Abstract
A perimenopausal woman presented with palpitations, hirsutism, and inability to lose weight. Laboratory tests revealed an unusual endocrine hormonal profile including pituitary hormones (TSH, ACTH, and prolactin) below reference intervals and gonadal (testosterone) and adrenal (cortisol) hormones above reference intervals. Ultimately, after a comprehensive workup including a scheduled surgical procedure, abnormal laboratories were determined due to biotin interference. Biotin (vitamin B7) is a water-soluble vitamin and essential cofactor for the metabolism of fatty acids, glucose, and amino acids. The recommended daily intake of biotin for adults is 30 µg/d. Many over-the-counter products, particularly those marketed for hair, skin, and nail growth, contain biotin 100-fold of recommended daily intake. This case is unique due to the abnormalities observed not only in the well-described TSH “sandwich” immunoassay, but also in tests for gonadal steroids, adrenal, and pituitary hormones. Falsely high as well as falsely low results can be ascribed to biotin. Competitive immunoassays (Fig. 1A)— in this case, tests used initially for serum cortisol and testosterone— can demonstrate falsely high results. Interference falsely lowers the immunometric “sandwich” immunoassay (Fig. 1B)—in this case, TSH. Biotin effect on our patient’s endocrine testing led to decidedly abnormal findings, unnecessary medical referrals and diagnostic studies, and comprehensible psychological distress. Interference with one immunoassay, TSH, persisted a full 2 weeks after discontinuation of biotin; indeed, some tests demonstrate sensitivity to lesser quantities of biotin. Improved communication between patients, health care providers, and laboratory professionals is required concerning the likelihood of biotin interference with immunoassays.
Keywords: biotin interference, cortisol, immunoassay, testosterone, thyroid stimulating hormone
We report a case of biotin interference with endocrine laboratory testing, which nearly resulted in an unnecessary surgical procedure.
1. Case Report
A 48-year-old white woman presented to the endocrinology clinic with palpitations, inability to lose weight, and hirsutism. The referring provider queried whether these symptoms might be related to her known thyroid dysfunction. Subclinical hyperthyroidism, diagnosed 3 years prior, remained of unclear etiology; workup included a normal 24-hour I-131 uptake of 20%, a homogenous I-131 scan, and a negative thyroid-stimulating immunoglobulin. Antithyroid therapy was prescribed; however, each time the patient began therapy, this was discontinued due to weight gain and fatigue. Past medical history was notable for lifelong infertility, Roux-en-Y gastric bypass, tobacco use, and Factor V Leiden requiring chronic anticoagulation. Review of systems was notable for right hip pain due to arthritis, amenorrhea, and hirsutism, which was markedly worse for 6 months. Pertinent negatives included lack of acne, central adiposity, and hyperglycemia. Medications included bupropion 200 mg, calcium/vitamin D3 600-200, duloxetine 30 mg, topiramate 100 mg twice daily warfarin 15 mg, oxycodone 5 mg, and tapentadol 50 mg as needed for hip pain. She denied family history of thyroid disease, hirsutism, or endocrinopathies.
Physical examination demonstrated a blood pressure of 110/77, pulse of 90, height of 175.3 cm (5 feet 9.02 inches), weight of 96.8 kg, and body mass index of 31.50 kg/m2. She was seated comfortably. No exophthalmos, chemosis, or lid lag were noted on ocular exam. Thick terminal hair was present on the upper lip, chin, and neck with a modified Ferriman-Gallwey score of 7. The thyroid was 30 g, smooth, and without nodules. Cardiovascular exam demonstrated a regular rate and rhythm without murmurs. Proximal muscle strength was intact. No violaceous striae or oncholysis was noted. Reflexes were 2+ bilaterally, and she lacked a tremor.
Due to hirsutism, weight gain, and potential thyroid dysfunction, additional endocrine testing was pursued. Thyroid function tests again indicated subclinical hyperthyroidism (Table 1). The am cortisol and total testosterone were elevated at 115 µg/dL and 232 ng/dL, respectively, whereas ACTH was low. FSH and LH concentrations were lower than expected for perimenopausal state. This multitude of aberrant hormone levels prompted an inquiry about potential use of medications and/or supplements. The patient disclosed that she was taking a biotin supplement at a dose of 5000 μg per day at initial presentation. She had been on a biotin supplement regularly for 6 months prior to initial presentation, but also endorsed using biotin intermittently for a period of about 5 years.
Table 1.
Summary of Patient Test Results
| Test | Initial | Weeks After Initial Presentation |
Reference Interval | Method | Biotin Interferencea | |||
|---|---|---|---|---|---|---|---|---|
| 3.5 | 4 | 10 | 12 | |||||
| Pituitary | ||||||||
| ACTH pg/mL | 8.3 (L) | 6.6 (L) | <5.0 (L) | 7.2–63 (am draws) | Sandwich IA, Roche cobas e601 | Yes | ||
| FSH mIU/mL | 5.1 | 4.8 | 10.1 | Follicular 1.9–11.6, luteal 1.4–9.6 | Sandwich IA, OCD Vitros5600 | Yes | ||
| Postmenopausal 21.5–131.0 | ||||||||
| LH mIU/mL | 1.8 | 1.4 | 7.8 | Follicular 2.6–12.1, luteal 0.8–15.5 | Sandwich IA, OCD Vitros5600 | Yes | ||
| Postmenopausal 13.1–86.5 | ||||||||
| IGF-1 ng/mL | 73 (z score: 1.04) | 44–227 (age matched) | LC-MS, LDT | No | ||||
| Prolactin ng/mL | <1.0 (L) | <1.0 (L) | 1.5 (L) | 3.0–19.0 | Sandwich IA, OCD Vitros5600 | Yes | ||
| TSH mIU/mL | 0.06 (L) | 0.09 (L) | 0.06 (L) | 0.6–3.3 | Sandwich IA, OCD Vitros5600 | Yes | ||
| TSH mIU/mL | 1.1 | 0.3–4.3 | Sandwich IA, Beckman Coulter UniCel DXI 800 | No | ||||
| Thyroid | ||||||||
| Free T3 pg/mL | 4.01 | 4.10 | 4.07 | 2.71–6.16 (age matched) | Sandwich IA, OCD Vitros5600 | No | ||
| Free T4 ng/dL | 1.02 | 1.24 | 0.97 | 0.7–1.4 | Sandwich IA, OCD Vitros5600 | No | ||
| Adrenal | ||||||||
| Cortisol µg/dL | 115 (H) | >123 (H) | 26.2b (H) | 2.9 | Before 10 am: 4.5–22.7, after 5 pm: 1.7–14.1 (LDST < 1.8 μg/dl)c | Competitive IA, OCD Vitros5600 | Yes | |
| Cortisol, salivary ng/dL | <50 | 11 pm–12 am: <100 | LC-MS/MS, LDT | No | ||||
| Cortisol (free), urine µg/24 h | 25 | 3.5–45 | LC-MS/MS, LDT | No | ||||
| DHEA-sulfate µg/dL | 21.1 | 28.9 | 18–244 (age matched) | Competitive IA, IMMULITE 2000 | No | |||
| Androstenedione ng/dL | 23 (L) | 38 | 30–200 | LC-MS/MS, LDT | No | |||
| Gonadal | ||||||||
| Total testosterone ng/dL | 232 (H) | 184 (H) | 13.8 | 6–77 | Competitive IA, OCD VITROS 5600 | Yes | ||
| Total testosterone ng/dL | 15 | 11 | 8–60 | LC-MS/MS, LDT | No | |||
| Free testosterone ng/dL | 0.20 | 0.13 | 0.06–0.95 | Equilibrium dialysis, LC-MS/MS, LDT | No | |||
| Other | ||||||||
| Biotin ng/mL | 38 | 3 | Undefined | LC-MS/MSd, LDT | No | |||
| SHBG nmol/L | 51 | 18–144 | Sandwich IA, IMMULITE 2000 | No | ||||
Conversion factors to International System of Units (SI). ACTH: 0.2 pmol/L; IGF-1: 1.0 µg/L; free T3: 1.5 pmol/L; free T4: 12.9 pmol/L; cortisol: 27.6 nmol/L; cortisol, urine: 2.8 nmol; dehydroepiandrosterone-sulfate: 0.03 µmol/L; testosterone/androstenedione: 0.035 nmol/L; free testosterone: 34.7 pmol/L; FSH/LH/TSH: 1.0 IU/mL.
Abbreviations: DHEA, dehydroepiandrosterone; H, above the upper reference limit; IA, immunoassay; L, below the lower reference limit; LC-MS, liquid chromatography tandem mass spectroscopy; LDST, low-dose dexamethasone suppression test; LDT, laboratory-developed test; OCD, Ortho Clinical Diagnostics.
Test contains biotin-based reagents and is susceptible to biotin interference.
am cortisol for low-dose dexamethasone suppression test.
Low-dose dexamethasone suppression test am cortisol <1.8 µg/dL (<49.7 nmol/L), per Endocrine Society clinical practice guidelines.
Research use only.
Our patient’s initial laboratory values were inconsistent with the clinical presentation and suggested biotin interference; she was, therefore, instructed to discontinue biotin in preparation for testing 3.5 weeks later. Surprisingly, these results similarly indicated an unusual hormone profile (Table 1). Due to a multitude of unexplained low pituitary hormone values, particularly her prolactin deficiency, which is rare and typically suggestive of pituitary pathology [1], a pituitary protocol MRI was performed to rule out a nonfunctioning adenoma. Pituitary Cushing disease was not part of the differential as the ACTH was low. Additionally, a CT scan was performed to rule out adrenal pathology. A low-dose dexamethasone suppression test revealed incomplete suppression of cortisol (26 µg/dL) [2], though the measured dexamethasone level was 44 ng/dL (expected 140 to 295 ng/dL after 1 mg dexamethasone), suggesting potential incomplete absorption. Urinary free cortisol and midnight salivary cortisol results were within reference intervals, which was discordant with serum cortisol findings, signifying possible elevation in cortisol binding globulin, as opposed to true hypercortisolemia (Table 1). The patient was referred to reproductive endocrinology to consider a potential testosterone-secreting tumor vs ovarian hyperthecosis as the source for the high testosterone. A pelvic ultrasound was unremarkable; however, a small testosterone-secreting tumor could not be ruled out, and hysterectomy with oophorectomy was recommended and scheduled.
Ten weeks after initial presentation, the patient’s free and total testosterone were measured using liquid chromatography tandem mass spectroscopy (LC-MS/MS) and found to be within reference intervals (Table 1). Health care professionals in the laboratory were consulted regarding the differing measured testosterone concentrations. A pattern was identified consisting of aberrant hormones by competitive and immunometric immunoassays compared with normal hormone measurements by LC-MS/MS (Table 1). Notably, tests via LC-MS/MS are not prone to biotin interference. Collectively, these findings suggested potential biotin interference with immunoassay testing as a cause for the patient’s aberrant hormonal profile. Ten weeks after presentation, our patient’s sample was thus tested for biotin by LC-MS/MS (research use only), which measured a biotin concentration of 38 ng/mL (Table 1).
Upon further questioning, at 10 weeks, the patient admitted to continued use of biotin supplements. She agreed to discontinue biotin supplements for repeat testing. Two weeks later (i.e., 12 weeks), the patient’s serum total testosterone measured by immunoassay and LC-MS/MS were within reference intervals; additionally, her serum biotin concentration reduced to 3 ng/mL (Table 1). Most hormones measured using immunoassays (susceptible to biotin interference) normalized, except for TSH. Repeat TSH testing in the same sample was performed utilizing a different manufacturer’s immunoassay that is not susceptible to biotin interference, which yielded a TSH concentration of 1.1 mIU/mL, within reference interval (Table 1). Clinical evidence suggests that the patient was, indeed, euthyroid.
2. Discussion
Seventy percent of older adults in the United States reported using more than one dietary supplement in the past 30 days [3]. Such patients may fail to share this information with medical providers. Supraphysiologic biotin concentrations in individuals ingesting biotin-containing supplements, exceeding the recommended daily intake of 30 µg, interfere with select laboratory tests, specifically immunoassays using biotinylated antibodies [4, 5]
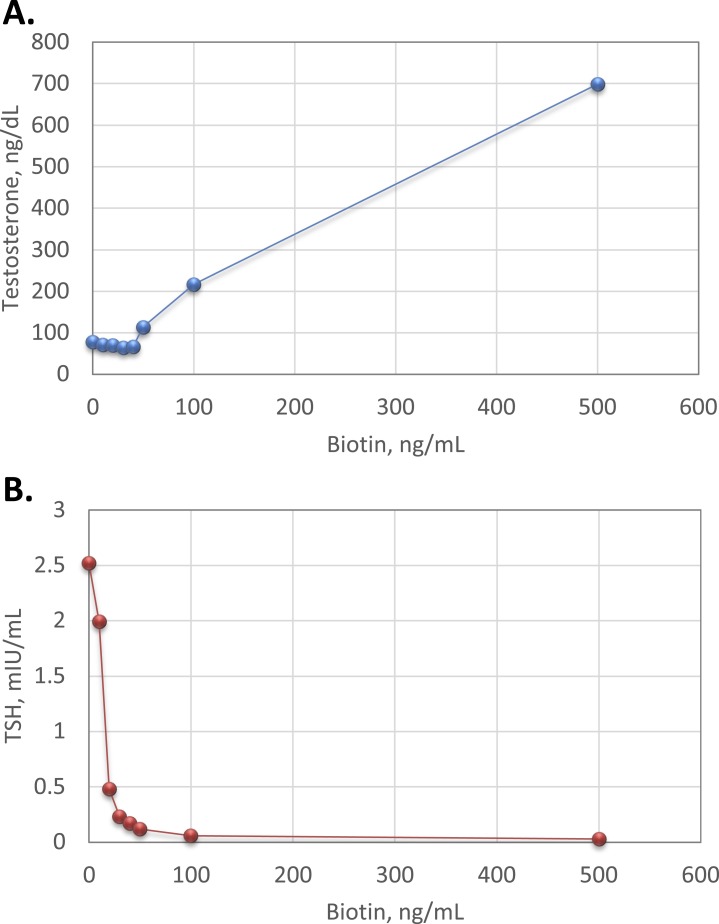
Biotin interference with competitive immunoassays can cause falsely increased results, whereas biotin interference with immunometric “sandwich” assays falsely lowers results (Fig. 1). As shown in Fig. 2, our internal studies using residual patient-pooled serum matrix and exogenous biotin demonstrate that biotin interference causes marked positive bias in testosterone measured by a competitive immunoassay and marked negative bias in TSH measured by an immunometric “sandwich” assay (Fig. 2). As such, biotin interference is analyte-specific and method dependent and varies by manufacturer.
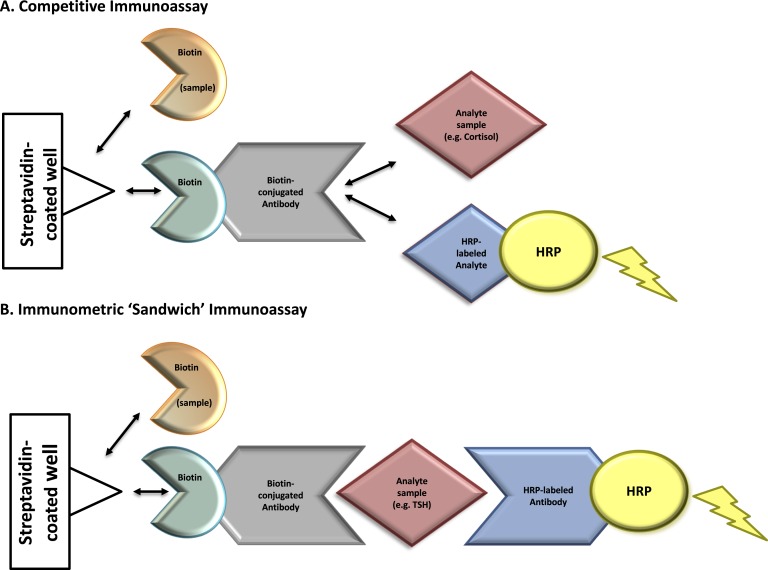
Figure 1.
Mechanism of biotin interference with immunoassay methodologies. Immunoassays in this case report contain a solid phase with a wash step that physically separates the label-bound analyte/antibody from the free label. Assays that contain this step are referred to as “heterogeneous” or “multistep” (vs homogeneous assays that do not contain a wash step). (A) Competitive immunoassays are comprised of exogenous biotin-conjugated antibodies that compete for binding with an analyte of interest in the patient’s sample as well as exogenous labeled analyte. Complexes of biotin-antibody-analyte are captured to a streptavidin-coated well through strong interactions between biotin and streptavidin. A wash step removes any unbound materials. The exogenous labeled analyte is conjugated to an enzyme [e.g., horseradish peroxidase (HRP)]. Substrate is added to the well and oxidized by horseradish peroxidase, producing a luminescence signal, measured by spectrophotometry. The measured signal is inversely proportional to the concentration of analyte in the patient’s sample. Elevated concentrations of biotin in a patient’s sample can compete with biotin-antibody-(labeled) analyte complexes for binding to the streptavidin-coated well. This leads to the detection of a diminished signal causing a falsely high analyte result. (B) Immunometric “sandwich” immunoassays contain an exogenous biotin-conjugated antibody and exogenous labeled antibody. Both antibodies bind to the same analyte of interest, forming a “sandwich”. Biotin-antibody-analyte-labeled antibody complexes are captured to streptavidin-coated wells through strong interactions between biotin and streptavidin. A wash step removes any unbound materials. Exogenous labeled antibody is conjugated to an enzyme (horseradish peroxidase). Substrate is added to the well and oxidized by horseradish peroxidase, producing a luminescence signal proportional to the analyte’s concentration. Elevated biotin in a patient’s sample can compete with biotin-antibody-analyte-labeled antibody complexes for binding to the streptavidin-coated well. This leads to the detection of a diminished signal causing a falsely low analyte result. Horseradish peroxidase–labeled analyte, tracer-analyte; horseradish peroxidase–labeled antibody, tracer-antibody.
Figure 2.
Effect of biotin interference with measured analyte concentrations. Increasing concentrations (0, 10, 20, 30, 40, 50, 100, and 500 ng/mL) of exogenous biotin (Sigma Aldrich, St. Louis, MO) were added into aliquots of residual patient serum matrix. Samples were tested for total testosterone and TSH using competitive immunoassay and immunometric “sandwich” immunoassay methodologies, respectively (Ortho Clinical Diagnostics, Raritan, NJ;Vitros5600). Plots show measured (A) total testosterone and (B) TSH vs biotin concentration.
This is an unusual case of biotin interference with immunoassay testing for multiple tests, including total testosterone. In this patient’s case, the initial test results were clinically misleading, prompting numerous consultations and unnecessary radiographic and laboratory testing. The negative clinical impact included weeks of psychological distress considering the possibilities of hypercortisolemia or a testosterone-producing tumor. Most significantly, these abnormal test results nearly resulted in an unnecessary invasive procedure for a complex patient with a hypercoagulable state. Other cases of biotin interference with laboratory testing have been described, most of which involve interference with thyroid function tests [6–10]; however, the prevalence of biotin interference in laboratory testing remains unknown. A prior case of falsely elevated sex steroid hormones resulted in unnecessary surgery, highlighting the impact of this clinical problem [11]. Collectively, biotin interference with laboratory testing presents serious risks to patient safety and may lead to unnecessary testing/procedures and health care costs.
Until manufacturers modify immunoassays to resist biotin interference, strategies to mitigate patient harm from biotin interference are required. The US Food and Drug Administration recently issued a safety communication regarding biotin interference with laboratory tests, recommending a multidisciplinary approach for lessening risks (Food and Drug Administration, November 2017). Education as well as communication between laboratorians, providers, and patients is vital. Laboratorians can inform providers regarding test limitations including biotin interference with select tests, as well as develop protocols for investigating potential interference in suspected samples (e.g., alternate testing procedures; biotin sequestration by streptavidin-coated microparticles) [4, 12]. Providers similarly can alert patients regarding biotin interference and inquire about supplement use. Patients are advised to discontinue biotin supplements prior to immunoassay testing, when feasible. In sum, unexpected laboratory results require providers and laboratory professionals to consider biotin interference. It is vital not only to recognize the interference, but also to prompt rigorous clinical and laboratory investigations regarding the prevalence of biotin interference and need for alternative laboratory assays.
Acknowledgments
Financial Support: This work was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases (AR062097 to M.S.) and the National Institute of Diabetes and Digestive and Kidney Diseases (P30 DK056350 to M.S.).
Disclosure Summary: The authors have nothing to disclose.
Glossary
Abbreviation:
- LC-MS/MS
liquid chromatography tandem mass spectroscopy
References and Notes
- 1. Mukherjee A, Murray RD, Columb B, Gleeson HK, Shalet SM. Acquired prolactin deficiency indicates severe hypopituitarism in patients with disease of the hypothalamic-pituitary axis. Clin Endocrinol (Oxf). 2003;59(6):743–748. [DOI] [PubMed] [Google Scholar]
- 2. Nieman LK, Biller BM, Findling JW, Newell-Price J, Savage MO, Stewart PM, Montori VM. The diagnosis of Cushing’s syndrome: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2008;93(5):1526–1540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Gahche JJ, Bailey RL, Potischman N, Dwyer JT. Dietary supplement use was very high among older adults in the United States in 2011-2014. J Nutr. 2017;147(10):1968–1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Li D, Radulescu A, Shrestha RT, Root M, Karger AB, Killeen AA, Hodges JS, Fan SL, Ferguson A, Garg U, Sokoll LJ, Burmeister LA. Association of biotin ingestion with performance of hormone and nonhormone assays in healthy adults. JAMA. 2017;318(12):1150–1160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Trambas C, Lu Z, Yen T, Sikaris K. Depletion of biotin using streptavidin-coated microparticles: a validated solution to the problem of biotin interference in streptavidin-biotin immunoassays. Ann Clin Biochem. 2018;55(2):216–226. [DOI] [PubMed] [Google Scholar]
- 6. Barbesino G. Misdiagnosis of Graves’ disease with apparent severe hyperthyroidism in a patient taking biotin megadoses. Thyroid. 2016;26(6):860–863. [DOI] [PubMed] [Google Scholar]
- 7. Willeman T, Casez O, Faure P, Gauchez AS. Evaluation of biotin interference on immunoassays: new data for troponin I, digoxin, NT-Pro-BNP, and progesterone. Clin Chem Lab Med. 2017;55(10):e226–e229. [DOI] [PubMed] [Google Scholar]
- 8. Minkovsky A, Lee MN, Dowlatshahi M, Angell TE, Mahrokhian LS, Petrides AK, Melanson SE, Marqusee E, Woodmansee WW. High-dose biotin treatment for secondary progressive multiple sclerosis may interfere with thyroid assays. AACE Clin Case Rep. 2016;2(4):e370–e373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Elston MS, Sehgal S, Du Toit S, Yarndley T, Conaglen JV. Factitious Graves’ disease due to biotin immunoassay interference-a case and review of the literature. J Clin Endocrinol Metab. 2016;101(9):3251–3255. [DOI] [PubMed] [Google Scholar]
- 10. Batista MC, Ferreira CES, Faulhaber ACL, Hidal JT, Lottenberg SA, Mangueira CLP. Biotin interference in immunoassays mimicking subclinical Graves’ disease and hyperestrogenism: a case series. Clin Chem Lab Med. 2017;55(6):e99–e103. [DOI] [PubMed] [Google Scholar]
- 11. Langlois F, Moramarco J, He G, Carr BR. Falsely elevated steroid hormones in a postmenopausal woman due to laboratory interference. J Endocr Soc. 2017;1(8):1062–1066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Lam L, Kyle CV. A simple method to detect biotin interference on immunoassays. Clin Chem Lab Med. 2017;55(6):e104–e106. [DOI] [PubMed] [Google Scholar]