Abstract
The gastrointestinal (GI) tract is not a common site of metastasis in primary lung cancer. The aim of the present study was to reveal the clinical and prognostic characteristics of gastrointestinal metastases of lung cancer (GMLC). Information on 366 cases of GMLC was collected and factors that affect severe GI complications were analyzed. Univariate and multivariate survival analyses were performed using the Cox proportional hazards model. Of the cases analyzed, the small intestine (59.6%) and colorectum (25.6%) were the two organs where lung cancer was most likely to metastasize in the GI tract. Squamous cell carcinoma (28.5%), adenocarcinoma (27.6%) and large cell carcinoma (20.9%) were the three most common histological types. However, compared with the histological distributions of primary lung cancer, patients with large cell carcinoma exhibited the highest elevated risk of GMLC [relative risk (RR), 4.07; P<0.001] and those with adenocarcinoma exhibited the lowest risk (RR, 0.58; P<0.001). Differences in organ involvement and in histological type led to varying GI complications. It was also indicated that chemotherapy was associated with a decreased risk of hemorrhage (P=0.006), but there was no reduction in the risk of hemorrhage associated with perforation and obstruction (P>0.05). The median overall survival time of GMLC patients was 2.8 months (range, 0–108 months). The survival analyses revealed that perforation and extra-GI metastasis were negative prognostic factors but abdominal surgery was identified a positive prognostic factor. In conclusion, the histological distribution of GMLC differed from that of primary lung cancer. Sufficient and careful patient evaluation, targeted surgeries and systemic therapies for specific patients are able to increase patient survival rate and improve the quality of life.
Keywords: gastrointestinal neoplasm, lung cancer, metastasis, complication, prognosis
Introduction
As a common cancer and the leading cause of cancer-associated mortality, lung cancer has always been universally associated with poor patient prognosis (1). The incidence and mortality rates of lung cancer occupy the first or second position in malignant tumors in China and the United States (2,3). Until 2015, the 5-year total survival rate for lung cancer was ≤20% in developed and developing countries (4). Between 40 and 50% of patients present with distant metastases upon diagnosis with lung cancer have a poor 5-year survival rate of <5% (1). Small cell lung cancer accounts for only 10% of lung cancer but non-small cell lung cancer accounts for ~90%, including squamous cell carcinoma, adenocarcinoma, large cell carcinoma, adenosquamous carcinoma and sarcomatoid carcinoma (4).
As demonstrated in autopsy studies, lung cancer metastases may be identified in every organ system (4). However, the gastrointestinal (GI) tract is not a common site of metastasis for primary lung cancer when compared with other sites, including bone, brain, liver and adrenal glands (5). Gastrointestinal metastases of lung cancer (GMLC) was caused by hematogenous spread and occurred at the end-stage of lung cancer. The incidence of GMLC was <2% in clinical studies, which was much lower compared with its prevalence identified during autopsies (5–8). In previous studies, GMLC was described primarily in case reports. Systemic survival analyses and association between clinicopathological factors, therapeutic factors and GI complications were seldom discussed. The present study reviewed >130 studies from the last 50 years and analyzed 366 cases of GMLC to reveal its clinical and prognostic characteristics by univariate and multivariate survival analyses.
Patients and methods
Patients
A total of 33 cases of GMLC from 32 Chinese articles (9–40) and 3 cases from the School of Medicine, Second Affiliated Hospital of Zhejiang University (Hangzhou, China). The 3 cases were presented to the hospital between 2008 and 2015 were collected in the present study. The Chinese literature was obtained from the VIP Journal Integration Platform (Chongqing, China; http://lib.cqvip.com/) published between 1993 and 2013. A total of 95 articles (5,8,41–133) from the PubMed database from 1961 to 2013 were reviewed, and 330 cases of GMLC were included. In all 366 cases of GMLC assessed, demographic and clinicopathological information was recorded and reviewed, including ethnicity, sex, age, pathology, initial lung cancer stage and therapy, interval between lung cancer diagnosis and identification of GMLC, clinical presentation, location, diameter and number of gross metastases, other metastatic site locations at the time of GMLC, GI surgeries, survival information and immunohistochemical information.
Disease classification
The histological classification of primary lung cancer was performed according to the 2004 World Health Organization classification system of lung tumors (134). The Tumor-Node-Metastasis staging was performed according to the 7th edition of the 2010 American Joint Committee on Cancer Staging manual (135). The histological distribution data of primary lung cancer was acquired from the International Lung Cancer Consortium (136).
Statistical analysis
χ2 test and Fisher's exact test were used to compare the incidence rates of different groups. Overall survival (OS) time was measured from the date of GMLC diagnosis to the date of mortality. This end point was computed using the Kaplan-Meier product limit method and the groups were compared using the log-rank test. Univariate and multivariate analyses of OS time were performed using Cox proportional hazards model. Only the variables with P<0.25 in the univariate analysis were included in the multivariate analysis model and, the backward sequencing method was used (137). The variables in the univariate and multivariate analyses included region (Asia vs. The Americas vs. Europe), histological type of lung cancer (squamous cell carcinoma vs. adenocarcinoma vs. large cell carcinoma vs. small cell lung cancer vs. adenosquamous carcinoma vs. sarcomatoid carcinoma), time (synchronous vs. metachronous), number (solitary vs. multiple), the organ where metastasis has been identified (esophagus vs. stomach vs. small intestine vs. colorectum vs. multiple organs), extra-GI metastasis (yes vs. no), perforation (yes vs. no), surgery of primary lung cancer (yes vs. no) and abdominal surgery (yes vs. no). Metastasis was considered synchronous if the interval of diagnosis between lung cancer and GI metastasis was <1 month. Metastasis was considered metachronous if the interval was >1 month. P<0.05 (two-sided) was considered statistically significant. The statistical analyses were performed using SPSS Statistics software (version 19.0; IBM Corp., Armonk, NY, USA).
Results
Patient and tumor characteristics
As presented in Table I, the majority of the patients were from Asia (62.3%) and >80% of them were male. The three countries with the greatest number of patients analyzed were Japan (139 cases), China (65 cases) and the United States (64 cases). The median age was 63 years (range, 31–92 years). The small intestine was the most frequently involved organ (59.6%) in the GI tract, and the colorectum ranked second (25.6%). Of the small intestine cases of GMLC, where the site of metastasis was specific, the majority of cases (63.4%) occurred in the jejunum. GI perforation (42.0%), hemorrhage (24.6%) and obstruction (20.4%) were the three most common complications. Synchronous and metachronous metastases were similarly probable (46.2% vs. 53.8%). The median time of metachronous patients from primary cancer diagnosis to GI metastasis was 6 months (range, 1–108 months). Solitary GMLCs were present in an increased number of patients compared with multiple metastases (69.4% vs. 30.6%). Furthermore, extra-GI metastases were present in the majority of patients (70.5%). The liver, brain, bone and adrenal gland were the four most prevalent sites of metastasis (data not shown), which was expected. In total, ~1/3 of the patients (33.3%) underwent surgical resection to remove the primary cancer and surgeries were performed in the GI tract in 75.8% of patients. Partial gastrectomy and enterectomy were the main types of abdominal resections performed (86.5%). These statistics are based solely upon cases where data was available.
Table I.
Patient, tumor and treatment characteristics of 366 cases of gastrointestinal metastases of lung cancer.
| A, Demographic factors | ||
|---|---|---|
| Characteristics | Patients, n | Percentage |
| Region | ||
| Asia | 228 | 62.3 |
| America | 67 | 18.3 |
| Europe | 71 | 19.4 |
| Sex (not reported in 114 cases) | ||
| Male | 208 | 82.5 |
| Female | 44 | 17.5 |
| Age, years (not reported in 13 cases) | ||
| ≤54 | 54 | 15.3 |
| 55–64 | 133 | 37.7 |
| 65–74 | 116 | 32.9 |
| ≥75 | 50 | 14.2 |
| B, Tumor factors | ||
| Characteristics | Patients, n | Percentage |
| Involved organ (not reported in 10 cases) | ||
| Esophagus | 1 | 0.3 |
| Stomach | 40 | 11.2 |
| Small intestinea | 212 | 59.6 |
| Colorectumb | 91 | 25.6 |
| Multiple organs | 12 | 3.4 |
| Complications (not reported in 9 cases) | ||
| Perforation | 150 | 42.0 |
| Hemorrhage | 88 | 24.6 |
| Obstruction | 73 | 20.4 |
| Histological type (not reported in 22 cases) | ||
| Small cell lung cancer | 46 | 13.4 |
| Squamous cell carcinoma | 98 | 28.5 |
| Adenocarcinoma | 95 | 27.6 |
| Large cell carcinoma | 72 | 20.9 |
| Sarcomatoid carcinoma | 15 | 4.4 |
| Adenosquamous carcinoma | 5 | 1.5 |
| Other types | 13 | 3.8 |
| Synchronous or metachronous metastasis (not reported in 208 cases) | ||
| Synchronous | 73 | 46.2 |
| Metachronous | 85 | 53.8 |
| Number of GI metastasis (not reported in 268 cases) | ||
| Solitary | 68 | 69.4 |
| Multiple | 30 | 30.6 |
| Extra-GI metastasis (not reported in 220 cases) | ||
| No | 43 | 29.5 |
| Yes | 103 | 70.5 |
| C, Treatment factors | ||
| Treatment | Patients, n | Percentage |
| Lung cancer surgery (not reported in 234 cases) | ||
| No | 88 | 66.7 |
| Yes | 44 | 33.3 |
| Abdominal surgery (not reported in 180 cases) | ||
| No | 45 | 24.2 |
| Yesc | 141 | 75.8 |
Metastasis in the small intestine: Duodenum, 19 cases; jejunum, 104 cases; ileum, 41 cases; not otherwise specified, 60 cases.
Metastasis in the colorectum: Appendix and ileocecus, 10 cases; colon, 78 cases; rectum, 5 cases.
Abdominal surgery: Partial gastrectomy and enterectomy, 122 cases; gastrointestinal fistula, bypass surgery and repair of the perforation, 7 cases; not otherwise specified, 12 cases.
Squamous cell carcinoma (28.5%), adenocarcinoma (27.6%) and large cell carcinoma (20.9%) were the three most common histological types of GMLC. When the histological distributions between GMLC and primary lung cancer were compared, it was observed that there was an increased frequency of GMLC of large cell carcinoma, small cell lung cancer and squamous cell carcinoma compared with GMLC of adenocarcinoma and other types of cancer (P<0.01; Table II).
Table II.
Comparison of the histological distributions between gastrointestinal metastases of lung cancer and primary lung cancer.
| Histological type | GMLC, % | Primary lung cancer, %a | χ2-value | P-value | RR (95% CI) |
|---|---|---|---|---|---|
| Small cell lung cancer | 13.37 | 9.28 | 6.61 | 0.01 | 1.44 (1.10–1.90) |
| Squamous cell carcinoma | 28.49 | 20.48 | 13.08 | <0.001 | 1.39 (1.17–1.65) |
| Adenocarcinoma | 27.62 | 47.34 | 52.31 | <0.001 | 0.58 (0.49–0.69) |
| Large cell carcinoma | 20.93 | 5.15 | 158.81 | <0.001 | 4.07 (3.27–5.06) |
| Other types | 9.59 | 17.76 | 15.43 | <0.001 | 0.54 (0.39–0.75) |
Data from the International Lung Cancer Consortium (133). GMLC, gastrointestinal metastases of lung cancer; RR, relative risk; 95% CI, 95% confidence interval.
Complications
The association between involved organs, histological type and GI complications is presented in Table III. Perforation of the small intestine (63.7%) occurred more frequently compared with perforation of stomach and colorectum (P<0.001). The organs at risk of hemorrhage, from most to least common, were the stomach, colorectum and small intestine (P<0.05 in comparisons between two organs). No obstruction occurred in all 36 patients with gastric metastasis, and the incidence of obstruction was much lower compared with colorectum (P=0.003) or small intestine (P=0.003). In all the histological types of lung cancer analyzed, sarcomatoid carcinoma exhibited the lowest likelihood of perforation and the greatest likelihood of hemorrhage (P<0.05). The risks of perforation of squamous cell carcinoma and small cell lung cancer were increased compared with the risks associated with adenocarcinoma (P<0.05). The risks of hemorrhage of squamous cell carcinoma and small cell lung cancer were lower compared with the risks for large cell carcinoma (P<0.05). No statistically significant difference was observed between two other organs or histological types (P>0.05). No significant association was identified between histological type and GI obstruction (P>0.05).
Table III.
Association between GI complications, organ and histological types of gastrointestinal metastases of lung cancer.
| Perforation, n (%) | Hemorrhage, n (%) | Obstruction, n (%) | |||||
|---|---|---|---|---|---|---|---|
| Organa/histological typeb | Yes | No | Yes | No | Yes | No | Total |
| Esophagus | 0 (0.0) | 1 (100.0) | 0 (0.0) | 1 (100.0) | 0 (0.0) | 1 (100.0) | 1 |
| Stomach | 5 (13.9) | 31 (86.1) | 19 (52.8) | 17 (47.2) | 0 (0.0) | 36 (100.0) | 36 |
| Small intestine | 135 (63.7) | 77 (36.3) | 33 (15.6) | 179 (84.4) | 43 (20.3) | 169 (79.7) | 212 |
| Colorectum | 10 (11.5) | 77 (88.5) | 28 (32.2) | 59 (67.8) | 18 (20.7) | 69 (79.3) | 87 |
| Multiple organs | 0 (0.0) | 12 (100.0) | 7 (58.3) | 5 (41.7) | 4 (33.3) | 8 (66.7) | 12 |
| Unreported organ | 0 (0.0) | 9 (100.0) | 1 (11.1) | 8 (88.9) | 8 (88.9) | 1 (11.1) | 9 |
| Squamous cell carcinoma | 49 (50.5) | 48 (49.5) | 20 (20.6) | 77 (79.4) | 20 (20.6) | 77 (79.4) | 97 |
| Adenocarcinoma | 33 (35.5) | 60 (64.5) | 23 (24.7) | 70 (75.3) | 16 (17.2) | 77 (82.8) | 93 |
| Large cell carcinoma | 26 (36.6) | 45 (63.4) | 26 (36.6) | 45 (63.4) | 18 (25.4) | 53 (74.6) | 71 |
| Small cell lung cancer | 24 (53.3) | 21 (46.7) | 8 (17.8) | 37 (82.2) | 6 (13.3) | 39 (86.7) | 45 |
| Adenosquamous carcinoma | 3 (60.0) | 2 (40.0) | 0 (0.0) | 5 (100.0) | 2 (40.0) | 3 (60.0) | 5 |
| Sarcomatoid carcinoma | 0 (0.0) | 15 (100.0) | 8 (53.3) | 7 (46.7) | 5 (33.3) | 10 (66.7) | 15 |
| Other types or unreported | 15 (48.4) | 16 (51.6) | 3 (9.7) | 28 (90.3) | 6 (19.4) | 25 (80.6) | 31 |
| Total | 150 (42.0) | 207 (58.0) | 88 (24.6) | 269 (75.4) | 73 (20.4) | 284 (79.6) | 357c |
Perforation of the small intestine was more likely to occur compared with perforation of the stomach and colorectum (P<0.001). The prevalence of hemorrhage descended in order of the stomach, colorectum and small intestine (P<0.05). Obstruction of the stomach was much less than that of other organs (P=0.003).
Perforation of SARC was the least in all the types (P<0.05). Perforations of squamous cell carcinoma and small cell lung cancer were more likely compared with adenocarcinoma (P<0.05). Hemorrhage of sarcomatoid carcinoma was more likely to happen compared with hemorrhage of squamous cell carcinoma (P=0.016), adenocarcinoma (P=0.049) and small cell lung cancer (P=0.018). Hemorrhage of large cell carcinoma was more likely to happen compared with hemorrhage of squamous cell carcinoma (P=0.022) and small cell lung cancer (P=0.030). No significant association was identified between histological type and gastrointestinal obstruction (P>0.05).
The complications in 9 cases were not reported.
The association between GI complications and chemotherapy is indicated in Table IV. In total, ~1/4 (26.5%, 97/366) of the cases analyzed contained patient data of chemotherapy. Hemorrhage was more common in patients with GMLC who received no chemotherapy (P=0.006). There was no significant difference in the risks of perforation and obstruction between patients who had received and had not received chemotherapy (P>0.05).
Table IV.
Association between gastrointestinal complications and chemotherapy.
| Chemotherapy, n (%) | |||
|---|---|---|---|
| Complications | Yes | No | P-value |
| Perforation | 0.405 | ||
| Yes | 7 (19.4) | 8 (13.1) | |
| No | 29 (80.6) | 53 (86.9) | |
| Hemorrhage | 0.006a | ||
| Yes | 7 (19.4) | 29 (47.5) | |
| No | 29 (80.6) | 32 (52.5) | |
| Obstruction | 0.448 | ||
| Yes | 7 (19.4) | 16 (26.2) | |
| No | 29 (80.6) | 45 (73.8) | |
| Totalb | 36 | 61 | |
Hemorrhages were less likely in patients who had received chemotherapy compared with patients who have not received chemotherapy (P=0.006).
Available data, 97 cases.
Immunohistochemical analysis
In total, there were 35 cases for which immunohistochemical data were available. Information from this limited number of cases illustrated that a typical immunohistochemical staining of GMLC was positive for thyroid transcription factor-1 (TTF-1; 84.4%, 27/32) and cytokeratin 7 (CK7, 96.6%, 28/29), but negative for CK20 (96.6%, 28/29) and caudal-related homeodomain transcription 2 (CDX2, 100%, 23/23).
Survival estimates
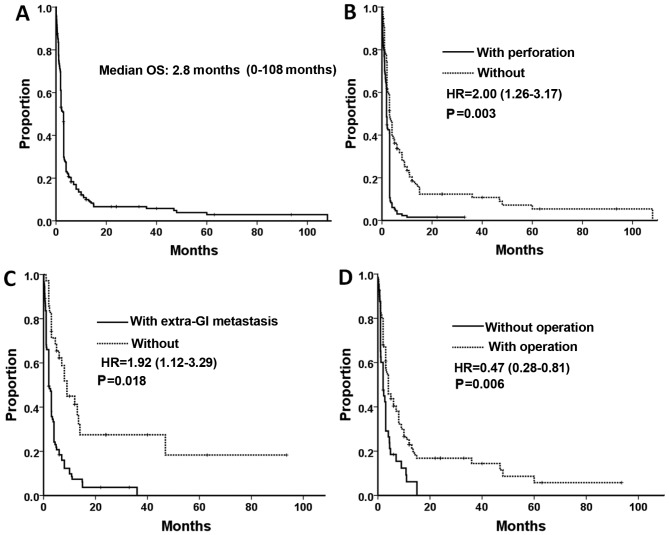
Survival rate data was available for 268 of the 366 patients studied. A total of 246 patients had succumbed to disease by the end of the duration of the study, and the data of the other 22 patients were censored data. The median OS time was 2.8 months (range, 0–108 months, Fig. 1A). Univariate Cox analysis (Table V) revealed that patients from the Americas exhibited poorer prognoses compared with patients from Asia (P<0.001). It was also indicated that the survival rates for patients with small cell lung cancer and sarcomatoid carcinoma were increased compared with patients with squamous cell carcinoma (unadjusted hazard ratios (HRs), 0.62 (P=0.03) and 0.34 (P=0.005), respectively). The other prognostic factors, which indicate a poor survival outcome, included synchronous metastasis, perforation of GMLC, extra-GI metastasis and lack of abdominal surgery (P<0.05). Patients with multiple metastases exhibited marginal poorer prognoses compared with patients with solitary metastases (P=0.06). There were no statistical significance between overall survival rate and other factors, (sex, age, organs of GI metastasis, and surgery on the primary lung cancer) (P>0.05).
Figure 1.
(A) OS of patients with gastrointestinal metastases of lung cancer. Variables affecting OS in multivariate Cox regression analysis: (B) Perforation, (C) extra-GI metastasis and (D) abdominal surgery. GI, gastrointestinal; HR, hazard ration; OS, overall survival.
Table V.
Univariate and multivariate Cox regression analyses of variables that may affect overall survival of patients with gastrointestinal metastases of lung cancer.
| A, Univariate analysis | |||
|---|---|---|---|
| Overall survival | |||
| Variable | HR | 95% CI | P-value |
| Demographic factors | |||
| Region | |||
| Asia | 1.00 | ||
| America | 3.27 | 2.32–4.59 | <0.001 |
| Europe | 1.27 | 0.92–1.76 | 0.148 |
| Sex | |||
| Male | 1.00 | ||
| Female | 0.81 | 0.53–1.25 | 0.351 |
| Age, years | |||
| ≤54 | 1.00 | ||
| 55–64 | 0.86 | 0.60–1.25 | 0.430 |
| 65–74 | 0.96 | 0.66–1.42 | 0.849 |
| ≥75 | 1.27 | 0.78–2.07 | 0.339 |
| Histological type | |||
| Squamous cell carcinoma | 1.00 | ||
| Adenocarcinoma | 0.78 | 0.55–1.10 | 0.150 |
| Large cell carcinoma | 0.75 | 0.52–1.08 | 0.123 |
| Small cell lung cancer | 0.62 | 0.41–0.96 | 0.031 |
| Adenosquamous carcinoma | 2.01 | 0.73–5.56 | 0.177 |
| Sarcomatoid carcinoma | 0.34 | 0.16–0.72 | 0.005 |
| Time of GI metastasis | |||
| Synchronous | 1.00 | ||
| Metachronous | 0.62 | 0.43–0.90 | 0.011 |
| Number of GI metastasis | |||
| Solitary | 1.00 | ||
| Multiple | 1.64 | 0.98–2.76 | 0.064 |
| GI metastasis organ | |||
| Esophagusa | N/A | ||
| Stomach | 1.00 | ||
| Small intestine | 1.19 | 0.82–1.73 | 0.367 |
| Colorectal | 0.78 | 0.48–1.28 | 0.321 |
| Multiple organs | 0.95 | 0.44–2.05 | 0.886 |
| Perforation | |||
| No | 1.00 | ||
| Yes | 2.13 | 1.61–2.78 | <0.001 |
| Extra-GI metastasis | |||
| No | 1.00 | ||
| Yes | 2.94 | 1.85–4.76 | <0.001 |
| Treatment factors | |||
| Surgical resection of the primary cancer | |||
| No | 1.00 | ||
| Yes | 0.68 | 0.43–1.04 | 0.078 |
| Abdominal surgery | |||
| No | 1.00 | ||
| Yes | 0.53 | 0.35–0.78 | 0.001 |
| B, Multivariate analysis | |||
| Overall survival | |||
| Variable | Adjusted HR | 95% CI | P-value |
| Demographic factors | |||
| Region | |||
| Asia | 1.00 | ||
| America | 3.91 | 2.41–6.34 | <0.001 |
| Tumor factors | |||
| Histological type | |||
| Squamous cell carcinoma | 1.00 | ||
| Adenosquamous carcinoma | 5.57 | 1.84–16.83 | 0.002 |
| Perforation | |||
| No | 1.00 | ||
| Yes | 2.00 | 1.26–3.17 | 0.003 |
| Extra-GI metastasis | |||
| No | 1.00 | ||
| Yes | 1.92 | 1.12–3.29 | 0.018 |
| Treatment factors | |||
| Surgical resection | |||
| No | 1.00 | ||
| Yes | 0.47 | 0.28–0.81 | 0.006 |
| Other factors | NS | ||
Esophageal metastasis in 1 case. HR, hazard ratio; CI, confidence interval; GI, gastrointestinal; NS, not significant.
In multivariate analysis, following adjustment for prognostic factors, the region where the patients were from, histological type, GI perforation, extra-GI metastasis and abdominal surgery remained prognostic factors for survival rate. Other factors were not significant (Table V; Fig. 1B-D).
Discussion
As the digestive tract remains a rare site of metastasis of primary lung cancer, GMLC was previously described primarily in case reports (8). The clinical incidence of GMLC was <2% in patients with primary lung cancer and >10% in autopsy-associated studies (5–8). The majority of GMLC cases occurred at the end-stage of lung cancer, and misdiagnosis and missed diagnosis occurred frequently (138). In the present study, the sex and age of the patients with GMLC was similar with cases reported in previous studies with small sample sizes (7,8). In the present study, the majority of the patients were elderly men.
The intestines, particularly the small intestine, were the main organs involved in GMLC, owing to their abundant blood supply. There were certain patients that presented with metastases in multiple GI organs, due to the lethality of the first involved organ and the involvement of additional GI organs. On account of the significant difference between the GMLC incidences from clinical and autopsy-associated studies, the majority of the patients with GMLC were subclinical with minimal or no symptoms. The symptoms of GMLC always appeared alongside clinical complications, including GI perforation, hemorrhage and obstruction. The incidence of severe complications differed between organs due to differences in individual anatomical structures of the organs. Previous studies (129,132,139) reported that the incidence of perforation in the small intestine was markedly increased compared with the incidence in the colorectum. The present study indicated similar findings. The probability and quantity of hemorrhage may depend on the incubation time of GMLC and the inner diameter of the involved organ. Metastases are able to grow for a longer period of time in the large gastric cavity, leading to more bleeding in the stomach compared with other organs. In the present study, no obstruction was identified in the stomach, and it was hypothesized that this may be associated with the wider inner diameter of the stomach compared with other GI organs.
In the present study, >3/4 of GMLC cases analyzed were squamous cell carcinoma, adenocarcinoma and large cell carcinoma. The high incidence of squamous cell carcinoma and adenocarcinoma was due to their high prevalence in primary lung cancer. The highest risk of GI metastasis was identified in large cell carcinoma, whereas the lowest risk of all the major histological types of GI metastasis was identified in adenocarcinoma. This finding is partly consistent with the autopsy data from Antler et al (6). On the other hand, different histological types of lung cancer exhibit different GI complications. A previous study on metastasis in the small intestine indicated that large cell carcinoma is associated with the greatest likelihood of perforation, whereas adenocarcinoma is most closely associated with the lowest rate of perforation in all the histological types (69). The present study partly confirmed this result and suggested that sarcomatoid carcinoma was associated with a lower rate of perforation and a greater rate of hemorrhages of the GI tract (Table III). This may be due to the relatively compact structure of sarcomatoid carcinoma with its differentiation of connective tissues. A number of previous studies (112,140) have suggested that GI perforation may be caused by chemotherapy, but other studies (106,141) indicated that GI perforation is brought about mainly by ischemia and necrosis of metastases, obstruction and high pressure in the GI cavity. In the present study, in which only a quarter of the total studies contained data on GI perforation, patients with or without chemotherapy experience statistically similar incidences of perforation and obstruction. The risk of hemorrhage may be decreased by chemotherapy, which may lead to tumor atrophy and ischemia.
The diagnosis of GMLC is not easy owing to its non-specific symptoms, although its complications are well known. When patients with lung cancer complain of potential GMLC symptoms (abdominal pain, nausea, vomiting, anemia, hematochezia, melena, constipation or other changes in bowel habits), particularly those that cannot be explained by primary lung cancer or undergoing chemotherapy or radiotherapy treatment, GMLC should be considered. As part of systemic metastases, GMLC is caused by hematogenous dissemination (8). In previous studies (88,141), 42.9–100.0% of the patients with GMLC had extra-GI metastasis, and this ratio was 70.5% in the present study. Therefore, it is necessary for patients with GMLC to undergo sufficient evaluations prior to initiation of treatment (142). On the other hand, it was reported that GMLC was more common in patients with metastases in adrenal glands, kidneys and celiac lymph nodes compared with in patients with other type of metastases (5). However, this finding was not supported by the data in the present study.
Laboratory examination, endoscopy, gastroenterography, computed tomography (CT) and positron-emission tomography (PET)-CT may aid the diagnosis of GMLC (120). In a study by Kim et al (141) on the signs of GMLC in CT, it was reported that positive signs could be identified in 93% of patients, including localized GI wall thickening, the presence of a mass in the GI cavity, regional glandular enlargement, indigitation and perforation. PET-CT was able to assist the detection of subclinical GMLC and systemic evaluation of extra-GI metastases (133,143,144). Pathology was a critical factor in diagnosis. The pathology of primary lung cancer should be reviewed and compared with that of GMLC. In the present study, in the majority of cases of GMLC considered, there was positive immunohistochemical staining for thyroid transcription factor 1 and cytokeratin-7 but negative staining for CK20 and CDX2, which was consistent with a previous study (8).
As the end-stage of lung cancer, GMLC was always associated with a poor prognosis (69). In the present study, the median OS time was 2.8 months and 53.4% of the patients succumbed to disease within 3 months of diagnosis of GMLC. Only 9.0% (24/268) of patients were reported to survive for >1 year and 2 patients (0.7%) with large cell carcinoma survived >7 years. Patients in the dataset analyzed by the present study generally exhibited longer overall survival times compared with patients assessed by Garwood et al (69). The patients that Garwood et al (69) studied are part of the present dataset. All patients in the study by Garwood et al (69) exhibited perforation of the small intestine, which appears to be associated with shorter OS (based on comparisons between the results of the present study and Garwood et al) (69). In the present study, there were five factors, including region, histological type, GI perforation, extra-GI metastasis and surgical resection, which formed the final components included in the multivariate Cox model. Although it was indicated that the survival times for Asian patients were longer compared with American patients, region was not a useful factor in determining survival times as American studies assessed in the present study were undertaken on average ~20 years earlier compared with the studies from Asia. With an adjusted hazard ratio of 5.57 (P=0.002), it was indicated that patients with adenosquamous carcinoma had a significantly poorer prognosis compared with patients with squamous cell carcinoma based on multivariate Cox analysis. However, only four cases of adenosquamous carcinoma were available in the present survival analysis, so this result is of limited value. Perforation of GMLC and extra-GI metastasis were indicated to be negative prognostic factors, but abdominal surgery appeared to be a positive prognostic factor. However, the selective bias of the patients, particularly in treatment choice, must be considered in the present retrospective study. The patients with less severe general conditions were more likely to undergo surgery than those in critical conditions.
The present study revealed that, due to the increasing incidence of lung cancer, as well as the ability of modern medicine to prolong the life of lung cancer patients, GMLC is no longer rare. On the basis of the assessment performed in the present study, the histological distribution of GMLC was different from that of primary lung cancer. Early detection, diagnosis and treatment are central to improving patient prognosis. Sufficient and careful evaluations, targeted surgeries and systemic therapies for specific patients following discussion between multi-disciplinary teams of medics are able to improve the survival rate and quality of life of patients (138,145). The main purpose of surgery is to relieve symptoms while causing the least trauma possible to the patient. Data from the present study revealed that >30% of the patients had multiple metastases in their GI tract, therefore careful investigations during the surgery are required to avoid subclinical metastases being missed.
Acknowledgements
The present study was funded by grants from the Natural Science Foundation of Zhejiang Province of China (nos. LY14H160030 and LY13H160011), the National Program on Key Basic Research Project of China (973 Program; no. 2014CB542003), the National Natural Science Foundation of China (no. 30801341) and the Zheng Shu Elite Scholarship for Clinical Medicine.
References
- 1.Mao Y, Yang D, He J, Krasna MJ. Epidemiology of Lung Cancer. Surg Oncol Clin N Am. 2016;25:439–445. doi: 10.1016/j.soc.2016.02.001. [DOI] [PubMed] [Google Scholar]
- 2.Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66:115–132. doi: 10.3322/caac.21338. [DOI] [PubMed] [Google Scholar]
- 3.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2017. CA Cancer J Clin. 2017;67:7–30. doi: 10.3322/caac.21387. [DOI] [PubMed] [Google Scholar]
- 4.Frank CD, Roy HD, Lynn T, Rogerio CL. DeVita, Hellman and Rosenberg's Cancer: Principles & Practice of Oncology. 10th Edition. Wolters Kluwer Health Corp.; Philadelphia: 2015. Non-small Cell Lung Cancer; pp. 495–502. [Google Scholar]
- 5.Yoshimoto A, Kasahara K, Kawashima A. Gastrointestinal metastases from primary lung cancer. Eur J Cancer. 2006;42:3157–3160. doi: 10.1016/j.ejca.2006.08.030. [DOI] [PubMed] [Google Scholar]
- 6.Antler AS, Ough Y, Pitchumoni CS, Davidian M, Thelmo W. Gastrointestinal metastases from malignant tumors of the lung. Cancer. 1982;49:170–172. doi: 10.1002/1097-0142(19820101)49:1<170::AID-CNCR2820490134>3.0.CO;2-A. [DOI] [PubMed] [Google Scholar]
- 7.Yang CJ, Hwang JJ, Kang WY, Chong IW, Wang TH, Sheu CC, Tsai JR, Huang MS. Gastro-intestinal metastasis of primary lung carcinoma: clinical presentations and outcome. Lung Cancer. 2006;54:319–323. doi: 10.1016/j.lungcan.2006.08.007. [DOI] [PubMed] [Google Scholar]
- 8.Rossi G, Marchioni A, Romagnani E, Bertolini F, Longo L, Cavazza A, Barbieri F. Primary lung cancer presenting with gastrointestinal tract involvement: Clinicopathologic and immunohistochemical features in a series of 18 consecutive cases. J Thorac Oncol. 2007;2:115–120. doi: 10.1097/01243894-200702000-00004. [DOI] [PubMed] [Google Scholar]
- 9.Chongbo S, Gaojia Z. A case report of small intestinal metastasis of lung cancer. Chin J Clin Oncol. 1993;20:319. (In Chinese) [Google Scholar]
- 10.Zhiqiang C, Chenghai Z. Lung cancer with its first symptom of gastrointestinal hemorrhage: a case report. J Jinan Univ (Med Ed) 1996;17:132. (In Chinese) [Google Scholar]
- 11.Yuyuan Y. Bowel obstruction caused by colonic metastasis of squamous cell lung cancer: a case report. Chin J Cancer. 1999;18:143. (In Chinese) [Google Scholar]
- 12.Jianguo S, Xiufeng Z, Qingmei J. A case report of rectal metastasis of small cell lung cancer. Chin J Cancer Prev Treat. 2000;7:369. (In Chinese) [Google Scholar]
- 13.Jun Q, Wenyong S, Dechuan L, Rongcan L, Guiquan L. A case report of lung cancer with right colonic metastasis. J Coloproctol Surg. 2001;7:49. (In Chinese) [Google Scholar]
- 14.Xiaoqiang L, Delong Z, Yimeng Z, Chunbao Z. Gastric metastasis of primary lung cancer: one case report. Suzhou Med. 2001;24:160. (In Chinese) [Google Scholar]
- 15.Xiaoning C, Jinlong C, Shilian Z. Bowel obstruction caused by ileum metastasis of lung cancer: a case report. Chin J Prim Med Pharm. 2002;9:795. (In Chinese) [Google Scholar]
- 16.Xin T, Wang W, Xu Y, Yang Y, Hao W, Li Y. A case report of rectal metastasis of lung cancer. Zhongguo Fei Ai Za Zhi. 2002;5:457. (In Chinese) [Google Scholar]
- 17.Jinlin L, Xiaoming L. Jejunal metastasis of lung cancer: one case report. J Abdom Surg. 2002;15:59. [Google Scholar]
- 18.Hui C, Yaoguang L, Yin L. A case report of gastric metastasis of lung cancer. J Guangxi Med Univ. 2004;21:766. (In Chinese) [Google Scholar]
- 19.Huili Z, Hanmeng Y. Bowel idigitation caused by intestinal metastasis of lung cancer: one case report. Med J CASC. 2004;6:35. [Google Scholar]
- 20.Yan L, Chongqin Y. Ileum metastasis of lung adenocarcinoma: a case report. Chin J Geriatr. 2005;24:382–383. [Google Scholar]
- 21.Xiangtao M, Liwei Y, Jing F, Shan W, Ruyi D, Zhirong C. A case report of colonic metastasis of undifferentiated lung cancer. Chin J Clin Oncol. 2006;33:358. (In Chinese) [Google Scholar]
- 22.Xiangtao M, Liwei Y, Jing F, Shan W, Ruyi D, Zhirong C. Colonic metastasis of squamous cell lung cancer: a case report and review. Cancer Res Prev Treat. 2006;33:631. [Google Scholar]
- 23.Yong L, Yuguo H, Yongcan L, Hongzhe Y, Fengxia J. A case of colonic metastasis of lung cancer. Chin J Thorac Cardiovasc Surg. 2007;23:113. [Google Scholar]
- 24.Jianzhong D, Pin Z, Yi W. A case of multiple bowel perforations caused by jejunum metastasis of lung cancer. Chin J Surg. 2008;46:77. (In Chinese) [Google Scholar]
- 25.Lixia L, Weimin Z. Small intestinal metastasis of lung cancer: a case report. J Prac Med. 2008;24:3853. [Google Scholar]
- 26.Xuan W, He H, Muyan C, Liping L, Yuanyuan Z, Li Z. Small intestinal metastasis of non-small cell lung cancer: Case report and literature review. Chin J Cancer. 2008;27:447–448. (In Chinese) [Google Scholar]
- 27.Wei H, Baojin H, Weiru X. A case report of gastric metastasis of primary lung cancer. Chin J Oncol. 2008;30:698. (In Chinese) [Google Scholar]
- 28.Yingman W, Xiangmin L, Haixia Z. Small intestinal metastasis of lung cancer: case report and literature review. J Fourth Mil Med Univ. 2009;30:96. [Google Scholar]
- 29.Qian L, JIangyang L, Xiaohong W, Yi Y, Lin L. Clinical and pathological analysis of lung giant cell carcinoma with neuroendocrine differentiation metastatic to small intestine. J Diag Pathol. 2009;16:100–103. [Google Scholar]
- 30.Miao Z, Li R, Guangqin Z. A case of gastrointestinal haemorrhage caused by small intestinal metastasis of squamous cell lung cancer. Med J Chin PAPF. 2010;21:605–606. [Google Scholar]
- 31.Fenghui Z, Yunping Z. A case of choroid membrane and rectal metastasis of lung cancer. Chin J Clin Oncol. 2010;37:878. (In Chinese) [Google Scholar]
- 32.Liu Y, Zhang L, Han X, Zhou T. A case report and literature review of small intestinal metastasis of large cell lung cancer. Zhongguo Fei Ai Za Zhi. 2010;13:655–658. doi: 10.3779/j.issn.1009-3419.2010.06.019. (In Chinese) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zhidong L, Weijun Z, Funan L, Yimin D, Jixian S. Bowel perforation caused by colonic metastasis of small cell lung cancer: a case report. J Basic Clin Oncol. 2010;23:451–452. [Google Scholar]
- 34.Mo S, Lei D, Wei J, Bin Y, Hongjiang W. Bowel indigitation caused by small intestinal metastasis of lung cancer. J Dalian Med Univ. 2010;32:374–375. [Google Scholar]
- 35.Lei Y, Xishan W, Xinshu D, Li L. Multiple small intestinal metastasis of lung cancer. Chin J Oncol. 2010;32:525. (In Chinese) [Google Scholar]
- 36.Wang Y, An T, Yang L, Wang Z, Zhuo M, Duan J, Wang J, Wu M. Primary lung cancer with gastrointestinal metastasis: 2 case report and literature review. Zhongguo Fei Ai Za Zhi. 2011;14:278–280. doi: 10.3779/j.issn.1009-3419.2011.03.23. (In Chinese) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wu W, Mu J, Tong L, Liu Z. Primary lung squamous cell carcinoma ileocecal metastasis: one case report and literature review. Int J Respir. 2011;31:1225–1227. [Google Scholar]
- 38.Xu L, Zijun L, Yahuan G, Juntao Y. Esophagus metastasis of mucoepidermoid lung carcinoma: case report and literature review. Cancer Res Prev Treat. 2011;38:1462. [Google Scholar]
- 39.Hong Z, Xiaojing L, Honglin W. Duodenal metastases from primary squamous cell carcinoma of the lung: one case report. J Chin Oncol. 2012;18:319–320. (In Chinese) [Google Scholar]
- 40.Junni C, Bo Y, Yanli Z, Jie L, Fen W. Gastric metastasis of primary lung cancer: one case report Guangdong. Med J. 2012;33:1369. [Google Scholar]
- 41.Pezzuto A, Mariotta S, Fioretti F, Uccini S. Metastasis to the colon from lung cancer presenting with severe hyponatremia and dyspnea in a young male: A case report and review of the literature. Oncol Lett. 2013;5:1477–1480. doi: 10.3892/ol.2013.1208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Huang YM, Hsieh TY, Chen JR, Chien HP, Chang PH, Wang CH, Huang JS. Gastric and colonic metastases from primary lung adenocarcinoma: A case report and review of the literature. Oncol Lett. 2012;4:517–520. doi: 10.3892/ol.2012.778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sakai H, Egi H, Hinoi T, Tokunaga M, Kawaguchi Y, Shinomura M, Adachi T, Arihiro K, Ohdan H. Primary lung cancer presenting with metastasis to the colon: A case report. World J Surg Oncol. 2012;10:127. doi: 10.1186/1477-7819-10-127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hsing CT, Kim HY, Lee JH, Han JS, Lee JH, Chang JS, Choi SR, Jeong JS. Gastrointestinal metastasis from a primary adenocarcinoma of the lung presenting with acute abdominal pain. Korean J Gastroenterol. 2012;59:382–385. doi: 10.4166/kjg.2012.59.5.382. [DOI] [PubMed] [Google Scholar]
- 45.Pratto D, Resial M, Wulfson A, Gennaro M, Brarda M, Schmidt A. Jejuno-jejunal intussusception as presentation of a primary lung carcinoma: a case report. Acta Gastroenterol Latinoam. 2012;42:50–52. (In Chinese) [PubMed] [Google Scholar]
- 46.Cedres S, Mulet-Margalef N, Montero MA, Martinez P, Martinez A, Felip E. Rectal metastases from squamous cell carcinoma: A case report and review of the literature. Case Rep Med. 2012;2012:947524. doi: 10.1155/2012/947524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lee PC, Lo C, Lin MT, Liang JT, Lin BR. Role of surgical intervention in managing gastrointestinal metastases from lung cancer. World J Gastroenterol. 2011;17:4314–4320. doi: 10.3748/wjg.v17.i38.4314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Fujiwara A, Okami J, Tokunaga T, Maeda J, Higashiyama M, Kodama K. Surgical treatment for gastrointestinal metastasis of non-small-cell lung cancer after pulmonary resection. Gen Thorac Cardiovasc Surg. 2011;59:748–752. doi: 10.1007/s11748-011-0811-3. [DOI] [PubMed] [Google Scholar]
- 49.Salemis NS, Nikou E, Liatsos C, Gakis C, Karagkiouzis G, Gourgiotis S. Small bowel perforation secondary to metastatic non-small cell lung cancer. A rare entity with a dismal prognosis. J Gastrointest Cancer. 2012;43:391–395. doi: 10.1007/s12029-011-9329-2. [DOI] [PubMed] [Google Scholar]
- 50.Yamada H, Akahane T, Horiuchi A, Shimada R, Shibuya H, Hayama T, Nozawa K, Ishihara S, Matsuda K, Watanabe T. A case of lung squamous cell carcinoma with metastases to the duodenum and small intestine. Int Surg. 2011;96:176–181. doi: 10.9738/1380.1. [DOI] [PubMed] [Google Scholar]
- 51.Ceretti AP, Goi G, Barabino M, De Nicola E, Strada D, Bislenghi G, Opocher E. Colonic metastasis from primary carcinoma of the lung. Case report. Ann Ital Chir. 2011;82:229–232. (In Italian) [PubMed] [Google Scholar]
- 52.Bugiantella W, Cavazzoni E, Graziosi L, Valiani S, Franceschini MS, Donini A. Small bowel metastasis from lung cancer: A possible cause of acute abdomen. Case report and literature review. G Chir. 2011;32:120–122. [PubMed] [Google Scholar]
- 53.Lin HC, Yu CP, Lin HA, Lee HS. A case of lung cancer metastasized to the gastrointestinal anastomosis site where the primary gastric cancer was resected 17 years ago. Lung Cancer. 2011;72:255–257. doi: 10.1016/j.lungcan.2011.02.005. [DOI] [PubMed] [Google Scholar]
- 54.Azevedo CR, Cezana L, Moraes ES, Begnami MD, Junior Paiva TF, Dettino AL, Fanelli MF. Synchronous thyroid and colon metastases from epidermoid carcinoma of the lung: Case report. Sao Paulo Med J. 2010;128:371–374. doi: 10.1590/S1516-31802010000600011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Trouillet N, Robert B, Charfi S, Bartoli E, Joly JP, Chatelain D. Gastric metastases. An endoscopic series of ten cases. Gastroenterol Clin Biol. 2010;34:305–309. doi: 10.1016/j.gcb.2010.01.019. [DOI] [PubMed] [Google Scholar]
- 56.Guerin E, Gilbert O, Dequanter D. Acute abdomen: A rare presentation of lung cancer metastasis. Case Rep Med. 2009;2009:903897. doi: 10.1155/2009/903897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Koch B, Tannapfel A, Vieth M, Grun R. Gastric metastasis from small cell lung cancer. Pneumologie. 2009;63:585–587. doi: 10.1055/s-0029-1214905. (In German) [DOI] [PubMed] [Google Scholar]
- 58.Yun IS, Lee JY, Lee JS, Lee JY, Byun JM, Kim EJ, Park JY, Park JK. Jejunal intussusception with gastrointestinal bleeding caused by metastatic lung cancer. Korean J Gastroenterol. 2008;51:377–380. (In Koren) [PubMed] [Google Scholar]
- 59.Aokage K, Yoshida J, Ishii G, Takahashi S, Sugito M, Nishimura M, Ochiai A, Nagai K. Long-term survival in two cases of resected gastric metastasis of pulmonary pleomorphic carcinoma. J Thorac Oncol. 2008;3:796–799. doi: 10.1097/JTO.0b013e31817c925c. [DOI] [PubMed] [Google Scholar]
- 60.Kim MS, Kook EH, Ahn SH, Jeon SY, Yoon JH, Han MS, Kim CH, Lee JC. Gastrointestinal metastasis of lung cancer with special emphasis on a long-term survivor after operation. J Cancer Res Clin Oncol. 2009;135:297–301. doi: 10.1007/s00432-008-0424-0. [DOI] [PubMed] [Google Scholar]
- 61.Goh BK, Yeo AW, Koong HN, Ooi LL, Wong WK. Laparotomy for acute complications of gastrointestinal metastases from lung cancer: Is it a worthwhile or futile effort? Surg Today. 2007;37:370–374. doi: 10.1007/s00595-006-3419-y. [DOI] [PubMed] [Google Scholar]
- 62.Kostakou C, Khaldi L, Flossos A, Kapsoritakis AN, Potamianos SP. Melena: A rare complication of duodenal metastases from primary carcinoma of the lung. World J Gastroenterol. 2007;13:1282–1285. doi: 10.3748/wjg.v13.i8.1282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ohashi K, Kiura K, Takigawa N, Mizushima T, Ino H, Tabata M, Ueoka H, Tanimoto M. Successful treatment of a patient with gastric and duodenal metastases from large cell carcinoma of the lung with carboplatin and gemcitabine. Anticancer Res. 2006;26:4695–4696. [PubMed] [Google Scholar]
- 64.Karamouzis MV, Linardou H, Papadopoulos G, Bousboukea E, Kanaloupiti D, Bitza M, Spourlis N, Bafaloukos D. Gastrointestinal solitary metastases from squamous cell lung cancer. Lung Cancer. 2007;55:251–252. doi: 10.1016/j.lungcan.2006.10.009. [DOI] [PubMed] [Google Scholar]
- 65.Kanemoto K, Kurishima K, Ishikawa H, Shiotani S, Satoh H, Ohtsuka M. Small intestinal metastasis from small cell lung cancer. Intern Med. 2006;45:967–970. doi: 10.2169/internalmedicine.45.1651. [DOI] [PubMed] [Google Scholar]
- 66.Casella G, Di Bella C, Cambareri AR, Buda CA, Corti G, Magri F, Crippa S, Baldini V. Gastric metastasis by lung small cell carcinoma. World J Gastroenterol. 2006;12:4096–4097. doi: 10.3748/wjg.v12.i25.4096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Altintas E, Sezgin O, Uyar B, Polat A. Acute upper gastrointestinal bleeding due to metastatic lung cancer: An unusual case. Yonsei Med J. 2006;47:276–277. doi: 10.3349/ymj.2006.47.2.276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Miyazaki K, Satoh H, Sekizawa K. Metastasis to appendix from lung adenocarcinoma. Int J Gastrointest Cancer. 2005;36:59–60. doi: 10.1385/IJGC:36:1:059. [DOI] [PubMed] [Google Scholar]
- 69.Garwood RA, Sawyer MD, Ledesma EJ, Foley E, Claridge JA. A case and review of bowel perforation secondary to metastatic lung cancer. Am Surg. 2005;71:110–116. [PubMed] [Google Scholar]
- 70.Morgan MW, Sigel B, Wolcott MW. Perforation of a metastatic carcinoma of the jejunum after cancer chemotherapy. Surgery. 1961;49:687–689. [PubMed] [Google Scholar]
- 71.Tillotson PM, Douglas RG., Jr Metastatic tumor of the small intestine. Three cases presenting unusual clinical and roentgenographic findings. Am J Roentgenol Radium Ther Nucl Med. 1962;88:702–706. [PubMed] [Google Scholar]
- 72.Hayashi K, Masuoka S, Kitade F. A case report of perforation of intestinal metastasis of lung cancer. Nihon Geka Hokan. 1965;34:816–819. (In Japanese) [PubMed] [Google Scholar]
- 73.Wootton DG, Morgan SC, Hughes RK. Perforation of a metastatic bronchogenic carcinoma to the jejunum. Ann Thorac Surg. 1967;3:57–59. doi: 10.1016/S0003-4975(10)66688-X. [DOI] [PubMed] [Google Scholar]
- 74.Wellmann KF, Chafiian Y, Edelman E. Small bowel perforation from solitary metastasis of clinically undetected pulmonary giant cell carcinoma. Am J Gastroenterol. 1969;51:145–150. [PubMed] [Google Scholar]
- 75.Midell AI, Lochman DJ. An unusual metastatic manifestation of a primary bronchogenic carcinoma. Cancer. 1972;30:806–809. doi: 10.1002/1097-0142(197209)30:3<806::AID-CNCR2820300332>3.0.CO;2-O. [DOI] [PubMed] [Google Scholar]
- 76.Inalsingh CH, Hazra T, Prempree T. Unusual metastases from carcinoma of the lung. J Can Assoc Radiol. 1974;25:242–244. [PubMed] [Google Scholar]
- 77.Ramanathan T, Skene-Smith H, Singh D, Sivanesan S. Small intestinal perforation due to secondaries from Bronchogenic carcinoma. Br J Dis Chest. 1976;70:121–124. doi: 10.1016/0007-0971(76)90018-8. [DOI] [PubMed] [Google Scholar]
- 78.Winchester DP, Merrill JR, Victor TA, Scanlon EF. Small bowel perforation secondary to metastatic carcinoma of the lung. Cancer. 1977;40:410–415. doi: 10.1002/1097-0142(197707)40:1<410::AID-CNCR2820400159>3.0.CO;2-0. [DOI] [PubMed] [Google Scholar]
- 79.Ejeckam GC, Abele R, Thomas J, Heringer R. Abdominal crisis due to metastasizing lung carcinoma to the small bowel. Can J Surg. 1979;22:351–353. [PubMed] [Google Scholar]
- 80.Sternberg A, Giler S, Segal I, Shmuter Z, Kott I. Small bowel perforation as the presenting symptom of squamous cell carcinoma of the lung. Clin Oncol. 1980;6:181–186. [PubMed] [Google Scholar]
- 81.Leidich RB, Rudolf LE. Small bowel perforation secondary to metastatic lung carcinoma. Ann Surg. 1981;193:67–69. doi: 10.1097/00000658-198101000-00011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Rosencrans DL. Small bowel perforation caused by metastatic lung carcinoma. Ann Surg. 1983;197:120. [PMC free article] [PubMed] [Google Scholar]
- 83.Quero Hernandez J, Tabernero Zubieta J, Martin Moreno A, Morales Caballero T, de la Higuera Torres-Puchol J, Parra Miras F, de la Higuera Rojas J. Pulmonary large-cell carcinoma with multiple metastases in the small intestine. Rev Clin Esp. 1982;165:285–286. [PubMed] [Google Scholar]
- 84.Catrambone G, Pesce L, Mazza M, Iurilli L. Perforation of the small intestine secondary to metastasis of pulmonary carcinoma. Minerva Chir. 1983;38:55–59. [PubMed] [Google Scholar]
- 85.Nicolosi A, Paderi R, Onnis P, Cafini D. Ileal perforation caused by metastasis of bronchogenic carcinoma. Minerva Chir. 1985;40:567–570. [PubMed] [Google Scholar]
- 86.Quayle AR, Holt S, Clark RG. Jejunal perforation secondary to metastatic bronchogenic carcinoma. Postgrad Med J. 1985;61:163–165. doi: 10.1136/pgmj.61.712.163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Vesth N, Jensen KH, Karkov J. Perforating small intestinal metastasis from a primary lung tumor. Ugeskr Laeger. 1985;147:3492. [PubMed] [Google Scholar]
- 88.McNeill PM, Wagman LD, Neifeld JP. Small bowel metastases from primary carcinoma of the lung. Cancer. 1987;59:1486–1489. doi: 10.1002/1097-0142(19870415)59:8<1486::AID-CNCR2820590815>3.0.CO;2-W. [DOI] [PubMed] [Google Scholar]
- 89.Pang JA, King WK. Bowel haemorrhage and perforation from metastatic lung cancer. Report of three cases and a review of the literature. Aust NZJ Surg. 1987;57:779–783. doi: 10.1111/j.1445-2197.1987.tb01261.x. [DOI] [PubMed] [Google Scholar]
- 90.Cavenaile JC, Blairon J, Limbosch JM. Jejunal perforation indicating the metastatic extension of a bronchial neoplasm. Acta Chir Belg. 1988;88:155–157. [PubMed] [Google Scholar]
- 91.Hwang GS, Yeh PF, Lee YC, Perng RP, Li WY. Small bowel perforation secondary to metastatic carcinoma of lung. Zhonghua Yi Xue Za Zhi (Taipei) 1988;41:159–164. (In Chinese) [PubMed] [Google Scholar]
- 92.Koury HI, Kenady D. Perforation of a metastatic lung adenocarcinoma of the jejunum. Am J Gastroenterol. 1988;83:462–463. [PubMed] [Google Scholar]
- 93.Shatikhin VA, Vasil'ev AV, Bychkov MB. Metastasis of lung cancer to the small intestine with its perforation. Klin Med (Mosk) 1988;66:135–136. [PubMed] [Google Scholar]
- 94.Shirani J, Brackett JW. Lung cancer presenting as small bowel perforation. Conn Med. 1989;53:455–456. [PubMed] [Google Scholar]
- 95.Joyce WP, Huddy SP, Corbishley C, Wright NL. Small bowel complications of metastatic lung carcinoma. Ir J Med Sci. 1990;159:149–150. doi: 10.1007/BF02937409. [DOI] [PubMed] [Google Scholar]
- 96.Woods JM, IV, Koretz MJ. Emergency abdominal surgery for complications of metastatic lung carcinoma. Arch Surg. 1990;125:583–585. doi: 10.1001/archsurg.1990.01410170029005. [DOI] [PubMed] [Google Scholar]
- 97.Nakano Y, Kamimori T, Shoji S, Taruya E, Tanaka I. A case of jejunal metastasis from pulmonary adenocarcinoma occurring as perforative peritonitis. Nihon Kyobu Shikkan Gakkai Zasshi. 1991;29:649–653. (In Japanese) [PubMed] [Google Scholar]
- 98.Yasunaga A, Shibata O, Sasaki T, Tohara K, Hadama T, Uchida Y, Yasunaga T, Adachi Y. A case of perforation of the metastatic site of lung carcinoma in the small bowel. Kyobu Geka. 1991;44:596–599. (In Japanese) [PubMed] [Google Scholar]
- 99.Beluffi L, Agostini M, Ruck F, Valenghi D, Morotti L, Marsetti M. Peritonitis caused by jejunal perforation resulting from metastasis of pulmonary carcinoma. Minerva Chir. 1992;47:1023–1026. (In Italian) [PubMed] [Google Scholar]
- 100.Gitt SM, Flint P, Fredell CH, Schmitz GL. Bowel perforation due to metastatic lung cancer. J Surg Oncol. 1992;51:287–291. doi: 10.1002/jso.2930510417. [DOI] [PubMed] [Google Scholar]
- 101.Ryo H, Sakai H, Ikeda T, Hibino S, Goto I, Yoneda S, Noguchi Y. Gastrointestinal metastasis from lung cancer. Nihon Kyobu Shikkan Gakkai Zasshi. 1996;34:968–972. (In Japanese) [PubMed] [Google Scholar]
- 102.Cossavella D, Paino O, Luc Realis A, Clerico G, Catania S, Pozzo M, Trompetto M. A rare form of intestinal perforation: adenocarcinoma of the ileum. Presentation of a clinical case. Minerva Chir. 1998;53:431–433. (In Italian) [PubMed] [Google Scholar]
- 103.Fischer M, Papp J, Kulka J, Zsiray M, Kempler P, Szalay F. Upper gastrointestinal bleeding and intestinal perforation due to multiple duodenojejunal metastases from a silent bronchogenic adenosquamous carcinoma. Endoscopy. 1998;30:S79. doi: 10.1055/s-2007-1001378. [DOI] [PubMed] [Google Scholar]
- 104.Shiraishi Y, Nakajima Y, Katsuragi N, Hanaoka T, Konno H, Tanaka S. Metastatic lung tumor: report of two cases. Kyobu Geka. 2003;56:47–50. [PubMed] [Google Scholar]
- 105.Yokota T, Yamada Y, Sakata N, Kikuchi S, Kunii Y, Tezuka F, Suzuki H, Yamauchi H. Emergency abdominal surgery for small bowel perforation secondary to metastatic lung cancer. Tohoku J Exp Med. 1999;188:265–270. doi: 10.1620/tjem.188.265. [DOI] [PubMed] [Google Scholar]
- 106.Ise N, Kotanagi H, Morii M, Yasui O, Ito M, Koyama K, Sageshima M. Small bowel perforation caused by metastasis from an extra-abdominal malignancy: Report of three cases. Surg Today. 2001;31:358–362. doi: 10.1007/s005950170161. [DOI] [PubMed] [Google Scholar]
- 107.Polak M, Kupryjańczyk J, Rell KW. A rare case of colonic perforation in a sole site of latent lung cancer metastasis. Pol Tyg Lek. 1990;45:179–181. (In Polish) [PubMed] [Google Scholar]
- 108.Rahman R, Bernstein Z, Vaickus L, Penetrante R, Arbuck S, Kopec I, Vesper D, Douglass HO, Jr, Foon KA. Unusual gastrointestinal complications of interleukin-2 therapy. J Immunother (1991) 1991;10:221–225. doi: 10.1097/00002371-199106000-00009. [DOI] [PubMed] [Google Scholar]
- 109.Fletcher MS. Gastric perforation secondary to metastatic carcinoma of the lung: a case report. Cancer. 1980;46:1879–1882. doi: 10.1002/1097-0142(19801015)46:8<1879::AID-CNCR2820460829>3.0.CO;2-A. [DOI] [PubMed] [Google Scholar]
- 110.Schmidt G, Börsch G, von Liebe S, Böhm E. Gastric perforation secondary to metastatic bronchogenic carcinoma. Hepatogastroenterology. 1985;32:103–105. [PubMed] [Google Scholar]
- 111.Opanasenko NS. Gastric perforation into the left pleural cavity after pleuropulmonectomy for pulmonary cancer. Klin Khir. 2000:56–57. (In Russian) [PubMed] [Google Scholar]
- 112.Suzaki N, Hiraki A, Ueoka H, Aoe M, Takigawa N, Kishino T, Kiura K, Kanehiro A, Tanimoto M, Harada M. Gastric perforation due to metastasis from adenocarcinoma of the lung. Anticancer Res. 2002;22:1209–1212. [PubMed] [Google Scholar]
- 113.Sanli Y, Adalet I, Turkmen C, Kapran Y, Tamam M, Cantez S. Small bowel metastases from primary carcinoma of the lung: Presenting with gastrointestinal hemorrhage. Ann Nucl Med. 2005;19:161–163. doi: 10.1007/BF03027397. [DOI] [PubMed] [Google Scholar]
- 114.Tomas D, Ledinsky M, Belicza M, Kruslin B. Multiple metastases to the small bowel from large cell bronchial carcinomas. World J Gastroenterol. 2005;11:1399–1402. doi: 10.3748/wjg.v11.i9.1399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Misra SP, Dwivedi M, Misra V, Dharmani S, Gupta M. Duodenal metastases from squamous cell carcinoma of the lung: Endoscopic management of bleeding and biliary and duodenal obstruction. Indian J Gastroenterol. 2004;23:185–186. [PubMed] [Google Scholar]
- 116.Katsinelos P, Paroutoglou G, Beltsis A, Pilpilidis I, Papaziogas B, Mimidis K, Tsolkas P. Hematemesis as a presenting symptom of lung cancer with synchronous metastases to the esophagus and stomach. A case report. Rom J Gastroenterol. 2004;13:251–253. [PubMed] [Google Scholar]
- 117.Kobayashi O, Murakami H, Yoshida T, Cho H, Yoshikawa T, Tsuburaya A, Sairenji M, Motohashi H, Sugiyama Y, Kameda Y. Clinical diagnosis of metastatic gastric tumors: Clinicopathologic findings and prognosis of nine patients in a single cancer center. World J Surg. 2004;28:548–551. doi: 10.1007/s00268-004-7216-8. [DOI] [PubMed] [Google Scholar]
- 118.Capasso L, Iarrobino G, D'Ambrosio R, Carfora E, Ventriglia R, Borsi E. Surgical complications for gastric and small bowel metastases due to primary lung carcinoma. Minerva Chir. 2004;59:397–403. [PubMed] [Google Scholar]
- 119.Renault PA, Arotçarena R, Calès V, Lippa A, Benichou M, Laurent P, Laborde Y. Metastatic obstruction of the small bowel revealing or complicating squamous-cell lung cancer. Two cases and a review of the literature. Rev Pneumol Clin. 2003;59:161–165. [PubMed] [Google Scholar]
- 120.Yamamoto M, Matsuzaki K, Kusumoto H, Uchida H, Mine H, Kabashima A, Maehara Y, Sugimachi K. Gastric metastasis from lung carcinoma. Case report. Hepatogastroenterology. 2002;49:363–365. [PubMed] [Google Scholar]
- 121.Ito Y, Suzuki M, Oyamada Y, Kou H, Takeshita K, Asano K, Yamaguchi K. A case of relapsed small cell lung cancer recognized by simple metastasis to the duodenum. Nihon Kokyuki Gakkai Zasshi. 2001;39:30–34. (In Japanese) [PubMed] [Google Scholar]
- 122.Fukata T, Fukino S, Hayashi E, Okada K, Tamai N, Nakashima H. A case of G-CSF-producing large cell carcinoma of the lung with gastric metastasis. Kyobu Geka. 2000;53:798–803. (In Japanese) [PubMed] [Google Scholar]
- 123.Berger A, Cellier C, Daniel C, Kron C, Riquet M, Barbier JP, Cugnenc PH, Landi B. Small bowel metastases from primary carcinoma of the lung: clinical findings and outcome. Am J Gastroenterol. 1999;94:1884–1887. doi: 10.1111/j.1572-0241.1999.01224.x. [DOI] [PubMed] [Google Scholar]
- 124.Centeno Cortés C, Clavero Borau MJ, Rubiales Sanz A, Martín López-Lara F. Intestinal bleeding in disseminated non-small cell lung cancer. Lung Cancer. 1997;18:101–105. doi: 10.1016/S0169-5002(97)00050-0. [DOI] [PubMed] [Google Scholar]
- 125.Akahoshi K, Chijiiwa Y, Hirota I, Ohogushi O, Motomatsu T, Nawata H, Sasaki I. Metastatic large-cell lung carcinoma presenting as gastrointestinal hemorrhage. Acta Gastroenterol Belg. 1996;59:217–219. [PubMed] [Google Scholar]
- 126.Raijman I. Duodenal metastases from lung cancer. Endoscopy. 1994;26:752–753. doi: 10.1055/s-2007-1009092. [DOI] [PubMed] [Google Scholar]
- 127.Gateley CA, Lewis WG, Sturdy DE. Massive lower gastrointestinal haemorrhage secondary to metastatic squamous cell carcinoma of the lung. Br J Clin Pract. 1993;47:276–277. [PubMed] [Google Scholar]
- 128.Maeda J, Miyake M, Tokita K, Iwahashi N, Nakano T, Tamura S, Hada T, Higashino K. Small cell lung cancer with extensive cutaneous and gastric metastases. Intern Med. 1992;31:1325–1328. doi: 10.2169/internalmedicine.31.1325. [DOI] [PubMed] [Google Scholar]
- 129.Mosier DM, Bloch RS, Cunningham PL, Dorman SA. Small bowel metastases from primary lung carcinoma: A rarity waiting to be found? Am Surg. 1992;58:677–682. [PubMed] [Google Scholar]
- 130.Park SW, Cho HJ, Choo WS, Chung KS, Kim HY, Yoo JY, Kim JS, Shin HS. A case of intestinal hemorrhage due to small intestinal metastases from primary lung cancer. Korean J Intern Med. 1991;6:79–84. doi: 10.3904/kjim.1991.6.2.79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Spedini C, Lombardi C, Buffoli F. Multiple mucosal gastrointestinal metastases from primary asymptomatic bronchogenic carcinoma. Recenti Prog Med. 1990;81:442–444. [PubMed] [Google Scholar]
- 132.Ono H, Okabe M, Kimura T, Kawakami M, Nakamura K, Danjo Y, Takasugi H, Nishihara H. Colonic metastasis from primary carcinoma of the lung: Report of a case and review of Japanese literature. Clin J Gastroenterol. 2009;2:89–95. doi: 10.1007/s12328-008-0053-9. [DOI] [PubMed] [Google Scholar]
- 133.Stinchcombe TE, Socinski MA, Gangarosa LM, Khandani AH. Lung cancer presenting with a solitary colon metastasis detected on positron emission tomography scan. J Clin Oncol. 2006;24:4939–4940. doi: 10.1200/JCO.2006.06.3354. [DOI] [PubMed] [Google Scholar]
- 134.Travis WD, Brambilla E, Nicholson AG, Yatabe Y, Austin JH, Beasley MB, Chirieac LR, Dacic S, Duhig E, Flieder DB, et al. The 2015 World Health Organization classification of lung tumors: impact of genetic, clinical and radiologic advances since the 2004 classification. J Thorac Oncol. 2015;10:1243–1260. doi: 10.1097/JTO.0000000000000630. [DOI] [PubMed] [Google Scholar]
- 135.Edge S, Byrd D, Compton C, Fritz A, Greene F, Trotti A. AJCC Cancer Stageing Manual. Springer; New York: 2010. [Google Scholar]
- 136.Kim CH, Lee YC, Hung RJ, McNallan SR, Cote ML, Lim WY, Chang SC, Kim JH, Ugolini D, Chen Y, et al. Exposure to secondhand tobacco smoke and lung cancer by histological type: A pooled analysis of the International Lung Cancer Consortium (ILCCO) Int J Cancer. 2014;135:1918–1930. doi: 10.1002/ijc.28835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Cox D, Oakes N. Analysis of Survival Data. Chapman and Hall; New York: 1984. [Google Scholar]
- 138.Guo Z, Han B, Wang Y. The status of diagnosis and therapy for gastrointestinal metastasis from primary lung cancer. Zhongguo Fei Ai Za Zhi. 2011;14:69–71. doi: 10.3779/j.issn.1009-3419.2011.01.14. (In Chinese) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Hillenbrand A, Sträter J, Henne-Bruns D. Frequency, symptoms and outcome of intestinal metastases of bronchopulmonary cancer. Case report and review of the literature. Int Semin Surg Oncol. 2005;2:13. doi: 10.1186/1477-7800-2-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Yuen JS, Chow PK, Ahmed Q. Metastatic lung cancer causing bowel perforations: Spontaneous or chemotherapy-related? ANZ J Surg. 2002;72:245–246. doi: 10.1046/j.1445-2197.2002.02236.x. [DOI] [PubMed] [Google Scholar]
- 141.Kim SY, Ha HK, Park SW, Kang J, Kim KW, Lee SS, Park SH, Kim AY. Gastrointestinal metastasis from primary lung cancer: CT findings and clinicopathologic features. AJR Am J Roentgenol. 2009;193:W197–W201. doi: 10.2214/AJR.08.1907. [DOI] [PubMed] [Google Scholar]
- 142.Di JZ, Peng JY, Wang ZG. Prevalence, clinicopathological characteristics, treatment, and prognosis of intestinal metastasis of primary lung cancer: A comprehensive review. Surg Oncol. 2014;23:72–80. doi: 10.1016/j.suronc.2014.02.004. [DOI] [PubMed] [Google Scholar]
- 143.Israel O, Yefremov N, Bar-Shalom R, Kagana O, Frenkel A, Keidar Z, Fischer D. PET/CT detection of unexpected gastrointestinal foci of 18F-FDG uptake: Incidence, localization patterns and clinical significance. J Nucl Med. 2005;46:758–762. [PubMed] [Google Scholar]
- 144.Kim MS, Cheon GJ, Lim SM, Kim CH, Lee JC. F-18 FDG PET-CT imaging of intestinal metastasis from primary lung cancer. Clin Nucl Med. 2008;33:870–871. doi: 10.1097/RLU.0b013e31818bf256. [DOI] [PubMed] [Google Scholar]
- 145.Kini S, Kapadia RM, Amarapurkar A. Intussusception due to intestinal metastasis from lung cancer. Indian J Pathol Microbiol. 2010;53:141–143. doi: 10.4103/0377-4929.59208. [DOI] [PubMed] [Google Scholar]