Abstract
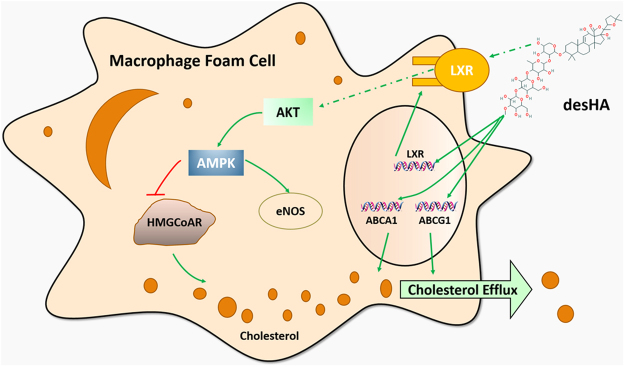
This work was designed to separate and purify the saponin from Thelenota ananas with the highest anti-cholesterol ability using multiple chromatography and mass spectrometry analyses, and to systematically investigate the effect of the Thelenota ananas saponin on cholesterol metabolism in oxidized low-density lipoprotein (ox-LDL) induced macrophage foam cells. Desulfated holothurin A (desHA), which was finally identified as the targeted saponin with the highest activity in decreasing low-density lipoprotein-cholesterol (LDL-C), markedly inhibited the formation of foam cells derived from macrophages based on Oil Red O staining. In addition, desHA significantly blocked the synthesis of fatty acid synthetase while promoted intracellular cholesterol efflux. Furthermore, desHA inhibited the effects of ox-LDL on macrophage mRNA expression, which enhanced the level of 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMG-CoAR) and suppressed the expression of SR-BI, ABCA1 and ABCG1, which further increased the levels of extracellular cholesterol and triglyceride. Blocking AKT and AMPK pathway and LXR synthesis revealed that desHA also regulated the contents of HMG-CoAR and eNOS via LXR/AKT/AMPK pathway. Thus, desHA played an essential role in cholesterol efflux and synthesis, which indicated desHA and Thelenota ananas are valuable resources to exploit new functional food and nutraceuticals.
Introduction
Saponins are the primary glycosylated secondary metabolites found in many major crops and certain sea foods, such as the sea cucumber, starfish and alcyonacea1. Thelenota ananas is an edible cucumber species popular in South China and some Southeast Asian countries, which contains the most abundant saponins both in the body wall and alimentary canal. The common structure of saponins has been identified as combination of triterpenoid or spirosterane sapogenins and oligosaccharides. Notably, active compositions of sea cucumber saponins have been clarified by using silica-gel column chromatography, high performance liquid chromatography (HPLC) and high performance centrifugal partition chromatography (HPCPC) and other newly developed technologies, including holothurin A1 (HA), holothurin B (HB), 24-dehydroechinoside A (DA), echinoside A (EA), argusides, coustesids, holothurinosides, impatiensides, marmoratoside A and philinopsides2–10.
Sea cucumber was the most special marine animal found existing some saponins with a lot of remarkable bioactivities in inhibiting tumor and inflammation, promoting hemolysis, and modulating circadian clock11–16. Moreover, sea cucumber saponins helped regulate cholesterol metabolism and further alleviated the development of obesity, hyperlipidaemia and diabetes, revealing that sea cucumber saponins potentially resistant against atherosclerosis, which is a chronic inflammatory disease induced mainly by lipid accumulation on middle and large vessels17–19. It was reported that saponins prevented atherogenesis mainly by regulating liver X receptor alpha (LXRα), ATP binding cassette transporter A1 (ABCA1) expressions and suppressing 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMG-CoAR) activity and further lowering cholesterol and triglyceride concentration20–22. However, systematic and comprehensive studies are still needed to elucidate the mechanism.
Therefore, the objective of the study was to screen and identify the Thelenota ananas saponin with the highest activity of lowering cholesterol. The target saponin was prepared using differential column chromatography, and the structure was subsequently determined by HPLC-mass spectroscopy (MS). The changes in key signal pathways involved in cholesterol metabolism were then detected to illuminate the bioactivity of sea cucumber saponin in modulating cholesterol metabolism in vitro, which may provide further theoretical support regarding the anti-atherosclerotic role of Thelenota ananas saponin.
Results and Analysis
Purification and identification of Thelenota ananas saponin
Crude Thelenota ananas saponins (29.8 g) were obtained using alcohol-extraction and liquid-liquid extraction, and were qualitatively determined using a foam test, precipitation reaction and Molisch reaction (data not shown). First, two fractions of the saponins were collected via the AB-8 macroporous resin column, i.e., 30% ethanol-eluting portion and 70% ethanol-eluting portion. Both portions decreased the content of LDL-C with the rates of 11.5% and 27.7%, respectively (data not shown). Thus, the 70% ethanol-eluting portion (16.1 g) was continuously purified using a Sephadex LH-20 column to obtain 8 fractions, and Fraction 8 (137.6 mg) exhibited excellent cholesterol decreasing activity (66.7%, data not shown). As fraction 8 was subjected to a semi-preparative HPLC, compound 1 (retention time 11.869 min), compound 2 (retention time 13.998 min), and compound 3 (retention time 16.320 min) were finally acquired, among which compound 3 (25.1 mg) possessed the highest content and anti-cholesterol properties (Supplementary Figure S1). Then, the results of the HPLC-MS analyses revealed that the molecular mass of compound 3 is 1119 Da, which is the same with desulfated holothurin A (NCBI PubChem CID: 3038027, CAS NO. 11060-73-4, desHA). DesHA has been previously detected in sea cucumber Holothuria forskali, also known as nobiliside 2A23. Therefore, it was inferred that the structure of compound 3 was 3-O-[3-O-methyl-β-D-glucopyranosyl-(1 → 3)-β-D-glucopyranosyl-(1 → 4)-β-D-quinovose-(1 → 2)-β-D-xylopyranosyl]-22,25-epoxyholost-9-ene-3β,12α,17α-trihydroxy. According to this, there might be two sequences of fragmentation of compound 3 at m/z 1141.6, showing the consecutive losses of 3-O-methylglucose, aglycone, and glucose-quinovose-xylose sodium or consecutive losses of the aglycone, 3-O-methylglucose, and glucose-quinovose-xylose sodium from the mass-selected compound 3 (Fig. 1). These findings enabled the identification of the m/z 1141.6 ions as desHA.
Figure 1.
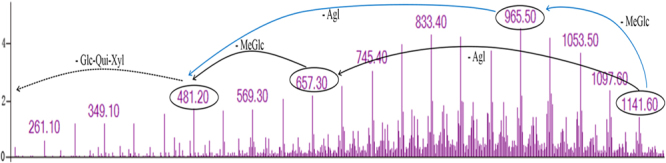
The collision-induced fragmentation patterns of compound 3 separated by semi-preparative HPLC. Blue and Black arrows are two possible fragmentation patterns. Note: Agl is short for Aglycone, MeGlc is short for 3-O-methylglucose, Glc is short for Glucose, Qui is short for Quinovose, Xyl is short for Xylose.
Effect of desHA on the viability of RAW264.7 cells
DesHA imapcted the cell viability of macrophage depended on its concentrations (Fig. 2). Specifically, 0.5 to 25 μg/mL of desHA prepared from Thelenota ananas gradually increased cell proliferation, while higher concentrations of saponin induced a weaker increase in the viability of RAW264.7 cells. Thus, low concentrations of 1, 3 and 5 μg/mL were used in the following experiments.
Figure 2.
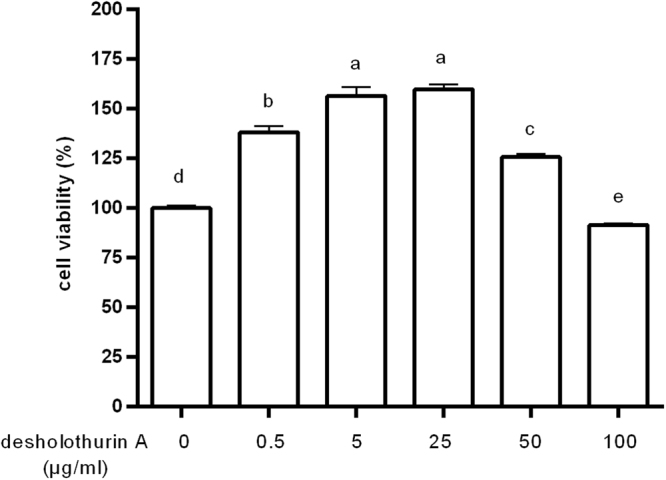
The effect of desHA on RAW264.7 cell viability. Results were presented as mean ± SD of three independent experiments. Values having different superscripts are significantly different, p < 0.05 (same to the below figures).
DesHA inhibited the formation of foam cells derived from macrophage
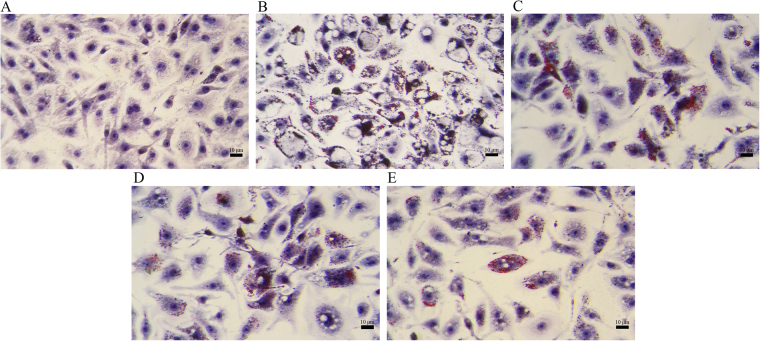
Figures 3A,B illustrate that ox-LDL apparently induced a larger proportion and size of foam cells than those in the control group, which primarily maintained the normal macrophages. However, desHA gradually decreased the lipid intake of macrophages and the formation of foam cells with increasing concentration (Fig. 3C–E).
Figure 3.
The effect of desHA on macrophage-derived foam cells formation with a magnification of ×400. (A) control group, (B) ox-LDL group, (C) ox-LDL + 1 µg/mL desHA group, (D) ox-LDL + 3 µg/mL desHA group, (E) ox-LDL + 5 µg/mL desHA group.
Effect of desHA on fatty acid synthetase (FAS), triglyceride (TG) and total cholesterol (TC)
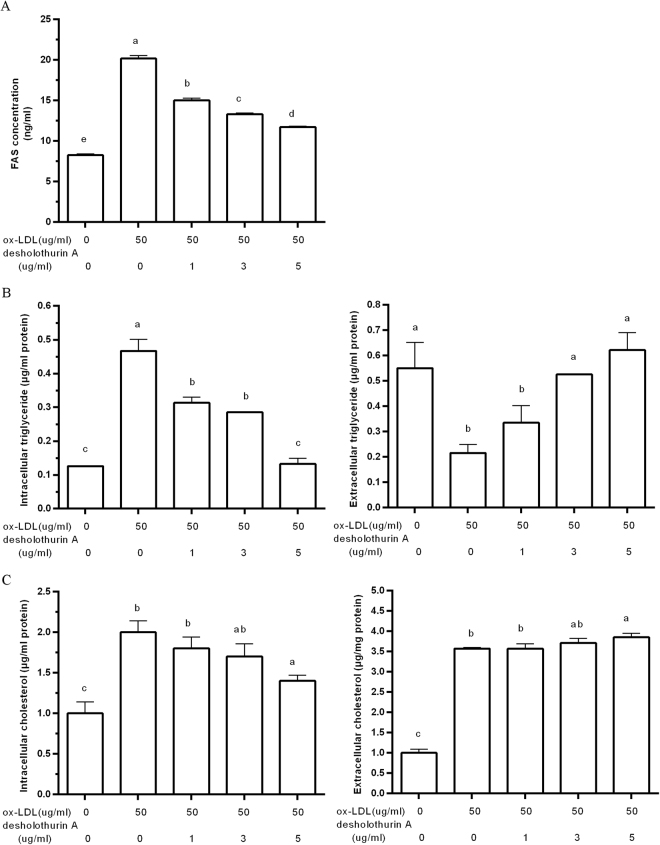
Ox-LDL has been shown to induce the transformation of RAW264.7 cells into foam cells. Thus, we detected the influence of desHA on ox-LDL induced foam cells. DesHA inhibited the content of ox-LDL-induced FAS with a dose-dependent manner (Fig. 4A). Figure 4B,C demonstrate that desHA repressed the intracellular triglyceride and cholesterol levels. Meanwhile, desHA increased the contents of extracellular triglyceride and cholesterol, thereby promoting the efflux of both triglyceride and cholesterol.
Figure 4.
The effect of desHA on the productions of FAS (A), TG (B) and TC (C) in RAW264.7 cells.
Effect of desHA on the expression of HMG-CoAR, SR-BI, ABCA1 and ABCG1
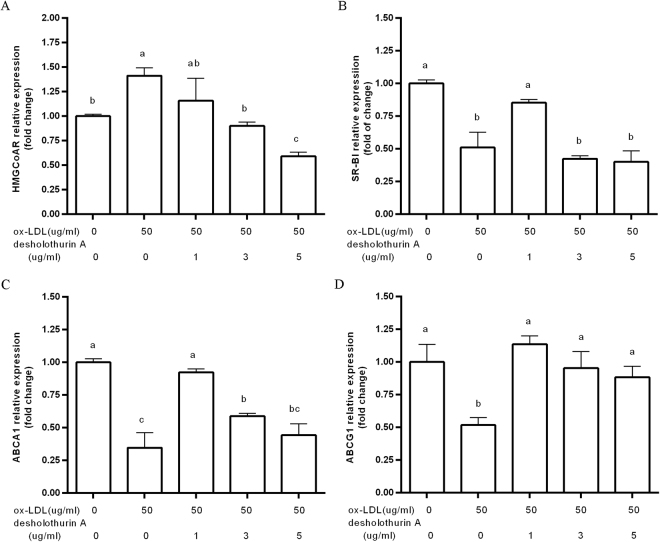
Figure 5 shows that the mRNA expression of HMG-CoAR elevated by 1.5-fold, while the expressions of SR-BI, ABCA1 and ABCG1 were markedly down-regulated by ox-LDL compared with that in the control group. In contrast, desHA significantly reversed the influence of ox-LDL. Specifically, the HMG-CoAR level gradually declined with increased desHA concentration (Fig. 5A). Compared with the ox-LDL treatment, the mRNA expressions of SR-BI, ABCA1 and ABCG1 treated by 1 μg/mL desHA were up-regulated by 2-fold, 2.4-fold and 2.3-fold, respectively (Fig. 5B,C,D).
Figure 5.
The effect of desHA on the expressions of cholesterol metabolism related factors HMG-CoAR (A), SR-BI (B), ABCA1 (C) and ABCG1 (D).
DesHA modulated AKT and AMPK signal pathways via regulating LXRα in RAW264.7
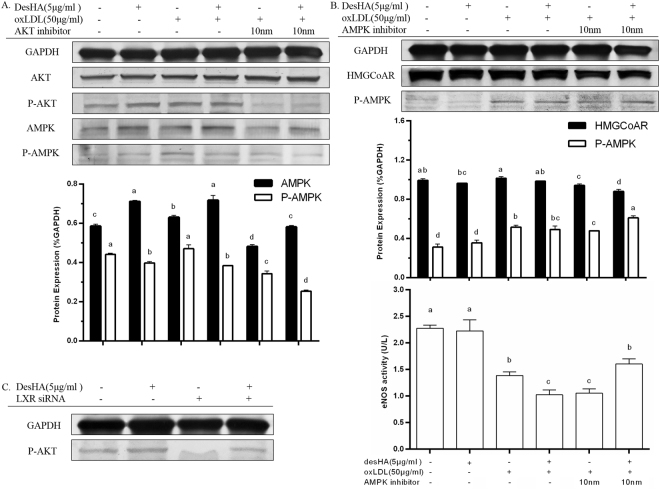
DesHA played an essential role in regulating the phosphorylated proteins of AKT and AMPK in ox-LDL induced macrophage foam cells (Fig. 6A). Obviously, an increment of phosphor-AKT and AMPK accompanied by suppressed phosphor-AMPK were observed under the treatment of desHA with or without ox-LDL.
Figure 6.
The cell signal pathways mediated by desHA in cholesterol metabolism. (A) The changes of AKT, P-AKT, AMPK and P-AMPK levels treated by AKT inhibitor with or without desHA. (B) The changes of P-AMPK and its downstream protein factors treated by AMPK inhibitor with or without desHA. (C) The changes of P-AKT expression treated by LXR siRNA with or without desHA.
Figure 6A also indicates that no significant change of AKT total protein content was observed with addition of AKT inhibitor MK-2206 2HCl, however, phosphor-AKT was almost entirely inhibited by AKT inhibitor and then being restored by desHA. Besides, the expressions of AMPK and phosphor-AMPK were changed when blocking AKT signal pathway, but desHA inhibited the transform of AMPK to phosphor-AMPK with or without the treatment of AKT inhibitor, which showing an important regulation effect of desHA to keep the homeostasis of AMPK content via AKT signal pathway.
Furthermore, the treatment of AMPK inhibitor Dorsomorphin markedly decreased phosphor-AMPK content which was then reversed by desHA (Fig. 6B). It also illustrates that blocking AMPK signal pathway decreased the levels of HMG-CoAR. Meanwhile, desHA suppressed the HMG-CoAR expression with or without the addition of AMPK inhibitor. In addition, the activity of eNOS was suppressed both by Dorsomorphin and desHA in comparison with the one treated by ox-LDL, but desHA finally enhanced the activity of eNOS in cells under the ox-LDL and Dorsomorphin treatment.
Figure 6C demonstrates that LXR promoted AKT activation because little expression of phosphor-AKT was accordingly found by knockdown LXR. However, desHA significantly revised the inhibition of phosphor-AKT. Consequently, desHA might exhibit its decreasing-cholesterol property by modulating LXR/AKT/AMPK pathway.
Discussion
Different extraction methods were used to identify saponins with different structures, and the discovery of new structures of saponins with various bioactivities in the sea cucumber have attracted more attention. In this work, desHA was separated from Thelenota ananas for the first time confirmed by the positive tandem ESI-MS results.
According to the latest statistics, scholars have isolated more than 700 types of sea cucumber saponins from nearly hundreds of species of sea cucumber, including Aspidochirotida: Holothuriidae, Aspidochirotida: Stichopodidae, Dendrochirotida: Cucumariidae24. HA and HB were identified in several sea cucumber Holothuria25. Friess, et al.26 initially discussed the desulfated derivate of HA, namely, holothurin A, desulfo- or desHA, purified from the sea cucumber Actinopyga agassizi Selenka, in terms of the mechanism of the saponin irreversible inactivation of the ganglion and the survival of ganglionic excitability. Subsequently, desHA was detected in Bohaadschia argus, Holothuria forskali, Holothuria leucospilota, Holothuria nobilis and Pearsonothuria graeffei27.
Because of the abundant resources in the ocean, much attention has been paid to exploiting new active substances with extended application in nutraceutical, cosmeceutical, medicinal and pharmaceutical products. Although, other active substances from Thelenota ananas, such as fucoidan, fucosylated chondroitin sulfate, and glycosaminoglycan, have gained more attention than saponins because of their abundant content and multiple functions28–31. Generally, desHA is secreted as a chemical defense when sea cucumbers are stressed32. The degrading-cholesterol effect of desHA has not been discussed thus far. The functions of saponins in Thelenota ananas and other marine animals were also attractive to investigate in depth.
On one hand, the structures of saponins prepared from the sea cucumber are different from those of plant saponins. Lanostane-type triterpene glycosides (e.g. holostane types, including HA and HB), which are the most important secondary metabolites of sea cucumbers, occupied a large proportion of the purified saponins33–35. Particularly, the presence of an acetoxy group plays a significant role in the bioactivity, including the induction of caspase, apoptosis, cytotoxicity, and the anti-cancer, anti-fungal and anti-bacterial activities36. However, few studies regarding the species and bioactivities of saponins from Thelenota ananas have been performed.
On the other hand, several identified saponins have been studied thoroughly to understand the underlying machenism of anti-atherosclerosis effects, including panax notoginseng saponin ginsenoside-Rd37, reinioside C from the the root of Polygala aureocauda Dunn38, buddlejasaponin IV extract of Pleurospermum kamtschaticum39, and sea cucumber saponin EA40. The sulfated holothurin A prepared from Pearsonothuria graeffei was demonstrated to significantly reduce the adipose tissue weight in C57BL/6 mice, thereby exhibited an anti-obesity effect in male Wistar rats9. It seems that both crude extraction of saponin and single saponin mentioned above may decrease the concentrations of plasma cholesterol, triglyceride in animal model, therefore may play an important role in preventing cholesterol dysfunction related diseases. By far, few study concerned on the mechanisms of single saponin working in cell model to further elucidating the anti-atherosclerosis activity of the sea cucumber saponin.
Based on these results, the underlying mechanism of the mediation effects of desHA to cholesterol content was examined in the present study, and the results indicated that desHA also showed excellent bioactivity in repressing the synthesis of cholesterol and promoting intracellular cholesterol efflux by regulating the mRNA expression of HMG-CoAR, SR-BI, ABCA1 and ABCG1. Our results also showed that desHA took part in the regulation of cholesterol metabolism via LXR/AKT/AMPK signaling pathway (Fig. 7), but there would be other signaling pathways to discover in the following study. Furthermore, most of the mechanisms of saponins effect on cholesterol metabolism and atherosclerosis progression was confirmed in cell models and direct proof in animals and human body is still need. To date, there are several mechanisms available to explain the activity of saponins apart from the regulation of cholesterol metabolism and lipase activity this work focused on, including the ability to form insoluble complexes with cholesterol and to interfere with micelle formation and bile acid metabolism41. And this called on researchers to further investigate the cardiovascular protection of saponins in animal models and unearth the comprehensive mechanisms.
Figure 7.
The molecular mechanism of desHA on regulating cholesterol metabolism in macrophage foam cells.
Since saponins exhibit various activities, reflecting the surfactant properties of these compounds, it is necessary to further investigate the relationship between the activity of desHA and its structure in following study.
Overall, Thelenota ananas is a marine animal with a high food and potential medicinal value, and the saponins of this species possess promising multiple biological activities. This work discussed the effects of desHA on lowering cholesterol, showing that desHA is of great interest due to its potential usefulness as a food ingredient, dietary supplement or food additive for preventing certain cardiovascular diseases that are associated with abnormal cholesterol levels.
Materials and Methods
Materials
Thelenota ananas was obtained from Guangxi Simaier Co. Ltd. (Guangxi, China), and generally 5–6 organisms per 500 g were identified as superior dried sea cucumber. AB-8 macroporous resin (Qingdao Marine Chemical Co. Ltd., Qingdao, Shandong, China) and Sephadex LH-20 (GE Healthcare Bio-Sciences AB, Uppsala, Sweden) were used for column chromatography. The chemicals were of analytical grade.
The semi-preparative HPLC separation was performed on a 1260 Infinity HPLC system (Santa Clara, CA) with a C18 column (150 × 10 mm i.d., 120 Å, S-5µm). HPLC-MS analyses were performed using Agilent 6460 liquid chromatography triple quadrupole mass spectrometry (Milford, MA).
Extraction and identification of Thelenota ananas saponins
The procedure was made references to Van Dyck, et al. and Zhu42,43. Dry Thelenota ananas (956 g) were first rehydrated by ultrapure water soaking for two days. After the homogenization, the crude saponins of Thelenota ananas were extracted three times using cold 70% ethanol. The combined and concentrated 70% ethanolic extraction was suspended in deionized water and successively extracted using petroleum ether and n-butanol. The n-butanol portion was concentrated by evaporation under a reduced pressure and dried by vacuum freeze-drying. Subsequently, 0.5 g/mL Thelenota ananas saponins was loaded and purified on a pre-treated AB-8 macroporous resin column (30 × 1000 mm) and successively eluted with 600 mL distilled water, 30% v/v, 70% v/v and 100% v/v ethanol solution at a flow rate of 3 mL/min. After rotating evaporation and freeze-drying, the collected eluent was obtained as a crude Thelenota ananas saponin, and the content of low-density lipoprotein-cholesterol (LDL-C) was determined by cell experiments. The 70% ethanol fraction was isolated on a Sephadex LH-20 column (10 × 500 mm) by isocratic elution with 50% to identify the best fraction with the highest cholesterol-degrading activity, which was further purified on semi-preparative HPLC with a gradient elution comprising acetonitrile and water (2 mL/min, 205 nm). The obtained compound was finally identified by the HPLC-MS analyses using an Agilent mass spectrometer. Mass data were obtained using electrospray ionization in positive ion mode.
Detection of the cholesterol-degrading activity of saponins
The RAW264.7 macrophage cell line (China Infrastructure of Cell Line Resource, Beijing, China) was used to evaluate the effect of saponins on cholesterol metabolism. The cells were incubated in the Gibco® DMEM, containing 10% Gibco® fetal bovine serum (North America, Thermo Fisher Scientific Inc., Waltham, USA), maintained at 37 °C under a humidified 5% CO2 environment. The homogeneous cell suspension (1 × 105/mL) were cultured in a 12 well-culture plate for 24 h. The plate was treated with different fractions of Thelenota ananas saponins (filtration by 0.45 μm filter membrane) for 24 h at 37 °C. The control group was treated with an equal volume of serum-free culture medium. Then, the supernatant was collected and used to detect the content of LDL-C via the LDL-C assay kit (Nanjing Jiancheng Bioengineering Institute, Nanjing, China).
Determination of the cell viability of RAW264.7 treated with Thelenota ananas saponin
The RAW264.7 cell suspension (200 μL 1 × 105 cell/mL) were uniformly inoculated in a 96-well cell culture plate overnight. The cells in the experimental groups were treated with different concentrations of the identified saponin from Thelenota ananas (0.5, 5, 25, 50, 100 μg/mL) for 6 h, and the cells in the control group were incubated in the serum-free culture medium containing 0.1% DMSO, while the blank group included only the cell culture (containing 0.1% DMSO) without cells. Each group was replicated five times. After the treatments, the cells were washed three times with ice-cold PBS, and then, 20 μL of the CCK-8 solution (Beijing Solarbio Science & Technology Co. Ltd., Beijing, China) was added to each well for 3 h. The absorbance was determined at OD570 nm using a Model 680 spectrophotometer.
Determination of macrophage foam cell formation
In total, 1 × 105/mL of RAW264.7 cells were incubated in a 6-well cell culture plate overnight. The experimental groups were stimulated with 50 μg/mL of ox-LDL. (Guangzhou Yiyuan Biotech. Co. Ltd., Guangzhou, China) for 24 h, and the control group was treated with serum-free culture medium containing 0.1% DMSO. Subsequently, the precipitate was removed, and the experimental groups were sequentially treated with or without the identified saponin at the final concentrations of 1, 3, 5 μg/mL for another 6 h. To visualize the cellular neutral lipid accumulation, the cells were fixed in 4% cold paraformaldehyde solution for 15 min, and the intracellular lipid droplet and cytomembrane were separately stained with 1% Oil Red O (in 60% isopropanol) and methylene blue at 37 °C for 10 min. The cell staining of cells was examined at 400 × magnification under an inverted microscope (Diaphot 200, Nikon, Garden City, NY).
Quantitation of FAS, TG and TC influenced by the Thelenota ananas saponin
The experiment was performed as described above. According to the manufacturer’s instruction for the FAS, TG and TC detection kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China), the cell medium was collected by centrifugation, and the cells were lysed by 1% Triton X-100. Both the supernatant and the cells were used to determine the contents of the target indices.
Genes expression involved in cholesterol metabolism influenced by the identified saponin
According to the manufacturer’s instructions for the RNA extraction kit (Tiangen Biotech Co. Ltd., Beijing, China), total RNA were extracted and the first chain of cDNA was synthesized using a reverse kit. Real-time quantitative PCR (RT-PCR) was used to detect the expression of HMG-CoAR, SR-BI, ABCA1 and ABCG1. The expression of the target genes was normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH). The primers were following: GAPDH-F, 5′-AGGCCGGTGCTGAGTATGTC-3′, GAPDH-R, 5′-TGCCTGCTTCACCACCTTCT-3′, HMGR-F, 5′-GCTTGGCCTCCATTGAGAT-3′, HMGR-R, 5′-ATGCATCCGGAAAAGTCTTG-3′, SRBI-F, 5′-AGGGATAGGGTTGGAGTCAGC-3′, SRBI-R, 5′-CGTTGTAATGGAAGCCAGAGG-3′, ABCA1-F, 5′-AAGCCAAGCATCTTCAGTTC-3′, ABCA1-R, 5′-CCATACAGCAAGAGCAGAAGG-3′, ABCG1-F, 5′- ATACAGGGGAAAGGTCTCCAAT-3′, ABCG1-R, 5′-CCCCCGAGGTCTCTCTTATAGT-3′.
Determination of cholesterol-controlling signal transduction pathways by western blot assay
To explore the possible cholesterol-controlling transduction pathway of the identified saponin, cells were pre-incubated with 50 μg/mL ox-LDL for 24 h, and further separately treated with 10 nM MK-2206 2HCl (an inhibitor of AKT pathway), 5 μg/mL Thelenota ananas saponin and the mixture of both for another 24 h. Cells containing serum-free culture medium was set as the control group. Total protein of each treatment group was extracted by the addition of M-PER™ Mammalian Protein Extraction Reagent (Thermo Fisher Scientific Inc., Waltham, USA) and high speed centrifugation at 4 °C. Then protein concentration of the supernatant was determined and adjusted to the same level. The expressions of AKT, phosphor-AKT, AMPK, phosphor-AMPK levels were analyzed by Western Bolt assay. Rabbit polyclonal antibody to GAPDH antibody was set as the internal standard and anti-rabbit secondary antibody at 1:200 were used for immunoblotting analysis. The protein was visualized with a BCIP/NBT Alkaline Phosphatase Color Development Kit (Beyotime, Haimen, China).
In addition, with the same treatment of ox-LDL, cells were co-cultured with 10 nM AMPK inhibitor Dorsomorphin, 5 μg/mL identified saponin and the mixture of both for 24 h. Other treatments were the same as above. The protein levels of phosphor-AMPK and HMG-CoAR were determined subsequently. Besides, the supernatant was collected to detect the activity of eNOS using ELISA Kit (Sangon Biotech, Shanghai, China).
Effect of LXR siRNA on cholesterol efflux regulation
To investigate the effect of LXR siRNA on cholesterol metabolism and related protein expression, the LXR siRNA was transfected into cells by Lipofectamine® RNAiMAX transfection reagent (Thermo Fisher Scientific Inc., Waltham, USA), then the identified saponin was added to the cells. The protein of each group was extracted and phosphor-AKT level was determined by Western blot assay.
Statistics
Significance differences between the means were assessed using One-Way ANOVA (SPSS 19.0, SPSS Inc., Chicago, USA). P < 0.05 was considered statistically significant.
Electronic supplementary material
Acknowledgements
This study was supported by Beijing Advanced Innovation Center for Food Nutrition and Human Health (201502910110942) and the National Nature Science Foundation of China (31401520).
Author Contributions
Qi-an Han designed and performed the experiments and wrote the manuscript. Kaifeng Li assisted to complete the experiments. Xiuping Dong helped a lot on data analyses and manuscript preparation. Beiwei Zhu and Yongkang Luo conceived the experiment and approved the final version of the manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-27932-x.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Yongkang Luo, Email: luoyongkang@263.net.
Beiwei Zhu, Email: zhubeiwei@163.com.
References
- 1.Osbourn, A. E., Wubben, J. P. & Daniels, M. J. Saponin detoxification by phytopathogenic fungi. [Stacey G., Keen N. T. (eds)] Plant-microbe Interactions 2. Chapter 4, 99–124. (Springer, Boston, MA, 1997).
- 2.Li M, et al. Echinoside A, a new marine-derived anticancer saponin, targets topoisomerase2alpha by unique interference with its DNA binding and catalytic cycle. Ann. Oncol. 2010;21:597–607. doi: 10.1093/annonc/mdp335. [DOI] [PubMed] [Google Scholar]
- 3.Yasumoto T, Nakamura K, Hashimoto Y. A new saponin, holothurin B, isolated from sea-cucumber, Holothuria vagabunda and Holothuria lubrica. Agric. Biol. Chem. 1967;31:7–10. doi: 10.1080/00021369.1967.10858767. [DOI] [Google Scholar]
- 4.Tong Y, et al. Philinopside A, a novel marine-derived compound possessing dual anti-angiogenic and anti-tumor effects. Int. J. Cancer. 2005;114:843–853. doi: 10.1002/ijc.20804. [DOI] [PubMed] [Google Scholar]
- 5.Tian F, et al. Philinopside E, a new sulfated saponin from sea cucumber, blocks the interaction between kinase insert domain-containing receptor (KDR) and αvβ3 integrin via binding to the extracellular domain of KDR. Mol. Pharmacol. 2007;72:545–552. doi: 10.1124/mol.107.036350. [DOI] [PubMed] [Google Scholar]
- 6.Zhang SL, Li L, Yi YH, Sun P. Philinopsides E and F, two new sulfated triterpene glycosides from the sea cucumber Pentacta quadrangularis. Nat. Prod. Res. 2006;20:399–407. doi: 10.1080/14786410500185584. [DOI] [PubMed] [Google Scholar]
- 7.Qin Z, et al. Differential effects of sulfated triterpene glycosides, holothurin A1, and 24-dehydroechinoside A, on antimetastasic activity via regulation of the MMP-9 signal pathway. J. Food Sci. 2010;75:280–288. doi: 10.1111/j.1750-3841.2010.01837.x. [DOI] [PubMed] [Google Scholar]
- 8.Elbandy M, Rho JR, Afifi R. Analysis of saponins as bioactive zoochemicals from the marine functional food sea cucumber Bohadschia cousteaui. Eur. Food Res. Technol. 2014;238:937–955. doi: 10.1007/s00217-014-2171-6. [DOI] [Google Scholar]
- 9.Wang Y, et al. Effects of two sulfated triterpene saponins echinoside A and holothurin A on the inhibition of dietary fat absorption and obesity reduction. Biosci. Biotechnol. Biochem. 2014;78:139–146. doi: 10.1080/09168451.2014.877830. [DOI] [PubMed] [Google Scholar]
- 10.Dong P, Xue CH, Du QZ. Separation of two main triterpene glycosides from sea cucumber Pearsonothuria graeffei by high-speed countercurrent chromatography. Acta. Chromatogr. 2008;20:269–276. doi: 10.1556/AChrom.20.2008.2.11. [DOI] [Google Scholar]
- 11.Takada K, Nakao Y, Matsunaga S, van Soest RWM, Fusetani N. Nobiloside, a new neuraminidase inhibitory triterpenoidal saponin from the marine sponge Erylus nobilis. J. Nat. Prod. 2002;65:411–413. doi: 10.1021/np010480n. [DOI] [PubMed] [Google Scholar]
- 12.El Barky AR, Ail E, Mohamed TM. Marine sea cucumber saponins and diabetes. Austin Pancreat Disord. 2017;1:1002–1008. [Google Scholar]
- 13.Su X, Xu C, Li Y, Gao X, Lou Y. Antitumor activity of polysaccharides and Saponin Extracted from Sea Cucumber. J. Clin. Cell. Immunol. 2011;2:105–109. doi: 10.4172/2155-9899.1000105. [DOI] [Google Scholar]
- 14.Soltani M, Parivar K, Baharara J, Kerachian MA, Asili J. Hemolytic and cytotoxic properties of saponin purified from Holothuria leucospilota sea cucumber. Rep. Biochem. Mol. Biol. 2014;3:43–50. [PMC free article] [PubMed] [Google Scholar]
- 15.Wen M, et al. Effects of dietary sea cucumber saponin on the gene expression rhythm involved in circadian clock and lipid metabolism in mice during nighttime-feeding. J. Physiol. Biochem. 2014;70:801–808. doi: 10.1007/s13105-014-0349-9. [DOI] [PubMed] [Google Scholar]
- 16.Soltani M, Baharara J, Namvar F. Evaluation of cytotoxicity and anti-inflammatory effects of saponin isolated from Holothuria leucospilota Sea cucumber. J. Cell. Immunother. 2015;1:17–18. doi: 10.1016/j.jocit.2015.10.019. [DOI] [Google Scholar]
- 17.Hu, X. Q. et al. Effect of dietary saponins of sea cucumber on lipids metabolism in rats. International Conference of National Product and Traditional Medicine 282–286 (2009).
- 18.Xiaoqian HU, et al. Effects of bioactive components of sea cucumber on the serum, liver lipid profile and lipid absorption. Biosci. Biotech. & Bioch. 2012;76:2214–2218. doi: 10.1271/bbb.120495. [DOI] [PubMed] [Google Scholar]
- 19.Guo L, et al. Saponin-enriched sea cucumber extracts exhibit an antiobesity effect through inhibition of pancreatic lipase activity and upregulation of LXR-β signaling. Pharm. Biol. 2015;54:1312–1325. doi: 10.3109/13880209.2015.1075047. [DOI] [PubMed] [Google Scholar]
- 20.Fan JS, et al. Panax notoginseng saponins attenuate atherosclerosis via reciprocal regulation of lipid metabolism and inflammation by inducing liver X receptor alpha expression. J. Ethnopharmacol. 2012;142:732–738. doi: 10.1016/j.jep.2012.05.053. [DOI] [PubMed] [Google Scholar]
- 21.Jia Y, et al. Panax notoginseng saponins decrease cholesterol ester via up-regulating ATP-binding cassette transporter A1 in foam cells. J. Ethnopharmacol. 2010;132:297–302. doi: 10.1016/j.jep.2010.08.033. [DOI] [PubMed] [Google Scholar]
- 22.Wu JH, et al. Suppression of diet-induced hypercholesterolaemia by saponins from Panax notoginseng in rats. J. Funct. Foods. 2013;5:1159–1169. doi: 10.1016/j.jff.2013.03.013. [DOI] [Google Scholar]
- 23.Wu J, Yi YH, Zou ZR, Wu HM, Tang HF. Two new triterpene glycosides from sea cucumber Holothuria nobilis. Chinese Traditional Herbal Drugs. 2006;37:497–500. [Google Scholar]
- 24.Zhang R, et al. Isolation, preparation and analysis of triterpene glycoside Holotoxin A1 in Apostichopus japonicus. Chinese Journal of Marine Drugs. 2013;32:8–14. [Google Scholar]
- 25.Caulier G, Dyck SV, Gerbaux P, Eeckhaut I, Flammang P. Review of saponin diversity in sea cucumbers belonging to the family Holothuriidae. SPC Beche-de-mer Inf. Bull. 2011;31:48–54. [Google Scholar]
- 26.Friess SL, Chanley JD, Hudak WV, Weems HB. Interactions of the echinoderm toxin holothurin A and its desulfated derivative with the cat superior cervical ganglion preparation. Toxicon. 1970;8:211–219. doi: 10.1016/0041-0101(70)90204-7. [DOI] [PubMed] [Google Scholar]
- 27.Bahrami Y, Zhang W, Chataway T, Franco C. Structural elucidation of novel saponins in the sea cucumber Holothuria lessoni. Mar. Drugs. 2014;12:4439–4473. doi: 10.3390/md12084439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wu M, Xu S, Zhao J, Kang H, Ding H. Preparation and characterization of molecular weight fractions of glycosaminoglycan from sea cucumber Thelenata ananas using free radical depolymerization. Carbohyd. Res. 2010;345:649–655. doi: 10.1016/j.carres.2009.11.030. [DOI] [PubMed] [Google Scholar]
- 29.Yu L, et al. Structure elucidation of fucoidan composed of a novel tetrafucose repeating unit from sea cucumber Thelenota ananas. Food Chem. 2014;146:113–119. doi: 10.1016/j.foodchem.2013.09.033. [DOI] [PubMed] [Google Scholar]
- 30.Wu M, et al. Structure and effect of sulfated fucose branches on anticoagulant activity of the fucosylated chondroitin sulfate from sea cucumber Thelenata ananas. Carbohyd. Polym. 2012;87:862–868. doi: 10.1016/j.carbpol.2011.08.082. [DOI] [PubMed] [Google Scholar]
- 31.Wu M, Xu S, Zhao J, Kang H, Ding H. Physicochemical characteristics and anticoagulant activities of low molecular weight fractions by free radical depolymerization of a fucosylated chondroitin sulfate from sea cucumber Thelenota ananas. Food Chem. 2010;122:716–723. doi: 10.1016/j.foodchem.2010.03.042. [DOI] [Google Scholar]
- 32.Van DS, et al. The triterpene glycosides of Holothuria forskali: usefulness and efficiency as a chemical defense mechanism against predatory fish. J. Exp. Biol. 2011;214:1347–1356. doi: 10.1242/jeb.050930. [DOI] [PubMed] [Google Scholar]
- 33.Kitagawa I, et al. Structures of echinoside A and B, two antifungal oligoglycosides from the sea cucumber Actinopyga echinites (Jaeger) Chem. Pharm. Bull. 1980;28:1651–1653. doi: 10.1248/cpb.28.1651. [DOI] [PubMed] [Google Scholar]
- 34.Kitagawa I, Kobayashi M, Inamoto T, Yasuzawa T, Kyogoku Y. The structures of six antifungal oligoglycosides, Stichlorosides A1, A2, B1, B2, C1 and C2 from the sea cucumber Stichopus chloronotus (Brandt) Chem. Pharm. Bull. 1981;29:2387–2391. doi: 10.1248/cpb.29.2387. [DOI] [Google Scholar]
- 35.Zhang SY, Yi YH, Tang HF. Bioactive triterpene glycosides from the sea cucumber Holothuria fuscocinerea. J. Nat. Prod. 2006;69:1492–1495. doi: 10.1021/np060106t. [DOI] [PubMed] [Google Scholar]
- 36.Bahrami Y, Franco C. Acetylated triterpene glycosides and their biological activity from holothuroidea reported in the past six decades. Mar. Drugs. 2016;14:147–184. doi: 10.3390/md14080147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li J, Xie ZZ, Tang YB, Zhou JG, Guan YY. Ginsenoside-Rd, a purified component from panax notoginseng saponins, prevents atherosclerosis in apoE knockout mice. Eur. J. Pharmacol. 2011;652:104–110. doi: 10.1016/j.ejphar.2010.11.017. [DOI] [PubMed] [Google Scholar]
- 38.Li H, Wang QJ, Zhu DN, Yang Y. Reinioside C, a triterpene saponin of Polygala aureocauda Dunn, exerts hypolipidemic effect on hyperlipidemic mice. Phytother. Res. 2008;22:159–164. doi: 10.1002/ptr.2262. [DOI] [PubMed] [Google Scholar]
- 39.Jung HJ, et al. The MeOH extract of Pleurospermum kamtschaticum and its active component buddlejasaponin (IV) inhibits intrinsic and extrinsic hyperlipidemia and hypercholesterolemia in the rat. J. Ethnopharmacol. 2007;112:255–261. doi: 10.1016/j.jep.2007.03.012. [DOI] [PubMed] [Google Scholar]
- 40.Wen M, et al. Sea cucumber saponin echinoside A (EA) stimulates hepatic fatty acid β-oxidation and suppresses fatty acid biosynthesis coupling in a diurnal pattern. J. Nutr. Sci. Vitaminol. 2016;62:170–177. doi: 10.3177/jnsv.62.170. [DOI] [PubMed] [Google Scholar]
- 41.Zhao D. Challenges associated with elucidating the mechanisms of the hypocholesterolaemic activity of saponins. J. Funct. Foods. 2016;23:52–65. doi: 10.1016/j.jff.2016.02.023. [DOI] [Google Scholar]
- 42.Van Dyck S, Gerbaux P, Flammang P. Elucidation of molecular diversity and body distribution of saponins in the sea cucumber holothuria forskali (echinodermata) by mass spectrometry. Comp. Biochem. Phys. B. 2009;152:124–134. doi: 10.1016/j.cbpb.2008.10.011. [DOI] [PubMed] [Google Scholar]
- 43.Zhu, B. W. Research on theory and technology of precious seafood processing. Chapter 1, 86. (Science Press, Beijing, China, 2010).
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.