Abstract
Purpose
This study was conducted to investigate the effects of an experimental bacterial keratitis model on the corneal collagen cross-linking treatment (CXL), and also to compare topical antibiotic treatment with the combined treatment.
Methods
The study involved 40 young adult female Sprague Dawley rats, which had a 2 mm scraped defect of the central corneal epithelium in both eyes. The rats were divided into two equal groups. The first group was inoculated in both eyes with standard Pseudomonas Aeruginosa (PA) from a strain suspension prepared from 0.05 ml (Group 1), and the second group was inoculated with standard Methicillin Resistance Staphylococcus Aureus (MRSA) strains from a suspension prepared from 0.05 ml (Group 2). Group 1 was divided into four sub-groups: Group 1A was treated by collagen cross-linking (CXL), Group 1C was treated with topical tobramycin drops CXL and also treated by collagen cross-linking (CXL), Group 1D was treated with topical tobramycin drops, and Group 1B was left untreated in order to create a control group. Similarly, Group 2 was also divided into four sub-groups: Group 2A was treated by CXL, Group 2C was treated with topical 5% fortified vancomycin drops CXL and also treated by CXL, Group 2D was treated with topical 5% fortified vancomycin drops, and Group 2B was left untreated in order to create a control group. CXL was performed on the third day following the inoculation and topical drop therapy. Biomicroscopy and microbiologic assessments were performed on the third and seventh days following the inoculation of microorganisms.
Results
In the treatment, which compared baselines in all groups before treatment, the diameter of keratitis infiltrations, corneal clouding, and corneal swab samples were obtained from the reduction in reproduction. The results were statistically significant (p < 0.01). Keratitis infiltration groups were conducted on the seventh day for Groups 1C and 1D according to Group 1B, whilst Groups 2A, 2C and 2D were conducted according to Group 2B, which showed a significant statistical reduction (p < 0.01). On the seventh day, focal groups were conducted in corneal clouding Group 1D according to Group 1B and in Groups 2A, 2C and 2D according to Group 2B, which revealed a significant statistical reduction (p < 0.01). On the seventh day, reproduction in culture was obtained from corneal swab samples in Groups 1C and 1D according to Group 1B; in Groups 1C and 1D according to Group 1A; in Groups 2A, 2C and 2D according to Group 2B; and in Group 2C according to Group 2A, where a significant statistical reduction was observed (p < 0.01).
Conclusions
The clinical and microbiological efficacy of the CXL treatment is evaluated in our study. In accordance with the conclusion reached an effective reduction in the density and severity of (infection), occurred as a result of CXL treatment, CXL treatment combined with topical antibiotic treatment and topical antibiotic treatment of Pseudomonas Aeruginosa (PA) and Metisilin Rezistant Staphylococcus Aureus (MRSA) keratitis infections. From these results, it is shown that topical antibiotics and CXL potentiate each other’s effects in the treatment of resistant bacterial keratitis.
Keywords: Cornea, Bacterial Keratitis, Cross-linking
Introduction
It is currently common practice to treat corneal infections with a broad range of antibiotics but, as the medical profession is faced with the problem of increased bacterial resistance to antibiotic medication, the need to introduce alternative treatments is becoming an issue of growing significance.1 This is particularly crucial when considering corneal infections, as serious damage to the patient’s vision, and even blindness, can ensue, leading to the necessity for more serious treatments, such as chemotherapy or surgery.
One alternative to antibiotic treatment is known as corneal collagen crosslinking (CXL). This treatment works by using UV–A at 365 or 370 nm to activate the photosensitive properties of riboflavin to initiate photochemical reactions which result in covalent bonds or crosslinks in the corneal stroma, which in turn can augment the cornea’s biomechanical strength and, thus, potentially arrest the further advancement of keratoconus.2, 3 The idea of using UV rays against microorganisms is certainly not new and is regularly found in research; it is also widely utilised in medical settings such as operating theatres to augment existing sterilisation techniques.4, 5
The first relevant study in this area was carried out in 1960, when it was revealed that, upon exposure to UV rays, riboflavin would inactivate the RNA of various viruses. Contemporary studies have found that riboflavin also behaves as a photomediator and has the capacity to neutralize not only pathogens in plasma, platelets, and red blood cells6, 7, 8 but also a variety of viruses, bacteria, and parasites.9
The treatment of corneal infections using CXL confers an additional benefit, in that resistance of the cornea to enzymatic digestion by microorganisms10 is increased.
It is the case that 90% of the thickness of the cornea consists of the stroma, which is made up of collagen fibrils caught up within an environment of proteoglycans, proteins, glycoproteins and keratocytes located amid the collagen lamellae.
Some microorganisms, including bacteria and fungi, emit enzymes that have the capacity to digest collagen, in turn leading to melting and perforation of the cornea. Notwithstanding microbiological remedy, deterioration of collagen and proteolysis as a result of enzymatic action may well still be apparent in the form of progressive ulceration. Use of the CXL treatment in porcine testing has had very positive results against enzymatic degeneration by collagenase, trypsin and pepsin. It can therefore be supposed that CXL is not only able to neutralise the microorganisms causing the infection, but also confer the benefit of corneal rigidity, thereby lessening vulnerability of the stroma to proteolysis and the advancement of corneal melt.10, 11, 12
Material and methods
Preparations of the strains
The standard strains PA ATCC 9027 and MRSA ATCC 33591 were utilized in this study. It was necessary for the strains to be kept at a temperature of −86 °C until required for testing purposes, at which point they were revived with agar containing 5% sheep blood; was necessary for them to be passaged twice with blood agar. The suspensions of the strains are dense and they were prepared in sterile physiological serum. The actual concentration of bacteria required was 1.0 × 108 CFU/ml in each 50 μl suspension to be introduced into the rat eyes; this was achieved by means of dilution using physiological saline solution spectrophotometrically.
Animals
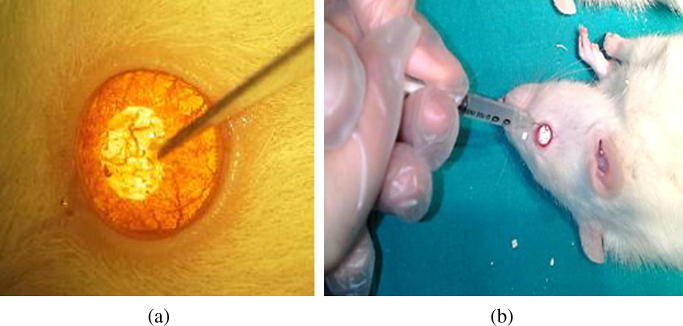
The study included 40 young adult female Sprague Dawley rats weighing between 230 and 250 kg. All rats were treated in accordance with the Association for Research in Vision and Ophthalmology statement for the use of animal in ophthalmic research, and the protocol was approved by the Institutional Ethics Committee. The rats were anesthetized with 50 mg/kg of intraperitoneal ketamine hydrochloride and 7 mg/kg of xylazine before all interventions. Corneal anesthesia was attained using 0.5% topical proparacaine hydrochloride. The central corneal epithelium was marked with a 2 mm disposable dermatological skin punch (Acu-Punch, Acuderm, Ft. Lauderdale, FL). The marked corneal epithelium was scraped using a number 11 scalpel (Fig. 1a). The rats were divided into two equal groups. The first group was inoculated into both eyes with standard Pseudomonas Aeruginosa (PA) from a strain suspension prepared from 0.05 ml (Group 1), and the second group was inoculated with standard Methicillin Resistance Staphylococcus Aureus (MRSA) strains from a suspension prepared from 0.05 ml (Group 2) (Fig. 1b). Three days later, bio microscopic examination revealed keratitis in both eyes of all rats. All rats were swallowed from the cornea of both eyes and microbiologically appeared to be infected.
Fig. 1.
(a) Abrasion of the corneal epithelium using a number 11 scalpel. (b) Introduction of microorganism suspension on the created epithelial defect area.
Group 1 was divided into four sub-groups: Group 1A was treated by collagen cross-linking (CXL), Group 1C was treated with topical tobramycin drops (CXL) and also treated by collagen cross-linking (CXL), Group 1D was treated with topical tobramycin drops, and Group 1B was left untreated in order to create a control group. Similarly, Group 2 was also divided into four sub-groups: Group 2A was treated by CXL, Group 2C was treated with topical 5% fortified vancomycin drops (CXL) and also treated by (CXL), Group 2D was treated with topical 5% fortified vancomycin drops, and Group 2B was left untreated in order to create a control group (Table 1).
Table 1.
Rats groups’ definition with treatment.
| Experimental rats | Eye | Groups | Group definition | Group treatment |
|---|---|---|---|---|
| Rat 1 | Left eye | Group 1 A | Crosslinking Groups | Treated by collagen cross-linking (CXL) |
| Right Eye | Group 1 B | Control Groups | Untreated in order to create a control group | |
| Rat 2 | Left eye | Group 1 C | Crosslinking Groups | Treated by collagen cross-linking (CXL), and also treated with topical tobramycin drops |
| Right Eye | Group 1 D | Control Groups | Treated with topical tobramycin drops | |
| Rat 1 | Left eye | Group 2 A | Crosslinking Groups | Treated by collagen cross-linking (CXL) |
| Right Eye | Group 2 B | Control Groups | Untreated in order to create a control group | |
| Rat 2 | Left eye | Group 2 C | Crosslinking Groups | Treated by collagen cross-linking (CXL), and also treated with topical tobramycin drops |
| Right Eye | Group 2 D | Control Groups | Treated with topical tobramycin drops | |
Treatment
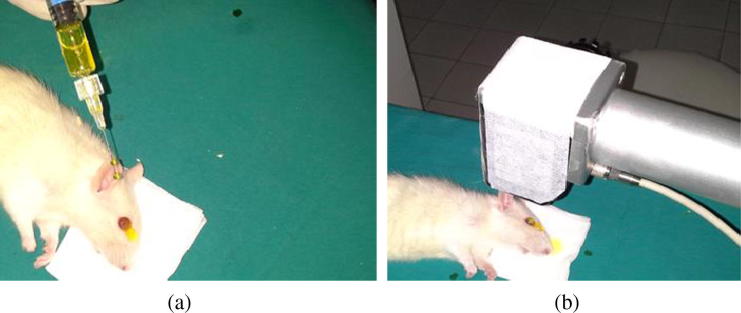
The CXL procedure was performed three days after the inoculation of Pseudomonas Aeruginosa (PA) and Methicillin Resistance Staphylococcus Aureus (MRSA), conducted under sterile conditions in an operating room. Topical drop therapy was undertaken within those groups which were exposed to CXL treatment after CXL (on the third day), and no CXL treatment (only topical antibiotics treatment the third day after introduction of the microorganism suspension). The rats were anesthetized with intramuscular 50 mg/kg of ketamine hydrochloride and 7 mg/kg of xylazine before all interventions and corneal anesthesia was attained using 0.5% topical proparacaine hydrochloride. Using a number 11 scalpel, a 2 mm central fragment of the corneal epithelium was carefully detached. As a photosensitizer, a riboflavin 0.1% solution (10 mg of riboflavin-5-phosphate in 10 mL of dextran–T-500 20% solution) was applied every five minutes for 30 min before irradiation and every five minutes during irradiation (Fig. 2a and b). After allowing the riboflavin to permeate through the cornea, UV-A irradiation was started. For the operation, a 370 nm UV light-emitting diode as well as CBM Vega X-Link equipment with a power density of irradiation of 3 mW/cm2, maximum radiated power of 4 mW, an irradiated area diameter of 4–11 mm and a single led UV-A as a UV source were used. The operation was performed on all eyes in Groups 1A, 1C, 2A and 2C from a distance of 54 mm for 30 min. The cornea of each rat was irradiated by use of the UV light diodes (370 nm) at a distance of 54 mm for 30 min using 3 mW/cm2 irradiance; this equals a dose of 5.4 J/cm2.
Fig. 2.
(a) Introduction of 0.1% riboflavin solution in preparation for irradiation. (b) Introduction of 0.1% riboflavin solution during irradiation.
Clinical examination
The eyes were examined on day 0 (bacterial inoculation), day 3 (prior to undergoing CXL and topical treatment), and day 7 (four days after CXL and topical treatment) using a portable slit-lamp biomicroscope. The extent of keratitis was evaluated by a masked observer. The diameter of corneal infiltration and corneal clouding were evaluated using the modified scoring system of Ozturk et al.13 The diameter of corneal infiltration was measured in millimeters. Corneal infiltration was graded as follows: corneal infiltration is unavailable, 0; corneal infiltration < 1.5 mm, 1; corneal infiltration 1.5–3 mm, 2; and corneal infiltration > 3 mm, 3. Corneal clouding was graded as follows: clear cornea, 0; minor edema, 1; corneal edema in two quadrants of the cornea, 2; and corneal edema in more than two quadrants of the cornea, 3.
Microbiological analysis
The eyes were examined on day 3 (prior to performing CXL and topical treatment), and on day 7 (four days after CXL and topical treatment) corneal swab samples placed into amies transport medium after anesthesia (as defined above) were cultivated with 5% sheep blood agar in the laboratory.
The plates were incubated overnight at 37 °C and then the density of reproductions were graded from 0 to 4 using the following system: no reproduction, 0; an insignificant number of colonies in the first area, 1; a significant number of colonies in the first area, 2; reproduction in the second area also, 3; and dense reproduction extending to the third area, 4.
Statistical analysis
The Statistical Package for Social Sciences (Version 17, Chicago IL, USA) (SPSS) was utilised to analyse the data and variables were designated as mean ± standard deviation. In order to evaluate observance of the data to a repeated measures analysis of variance, Mauchly's sphericity test and Box-M’s test of Homogeneity of Variances was applied. Repeated measures variance, which was a factor included in the factorial design, was used to compare the means. Where preconditions of parametric tests (repeated measures variance analysis on factorial design) were unfulfilled, the Greenhouse-Geisser test (1959), correcting the degrees of freedom, or Huynh-Feldt (1976), were utilized, and the Corrected Bonferroni test was implemented to perform multiple comparisons. The values p < 0.05 and p < 0.01 were followed for use in the significance level of the tests.
Results
Clinical examination results
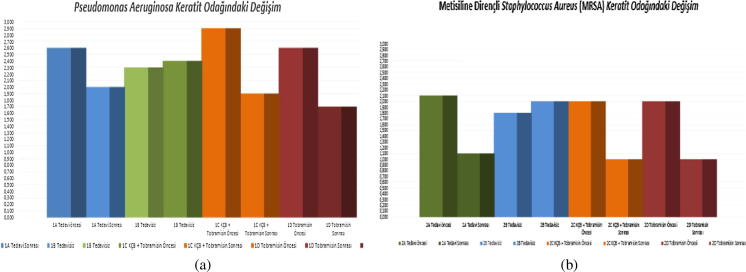
By day 3 (72 h after inoculation), it was found that Pseudomonas Aeruginosa (PA) and Methicillin Resistance Staphylococcus Aureus (MRSA) keratitis had become established in all inoculated eyes. At day 0, no statistically significant variation between groups existed (p > 0.05); however, at day 3, the diameter of keratitis infiltrations keratitis were statistically and significantly higher in Group 1C (CXL+ tobramycin) than in Group 1B (untreated) (p = 0.04). No such statistically significant difference existed within the remaining groups on day 3 (p > 0.05). Subsequently, a statistically significant reduction in the diameter of keratitis infiltrations in Groups 1A (CXL), 1C (CXL+ tobramycin), 1D (tobramycin), 2A (CXL), 2C (CXL+ vancomycin) and 2D (vancomycin) was in evidence after treatment (day 7) in comparison to that seen prior to treatment (day 3) (p < 0.01) (Fig. 3a and b).
Fig. 3.
(a) Group 1 changes in keratitis focus (days 3 and 7). (b) Group 2 changes in keratitis focus (days 3 and 7).
A statistically significant decrease in the diameter of keratitis infiltrations were noted on day 7 between Group 1C, which had undergone topical tobramycin treatment combined with CXL, and Group 1B (control group), which had gone untreated (p = .039). Similarly, a decrease was noted between Group 1D, which had undergone only topical tobramycin treatment combined with CXL, and Group 1B (control group), which had gone untreated (p = 0.002). A statistically significant decrease in the diameter of keratitis infiltrations were also noted on day 7 between Group 2A, which had only undergone CXL treatment, and Group 2B (control group), which had gone untreated (p = 0.001), as well as between Group 2C, which had undergone topical fortified vancomycin therapy combined with CXL, and Group 2B (control group), which had gone untreated (p = 0.001), and also between Group 2D, which had undergone only topical fortified vancomycin therapy, and Group 2B (control group), which had gone untreated (p = 0.001). No statistically significant difference was found in terms of the diameter of keratitis infiltrations on day 7 between the remaining groups (p > 0.05).
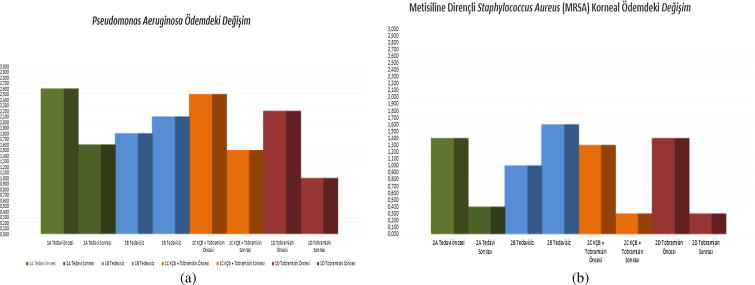
On day 3, prior to the treatment, corneal clouding was observed to be statistically significant in Group 1A (CXL) compared to Group 1B (untreated) (p = 0.11) and in Group 1C (CXL+ tobramycin) compared to Group 1B (untreated) (p = 0.33). No such statistical significance was found on day 3 between the remaining groups (p > 0.05). A statistically significant reduction in corneal clouding was observed in Groups 1A (CXL), 1C (CXL+ tobramycin), 2A (CXL), 2C (CXL+ vancomycin) and 2D (vancomycin) subsequent to treatment (day 7) compared to prior to treatment (day 3) (p < 0.01)) (Fig. 4a and b).
Fig. 4.
(a) Group 1 change in corneal edema (days 3 and 7). (b) Group 2 change in corneal edema (days 3 and 7).
Group 1D, which had been treated only with topical tobramycin, exhibited statistically significant deterioration in corneal clouding on day 7 compared to Group 1B (control group), which had gone untreated (p = 0.004). Group 2A, which had been treated only with CXL, exhibited a statistically significant deterioration in corneal clouding on day 7 compared to Group 2B (control group), which had gone untreated (p = .001), the deterioration was also found in Group 2C, which had been treated with topical fortified vancomycin combined with CXL, compared to Group 2B (control group), which had gone untreated (p = 0.001) and also in Group 2D, which had been treated with only topical fortified vancomycin, compared to Group 2B (control group), which had gone untreated (p = 0.001). No such statistical significance was found between the remaining groups on day 7 (p > 0.05) (Fig. 5a–h).
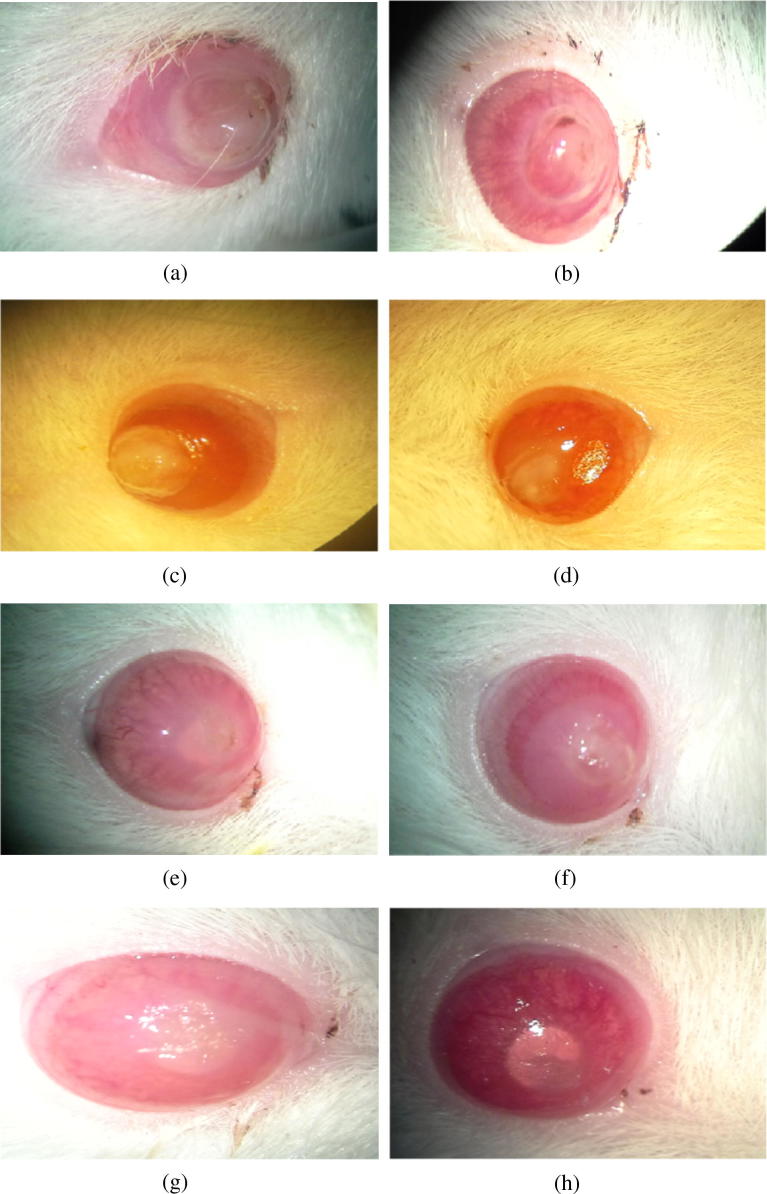
Fig. 5.
(a) GROUP 1A before CXL. (b) GROUP 1A after CXL. (c) GRUP 1C before CXL+ Topical Tobramycin. (d) GRUP 1C after CXL+ Topical Tobramycin. (e) GRUP 2B 3 days after bacterial inoculation. (f) GRUP 2B 7 days after bacterial inoculation. (g) GRUP 2D before Topical vancomycin. (h) GRUP 2D after Topical vancomycin.
Microbiological analysis results
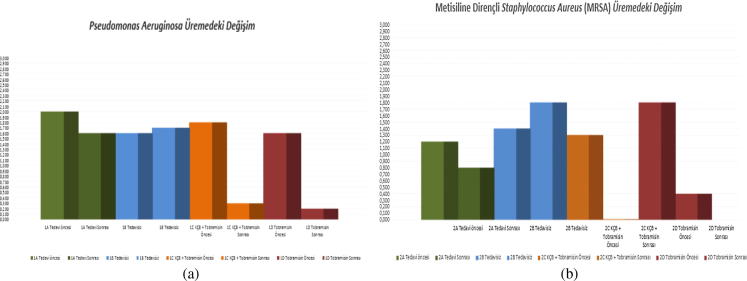
Appraisal of each of the cultures derived from the corneal swabs taken from the eyes of each of the rats on days 3 and 7 was undertaken. All cultures collected on day 3 exhibited reproduction. No statistically significant difference in the cultures had existed between the groups within the context of reproduction prior to the treatment (day 3) (p > 0.05). However, a statistically significant reduction within the context of reproduction was in evidence (p < 0.01) subsequent to treatment (day 7) in contrast to levels in existence before treatment (day 3) in Groups 1A (CXL), 1C (CXL+ tobramycin), 1D (tobramycin), 2A (CXL), 2C (CXL+ vancomycin) and 2D (vancomycin) (Fig. 6a and b).
Fig. 6.
(a) Group 1 change in reproduction (days 3 and 7). (b) Group 2 change in reproduction (days 3 and 7).
On day 7, statistically significant reductions in the reproduction of cultures were observed between Group 1C (CXL+ tobramycin) and Group 1B (untreated) (p = 0.001), Group 1D (tobramycin) and Group 1B (untreated) (p = 0.001), Group 1C (CXL+ tobramycin) and Group 1A (CXL) (p = 0.001) and Group 1D (tobramycin) and Group 1A (CXL) (p = 0.001). On day 7, statistically significant reductions in the reproduction of cultures were also observed between Group 2A (CXL) and Group 2B (untreated) (p = 0.001), Group 2C (CXL+ vancomycin) and Group 2B (untreated) (p = 0.001), Group 2D (vancomycin) and Group 2B (untreated) (p = 0.001) and Group 2C (vancomycin) and Group 2A (CXL) (p = 0.008). No statistically significant difference in the reproduction of cultures on day 7 was found to exist between the remaining groups (p > 0.05).
Discussion
Studies relating to in vitro have demonstrated wide-ranging antimicrobial effects to be gained via the combined use of riboflavin and UV–A, which has proved effectual in pre-clinical studies of platelets and plastma against pathogens such as bacteria, intracellular human immunodeficiency virus-1, West Nile virus and porcine parvovirus. It is thought that the combination of riboflavin and UV–A causes nucleic acids to degenerate by direct electron transfer, the production of singlet oxygen and the generation of hydrogen peroxide with the formation of hydroxyl radicals. It is therefore clear that the riboflavin UV-A combination has huge potential in terms of offering an effective medical treatment with low protein damage and little toxicity.15
In tests, relief of the symptoms of acanthamoeba keratitis have been evident within 24 h of the implementation of CXL treatment, including vision improvement and healing. In addition, the corneal stability gained via CXL prevents continued tissue damage and disengages the amoebae, thereby preventing further spread.16, 17, 18 CXL treatment administered to patients with corneal melt attributable to contact lens use with infectious keratitis caused by bacteria, fungi, and acanthamoeba also appears to offer improved healing and improved overall outcomes, subsequent to such surgical procedures as corneal transplantation.19
Recent research has been carried out in relation to a sample of 16 cases of microbial keratitis without prior antibiotic management. These patients undertook CXL treatment as their primary mode of treatment. In all cases, abatement of inflammation and epithelial healing occurred; only two patients required additional treatment in the form of a standard topical antibiotic.20
Current research suggests that, whilst CXL treatment is a potent treatment for bacterial corneal infections, it is not necessarily effective for fungal infection, while, for herpes simplex keratitis, is potentially best avoided altogether as a treatment method. Clearly, further study is required further study into the use of CXL, and comparisons made with ocular antibiotics for validation purposes; nevertheless, CXL demonstrates very positive effects and potential benefits in terms of use on the corneal surface.21
As far as side effects are concerned, much research has shown that CXL with riboflavin treatment gives only minimal cause for concern. Wollensak et al. undertook a pilot study on the subject, which noted that corneal and lens transparency, endothelial cell density, and intraocular pressure all remained unaltered following treatment for keratoconus.3 A further study on the safety of CXL, carried out by Spoerl et al. to assess possible damage to ocular tissues,22 determined that no impairment to the corneal endothelium, the lens, or the retina was likely in cases where the fundamental criteria for the treatment was met.23 Certainly, no serious side effects were apparent in the CXL group of this study.
The experimental rabbit model of Galperin et al. in relation to fusarium keratitis assessed CXL with riboflavin in terms of its clinical and microbiological effectiveness, and reported that although the treatment is effectual in diminishing the density and severity of fungal keratitis infection, the treatment is insufficient in cases of fungal keratitis, and therefore maybe of value only a complementary treatment for fungal keratitis that is resistant to treatment.1 The experimental rabbit model of Berra et al. in relation to acanthamoeba keratitis assessed CXL with riboflavin in terms of its clinical and microbiological effectiveness and reported the treatment to be ineffectual in diminishing the density and severity of acanthamoeba keratitis infection in addition to clinical and microbiological degeneration in the affected corneas. In conclusion, CXL was deemed to be inappropriate in the treatment in acanthamoeba keratitis.16
A publication by Alio et al. concluded that CXL may well offer an effective contemporary approach to dealing with infectious keratitis and, in their opinion, the treatment successfully controls infectious keratitis by inhibiting corneal melting. Unfortunately, the lack of availability of control groups will hinder the further development of this treatment in the immediate future, although the researchers highlighted the potential for studies which compare the application of CXL with typical topical antibiotic therapy.24
The clinical and microbiological efficacy of the CXL treatment is evaluated in this study. CXL treatment, CXL treatment combined with topical antibiotic treatment and the topical antibiotic treatment of Pseudomonas Aeruginosa (PA) and Metisilin Rezistant Staphylococcus Aureus (MRSA) keratitis infections led to an effective reduction in the density and severity (of infection), in accordance with the conclusion reached by the researchers. From these results, it is shown that topical antibiotics and CXL potentiate each other’s effects in the treatment of resistant bacterial keratitis. It is concluded that these findings should be supported by a more extensive series of tests and other practices on humans. Further investigation of this treatment both by in vitro and in vivo studies, is essential before it can be incorporated into everyday clinical practice.
Conflict of interest
The authors declare that there is no conflict of interest.
Footnotes
Peer review under responsibility of Saudi Ophthalmological Society, King Saud University.
References
- 1.Galperin G., Berra M., Tau J., Boscaro G., Zarate J., Berra A. Treatment of fungal keratitis from Fusarium infection by corneal cross-linking. Cornea. 2012;31:176–180. doi: 10.1097/ICO.0b013e318221cec7. [DOI] [PubMed] [Google Scholar]
- 2.Wollensak G. Crosslinking treatment of progressive keratoconus: new hope. Curr Opin Ophthalmol. 2006;17:356–360. doi: 10.1097/01.icu.0000233954.86723.25. [DOI] [PubMed] [Google Scholar]
- 3.Wollensak G., Spoerl E., Seiler T. Riboflavin/ultraviolet-a-induced collagen crosslinking for the treatment of keratoconus. Am J Ophthalmol. 2003;135:620–627. doi: 10.1016/s0002-9394(02)02220-1. [DOI] [PubMed] [Google Scholar]
- 4.Rau H. A transportable apparatus for UV irradiation in final disinfection of patient rooms. Dtsch Gesundheitsw. 1960;15:148–151. [in German] [PubMed] [Google Scholar]
- 5.Müller G., Goethe H., Herrmann R. Ship drinking water disinfection using UV irradiation. II. Zentralbl Bakteriol Orig B. 1972;156:361–372. [in German] [PubMed] [Google Scholar]
- 6.Tsugita A., Okada Y., Uehara K. Photosensitized inactivation of ribonucleic acids in the presence of riboflavin. Biochim Biophys Acta. 1965;103:360–363. doi: 10.1016/0005-2787(65)90182-6. [DOI] [PubMed] [Google Scholar]
- 7.Asano H., Lee C.Y., Fox-Talbot K. Treatment with riboflavin and ultraviolet light prevents alloimmunization to platelet transfusions and cardiac transplants. Transplantation. 2007;84:1174–1182. doi: 10.1097/01.tp.0000287318.94088.d7. [DOI] [PubMed] [Google Scholar]
- 8.Goodrich R.P. The use of riboflavin for the inactivation of pathogens in blood products. Vox Sang. 2000;78(suppl 2):211–215. [PubMed] [Google Scholar]
- 9.Makdoumi K., Mortensen J., Crafoord S. Infectious keratitis treated with corneal crosslinking. Cornea. 2010;29:1353–1358. doi: 10.1097/ICO.0b013e3181d2de91. [DOI] [PubMed] [Google Scholar]
- 10.Spoerl E., Wollensak G., Seiler T. Increased resistance of crosslinked cornea against enzymatic digestion. Curr Eye Res. 2004;29:35–40. doi: 10.1080/02713680490513182. [DOI] [PubMed] [Google Scholar]
- 11.American Academy of Ophthalmology. Basic and clinical science course: fundamentals and principles of ophthalmology, 2011–2012. San Fransisco (California): American Academy of Ophthalmology; 2011. p. 43–47 [Chapter 2].
- 12.Makdoumi K., Mortensen J., Crafoord S. Corneal cross-linking for the treatment of keratitis. Cataract Refract Surg Today Eur. 2011:34–38. [Google Scholar]
- 13.Ozturk F., Yavas G.F., Kusbeci T. Efficacy of topical caspofungin inexperimental Fusarium keratitis. Cornea. 2007;26:726–728. doi: 10.1097/ICO.0b013e3180553b9d. [DOI] [PubMed] [Google Scholar]
- 15.Martins S.A., Combs J.C., Noguera G. Antimicrobial efficacy of riboflavin/UVA combination (365 nm) in vitro for bacterial and fungal isolates: a potential new treatment for infectious keratitis. Invest Ophthalmol Vis Sci. 2008;49:3402–3408. doi: 10.1167/iovs.07-1592. [DOI] [PubMed] [Google Scholar]
- 16.Martins S.A., Combs J.C., Noguera G. Antimicrobial efficacy of riboflavin/UVA combination (365nm) in vitro for bacterial and fungal isolates: a potential new treatment for infectious keratitis. Invest Ophthalmol Vis Sci. 2008;49:3402–3408. doi: 10.1167/iovs.07-1592. [DOI] [PubMed] [Google Scholar]
- 17.Garduño-Vieyra L, Gonzalez-Sanchez CR, Hernandez-Da Mota SE. Ultraviolet-A light and riboflavin therapy for acanthamoeba keratitis: a case report. Case Rep Ophthalmol 2011; 2:291–295. [DOI] [PMC free article] [PubMed]
- 18.Khan Y.A., Kashiwabuchi R.T., Martins S.A. Riboflavin and ultraviolet light A therapy as an adjuvant treatment for medically refractive acanthamoeba keratitis: report of 3 cases. Ophthalmology. 2010;118:324–331. doi: 10.1016/j.ophtha.2010.06.041. [DOI] [PubMed] [Google Scholar]
- 19.Muller L., Thiel M.A., Kipfer-Kauer A.I., Kaufmann C. Corneal cross-linking as supplementary treatment option in melting keratitis: a case series. Klinische Monatsblatter fur Augenheilkunde. 2012;229:411–415. doi: 10.1055/s-0031-1299420. [DOI] [PubMed] [Google Scholar]
- 20.Makdoumi K., Mortensen J., Sorkhabi O. UVA-riboflavin photochemical therapy of bacterial keratitis: a pilot study. Graefes Arc Clin Exp Ophthalmol. 2012;250:95–102. doi: 10.1007/s00417-011-1754-1. [DOI] [PubMed] [Google Scholar]
- 21.Price M.O., Tenkman L.R., Schrier A. Photoactivated riboflavin treatment of infectious keratitis using collagen cross-linking technology. J Refract Surg. 2012;28:706–713. doi: 10.3928/1081597X-20120921-06. [DOI] [PubMed] [Google Scholar]
- 22.Spoerl E., Mrochen M., Sliney D. Safety of UVA-riboflavin crosslinking of the cornea. Cornea. 2007;26:385–389. doi: 10.1097/ICO.0b013e3180334f78. [DOI] [PubMed] [Google Scholar]
- 23.Berra M., Galperín G., Boscaro G., Zarate J., Tau J., Chiaradia P. Treatment of Acanthamoeba keratitis by corneal cross-linking. Cornea. 2013;32(2):174–178. doi: 10.1097/ICO.0b013e31825cea99. [DOI] [PubMed] [Google Scholar]
- 24.Alio J.L., Abbouda A., Valle D.D., Del Castillo J.M., Fernandez J.A. Corneal cross linking and infectious keratitis: a systematic review with a meta-analysis of reported cases. J Ophthalmic Inflamm Infect. 2013;3(1):47. doi: 10.1186/1869-5760-3-47. [DOI] [PMC free article] [PubMed] [Google Scholar]