar coma. NSTE-ACS was defined as ischemic symptoms within 24 hours of presentation lasting for at least 10 minutes, combined with high-risk features such as ischemic ST segment electrocardiographic changes (ST depression ≥0.5 mm, transient ST elevation of 0.5-10 mm lasting for <10 minutes) and/or positive cardiac biomarkers (elevated troponin I or T and/or creatine kinase-MB>upper limit of normal) within 24 hours of hospital admission. T2DM was diagnosed according to the American Diabetes Association criteria or the use of insulin or glucose-lowering medication. The exclusion criteria were severe non-cardiac disease with expected survival of less than 3 months and unwillingness to participate. Patients were categorized according to MAGE levels (<3.9 and ≥3.9 mmol/L) based on reference values for continuous glucose monitoring in Chinese subjects (12). The study protocol was approved beforehand by the Medical Ethics Committee of Beijing An Zhen Hospital of Capital Medical University, and the procedures followed were in accordance with the institutional guidelines. The study complied with the declaration of Helsinki, and informed consent was obtained from all patients.
Continuous glucose monitoring
All patients were equipped with a continuous glucose monitoring system (CGMS, Medtronic MiniMed, USA) and monitored for 24-72 consecutive hours after admission. A CGMS sensor was inserted into the subcutaneous abdominal fat tissue, calibrated according to the standard Medtronic MiniMed operating guidelines. During CGMS monitoring, patients were checked for their blood glucose level with a self-monitoring of blood glucose (SMBG) device (Medisafe Mini, Terumo, Japan) at least 4 times per day. Then, the SMBG data and time of each meal were entered into CGMS. After monitoring, the recorded data were downloaded onto a personal computer for analysis of the glucose profile and glycemic excursion parameters with MiniMed Solutions software. MAGE was calculated from the first 24 hours of recording. Because the measurable range of glucose by CGMS was mechanically limited from 2.2 to 22.2 mmol/L, patients showing values beyond this range were excluded from the study. MAGE was calculated by measuring the arithmetic mean of the differences between consecutive peaks and nadirs, provided that the differences were greater than one standard deviation of the mean glucose value. If patients did not maintain anti-hyperglycemic therapy as usual and avoid glucose infusion during CGMS monitoring period, they would have been excluded from the study.
Coronary intervention
All enrolled patients underwent subsequent PCI when indicated as part of the routine treatment for ACS. Coronary intervention was performed using standard techniques, including percutaneous transluminal coronary angioplasty, intracoronary stenting, and/or mechanical rotational atherectomy. The PCI strategy was at the operator’s discretion. All patients received aspirin (100-300 mg) and clopidogrel (300-600 mg) before or on admission and were treated with aspirin (100 mg) and clopidogrel (75 mg) daily after PCI (13). Other adjunctive pharmacotherapies were administered at the discretion of the operator.
In-hospital MACE
All patients meeting criteria for this analysis were invited to participate in the study after informed consent was obtained from the patient or a family member. During the in-hospital period, incidences of MACE were registered, including all-cause mortality, new-onset myocardial infarction, acute heart failure, and stroke. Secondary outcomes were each of these conditions considered separately. All MACE data were adjudicated by an experienced cardiovascular physician blinded to clinical details and outcomes.
Statistical analysis
Normally distributed variables were presented as mean±SD and compared using independent samples t-test. Non-normally distributed variables were expressed in medians with interquartile range, and Mann-Whitney U test was used to determine significant differences among the groups. Categorical variables were expressed in frequencies and percentages and compared using chi-square test. To ascertain the independent contribution of MAGE to MACE, multiple regression analysis was performed. Variables adjusted in the model were age ≥65 years, sex male, body mass index (BMI) ≥ 30 kg/m2, HbA1c ≥7%, hypertension, hyperlipidemia, diabetes duration ≥60 months, current smoker, previous myocardial infarction (MI), previous PCI, previous coronary artery bypass graft surgery (CABG), left ventricular ejection fraction (LVEF) ≤ 40%, renal insufficiency (estimated glomerular filtration rate, eGFR<60 ml/min/1.73 m2), and positive cardiac biomarkers. Cox proportional hazards model and receiver-operating characteristic (ROC) curve were used to determine independent predictors of in-hospital MACE. Hazard ratios with 95% confidence intervals (CI) were determined. A value of p<0.05 was considered statistically significant. All statistical analyses were performed using SPSS for Windows 19.0 (SPSS Inc, Chicago, IL, USA).
Results
Baseline characteristics
During the study period, 759 patients with complete data were enrolled. Mean age was 62.8±9.5 years, and 61.3% were male. MAGE level was <3.9 mmol/L in 496 patients (65.3%) and ≥3.9 mmol/L in 263 patients (34.7%). The GRACE risk score ranged from 60 to 237, with a mean of 136±38. The patients with a high MAGE level were older and had longer duration of diabetes, higher GRACE scores, and BMI and lower LVEF and eGFR values than those with a low MAGE level. Baseline characteristics of patient groups based on the presence of MACE are shown in Table 1.
Table 1.
Baseline characteristics of diabetic patients with NSTE-ACS based on MAGE levels
| Variables | MAGE (mmol/L) | P | |
|---|---|---|---|
| <3.9 | ≥3.9 | ||
| n | 496 | 263 | |
| Patient demographics | |||
| Age (years) | 62 (34, 85) | 64 (39, 87) | 0.004 |
| Males | 177 (61.0) | 107 (63.3) | 0.690 |
| Medical history | |||
| Prior MI | 74 (14.9) | 52 (19.8) | 0.101 |
| Prior PCI | 91 (18.3) | 61 (23.3) | 0.127 |
| Prior CABG | 39 (7.9) | 31 (11.8) | 0.087 |
| Duration of DM (months) | 25 (0.2, 288) | 38 (0.4, 300) | <0.001 |
| Risk factors | |||
| Hypertension | 389 (78.4) | 205 (77.9) | 0.926 |
| Hyperlipidemia | 236 (47.6) | 132 (50.2) | 0.542 |
| Current smoking | 273 (55.0) | 151 (57.4) | 0.540 |
| BMI (kg/m2) | 26.2 (19.4, 40.3) | 26.6 (21.5, 38.9) | 0.027 |
| LVEF (%) | 54.39±11.35 | 51.55±10.89 | <0.001 |
| eGFR (ml/min/1.73 m2) | 74.46±28.86 | 67.84±19.77 | <0.001 |
| Peak CK (U/L) | 302.4 (9, 3025) | 309.5 (15, 2119) | 0.653 |
| TG (mmol/L) | 2.01 (0.57, 11.42) | 2.25 (0.72, 13.28) | 0.056 |
| TC (mmol/L) | 4.75 (2.53, 8.61) | 4.81 (2.61, 9.62) | 0.145 |
| LDL-C (mmol/L) | 2.67 (2.27, 3.27) | 2.82 (2.40, 3.38) | 0.105 |
| HDL-C (mmol/L) | 1.02 (0.88, 1.21) | 1.00 (0.89, 1.12) | 0.528 |
| ABG (mmol/L) | 8.18±3.28 | 8.87±3.06 | <0.001 |
| HbA1c (%) | 6.54±1.50 | 7.34±1.51 | <0.001 |
| Statin therapy | 453 (91.3) | 243 (92.4) | 0.613 |
| Hypoglycemic agents | |||
| Insulin secretagogues | 192 (38.7) | 109 (41.4) | 0.483 |
| Metformin | 258 (52.0) | 149 (56.7) | 0.251 |
| Insulin sensitizers | 104 (21.0) | 64 (24.3) | 0.312 |
| Glucosidase inhibitors | 304 (61.3) | 152 (57.8) | 0.351 |
| Insulin | 202 (40.7) | 119 (45.2) | 0.247 |
| PCI data | |||
| Culprit vessel, LAD | 209 (42.1) | 120 (45.6) | 0.357 |
| Multi-vessel CAD | 237 (47.8) | 140 (53.2) | 0.170 |
| TIMI grade 3 before PCI | 390 (78.6) | 202 (76.8) | 0.581 |
| TIMI grade 3 after PCI | 477 (96.2) | 244 (92.8) | 0.053 |
| Stents | 1.7±1.2 | 1.9±1.3 | 0.022 |
| GRACE Score | 132±37 | 143±35 | <0.001 |
ABG - admission blood glucose; BMI - body mass index; CABG - coronary artery bypass graft surgery; CAD - coronary artery disease; CK - creatine kinase; DM - diabetes mellitus; eGFR - estimated glomerular filtration rate; HDL-C - high-density lipoprotein cholesterol; HbA1c - hemoglobin A1c; NSTE-ACS - non-ST segment elevation acute coronary syndrome; MAGE - mean amplitude of glycemic excursion; MI - myocardial infarction; PCI - percutaneous coronary intervention; LVEF - left ventricular ejection fraction; TG - triglyceride; TC - total cholesterol; LDL-C - low-density lipoprotein cholesterol; LAD - left anterior descending artery. Data are mean±SD, median, and number (%)
Incidences of MACE
In total, 48 patients experienced an adverse cardiac event. Six patients died (0.8%), 12 patients had new-onset MI (1.6%), 21 patients experienced acute heart failure (2.8%), and 9 patients had stroke (1.2%). Compared with the low-MAGE group, the high-MAGE group had a higher all-cause mortality (2.3% vs. 0.4%, p=0.023) and incidence of total MACE (9.9% vs. 4.8%, p=0.009). Differences in rates of new-onset infarction (1.9% vs. 1.4%), acute heart failure (3.8% vs. 1.4%), and stroke (1.9% vs. 0.8%) were not statistically significant between the high- and low-MAGE groups (all p>0.05) (Fig. 1).
Figure 1.
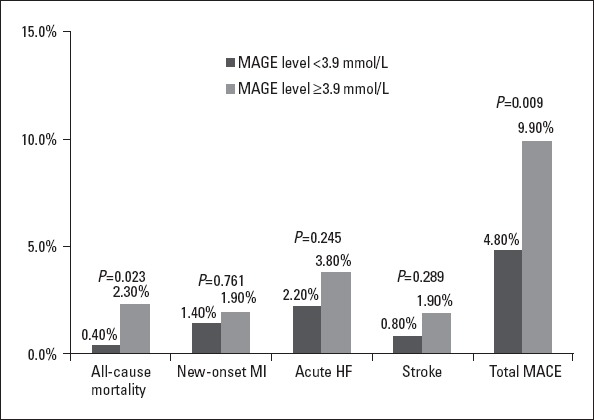
Comparison of incidences of in-hospital MACE between two MAGE groups. Patients with a higher MAGE level had a higher incidence of total MACE (black bars: MAGE level <3.9 mmol/L; grey bars: MAGE level ≥3.9 mmol/L)
Multiple analysis
Cox proportional hazard regression model was used to investigate the associations of MAGE with incidences of in-hospital MACE with adjustment for age, sex, CVD risk factors, and complications. The analysis showed that age ≥65 years, previous MI, LVEF ≤40%, positive cardiac biomarkers, and MAGE ≥3.9 mmol/L were significantly associated with in-hospital MACE. Significant predictors are presented in Table 2.
Table 2.
Multiple Cox proportional hazard regression models for total in-hospital MACE
| Variables | Total in-hospital MACE | ||
|---|---|---|---|
| OR | 95% CI | P | |
| Age ≥65 years | 1.982 | 1.033-3.915 | 0.049 |
| MAGE ≥3.9 mmol/L | 2.024 | 1.105-3.704 | 0.022 |
| LVEF ≤ 40% | 2.227 | 1.072-4.630 | 0.032 |
| Prior MI | 3.259 | 1.341-7.923 | 0.009 |
| Positive cardiac biomarkers | 2.695 | 1.182-6.135 | 0.018 |
CI – confidence interval; LVEF- left ventricular ejection fraction; MACE - major adverse cardiac events; MAGE - the mean amplitude of glycemic excursions; MI - myocardial infarction; OR – odds ratio
ROC curve analysis
ROC analysis was used to discriminate the distributions of MAGE and HbA1c in predicting in-hospital MACE. The area under the ROC curve for MAGE (0.608, 95% CI 0.524-0.692, p=0.012) was superior to that for HbA1c (0.556, 95% CI 0.475-0.637, p=0.193) (Fig. 2). MAGE, but not HbA1c, displayed significant value in predicting in-hospital outcomes in patients.
Figure 2.
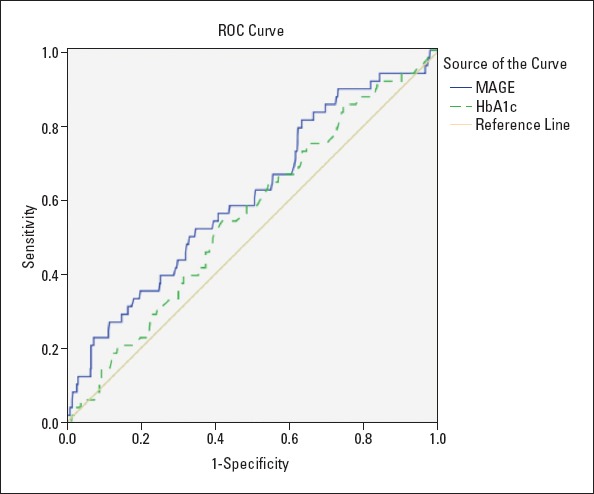
Area under the receiver-operating characteristic (ROC) curve in predicting in-hospital MACE: MAGE (0.608, 95% CI 0.524-0.692, P=0.012); HbA1c (0.556, 95% CI 0.475-0.637, P=0.193)
Discussion
GV involves two aspects: acute (short term) glucose fluctuations and chronic (long term) glucose variability. Long-term GV as assessed by variability of HbA1c levels has been reported to be a risk factor for all-cause mortality in patients with type 2 diabetes (11, 14). The Verona Diabetes study reported that fasting GV is an independent predictor of mortality in T2DM patients (15). However, the relationship between AGV and in-hospital outcomes has not been fully evaluated. In this study, our data analysis demonstrated that an elevated MAGE level (defined as a MAGE level ≥3.9 mmol/L) on admission is associated with a significantly higher risk of all-cause mortality and total in-hospital MACE after elective PCI in diabetic patients with NSTE-ACS. These results indicate that greater AGV may have an important prognostic significance in diabetic patients with ACS.
Increasing evidence is available regarding GV possibly playing an important role in resolving potential cardiovascular problems in diabetes. In the Action to Control Cardiovascular Risk in Diabetes study, retrospective analysis showed that reduction of HbA1c, targeting near normoglycemia, failed to decrease mortality, for which hypoglycemia was not fully responsible (16). It seems that increased GV has a disadvantageous effect on survival. In our previous study, we found that acute glucose excursions seem to be of greater importance than admission glucose and long-term derangements of glucose metabolism in predicting 1-year outcomes following AMI (5). Some studies concluded that short-term GV was a significant predictor of mortality in critically ill patients independently from mean glucose level and severity of illness (17-19). In the present study, significantly higher all-cause mortality and incidence of total in-hospital MACE were found in patients with a higher admission MAGE level. Multivariate analysis disclosed that MAGE was associated with in-hospital MACE but HbA1c was not. The results indicate that acute glucose fluctuations may be an important risk factor of in-hospital MACE in diabetic patients with NSTE-ACS.
Although we did not address the underlying mechanisms of the relationship between GV and cardiovascular outcomes in diabetic patients with ACS, both oxidative stress and inflammation may be involved in the association between acute GV and the outcomes. As a well-known pathogenic factor in diabetes complications, oxidative stress is correlated with glycemic excursions in subjects with type 2 diabetes (20, 21). Some studies indicate that glycemic fluctuation has been shown to cause an increase in inflammatory cytokines and monocyte and macrophage adhesion to endothelial cells in animals and humans (22, 23). Additionally, Quagliaro et al. (24) found that the exposure to intermittent high glucose level leads to apoptosis of endothelial cells. These findings suggest that glucose fluctuations augment inflammation via oxidative mechanisms closely linked to adverse outcomes. Some studies showed that glucose excursions were independently related to the development of atherosclerosis in individuals with T2DM (25, 26). In another previous study, we found that GV is an important contributing factor in the presence and severity of coronary artery disease, which is independent of the average level of blood glucose (27). In this study, more stents were implanted in patients with a high MAGE level than in those with a low MAGE level (1.9 vs. 1.7, p=0.022). The rate of TIMI grade 3 flow after PCI in the high-MAGE group showed a trend toward lower than that in the low-MAGE group (92.8% vs. 96.2%, p=0.053). Meanwhile, patients with a high MAGE level have higher GRACE scores, the worse heart function, and renal insufficiency. These data indicate that patients with higher GV may have severer cardiovascular conditions. Furthermore, severe glycemic excursions may adversely affect sympathetic dysfunction and increase the thrombotic properties of platelets, which can result in additional cardiovascular mortality and morbidity (28, 29).
Several well-conducted studies demonstrated that patients with persistent hyperglycemia tend to suffer from the worse long-term outcomes. However, our study shows that increased acute GV should be more important in predicting in-hospital outcomes of diabetic patients with NSTE-ACS. The analysis shows that high MAGE level was a significant predictor of the presence of MACE but HbA1c was not. In the ROC curve analysis for MAGE and HbA1c for predicting in-hospital outcomes, the area under the ROC curve for MAGE (0.608, p=0.012) was superior to that for HbA1c (0.556, p=0.193). Increased HbA1c represents abnormal long-term glucose regulation, whereas elevated admission GV is not only a symptom of glucose dysregulation but also that of stress and general poor health. There was a clear association between HbA1c and long-term outcomes in AMI patients after a 3.3-year follow-up (30). Thus, HbA1c may have limited predictive capability pertaining to short-term prognosis in patients, but its association with long-term prognosis may be stronger.
Several study limitations should be considered in the interpretation of the results. First, this is a single-center study, and it is uncertain whether our findings can be generalized to other centers or hospitals. Second, due to the lack of microvascular complications data, we did not include these risk factors in the analysis. Third, the sample size was relatively small; thus, some subgroup comparisons may have lacked the power to detect significant differences for selected variables. In addition, although we had maintained patients’ anti-hyperglycemic therapy as usual and avoided glucose infusion during CGMS monitoring, some other factors, such as different diets and physical and emotional factors, which may affect glucose fluctuations could not be prevented. Hence, we think that the results of the present study should be interpreted with caution. This study is hypothesis-generating and should stimulate a larger multicenter evaluation.
Conclusion
Although the detailed underlying mechanism is unclear to date, our findings suggest the importance of stabilization of blood glucose level in diabetic patients, especially in those with ACS, to prevent in-hospital adverse cardiac events. In diabetic patients with NSTE-ACS who underwent PCI, AGV seems to be of greater importance than HbA1c in predicting in-hospital poor outcomes. The results of this study further support the view that GV in ACS patients may be an important marker for risk stratification while potentially influencing therapeutic strategies.
Footnotes
Conflict of interest: None declared.
Peer-review: Externally peer-reviewed.
Authorship contributions: Concept – G.S., S.M.; Design – G.S., T.Z.; Supervision – G.S., S.M.; Fundings – This work was supported by a key grant from Beijing Health Special Foundation (JING 15-10). Materials – G.S., H.Y., W.D.; Data collection &/or processing – T.Z., H.Y., W.D., L.T.; Analysis &/or interpretation – G.S., T.Z., H.Y.; Literature search – G.S., H.Y.; Writing – G.S., T.Z.; Critical review – H.T., T.W., S.M.
References
- 1.Deedwania P, Kosiborod M, Barrett E, Ceriello A, Isley W, Mazzone T, et al. American Heart Association Diabetes Committee of the Council on Nutrition, Physical Activity, and Metabolism. Hyperglycemia and acute coronary syndrome:a scientific statement from the American Heart Association Diabetes Committee of the Council on Nutrition, Physical Activity, and Metabolism. Circulation. 2008;117:1610–9. doi: 10.1161/CIRCULATIONAHA.107.188629. [DOI] [PubMed] [Google Scholar]
- 2.Stranders I, Diamant M, van Gelder RE, Spruijt HJ, Twisk JW, Heine RJ, et al. Admission blood glucose level as risk indicator of death after myocardial infarction in patients with and without diabetes mellitus. Arch Intern Med. 2004;164:982–8. doi: 10.1001/archinte.164.9.982. [DOI] [PubMed] [Google Scholar]
- 3.Kosiborod M, Rathore SS, Inzucchi SE, Masoudi FA, Wang Y, Havranek EP, et al. Admission glucose and mortality in elderly patients hospitalized with acute myocardial infarction:implications for patients with and without recognized diabetes. Circulation. 2005;111:3078–86. doi: 10.1161/CIRCULATIONAHA.104.517839. [DOI] [PubMed] [Google Scholar]
- 4.Mi SH, Su G, Yang HX, Zhou Y, Tian L, Zhang T, et al. Comparison of in-hospital glycemic variability and admission blood glucose in predicting short-term outcomes in non-diabetes patients with ST elevation myocardial infarction underwent percutaneous coronary intervention. Diabetol Metab Syndr. 2017;9:20. doi: 10.1186/s13098-017-0217-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Su G, Mi SH, Tao H, Li Z, Yang HX, Zheng H, et al. Impact of admission glycemic variability, glucose, and glycosylated hemoglobin on major adverse cardiac events after acute myocardial infarction. Diabetes Care. 2013;36:1026–32. doi: 10.2337/dc12-0925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mellbin LG, Malmberg K, Rydén L, Wedel H, Vestberg D, Lind M. The relationship between glycaemic variability and cardiovascular complications in patients with acute myocardial infarction and type 2 diabetes:a report from the DIGAMI 2 trial. Eur Heart J. 2013;34:374–9. doi: 10.1093/eurheartj/ehs384. [DOI] [PubMed] [Google Scholar]
- 7.Mendez CE, Mok KT, Ata A, Tanenberg RJ, Calles-Escandon J, Umpierrez GE. Increased glycemic variability is independently associated with length of stay and mortality in noncritically ill hospitalized patients. Diabetes Care. 2013;36:4091–7. doi: 10.2337/dc12-2430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zoppini G, Verlato G, Targher G, Bonora E, Trombetta M, Muggeo M. Variability of body weight, pulse pressure and glycaemia strongly predict total mortality in elderly type 2 diabetic patients. The Verona Diabetes Study. Diabetes Metab Res Rev. 2008;24:624–8. doi: 10.1002/dmrr.897. [DOI] [PubMed] [Google Scholar]
- 9.Kilpatrick ES, Rigby AS, Atkin SL. A1c variability and the risk of microvascular complications in type 1 diabetes:data from the diabetes control and complications trial. Diabetes Care. 2008;31:2198–202. doi: 10.2337/dc08-0864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wadén J, Forsblom C, Thorn LM, Gordin D, Saraheimo M, Groop PH Finnish Diabetic Nephropathy Study Group. A1c variability predicts incident cardiovascular events, microalbuminuria, and overt diabetic nephropathy in patients with type 1 diabetes. Diabetes. 2009;58:2649–55. doi: 10.2337/db09-0693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Skriver MV, Sandbæk A, Kristensen JK, Støvring H. Relationship of HbA1c variability, absolute changes in HbA1c, and all-cause mortality in type 2 diabetes:a Danish population-based prospective observational study. BMJ Open Diabetes Res Care. 2015;3:e000060. doi: 10.1136/bmjdrc-2014-000060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhou J, Li H, Ran X, Yang W, Li Q, Peng Y, et al. Reference values for continuous glucose monitoring in Chinese subjects. Diabetes Care. 2009;32:1188–93. doi: 10.2337/dc09-0076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Section of Interventional Cardiology of Chinese Society of Cardiology of Chinese Medical Association;Specialty Committee on Prevention and Treatment of Thrombosis of Chinese College of Cardiovascular Physicians;Editorial Board of Chinese Journal of Cardiology. [Chinese guideline for percutaneous coronary intervention (2016)] Zhonghua Xin Xue Guan Bing Za Zhi. 2016;44:382–400. doi: 10.3760/cma.j.issn.0253-3758.2016.05.006. [DOI] [PubMed] [Google Scholar]
- 14.Ma WY, Li HY, Pei D, Hsia TL, Lu KC, Tsai LY, et al. Variability in hemoglobin A1c predicts all-cause mortality in patients with type 2 diabetes. J Diabetes Complications. 2012;26:296–300. doi: 10.1016/j.jdiacomp.2012.03.028. [DOI] [PubMed] [Google Scholar]
- 15.Muggeo M, Zoppini G, Bonora E, Brun E, Bonadonna RC, Moghetti P, et al. Fasting plasma glucose variability predicts 10-year survival of type 2 diabetic patients:the Verona Diabetes Study. Diabetes Care. 2000;23:45–50. doi: 10.2337/diacare.23.1.45. [DOI] [PubMed] [Google Scholar]
- 16.Bonds DE, Miller ME, Bergenstal RM, Buse JB, Byington RP, Cutler JA, et al. The association between symptomatic severe hypoglycaemia and mortality in type 2 diabetes:retrospective epidemiological analysis of the ACCORD study. BMJ. 2010;340:b4909. doi: 10.1136/bmj.b4909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Krinsley JS. Glycemic variability:a strong independent predictor of mortality in critically ill patients. Crit Care Med. 2008;36:3008–13. doi: 10.1097/CCM.0b013e31818b38d2. [DOI] [PubMed] [Google Scholar]
- 18.Dossett LA, Cao H, Mowery NT, Dortch MJ, Morris JM, Jr, May AK. Blood glucose variability is associated with mortality in the surgical intensive care unit. Am Surg. 2008;74:679–85. doi: 10.1177/000313480807400802. [DOI] [PubMed] [Google Scholar]
- 19.Hirshberg E, Larsen G, Van Duker H. Alterations in glucose homeostasis in the pediatric intensive care unit:Hyperglycemia and glucose variability are associated with increased mortality and morbidity. Pediatr Crit Care Med. 2008;9:361–6. doi: 10.1097/PCC.0b013e318172d401. [DOI] [PubMed] [Google Scholar]
- 20.Ceriello A, Ihnat MA. Glycaemic variability:A new therapeutic challenge in diabetes and the critical care setting. Diabetic Med. 2010;27:862–7. doi: 10.1111/j.1464-5491.2010.02967.x. [DOI] [PubMed] [Google Scholar]
- 21.Monnier L, Mas E, Ginet C, Michel F, Villon L, Cristol JP, et al. Activation of oxidative stress by acute glucose fluctuations compared with sustained chronic hyperglycemia in patients with type 2 diabetes. JAMA. 2006;295:1681–7. doi: 10.1001/jama.295.14.1681. [DOI] [PubMed] [Google Scholar]
- 22.Saisho Y. Glycemic variability and oxidative stress:a link between diabetes and cardiovascular disease? Int J Mol Sci. 2014;15:18381–406. doi: 10.3390/ijms151018381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ceriello A, Esposito K, Piconi L, Ihnat MA, Thorpe JE, Testa R, et al. Oscillating glucose is more deleterious to endothelial function and oxidative stress than mean glucose in normal and type 2 diabetic patients. Diabetes. 2008;57:1349–54. doi: 10.2337/db08-0063. [DOI] [PubMed] [Google Scholar]
- 24.Quagliaro L, Piconi L, Assaloni R, Martinelli L, Motz E, Ceriello A. Intermittent high glucose enhances apoptosis related to oxidative stress in human umbilical vein endothelial cells:the role of protein kinase C and NAD(P)H-oxidase activation. Diabetes. 2003;52:2795–804. doi: 10.2337/diabetes.52.11.2795. [DOI] [PubMed] [Google Scholar]
- 25.Mo Y, Zhou J, Li M, Wang Y, Bao Y, Ma X, et al. Glycemic variability is associated with subclinical atherosclerosis in Chinese type 2 diabetic patients. Cardiovasc Diabetol. 2013;12:15. doi: 10.1186/1475-2840-12-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hu Y, Liu W, Huang R, Zhang X. Postchallenge plasma glucose excursions, carotid intima-media thickness, and risk factors for atherosclerosis in Chinese population with type 2 diabetes. Atherosclerosis. 2010;210:302–6. doi: 10.1016/j.atherosclerosis.2009.11.015. [DOI] [PubMed] [Google Scholar]
- 27.Su G, Mi S, Tao H, Li Z, Yang H, Zheng H, et al. Association of glycemic variability and the presence and severity of coronary artery disease in patients with type 2 diabetes. Cardiovasc Diabetol. 2011;10:19. doi: 10.1186/1475-2840-10-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Takei Y, Tomiyama H, Tanaka N, Yamashina A. Close relationship between sympathetic activation and coronary microvascular dysfunction during acute hyperglycemia in subjects with atherosclerotic risk factors. Circ J. 2007;71:202–6. doi: 10.1253/circj.71.202. [DOI] [PubMed] [Google Scholar]
- 29.Gresele P, Guglielmini G, De Angelis M, Ciferri S, Ciofetta M, Falcinelli E, et al. Acute, short-term hyperglycemia enhances shear stress-induced platelet activation in patients with type II diabetes mellitus. J Am Coll Cardiol. 2003;41:1013–20. doi: 10.1016/s0735-1097(02)02972-8. [DOI] [PubMed] [Google Scholar]
- 30.Timmer JR, Hoekstra M, Nijsten MW, van der Horst IC, Ottervanger JP, Slingerland RJ, et al. Prognostic value of admission glycosylated hemoglobin and glucose in nondiabetic patients with ST-segment-elevation myocardial infarction treated with percutaneous coronary intervention. Circulation. 2011;124:704–11. doi: 10.1161/CIRCULATIONAHA.110.985911. [DOI] [PubMed] [Google Scholar]


