Abstract
Streptococcus infantarius subsp. infantarius (Sii), a member of the Streptococcus bovis/Streptococcus equinus complex (SBSEC), predominates as dairy-adapted and non-adapted variants in fermented dairy products (FDP) in East and West Africa. Epidemiologic data suggest an association with colorectal cancer for most SBSEC members, including Sii from Kenyan patients. Phylogenetic relationships of East African human (EAH) isolates to those of dairy and pathogenic origin were analysed to better estimate potential health implications via FDP consumption. The MLST-derived population structure was also evaluated to provide host, disease, geography and dairy adaptation associations for 157 SBSEC isolates, including 83 novel Sii/SBSEC isolates of which 40 originated from Kenyan colonoscopy patients. Clonal complex (CC) 90 was delineated as potential pathogenic CC for Sii. Single EAH, West African dairy (WAD), food and animal Sii isolates clustered within CC-90, suggesting a potential link to pathogenic traits for CC-90. The majority of EAH and WAD Sii were clustered in a shared clade distinct from CC-90 and East African dairy (EAD) isolates. This indicates shared ancestry for the EAH and WAD clade and limitations to translate disease associations of EAH and CC-90 to EAD Sii, which could support the separation of pathogenic, pathobiont/commensal and food lineages.
Introduction
Streptococcus infantarius subsp. infantarius (Sii) belongs to the Streptococcus bovis/Streptococcus equinus complex (SBSEC). SBSEC is a diverse group of bacteria including commensal inhabitants of the human and animal gastrointestinal tract, opportunistic pathogens and variants in food1,2. The SBSEC is composed of seven subspecies: Streptococcus gallolyticus subsp. gallolyticus (Sgg), Streptococcus gallolyticus subsp. macedonicus (Sgm), Streptococcus gallolyticus subsp. pasteurianus (Sgp), Streptococcus equinus, Streptococcus infantarius subsp. infantarius (Sii), Streptococcus lutetiensis and Streptococcus alactolyticus. SBSEC provides enhanced discrimination power in contrast to the former Streptococcus bovis designation that is still used for its wide recognition2,3.
SBSEC group members are associated with bacteraemia, infective endocarditis, urinary tract infections, meningitis, sepsis, gastroenteritis, endophthalmitis and carcinoma of the colon4–6. Sgg feature a strong association with colorectal cancer, particularly in patients affected by Sgg-related bacteraemia or infective endocarditis7. To a lesser extent, this association is also observed for S. lutetiensis, Sgp and possibly even Sii and Sgm. However, the causality of SBSEC members in colorectal cancer is still questioned and rather explained by a bacterial driver-passenger model7–10. In contrast to Sgg, Sgp and S. lutetiensis, Sii is a rare isolate in clinical specimens11,12, partially because Sii was only recently separated from S. lutetiensis (former S. infantarius subsp. coli) within the S. infantarius branch2,3. This separation provides clarity regarding the involvement of subspecies of S. infantarius in disease that was rarely available in studies conducted prior to this new taxonomy. Sii is implicated in human infections including infective endocarditis13, biliary tract infection, cirrhotic bacteraemia and non-colorectal cancer8,10,14–18.
The habitat of Sii as well as most other SBSEC members is mainly in the gastrointestinal tract of humans as well as animals such as ruminants, sea otters and birds2,3, where they are considered to be commensals but also fit as putative representatives of pathobionts19. Furthermore, Sii and Sgm are regularly found as predominant organisms in fermented dairy products (FDP)2. Particularly, Sii is highly prevalent as a predominant bacterium in traditional FDP in sub-Saharan Africa20–23. Thus, live Sii are ingested by millions of FDP consumers in sub-Saharan Africa at levels over 108 CFU mL−1. However, East African and many West African dairy Sii are clearly different from the human Sii analysed to date24–26. African dairy Sii feature dairy adaptations such as a modified lactose metabolism via a lacS and lacZ encoded lactose uptake instead of the SBSEC-typical lactose phosphotransferase system24,25. Phylogenetic analysis further supports the differentiation of specific African dairy lineages by multi locus sequence typing (MLST)24. Several of these Sii lineages seem to be clearly separating from human commensal and human pathogenic lineages while other dairy isolates share a closer relationship with potentially harmful strains24.
Knowledge of phylogeny of human commensal and pathogenic lineages is currently limited to strains isolated from humans in Europe and Asia; analogous knowledge of African Sii is limited to dairy strains only24. Representative human Sii isolates from African countries such as Kenya, Somalia or Côte d’Ivoire with a documented presence of dairy Sii were missing and therefore did not allow for a more comprehensive phylogenetic evaluation of lineages, strains and their relationships. In a recent hospital-based study on patients undergoing colonoscopy at Kenyatta National Hospital in Nairobi, Kenya, strains to investigate this missing link were obtained27. Among these colonoscopy patients, Sii and other SBSEC members were isolated from faecal samples and rectal swabs, and in conjunction, the socioeconomic aspects and dietary habits of the patients were assessed. The result was a set of isolates that included comprehensive descriptions of the patients27. In that study, SBSEC and Sii carriage rate among 273 participants was about 20%27. Furthermore, Sii isolates among these study participants indicated associations with colorectal cancer and haemorrhoids27. However, the population structure and relationships to human and dairy strains is unknown, leaving an important knowledge gap in the epidemiology of Sii-associated diseases and the role of dairy Sii in Africa, particularly in the example of Kenya. Given the large number of daily consumers of FDP and thus Sii consumers in Kenya, closing this knowledge gap of dairy vs human Sii in Kenya also has a significant public health impact.
Therefore, the objective of this study was to evaluate the phylogenetic relationships among the first African human Sii and other SBSEC members in comparison with dairy, commensal and pathogenic Eurasian strains through the SBSEC-specific MLST approach. Thereby, we investigated the evolution of Sii and SBSEC with a global and an African perspective. Particularly for Africa, a thorough safety assessment of dairy Sii and their relationship to potentially pathogenic and human commensal lineages is of high priority, given the role of Sii in the daily diet of millions of pastoralists in sub-Saharan Africa28. This study represents the first phylogenetic analysis of African human Sii and SBSEC isolates.
Results
MLST analysis of novel SBSEC isolates and update on key characteristics of the SBSEC MLST scheme
This study marks the first comparison of human SBSEC and Sii isolates of African origin to those of human commensal, human pathogen, animal commensal and dairy origin of a global collection. A total of 83 SBSEC and Sii isolates were integrated into the SBSEC multi locus sequence typing (MLST) scheme to yield 157 isolates for the subsequent assessment of evolutionary and phylogenetic relationships (Fig. 1). All isolates originating from human, animal and food collections were previously identified as SBSEC members using SBSEC-specific PCR assays targeting the 16S rRNA gene and to species level using partial groEL sequencing6,20,21,24,27,29–33.
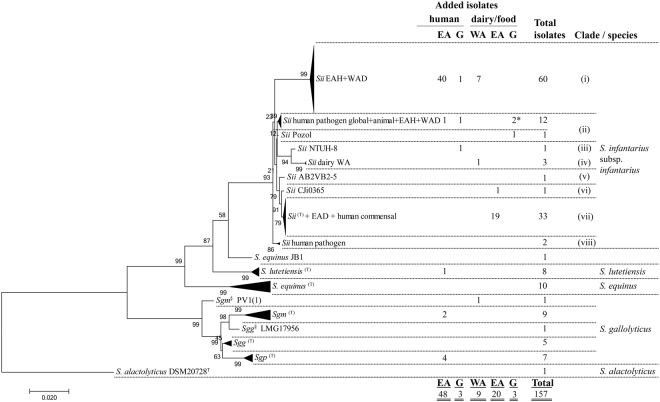
Figure 1.
Phylogenetic MLST tree of 157 SBSEC isolates including 83 new isolates. Maximum likelihood phylogenetic tree calculated from the concatenated sequences of the 10 MLST loci of 157 SBSEC isolates including a total of 83 new isolates of East African (EA) human, EA dairy, West African (WA) dairy and global (G) isolates of human, dairy, food or animal origin. The tree is rooted to S. alactolyticus DSM20728T. Validation was performed using 200 bootstrap replications for which the percentage of clustered trees is given next to the branches. The horizontal bar at the bottom indicates the evolutionary distance in the same units as used for branch length. * includes one isolate of animal origin; §marks strains with species designations according to groEL sequence but tree position not within the expected subspecies clades.
The SBSEC MLST scheme currently contains 120 STs out of these 157 isolates (Table 1).
Table 1.
Key characteristics of the SBSEC-MLST scheme for 157 SBSEC isolates calculated for the overall complex and subspecies with newly incorporated alleles.
| SBSEC | n = 157 | Sii | n = 114 | S. lutetiensis | n = 9 | Sgm | n = 10 | Sgp | n = 7 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IA = 2.784 | dN/dS | IA = 1.864 | dN/dS | IA = 1.959 | dN/dS | IA = 2.195 | dN/dS | IA = 0.534 | dN/dS | ||||||
| IAS = 0.309 | IAS = 0.207 | IAS = 0.218 | IAS = 0.244 | IAS = 0.059 | |||||||||||
| ST and alleles | ST and alleles | ST and alleles | ST and alleles | ST and alleles | |||||||||||
| New | Total | New | Total | New | Total | New | Total | New | Total | ||||||
| ST | 54 | 120 | 47 | 78 | 1 | 9 | 3 | 10 | 3 | 6 | |||||
| ddl | 10 | 38 | 0.080 | 8 | 13 | 0.100 | 0 | 6 | 0.149 | 2 | 4 | 0.121 | 1 | 3 | 0.072 |
| gki | 7 | 34 | 0.057 | 4 | 9 | 0.125 | 0 | 5 | 0.089 | 3 | 6 | 1.368 | 1 | 2 | 0.328 |
| glnA | 6 | 27 | 0.038 | 4 | 11 | 0.129 | 0 | 6 | 0.080 | 1 | 2 | 0.141 | 0 | 3 | 0 |
| mutS | 7 | 37 | 0.028 | 4 | 9 | 0.213 | 0 | 5 | 0.600 | 2 | 6 | 0.065 | 1 | 3 | 0.146 |
| mutS2 | 11 | 47 | 0.030 | 8 | 18 | 0.179 | 0 | 5 | 0 | 3 | 5 | 0 | 1 | 3 | 0.036 |
| pheS | 8 | 36 | 0.023 | 5 | 15 | 0.011 | 0 | 4 | 0.046 | 1 | 3 | 0.017 | 0 | 3 | 0 |
| proS | 12 | 40 | 0.063 | 9 | 16 | 0.322 | 1 | 6 | 1.089 | 2 | 3 | 0.398 | 2 | 2 | 0 |
| pyrE | 30 | 66 | 0.022 | 22 | 38 | 0.043 | 1 | 6 | 0 | 1 | 4 | 0.058 | 1 | 2 | 0 |
| thrS | 4 | 35 | 0.082 | 3 | 14 | 0.107 | 0 | 5 | 0.074 | 2 | 4 | 0.099 | 1 | 2 | 0.140 |
| tpi | 7 | 24 | 0.148 | 4 | 8 | 0.761 | 0 | 5 | 0 | 2 | 4 | 0.575 | 0 | 2 | 0 |
New alleles include all new alleles incorporated into the SBSEC MLST scheme. For individual species, the number corresponds to new alleles assigned to a species, which includes also existing SBSEC alleles that were previously not assigned to a specific. IA: index of association; IAS: standardized index of association; dN/dS: ratio of non-synonymous sites over synonymous sites.
MLST analysis confirmed the subspecies of all 83 isolates, including 75 Sii, three Sgm, four Sgp, and one S. lutetiensis previously determined by groEL sequencing (Table 1). The new isolates were responsible for a total of 54 new STs of which 47 were due to Sii isolates in addition to one S. lutetiensis, three Sgm and three Sgp isolates as a result of four to 30 new alleles for the 10 different loci (Table 1). IA and IAS were calculated to be 0.534–2.784 and 0.059–0.309, respectively, indicating significant linkage disequilibrium for most species except Sgp (Table 1). The values of dN/dS < 1 indicate no selective pressure for most alleles except for gki in Sgm (Table 1). However, while overall SBSEC (n = 157) and Sii (n = 114) provided sufficient data for calculations, the number of alleles for S. lutetiensis, Sgm and Sgp is still comparably low for reliable calculations.
Phylogenetic analysis of Sii by MLST in relation to host, source, geography and genetic markers
A total of 114 Sii isolates were included in this analysis, of which 41 originated from human faecal samples and rectal swabs obtained in Kenya, referred hereafter as East African human (EAH) isolates (colonoscopy patient Sii isolates n = 34, infant faecal sample Sii isolates n = 7). Four out of the 33 Kenyatta National Hospital (KNH) patients delivered Sii isolate pairs. The isolate pairs of three patients were determined as potential isolate duplicates per patient (D261/D933, D1201/D1202, D1396/D1437), whereas one patient delivered two unique isolates D1095 and D1098 (Supplementary Fig. S1).
The Sii were separated into eight distinct clades with the major clusters comprised of (i) isolates of West African Dairy (WAD) and EAH, (ii) isolates of human blood (pathogenic), animals, EAH, WAD and pozol; and (vii) isolates of East Africa dairy (EAD) origin as well as the Sii type strain and presumptive commensal Sii (Fig. 1 and Supplementary Fig. S1). Sii strains added in this study were clustered in all Sii clades except clade (v).
Geography and sample origin were observed as important aspects for clade structure in relation to dairy isolates. WAD and EAD Sii isolates analysed in this study clustered predominantly in the clades (i) and (vii), respectively. In contrast, EAH isolates clustered in clade (i) with the exception of isolate D1266, which clustered in clade (ii). EAH Sii isolates obtained from colonoscopy patients at KNH, Nairobi, Kenya (33 isolates) and from infant faecal samples in Msambweni, Kenya (7 isolates), clustered in clade (i) shared with WAD/Asian Sii isolates (19 isolates), whereas one EAH Sii isolate (D1266) clustered in clade (ii) of human pathogens (Fig. 1 and Supplementary Fig. S1). Subdivisions of individual clades were not further followed due to limited bootstrap reliability.
Analysis of alleles and loci revealed that clustering of WAD/Asian Sii (n = 19) with EAH Sii (n = 40) in clade (i) was due to shared or highly similar alleles for glnA, mutS2, proS, thrS and tpi, despite different geographical origins and biological sources. The consensus allele array (ddl, gki, glnA, mutS, mutS2, pheS, proS, pyrE, thrS, tpi) for WAD and EAH was (2,2,2,6,2,2,2,3,2,2) and (3,3,2,1,2,3,2,41,2,2), respectively. This consensus is clearly different from that of EAD isolates and human commensal isolates of clade (iii) featuring the consensus allele array (1,1,1,4,6,1,1,1,4,1). DNA sequence analysis of all shared alleles (glnA, mutS2, proS, thrS and tpi) among WAD and EAH isolates in clade (i) displayed highest sequence identity to other Sii isolates for glnA, mutS2, proS and tpi. In contrast, thrS allele sequences of all Sii isolates of clade (i) featured highest sequence identity to S. thermophilus. This highest sequence identity to S. thermophilus was observed for all thrS alleles (2, 8, 18, 32 and 34) comprised in isolates of clade (i), and confirms previous indications of potential horizontal gene transfer (HGT) in this clade for thrS24. In addition, thrS displayed also characteristics of HGT among other SBSEC members. thrS alleles 23 and 31, as well as 33, shared highest sequence identity with S. salivarius and S. thermophilus, respectively. Alleles 23 and 33 were observed in Kenyan Sgm isolates of human origin and Sgm DSM15879T from Greek cheese. Allele 31 was present in S. alactolyticus DSM 20728 T.
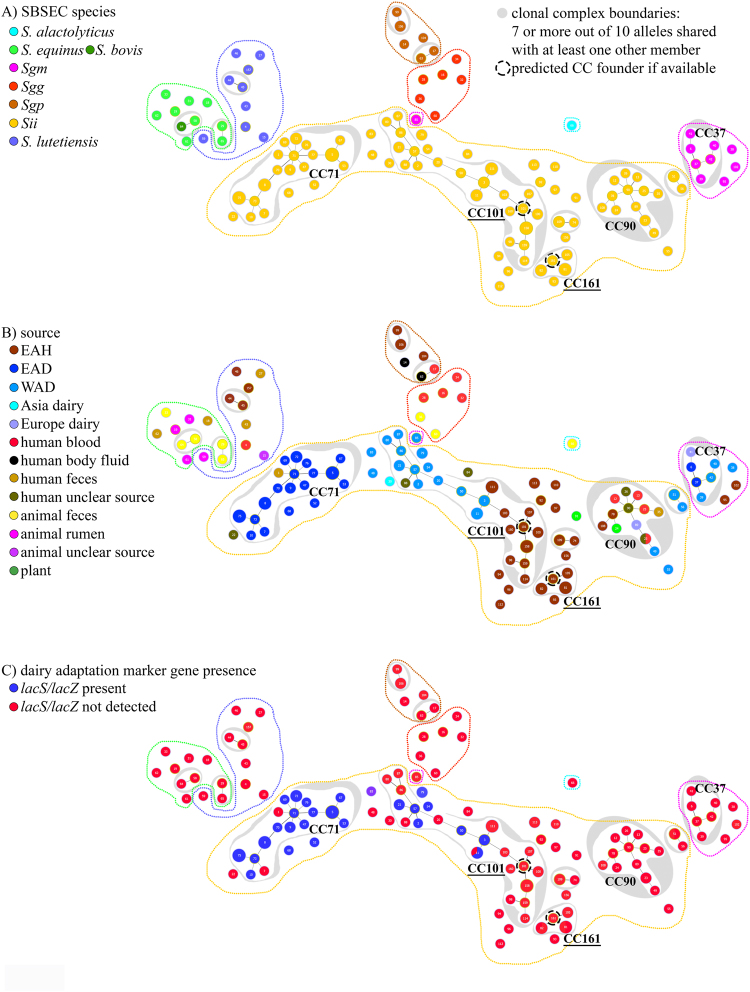
Clonal Complex (CC) calculations, with a CC definition based on STs sharing seven or more out of 10 alleles24, revealed six CCs for Sii, two for S. equinus/S. bovis, two for Sgp and one each for S. lutetiensis and Sgm (Fig. 2). Among these, five CCs were comprised of more than two STs and were labelled CC-101, CC-71, CC-90 and CC-161 for Sii and CC-37 for Sgm. Out of the Sii CCs, CC-101 was comprised of 32 isolates yielding 21 different sequence types (STs) (Fig. 2 and Supplementary Fig. S1). The predicted founder of CC-101 was ST 101. The other Sii CC-71, CC-90 and CC-161 were comprised of 17, 12 and 4 STs representing 31, 13 and 10 isolates that were centred around ST71, ST90 and ST161, respectively. However, a founding ST prediction was only possible for ST161 in CC-161, while for the CCs comprising ST71 or ST90, no founding ST could be calculated.
Figure 2.
Schematic relationships of SBSEC isolates by MLST sequence types coloured according to isolate metadata. Philoviz/eBURST clustering of SBSEC MLST sequence types (STs) of human, animal and dairy sources calculated from ST profiles. ST bullet points were coloured according to schemes for (A) species and subspecies tree including dotted lines in the same colour for graphs A-C for species and subspecies; (B) origin and source of isolates and STs; and (C) presence of dairy adaptation marker genes lacS and lacZ. Clustering was initially calculated using eBURST algorithm set to fullMST specifications in Phyloviz 2.0 and subsequently reduced to clonal complex (CC) definition of 7 or more alleles shared with at least one other member of the CC to yield this figure. For CC with >2 STs and predicted founder, the CC is labelled by underlined font. ST bullet point positions and cluster layout correspond to nearest phylogenetic relationships with the exception of S. alactolyticus DSM 20728T (ST66), which was placed outside of other species for improved visibility. ST bullet point size corresponds to the number of isolates for each specific ST with the smallest bullet points representing single isolates.
Origin and source were strongly associated with Sii CCs (Fig. 2). CC-90 encompassed all Sii human blood isolates of potential pathogenic backgrounds from worldwide sources, human clinical isolates of unknown body sites as well as several EAH, WAD, European dairy and plant isolates, raising safety concerns for members of CC-90. CC-71 was predominantly comprised of EAD Sii isolates. EAD Sii isolates and STs were all comprised within CC-71 with the exception of two singletons. CC-101 was a mix of WAD and EAH Sii isolates, including a Spanish hospital isolate (ST88). In contrast to the focused clustering of EAD Sii isolates in CC-71, WAD and EAH Sii isolates displayed wider distribution among three different CC-s and numerous singletons. This higher diversity of WAD vs. EAH and EAD Sii isolates was supported by SID analysis of the Sii STs for EAD, EAH and WAD of 0.940, 0.970 and 0.975, respectively, and also for the individual ten alleles (Supplementary Table S1). The higher SID of EAH and WAD Sii was mainly due to isolates being spread over different clades. Focusing only on clade (i), SID indicated a lower diversity of 0.968 and 0.961 for EAH and WAD Sii, respectively (Supplementary Table S1). Similarly, sequence identity analysis of the concatenated sequences of the 10 alleles of all EAH Sii isolates featured an average identity of 99.6% vs. 99.8 for EAD and 98.7 for WAD Sii (Supplementary Table S2). Within clade (i), EAH and WAD Sii featured 99.7% identity, showing that within a clade, sequence conservation was rather high (Supplementary Table S2).
The presence of the dairy adaptation marker genes lacS/lacZ was strongly associated with Sii CCs and MLST clades (Supplementary Fig. S1 and Fig. 2). Marker genes were only detected in EAD and WAD Sii isolates. None were detected in EAH Sii isolates (Supplementary Fig. S1). EAD Sii isolates shared 96.9% prevalence of lacS/lacZ genes among 32 isolates. In contrast, WAD Sii isolates (n = 24) only presented 54.2% lacS/lacZ prevalence that was focused on isolates clustered in four small clades and two single branches. In relation to CCs, CC-71 was mainly comprised of EAD Sii isolates featuring lacS/lacZ adaptation in 15 out of 17 STs, with Sii type strain (ST1, human faecal isolate) and Sii 150A (ST7, EAD isolate) being the exceptions. CC-101 including WAD Sii isolates only presented lacS/lacZ in six out of 11 WAD STs, but not in any of the EAH STs. Similarly, no lacS/lacZ adaptation was detected in human blood isolates or any STs of CC-90 and CC-161.
Phylogenetic analysis of S. gallolyticus subspecies
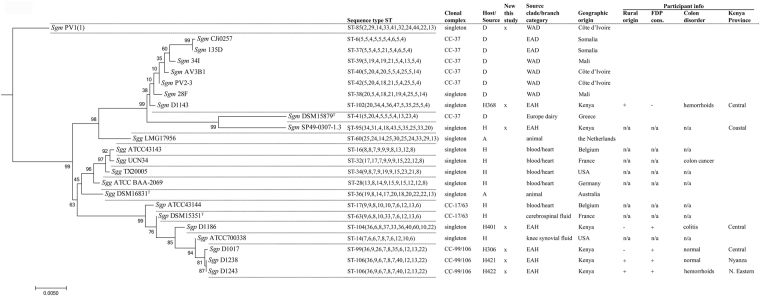
A total of seven new S. gallolyticus isolates and STs were added to the SBSEC MLST scheme (Figs 1 and 3). The three Sgm isolates comprised of one WAD and two EAH isolates clustered clearly distinct. They did not match existing ST or CC definitions and therefore formed singletons. Sgm PV1(1) (WAD) branched as a single isolate and separated from the main S. gallolyticus clade. Within the main Sgm clade, a clear division between the Sgm type strain clade and the majority of African Sgm isolates was observed using sequence-based analysis (Fig. 3). However, profile-based analysis assigned the majority of EAD and WAD Sgm to CC-37, together with the Sgm type strain. Sgm CC-37 was defined around ST37, although no direct founder was predicted. CC-37 was comprised of six isolates each representing a unique ST, but all members of CC-37 shared a dairy origin suggesting CC-37 as a main dairy CC for Sgm.
Figure 3.
Phylogenetic MLST tree of the S. gallolyticus branch. Maximum likelihood phylogenetic tree of the S. gallolyticus branch extracted from the overall rooted SBSEC MLST tree of the concatenated sequences of 10 MLST loci. New isolate additions were: Sgm PV1(1), Sgm SP49-0307-1.3, Sgm D1143, Sgp D1017, Sgp D1186, Sgp D1238 and Sgp D1243. The horizontal bar at the bottom indicates the evolutionary distance in the same units as used for branch length. Isolate information is given for host/source of animal (A), dairy (D), or human (H) origin followed by participant ID for isolates obtained from colonoscopy patients of this study. “n/a” indicates that this information is not available for this strain. Participant information was only filed for human samples. Sequence type (ST) and clonal complex (CC) assignment was indicated based on MLST profile analysis.
EAH Sgp isolates formed subclades with Sgp ATCC700338 and Sgp DSM15351T. CC definition among Sgp was only possible for two pairs while the other STs remained singletons. Sgg represented only singletons without any CCs. This finding of a majority of singleton isolates was also reflected in highest SID values of 1 for Sgm, Sgg and Sgp indicating absolute diversity (Supplementary Table S1). DNA sequence identity across the 10 alleles showed high conservation of 99.2 and 99.5% for Sgg and Sgp, respectively, whereas Sgm featured 98.4% (Supplementary Table S2).
Distribution of Sii, Sgm and Sgp isolates in relation to colonic disorders and consumption of traditionally fermented dairy products
A total of 40 SBSEC isolates originating from 33 colonoscopy patients were analysed. Of these, EAH Sii isolates (n = 34) from 30 different colonoscopy patients were integrated into the MLST scheme (Supplementary Fig. S1). These 34 Sii isolates were obtained from eight participants with colorectal cancer (CRC) (8/34 isolates), 12 participants with haemorrhoids (14/34 isolates) and nine participants with normal colons (11/34 isolates).
EAH Sii were distributed among clade (i), with the exception of Sii D1266 clustering in clade (ii) yielding a clear separation for most EAH Sii isolates from those of blood origin of clade (ii)24 and EAD Sii of clade (vii) (Supplementary Fig. S1). There was no apparent allocation of diagnoses as well as normal colon types or patient origin to specific subclades within clade (i) due to a high degree of sequence identity (Supplementary Fig. S1). Isolate D1266 of a patient suffering from haemorrhoids clustered among these presumptive pathogens of clade (ii).
Consumption of FDP was a rare trait among participants and only recorded for seven patients (Fig. 3 and Supplementary Fig. S1). This included two exclusive Sii carriers, three exclusive Sgp carriers and two carriers of either Sii and Sgp or Sii and S. lutetiensis. None of these isolates of FDP consumers were found to harbour dairy adaptation markers lacS or lacZ. Furthermore, MLST clustering of Sii and Sgm of these EAH isolates was clearly distinct from the corresponding species among the dairy adapted EAD isolates, indicating that EAH Sii and possibly also Sgm represent different lineages from EAD Sii and Sgm, respectively.
For Sgm, Sgp and S. lutetiensis, none of the patients reported to carry Sgm (one patient, one isolate), Sgp (four patients, four isolates) or S. lutetiensis (one patient, one isolate) were diagnosed with CRC, whereas haemorrhoids was the most common finding in three patients, followed by normal colon in two patients (Fig. 3).
Discussion
Sub-Saharan Africa has unique lineages of dairy adapted Sii isolated from traditional FDP that are clearly distinct from human and animal isolates obtained from Europe or Asia24. Previous epidemiologic data of patients undergoing colonoscopy at KNH suggested an association between CRC and Sii as well as haemorrhoids and Sii. Although no causality is proven, this finding raises concerns considering the large population consuming Sii on a daily basis via FDP. However, before this study, no African human Sii isolates had been analysed to reveal the population structure, phylogenetic relationships and potential evolution of African human Sii vs other human and particularly dairy Sii. In addition, this study reports the first human Sgm and Sgp isolates obtained in Africa to give a more comprehensive insight into SBSEC in Africa.
A total of 83 new SBSEC isolates including 75 Sii isolates of human, dairy and animal origins were therefore included in this study for a total of 157 SBSEC isolates in the MLST scheme. Of these 75 Sii, 34 Sii isolates originated from human patients undergoing colonoscopy at KNH, Kenya while others were obtained from Kenyan infants as well as food and human sources in Africa, Europe, Asia and America. Among the Sii, CC-90 was clearly delineated from all other CCs, and encompassed all human blood isolates and thus potential human pathogens. CC-90 could therefore be considered as a key pathogen CC of Sii. The presence of single EAH and WAD Sii among CC-90 raises concerns for human carriers and food products containing these strains. However, the clear delineation of CC-90 from the EAD CC-71 and EAH clade (vii) as well as most other WAD/EAH CCs and clades supports further in-depth functional and genomic comparisons between key representative isolates of these CCs.
The unexpectedly close relationship between EAH Sii and WAD Sii raised further concerns and questions. The EAH Sii isolates were, with the exception of one isolate, closest related to WAD Sii isolates and clearly distinct from EAD Sii isolates. The majority of EAH and WAD Sii isolates clustered in shared CC-101 and CC-161 instead of CC-71 of EAD. This phylogenetic division was related to highly similar or shared alleles in five out of ten loci, suggesting a common ancestor for most EAH and WAD Sii. Furthermore, EAH and WAD Sii isolates shared the unique feature of thrS alleles with highest sequence identity to S. thermophilus/S. salivarius. This could be a result of genetic exchange at the base of this clade to define this new lineage. As observed for Sgp and Sgm in comparison to Sgg, this might further suggest a common ancestral Sii, possibly of human or animal origin, from which these lineages diverged and undergo niche-adaptation, particularly for EAD Sii7,34.
The combined clustering of EAH Sii and WAD Sii isolates also raised questions regarding the original reservoir of these lineages as well as the possibility for regular exchange of isolates between humans and dairy products. EAH isolates were obtained from several participants consuming traditional FDP. Kenyan FDP were observed to contain dairy adapted Sii in over 90% of samples, of which strains clustered exclusively in the EAD Sii clade (vii)21,24. The absence of the dairy adaptation marker genes lacS/lacZ in all EAH Sii isolates as well as a clear MLST separation from EAD Sii isolates strongly suggests that none of the EAH Sii isolates originated from FDP. This also suggests that the observed association between EAH Sii27, haemorrhoids and CRC does not directly apply to the EAD Sii lineage without further strain-to-strain evaluation.
Whether the reason for the absence of EAD Sii in FDP consumers is related to the ability of EAD Sii to compete against intestinal Sii and then colonize the gastrointestinal tract remains to be investigated. Colonization is often related to an initial adhesion step. Adhesion to extracellular matrix proteins of the gut such as mucin was limited among EAD in contrast to human isolates24, and could be an indication of reduced competitiveness within the gut microbiota. This will of course require further functional comparisons between isolates and additional field-based studies to directly link dairy product analysis with consumer stool analysis, as well as comparisons between East and West Africa.
This East-West comparison seems crucial due to the different dairy Sii lineages and potentially different human Sii lineages. It also has important safety implications, as the clear division between the EAH/WAD Sii lineages of clade (i), the potential pathobiont human Sii lineage of clade (ii) and the dairy adapted EAD lineage of clade (vii) might enable better definition of possible virulence factors and dairy marker genes for a thorough safety assessment. However, this also means that food isolates (WAD, Asian and Mexican Sii) clustered in clades (i) and (ii) will require even more detailed examinations to provide recommendations for fermented food production.
The causality of SBSEC to CRC is an ongoing investigation9. In this study, no apparent link between EAH Sii isolates, geographic patient origin and CRC or haemorrhoids could be established (Supplementary Fig. S1). This was certainly limited by the available number of strains for analysis. It is however an important step towards establishing better epidemiologic data for Sii. Sii showed an association with CRC and haemorrhoids among colonoscopy patients at KNH, Nairobi, Kenya27. This finding therefore supports the recommendation that the detection of any SBSEC member justifies colorectal examination8. However, these EAH Sii were not related to EAD Sii, and thus disease associations found for EAH in colonoscopy patients might not apply to EAD Sii. Further epidemiologic and comparative investigations are required to determine the role of EAD Sii lineage strains in relation to EAH and WAD, particularly among larger sample numbers of FDP consumers. These assessments will also need to consider recent findings relating to rotavirus vaccine responses among Ghanaian infants, which showed positive correlation with SBSEC titres in gut microbiota35, and could thus further highlight a special role of SBSEC members in sub-Saharan Africa.
Within the S. gallolyticus branch, Sgm lineages provided several indications for unexpected phylogenetic differentiations. Sgm PV1(1) isolated from WAD branched off early from all other S. gallolyticus members. This finding will require in-depth analysis via whole genome sequencing to determine the reasons for this branching and assist in species classification. Within the Sgm main clade, only one EAH isolate (SP49-0307-1.3) clustered close to the Sgm type strain, whereas all others formed a clearly-separated African lineage when using sequence-based analysis. However, profile-based analysis indicated the formation of a possible dairy CC-37 together with the Sgm type strain while EAH Sgm and PV1(1) represented singletons, suggesting possible diverging human and dairy lineages as for Sii but on a more global scale. However, the differentiation between EAH, EAD and WAD Sgm is less established compared to Sii and too early for further interpretation. In contrast, Sgp displayed a single African lineage clearly integrated into a separated branch and CC-99/106 within the Sgp clade. The number of available strains and the presence of mostly singletons for Sgp as well as Sgg are too little for further interpretation at this stage.
These different observations in MLST trees calculated from profiles (Fig. 2) vs concatenated sequences (Fig. 3 and Supplementary Fig. S1) relate to the effect of recombination as a major driver for intrastrain variations within the SBSEC24,36. A profile-based approach in the case of a recombinant population provides equal weight for any kind of mutation and thus reduces the impact of genetic exchange among lineages36. In recombinant populations, this can lead to discrepancies between CC definitions according to profiles and concatenated sequence phylogenies as observed in this study (Fig. 3 and Supplementary Fig. S1). Often, species do not evolve as strictly clonal or recombinant populations but rather feature a combination of clonal and recombinant evolution36. Profile and sequence-based analysis in parallel helped to reveal the thrS-allele influenced delineation of clade (i) among Sii, but also the clear definition of key CCs for EAD Sii (CC-71), EAH/WAD Sii (CC-101 and CC-161), potential human pathogens Sii (CC-90) as well as potential Sgm dairy lineage (CC-37) (Fig. 3 and Supplementary Fig. S1).
Conclusively, MLST analysis of 157 SBSEC isolates including the first set of Sii, Sgm and Sgp isolates obtained from patients undergoing colonoscopy at KNH, Nairobi, Kenya, revealed unexpected insights into the phylogeny and population structure of the SBSEC including multiple recombination events. Africa-specific lineages were obtained for Sii, Sgm and Sgp requiring in-depth comparisons to described pathogens. Among Sii, CC-90 was delineated as a potential main pathogen lineage comprised of all human blood isolates but also single EAH, WAD, animal and food isolates, indicating health risks for these food and faecal isolates. Unexpectedly, most EAH Sii isolates shared closest phylogeny, allele sequences and absence of dairy adaption markers with most WAD Sii isolates to form one major clade comprised of two mixed CCs and multiple singletons. This clade structure indicates a shared ancestor for EAH and WAD Sii isolates that is clearly distinct from the EAD Sii. In East Africa, this finding seems to suggest the evolution of a unique Sii dairy lineage, which is significantly separated from any African human Sii isolate, even those obtained from East African FDP consumers and colonoscopy patients. The association of EAH Sii with CRC and haemorrhoids should therefore not be directly linked to EAD Sii without further functional and genomic comparison to potentially separate pathogenic, pathobiont and food lineages. Consequently, clade- and CC-specific marker genes will need to be identified to enable a thorough safety assessment per lineage. Furthermore, detailed epidemiological data on human SBSEC isolates paired with dairy product analysis from the regular population in Africa, particularly West Africa due to the EAH-WAD Sii link, and other continents will be required for a more comprehensive picture beyond the hospital setting. The daily consumption of Sii-containing food products by approximately 200 million people in sub-Saharan Africa demands such actions given the general recent pathogenicity implication postulated for all SBSEC members8, but under consideration of the novel population structure findings of this study.
Methods
Ethical approval of the study
This study was approved in Kenya by the Kenyatta National Hospital/University of Nairobi, Ethics and Research Committee (KNH/UoN ERC)-approval number P389/07/2012. In Switzerland, the Ethics Committees of ETH Zurich and Kantonale Ethik Kommission Zurich (KEK) approved the study under decision numbers EK 2013-N-78 and KEK-StV-Nr. 47/14, respectively. Strains originating from Msambweni, Kenya, were obtained from human faecal samples covered by KNH-ERC/A/337, the University of KwaZulu-Natal (BF121/08), the ETH Zurich (EK 2009-N-53) and registered at clinicaltrials.gov (NCT01111864)29. Informed consent was obtained from all study participants and/or their legal guardians. The study was conducted in accordance with the Declaration of Helsinki37.
Origin and overview of bacterial strains and criteria for isolate selection
This study was based on the pool of 130 SBSEC isolates obtained during the colonoscopy study performed at Kenyatta National Hospital (KNH), Nairobi, Kenya involving 273 participants recruited from 2013 until 201527. The 130 isolates were obtained from 56 out of 228 unique study participants. These isolates represent the first human SBSEC isolates of African origin to be analysed for their phylogeny and evolutionary relationships. For each isolate, corresponding participant hospital diagnosis of colon disorders and responses from a guided interview on lifestyle, socio-demographics and dietary habits is available27.
The criteria for isolate selection among KNH human isolates for this study were based on patients’ colonic disorder, FDP consumption status, residence (urban or rural) as well as isolate rep-PCR fingerprint and presence of lacS/lacZ marker gene. This detailed selection was performed to achieve a wide variety of isolates with different patient backgrounds. A total of 40 SBSEC and Sii from 33 different patients were thus selected. Species and subspecies status of these isolates was previously determined using a 16S rRNA gene-based assay and partial groEL sequencing21,24,27. The species distribution was 34 Sii (30 patients), one Sgm (one patient), four Sgp (four patients) and one S. lutetiensis (one patient) of which one patient each was determined as combined carrier of Sii and Sgm, Sii and Sgp or Sii and S. lutetiensis.
As an outgroup to the hospital-collected samples, Sii (n = 7) and Sgm (n = 1) isolates from faecal samples of eight infants participating in a gut microbiota study in Mswambeni, Kenya were incorporated29. In addition, further SBSEC and Sii isolates were incorporated to increase the diversity for subsequent comparisons. Among these additional isolates, Sii and Sgm dairy isolates were previously collected from traditionally fermented dairy products in Kenya (Sii n = 18), Somalia (Sii n = 2) and Côte d’Ivoire (Sii n = 8, Sgm n = 1)20,21. Further diversity among the isolate pool was achieved through SBSEC isolates kindly donated by other research collections including single isolates from external culture collections from hospitals in Spain (n = 2; Sii P-9 and JIM9345)32, Taiwan (n = 1, Sii NTUH-8)6, animal bovine milk isolate from France (n = 1; Sii ANSES 6953)33, Italian Grana cheese (n = 1; Sii 42)31 and Mexican fermented maize (n = 1, Sii pozol)30. Overall, 83 SBSEC isolates were processed in this study and included in the SBSEC MLST scheme.
For validation of assays, the following reference strains were obtained for this study from the Culture Collection of the Laboratory of Food Biotechnology (FBT) of ETH Zurich, Zurich, Switzerland, Deutsche Sammlung von Mikroorganismen und Zellkulturen (DSMZ) (Braunschweig, Germany) and the Culture Collection of the University of Gothenburg (CCUG) (Gothenburg, Sweden): Sii CJ1825, Streptococcus thermophilus DSM20259 (yoghurt isolate, DSMZ), Sgg DSM16831T 38, Enterococcus faecium DSM20477T39 and Sii CCUG43820T 3.
Growth media and growth conditions
Cultivation of SBSEC strains was performed overnight in Brain Heart Infusion broth (BHI, Biolife, Milan, Italy) at 37 oC under aerobic conditions. The purity of strains was evaluated by streak plating onto Mitis Salivarius agar medium (Becton Dickinson, Allschwil, Switzerland) under aerobic incubation for 1–2 days at 37 °C. The stock cultures were stored at −80 °C in BHI broth supplemented with 33% (v/v) glycerol.
Molecular characterization of SBSEC isolates
General DNA isolation procedures, PCR conditions, downstream processing and sequencing of amplified DNA fragments
DNA from single colonies was extracted by lysis and storage in an EDTA, Trizma-base, Triton-X 100-based buffer24,40. All PCR assays were performed using 2× concentrated PCR Master Mix (Thermo Scientific, St. Leon-Rot, Germany), 1 µM primer concentration and sterile ddH2O to a final volume of 20 µL21. All primers were obtained from Microsynth (Balgach, Switzerland). Purification of DNA amplicons was performed using the GFX PCR DNA and Gel Band Purification Kit (GE Healthcare, Buckinghamshire, UK) or direct filtration by Microsynth AG (Switzerland). Sanger sequencing of purified DNA amplicons was performed at GATC (Konstanz, Germany) and Microsynth AG (Switzerland) using the same primers as for PCR amplification.
Screening for dairy adaptation marker genes lacS and lacZ
All strains were subjected to lacS and lacZ-specific PCR assays to determine the presence of a lacS/lacZ-mediated lactose uptake system using lacS and lacZ as marker genes for dairy adaption24.
SBSEC MLST assay
SBSEC and Sii phylogeny was assessed using the SBSEC MLST assay24. A total of 83 SBSEC and Sii strains were processed and submitted to the public repository on pubmlst.org. Processing of DNA sequencing chromatograms was performed in CLC Genomic workbench 7.5 (Qiagen Aarhus A/S, Denmark). Sequence quality trimming was performed with a parameter setting of 0.1 followed by paired-read assembly per strain and loci. Curation, allele and ST assignment was performed as previously described using MEGA7.0 and START224.
Analysis of phylogeny was performed in MEGA7.0 and START2 based on the Maximum likelihood algorithm (200 bootstrap replications) to construct phylogenetic trees using the concatenated sequence-based MLST data of all 10 alleles for all 157 SBSEC isolates.
To investigate the genetic diversity of the ten housekeeping genes and to elucidate the impact of the newly added strains, key performance indices such as dN/dS ratio, index of association (IA and IAS for the standardized index) were calculated using START224. For dN/dS, N is the number of non-synonymous sites (nucleotide substitutions change the amino acid) and S is the number of synonymous sites (nucleotide substitutions do not change the amino acid). dN is the proportion of non-synonymous sites and dS the proportion of synonymous sites. Investigating the dN/dS ratio provides information about the degree of selection; in our case the degree of selection within the ten housekeeping genes. A ratio with a value less than 1 indicates that the respective gene is under stabilizing selective pressure, which means that the population mean stabilizes and the genetic diversity decreases41. IA and IAS quantify the amount of linkage disequilibrium between the alleles of the ten loci. The index of association has an expected value of 0, which means that no association between the different loci exists and indicates free recombination42. IA and IAS were calculated using a single isolate per ST to avoid bias. Clonal complex (CC) calculations were performed in Phyloviz 2.0 and eBURSTv3 for groups of STs sharing 7 or more out of 10 alleles with at least one other member of this group24. Simpson’s Index of Diversity (SID) was calculated for allele profiles to indicate infinite diversity for values of 1 and no diversity for values of 043.
Data availability
All data generated or analysed during this study are included in this published article and pubmlst.org. Additional information and access to raw data is available from the corresponding author on reasonable request.
Electronic supplementary material
Acknowledgements
This study was funded by the ERAfrica program theme “New Ideas” project No ERAfrica_NI_021 (Swiss Partners funded by the Swiss National Science Foundation Project No IZEAZ0_154308). Further fundings were received from the UBS Optimus Foundation, Switzerland. This paper is partially based also on work supported by the DELTAS Africa Initiative [Afrique One-ASPIRE /DEL-15-008]. Afrique One-ASPIRE is funded by a consortium of donor including the African Academy of Sciences (AAS) Alliance for Accelerating Excellence in Science in Africa (AESA), the New Partnership for Africa’s Development Planning and Coordinating (NEPAD) Agency, the Wellcome Trust [107753/A/15/Z] and the UK government. The funders had no role in study design, data collection, analysis, interpretation, decision to publish, or preparation of the manuscript. The authors would like to acknowledge invaluable support by Jonathan Oloo, Mercy Kathio John, Ms. Stella Kibet, Ms. Miriam Arandi, Geoffrey Nzili, and Kenyatta National Hospital staff; Jane Mwangi, Edward Kanata, Ann Salo and Jackie Ngige (Nurses), Dr. Edna Kamau, Dr. Stephen Onyango and Prof. Elly Ogutu (medical doctors/Gastroenterologists); and Prof. Christophe Lacroix, PD Dr. Esther Schelling and Prof. Jakob Zinsstag for contributing to this study. The authors greatly acknowledge strains or DNA received for analysis from Carmen Wacher (Departamento de Alimentos y Biotecnología, Facultad de Química, Mexico), Diego Mora (Dipartimento di Scienze e Tecnologie Alimentari e Microbiologiche, sezione Microbiologia Industriale Universita degli Studi di Milano, Milano, Italy), Marisa Haenni/Jean-Yves Madec (Anses - Agence nationale de sécurité sanitaire de l’alimentation, de l’environnement et du travail, France), Po-Ren Hsueh (Department of Internal Medicine, National Taiwan University Hospital, National Taiwan University College of Medicine, Taipei, Taiwan) and Rosa del Campo/Beatriz Romero Hernández (Servicio de Microbiología, Hospital Universitario Ramón y Cajal, Madrid, Spain).
Author Contributions
D.W.M.K., N.O. and T.S. performed the experiments. C.J. and L.M. designed and supervised the experiments. All authors contributed to data analysis and interpretation under the lead of C.J., J.H., D.W.M.K. and L.M. The main draft of the manuscript was written by D.W.M.K. Critical review and major revisions of the manuscript were performed by D.W.M.K., W.K.M., G.N.L., B.K., P.R., B.B., L.M., J.H. and C.J. under the coordination of C.J. All authors read and approved the final manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-27383-4.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Booijink CC, et al. High temporal and inter-individual variation detected in the human ileal microbiota. Environ. Microbiol. 2010;12:3213–3227. doi: 10.1111/j.1462-2920.2010.02294.x. [DOI] [PubMed] [Google Scholar]
- 2.Jans C, Meile L, Lacroix C, Stevens MJA. Genomics, evolution, and molecular epidemiology of the Streptococcus bovis/Streptococcus equinus complex (SBSEC) Infect., Genet. Evol. 2015;33:419–436. doi: 10.1016/j.meegid.2014.09.017. [DOI] [PubMed] [Google Scholar]
- 3.Schlegel L, Grimont F, Ageron E, Grimont PAD, Bouvet A. Reappraisal of the taxonomy of the Streptococcus bovis/Streptococcus equinus complex and related species: description of Streptococcus gallolyticus subsp. gallolyticus subsp. nov., S. gallolyticus subsp. macedonicus subsp. nov. and S. gallolyticus subsp. pasteurianus subsp. nov. Int. J. Syst. Evol. Microbiol. 2003;53:631–645. doi: 10.1099/ijs.0.02361-0. [DOI] [PubMed] [Google Scholar]
- 4.Beck M, Frodl R, Funke G. Comprehensive study of strains previously designated Streptococcus bovis consecutively isolated from human blood cultures and emended description of Streptococcus gallolyticus and Streptococcus infantarius subsp. coli. J. Clin. Microbiol. 2008;46:2966–2972. doi: 10.1128/JCM.00078-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chirouze C, et al. Streptococcus bovis/Streptococcus equinus complex fecal carriage, colorectal carcinoma, and infective endocarditis: a new appraisal of a complex connection. Eur. J. Clin. Microbiol. Infect. Dis. 2013;32:1171–1176. doi: 10.1007/s10096-013-1863-3. [DOI] [PubMed] [Google Scholar]
- 6.Sheng WH, Chuang YC, Teng LJ, Hsueh PR. Bacteraemia due to Streptococcus gallolyticus subspecies pasteurianus is associated with digestive tract malignancies and resistance to macrolides and clindamycin. J. Infect. 2014;69:145–153. doi: 10.1016/j.jinf.2014.03.010. [DOI] [PubMed] [Google Scholar]
- 7.Jans, C. & Boleij, A. The road to infection: Host-microbe interactions defining the pathogenicity of Streptococcus bovis/Streptococcusequinus complex members Front. Microbiol. 9, 603 (2018). [DOI] [PMC free article] [PubMed]
- 8.Ben-Chetrit E, Wiener-Well Y, Kashat L, Yinnon AM, Assous MV. Streptococcus bovis new taxonomy: does subspecies distinction matter? Eur. J. Clin. Microbiol. Infect. Dis. 2017;36:387–393. doi: 10.1007/s10096-016-2814-6. [DOI] [PubMed] [Google Scholar]
- 9.Tjalsma H, Boleij A, Marchesi JR, Dutilh BE. A bacterial driver-passenger model for colorectal cancer: beyond the usual suspects. Nat. Rev. Microbiol. 2012;10:575–582. doi: 10.1038/nrmicro2819. [DOI] [PubMed] [Google Scholar]
- 10.Corredoira-Sánchez J, et al. Association between bacteremia due to Streptococcus gallolyticus subsp. gallolyticus (Streptococcus bovis I) and colorectal neoplasia: a case-control study. Clin. Infect. Dis. 2012;55:491–496. doi: 10.1093/cid/cis434. [DOI] [PubMed] [Google Scholar]
- 11.Corredoira J, et al. Characteristics of Streptococcus bovis endocarditis and its differences with Streptococcus viridans endocarditis. Eur. J. Clin. Microbiol. Infect. Dis. 2008;27:285–291. doi: 10.1007/s10096-007-0441-y. [DOI] [PubMed] [Google Scholar]
- 12.Corredoira J, et al. Advanced intestinal cancer associated with Streptococcus infantarius (former S. bovis II/1) sepsis. Int. J. Clin. Pract. 2013;67:1358–1359. doi: 10.1111/ijcp.12190. [DOI] [PubMed] [Google Scholar]
- 13.Schlegel L, et al. Streptococcus infantarius sp. nov., Streptococcus infantarius subsp. infantarius subsp. nov. and Streptococcus infantarius subsp. coli subsp. nov., isolated from humans and food. Int. J. Syst. Evol. Microbiol. 2000;50:1425–1434. doi: 10.1099/00207713-50-4-1425. [DOI] [PubMed] [Google Scholar]
- 14.Corredoira JC, et al. Clinical characteristics and significance of Streptococcus salivarius bacteremia and Streptococcus bovis bacteremia: a prospective 16-year study. Eur. J. Clin. Microbiol. Infect. Dis. 2005;24:250–255. doi: 10.1007/s10096-005-1314-x. [DOI] [PubMed] [Google Scholar]
- 15.Corredoira J, Alonso MP, Coira A, Varela J. Association between Streptococcus infantarius (formerly S. bovis II/I) bacteremia and noncolonic cancer. J. Clin. Microbiol. 2008;46:1570. doi: 10.1128/JCM.00129-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lazarovitch T, et al. The relationship between the new taxonomy of Streptococcus bovis and its clonality to colon cancer, endocarditis, and biliary disease. Infection. 2013;41:329–337. doi: 10.1007/s15010-012-0314-x. [DOI] [PubMed] [Google Scholar]
- 17.Lee RA, et al. Geographical difference of disease association in Streptococcus bovis bacteraemia. J. Med. Microbiol. 2003;52:903–908. doi: 10.1099/jmm.0.05199-0. [DOI] [PubMed] [Google Scholar]
- 18.Ruoff KL, Miller SI, Garner CV, Ferraro MJ, Calderwood SB. Bacteremia with Streptococcus bovis and Streptococcus salivarius: clinical correlates of more accurate identification of isolates. J. Clin. Microbiol. 1989;27:305–308. doi: 10.1128/jcm.27.2.305-308.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chow J, Tang H, Mazmanian SK. Pathobionts of the gastrointestinal microbiota and inflammatory disease. Curr. Opin. Immunol. 2011;23:473–480. doi: 10.1016/j.coi.2011.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jans C, Bugnard J, Njage PMK, Lacroix C, Meile L. Lactic acid bacteria diversity of African raw and fermented camel milk products reveals a highly competitive, potentially health-threatening predominant microflora. LWT-Food Sci. Technol. 2012;47:371–379. doi: 10.1016/j.lwt.2012.01.034. [DOI] [Google Scholar]
- 21.Jans C, et al. Prevalence and comparison of Streptococcus infantarius subsp. infantarius and Streptococcus gallolyticus subsp. macedonicus in raw and fermented dairy products from East and West Africa. Int. J. Food Microbiol. 2013;167:186–195. doi: 10.1016/j.ijfoodmicro.2013.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Abdelgadir W, Nielsen DS, Hamad S, Jakobsen M. A traditional Sudanese fermented camel’s milk product, Gariss, as a habitat of Streptococcus infantarius subsp. infantarius. Int. J. Food Microbiol. 2008;127:215–219. doi: 10.1016/j.ijfoodmicro.2008.07.008. [DOI] [PubMed] [Google Scholar]
- 23.Wullschleger S, et al. Analysis of lactic acid bacteria communities and their seasonal variations in a spontaneously fermented dairy product (Malian fènè) by applying a cultivation/genotype-based binary model. Int. Dairy J. 2013;29:28–35. doi: 10.1016/j.idairyj.2012.08.001. [DOI] [Google Scholar]
- 24.Jans C, et al. Phylogenetic, epidemiological and functional analyses of the Streptococcus bovis/Streptococcus equinus complex through an overarching MLST scheme. BMC Microbiol. 2016;16:117. doi: 10.1186/s12866-016-0735-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jans C, et al. Comparative genome analysis of Streptococcus infantarius subsp. infantarius CJ18, an African fermented camel milk isolate with adaptations to dairy environment. BMC Genomics. 2013;14:200. doi: 10.1186/1471-2164-14-200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jans C, et al. Novel Streptococcus infantarius subsp. infantarius variants harboring lactose metabolism genes homologous to Streptococcus thermophilus. Food Microbiol. 2012;31:33–42. doi: 10.1016/j.fm.2012.02.001. [DOI] [PubMed] [Google Scholar]
- 27.Kaindi DWM, et al. Investigating the association between African spontaneously fermented dairy products, faecal carriage of Streptococcus infantarius subsp. infantarius and colorectal adenocarcinoma in Kenya. Acta Trop. 2017;178:10–18. doi: 10.1016/j.actatropica.2017.10.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jans C, et al. African fermented dairy products – overview of predominant technologically important microorganisms focusing on African Streptococcus infantarius variants and potential future applications for enhanced food safety and security. Int. J. Food Microbiol. 2017;250:27–36. doi: 10.1016/j.ijfoodmicro.2017.03.012. [DOI] [PubMed] [Google Scholar]
- 29.Jaeggi T, et al. Iron status and systemic inflammation, but not gut inflammation, strongly predict gender-specific concentrations of serum hepcidin in infants in rural Kenya. PLoS One. 2013;8:e57513. doi: 10.1371/journal.pone.0057513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Díaz-Ruiz G, Guyot JP, Ruiz-Teran F, Morlon-Guyot J, Wacher C. Microbial and physiological characterization of weakly amylolytic but fast-growing lactic acid bacteria: a functional role in supporting microbial diversity in pozol, a Mexican fermented maize beverage. Appl. Environ. Microbiol. 2003;69:4367–4374. doi: 10.1128/AEM.69.8.4367-4374.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mora D, Ricci G, Guglielmetti S, Daffonchio D, Fortina MG. 16S–23S rRNA intergenic spacer region sequence variation in Streptococcus thermophilus and related dairy streptococci and development of a multiplex ITS-SSCP analysis for their identification. Microbiology. 2003;149:807–813. doi: 10.1099/mic.0.25925-0. [DOI] [PubMed] [Google Scholar]
- 32.Romero B, et al. Reidentification of Streptococcus bovis isolates causing bacteremia according to the new taxonomy criteria: still an issue? J. Clin. Microbiol. 2011;49:3228–3233. doi: 10.1128/JCM.00524-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chuzeville S, Puymège A, Madec JY, Haenni M, Payot S. Characterization of a new CAMP factor carried by an integrative and conjugative element in Streptococcus agalactiae and spreading in streptococci. PLoS One. 2012;7:e48918. doi: 10.1371/journal.pone.0048918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Papadimitriou K, et al. Comparative genomics of the dairy isolate Streptococcus macedonicus ACA-DC 198 against related members of the Streptococcus bovis/Streptococcus equinus complex. BMC Genomics. 2014;15:272. doi: 10.1186/1471-2164-15-272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Harris VC, et al. Significant correlation between the infant gut microbiome and rotavirus vaccine response in rural Ghana. J. Infect. Dis. 2017;215:34–41. doi: 10.1093/infdis/jiw518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Maiden MC. Multilocus sequence typing of bacteria. Annu. Rev. Microbiol. 2006;60:561–588. doi: 10.1146/annurev.micro.59.030804.121325. [DOI] [PubMed] [Google Scholar]
- 37.World Medical Association. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA310, 2191–2194 (2013). [DOI] [PubMed]
- 38.Osawa R, Fujisawa T, Sly LI. Streptococcus gallolyticus sp. nov.; gallate degrading organisms formerly assigned to Streptococcus bovis. Syst. Appl. Microbiol. 1995;18:74–78. doi: 10.1016/S0723-2020(11)80451-0. [DOI] [Google Scholar]
- 39.Schleifer KH, Kilpper-Bälz R. Transfer of Strepcococcus faecalis and Streptococcus faecium to the genus Enterococcus nom. rev. as Enterococcus faecalis comb. nov. and Enterococcus faecium comb. nov. Int. J. Syst. Bacteriol. 1984;34:31–34. doi: 10.1099/00207713-34-1-31. [DOI] [Google Scholar]
- 40.Goldenberger D, Perschil I, Ritzler M, Altwegg M. A simple universal DNA extraction procedure using SDS and proteinase K is compatible with direct PCR amplification. PCR Methods Appl. 1995;4:368–370. doi: 10.1101/gr.4.6.368. [DOI] [PubMed] [Google Scholar]
- 41.Nei M, Gojobori T. Simple methods for estimating the numbers of synonymous and nonsynonymous nucleotide substitutions. Mol. Biol. Evol. 1986;3:418–426. doi: 10.1093/oxfordjournals.molbev.a040410. [DOI] [PubMed] [Google Scholar]
- 42.Smith JM, Smith NH, O’Rourke M, Spratt BG. How clonal are bacteria? Proc. Natl. Acad. Sci. USA. 1993;90:4384–4388. doi: 10.1073/pnas.90.10.4384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Grundmann H, Hori S, Tanner G. Determining confidence intervals when measuring genetic diversity and the discriminatory abilities of typing methods for microorganisms. J. Clin. Microbiol. 2001;39:4190–4192. doi: 10.1128/JCM.39.11.4190-4192.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data generated or analysed during this study are included in this published article and pubmlst.org. Additional information and access to raw data is available from the corresponding author on reasonable request.