Abstract
Plantar warts account for 30% of all cutaneous warts. These lesions could be very painful, especially if the lesion is located over pressure sites such as the metatarsal head. Plantar wart treatment remains a challenging therapeutic problem. A 67-year-old immunocompetent nonsmoking man presented with a large mosaic plantar wart on his right foot. The lesion had been present for 5 years. Several cryotherapy sessions (a total of 6 procedures) had been performed with no success. The lesion was therefore treated with a 5-fluorouracil (5-FU) regimen and then with a topical combination of 5-FU and salicylic acid, but also these approaches failed. At the initial visit, a large (16 cm2) mosaic wart lesion was present. Treatment with topical Polyphenon E, 10%, twice daily was prescribed and started. After 3 months of treatment, the lesion completely disappeared. Interestingly, no curettage or mechanical pickling of the hyperkeratotic parts of the lesion were performed before the start of the treatment. Local tolerability was evaluated as very good by the patient.
Keywords: Warts, Sinecatechins, Case report, Green tea extract
Introduction
Plantar warts are a common viral skin disease caused by human papillomavirus (HPV) infection [1]. HPV has a specific tropism for squamous stratified epithelium [2] and this infection leads to proliferation of keratinocytes. HPV infection is characterized by a relevant immune evasion mechanism that inhibits and delays the host immune response to the virus [3]. HPV genotypes types 2, 4, 27, 57, and 60 are responsible for common and plantar warts and myrmecia [4]. On the other hand, genotypes 6 and 11 are the most common HPV involved in genital wart [5] development. Curettage, topical salicylic acid, and cryotherapy are often used in plantar warts [6]. Liquid nitrogen is the most frequently used treatment for this type of warts; however, this procedure is in general very painful [7]. Excision is not a good option owing to frequent recurrences or scarring [8]. In case of therapy-resistant lesions, laser and photodynamic therapy approaches could be used [9] but these methods are time- and cost-consuming. Therefore, in case of plantar warts, both first- and second-line treatments are in general still unsatisfactory due to their low efficacy, low local tolerability, and high recurrence rate [10]. Topical green tea extract containing catechins (Polyphenon E) is indicated as a first-line treatment for genital warts (condylomata acuminata) [11]. In subjects with genital warts, Polyphenon E, applied 3 times a day and up to 16 weeks, induced a complete clearance rate in more than 50% of patients [12]. In addition, the recurrence rate at 3 months after the conclusion of the therapy is as low as < 6% [9]. Polyphenon E is a quantified extract of green tea leaves (Camellia sinensis) where the main active substance is epigallocatechin gallate (EGCG), a polyphenol substance which represents the major component of the catechins fraction [13]. EGCG has potent anti-inflammatory, antioxidant, and proapoptotic actions. These mechanisms could explain its antiviral properties [14]. Recent data have shown that EGCG is able to modulate the cellular genes involved in Toll-like receptor production and in the apoptosis mechanisms [15]. The product has shown a good safety and tolerability profile. Local skin reactions like erythema and edema, in general mild to moderate, have been reported in most treated subjects [16]. Polyphenon E has shown to be an effective and very well-tolerated treatment for common [17] and plane [18] warts. Data regarding the efficacy of Polyphenon E in plantar warts are lacking so far. We report the clinical efficacy and the tolerability of Polyphenon E 10% ointment (VeregenTM, Medigene, Germany) in a subject with a “difficult-to-treat”, treatment-resistant plantar wart.
Case Description
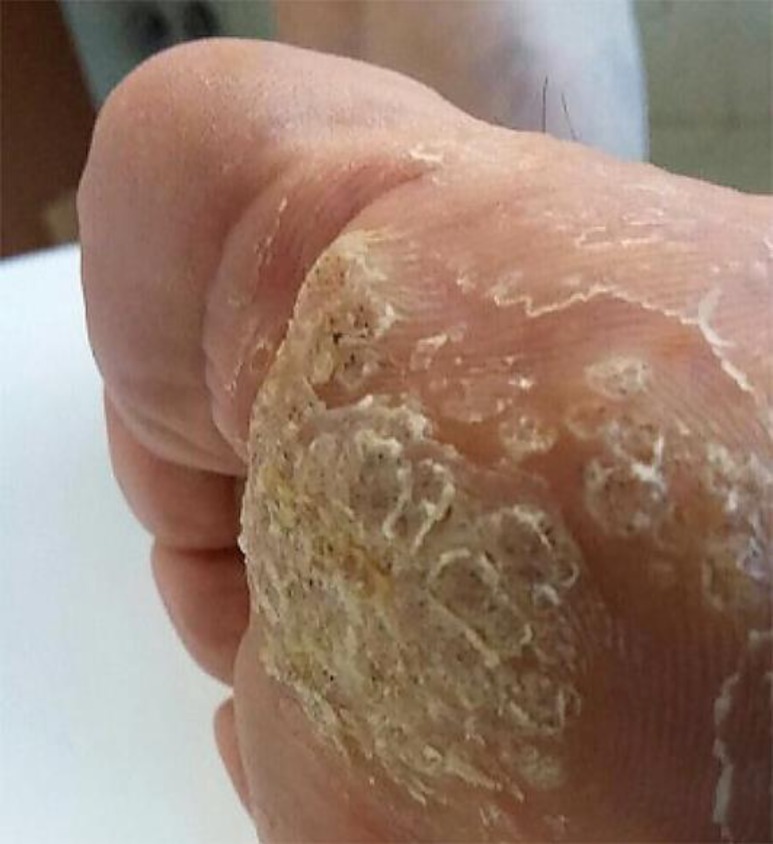
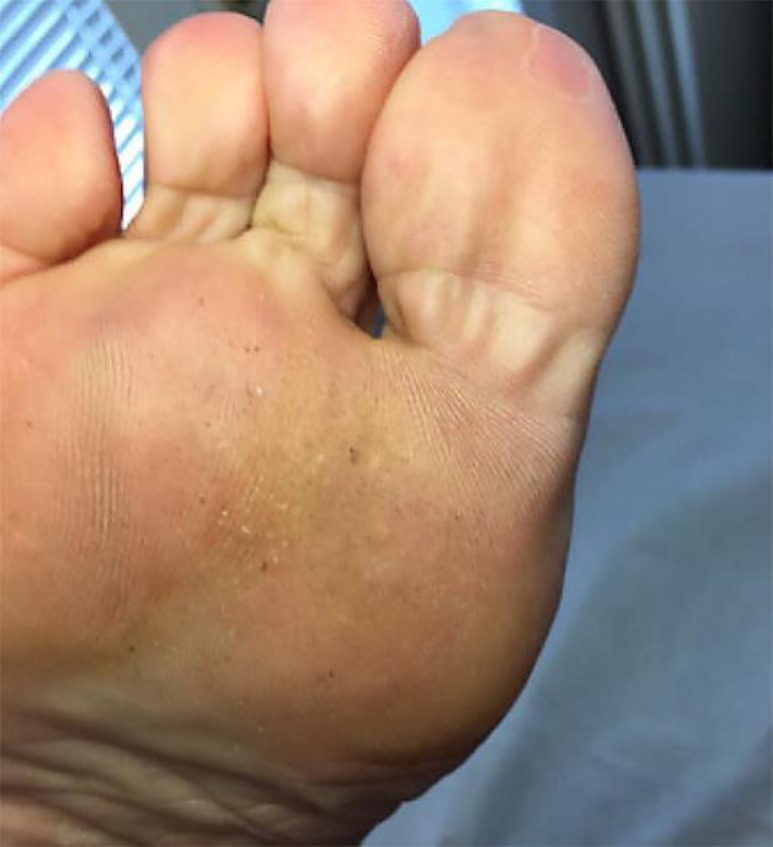
A 67-year-old immunocompetent nonsmoking man presented with a large mosaic plantar wart on his right foot. The lesion had been present for 5 years. Several cryotherapy sessions (a total of 6 procedures) had been performed with no success. The lesion was therefore treated with a 5-fluorouracil regimen and then with a topical combination of 5-fluorouracil and salicylic acid, but also these approaches failed. At the initial visit, a large (16 cm2) mosaic wart lesion was present (Fig. 1). Treatment with topical Polyphenon E 10% twice daily was prescribed and started. After 3 months of treatment, the lesion completely disappeared (Fig. 2). Interestingly, no curettage or mechanical pickling of the hyperkeratotic parts of the lesion were performed before the start of the treatment. Local tolerability was evaluated as very good by the patient.
Fig. 1.
Plantar mosaic wart lesion on the right foot before treatment with green tea extract.
Fig. 2.
Plantar region after a 3-month treatment with green tea extract.
Discussion
Plantar warts account for 30% of all cutaneous warts [19]. Large lesions are in general plaques with rough hyperkeratotic surface with some brown-black dots representing thrombosed capillaries [20]. These lesions could be very painful, especially if the lesion is located over pressure sites such as the metatarsal head [21]. Plantar wart treatment remains a challenging therapeutic problem [22]. Some procedures could be very painful with a long downtime posttreatment period [23]. Polyphenon E ointment is considered an effective, well-tolerated, self-applicable topical treatment of genital warts [24]. Clinical efficacy and tolerability was evaluated in randomized vehicle-controlled phase III trials in more than 1,000 patients [25]. Complete clearance rate is observed in 51% of the treated subjects. Mean treatment duration for complete clearance was between 14 and 16 weeks [26]. Polyphenon E has an antiviral action trough pathway triggering cell growth arrest and increasing proapoptotic molecules like p53 and p16 [27]. In addition, this compound interferes with cellular activity of E6 and E7, two HPV virus proteins [28]. E6 and E7are involved in the process of apoptosis inhibition and in the immune evasion mechanisms [29]. Therefore, Polyphenon E could express anti-HPV action through multiple mechanisms. The gene modulation expressed by Polyphenon could be of help in fighting therapy-resistant HPV lesions. Polyphenon E has shown to be an effective treatment of plane warts of the face [30]. An ideal wart therapy should be easy to apply (preferably home-based), painless, able to resolve all the treated lesions, not causing scarring, and with no or very short downtime period [23]. In this case report, we have shown a high clinical efficacy of Polyphenon E in the treatment of a therapy-resistant large mosaic plantar wart lesion. It is remarkable that complete clearance of the lesion was achieved with a 12-week treatment without any pretreatment curettage or pickling procedures and without a downtime period and without scar formation. This case report suggests that topical Polyphenon E 10% could be an effective therapeutic strategy in subjects with “difficult-to-treat” plantar wart lesions. Specific controlled trials in this specific population are warranted.
Statement of Ethics
The authors have no ethical conflicts to disclose.
Disclosure Statement
M.M. is an employee of Cantabria Labs Difa Cooper; Caronno P., Italy.
References
- 1.Jenson AB, Sommer S, Payling-Wright C, Pass F, Link CC, Jr, Lancaster WD, Human papillomavirus Frequency and distribution in plantar and common warts. Lab Invest. 1982 Nov;47((5)):491–7. [PubMed] [Google Scholar]
- 2.Majewski S, Jablonska S. Human papillomavirus-associated tumors of the skin and mucosa. J Am Acad Dermatol. 1997 May;36((5 Pt 1)):659–85. doi: 10.1016/s0190-9622(97)80315-5. [DOI] [PubMed] [Google Scholar]
- 3.Tindle RW. Immune evasion in human papillomavirus-associated cervical cancer. Nat Rev Cancer. 2002 Jan;2((1)):59–65. doi: 10.1038/nrc700. [DOI] [PubMed] [Google Scholar]
- 4.Pfister H, zur Hausen H. Seroepidemiological studies of human papilloma virus (HPV-1) infections. Int J Cancer. 1978 Feb;21((2)):161–5. doi: 10.1002/ijc.2910210206. [DOI] [PubMed] [Google Scholar]
- 5.Gissmann L, Wolnik L, Ikenberg H, Koldovsky U, Schnürch HG, zur Hausen H. Human papillomavirus types 6 and 11 DNA sequences in genital and laryngeal papillomas and in some cervical cancers. Proc Natl Acad Sci USA. 1983 Jan;80((2)):560–3. doi: 10.1073/pnas.80.2.560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sterling JC, Handfield-Jones S, Hudson PM. British Association of Dermatologists Guidelines for the management of cutaneous warts. Br J Dermatol. 2001 Jan;144((1)):4–11. doi: 10.1046/j.1365-2133.2001.04066.x. [DOI] [PubMed] [Google Scholar]
- 7.Gibbs S, et al. Local treatments for cutaneous warts systematic review. BMJ. 2002;325:461. [PMC free article] [PubMed] [Google Scholar]
- 8.Sterling JC, Handfield-Jones S, Hudson PM, British Association of Dermatologists Guidelines for the management of cutaneous warts. Br J Dermatol. 2001 Jan;144((1)):4–11. doi: 10.1046/j.1365-2133.2001.04066.x. [DOI] [PubMed] [Google Scholar]
- 9.Fuchs SM, et al. Photodynamic therapy (PDT) and waterfiltered infrared A (wIRA) in patients with recalcitrant common hand and foot warts. Ger Med Sci. 2004;2 Doc08. [PMC free article] [PubMed] [Google Scholar]
- 10.Kodner CM, Nasraty S. Management of genital warts. Am Fam Physician. 2004 Dec;70((12)):2335–42. [PubMed] [Google Scholar]
- 11.Workowski KA, Berman SM. Centers for Disease Control and Prevention sexually transmitted disease treatment guidelines. Clin Infect Dis. 2011;53(suppl 3):S59–S63. doi: 10.1093/cid/cir694. [DOI] [PubMed] [Google Scholar]
- 12.Stockfleth E, Beti H, Orasan R, Grigorian F, Mescheder A, Tawfik H, et al. Topical Polyphenon E in the treatment of external genital and perianal warts a randomized controlled trial. Br J Dermatol. 2008 Jun;158((6)):1329–38. doi: 10.1111/j.1365-2133.2008.08520.x. [DOI] [PubMed] [Google Scholar]
- 13.Waltner-Law ME, Wang XL, Law BK, Hall RK, Nawano M, Granner DK. Epigallocatechin gallate. a constituent of green tea represses hepatic glucose production. J Biol Chem. 2002 Sep;277((38)):34933–40. doi: 10.1074/jbc.M204672200. [DOI] [PubMed] [Google Scholar]
- 14.Valcic S, Muders A, Jacobsen NE, Liebler DC, Timmermann BN, Antioxidant chemistry of green tea catechins Identification of products of the reaction of (-)-epigallocatechin gallate with peroxyl radicals. Chem Res Toxicol. 1999 Apr;12((4)):382–6. doi: 10.1021/tx990003t. [DOI] [PubMed] [Google Scholar]
- 15.Masuda M, Suzui M, Weinstein IB. Effects of epigallocatechin-3-gallate on growth epidermal growth factor receptor signaling pathways gene expression and chemosensitivity in human head and neck squamous cell carcinoma cell lines. Clin Cancer Res. 2001 Dec;7((12)):4220–9. [PubMed] [Google Scholar]
- 16.Tatti S, Swinehart JM, Thielert C, Tawfik H, Mescheder A, Beutner KR. Sinecatechins a defined green tea extract in the treatment of external anogenital warts a randomized controlled trial. Obstet Gynecol. 2008 Jun;111((6)):1371–9. doi: 10.1097/AOG.0b013e3181719b60. [DOI] [PubMed] [Google Scholar]
- 17.Gerlero P, Hernández-Martín Á. Treatment of Warts in Children an Update [English Edition] Actas Dermosifiliogr. 2016 Sep;107((7)):551–8. doi: 10.1016/j.ad.2016.04.010. [DOI] [PubMed] [Google Scholar]
- 18.Clouth A, Schöfer H. Treatment of recalcitrant facial verrucae vulgares with sinecatechins (greentea catechins) ointment. J Eur Acad Dermatol Venereol. 2015 Jan;29((1)):178–9. doi: 10.1111/jdv.12341. [DOI] [PubMed] [Google Scholar]
- 19.Kilkenny M, Marks R. The descriptive epidemiology of warts in the community. Australas J Dermatol. 1996 May;37((2)):80–6. doi: 10.1111/j.1440-0960.1996.tb01010.x. [DOI] [PubMed] [Google Scholar]
- 20.Jabłońska S, Majewski S, Obalek S, Orth G. Cutaneous warts. Clin Dermatol. 1997 May-Jun;15((3)):309–19. doi: 10.1016/s0738-081x(96)00170-8. [DOI] [PubMed] [Google Scholar]
- 21.Bacelieri R, Johnson SM. Cutaneous warts an evidence-based approach to therapy. Am Fam Physician. 2005 Aug;72((4)):647–52. [PubMed] [Google Scholar]
- 22.Stender IM, Na R, Fogh H, Gluud C, Wulf HC. Photodynamic therapy with 5-aminolaevulinic acid or placebo for recalcitrant foot and hand warts randomised double-blind trial. Lancet. 2000 Mar;355((9208)):963–6. doi: 10.1016/S0140-6736(00)90013-8. [DOI] [PubMed] [Google Scholar]
- 23.Vlahovic TC, Khan MT, VLAHOVIC Tracey C., KHAN M. Tariq. The Human Papillomavirus and Its Role in Plantar Warts. Clin Podiatr Med Surg. 2016;33((3)):337–53. doi: 10.1016/j.cpm.2016.02.003. [DOI] [PubMed] [Google Scholar]
- 24.Stockfleth E, Meyer T. The use of sinecatechins (polyphenon E) ointment for treatment of external genital warts. Expert Opin Biol Ther. 2012 Jun;12((6)):783–93. doi: 10.1517/14712598.2012.676036. [DOI] [PubMed] [Google Scholar]
- 25.Gupta AK, Daigle D. Sinecatechins 10% ointment a green tea extract for the treatment of external genital warts. Skin Therapy Lett. 2015 Jan-Feb;20((1)):6–8. [PubMed] [Google Scholar]
- 26.Stockfleth E, Meyer T. The use of sinecatechins (polyphenon E) ointment for treatment of external genital warts. Expert Opin Biol Ther. 2012 Jun;12((6)):783–93. doi: 10.1517/14712598.2012.676036. [DOI] [PubMed] [Google Scholar]
- 27.Thakur VS, Ruhul Amin AR, Paul RK, Gupta K, Hastak K, Agarwal MK, et al. p53-Dependent p21-mediated growth arrest pre-empts and protects HCT116 cells from PUMA-mediated apoptosis induced by EGCG. Cancer Lett. 2010 Oct;296((2)):225–32. doi: 10.1016/j.canlet.2010.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Qiao Y, Cao J, Xie L, Shi X. Cell growth inhibition and gene expression regulation by (-)-epigallocatechin-3-gallate in human cervical cancer cells. Arch Pharm Res. 2009 Sep;32((9)):1309–15. doi: 10.1007/s12272-009-1917-3. [DOI] [PubMed] [Google Scholar]
- 29.Park JS, Kim EJ, Kwon HJ, Hwang ES, Namkoong SE, Um SJ, Inactivation of interferon regulatory factor-1 tumor suppressor protein by HPV E7 oncoprotein Implication for the E7-mediated immune evasion mechanism in cervical carcinogenesis. J Biol Chem. 2000 Mar;275((10)):6764–9. doi: 10.1074/jbc.275.10.6764. [DOI] [PubMed] [Google Scholar]
- 30.Gerlero P, Hernández-Martín Á. Treatment of Warts in Children an Update [English Edition] Actas Dermosifiliogr. 2016 Sep;107((7)):551–8. doi: 10.1016/j.ad.2016.04.010. [DOI] [PubMed] [Google Scholar]