Abstract
Amphetamine maintenance is effective clinically to reduce the consumption of the monoamine uptake inhibitor cocaine but not of the monoamine releaser methamphetamine, and its effectiveness in treating the abuse of other psychostimulants is not known. The mechanisms for differential amphetamine-maintenance effectiveness to treat different types of psychostimulant abuse are also not known. Accordingly, the present study compared the effects of amphetamine maintenance on abuse-related behavioral and neurochemical effects of cocaine, methamphetamine, and the “bath salts” constituent 3,4-methylenedioxypyrovalerone (MDPV) in rats. In behavioral studies, rats were trained to lever press for electrical brain stimulation in an intracranial self-stimulation (ICSS) procedure. In neurochemical studies, nucleus accumbens (NAc) levels of dopamine (DA) and serotonin (5-HT) were monitored by in vivo microdialysis. Cocaine, methamphetamine, and MDPV each produced dose-dependent ICSS facilitation and increases in NAc DA; cocaine and methamphetamine also increased NAc 5-HT. Amphetamine maintenance (0.32 mg/kg/h × 7 days) produced (1) sustained increases in basal ICSS and NAc DA with no change in NAc 5-HT, (2) blockade of cocaine but not methamphetamine effects on ICSS and NAc DA, and (3) no blockade of cocaine- or methamphetamine-induced increases in NAc 5-HT. Amphetamine maintenance blocked the increases in NAc DA produced by the selective DA uptake inhibitor MDPV, but it did not block MDPV-induced ICSS facilitation. These results show different effects of amphetamine maintenance on behavioral and neurochemical effects of different psychostimulants. The selective effectiveness of amphetamine maintenance to treat cocaine abuse may reflect attenuation of cocaine-induced increases in NAc DA while preserving cocaine-induced increases in NAc 5-HT.
Introduction
There are more than 2 million current (i.e., past month) psychostimulant users in the United States [1], and no pharmacotherapies are currently approved by the Food and Drug Administration for the treatment of psychostimulant abuse. According to the 2016 National Survey on Drug Use and Health [1], cocaine and methamphetamine are the two most commonly abused psychostimulants, and other drugs such as 3,4-methylenedioxypyrovalerone (MDPV) have emerged as problem drugs of abuse during the last decade [2]. Many psychostimulants produce their effects by interacting with transporters for the monoamine neurotransmitters dopamine (DA), serotonin (5-HT), and norepinephrine (NE) (DAT, SERT, and NET, respectively) to increase synaptic monoamine levels in brain areas such as nucleus accumbens (NAc), and drugs that are more potent to increase DA vs. 5-HT tend to produce more robust abuse-related effects [3–6]. Moreover, there are two broad classifications of monoamine transporter ligands based on their transporter interactions. Uptake inhibitors like cocaine and MDPV bind to and block transporters to promote extracellular accumulation of the associated monoamine, whereas releasers like methamphetamine and amphetamine pass through the transporters and cause a cascade of events leading to monoamine efflux [7, 8].
Although no pharmacotherapies are approved to treat psychostimulant use disorders, amphetamine maintenance decreases cocaine use in double-blind placebo-controlled clinical trials [9–13] and also decreases the choice of cocaine over an alternative reinforcer in laboratory studies in humans, non-human primates, and rats [14–16]. In contrast, amphetamine maintenance is not effective in decreasing methamphetamine use in either clinical trials or preclinical studies [17, 18], and the effects of amphetamine maintenance on abuse-related effects of other psychostimulants like MDPV are unknown. Additionally, the mechanisms that underlie selective amphetamine-maintenance effects on cocaine vs. methamphetamine use remain to be determined. Amphetamine maintenance is thought to function as an agonist-type therapy for cocaine abuse because both drugs produce similar behavioral effects and increase NAc DA levels [19, 20]; however, it is not clear why such an agonist-type effect would be selective for cocaine but not methamphetamine.
Intracranial self-stimulation (ICSS) is one preclinical procedure that can be used to evaluate the effects of candidate medications on abuse-related drug effects [21, 22], and we reported previously that an amphetamine-maintenance regimen sufficient to reduce cocaine-vs.-food choice in rats [16] also blunted cocaine-induced ICSS facilitation in rats [23]. The present study extended this finding in three ways. First, amphetamine-maintenance effects were compared on ICSS facilitation produced by cocaine, MDPV, and methamphetamine. Second, ICSS facilitation by monoamine transporter ligands correlates with selectivity to increase DA vs. 5-HT levels in NAc [5, 24]. Accordingly, we also used in vivo microdialysis to compare amphetamine-maintenance effects on cocaine-, MDPV-, and methamphetamine-induced changes in NAc DA and 5-HT levels. Given the preclinical and clinical effects of amphetamine maintenance on cocaine and methamphetamine self-administration, we hypothesized that amphetamine maintenance would block the effects of cocaine, but not of methamphetamine, on both ICSS and NAc DA levels, whereas changes in MDPV effects were more difficult to predict. Lastly, effects of amphetamine maintenance on striatal DAT density were also determined, because DAT downregulation is one possible mechanism of amphetamine maintenance-induced decreases in cocaine effects [25, 26].
Methods
Subjects
Studies were conducted in 119 male Sprague–Dawley rats (Envigo, Frederick, MD), weighing 300–350 g at the time of surgery. Rats were individually housed on a 12-h light–dark cycle (lights on 6 a.m.–6 p.m.) in a facility accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care. All rats had ad libitum access to food and water in their home cages. Animal maintenance accorded with The National Institutes of Health guidelines on care and use of research animals, and experimental protocols were approved by the Virginia Commonwealth University Institutional Animal Care and Use Committee. Procedures for implantation of microelectrodes, guide cannulae, and minipumps were identical to those published previously [5, 23]. Separate groups of rats were used for studies of ICSS, microdialysis, and receptor binding.
Intracranial self-stimulation
Training
Studies were conducted in operant chambers (Med Associates, St. Albans, VT) using procedures for training, testing, and data analysis identical to those described previously [4, 23, 27, 28]. Daily behavioral sessions consisted of three 10-min components, each consisting of ten 1-min trials. Each trial presented a different frequency of electrical stimulation available for brain stimulation, and the frequency decreased in 0.05 log increments across trials from 158 to 56 Hz. During the first 10 s of each trial, five non-contingent stimulations were delivered. During the remaining 50 s, lever presses produced brain stimulation and illumination of the stimulus lights over the lever under an FR 1 reinforcement schedule. Training continued until stabilization (3 days during which the maximum control rate (MCR) of stimulations per trial and total stimulations per component on each day were within 15% of the running mean across days). This was completed within 12 weeks of surgery, and the final 3 days of training served to establish the “Pre-pump” baseline for ICSS performance.
Testing
Once the pre-pump baseline performance was established, drug testing was conducted using an 8-day treatment protocol. On Days 0 and 7, the rats received a series of increasing intraperitoneal (IP) doses of cocaine (1, 3.2, 10 mg/kg), MDPV (0.1, 0.32, 1.0 mg/kg), or methamphetamine (0.1, 0.32, 1.0 mg/kg). Dose ranges of each drug were selected based on previous studies that showed dose-dependent ICSS facilitation by each drug [4, 27, 29]. Dose-effect sessions consisted of three baseline ICSS components followed by three drug injections administered at 30-min intervals. Two ICSS test components began 10 min after each injection. On Day 1, osmotic minipumps containing either saline, 0.1 or 0.32 mg/kg/h amphetamine were surgically implanted, and three-component ICSS sessions were conducted on Days 2–6 before the second dose-effect session on Day 7. Groups of N = 6 rats were used to test each minipump treatment in combination with each test drug, and only cocaine was tested with 0.1 mg/kg/h amphetamine.
Data analysis
Data from the first ICSS component on each day were discarded. The principal dependent measure was the number of stimulations per trial. These data were normalized for each rat to a percent maximum control rate (%MCR), with MCR defined as the average of the maximal number of stimulations in any trial of the 2nd and 3rd components of the three pre-pump baseline sessions, and %MCR is calculated using the equation %MCR = (stimulations per trial/MCR) × 100. Data from the three pre-pump baseline sessions were averaged within each rat and then across rats to generate a group pre-pump baseline “frequency–rate” curve. For dose-effect sessions, data from each pair of baseline and test components were averaged within each rat and then across rats to generate group mean frequency-rate curves for baseline and each drug dose. Frequency-rate curves were compared by repeated measures two-way ANOVA, with ICSS frequency and the experimental manipulation as the two factors. A significant interaction was followed by Holm–Sidak post-hoc test. The criterion for significance for this and all other analyses was set at p < 0.05. Additionally, the EF50 for each ICSS curve was defined as the “effective frequency” that maintained 50% MCR. EF50 values and 95% confidence limits were interpolated by linear regression from the linear portion of each ICSS curve, and EF50 values were considered to be different if 95% confidence limits did not overlap. In some cases, all points were above 50% MCR; in these cases, interpolation of EF50 values was not possible, and EF50 is shown as “<1.75” because this was the lowest frequency tested.
Microdialysis
Procedure
Procedures for the collection and analysis of NAc microdialysis samples were similar to those described previously [5]. Baseline samples were collected at 10-min intervals until DA and 5-HT levels stabilized (six consecutive baseline samples with <25% variability around the running mean). Subsequently, an injection was administered IP, and samples were collected for another 100 min. Initial studies evaluated the effects of saline and multiple doses of cocaine (1.0, 3.2, 10 mg/kg), MDPV (0.1, 0.32, 1.0 mg/kg), or methamphetamine (0.1, 0.32, 1.0 mg/kg) identical to those tested in ICSS. Each treatment was tested in six rats. Next, to determine the effects of amphetamine maintenance on test-drug effects, new groups were surgically implanted with minipumps containing either saline or 0.32 mg/kg/h amphetamine and tested between 7 and 13 days after minipump implantation with a dose of cocaine (10 mg/kg), MDPV (0.32 mg/kg), or methamphetamine (0.32 mg/kg) that produced similar 200–250% increases in NAc DA levels during initial dose-effect studies. Each minipump treatment and test-drug group contained six rats.
Data analysis
The dependent variables were DA and 5-HT concentrations in each dialysate fraction. For dose-effect studies, data at each time point were expressed as a percentage of the baseline concentration for each neurotransmitter: %Baseline = (test concentration/baseline concentration) × 100. Results were averaged across the rats and analyzed at each drug dose using repeated-measures one-way ANOVA, with time as a fixed effect and subject as a random effect. A significant ANOVA was followed by Dunnett’s post-hoc test to compare the monoamine concentrations at each time point with control concentrations determined 10 min after the drug administration. This sample was selected as the control because preliminary experiments indicated a lag time of ~20 min for dialysate to traverse the tubing from the probe to the electrochemical detector at the 1 μL/min flow rate. Accordingly, the 10-min sample was collected prior to drug administration, had advanced into the auto-injector tubing at the time of drug injection, and was evaluated after drug injection.
For minipump studies, baseline DA and 5-HT levels in saline-treated and amphetamine-treated rats were compared by Student’s t-test with Welch’s correction. Because amphetamine maintenance significantly increased the baseline DA levels (see Results), data from minipump studies were expressed as a difference from baseline: Delta baseline = test concentration−baseline concentration. The effects of cocaine, MDPV, and methamphetamine over time during saline or amphetamine maintenance were analyzed by one-way ANOVA as above. Additionally, results of each drug across maintenance treatments were compared by two-way ANOVA with time and treatment as the two factors. A significant interaction was followed by a Holm–Sidak post-hoc test.
[3H]WIN35,428 saturation binding
Membrane preparation
The rats were implanted with minipumps containing saline (N = 6) or 0.32 mg/kg/h amphetamine (N = 8). After 7 days of treatment, the rats were euthanized by rapid decapitation, and whole striatum, including NAc and caudate/putamen, were dissected on ice and frozen at −80 °C until use. On the day of each binding assay, the samples were thawed in cold assay buffer (20 mM sodium phosphate buffer, pH 7.9 with 0.32 M sucrose), homogenized with a Polytron homogenizer, and centrifuged at 50,000×g at 4 °C for 10 min. The supernatant was discarded, the pellet was re-suspended by homogenization in assay buffer, and the protein concentration was determined by the Bradford method.
Binding assay
The DA transporter inhibitors WIN35,428 [(–)-2β-carbomethoxy-3β-(4-fluorophenyl)tropane] and RTI-112 [2β-carbomethoxy-3β-(3-methyl-4-chlorophenyl)tropane] were used as the radiolabeled and non-labeled ligands, respectively [30]. Six concentrations of [3H]WIN35,428 (approximately 0.4–30 nM) were incubated in assay buffer containing 40 µg membrane protein for 90 min at 30 °C in a final volume of 0.25 ml. Non-specific binding was determined at each concentration of radioligand in the presence of 30 µM unlabeled RTI-112. The incubation was terminated by rapid filtration under vacuum through GF/B glass fiber filters using a 48-well Brandel harvester and rinsed three times with 3 ml ice-cold 50 mM Tris-HCl, pH 7.4. Bound radioactivity was determined by liquid scintillation spectrophotometry at 45% efficiency for 3H after overnight equilibration of the filters in Econosafe scintillation fluid (Research Products International, Prospect, IL).
Data analysis
Data are reported as specific binding, derived from duplicate determinations. Bmax and KD values were determined by iterative curve fitting using single-site saturation analysis (nH = 1) and compared in saline- and amphetamine-treated groups by two-tailed unpaired Student’s t-test.
Drugs
For behavioral and microdialysis studies, (−)-cocaine HCl, (±)-MDPV HCl, and (+)-amphetamine hemisulfate were obtained from the National Institute for Drug Abuse Drug Supply Program (Bethesda, MD). (+)-Methamphetamine HCl was purchased from Sigma Chemical Co. (St. Louis, MO). All drugs were dissolved in bacteriostatic saline. Cocaine, MDPV, and methamphetamine were administered IP in a volume of 1 ml/kg. Amphetamine was delivered subcutaneously (SC) via osmotic minipump (2ML2, Alzet, Cupertino, CA) at a rate of 5 µl/h. For receptor binding studies, [3H]WIN35,428 (82.6 Ci/mmol) was purchased from Perkin–Elmer (Waltham, MA). RTI-112 was kindly provided by Dr. F. Ivy Carroll of Research Triangle Institute (Research Triangle Park, NC).
Results
Intracranial self-stimulation
Pre-pump baseline performance
During pre-pump baseline sessions for all rats in the study, the mean ± SEM MCR was 55.06 ± 1.5 reinforcements per trial and the mean EF50 (95% confidence limits) was 2.02 (2.00–2.03) log Hz. One-way ANOVA indicated no difference in MCRs across treatment groups (F(6,35) = 1.35, n.s.), and overlapping confidence limits indicated no difference in pre-pump baseline EF50 values (Table 1).
Table 1.
EF50 values (95% confidence limits) in log Hz after the administration of cocaine, MDPV, or methamphetamine in rats tested before implantation of minipumps (Pre-pump) or 7 days after implantation of minipumps that delivered either saline or 0.32 mg/kg/h amphetamine
| Drug | Dose (mg/kg) | Pre-pump | Saline | Amphetamine |
|---|---|---|---|---|
| Cocaine | Baseline | 2.02 (1.99–2.05) | 2.02 (2.02–2.03) | 1.90 (1.88–1.92) |
| 1.0 | 1.93 (1.91–1.94)a | 1.96 (1.93–1.99)a | 1.87 (1.83–1.90) | |
| 3.2 | 1.89 (1.88–1.90)a | 1.88 (1.83–1.92)a | 1.91 (1.88–1.93) | |
| 10 | 1.75 (1.49–1.80)a | 1.77 (1.57–1.83)a | 1.83 (1.82–1.84)a | |
| MDPV | Baseline | 2.02 (1.97–2.06) | 2.01 (2.01–2.02) | 1.86 (1.80–1.89) |
| 0.1 | 1.97 (1.96–1.99) | 1.98 (1.94–2.02) | 1.84 (1.76–1.89) | |
| 0.32 | 1.80 (1.78–1.82)a | <1.75b | <1.75b | |
| 1.0 | <1.75b | <1.75b | <1.75b | |
| Methamphetamine | Baseline | 2.01 (1.99–2.04) | 2.03 (2.00–2.05) | 1.92 (1.91–1.94) |
| 0.1 | 1.95 (1.94–1.95)a | 1.93 (1.92–1.94)a | 1.89 (1.88–1.90)a | |
| 0.32 | 1.83 (1.82–1.83)a | 1.83 (1.78–1.87)a | 1.77 (1.75–1.79)a | |
| 1.0 | <1.75b | <1.75b | <1.75b |
aIndicates significantly different from “Baseline” as determined by non-overlapping confidence limits
bIndicates all points >50% MCR, and EF50 could not be calculated. This outcome was also considered to be significantly different from baseline
Pre-pump effects of cocaine, MDPV, and methamphetamine
Supplemental Fig. 1 and Table 1 show the effect of cocaine (1.0–10.0 mg/kg), MDPV (0.1–1.0 mg/kg), and methamphetamine (0.1–1.0 mg/kg) on Day 0, before the minipumps were implanted. Data are combined for saline- and amphetamine-treated rats because these data were collected before the minipumps were implanted and before the treatments had started. Brain stimulation maintained a frequency-dependent increase in reinforcement rates under baseline conditions, and all three drugs produced dose-dependent leftward/upward shifts in ICSS frequency–rate curves. Table 1 shows that all three drugs also produced dose-dependent decreases in EF50 values. EF50 values could not be determined for the highest doses of MDPV and methamphetamine, because facilitation was so robust that all points on the frequency–rate curves were above 50% MCR.
Effects of saline or amphetamine maintenance on baseline ICSS
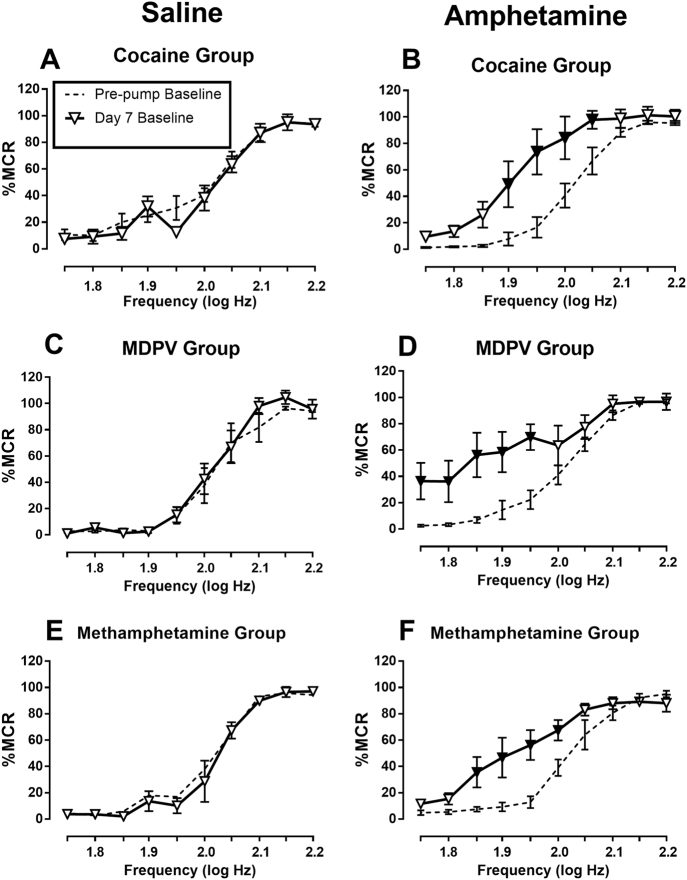
Figure 1 shows the effects of saline or 0.32 mg/kg/h amphetamine maintenance on baseline ICSS. In saline-treated rats, the Day 7 baseline frequency–rate curves were not different from the pre-pump baseline in any group (no main effect of day and no significant day × frequency interaction, panels a, c, and e). Conversely, 0.32 mg/kg/h amphetamine maintenance facilitated ICSS in all three groups [day × frequency interactions: (b) F(9,45) = 3.45, p = 0.0027, (d) F(9,45) = 4.18, p = 0.0006, (f) F(9,45) = 3.24, p = 0.0041]. Additionally, Table 1 shows that EF50 values in saline-treated rats were similar to pre-pump baselines, whereas 0.32 mg/kg/h amphetamine reduced EF50 values. Supplemental Fig. 2A shows that a lower maintenance dose of 0.1 mg/kg/h amphetamine also facilitated ICSS in rats that were subsequently treated with cocaine.
Fig. 1.
Effects of maintenance on saline or 0.32 mg/kg/h amphetamine on baseline ICSS performance in rats subsequently tested with cocaine (a, b), MDPV (c, d), or methamphetamine (e, f). Abscissae: brain stimulation frequency in log Hz. Ordinates: % maximum control rate. All points show mean ± SEM from N = 6 rats, and filled points indicate significantly different from “Pre-pump Baseline” as determined by two-way ANOVA followed by the Holm–Sidak post-hoc test (p < 0.05)
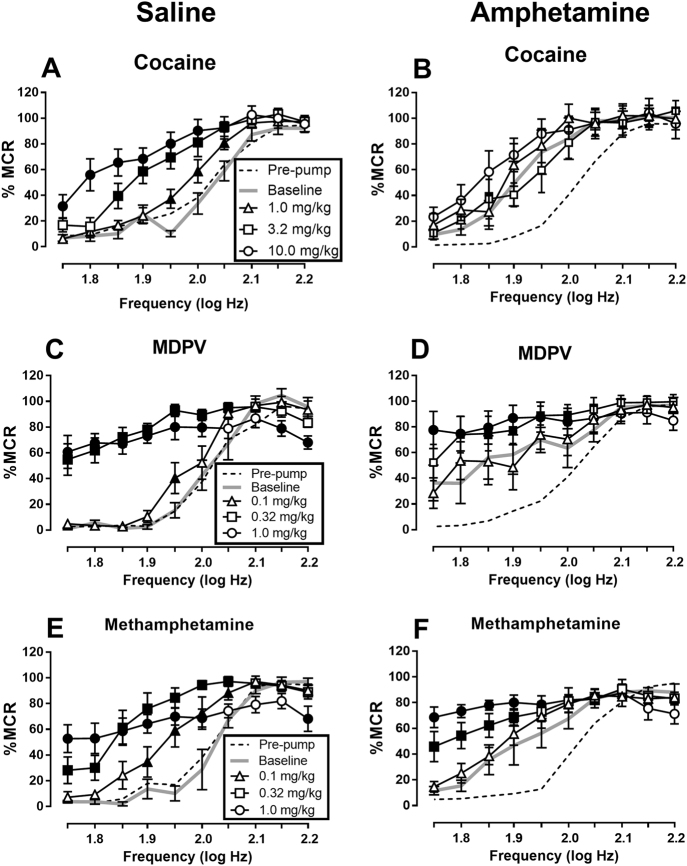
Effects of cocaine, MDPV, and methamphetamine on ICSS during saline or amphetamine maintenance
Figure 2 shows the effects of cocaine, MDPV, and methamphetamine in rats treated with saline or 0.32 mg/kg/h amphetamine, and EF50 values are shown in Table 1. Effects of all three drugs during saline maintenance were similar to pre-pump effects. Amphetamine maintenance produced at least a 10-fold decrease in the potency of cocaine to facilitate ICSS. Thus, during saline treatment, cocaine dose-dependently facilitated ICSS at all three doses as indicated both by two-way ANOVA of frequency–rate data (Fig. 2a: dose × frequency interaction F(27,135) = 3.93, p < 0.0001) and by reductions in EF50 values (Table 1). However, during maintenance on 0.32 mg/kg/h amphetamine, there was a main effect of cocaine dose on ICSS frequency–rate curves (Fig. 2b: F(3,15) = 3.92, p = 0.030) but no frequency × dose interaction (F(27,135) = 1.52, n.s.). Dunnett’s post-hoc test revealed that 10 mg/kg cocaine was different than baseline (p < 0.05). Moreover, only 10 mg/kg cocaine significantly reduced EF50 values (Table 1). Supplemental Fig. 2B shows that maintenance on a lower amphetamine dose (0.1 mg/kg/h) failed to blunt cocaine-induced ICSS facilitation.
Fig. 2.
Effects of cocaine (a, b), MDPV (c, d), and methamphetamine (e, f) on ICSS on Day 7 of treatment with either saline (a, c, e) or 0.32 mg/kg/h amphetamine (b, d, f). Abscissae: brain stimulation frequency in log Hz. Ordinates: % maximum control rate. All points show mean ± SEM from N = 6 rats, and filled points indicate significantly different from “Baseline” as determined by two-way ANOVA followed by the Holm–Sidak post-hoc test (p < 0.05)
Maintenance on 0.32 mg/kg/h amphetamine had lesser effects on MDPV-induced ICSS facilitation. In saline-treated rats, the lowest dose of 0.1 mg/kg MDPV significantly increased ICSS at only one frequency (1.95 log Hz) and failed to alter the EF50, and higher doses of 0.32 and 1.0 mg/kg MDPV produced robust ICSS facilitation across a broad range of frequencies (Fig. 2c: dose × frequency interaction F(27,135) = 11.03, p < 0.0001) and also significantly reduced EF50 values (Table 1). In rats treated with 0.32 mg/kg/h amphetamine, 0.1 mg/kg MDPV did not facilitate ICSS at any frequency or reduce the EF50, but higher doses still facilitated ICSS by both analyses (Fig. 2d: dose × frequency interaction F(27,135) = 3.32, p < 0.0001; Table 1).
Amphetamine maintenance also had only modest effects on methamphetamine-induced ICSS facilitation. In saline-treated rats, all methamphetamine doses facilitated ICSS both by two-way ANOVA of frequency–rate data (Fig. 2e: dose × frequency interaction F(27,135) = 9.13, p < 0.0001) and by significant reductions in EF50 values (Table 1). In rats treated with 0.32 mg/kg/h amphetamine, the lowest dose of 0.1 mg/kg methamphetamine no longer facilitated ICSS by analysis of frequency–rate curves, but this dose did still produce a significant, if small, decrease in EF50 value; moreover, higher doses of 0.32 and 1.0 mg/kg methamphetamine produced robust ICSS facilitation by both analyses (Fig. 2f: dose × frequency interaction F(27,135) = 6.05, p < 0.0001; Table 1).
Microdialysis
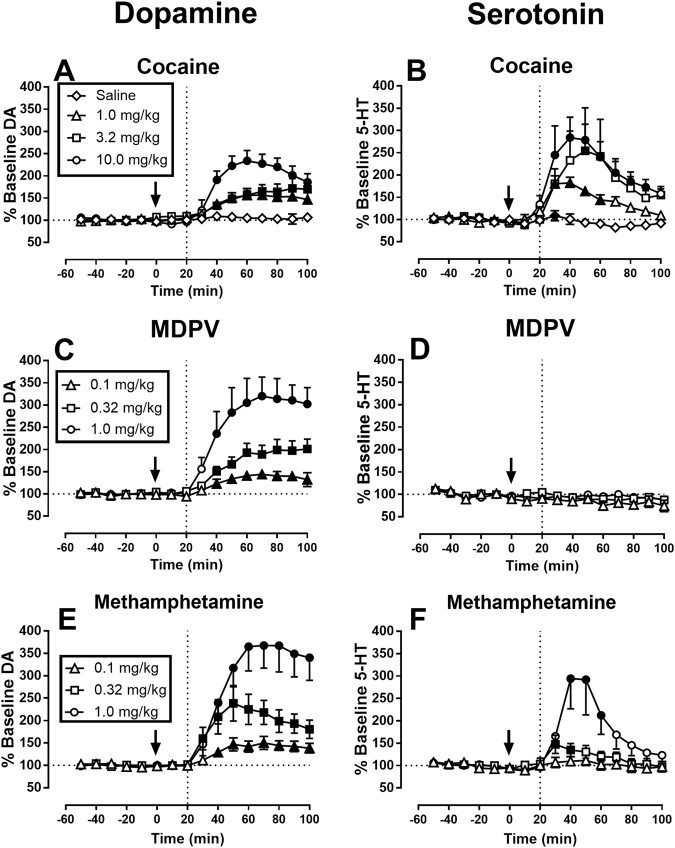
Effects of cocaine, MDPV, and methamphetamine on NAc DA and 5-HT in rats without minipumps
Supplemental Fig. 3 shows that all microdialysis probe placements were in the NAc. Figure 3 shows the effects of saline, cocaine, MDPV, and methamphetamine on NAc DA and 5-HT levels in rats without minipumps. Baseline DA and 5-HT levels were 1.46 ± 0.09 and 0.27 ± 0.01 pg/9 µl, respectively. After saline injection, one-way ANOVA indicated that DA levels did not significantly change (Fig. 3a), but 5-HT levels increased slightly at 30 min to 109% of baseline (Fig. 3b: F(9,45) = 3.97, p = 0.0009). Cocaine produced a dose- and time-dependent increase in both DA levels (Fig. 3a: 1.0 mg/kg: F(9,45) = 10.33, p < 0.0001; 3.2 mg/kg: F(9,45) = 9.28, p < 0.0001; 10.0 mg/kg: F(9,45) = 16.62, p < 0.0001) and 5-HT levels (Fig. 3b: 1.0 mg/kg: F(9,45) = 7.74, p < 0.0001; 3.2 mg/kg: F(9,45) = 2.32, p = 0.0305; 10.0 mg/kg: F(9,45) = 6.91, p < 0.0001). The 10 mg/kg cocaine dose increased DA levels to a maximum of 233% of baseline after 60 min, and 5-HT levels to a maximum of 284% of baseline after 40 min. MDPV produced a dose- and time-dependent increase in DA (Fig. 3c: 0.1 mg/kg: F(9,45) = 13.13, p < 0.0001; 0.32 mg/kg: F(9,45) = 24.64, p < 0.0001; 1.0 mg/kg: F(9,45) = 10.33, p < 0.0001) but not 5-HT (Fig. 3d). The 0.32 mg/kg MDPV dose used for subsequent studies increased DA levels to a maximum of 201% of baseline after 100 min. Methamphetamine produced a dose- and time-dependent increase in both DA (Fig. 3e: 0.1 mg/kg: F(9,45) = 9.27, p < 0.0001; 0.32 mg/kg: F(9,45) = 11.89, p < 0.0001; 1.0 mg/kg: F(9,45) = 10.26, p < 0.0001) and 5-HT (Fig. 3f: 0.1 mg/kg: n.s.; 0.32 mg/kg: F(9,45) = 4.44, p = 0.0003; 1.0 mg/kg: F(9,45) = 7.37, p < 0.0001). The 0.32 mg/kg methamphetamine dose used for subsequent studies increased DA levels to a maximum of 238% of baseline after 50 min, and 5-HT levels to a maximum of 147% of baseline after 30 min.
Fig. 3.
Effects of cocaine (a, b), MDPV (c, d), and methamphetamine (e, f) on NAc DA (a, c, e) and 5-HT (b, d, f) levels. Abscissae: time in minutes relative to test-drug injection. Vertical line at 20 min indicates earliest time of drug effect (see Methods). Ordinates: % of baseline DA or 5-HT. All points show mean ± SEM for N = 6 rats, and filled points indicate significantly different from the 10-min time point as determined by 1-way ANOVA followed by Dunnett’s post-hoc test (p < 0.05)
Effects of saline or amphetamine maintenance on baseline DA and 5-HT levels
Mean ± SEM DA levels in the NAc were increased by 7 days of 0.32 mg/kg/h amphetamine treatment (9.28 ± 0.75 pg/9 µl) as compared to 7 days of saline treatment (1.67 ± 0.52 pg/9 µl; t(17.26) = 7.68, p < 0.001). Conversely, mean ± SEM 5-HT levels in the NAc were similar in amphetamine-treated rats (0.38 ± 0.08 pg/9 µl) as compared to the saline-treated rats (0.28 ± 0.06 pg/9 µl; t(28.04) = 1.69, n.s.).
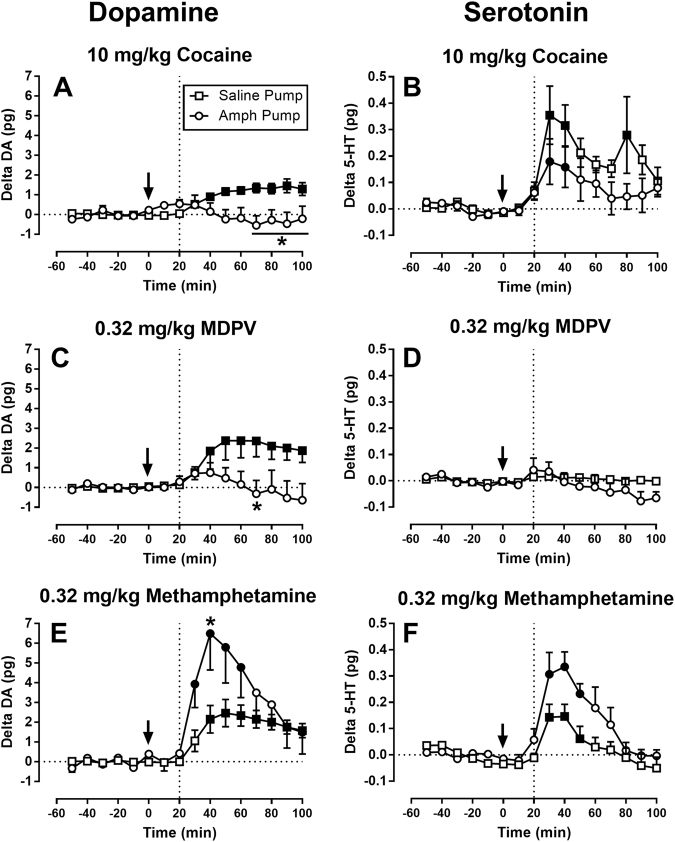
Effects of cocaine, MDPV, and methamphetamine during saline or amphetamine maintenance
Figure 4 shows effects of cocaine, MDPV, and methamphetamine on NAc DA and 5-HT levels after 7 treatment days with saline or 0.32 mg/kg/h amphetamine. Data are expressed as change from baseline (delta baseline) rather than % baseline due to the significant difference in DA baselines between the groups. However, for comparison, Supplemental Figs. 4 and 5 show the same data graphed as absolute concentrations or as % baseline, respectively.
Fig. 4.
Effects of cocaine (a, b), MDPV (c, d), and methamphetamine (e, f) on NAc DA (a, c, e) and 5-HT (b, d, f) levels after saline or amphetamine treatment. Abscissae: time in minutes relative to test-drug injection. Vertical line at 20 min indicates earliest time of drug effect (see Methods). Ordinates: change from baseline DA or 5-HT in pg/9 µl. Note that there is a different range used in DA panels (a, c, e) versus 5-HT panels (b, d, f). All points show mean ± SEM for N = 6 rats. Filled points indicate a significant difference from the 10-min time point within a given group as assessed by one-way ANOVA. Asterisks indicate a significant difference between saline and amphetamine-maintenance conditions within a given time point as assessed by two-way ANOVA
One-way ANOVA of data in Fig. 4 indicated significant increases in DA only during saline maintenance for cocaine (Fig. 4a: F(9,45) = 10.65, p < 0.0001) and MDPV (Fig. 4c: F(9,45) = 5.13, p < 0.0001), but during both saline and amphetamine maintenance for methamphetamine (Fig. 4e: F(9,45) = 10.31, p < 0.0001, F(9,45) = 5.88, p < 0.0001). 5-HT levels were increased during both saline and amphetamine maintenance by both cocaine (Fig. 4b: F(9,45) = 3.68, p = 0.0016, F(9,45) = 2.68, p = 0.0224) and methamphetamine (Fig. 4f: F(9,45) = 13.79, p < 0.0001, F(9,45) = 6.63, p < 0.0001), whereas MDPV did not alter 5-HT levels during either saline or amphetamine maintenance (Fig. 4d).
Two-way ANOVA of data in each panel of Fig. 4 indicated that amphetamine maintenance significantly attenuated the effects of cocaine on DA (Fig. 4a: treatment × time F(9,90) = 5.39, p < 0.0001), but it did not significantly alter cocaine effects on 5-HT (Fig. 4b: no main effect of treatment F(1,10) = 2.36, n.s. or treatment × time interaction F(9,90) = 1.22, n.s.). Amphetamine maintenance also attenuated effects of MDPV on DA (Fig. 4c: treatment × time F(9,90) = 4.10, p = 0.0002) without altering the lack of MDPV effect on 5-HT (Fig. 4d: no main effect of treatment: F(1,10) = 0.78, n.s.; significant treatment × time interaction F(9,90) = 2.40, p = 0.0174, but post-hoc test indicated no difference between saline and amphetamine maintenance at any time). In contrast, amphetamine maintenance enhanced the effects of methamphetamine on both DA (Fig. 4e: treatment × time F(9,90) = 2.76, p = 0.0067) and 5-HT (Fig. 4f: main effect of treatment F(1,10) = 7.09, p = 0.023).
[3H]WIN35,428 saturation binding
Mean ± SEM Bmax values in rats maintained on saline or 0.32 mg/kg/h amphetamine were 1.53 ± 0.12 pmol/mg and 1.33 ± 0.07 pmol/mg of membrane protein, respectively. Mean ± SEM KD values in rats maintained on saline or 0.32 mg/kg/h amphetamine were 14.7 ± 1.8 nM and 14.5 ± 1.3 nM, respectively. Neither Bmax nor KD values differed significantly between groups, indicating that amphetamine maintenance at this dose did not significantly alter striatal DAT levels or radioligand binding affinity.
Discussion
This study compared the effects of amphetamine maintenance on abuse-related behavioral and neurochemical effects of cocaine, MDPV, and methamphetamine in rats. There were three main findings. First, cocaine, MDPV, and methamphetamine all produced dose-dependent increases in ICSS facilitation and NAc DA levels before treatment, and cocaine and methamphetamine also increased NAc 5-HT. Second, amphetamine maintenance for 7 days produced sustained and significant increases in both basal ICSS and NAc DA levels without producing a significant change in basal 5-HT levels or in the density or binding affinity of striatal DAT. Finally, amphetamine maintenance blunted the effects of cocaine on both ICSS and NAc DA levels, it did not blunt methamphetamine-induced increases in ICSS or NAc DA, and it did not block the effects of either cocaine or methamphetamine on NAc 5-HT levels. For MDPV, amphetamine maintenance had little effect on ICSS facilitation, but did block the increases in NAc DA. Taken together, these results are consistent with the conclusion that amphetamine maintenance attenuates abuse-related behavioral effects of cocaine by reducing cocaine effects on NAc DA but conserving cocaine effects on NAc 5-HT. These results also suggest that amphetamine maintenance might be more effective as a pharmacotherapy for cocaine abuse than for MDPV or methamphetamine abuse.
Effects of cocaine, MDPV, and methamphetamine alone
Effects of cocaine, MDPV, and methamphetamine in the present study are consistent with previously published effects of these compounds on ICSS [4, 23, 27] and in microdialysis studies of NAc DA and 5-HT [31–33]. The effectiveness of these drugs to facilitate ICSS is consistent with other evidence for their abuse-related effects in preclinical studies (e.g., from drug self-administration procedures; [34–36]) and for their abuse by humans. Additionally, the relative potencies of these drugs to facilitate ICSS were similar to their relative potencies to increase NAc DA levels, supporting a role for NAc DA in mediating ICSS facilitation by these compounds. Both cocaine and methamphetamine also dose-dependently increased NAc 5-HT levels, and we and others have reported previously that either increases in 5-HT levels or activation of some 5-HT receptors (e.g., 5-HT2C receptors) can depress baseline ICSS and oppose DA-mediated ICSS facilitation and other stimulant effects [37–40]. However, this type of behavioral depression is usually associated with relatively large increases in NAc 5-HT levels (i.e., >400%; [5, 40]). The cocaine and methamphetamine doses tested here did not increase 5-HT levels to that degree, consistent with the absence of ICSS rate-decreasing effects.
Amphetamine maintenance facilitates ICSS and increases NAc DA
The sustained facilitation of ICSS during amphetamine maintenance replicated findings from a previous publication [23] and further demonstrated that this sustained behavioral effect was associated with a sustained elevation of NAc DA but not 5-HT. This profile of behavioral and neurochemical effects during 7-day amphetamine maintenance is consistent with both acute amphetamine effectiveness to facilitate ICSS and increase NAc DA but not 5-HT [4, 5] and with in vitro evidence that amphetamine is selective as a substrate for DAT relative to SERT [8]. The similarity in acute and chronic amphetamine effects suggests that little, if any, tolerance developed to the ICSS-facilitating or NAc DA-increasing effects of amphetamine-maintenance doses used here.
Amphetamine maintenance blocks cocaine effects on ICSS and NAc DA
The effectiveness of 0.32 mg/kg/h amphetamine to blunt ICSS facilitation by 10 mg/kg cocaine replicated findings from a previous study [23] and expanded on this finding in three ways. First, the present study evaluated the effects of 0.32 mg/kg/h amphetamine maintenance on ICSS-facilitation produced by a broader range of cocaine doses, and ICSS facilitating effects of 1.0 and 3.2 mg/kg cocaine doses were eliminated. This suggests that 0.32 mg/kg/h amphetamine produced at least a 10-fold rightward shift in the cocaine dose–effect curve for ICSS facilitation. Second, the present study also evaluated the effects of a lower amphetamine maintenance dose (0.1 mg/kg/h) on cocaine-induced ICSS facilitation, and this dose was not effective to alter ICSS facilitation by cocaine. The overall potency of amphetamine maintenance to attenuate ICSS facilitation in this study was similar to amphetamine-maintenance potency to reduce cocaine self-administration in rats responding in various drug self-administration procedures, including progressive-ratio, long-access, and cocaine-vs.-food choice procedures [16, 41, 42]. These effects are also consistent with amphetamine-maintenance effectiveness to decrease cocaine self-administration in monkeys and humans and to decrease metrics of cocaine use in clinical trials [9, 43, 44].
In a third extension of previous results, the present study also found that attenuation of cocaine-induced ICSS facilitation by amphetamine maintenance was associated with a selective attenuation of cocaine-induced increases in NAc DA. This correspondence between behavioral and neurochemical results further supports a role for NAc DA in mediating abuse-related effects of cocaine in general and ICSS facilitation in particular. However, two caveats warrant mention. First, although 10 mg/kg cocaine failed to increase NAc DA during 0.32 mg/kg/h amphetamine maintenance, it still produced significant, though blunted, ICSS facilitation. This is consistent with other data to suggest that ICSS may be more sensitive than in vivo microdialysis to small changes in synaptic NAc DA sufficient to alter behavior but insufficient to increase DA levels at extrasynaptic sites sampled by the microdialysis probe [5]. The use of brain stimulation to activate mesolimbic DA neurons during ICSS (Negus and Miller, 2014) [22], but not during in vivo microdialysis, may contribute to higher sensitivity of ICSS by generating higher levels of mesolimbic DA neuronal activity during ICSS than microdialysis. Second, amphetamine maintenance did not block cocaine-induced increases in NAc 5-HT. This is consistent with the lack of amphetamine maintenance effects on baseline 5-HT. Moreover, the retained effectiveness of cocaine to increase NAc 5-HT may also have opposed any residual cocaine effects on NAc DA [40]. Overall, cocaine increased only 5-HT during amphetamine maintenance, and selective SERT inhibitors neither facilitate ICSS [45] nor maintain self-administration [46].
Amphetamine maintenance blocks MDPV effects on NAc DA but not ICSS
As with cocaine, amphetamine maintenance also blocked increases in NAc DA produced by the other DAT inhibitor MDPV; however, in contrast to results with cocaine, amphetamine maintenance had little effect on MDPV-induced ICSS facilitation. There are at least three possible reasons for this discrepancy. First, as noted above, ICSS may be more sensitive than microdialysis to small changes in synaptic NAc DA levels. Second, consistent with its high selectivity to inhibit DAT over SERT [47, 48], MDPV did not increase NAc 5-HT in the absence or presence of amphetamine maintenance. Consequently, in contrast to cocaine, any residual DA-mediated effects of MDPV during amphetamine maintenance would not be opposed by concurrent increases in 5-HT. Lastly, it is possible that MDPV might facilitate ICSS via some mechanism other than DAT inhibition. At present, no evidence for other plausible mechanisms exists; for example, MDPV also has moderately high affinity for NET, but NET inhibitors do not facilitate ICSS, and MDPV does not have affinity for targets such as D1 DA receptors that can mediate ICSS facilitation [45, 47, 49]. Nonetheless, this possibility warrants consideration. Amphetamine-maintenance effects on MDPV self-administration or abuse have not been evaluated, but the present results suggest that amphetamine maintenance would be less effective clinically to reduce MDPV use than cocaine use.
Amphetamine maintenance fails to block methamphetamine effects
The failure of amphetamine maintenance to block methamphetamine-induced ICSS facilitation agrees with its failure to block the increases in NAc DA in the present study and with its failure to block methamphetamine self-administration in preclinical studies or methamphetamine abuse in clinical trials [17, 18]. Indeed, the present microdialysis data indicate that amphetamine maintenance enhanced rather than reduced methamphetamine effects on NAc DA levels, and also enhanced methamphetamine effects on 5-HT levels. Thus, amphetamine maintenance differentially modulated the neurochemical effects of monoamine uptake inhibitors (cocaine, MDPV) and a monoamine releaser (methamphetamine) on NAc DA and 5-HT levels.
Amphetamine maintenance does not reduce DAT density
The mechanisms by which amphetamine maintenance selectively reduces cocaine but not methamphetamine effects on ICSS and NAc DA levels remain to be determined. Although amphetamine-type releasers can reduce DAT density under some circumstances [25], the amphetamine-maintenance regimen used here did not significantly reduce DAT density as measured with the DAT inhibitor ligand WIN35,428, suggesting that decreased cocaine effects cannot be attributed to decreased cocaine binding sites on DAT. An alternative possibility is that amphetamine maintenance produces a sustained inhibition of DAT function due to competition between amphetamine and DA for DAT and/or effectiveness of amphetamine maintenance to promote intracellular-facing conformations of DAT that cannot mediate the uptake of extracellular DA [26, 50]. This might attenuate NAc DA-increasing effects of uptake inhibitors like cocaine and MDPV because transporter function is already inhibited; however, it is currently unknown why transporter substrates like methamphetamine would retain their effectiveness to increase DA levels.
Electronic supplementary material
Acknowledgements
The research reported in this publication was supported by the National Institute on Drug Abuse of the National Institutes of Health under Award Number R01 DA033930 and T32 DA007027. The content is solely the responsibility of the author and does not necessarily represent the official views of the National Institutes of Health.
Author contributions
ARJ conducted all the experiments, the initial graphing and analysis of all the data, and drafted the text. MLB supervised the microdialysis studies, SSN supervised the ICSS studies, and DES supervised the receptor binding studies. MLB, SSN, and DES contributed to the experimental design and generation of final data analysis, graphs, and text. All authors approved the final text.
Competing interests
The authors declare no competing interests.
Footnotes
Electronic supplementary material
The online version of this article (10.1038/s41386-018-0071-3) contains supplementary material, which is available to authorized users.
References
- 1.SAMSHA, 2017: Substance Abuse and Mental Health Services Administration. (2017). Key substance use and mental health indicators in the United States: Results from the 2016 National Survey on Drug Use and Health (HHS Publication No. SMA 17-5044, NSDUH Series H-52). Rockville, MD: Center for Behavioral Health Statistics and Quality, Substance Abuse and Mental Health Services Administration. Retrieved from https://www.samhsa.gov/data/
- 2.Karila L, Lafaye G, Scocard A, Cottencin O, Benyamina A. MDPV and α-PVP use in humans: the twisted sisters. Neuropharmacology. 2017:1–8. 10.1016/j.neuropharm.2017.10.007. [DOI] [PubMed]
- 3.Rothman RB, Baumann MH. Balance between dopamine and serotonin release modulates behavioral effects of amphetamine-type drugs. Ann N Y Acad Sci. 2006;1074:245–60. doi: 10.1196/annals.1369.064. [DOI] [PubMed] [Google Scholar]
- 4.Bauer CT, Banks ML, Blough BE, Negus SS. Use of intracranial self-stimulation to evaluate abuse-related and abuse-limiting effects of monoamine releasers in rats. Br J Pharmacol. 2013;168:850–62. doi: 10.1111/j.1476-5381.2012.02214.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Suyama JA, Sakloth F, Kolanos R, Glennon RA, Lazenka MF, Negus SS, et al. Abuse-related neurochemical effects of para-substituted methcathinone analogs in rats: microdialysis studies of nucleus accumbens dopamine and serotonin. J Pharmacol Exp Ther. 2016;356:182–90. doi: 10.1124/jpet.115.229559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wee S, Anderson KG, Baumann MH, Rothman RB, Blough BE, Woolverton WL. Relationship between the serotonergic activity and reinforcing effects of a series of amphetamine analogs. J Pharmacol Exp Ther. 2005;313:848–54. doi: 10.1124/jpet.104.080101. [DOI] [PubMed] [Google Scholar]
- 7.De Felice LJ, Glennon RA, Negus SS. Synthetic cathinones: chemical phylogeny, physiology, and neuropharmacology. Life Sci. 2014;97:20–26. doi: 10.1016/j.lfs.2013.10.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rothman RB, Baumann MH, Dersch CM, Romero DV, Rice KC, Carroll FIVY, et al. Amphetamine-type central nervous norepinephrine more potently than they release dopamine and serotonin. Synapse. 2001;39:32–41. doi: 10.1002/1098-2396(20010101)39:1<32::AID-SYN5>3.0.CO;2-3. [DOI] [PubMed] [Google Scholar]
- 9.Castelles X, Cunill R, Pérez-Mañá C, Vidal X, Capellà D. (2016). Psychostimulant drugs for cocaine dependence. Cochrane Database Syst Rev. 2016;27:CD007380. [DOI] [PMC free article] [PubMed]
- 10.Grabowski J, Rhoades H, Schmitz J, Stotts A, Daruzska LA, Creson D, et al. Dextroamphetamine for cocaine-dependence treatment: a double-blind randomized clinical trial. J Clin Psychopharmacol. 2001;21:522–6. doi: 10.1097/00004714-200110000-00010. [DOI] [PubMed] [Google Scholar]
- 11.Greenwald MK, Lundahl LH, Steinmiller CL. Sustained release d-amphetamine reduces cocaine but not “speedball”-seeking in buprenorphine-maintained volunteers: a test of dual-agonist pharmacotherapy for cocaine/heroin polydrug abusers. Neuropsychopharmacology. 2010;35:2624–37. doi: 10.1038/npp.2010.175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Levin FR, Mariani JJ, Specker S, Mooney M, Mahony A, Brooks DJ, et al. Extended-release mixed amphetamine salts vs. placebo for comorbid adult attention-deficit/hyperactivity disorder and cocaine use disorder. JAMA Psychiatry. 2015;72:593–602. doi: 10.1001/jamapsychiatry.2015.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schmitz JM, Rathnayaka N, Green CE, Moeller FG, Dougherty AE, Grabowski J. Combination of modafinil and d-amphetamine for the treatment of cocaine dependence: a preliminary investigation. Front Psychiatry. 2012;3:1–6. doi: 10.3389/fpsyt.2012.00077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Banks ML, Pharm D, Hutsell BA, Blough BE, Poklis JL, Stevens Negus S. Preclinical assessment of lisdexamfetamine as an agonist medication candidate for cocaine addiction: effects in rhesus monkeys trained to discriminate cocaine or to self-Administer cocaine in a cocaine versus food choice procedure. Int J Neuropsychopharmacol. 2015;18:1–10. doi: 10.1093/ijnp/pyv009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Rush CR, Stoops WW, Sevak RJ, Hays LR. Cocaine choice in humans during D-amphetamine maintenance. J Clin Psychopharmacol. 2010;30:152–9. doi: 10.1097/JCP.0b013e3181d21967. [DOI] [PubMed] [Google Scholar]
- 16.Thomsen M, Barrett AC, Negus SS, Caine SB. Cocaine versus food choice procedure in rats: environmental manipulations and effects of amphetamine. J Exp Anal Behav. 2013;99:211–33. doi: 10.1002/jeab.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Galloway GP, Buscemi R, Coyle JR, Flower K, Siegrist JD, Fiske LA, et al. A randomized, placebo-controlled trial of sustained-release dextroamphetamine for treatment of methamphetamine addiction. Clin Pharmacol Ther. 2011;89:276–82. doi: 10.1038/clpt.2010.307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Schwienteck KL, Banks ML. Effects of 7-day continuous d-amphetamine, methylphenidate, and cocaine treatment on choice between methamphetamine and food in male rhesus monkeys. Drug Alcohol Depend. 2015;155:16–23. doi: 10.1016/j.drugalcdep.2015.08.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Grabowski J, Rhoades H, Stotts A, Cowan K, Kopecky C, Dougherty A, et al. Agonist-like or antagonist-like treatment for cocaine dependence with methadone for heroin dependence: two double-blind randomized clinical trials. Neuropsychopharmacology. 2004;29:969–81. doi: 10.1038/sj.npp.1300392. [DOI] [PubMed] [Google Scholar]
- 20.Rothman RB, Blough BE, Baumann MH. Appetite suppressants as agonist substitution therapies for stimulant dependence. Ann N Y Acad Sci. 2002;965:109–26. doi: 10.1111/j.1749-6632.2002.tb04155.x. [DOI] [PubMed] [Google Scholar]
- 21.Tomasiewicz HC, Todtenkopf MS, Chartoff EH, Cohen BM, Carlezon WA., Jr The kappa-opioid agonist U69,593 blocks cocaine-induced enhancement of brain stimulation reward. Biol Psychiatry. 2008;64:982–8. doi: 10.1016/j.biopsych.2008.05.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Negus SS, Miller LL. Intracranial self-stimulation to evaluate abuse potential of drugs. Pharmacol Rev. 2014;66:869–917. doi: 10.1124/pr.112.007419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bauer CT, Banks ML, Negus SS. The effect of chronic amphetamine treatment on cocaine-induced facilitation of intracranial self-stimulation in rats. Psychopharmacology (Berl) 2014;231:2461–70. doi: 10.1007/s00213-013-3405-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Negus SS, Banks ML. Decoding the structure of abuse potential for new psychoactive substances: structure--activity relationships for abuse-related effects of 4-substituted methcathinone analogs. Curr Top Behav Neurosci. 2017;32:119–31. doi: 10.1007/7854_2016_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Boudanova E, Navaroli DM, Melikian HE. Amphetamine-induced decreases in dopamine transporter surface expression are protein kinase C-independent. Neuropharmacology. 2008;54:605–12. doi: 10.1016/j.neuropharm.2007.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.German CL, Baladi MG, McFadden LM, Hanson GR, Fleckenstein AE. Regulation of the dopamine and vesicular monoamine transporters: pharmacological targets and implications for disease. Pharmacol Rev. 2015;67:1005–24. doi: 10.1124/pr.114.010397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bonano JS, Glennon RA, De FeliceLJ, Banks ML, Negus SS. Abuse-related and abuse-limiting effects of methcathinone and the synthetic “bath salts” cathinone analogs methylenedioxypyrovalerone (MDPV), methylone and mephedrone on intracranial self-stimulation in rats. Psychopharmacology (Berl) 2014;231:199–207. doi: 10.1007/s00213-013-3223-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pereira Do Carmo G, Stevenson GW, Carlezon WA, Negus SS. Effects of pain- and analgesia-related manipulations on intracranial self-stimulation in rats: further studies on pain-depressed behavior. Pain. 2009;144:170–7. doi: 10.1016/j.pain.2009.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bonano JS, Runyon SP, Hassler C, Glennon RA, Negus SS. Effects of the neuropeptide S receptor antagonist RTI-118 on abuse-related facilitation of intracranial self-stimulation produced by cocaine and methylenedioxypyrovalerone (MDPV) in rats. Eur J Pharmacol. 2014;743:98–105. doi: 10.1016/j.ejphar.2014.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kuhar MJ, McGirr KM, Hunter RG, Lambert PD, Garrett BE, Carroll FI. Studies of selected phenyltropanes at monoamine transporters. Drug Alcohol Depend. 1999;56:9–15. doi: 10.1016/S0376-8716(99)00005-8. [DOI] [PubMed] [Google Scholar]
- 31.Andrews CM, Lucki I. Effects of cocaine on extracellular dopamine and serotonin levels in the nucleus accumbens. Psychopharmacology (Berl) 2001;155:221–9. doi: 10.1007/s002130100704. [DOI] [PubMed] [Google Scholar]
- 32.Schindler CW, Thorndike EB, Goldberg SR, Lehner KR, Cozzi NV, Brandt SD, et al. Reinforcing and neurochemical effects of the “bath salts” constituents 3,4-methylenedioxypyrovalerone (MDPV) and 3,4-methylenedioxy-N-methylcathinone (methylone) in male rats. Psychopharmacology (Berl) 2016;233:1981–90. doi: 10.1007/s00213-015-4057-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Baumann MH, Ayestas MA, Partilla JS, Sink JR, Shulgin AT, Daley PF, et al. The designer methcathinone analogs, mephedrone and methylone, are substrates for monoamine transporters in brain tissue. Neuropsychopharmacology. 2012;37:1192–203. doi: 10.1038/npp.2011.304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Aarde SM, Huang PK, Creehan KM, Dickerson TJ, Taffe MA. The novel recreational drug 3,4-methylenedioxypyrovalerone (MDPV) is a potent psychomotor stimulant: self-administration and locomotor activity in rats. Neuropharmacology. 2013;71:130–40. doi: 10.1016/j.neuropharm.2013.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gannon BM, Galindo KI, Rice KC, Collins GT. Individual differences in the relative reinforcing effects of 3,4-methylenedioxypyrovalerone under fixed and progressive ratio schedules of reinforcement in rats. J Pharmacol Exp Ther. 2017;361:181–9. doi: 10.1124/jpet.116.239376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Watterson LR, Kufahl PR, Nemirovsky NE, Sewalia K, Grabenauer M, Thomas BF, et al. Potent rewarding and reinforcing effects of the synthetic cathinone 3,4-methylenedioxypyrovalerone (MDPV) Addict Biol. 2014;19:165–74. doi: 10.1111/j.1369-1600.2012.00474.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lazenka MF, Blough BE, Negus SS. Preclinical Abuse potential assessment of flibanserin: effects on intracranial self-stimulation in female and male rats. J Sex Med. 2016;13:338–49. doi: 10.1016/j.jsxm.2015.12.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Negus SS, Banks ML (2017). Decoding the structure of abuse potential for new psychoactive substances: structure--activity relationships for abuse-related effects of 4-substituted methcathinone analogs. Curr Top Behav Neurosci. 2017;32:119–31. http://doi.org/10.1007/7854_2016_18. [DOI] [PMC free article] [PubMed]
- 39.Bauer CT, Banks ML, Blough BE, Negus SS. Role of 5-HT2C receptors in effects of monoamine releasers on intracranial self-stimulation in rats. Psychopharmacology (Berl) 2015;232:3249–58. doi: 10.1007/s00213-015-3982-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Baumann MH, Clark RD, Woolverton WL, Wee S, Blough BE, Rothman RB. In vivo effects of amphetamine analogs reveal evidence for serotonergic inhibition of mesolimbic dopamine transmission in the rat. J Pharmacol Exp Ther. 2011;337:218–25. doi: 10.1124/jpet.110.176271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chiodo KA, Läck CM, Roberts DCS. Cocaine self-administration reinforced on a progressive ratio schedule decreases with continuous D-amphetamine treatment in rats. Psychopharmacology (Berl) 2008;200:465–73. doi: 10.1007/s00213-008-1222-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Siciliano CA, Saha K, Calipari ES, Fordahl SC, Chen R, Khoshbouei H, Jones SR. Amphetamine reverses escalated cocaine intake via resortation of dopamine transporter conformation. J Neurosci. 2018;38:484–97. doi: 10.1523/JNEUROSCI.2604-17.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Grabowski J, Shearer J, Merrill J, Negus SS. Agonist-like, replacement pharmacotherapy for stimulant abuse and dependence. Addict Behav. 2004;29:1439–64. doi: 10.1016/j.addbeh.2004.06.018. [DOI] [PubMed] [Google Scholar]
- 44.Negus SS, Henningfield J. Agonist medications for the treatment of cocaine use disorder. Neuropsychopharmacology. 2015;40:1815–25. doi: 10.1038/npp.2014.322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rosenberg MB, Carroll FI, Negus SS. Effects of monoamine reuptake inhibitors in assays of acute pain-stimulated and pain-depressed behavior in rats. J Pain. 2013;14:246–59. doi: 10.1016/j.jpain.2012.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Roberts DCS, Phelan R, Hodges LM, Hodges MM, Bennett B, Childers S, et al. Self-administration of cocaine analogs by rats. Psychopharmacology (Berl) 1999;144:389–97. doi: 10.1007/s002130051022. [DOI] [PubMed] [Google Scholar]
- 47.Simmler LD, Buser TA, Donzelli M, Schramm Y, Dieu LH, Huwyler J, et al. Pharmacological characterization of designer cathinones in vitro. Br J Pharmacol. 2013;168:458–70. doi: 10.1111/j.1476-5381.2012.02145.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Baumann MH, Partilla JS, Lehner KR, Thorndike EB, Hoffman AF, Holy M, et al. Powerful cocaine-like actions of 3,4-methylenedioxypyrovalerone (MDPV), a principal constituent of psychoactive “ bath salts” products. Neuropsychopharmacology. 2013;38:552–62. doi: 10.1038/npp.2012.204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lazenka MF, Legakis LP, Stevens Negus S. Opposing effects of dopamine D1-and D2-Like agonists on intracranial self-stimulation in male rats. Exp Clin Psychopharmacol. 2016;24:193–205. doi: 10.1037/pha0000067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Jones SR, Joseph JD, Barak LS, Caron MG, Wightman RM. Dopamine neuronal transport kinetics and effects of amphetamine. J Neurochem. 1999;73:2406–14. doi: 10.1046/j.1471-4159.1999.0732406.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.