Abstract
Previous clinical trials have addressed the beneficial effects of fish oil supplementation on atopic dermatitis. Recently, we reported that fat-1 mice, which can convert n-6 to n-3 polyunsaturated fatty acids (PUFAs), are protected against allergic airway inflammation because their Th2 immune responses are suppressed. Here, we examined the effects of endogenously synthesized n-3 PUFAs on atopic dermatitis, a representative Th2-dominant allergic inflammatory disease. Mouse models of atopic dermatitis-like skin lesions were prepared by epicutaneous application of 2,4-dinitrochlorobenzene (DNCB) or house dust mite (HDM) extract to the ears. DNCB-treated fat-1 mice exhibited markedly reduced epidermal thickening, lower mast cell infiltration, and lower serum IgE and histamine compared with wild-type mice. The draining lymph nodes of fat-1 mice were substantially smaller and contained significantly smaller proportions of activated CD4+ T cells and IL-4-producing Th2 cells than those of wild-type mice. Consistent with these findings, the mRNA levels of Th2 cytokines were significantly decreased in DNCB-sensitized skin lesions of fat-1 mice. Lastly, inflammasome activation, IL-1β production, and pyroptotic cell injury were suppressed in fat-1 mice. Similar results were observed in HDM-challenged fat-1 mice. This study confirms the results of previous clinical studies and suggests fish oil supplementation as a therapeutic strategy for atopic dermatitis-like skin lesions.
Subject terms: Experimental models of disease, Atopic dermatitis
Skin disease: Omega-3 fatty acid reduces inflammation
A component of fish oil reduces the severity of atopic dermatitis in mice by dampening the immune response. Omega-3 fatty acids found in fish oil are essential nutrients that humans and other mammals need but cannot produce. Byung-Hyun Park at Chonbuk National University Medical School, Jeonju, South Korea, and colleagues show that genetically engineered mice expressing a gene which enables them to synthesize their own omega-3 fatty acids are protected from atopic dermatitis. Following exposure to a dermatitis-inducing chemical or house dust mite extract these mice developed smaller skin lesions and mounted a weaker inflammatory response than wild-type mice. This study confirms the anti-inflammatory effects of omega-3 fatty acids and highlights the use of fish oil supplements as a useful strategy for the treatment of atopic dermatitis and potentially other chronic inflammatory diseases.
Introduction
Atopic dermatitis is a multifactorial chronic inflammatory disease that results from complex interactions between genetic and environmental factors1. Atopic dermatitis is also considered a T-helper 2 (Th2)-mediated allergic disease because interleukin (IL)-4, IL-5, and IL-13 produced by Th2 cells play key roles in its onset and development2–4. These cytokines stimulate B cells, mast cells, and epidermal cells, leading to IgE production and mast cell degranulation as well as the production of various cytokines. Thymic stromal lymphopoietin (TSLP), a cytokine produced mainly by epidermal keratinocytes and mast cells, promotes the differentiation of naїve T cells into Th2 cells5. TSLP is highly expressed in skin lesions of atopic dermatitis patients6,7. Interestingly, T cell polarization in atopic dermatitis is biphasic: there is a predominantly a Th2 response in the acute phase, while high levels of interferon (IFN)-γ and the appearance of Th1 cells have been observed in the chronic phase8. In addition to adaptive immune responses, the innate immune system is activated via evolutionarily conserved pathogen-recognition receptors, such as Toll-like receptors (TLRs) and nucleotide-binding oligomerization domain receptors (NLRs)6,9. The most well known of these is NLR family pyrin domain-containing protein 3 (NLRP3), which recruits apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC) and pro-caspase-1. Inflammasome assembly then activates caspase-1 and leads to the release of IL-1β and IL-1810. These ILs are important in innate immune functions and in the regulation of adaptive immune responses. Indeed, single-nucleotide polymorphisms (SNPs) in the Nlrp3 gene are associated with atopic dermatitis11–13.
Based on the pathology of atopic dermatitis, immunomodulating strategies have been developed to treat the disease. These strategies include corticosteroids, calcineurin inhibitors, and IgE–neutralizing antibodies14–16. However, prolonged use of these drugs often produces adverse effects in atopic dermatitis patients. Therefore, a safe and long-term therapeutic strategy is required. Several epidemiological studies have shown that dietary fish oil supplementation decreases the prevalence of atopic dermatitis and alleviates the severity of the disease17. Fish oil is an important source of n-3 polyunsaturated fatty acids (PUFAs), such as eicosapentaenoic acid (EPA, 20:5) and docosahexaenoic acid (DHA, C22:6). The incorporation of n-3 PUFAs into the membranes of immune cells, at the expense of arachidonic acid, is thought to be important for the anti-inflammatory effects of n-3 PUFAs18, which gives rise to fewer inflammatory eicosanoids. This shift ultimately affects cytokine production. n-3 PUFAs suppress T-cell activation19, monocyte chemotaxis20, macrophage M1 polarization21, and dendritic cell maturation22, resulting in a decrease in local tissue inflammation. However, it is difficult to ascertain the contribution of n-3 PUFAs per se without the potential confounding effects of other dietary components, such as the nutrient composition, total caloric intake, duration of feeding, and contamination of trace elements. To address this issue, Kang et al.23. developed fat-1 mice, which contain the fat-1 gene from Caenorhabditis elegans and are able to convert n-6 to n-3 PUFAs in vivo. As a result of the endogenously synthesized n-3 PUFAs in blood and tissues, these mice exhibit the beneficial effects of n-3 PUFAs in various inflammatory disease models, such as pancreatitis24, allergic airway inflammation19, and rheumatoid arthritis25. In this study, we investigated whether atopic dermatitis-like symptoms and skin lesions observed in 2,4-dinitrochlorobenzene (DNCB)- or house dust mite (HDM) extract-exposed mice would be alleviated in fat-1 mice. Special attention was paid to the effects exerted by endogenously synthesized n-3 PUFAs on inflammasome activation in skin tissues.
Materials and methods
Animals
Dr. JX Kang of Harvard Medical School (Boston, MA, USA) kindly provided the fat-1 transgenic mice. Mice were housed in a laminar flow cabinet and maintained on an AIN-76A diet containing 5% corn oil provided ad libitum. All animal experiments were performed in accordance with the Guide for the Care and Use of Laboratory Animals, published by the US National Institutes of Health (NIH Publication No. 85-23, revised 2011). The current study protocol was also approved by the Institutional Animal Care and Use Committee of Chonbuk National University (Permit No. CBNU 2017-0085).
Induction of experimental atopic dermatitis
To induce experimental atopic dermatitis-like skin lesions, the surfaces of both ears were very gently stripped five times with surgical tape (Tegaderm; 3 M, St Paul, MN, USA). Mice were sensitized by applying 20 μl of 1% DNCB (Sigma-Aldrich, St. Louis, MO, USA) or vehicle alone to the ear on Day -7. On Day 0, mice were challenged again by applying 20 μl of 0.5% DNCB to the ears every other day for up to 42 days. Changes in ear thickness were measured weekly. The mice were sacrificed on Day 43. In the HDM (Dermatophagoides farina extract, Greer Laboratories, Lenoir, NC, USA)-induced atopic dermatitis model, both surfaces of the ear lobes were very gently stripped three times with Tegaderm. After stripping, 20 μl of HDM (10 mg/ml) was painted on each ear. The application of HDM was repeated 3 times per week for 8 weeks. Two weeks after the first induction, blood was collected from the tail vein to measure serum IgE.
Histology
Ear tissues were fixed with 10% formalin and embedded in paraffin. Fixed tissues were cut into 5 μm sections and deparaffinized. Sections were stained with hematoxylin-eosin (H&E) or toluidine blue for light-microscopic examinations or mast cell infiltration assays, respectively. The epidermis and dermis thicknesses were measured using the iSolution DT36 software (Carl Zeiss, Oberkochen, Germany). The inflammation severity was scored as follows:26 0, none; 1, minimal; 2, mild; 3, moderate; and 4, marked. Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining was performed using a commercial kit (Promega, Madison, WI, USA). For confocal microscopic analysis, sections were stained with antibodies against caspase-1 (14F468) or ASC (N-15) (both from Santa Cruz Biotechnology). Images were acquired with an LSM510 confocal laser scanning microscope (Carl Zeiss).
Enzyme-linked immunosorbent assay (ELISA)
The levels of IgE, IgG1, IgG2a, IL-1β, TNF-α, IFN-γ IL-4, IL-13, IL-17 (eBioscience, San Diego, CA, USA), histamine (Enzo Life Sciences), high-mobility group box 1 (HMGB1, Chondrex, Redmond, WA, USA), and lactate dehydrogenase (LDH) activity (Biovision, Milpitas, CA, USA) in mouse serum were measured using commercial ELISA kits.
Cell surface staining and intracellular cytokine staining for flow cytometry
Single-cell suspensions prepared from draining lymph nodes (dLNs) were stained with anti-CD4-PerCP/Cy5.5 and anti-CD69-FITC antibodies (eBioscience) for 30 min on ice and washed with fluorescence-activated cell sorting buffer (2% FBS in PBS). The cell surface expression of CD69 and percentage of IL-4-producing CD4+ T cells were evaluated by flow cytometry on an Accuri flow cytometer (BD Biosciences, San Jose, CA, USA). All of the antibodies used were purchased from eBioscience.
Statistical analysis
Data are expressed as the means ± SEMs. Significant differences between groups were determined using Student’s unpaired t-test followed by Fisher’s post-hoc analysis. A value of p < 0.05 was accepted as an indication of statistical significance.
Results
Liquid chromatographic analysis shows increased n-3 PUFA content in fat-1 mice
We first quantified the major n-3 and n-6 PUFAs in skin tissues of fat-1 mice and their wild-type (WT) littermates. The n-3 PUFA (ALA, EPA, and DHA) content was significantly different between the genotypes, with higher levels in the serum and skin tissues of fat-1 mice (Table S1). Conversely, the n-6 PUFA (AA and LA) contents were lower in fat-1 mice compared with WT mice. Fat-1 mice had a skin tissue n-6/n-3 ratio of 5.6 compared to the ratio of 12.5 in WT mice.
Fat-1 gene overexpression ameliorates the symptoms of atopic dermatitis in mice
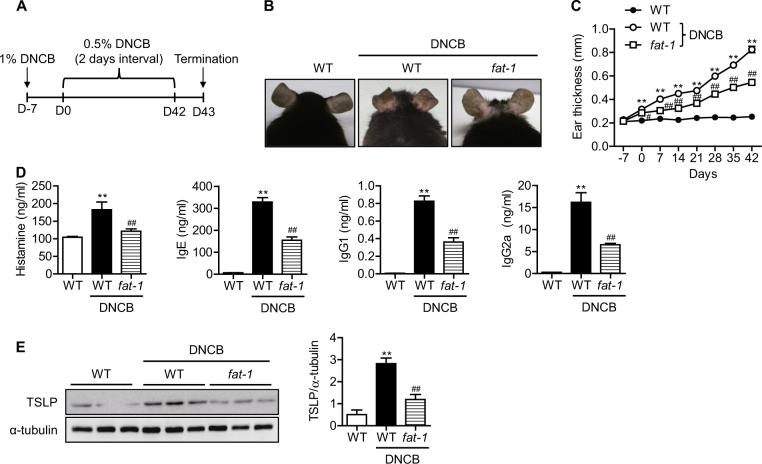
To validate the effects of endogenously synthesized n-3 PUFAs on atopic dermatitis, a DNCB-induced atopic dermatitis model was used (Fig. 1a). Following DNCB treatment, the ears of wild-type mice became red and swollen. On day 42, the ears of DNCB-treated wild-type mice showed severe erythema, erosion, and dryness (Fig. 1b). However, these atopic skin lesions were markedly attenuated in fat-1 mice. When ear thickness was measured over time and compared between genotypes, we found that thickness increased as atopic dermatitis developed. Consistent with the photographic images of the skin lesions, ear thickness was significantly reduced beginning on Day 7 (Fig. 1c). As IgE production, histamine release, and TSLP expression are markers for the development of atopic dermatitis, we also measured those levels in serum. As expected, fat-1 mice produced significantly less histamine, IgE, IgG1, IgG2a, and TSLP than wild-type mice at Day 43 (Fig. 1d, e). These data indicate that endogenously synthesized n-3 PUFAs effectively ameliorate atopic dermatitis-like skin lesions in mice.
Fig. 1. DNCB-induced atopic dermatitis-like symptoms in mice.
a A schematic diagram showing the induction of atopic dermatitis-like skin lesions by DNCB treatment in mice. b Representative pictures of mouse ears on Day 42. c Ear thickness was measured weekly from day 7 to 42 (n = 4). d Serum histamine, IgE, IgG1, and IgG2a in mice were measured by ELISA (n = 7–9). e TSLP in ear skin was measured by western blotting with densitometry (n = 3). Values are means ± SEMs. **p < 0.01 vs. WT; #p < 0.05 and ##p < 0.01 vs. WT+DNCB
Tissue inflammation and immune cell infiltration are diminished in fat-1 mice
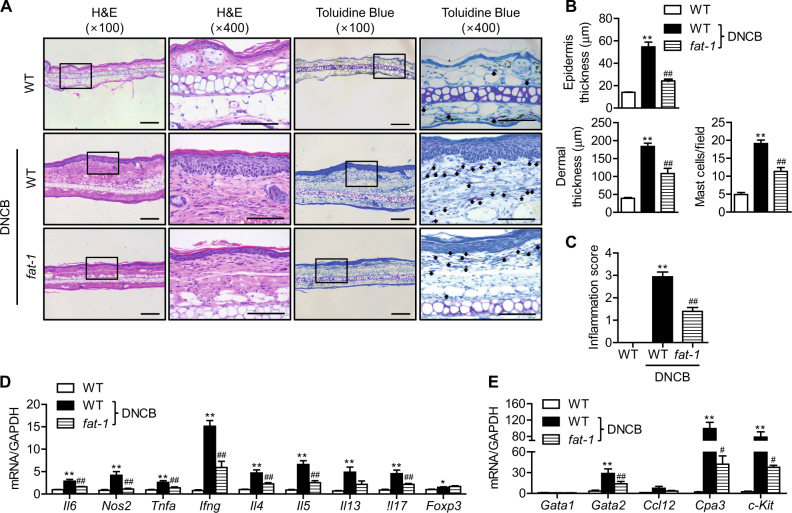
Improvements in the atopic symptoms of fat-1 mice were also confirmed by microscopic analysis of H&E-stained sections from ears removed at the end of the study. Wild-type mice under DNCB treatment exhibited the typical microscopic characteristics of atopic dermatitis, which include hyperkeratosis, parakeratosis, and acanthosis, along with infiltration of inflammatory cells (Fig. 2a, b). Toluidine blue staining of ear tissue sections revealed mast cell infiltration into the dermis of wild-type mice (Fig. 2a, b). However, in skin tissues from fat-1 mice, the epidermal and dermal tissues were significantly thinner and the infiltration of inflammatory cells into the dermis was significantly reduced. Accordingly, the inflammation score and mRNA levels of inflammation-related genes were changed (Fig. 2c, d). These results suggest that reductions in inflammation and mast cell infiltration in fat-1 mice might attenuate the symptoms of atopic dermatitis. Since histamine release was markedly downregulated in fat-1 skin tissues (Fig. 1d), we hypothesized that endogenously synthesized n-3 PUFAs improved atopic dermatitis-like skin symptoms through downregulation of mast cell function. To test this possibility, we focused on the transcription factors required for mast cell activation. Gene expression analysis showed that Gata2, but not Gata1, was markedly increased by DNCB treatment in WT mice and significantly downregulated in fat-1 mice (Fig. 2e). The expression of the GATA2 target genes Cpa3 and c-Kit was altered accordingly.
Fig. 2. Histological analysis of ear skin lesions.
Ears were excised from each group of mice on Day 43, fixed with formalin, embedded in paraffin, and cut into thin sections. The ear sections were stained with hematoxylin and eosin (H&E) and toluidine blue. Bars = 100 μm. a Representative photomicrographs of ear sections. The arrow indicates mast cells. b Epidermal and dermal thicknesses were measured in H&E-stained microphotographs, and mast cells were counted after toluidine blue staining. c The inflammation index was determined as described in the Methods (n = 9). d, e mRNA expression levels of inflammation-related genes (n = 7) and mast cell transcription factors (n = 3) were determined by real-time RT-PCR. Values are means ± SEMs. **p < 0.01 vs. WT; #p < 0.05 and ##p < 0.01 vs. WT+DNCB
T cell activation and Th2 cytokine production are suppressed in draining lymph nodes of fat-1 mice
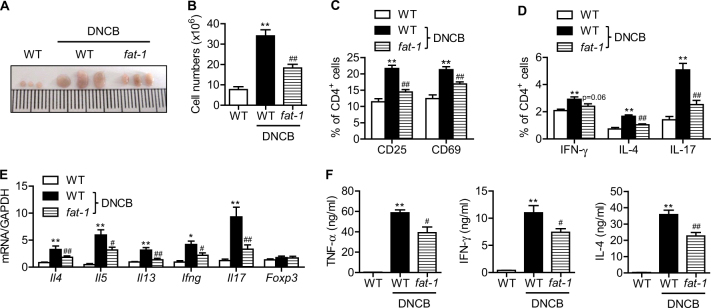
To investigate whether skin inflammation increased the number of Th2 cells in the dLNs of the ear, submandibular lymph nodes were collected. We first observed that the lymph nodes in fat-1 mice were substantially smaller than those from wild-type mice (Fig. 3a). Lymph nodes were homogenized, and single cells were prepared and counted. Consistent with the lymph node size, the total cell numbers were significantly decreased in the lymph nodes of fat-1 mice (Fig. 3b). Next, single cells prepared from lymph nodes were analyzed for T cell activation. Flow-cytometric analysis showed that the percentages of T cells expressing CD25+ and CD69+ were significantly lower in the dLNs of fat-1 mice (Fig. 3c), indicating that fat-1 expression suppressed DNCB-induced CD4+ T cell activation in dLNs. Moreover, CD4+ T cells from fat-1 mice showed lower percentages of Th1, Th2, and Th17 cells after DNCB treatment (Fig. 3d), but no difference in the percentage of Tregs (data not shown). Real-time RT-PCR analysis of cytokines in dLNs revealed an inhibition of CD4+ T cell differentiation into Th1, Th2, or Th17 in fat-1 mice (Fig. 3e). ELISA analysis in serum further revealed an inhibition of CD4+ T cell differentiation in fat-1 mice (Fig. 3f).
Fig. 3. T cell activation and differentiation in the draining lymph nodes.
a At the end of the study, submandibular lymph nodes were collected, and photographs were taken. b Single-cell suspensions were prepared from dLNs, and total cells were counted (n = 9). c CD4+ T cells were analyzed for CD25 and CD69 by flow cytometry (n = 9). d CD4+ T cells were stained for IL-4, IL-17, and IFN-γ and analyzed by flow cytometry. The percentage of CD4+ cells expressing each cytokine is shown (n = 9). e dLNs were homogenized, and the mRNA levels of cytokines were analyzed by real-time RT-PCR (n = 7). f Serum cytokines were measured by ELISA (n = 7). Values are means ± SEMs. **p < 0.01 vs. WT;#p < 0.05 and ##p < 0.01 vs. WT+DNCB
n-3 PUFAs alleviate NLRP3 inflammasome-mediated pyroptosis by inhibiting NF-κB activation
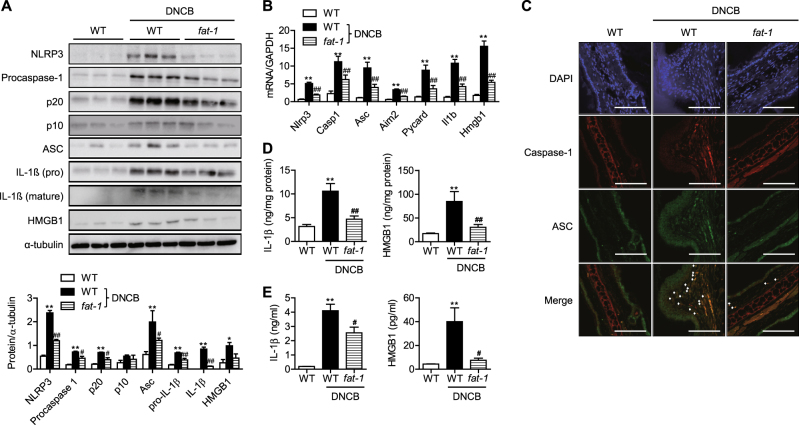
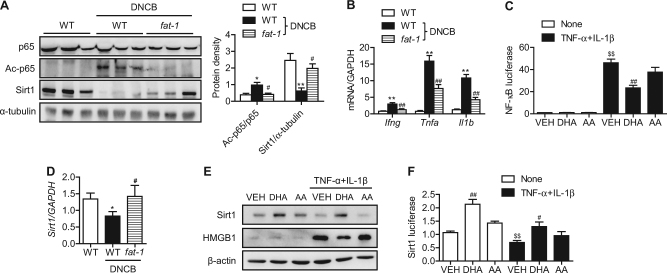
Since inflammasomes play important roles in skin inflammation, including atopic dermatitis27,28, we hypothesized that endogenously synthesized n-3 PUFAs would ameliorate the symptoms of atopic dermatitis by suppressing inflammasome activation. To test this hypothesis, we compared the protein and mRNA levels of NLRP3, ASC, and caspase-1, which are three key components of inflammasomes, in the skin of ears. NLRP3, procaspase-1, its proteolytic cleavage products p20 and p10, and ASC were markedly increased by DNCB treatment of WT mice (Fig. 4a, b). In agreement with the results of western blotting, immunofluorescence staining showed that p20 caspase-1 and ASC were elevated in DNCB-treated WT skin tissues (Fig. 4c). We further determined the protein levels of IL-1β and HMGB1, hallmarks of inflammasome activation. DNCB treatment increased the expression of IL-1β and HMGB1 in the skin tissues as well as their secretion into the systemic circulation (Fig. 4d, e). However, all of the aforementioned findings of inflammasome activation were remarkably suppressed in fat-1 mice.
Fig. 4. Inflammasome activation.
a, b At the end of the study, inflammasome components in the skin lesions were examined by western blotting and real-time RT-PCR. Protein levels were quantified by densitometry (n = 3). c Skin tissues were double-stained for caspase-1 and ASC. Signals are digitally merged in the lower panels. Bars = 50 μm. IL-1β and HMGB1 in skin tissues (d) and serum (e) were measured by ELISA (n = 5). Values are means ± SEMs. *p < 0.05 and **p < 0.01 vs. WT; #p < 0.05 and ##p < 0.01 vs. WT+DNCB
Activation of inflammasomes culminates in a special type of cell death called pyroptosis, a proinflammatory programmed cell death. Cell death by pyroptosis is characterized by the loss of the plasma membrane integrity and eventual cell lysis29. We observed a significant decrease in TUNEL-positive cells in fat-1 mice compared with WT mice (Fig. S1A). LDH activity was also decreased in both the skin and serum of fat-1 mice, suggesting maintenance of membrane integrity (Figs. S1B and S1C).
Because NLRP3 expression is under transcriptional control of NF-κB, we next evaluated changes in NF-κB under our experimental conditions. Acetylated p65 (Ac-p65) was markedly increased by DNCB, while no change was observed for total p65 (Fig. 5a). DNCB-mediated NF-κB activation was followed by an increase in the mRNA levels of two of the NF-κB-dependent proinflammatory molecules, Tnfa and Il1b (Figs. 4b and 5b). We further confirmed NF-κB activation by these cytokines in HaCaT cells (Fig. 5c). Endogenously synthesized n-3 PUFAs in fat-1 mice and exogenous treatment with DHA in HaCaT cells significantly suppressed DNCB- and cytokine-induced NF-κB activation, respectively (Figs. 5a and 5c). However, arachidonic acid, an n-6 PUFA, had no effect on NF-κB activation.
Fig. 5. The Sirt1-NF-κB axis in skin lesions and HaCaT cells.
a At the end of the study, p65, acetylated p65, and Sirt1 in the skin lesions were measured by western blotting. b mRNA levels of cytokines in the skin lesions were determined by real-time RT-PCR (n = 5). c HaCaT cells were transiently transfected with an NfkB luciferase construct and then treated with 10 nM TNF-α and 10 nM IL-1β in the presence or absence of 20 μM DHA or 20 μM arachidonic acid. After 24 h, cells were harvested in reporter lysis buffer, and luciferase activity in cell lysates was assayed (n = 7–9). d mRNA levels of Sirt1 in the skin lesions were determined by real-time RT-PCR (n = 5). e, f Protein levels of Sirt1 and HMGB1 and Sirt1 luciferase activity in HaCaT cells. Values are means ± SEMs. *p < 0.05 and **p < 0.01 vs. WT; #p < 0.05 and ##p < 0.01 vs. WT+DNCB or VEH; $$p < 0.01 vs. none+VEH. VEH vehicle, DHA docosahexaenoic acid, AA arachidonic acid
We previously reported that fat-1 gene overexpression increased Sirt1 expression and decreased p65 acetylation21. We therefore investigated whether Sirt1 expression was linked to the acetylation status of p65 in our experimental conditions. Our results revealed a significant reduction in Sirt1 expression and/or activity in DNCB-treated skin tissues and cytokine-treated HaCaT cells (Fig. 5d–f). Again, endogenous and exogenous n-3 PUFAs significantly upregulated the level of Sirt1 mRNA or protein.
HDM-induced atopic dermatitis-like skin lesions are suppressed in fat-1 mice
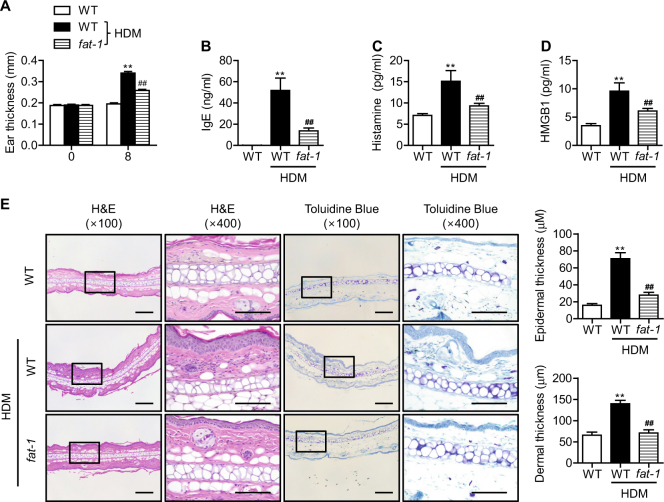
To explore the relevance of our findings to human disease, we reproduced our experiments using the HDM antigen, one of the most clinically important allergens for atopic dermatitis. HDM exposure induced increases in ear thickness; increases in serum HMGB1, IgE, and histamine; and histological abnormalities (Fig. 6a–e). Similar to the results obtained with the DNCB model, all of these parameters were significantly suppressed in fat-1 mice. The overall atopic symptoms and skin lesions observed in the HDM model were weaker than those in the DNCB model. Collectively, our experiments with HDM confirm our results with the DNCB model of atopic dermatitis.
Fig. 6. Allergic inflammation in response to HDM.
Mice were challenged with HDM as described in the Materials and Methods. a Ear thickness and (b-d) serum IgE, histamine, and HMGB1were analyzed (n = 6). e Paraffin-embedded skin sections were prepared and stained with H&E and toluidine blue. Bars = 100 μm. Epidermal and dermal thicknesses were measured in H&E-stained microphotographs (n = 6). Values are means ± SEMs. **p < 0.01 vs. WT; ##p < 0.01 vs. WT+HDM
Discussion
Several human clinical trials have addressed the beneficial effects of n-3 PUFAs on atopic dermatitis. Fish oil supplementation during pregnancy has resulted in improvements in the clinical severity of atopic outcomes for infants30,31. Similarly, infants supplemented with DHA-rich formula during the first year of life had a reduced incidence of atopic dermatitis during early childhood32. Regulation of epidermal barrier function and modulation of the immune system have been suggested as underlying mechanisms33. Since DHA and EPA are not major constituents of the epidermis because of insufficient dietary consumption or increased cellular utilization, the restoration of skin permeability-barrier abnormality is not likely the main mechanism. Instead, substantial attention has been paid to the immune-modulatory function of n-3 PUFAs, especially T cell regulation34,35. However, to our knowledge, a study on the inflammasome, a component of the innate immune system, has not been reported.
An Nlrp3 gene polymorphism has been associated with increased risk of atopic dermatitis11–13, and activation of inflammasomes triggers the allergic inflammatory response36. Consequently, targeting the inhibition of NLRP3 inflammasomes is an attractive strategy for treating allergic diseases, including atopic dermatitis. Although there is growing evidence that nutrition is an effective modulator of the inflammasome, few attempts have been made to inhibit the NLRP3 inflammasome through diet. Interestingly, saturated fatty acids activate the NLRP3 inflammasome37,38, while unsaturated fatty acids prevent its activation39,40. Herein, we investigated whether endogenously synthesized n-3 PUFAs are effective in suppressing the NLRP3 inflammasome and mitigating the pathophysiological consequences of inflammation in mouse models of DNCB- and HDM-induced atopic dermatitis-like skin lesions. Our results showed that DNCB treatment resulted in activation of NLRP3 inflammasome signaling pathways, as evidenced by increases in NLRP3 and ASC expression, enhanced maturation of caspase-1 and IL-1β, and induction of pyroptosis. However, fat-1 gene overexpression increases Sirt1 expression, which allows for deacetylation of NF-κB, resulting in the loss of its transcriptional activity. Given that NF-κB activation is required for the transcription of inflammasome components, including Nlrp341, n-3 PUFAs seem to inhibit the priming of the NLRP3 inflammasome based on its ability to suppress NF-κB.
Most of the mechanistic studies on atopic dermatitis point to an imbalance between Th1 and Th2 responses in favor of Th2 responses2,3. The characteristic Th2 responses observed in atopic dermatitis patients include abnormal IgE production, mast cell infiltration, peripheral eosinophilia, and induction of Th2 cells expressing IL-4 and IL-13. Interestingly, sequential biopsies from atopic dermatitis patients after exposure to aeroallergens show a biphasic immunologic response characterized by switching from a Th2 phenotype in the earlier phase to a Th1 phenotype in the later phase of the disease8. Concordantly, B cells switch their expression of immunoglobulin classes from IgE to IgG2a. Consistent with our previous report19, fat-1 gene overexpression decreased IL-4, IL-5, IL-13, and IFN-γ as well as IgE, IgG1, and IgG2a, suggesting that n-3 PUFAs inhibit both Th2 and Th1 immune responses. In addition to the Th1/Th2 paradigm, the roles of Th17 and Treg cells in the pathogenesis of atopic dermatitis have also been highlighted42,43. IL-17 modulates innate immunity via keratinocytes, as IL-17 together with IFN-γ amplifies a nonspecific cytotoxic cascade that results in a severe and sustained cutaneous inflammatory reaction44. To clarify this point, it was necessary to examine Th17 cell infiltration into skin lesions of fat-1 mice. In the present study, fat-1 gene overexpression successfully inhibited Th17 cell infiltration in the skin tissues and IL-17 production by draining lymph nodes, which was related to the decreased production of the inflammatory cytokines IFN-γ, TNF-α, and IL-6. Consistent with our previous report19, the present study strongly suggests that fat-1 gene overexpression suppresses Th17 cell-mediated cutaneous inflammation in DNCB-treated mice. However, in contrast to the report by Han et al.34, we did not observe an increase in Treg cells in fat-1 mice. In their study, DHA upregulated the function of M2 macrophages and promoted the generation of Treg cells. Since we also observed an increase in M2 macrophages in fat-1 mice21, differences in the n-3 PUFAs (DHA in their study vs. n-3 PUFAs in our study) might have produced the different outcomes.
In summary, our data demonstrate that DNCB treatment activates the NLRP3 inflammasome pathway, which induces the release of inflammatory cytokines and amplifies the inflammatory response to cause atopic dermatitis symptoms. Modulation of this pathway with n-3 PUFAs has clinical relevance and can be a useful strategy to attenuate atopic dermatitis.
Electronic supplementary material
Acknowledgements
This work was supported by the Basic Science Research Program (grant numbers NRF-2017R1A2B2005730 and 2018R1D1A1B07046553) and by the Medical Research Center Program (grant number NRF-2017R1A5A2015061) through the National Research Foundation (NRF) funded by the Korean government (MSIP). We thank Sang-Hyun Kim (School of Medicine, Kyungpook National University, Korea) for critical reading of the manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The authors contributed equally: Hyun-Young Jang, Jeung-Hyun Koo.
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s12276-018-0104-3.
References
- 1.Liang Y, Chang C, Lu Q. The genetics and epigenetics of atopic dermatitis-filaggrin and other polymorphisms. Clin. Rev. Allergy Immunol. 2016;51:315–328. doi: 10.1007/s12016-015-8508-5. [DOI] [PubMed] [Google Scholar]
- 2.Brandt EB, Sivaprasad U. Th2 cytokines and atopic dermatitis. J. Clin. Cell Immunol. 2011;2:110. doi: 10.4172/2155-9899.1000110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kabashima K. New concept of the pathogenesis of atopic dermatitis: interplay among the barrier, allergy, and pruritus as a trinity. J. Dermatol. Sci. 2013;70:3–11. doi: 10.1016/j.jdermsci.2013.02.001. [DOI] [PubMed] [Google Scholar]
- 4.Weidinger S, Novak N. Atopic dermatitis. Lancet. 2016;387:1109–1122. doi: 10.1016/S0140-6736(15)00149-X. [DOI] [PubMed] [Google Scholar]
- 5.Ito T, et al. TSLP-activated dendritic cells induce an inflammatory T helper type 2 cell response through OX40 ligand. J. Exp. Med. 2005;202:1213–1223. doi: 10.1084/jem.20051135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Soumelis V, et al. Human epithelial cells trigger dendritic cell mediated allergic inflammation by producing TSLP. Nat. Immunol. 2002;3:673–680. doi: 10.1038/ni805. [DOI] [PubMed] [Google Scholar]
- 7.Tatsuno K, Fujiyama T, Yamaguchi H, Waki M, Tokura Y. TSLP directly interacts with skin-homing Th2 cells highly expressing its receptor to enhance IL-4 production in atopic dermatitis. J. Invest Dermatol. 2015;135:3017–3024. doi: 10.1038/jid.2015.318. [DOI] [PubMed] [Google Scholar]
- 8.Grewe M, et al. Analysis of the cytokine pattern expressed in situ in inhalant allergen patch test reactions of atopic dermatitis patients. J. Invest Dermatol. 1995;105:407–410. doi: 10.1111/1523-1747.ep12321078. [DOI] [PubMed] [Google Scholar]
- 9.Jiao D, et al. NOD2 and TLR2 ligands trigger the activation of basophils and eosinophils by interacting with dermal fibroblasts in atopic dermatitis-like skin inflammation. Cell Mol. Immunol. 2016;13:535–550. doi: 10.1038/cmi.2015.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Arend WP, Palmer G, Gabay C. IL-1, IL-18, and IL-33 families of cytokines. Immunol. Rev. 2008;223:20–38. doi: 10.1111/j.1600-065X.2008.00624.x. [DOI] [PubMed] [Google Scholar]
- 11.Macaluso F, et al. Polymorphisms in NACHT-LRR (NLR) genes in atopic dermatitis. Exp. Dermatol. 2007;16:692–698. doi: 10.1111/j.1600-0625.2007.00589.x. [DOI] [PubMed] [Google Scholar]
- 12.Zhang Q, Fan HW, Zhang JZ, Wang YM, Xing HJ. NLRP3rs35829419 polymorphism is associated with increased susceptibility to multiple diseases in humans. Genet Mol. Res. 2015;14:13968–13980. doi: 10.4238/2015.October.29.17. [DOI] [PubMed] [Google Scholar]
- 13.Bivik C, et al. Genetic variation in the inflammasome and atopic dermatitis susceptibility. J. Invest Dermatol. 2013;133:2486–2489. doi: 10.1038/jid.2013.168. [DOI] [PubMed] [Google Scholar]
- 14.Belloni B, et al. Low-dose anti-IgE therapy in patients with atopic eczema with high serum IgE levels. J. Allergy Clin. Immunol. 2007;120:1223–1225. doi: 10.1016/j.jaci.2007.08.060. [DOI] [PubMed] [Google Scholar]
- 15.He A, Feldman SR, Fleischer AB. Trends in atopic dermatitis management: comparison of 1990-1997 to 2003-2012. J. Drugs Dermatol. 2018;17:135–140. [PubMed] [Google Scholar]
- 16.Chong M, Fonacier L. Treatment of eczema: corticosteroids and beyond. Clin. Rev. Allergy Immunol. 2016;51:249–262. doi: 10.1007/s12016-015-8486-7. [DOI] [PubMed] [Google Scholar]
- 17.Hageman JH, et al. The impact of dietary long-chain polyunsaturated fatty acids on respiratory illness in infants and children. Curr. Allergy Asthma Rep. 2012;12:564–573. doi: 10.1007/s11882-012-0304-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Faber J, et al. Supplementation with a fish oil-enriched, high-protein medical food leads to rapid incorporation of EPA into white blood cells and modulates immune responses within one week in healthy men and women. J. Nutr. 2011;141:964–970. doi: 10.3945/jn.110.132985. [DOI] [PubMed] [Google Scholar]
- 19.Jang HY, Lim K, Lee SM, Park BH. Effects of n-3 PUFA on the CD4+ type 2 helper T-cell-mediated immune responses in Fat-1 mice. Mol. Nutr. Food Res. 2014;58:365–375. doi: 10.1002/mnfr.201300194. [DOI] [PubMed] [Google Scholar]
- 20.Brown AL, et al. Omega-3 fatty acids ameliorate atherosclerosis by favorably altering monocyte subsets and limiting monocyte recruitment to aortic lesions. Arterioscler. Thromb. Vasc. Biol. 2012;32:2122–2130. doi: 10.1161/ATVBAHA.112.253435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Song MY, et al. Enhanced M2 macrophage polarization in high n-3 polyunsaturated fatty acid transgenic mice fed a high-fat diet. Mol. Nutr. Food Res. 2016;60:2481–2492. doi: 10.1002/mnfr.201600014. [DOI] [PubMed] [Google Scholar]
- 22.Teague H, Rockett BD, Harris M, Brown DA, Shaikh SR. Dendritic cell activation, phagocytosis and CD69 expression on cognate T cells are suppressed by n-3 long-chain polyunsaturated fatty acids. Immunology. 2013;139:386–394. doi: 10.1111/imm.12088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kang JX, Wang J, Wu L, Kang ZB. Transgenic mice: fat-1 mice convert n-6 to n-3 fatty acids. Nature. 2004;427:504. doi: 10.1038/427504a. [DOI] [PubMed] [Google Scholar]
- 24.Weylandt KH, et al. Reduction of inflammation and chronic tissue damage by omega-3 fatty acids in fat-1 transgenic mice with pancreatitis. Biochim Biophys. Acta. 2008;1782:634–641. doi: 10.1016/j.bbadis.2008.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Woo SJ, et al. Endogenous conversion of n-6 to n-3 polyunsaturated fatty acids attenuates K/BxN serum-transfer arthritis in fat-1 mice. J. Nutr. Biochem. 2015;26:713–720. doi: 10.1016/j.jnutbio.2015.01.011. [DOI] [PubMed] [Google Scholar]
- 26.Kurashima Y, et al. The enzyme Cyp26b1 mediates inhibition of mast cell activation by fibroblasts to maintain skin-barrier homeostasis. Immunity. 2014;40:530–541. doi: 10.1016/j.immuni.2014.01.014. [DOI] [PubMed] [Google Scholar]
- 27.Dai X, et al. Mite allergen is a danger signal for the skin via activation of inflammasome in keratinocytes. J. Allergy Clin. Immunol. 2011;127:806–14. e1-4. doi: 10.1016/j.jaci.2010.12.006. [DOI] [PubMed] [Google Scholar]
- 28.Iversen L, Johansen C. Inflammasomes and inflammatory caspases in skin inflammation. Expert Rev. Mol. Diagn. 2008;8:697–705. doi: 10.1586/14737159.8.6.697. [DOI] [PubMed] [Google Scholar]
- 29.Miao EA, Rajan JV, Aderem A. Caspase-1-induced pyroptotic cell death. Immunol. Rev. 2011;243:206–214. doi: 10.1111/j.1600-065X.2011.01044.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Furuhjelm C, et al. Allergic disease in infants up to 2 years of age in relation to plasma omega-3 fatty acids and maternal fish oil supplementation in pregnancy and lactation. Pediatr. Allergy Immunol. 2011;22:505–514. doi: 10.1111/j.1399-3038.2010.01096.x. [DOI] [PubMed] [Google Scholar]
- 31.Denburg JA, et al. Fish oil supplementation in pregnancy modifies neonatal progenitors at birth in infants at risk of atopy. Pediatr. Res. 2005;57:276–281. doi: 10.1203/01.PDR.0000148279.72611.1D. [DOI] [PubMed] [Google Scholar]
- 32.Birch EE, et al. The impact of early nutrition on incidence of allergic manifestations and common respiratory illnesses in children. J. Pediatr. 2010;156:902–906. doi: 10.1016/j.jpeds.2010.01.002. [DOI] [PubMed] [Google Scholar]
- 33.McCusker MM, Grant-Kels JM. Healing fats of the skin: the structural and immunologic roles of the omega-6 and omega-3 fatty acids. Clin. Dermatol. 2010;28:440–451. doi: 10.1016/j.clindermatol.2010.03.020. [DOI] [PubMed] [Google Scholar]
- 34.Han SC, et al. Docosahexaenoic acid alleviates atopic dermatitis by generating Tregs and IL-10/TGF-β-modified macrophages via a TGF-β-dependent mechanism. J. Invest Dermatol. 2015;135:1556–1564. doi: 10.1038/jid.2014.488. [DOI] [PubMed] [Google Scholar]
- 35.Park BK, et al. Omega-3 fatty acids suppress Th2-associated cytokine gene expressions and GATA transcription factors in mast cells. J. Nutr. Biochem. 2013;24:868–876. doi: 10.1016/j.jnutbio.2012.05.007. [DOI] [PubMed] [Google Scholar]
- 36.Herberth G, et al. Endogenous metabolites and inflammasome activity in early childhood and links to respiratory diseases. J. Allergy Clin. Immunol. 2015;136:495–497. doi: 10.1016/j.jaci.2015.01.022. [DOI] [PubMed] [Google Scholar]
- 37.Wen H, et al. Fatty acid-induced NLRP3-ASC inflammasome activation interferes with insulin signaling. Nat. Immunol. 2011;12:408–415. doi: 10.1038/ni.2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Reynolds CM, et al. Dietary saturated fatty acids prime the NLRP3 inflammasome via TLR4 in dendritic cells-implications for diet-induced insulin resistance. Mol. Nutr. Food Res. 2012;56:1212–1222. doi: 10.1002/mnfr.201200058. [DOI] [PubMed] [Google Scholar]
- 39.Yan Y, et al. Omega-3 fatty acids prevent inflammation and metabolic disorder through inhibition of NLRP3 inflammasome activation. Immunity. 2013;38:1154–1163. doi: 10.1016/j.immuni.2013.05.015. [DOI] [PubMed] [Google Scholar]
- 40.Finucane OM, et al. Monounsaturated fatty acid-enriched high-fat diets impede adipose NLRP3 inflammasome-mediated IL-1β secretion and insulin resistance despite obesity. Diabetes. 2015;64:2116–2128. doi: 10.2337/db14-1098. [DOI] [PubMed] [Google Scholar]
- 41.Bauernfeind FG, et al. Cutting edge: NF-κB activating pattern recognition and cytokine receptors license NLRP3 inflammasome activation by regulating NLRP3 expression. J. Immunol. 2009;183:787–791. doi: 10.4049/jimmunol.0901363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Guttman-Yassky E, Krueger JG. Atopic dermatitis and psoriasis: two different immune diseases or one spectrum? Curr. Opin. Immunol. 2017;48:68–73. doi: 10.1016/j.coi.2017.08.008. [DOI] [PubMed] [Google Scholar]
- 43.Di Cesare A, Di Meglio P, Nestle FO. A role for Th17 cells in the immunopathogenesis of atopic dermatitis? J. Invest Dermatol. 2008;128:2569–2571. doi: 10.1038/jid.2008.283. [DOI] [PubMed] [Google Scholar]
- 44.Pennino D, et al. IL-17 amplifies human contact hypersensitivity by licensing hapten nonspecific Th1 cells to kill autologous keratinocytes. J. Immunol. 2010;184:4880–4888. doi: 10.4049/jimmunol.0901767. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.