Abstract
Background
Hearing voices can be a distressing and disabling experience for some, whilst it is a valued experience for others, so-called ‘healthy voice-hearers’. Cognitive models of psychosis highlight the role of memory, appraisal and cognitive biases in determining emotional and behavioural responses to voices. A memory bias potentially associated with distressing voices is the Overgeneral Memory Bias (OGM), namely the tendency to recall a summary of events rather than specific occasions. It may limit access to autobiographical information that could be helpful in re-appraising distressing experiences, including voices.
Methods
We investigated the possible links between OGM and distressing voices in psychosis by comparing 3 groups: 1) clinical voice-hearers (N=39), 2) non-clinical voice-hearers (N=35) and 3) controls without voices (N=77) on a standard version of the autobiographical memory test (AMT). Clinical and non-clinical voice-hearers also completed a newly adapted version of the task, designed to assess voices-related memories (vAMT).
Results
As hypothesised, the clinical group displayed an OGM bias by retrieving fewer specific autobiographical memories on the AMT compared to both the non-clinical and control groups, who did not differ from each other. The clinical group also showed an OGM bias in recall of voice-related memories on the vAMT, compared to the non-clinical group.
Conclusions
Clinical voice-hearers display an OGM bias when compared to non-clinical voice-hearers on both general and voices-specific recall tasks. These findings have implications for the refinement and targeting of psychological interventions for psychosis.
Keywords: psychosis, auditory hallucinations, autobiographical memory, cognitive behavioural therapy
In the context of a psychiatric diagnosis, hearing voices is labelled as a psychotic symptom commonly known as auditory verbal hallucinations. However, there is an increasing body of evidence that hearing voices can also occur in the general population, is not always associated with distress, and does not necessarily indicate a current or future need for psychiatric care (for review see (Baumeister et al., 2017, Johns et al., 2014). What determines whether hearing voices is distressing and disabling for the individual experiencing them? Cognitive models of psychosis highlight the importance of appraisals in determining emotional and behavioural consequences to voices (Garety et al., 2001). Specifically, Chadwick and Birchwood (1994) highlighted four key beliefs: power, malevolence/benevolence, omniscience, and controllability. They found that people who believed their voices were very powerful, knowledgeable, and uncontrollable reacted with fear and resistance when the voices were perceived to be malevolent in nature (e.g. commands from an evil spirit). Similarly, studies have also found that people who made benevolent attributions about their voices were more likely to engage with their voices (Chadwick & Birchwood, 1995, Peters et al., 2012, Sayer et al., 2000).
According to cognitive models of positive psychotic symptoms (Garety et al., 2001) cognitive biases in reasoning and attributional processes contribute to the maintenance of distressing beliefs about voices. Findings that healthy and clinical voice-hearers differ on the Cognitive Biases Questionnaire for psychosis (Peters et al., 2014) provides some, albeit indirect, evidence for this claim (Daalman et al., 2013). Memory functioning is a further cognitive domain that may contribute to maintenance of beliefs about voices: a recent meta-analysis of 14 studies of memory functioning in schizophrenia reported a large effect size (g=-0.97) for reduced specificity in autobiographical recall (Berna et al., 2015). However, the diagnosis of schizophrenia covers a heterogeneous range of symptoms, and consequently these studies tell us little about the association between specific memory bias and specific psychotic symptoms. There have been only a few studies to date that have taken a symptom-specific approach, mostly examining the links between the overgeneral memory bias (OGM) and delusions. The OGM is defined as the tendency to recall, when prompted, a summary of events rather than a specific occasion (Williams, 1996). The OGM bias was first identified in people who had recently attempted suicide (Williams & Broadbent, 1986) and later in people with a diagnosis of major depressive disorder (for review, see Williams et al. (2007)). A difficulty in recalling specific autobiographical memories is thought to be clinically relevant in depression as it is linked to impairment on problem-solving tasks (Goddard et al., 1996) and difficulties in imagining future events in detail (Williams et al., 1996). Patients with a diagnosis of schizophrenia presenting with delusions were found to recall fewer specific memories compared to those without delusions (Baddeley et al., 1996), and compared to patients with depression and healthy controls (Kaney et al., 1999, Vorontsova et al., 2013). Vorontsova et al. further demonstrated this was the case regardless of concurrent depression.
Similarly, for people who experience distressing voices, a difficulty in recalling specific memories both in everyday life, and also specifically in relation to their voices, would be hypothesised to perpetuate poor problem solving, low mood and hopelessness, as well as helping to maintain a perception of voices as omnipotent, all-knowing and uncontrollable, by limiting access to disconfirmatory information. This hypothesised role in belief-maintenance is consistent with clinical observation, in that people who are distressed by voices often struggle to remember disconfirmatory evidence that might be helpful in both changing their beliefs about the power of their voices and increasing a sense of personal control and autonomy. Although the OGM bias has been well-established in various clinical groups, less is known about the underlying mechanisms of this process. It has been suggested that overgeneral memory recall arises from a premature termination of the memory search, when top-down retrieval is stopped at too high a level when only general categories of memory have been accessed (Conway & Pleydell-Pearce, 2000). Conway and Pleydell-Pearce suggest that this truncated search is the result of a passive avoidance strategy to avoid emotionally distressing memories. Williams and colleagues conceptualise this within their model of overgeneral memory as functional avoidance, because it protects against short-term emotional disturbance, whilst being an ineffectual emotion regulation strategy in the longer-term (Williams et al., 2007). Based on this theory, we therefore expected that clinical voice hearers, whose voices are known to be more distressing and disturbing (Daalman et al., 2011), would be more likely to be over-general in autobiographical memory recall in general compared with non-clinical groups, and to an even greater degree in relation to voices-specific memories.
The main aim of the present study was therefore to test whether an OGM bias distinguishes people with distressing voices from those who are healthy voice-hearers, adding to our understanding of what may maintain voices that cause distress. Comparing healthy and clinical individuals with similar anomalous experiences is uniquely helpful in disentangling the presence of voices, which is usually viewed solely as the symptom of a psychotic illness, from the resulting distress and disability. A secondary aim was to pilot an adapted ‘voices’ version of the standard Autobiographical Memory Test (AMT), to explore differences in recall on the general and voices-specific AMT.
The three hypotheses for the study were as follows:-
Clinical voice hearers will show a greater overgeneral memory bias (OGM) compared to both the non-clinical voice hearers and controls on the standard task (AMT), with the latter groups not differing from each other.
Clinical voice hearers will show a greater overgeneral memory bias (OGM) compared to non-clinical voice hearers on the voices-specific task (vAMT).
Clinical voice hearers will show a relatively greater exacerbation of the OGM on the voices-specific task (vAMT) compared to the standard task (AMT), than the non-clinical voice hearers.
Method
Participants
Three groups of participants were recruited across two sites (South London and Bangor, North Wales): 1) people who hear voices with a diagnosis of psychosis-spectrum disorder (clinical) (N=39), 2) people who hear voices but are not distressed and have never needed mental health care (non-clinical) (N=35), and 3) controls (N=77). Participants were recruited as part of the wider UNIQUE (Unusual Experiences Inquiry study) which investigated cognitive and social processes in the pathway to psychosis by comparing clinical and non-clinical groups of people with persistent psychotic experiences (Peters et al., 2016). The current sample (N=151) was selected from the larger sample (N=259) on the basis of whether or not the participants reported hearing voices currently. Hearing voices was defined by reporting at least occasional voices on the screening measures for voices (Scale for the Assessment of Positive Symptoms (SAPS; Andreasen (1984)). The OGM bias study was planned in advance, and the data presented in this paper are not reported elsewhere.
1). Clinical group
Inclusion criteria were i) established diagnosis of psychosis-spectrum disorder (ICD-10 diagnoses F20-39), ii) psychotic symptoms in the form of voices (score of ≥2 on auditory hallucinations item on the SAPS) iii) no history of cognitive behaviour therapy for psychosis (defined by the National Institute for Health and Care Excellence as a minimum of 16 planned sessions over at least 6 months; NICE guidelines, update 2014). Participants were recruited from both inpatient and outpatient services, and from urban (South London and Maudsley NHS Foundation Trust) and rural (Betsi Cadwaldr University Health Board, North Wales) locations. The most common diagnosis was schizophrenia (F20; N=24). Other diagnoses included schizoaffective disorder (F25; N=6), psychosis NOS (F28-29; N=3), acute and transient psychosis (F23; N=2), bipolar affective disorder (F31; N=2) and other affective disorders (F32-34; N=2). All participants were prescribed anti-psychotic medication (most commonly an atypical), and they reported an average of 4.5 previous hospital admissions.
2). Non-clinical group
Inclusion criteria were (i) reported one or more psychotic experiences (secondary item) on the Psychosis Screening Questionnaire (PSQ; Bebbington and Nayani (1995)), and ‘occasional’ (at least monthly) experiences of any positive and Schneiderian first-rank symptom on the Unusual Experiences Screening Questionnaire (UESQ), within the last month and in the absence of drug use/clear consciousness; ii) enduring psychotic experiences in the form of voices (score of >2 on auditory hallucinations item of SAPS), but in the absence of diagnosis or treatment for a psychotic disorder, iii) experiences started at least 5 years ago (to avoid inclusion of prodromal individuals), iv) scores less than 2 (‘unmet need’) on the Camberwell Assessment of Need ‘psychological distress’ (in relation to voices) dimension (CAN; Phelan et al. (1995)). Participants were recruited from specialist sources in the community (see Peters et al. (2016) for further details).
3). Controls
Inclusion criteria were i) no anomalous experiences (endorsed no items on UESQ and PSQ) or psychosis diagnosis ii) scoring below 1 standard deviation of the mean (score of 15) of the ‘Unusual Experiences’ sub-scale of the Oxford-Liverpool Inventory of Feelings and Experiences (O-LIFE; Mason & Claridge (2006)). Participants were recruited through general community settings.
Exclusion criteria for all groups were: i) less than 18 years old ii) insufficient command of English iii) neurological history, head injury or epilepsy iv) primary substance dependence.
Procedure
Ethical approval
Ethical approval was obtained from NRES Committee London – Westminster (Reference 12/LO/0766), and from the South London & Maudsley/Institute of Psychiatry (SLAM/IoP) Research and Development (R&D) Office (reference: R&D2012/047), and the BCUHB R&D Office (reference: Jackson/LO/0766). Participants were screened by research workers (RWs) over the phone, or face to face in the case of clinical inpatients. Following written informed consent, participants completed the assessments and experimental tasks with the RWs as outlined below, in addition to other measures (not reported in this paper). At the end of the study participants were debriefed, given a small honorarium, and offered a one week follow-up phone call in case of any distress.
Design
The main independent variable consisted of 3 levels of group (between-participants factor). For the clinical and non-clinical groups only, there was a further within-participants factor for the AMT, with 2 levels of task (general vs. voices-specific memories). AMT scores (i.e., number of specific memories retrieved in response to cue words) was the dependent variable.
Experimental Tasks
All participants completed the AMT, administered in line with the standard procedure established by Williams and Broadbent (1986). Participants were given 60s to retrieve a specific autobiographical memory in response to ten cue words (defined as an event lasting a day or less, which occurred at a certain place and time even if the subject could not remember when). If the participant retrieved a memory that was not specific, they were given a prompt as follows:-“Can you think of a specific time in the past – one particular memory?” The prompt was repeated as required within the 60s time limit. In addition to the standard task, the clinical and non-clinical groups completed a novel version of the AMT, using the same prompt words and general procedure, but asking people to recall only memories relating to their voices (vAMT). The standard AMT was always completed first, since only two of the three groups (clinical and non-clinical) completed the vAMT. See supplementary materials for full task instructions.
Scoring and analysis
All responses were audio-taped and later transcribed verbatim. In line with standard scoring procedure (Williams & Dritschel, 1992) 1 point was given per item for which a specific memory was retrieved within the memory limit (range 0-10). Twelve participants were sampled by PJ for the purposes of calculating inter-rater reliability (3 per each of 4 RWs). These were stratified with 4 each from the clinical, non-clinical and control groups. Samples of both standard and voices-specific AMT were included. Inter-rater reliability was acceptable for all RWs, with kappa values ranging from 0.69-0.79 indicating “substantial agreement” (Landis & Koch, 1977).
Measures
Psychotic experiences
The Appraisals of Anomalous Experiences (AANEX; Brett et al. (2007)) semi-structured interview was used to elicit participants’ psychotic experiences. The first part of the interview (AANEX-Inventory, short form; Lovatt et al. (2010)) consists of 17 anomalous experiences that are rated for both presence and severity in the person’s lifetime and currently (within the last month). Each item is rated on a 3-point scale (1=not present; 2=unclear; 3=present). Possible total scores range from 17 to 51 for both lifetime and current experiences. This scale was not administered to the control group.
Screening measure for voices
The Scale for the Assessment of Positive Symptoms (SAPS; Andreasen (1984)) is a clinician-assessed, standardised scale which is widely used to assess positive symptoms, with scores for each item reflecting level of severity and frequency, ranging from ‘0’ (None) to ‘5’ (Severe). We defined “hearing voices” in the clinical and the non-clinical group as scoring ≥2 on the auditory hallucination item, which indicates at least occasional frequency of voices. This scale was not administered to the control group.
IQ measure
All participants completed a brief IQ measure (Wechsler Adult Intelligence Scale – 3rd edition; WAIS-III; Wechsler (1997)). The WAIS-III is a widely-used test of adult intelligence, with good normative data available and established reliability and validity. Four sub-tests of the WAIS were used in order to calculate a full-scale IQ (digit symbol, arithmetic, block design and information).
Depression measure
All participants completed the Beck Depression Inventory – 2nd edition (BDI-II; Beck et al. (1996)). The BDI-II is a 21 item self-report measure of depressive symptoms. Each item is scored from 0-3, with a total score out of 63. Higher scores indicate more depressive symptoms. The BDI-II has well-established reliability and validity as a depression measure and is widely used in both research and clinical practice.
Data analysis
In line with standard practice (Williams et al., 2007) the primary data analysis was based on first memory response as the dependent variable, with a secondary analysis based on best memory response. A degree of skew in the distribution of the AMT data was tolerated given the robustness of the ANOVA to violation of the assumption of a normally distributed data-set (Schmider et al., 2010). Performance between the 3 groups (clinical, non-clinical and controls) on the standard AMT was compared using a one-way ANOVA, with post-hoc comparisons using the Games-Howell procedure (due to unequal sample sizes and non-homogeneity of variance). Performance between groups (clinical vs. non-clinical) and tasks (standard AMT vs. vAMT) was analysed using a mixed-effects 2X2 ANOVA.
Results
Participant Demographic and Clinical Characteristics
Demographic and clinical data are shown in Table 1. There was no statistically significant difference in age between the three groups, however the groups differed on gender, ethnicity, IQ, and employment status, as is typical for these samples (Ward et al., 2014; Brett et al., 2007). On psychotic symptoms measures, the clinical and non-clinical groups did not differ on the AANEX (both current and lifetime score), but the clinical group scored significantly higher on both the auditory hallucinations and global delusions items of the SAPS compared to the non-clinical group. A common concern when comparing clinical with non-clinical groups is that differences in general cognitive ability between groups may be a confounding variable in interpreting apparent differences on task performance. As would be expected, IQ differed significantly across the three groups (Table 1), with the clinical group scoring lower than both the non-clinical and control group, who did not differ from each other. However, a recent meta-analysis of memory functioning in schizophrenia (20 studies, N=571), found that memory specificity, the variable of interest in this study, was not related to any of several factors that influence cognitive functioning, including IQ, level of education and duration of illness (Berna et al., 2016). Nevertheless, we ran bivariate Pearson’s correlations between IQ and AMT score in the three groups separately to check whether IQ was related to task performance in this sample. In line with previous findings, IQ was not related to AMT score in any of the groups (controls (N=77): r=0.18, p=0.12; non-clinical (N=35): r=0.21, p=0.23; clinical (N=39) r=0.29; p=0.08). There was therefore no rationale for entering IQ as a covariate in the main analysis as there was no evidence it was a confounding variable on task performance. To address the question of whether task performance may also be affected by the additional presence of delusions (Vorontsova et al., 2013), we also ran bivariate Pearson’s correlations between SAPS global delusions score and AMT score in the clinical and non-clinical group. Delusions score was not related to AMT score in either group (non-clinical (N=35): r=0.02, p=0.90; clinical (N=39) r=0.13; p=0.44), therefore delusions score was also not entered as a co-variate. However, as the clinical group had substantially higher scores on depression than the other 2 groups, BDI scores were entered as a covariate in a secondary analysis as a potentially confounding variable, given the strong association between OGM and depression found in previous studies (Williams et al., 2007).
Table 1. Participant demographic and clinical characteristics (sig. differences in bold).
| Clinical (N=39) |
Non-Clinical (N=35) |
Controls (N=77) |
Statistics | |
|---|---|---|---|---|
| Age (mean) | 41 (range 21-65) | 45 (range 20-68) | 45 (range 21-76) | F2,148=1.59, p=0.207 |
|
Gender % - Male - Female |
64 36 |
26 74 |
31 69 |
χ2=15.0, df=2, p=0.001 (clinical>non-clinical=controls) |
|
Ethnicity % - White - BME1 |
56 44 |
89 11 |
92 8 |
χ2=23.9, df=2, p<0.001 (non-clinical=controls>cl inical) |
|
Currently in employment or education? % - Yes - No |
15 85 |
63 37 |
81 19 |
χ2=45.8, df=2, p<0.001 (controls>nonclinical >clinical) |
| IQ (Full-scale) (mean and (SD)) | 82 (13.4) | 105 (11.3) | 112 (16.6) |
F2,148=54.4, p<0.001 (non-clinical=controls>cl inical) |
| Beck Depression Inventory (mean and (SD)) | 26.2 (13.9) | 7.6 (7.5) | 6.0 (8.4)2 |
F2,147=56.9, p<0.001 (clinical>non-clinical=control) |
| Age of onset of voices (years) | 20 (8.9) | 14 (12.1) | U72=346, P<0.001 | |
| Duration of hearing voices (years) | 20 (13.7) | 30 (14.6) | t72=3.12, p=0.003 | |
| SAPS – auditory hallucinations (mean and (SD)) | 4.28 (1.03) | 2.83 (0.954) | U72=233, P<0.001 | |
| SAPS – global delusions rating | 3.69 (1.13) | 2.31 (1.47) | U72=318, P<0.001 | |
| AANEX – total lifetime (mean and (SD)) | 35.5 (5.7) | 35.7 (5.36) | t72=0.158, p=0.875 | |
| AANEX – total current (mean and (SD)) | 31.1 (6.04) | 30.5 (5.4) | t72= -0.402, p=0.689 |
BME=Black and minority ethnic background
Missing data for 1 participant
Autobiographical Memory Test (AMT) analysis
Hypothesis 1: Clinical voice hearers will show a greater overgeneral memory bias (OGM) compared to both the non-clinical voice hearers and controls on the standard task (AMT), with the latter groups not differing from each other.
A one-way ANOVA showed a significant effect of group (F(2,148)=13.32, p<0.001). As predicted, post-hoc comparisons indicated that the clinical group (mean =5.62, standard deviation (sd)=2.46) scored significantly lower than both the controls (mean=7.48, sd=2.14; p<0.001) and the non-clinical group (mean=8, sd=1.86; p<0.001), who did not differ from each other (p=0.40). The secondary analysis with best response on the AMT as the dependent variable rather than first response, showed the same pattern of results. Co-varying out depression (BDI score) did not change the results (F(2,146)=5.32, p=0.006).
Hypothesis 2:Clinical voice hearers will show a greater overgeneral memory bias (OGM) compared to non-clinical voice hearers on the voices-specific task (vAMT).
Four participants in the clinical group, and three in the non-clinical group did not complete the vAMT task, and were excluded. A two-way mixed factorial ANOVA, with task (standard vs vAMT) as the within-participants factor and group (non-clinical vs. clinical) as a between-participants variable revealed a significant main effect of group (F(1,65)=28.4, p<0.001), with AMT scores being higher in the non-clinical group (estimated marginal mean (EMM)=7.25, standard error (SE)=0.39) than the clinical group (EMM=4.39, SE=0.37).
Hypothesis 3:Clinical voice hearers will show a relatively greater exacerbation of the OGM on the voices-specific task (vAMT) compared to the standard task (AMT), than the non-clinical voice hearers.
The two-way mixed factorial ANOVA also showed there was a significant main effect of task on AMT scores (F(1,65)=47.7, p<0.001), with higher scores for the standard task (EMM=6.84, SE=0.27) than the vAMT task (EMM=4.79, SE=0.34). As predicted, the group x task interaction was also significant (F(1,65)=4.23, p=0.044): while both groups scored higher on the standard AMT than the vAMT, the relative difference was greater in the clinical group.
Discussion
The primary focus in the present study was to investigate the overgeneral memory (OGM) bias in clinical and non-clinical groups of voice hearers, and controls, using the standard autobiographical memory test (AMT). As predicted, the clinical group retrieved fewer specific memories on both the standard AMT and vAMT than the non-clinical group, and fewer specific memories on the standard task than the control group, thereby displaying an OGM bias in relation to both general and voices-specific autobiographical memories. Furthermore, the clinical group showed a relatively greater OGM bias when recalling voices-specific memories compared to the non-clinical group. Also as hypothesised, there was no evidence of impairment in autobiographical memory recall in the non-clinical group in comparison with the control group. Moreover, in line with a recent meta-analysis of memory functioning in schizophrenia (Berna et al., 2016), the OGM bias seen in the clinical group was not attributable to observed differences in IQ (IQ and standard AMT scores were unrelated in all three groups); nor was it due to differences in delusions or depression scores.
These findings suggest that the OGM bias is not related to hearing voices per se. However, the clinical group showed a clear bias towards overgeneral autobiographical recall. The OGM bias may therefore contribute to problematic voices by the maintenance of unhelpful beliefs about voices, by potentially limiting access to vital information that might help to disconfirm or counter their beliefs about their voices, for example that voices must always be obeyed. Models of overgeneral memory suggest that the memory search is sometimes terminated prematurely as a way of avoiding emotionally disturbing memories (Williams et al., 2007). The clinical group may use this avoidance strategy as an emotion-regulation technique more frequently compared to non-clinical groups (Brett et al., 2007), and particularly for voices-related memories that are likely to be more distressing.
This study successfully builds on previous work comparing clinical and non-clinical groups of people with psychotic experiences in order to highlight key processes which may protect against the development of need-for-care (Brett et al., 2014, Daalman et al., 2013, Gaynor et al., 2013, Lovatt et al., 2010, Peters et al., 2017, Ward et al., 2014). In terms of the comparability of the clinical and non-clinical groups in their experience of hearing voices, there were some observed similarities (e.g. AANEX current & lifetime scores) and differences between the groups (e.g. longer duration of hearing voices in the non-clinical group). These group differences are consistent with the analysis of the larger data-set for the UNIQUE study (Peters et al., 2016), and are likewise noted in the review by Johns et al. (2014) and Baumeister et al. (2017). As would be predicted, the clinical group scored more highly on the depression measure compared to both the non-clinical and control groups, which did not differ from each other. However, we found that there was still a significant difference on AMT score even after controlling for depression, showing that the observed group differences were not attributable to group differences in depression alone. This is consistent with another recent study on autobiographical memory impairment in psychosis, which also found that patients retrieved fewer specific memories compared with controls even after controlling for depression score (Ricarte et al., 2014). However, we recognise the methodological challenges arising from the fact that clinical and non-clinical individuals do show differences in a range of demographic and clinical variables, which are inherent to their group status. For example, naturally occurring group differences include variables that reflect established risk factors for need-for-care, such as low IQ/poorer pre-morbid functioning and ethnicity (Coid et al., 2008, Kendler et al., 2015, Kirkbride et al., 2012), and/or are sequelae of group status (e.g. impaired functioning, lack of education, and anxiety). Our approach has been to draw on empirical evidence on plausible confounders on task performance, and to examine each in turn rather than attempting to simply ‘control’ for all group differences. In this case, we have examined the effect of IQ, depression, and delusions and found that none of them accounted for the differences in task performance between groups. However, given these methodological limitations, our findings should be interpreted with an appropriate degree of caution, and placed in the wider context of previous findings in the field comparing clinical and non-clinical voices hearers on experimental tasks (Underwood et al., 2016, Ward et al., 2017, Ward et al., 2014).
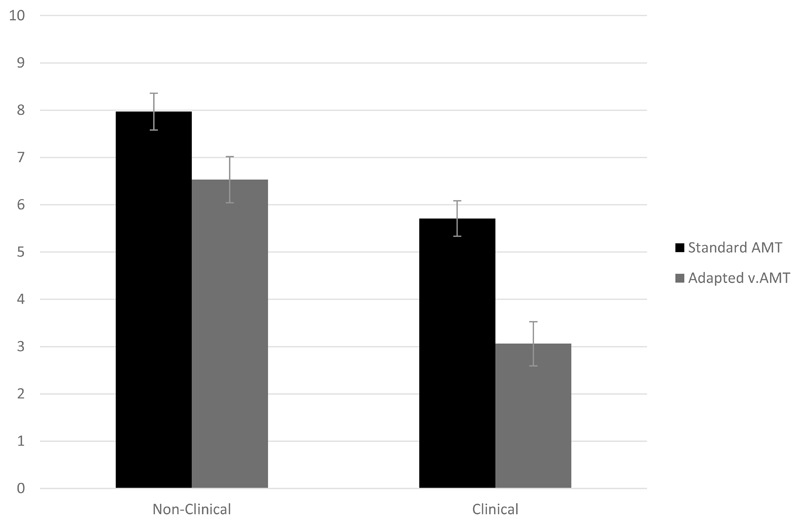
This study is also the first to pilot a novel voices-specific version of the AMT. We suggest this might be a useful tool for further research in exploring autobiographical recall for voices-specific memories, as our data showed that only 2% of responses from the clinical and non-clinical groups on the standard AMT were voices-related. However, we found that people were able to recall voices-related memories when specifically prompted to, using the newly adapted vAMT – and the OMG bias in recall of voice-related memories was greater in the clinical group. We also found an interesting interaction effect, in that although both clinical and non-clinical groups were more overgeneral in their memory recall on the vAMT compared to the AMT, this was significantly more pronounced in the clinical group (Figure 1). This is consistent with a hypothesis derived from Conway and Pleydell-Pearce (2000) model that memory searching is curtailed earlier in the clinical voice hearers because their memories are more distressing. However, we would still interpret the interaction effect with caution, as an alternative explanation is that the vAMT is simply ‘harder’ than the standard task, as the potential pool of eligible memories is restricted by the additional instruction to recall only voices-related memories. It could therefore also be possible that there was a difference in how the clinical and non-clinical groups responded to the change in task difficulty, with the non-clinical group perhaps finding the vAMT more effortful compared to the clinical group. This potential caveat in interpreting the observed interaction effect could be addressed in future studies by the inclusion of a 3rd “control” task, matched for required effort with the vAMT, but relating to another neutral category of memories. Another methodological limitation of the study is that the voices-specific version of the AMT always followed the standard version, so the impact of possible order effects on the vAMT task cannot be ruled out.
Figure 1. Mean AMT score on standard and adapted task by group (Non-Clinical vs. Clinical) with error bars: +/- 1 standard error.
The present study has implications for psychological therapies for distressing voices. Cognitive-behavioural therapy for psychosis (CBTp) is now a well-established treatment and is recommended by both the NICE guidelines in the United Kingdom (National Institute for Health and Care Excellence; 2014) and the PORT guidelines in the United States (Schizophrenia Patient Outcomes Research Team; Dixon et al. (2010)). CBTp already includes therapeutic techniques that target the processes highlighted in this paper. For example, patients learn to recall and monitor specific instances where they act in accordance with their own goals and values, against explicit advice from omnipotent voices (Chadwick et al., 2000). Also, cognitive therapy for command hallucinations includes the keeping of detailed diaries to gather evidence that challenges unhelpful beliefs about the power and infallibility of the voices (Birchwood et al., 2014). The use of competitive memory training for people with distressing voices has also been investigated (van der Gaag et al., 2012).
This therapy involves invoking memories associated with positive self-esteem to compete with the content of the voices, with the aim of weakening the association between negative voice content and negative self-evaluation. Van der Gaag and colleagues found that increased self-esteem and acceptance of voices led to reductions in depression, and this was partially mediated by the power attributed to the voices. This supports the idea that targeting memory biases is important because of their impact on beliefs about voices, and consequently the distress and disability arising from voices. Finally, mindfulness based cognitive therapy for people with recurrent depression has been shown to reduce the OGM bias (Williams et al., 2000). There is an emerging literature on the efficacy of mindfulness for psychosis (Chadwick et al., 2016, Khoury et al., 2013, Lopez-Navarro et al., 2015), but we do not yet know whether mindfulness training for people with distressing voices may be beneficial in reducing overgeneral autobiographical memory recall in a similar way. Future trials could investigate this by assessing memory specificity on the AMT pre- and post a mindfulness intervention for voices. The findings of the current study could also be extended by looking at whether the relationship between memory specificity and beliefs about voices is mediated by limiting access to disconfirmatory information as hypothesised, or whether there are alternative routes between these variables. Finally, the clinical and non-clinical groups in this study were selected on the basis participants reported hearing voices, but we did not control for the possible additional presence of other psychotic symptoms, such as delusional beliefs, although we did not find significant relationships between AMT and delusions in this study. Likewise, previous studies taking a symptom-specific approach in psychosis have selected participants reporting persecutory beliefs, but have not controlled for the presence of voices (Vorontsova et al., 2013). Future studies could therefore explore further the impact of the presence of psychotic symptoms on autobiographical memory functioning, in groups of people who report just voices or delusions, or both together.
In summary, as predicted we found that a group of clinical voice-hearers were more overgeneral in autobiographical memory recall on the AMT, compared to both a group of non-clinical voice-hearers and a control group who did not hear voices. These findings add to our knowledge of what distinguishes clinical from non-clinical groups of people who experience psychotic symptoms, and has implications for the refinement of psychological therapies for people who experience distressing and disabling symptoms.
Supplementary Material
Acknowledgements
We would like to acknowledge Eleonore Bristow, Justine Walker, Paul Green, Emma Saddleton, and Louise Brookwell, who collected the data as research workers employed on the study. We thank Stephani Hatch, Souci Frissa, and Matthew Hotopf for access to the SeLCOH database.
Financial support: This work was supported by the Medical Research Council (Project Grant; Reference: G1100568), awarded to Peters (Principal Investigator), Jackson & Garety (co-applicants).
Footnotes
Conflict of Interest: None.
Ethical standards: The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
References
- Andreasen NC. Scale for the Assessment of Positive Symptoms (SAPS) University of Iowa; Iowa City, IA: 1984. [Google Scholar]
- Baddeley A, Thornton A, Chua SE, McKenna P. Schizophrenic delusions and the construction of autobiographical memory. In: Rubin DC, editor. Remembering our past: Studies in autobiographical memory. Cambridge University Press; US: New York, NY: 1996. pp. 384–428. [Google Scholar]
- Baumeister D, Sedgwick O, Howes O, Peters E. Auditory verbal hallucinations and continuum models of psychosis: A systematic review of the healthy voice-hearer literature. Clinical Psychology Review. 2017;51:125–141. doi: 10.1016/j.cpr.2016.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bebbington P, Nayani T. The Psychosis Screening Questionnaire. International Journal of Methods in Psychiatric Research. 1995;5:11–19. [Google Scholar]
- Beck AT, Steer RA, Brown GK. Beck Depression Inventory (2nd edition) The Psychological Corporation; San Antonio, TX: 1996. [Google Scholar]
- Berna F, Potheegadoo J, Aouadi I, Ricarte JJ, Allé MC, Coutelle R, Boyer L, Cuervo-Lombard CV, Danion J-M. A Meta-Analysis of Autobiographical Memory Studies in Schizophrenia Spectrum Disorder. Schizophrenia Bulletin. 2015 doi: 10.1093/schbul/sbv099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birchwood M, Michail M, Meaden A, Tarrier N, Lewis S, Wykes T, Davies L, Dunn G, Peters E. Cognitive behaviour therapy to prevent harmful compliance with command hallucinations (COMMAND): a randomised controlled trial. Lancet Psychiatry. 2014;1:23–33. doi: 10.1016/S2215-0366(14)70247-0. [DOI] [PubMed] [Google Scholar]
- Brett C, Heriot-Maitland C, McGuire P, Peters E. Predictors of distress associated with psychotic-like anomalous experiences in clinical and non-clinical populations. British Journal of Clinical Psychology. 2014;53:213–227. doi: 10.1111/bjc.12036. [DOI] [PubMed] [Google Scholar]
- Brett C, Peters E, Johns LC, Tabraham P, Valmaggia LR, McGuire P. Appraisals of Anomalous Experiences Interview (AANEX): a multidimensional measure of psychological responses to anomalies associated with psychosis. British Journal of Psychiatry. 2007;191:S23–S30. doi: 10.1192/bjp.191.51.s23. [DOI] [PubMed] [Google Scholar]
- Chadwick P, Birchwood M. The Omnipotence of Voices - a Cognitive Approach to Auditory Hallucinations. British Journal of Psychiatry. 1994;164:190–201. doi: 10.1192/bjp.164.2.190. [DOI] [PubMed] [Google Scholar]
- Chadwick P, Birchwood M. The omnipotence of voices: II. The Beliefs About Voices Questionnaire (BAVQ) The British Journal of Psychiatry. 1995;166:773–776. doi: 10.1192/bjp.166.6.773. [DOI] [PubMed] [Google Scholar]
- Chadwick P, Sambrooke S, Rasch S, Davies E. Challenging the omnipotence of voices: group cognitive behavior therapy for voices. Behaviour Research and Therapy. 2000;38:993–1003. doi: 10.1016/s0005-7967(99)00126-6. [DOI] [PubMed] [Google Scholar]
- Chadwick P, Strauss C, Jones AM, Kingdon D, Ellett L, Dannahy L, Hayward M. Group mindfulness-based intervention for distressing voices: A pragmatic randomised controlled trial. Schizophrenia Research. 2016;175:168–173. doi: 10.1016/j.schres.2016.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coid JW, Kirkbride JB, Barker D, Cowden F, Stamps R, Yang M, Jones PB. Raised incidence rates of all psychoses among migrant groups: Findings from the east london first episode psychosis study. Archives of General Psychiatry. 2008;65:1250–1258. doi: 10.1001/archpsyc.65.11.1250. [DOI] [PubMed] [Google Scholar]
- Conway MA, Pleydell-Pearce CW. The construction of autobiographical memories in the self-memory system. Psychological Review. 2000;107:261–288. doi: 10.1037/0033-295x.107.2.261. [DOI] [PubMed] [Google Scholar]
- Daalman K, Boks MPM, Diederen KMJ, de Weijer AD, Blom JD, Kahn RS, Sommer IEC. The Same or Different? A Phenomenological Comparison of Auditory Verbal Hallucinations in Healthy and Psychotic Individuals. Journal of Clinical Psychiatry. 2011;72:320–325. doi: 10.4088/JCP.09m05797yel. [DOI] [PubMed] [Google Scholar]
- Daalman K, Sommer IEC, Derks EM, Peters ER. Cognitive biases and auditory verbal hallucinations in healthy and clinical individuals. Psychological Medicine. 2013;43:2339–2347. doi: 10.1017/S0033291713000275. [DOI] [PubMed] [Google Scholar]
- Dixon LB, Dickerson F, Bellack AS, Bennett M, Dickinson D, Goldberg RW, Lehman A, Tenhula WN, Calmes C, Pasillas RM, Peer J, et al. The 2009 schizophrenia PORT psychosocial treatment recommendations and summary statements. Schizophrenia Bulletin. 2010;36:48–70. doi: 10.1093/schbul/sbp115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garety PA, Kuipers E, Fowler D, Freeman D, Bebbington PE. A cognitive model of the positive symptoms of psychosis. Psychological Medicine. 2001;31:189–195. doi: 10.1017/s0033291701003312. [DOI] [PubMed] [Google Scholar]
- Gaynor K, Ward T, Garety P, Peters E, Gaynor K, Ward T, Garety P, Peters E. The role of safety-seeking behaviours in maintaining threat appraisals in psychosis. Behaviour Research and Therapy. 2013;51:75–81. doi: 10.1016/j.brat.2012.10.008. [DOI] [PubMed] [Google Scholar]
- Goddard L, Dritschel B, Burton A. Role of autobiographical memory in social problem solving and depression. Journal of Abnormal Psychology. 1996;105:609–616. doi: 10.1037//0021-843x.105.4.609. [DOI] [PubMed] [Google Scholar]
- Johns LC, Kompus K, Connell M, Humpston C, Lincoln TM, Longden E, Preti A, Alderson-Day B, Badcock JC, Cella M, Fernyhough C, et al. Auditory Verbal Hallucinations in Persons With and Without a Need for Care. Schizophrenia Bulletin. 2014;40:S255–S264. doi: 10.1093/schbul/sbu005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaney S, Bowen-Jones K, Bentall RP. Persecutory delusions and autobiographical memory. British Journal of Clinical Psychology. 1999;38:97–102. doi: 10.1348/014466599162692. [DOI] [PubMed] [Google Scholar]
- Kendler KS, Ohlsson H, Sundquist J, Sundquist K. IQ and schizophrenia in a Swedish national sample: their causal relationship and the interaction of IQ with genetic risk. American Journal of Psychiatry. 2015;172:259–65. doi: 10.1176/appi.ajp.2014.14040516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khoury B, Lecomte T, Gaudiano BA, Paquin K. Mindfulness interventions for psychosis: A meta-analysis. Schizophrenia Research. 2013;150:176–184. doi: 10.1016/j.schres.2013.07.055. [DOI] [PubMed] [Google Scholar]
- Kirkbride JB, Errazuriz A, Croudace TJ, Morgan C, Jackson D, Boydell J, Murray RM, Jones PB. Incidence of schizophrenia and other psychoses in England, 1950-2009: A systematic review and meta-analyses. PLoS ONE. 2012;7 doi: 10.1371/journal.pone.0031660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landis JR, Koch GG. An application of hierarchical kappa-type statistics in the assessment of majority agreement among multiple observers. Biometrics. 1977;33:363–74. [PubMed] [Google Scholar]
- Lopez-Navarro E, Del Canto C, Belber M, Mayol A, Fernandez-Alonso O, Lluis J, Munar E, Chadwick P. Mindfulness improves psychological quality of life in community-based patients with severe mental health problems: A pilot randomized clinical trial. Schizophrenia Research. 2015;168:530–536. doi: 10.1016/j.schres.2015.08.016. [DOI] [PubMed] [Google Scholar]
- Lovatt A, Mason O, Brett C, Peters E. Psychotic-Like Experiences, Appraisals, and Trauma. Journal of Nervous and Mental Disease. 2010;198:813–819. doi: 10.1097/NMD.0b013e3181f97c3d. [DOI] [PubMed] [Google Scholar]
- Mason O, Claridge G. The Oxford-Liverpool Inventory of Feelings and Experiences (O-LIFE): Further description and extended norms. Schizophrenia Research. 2006;82:203–211. doi: 10.1016/j.schres.2005.12.845. [DOI] [PubMed] [Google Scholar]
- NICE. Psychosis and schizophrenia in adults: prevention and management. 2014. [Google Scholar]
- Peters E, Ward T, Jackson M, Morgan C, Charalambides M, McGuire P, Woodruff P, Jacobsen P, Chadwick P, Garety P. Clinical, socio-demographic and psychological characteristics in individuals with persistent psychotic experiences with and without a “need for care”. World Psychiatry. 2016;15:41–52. doi: 10.1002/wps.20301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters E, Ward T, Jackson M, Woodruff P, Morgan C, McGuire P, Garety PA. Clinical relevance of appraisals of persistent psychotic experiences in people with and without a need for care: an experimental study. Lancet Psychiatry. 2017;4:927–936. doi: 10.1016/S2215-0366(17)30409-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters E, Williams S, Cooke M, Kuipers E. It's not what you hear, it's the way you think about it: appraisals as determinants of affect and behaviour in voice hearers. Psychological Medicine. 2012;42:1507–1514. doi: 10.1017/S0033291711002650. [DOI] [PubMed] [Google Scholar]
- Peters ER, Moritz S, Schwannauer M, Wiseman Z, Greenwood KE, Scott J, Beck AT, Donaldson C, Hagen R, Ross K, Veckenstedt R, et al. Cognitive Biases Questionnaire for psychosis. Schizophrenia Bulletin. 2014;40:300–13. doi: 10.1093/schbul/sbs199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phelan M, Slade M, Thornicroft G, Dunn G, Holloway F, Wykes T, Strathdee G, Loftus L, Mccrone P, Haywar P. The Camberwell Assessment of Need - the Validity and Reliability of an Instrument to Assess the Needs of People with Severe Mental-Illness. British Journal of Psychiatry. 1995;167:589–595. doi: 10.1192/bjp.167.5.589. [DOI] [PubMed] [Google Scholar]
- Ricarte JJ, Hernandez JV, Latorre JM, Danion JM, Berna F. Rumination and autobiographical memory impairment in patients with schizophrenia. Schizophrenia Research. 2014;160:163–8. doi: 10.1016/j.schres.2014.10.027. [DOI] [PubMed] [Google Scholar]
- Sayer J, Ritter S, Gournay K. Beliefs about voices and their effects on coping strategies. Journal of Advanced Nursing. 2000;31:1199–1205. doi: 10.1046/j.1365-2648.2000.01375.x. [DOI] [PubMed] [Google Scholar]
- Schmider E, Ziegler M, Danay E, Beyer L, Buhner M. Is It Really Robust? Reinvestigating the Robustness of ANOVA Against Violations of the Normal Distribution Assumption. Methodology-European Journal of Research Methods for the Behavioral and Social Sciences. 2010;6:147–151. [Google Scholar]
- Underwood R, Kumari V, Peters E. Appraisals of psychotic experiences: An experimental investigation of symptomatic, remitted and non-need-for-care individuals. Psychological Medicine. 2016;46:1249–1263. doi: 10.1017/S0033291715002780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Gaag M, van Oosterhout B, Daalman K, Sommer IE, Korrelboom K. Initial evaluation of the effects of competitive memory training (COMET) on depression in schizophrenia-spectrum patients with persistent auditory verbal hallucinations: a randomized controlled trial. British Journal of Clinical Psychology. 2012;51:158–71. doi: 10.1111/j.2044-8260.2011.02025.x. [DOI] [PubMed] [Google Scholar]
- Vorontsova N, Garety P, Freeman D. Cognitive Factors Maintaining Persecutory Delusions in Psychosis: The Contribution of Depression. Journal of Abnormal Psychology. 2013;122:1121–1131. doi: 10.1037/a0034952. [DOI] [PubMed] [Google Scholar]
- Ward T, Peters E, Jackson M, Day F, Garety PA. Data-Gathering, Belief Flexibility, and Reasoning Across the Psychosis Continuum. Schizophrenia Bulletin. 2017 doi: 10.1093/schbul/sbx029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward TA, Gaynor KJ, Hunter MD, Woodruff PWR, Garety PA, Peters ER. Appraisals and Responses to Experimental Symptom Analogues in Clinical and Nonclinical Individuals With Psychotic Experiences. Schizophrenia Bulletin. 2014;40:845–855. doi: 10.1093/schbul/sbt094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wechsler D. Wechsler Adult Intelligence Scale - Third Edition. The Psychological Corporation; San Antonio TX: 1997. [Google Scholar]
- Williams J, Broadbent K. Autobiographical memory in suicide attempters. Journal of Abnormal Psychology. 1986;95:144–149. doi: 10.1037//0021-843x.95.2.144. [DOI] [PubMed] [Google Scholar]
- Williams JMG. Autobiographical memory in depression. In: Rubin D, editor. Remembering our past: Studies in autobiographical memory. Cambridge: Cambridge University Press; 1996. pp. 244–267. [Google Scholar]
- Williams JMG, Barnhofer T, Crane C, Hermans D, Raes F, Watkins E, Dalgleish T. Autobiographical memory specificity and emotional disorder. Psychological Bulletin. 2007;133:122–148. doi: 10.1037/0033-2909.133.1.122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams JMG, Dritschel BH. Categoric and Extended Autobiographical Memories. Theoretical Perspectives on Autobiographical Memory. 1992;65:391–410. [Google Scholar]
- Williams JMG, Ellis NC, Tyers C, Healy H, Rose G, MacLeod AK. The specificity of autobiographical memory and imageability of the future. Memory & Cognition. 1996;24:116–125. doi: 10.3758/bf03197278. [DOI] [PubMed] [Google Scholar]
- Williams JMG, Teasdale JD, Segal ZV, Soulsby J. Mindfulness-based cognitive therapy reduces overgeneral autobiographical memory in formerly depressed patients. Journal of Abnormal Psychology. 2000;109:150–155. doi: 10.1037//0021-843x.109.1.150. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.