Abstract
Background
The mechanism of inappropriate sinus tachycardia (IST) remains incompletely understood.
Methods and Results
We prospectively compared 3 patient groups: 11 patients with IST (IST Group), 9 control patients administered isoproterenol (Isuprel Group), and 15 patients with cristae terminalis atrial tachycardia (AT Group). P‐wave amplitude in lead II and PR interval were measured at a lower and higher heart rate (HR1 and HR2, respectively). P‐wave amplitude increased significantly with the increase in HR in the IST Group (0.16±0.07 mV at HR1=97±12 beats per minute versus 0.21±0.08 mV at HR2=135±21 beats per minute, P=0.001). The average increase in P‐wave amplitude in the IST Group was similar to the Isuprel Group (P=0.26). PR interval significantly shortened with the increases in HR in the IST Group (146±15 ms at HR1 versus 128±16 ms at HR2, P<0.001). A similar decrease in the PR interval was noted in the Isuprel Group (P=0.6). In contrast, patients in the atrial tachycardia Group experienced PR lengthening during atrial tachycardia when compared with baseline normal sinus rhythm (153±25 ms at HR1=78±17 beats per minute versus 179±29 ms at HR2=140±28 beats per minute, P<0.01).
Conclusions
We have shown that HR increases in patients with IST were associated with an increase in P‐wave amplitude in lead II and PR shortening similar to what is seen in healthy controls following isoproterenol infusion. The increase in P‐wave amplitude and absence of PR lengthening in IST support an extrinsic mechanism consistent with a state of sympatho‐excitation with cephalic shift in sinus node activation and enhanced atrioventricular nodal conduction.
Keywords: atrial tachycardia, atrio‐ventricular conduction, inappropriate sinus tachycardia
Subject Categories: Arrhythmias, Electrophysiology
Clinical Perspective
What Is New?
The mechanism of inappropriate sinus tachycardia (IST) remains incompletely understood.
We prospectively measured P‐wave amplitude in lead II and PR interval at a slower and faster heart rate in 11 patients with IST, 9 control patients who were administered isoproterenol, and 15 patients with cristae atrial tachycardia (PR interval only).
Heart rate increases in patients with IST were associated with an increase in P‐wave amplitude and PR shortening similar to what is seen in the control group following isoproterenol infusion. PR lengthening was noted in the atrial tachycardia group.
Our findings support an extrinsic mechanism in patients with IST consistent with cephalic shift in sinus node activation and enhanced atrioventricular nodal conduction.
What Are the Clinical Implications?
The differential diagnosis in patients with palpitations and ECG findings consistent with a sinus mechanism includes IST versus focal atrial tachycardia originating from the high cristae terminalis.
The absence of an increase in P‐wave amplitude in the inferior leads and PR shortening during tachycardia when compared with slower heart rate strongly argues against IST and favors atrial tachycardia originating from the high cristae terminalis as the underlying mechanism.
The sensitivity and specificity of these findings remain to be determined.
The syndrome of inappropriate sinus tachycardia (IST) is defined as a sinus heart rate (HR) >100 beats per minute (bpm) at rest not caused by primary causes and is associated with symptoms of palpitations, fatigue, and poor exercise tolerance.1 Patients with IST frequently have a mean 24‐hour HR >90 bpm during Holter monitoring with large increases in HR following minimal exertion, emotional stress, or isoproterenol infusion. The mechanisms of IST are not completely understood but are thought to be caused by either extrinsic causes (increased sympathetic activity, decreased parasympathetic activity, impaired neurohumoral modulation) or intrinsic causes (increased sinus node automaticity or β‐adrenergic hypersensitivity).2, 3
The normal behavior of the sinus node with increased sympathetic drive is a gradual increase in HR associated with an increase in P‐wave amplitude in the inferior leads as the impulse travels superiorly along the cristae terminalis (CT).4 The increase in sympathetic drive also results in shortening of the PR interval because of enhanced atrioventricular nodal conduction. The opposite is seen during focal atrial tachycardia (AT) where, regardless of the site of origin, the P‐wave amplitude remains unchanged at different rates, and the PR interval is prolonged relative to the PR interval during sinus rhythm because of the decremental conduction properties of the atrioventricular node. Furthermore, in patients with preserved left ventricular function, AT rates <160 bpm often result in an increase in cardiac output and blood pressure, leading to a reflex decrease in sympathetic activity and further slowing of atrioventricular nodal conduction.5
The purpose of this study was to assess the changes in P‐wave amplitude and PR interval in a group of patients with IST when compared with (1) a control group of patients following the administration of isoproterenol and (2) a group of patients with focal AT originating from the CT. If the mechanism of IST is extrinsic, one would expect the changes in P‐wave amplitude and PR interval to mirror what is seen in healthy controls following isoproterenol infusion (ie, increase in P‐wave amplitude in the inferior leads and shortening in the PR interval). If the mechanism of IST is intrinsic because of a focal increase in automaticity, one would expect to see no significant changes in P‐wave amplitude and PR lengthening at faster rates, similar to what is seen in patients with focal AT.
Methods
The data, analytic methods, and study materials will not be made available to other researchers for purposes of reproducing the results or replicating the procedure as this was not part of our original Institutional Review Board approval.
Study Design and Patient Selection
This was a prospective study conducted between 2013 and 2017 at the University of Wisconsin Hospitals and Clinics in Madison, Wisconsin, and the Ospedali del Tigullio in Lavagna, Italy and Arcispedale S. Maria Nuova, Reggio Emilia, Italy. The study was approved by the institutional review boards at the respective institutions. Informed consent was obtained from all patients and all procedures were conducted in accordance with institutional guidelines.
We enrolled 11 consecutive patients referred for further evaluation of IST (IST Group). The diagnosis of IST was based on (1) history of palpitations and exercise intolerance, (2) resting HR >100 bpm or average HR >90 bpm on 24‐hour Holter monitoring with a P‐wave morphology consistent with a sinus mechanism, and (3) exclusion of other causes such as hyperthyroidism, drug abuse, and orthostatic hypotension. Two ECGs were obtained from all 11 patients with IST: 1 with a resting HR ≈100 bpm (HR1‐IST Group) and another with a HR >120 bpm (HR2‐IST Group). Using electronic calipers, a blinded observer measured the P‐wave amplitude and PR interval in lead II during baseline (HR1‐IST Group) and tachycardia (HR2‐IST Group). In 4 patients, an electrophysiologic study (EPS) was performed to exclude high cristae AT. The diagnosis of IST was confirmed in all 4 patients. During EPS, assessment of atrioventricular nodal conduction was performed including atrioventricular node block cycle length and atrioventricular node effective refractory period at different cycle lengths.
During the same time period, we also enrolled 9 consecutive patients, age ≥18 years, referred for EPS for the evaluation of palpitations or supraventricular tachycardia (Isuprel Group). Patients were excluded if they had anterograde accessory pathway conduction or dual atrioventricular nodal physiology, history of pacemaker or implantable cardioverter‐defibrillator implantation, first‐degree atrioventricular delay (baseline PR >200 ms), or left ventricular dysfunction (LVEF <50%). Patients on atrioventricular nodal‐slowing agents had their medications discontinued 5 half‐lives before the EPS. Isoproterenol was administered and titrated to achieve a target HR of 140 bpm. The P‐wave amplitude and PR interval in lead II were measured at baseline (HR1‐Isuprel Group) and following isoproterenol infusion (HR2‐Isuprel Group).
For comparison, we included 15 consecutive patients identified over the same time period with a focal AT originating from the high CT (AT Group). The diagnosis of focal AT was made according to established criteria.6 The P‐wave morphology during AT required a negative or a biphasic (+/−) polarity P wave in lead V1, positive in leads I and II, and negative in aVR. Furthermore, the localization was confirmed by activation mapping at the time of EPS. Patients who required isoproterenol for AT induction were excluded. The PR interval was measured during normal sinus rhythm (HR1‐AT Group) and during atrial tachycardia (HR2‐AT Group) in the absence of isoproterenol infusion. P‐wave changes were not assessed in the AT group since this is determined by the AT site of origin.
Statistical Analysis
All data were reported as mean±SD for continuous variables and as frequencies for categorical data. Continuous variables were compared using unpaired or paired 2‐tail t tests as appropriate. The Fisher exact test was used to compare proportions. Statistical analysis was performed with Microsoft Excel (Microsoft Corp, Redmond, WA).
Results
Baseline Characteristics
The baseline characteristics of the study population are shown in the Table. The patients with IST were younger when compared with patients in the Isuprel and AT Groups (33±14 years old in the IST Group versus 44±16 and 51±18 in the Isuprel and AT Groups, P=0.11 and 0.01, respectively). There were no significant clinical differences among the groups. The resting HR was significantly higher in the IST Group when compared with the Isuprel and AT Groups (97±12 bpm in the IST Group versus 74±8 and 75±15 bpm in the Isuprel and AT Groups, P<0.01). On Holter monitoring, the mean HR in the IST Group was 100±22 bpm with a minimum HR of 70±20 bpm and a maximum HR of 156±22 bpm. The baseline electrophysiologic measurements including atrioventricular node block cycle length and atrioventricular node effective refractory period were not different in the 4 patients with IST who underwent EPS when compared with patients in the Isuprel and AT Groups.
Table 1.
Baseline Patient Characteristics
| Demographics | IST Group | Isuprel Group | AT Group |
|---|---|---|---|
| n=11 | n=9 | n=15 | |
| Age, y | 33±14 | 44±16 | 51±18a |
| Female, n (%) | 9 (82) | 7 (78) | 10 (67) |
| Hypertension, n (%) | 1 (9) | 1 (11) | 3 (20) |
| Diabetes mellitus, n (%) | 0 (0) | 0 (0) | 1 (7) |
| BMI, kg/m2 | 26±6 | 28±5 | 25±4 |
| LVEF, % | 66±5 | 62±4 | 60±6a |
| LA size, mm | 32±4 | 33±4 | 36±3a |
| LVEDD, mm | 41±6 | 44±5 | 47±7a |
| Sinus rate, bpm | 97±12 | 71±11a | 75±15a |
| PR interval, ms | 146±15 | 158±22 | 157±27 |
| QRS interval, ms | 84±11 | 94±13 | 90±16 |
| QT interval, ms | 355±27 | 404±27 | 390±38 |
| QTc interval, ms | 465±60 | 448±29 | 432±46 |
| EPS baseline CL, ms | 578±71 | 816±128a | 800±193a |
| AH interval, ms | 59±20 | 79±24 | 86±25 |
| AV WCL, ms | 298±42 | 351±64 | 344±52 |
| AVNERP, ms | 250±41 | 310±49 | 275±46 |
Values are mean±SD for continuous data and n (%) for categorical data. AH, atrial‐His interval; ANVERP, AV nodal effective refractory period at a drive cycle length of 600 ms; AV, atrioventricular; AT, atrial tachycardia; BMI, body mass index; bpm, beats per minute; CL, cycle length; EPS, electrophysiology study; IST, inappropriate sinus tachycardia; LA, left atrial; LVEDD, left ventricular end‐diastolic diameter; LVEF, left ventricular ejection fraction.
P<0.05 vs IST group.
P‐Wave Amplitude and PR Changes in the IST Group Compared With the Isuprel Group
The P‐wave amplitude (lead II) increased significantly with the increase in HR in the IST Group (0.16±0.07 mV at HR1=97±12 bpm versus 0.21±0.08 mV at HR2=135±21 bpm, P=0.001). Similarly, there was a significant increase in the P‐wave amplitude in the Isuprel Group following Isuprel infusion (0.14±0.03 mV at HR1=74±8 bpm versus 0.20±0.05 mV at HR2=123±12 bpm, P=0.02). The average increase in P‐wave amplitude in the IST and Isuprel Groups were similar (0.05 mV versus 0.07 mV, respectively, P=0.26).
The PR interval significantly shortened as the HR increased in the IST Group (146±15 ms at HR1=97±12 bpm versus 128±16 ms at HR2=135±21 bpm, P<0.001). A similar shortening in the PR interval was noted in the Isuprel Group as the HR increased (158±22 ms at HR1=74±8 bpm versus 136±16 ms at HR2=123±12 bpm, P<0.01). Again, the average shortening in PR interval in the 2 groups was similar (18±10 ms in the IST Group and 22±22 ms in the Isuprel Group, P=0.6).
PR Changes in the IST Group Compared With the AT Group
As described above, the PR interval shortened with the increases in HR in all patients in the IST Group (146±15 ms at HR1=97±12 bpm versus 128±16 ms at HR2=135±21 bpm, P<0.001). In contrast, patients in the AT Group experienced PR lengthening during AT when compared with baseline normal sinus rhythm. Specifically, the PR interval increased from 153±25 ms at HR1=78±17 bpm to 179±29 ms at HR2=140±28 bpm (P<0.01). The PR interval changes as the HR increased were significantly different between the 2 groups. PR interval shortened by an average of 18±10 ms in the IST Group, while it lengthened by an average of 21±30 ms in the AT Group (P<0.001).
The PR intervals at lower and higher HR in each group are shown in Figure 1. Representative tracings of the P‐wave amplitude and PR intervals associated with the changes in HR in each of the 3 groups are provided in Figure 2.
Figure 1.

PR intervals during lower and higher heart rate (HR) in patients with inappropriate sinus tachycardia (IST Group, left), healthy controls at baseline and following isoproterenol infusion (Isuprel Group, middle), and sinus rhythm and atrial tachycardia (AT Group, right). The PR interval significantly shortened with the increases in HR in the IST Group, with a similar decrease noted in the Isuprel Group. In contrast, patients in the AT Group experienced PR lengthening during AT when compared with baseline normal sinus rhythm (*P<0.05, lower HR vs higher HR within each group). The larger dots indicate sample means. Bars represent 1 SD.
Figure 2.
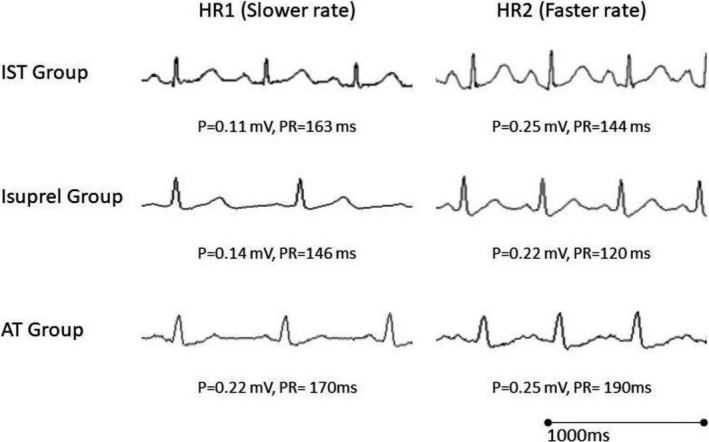
Sample tracings showing changes in P‐wave amplitude and PR interval in (1) a patient with inappropriate sinus tachycardia (top tracings), (2) a healthy control patient before and after isoproterenol infusion (middle tracings), and (3) a patient during sinus rhythm and high cristae atrial tachycardia (lower tracings). Note the increase in P‐wave amplitude in lead II and PR shortening at faster rates when compared with slower rates in the patient with inappropriate sinus tachycardia (IST) and healthy control following isoproterenol infusion. In the patient with atrial tachycardia (AT), the PR interval increased at faster rates when compared with slower rates.
Discussion
We have shown that HR increases in patients with IST were associated with an increase in P‐wave amplitude in lead II similar to that which is seen in healthy controls following isoproterenol infusion. Furthermore, we have shown that, unlike patients with focal AT, the PR interval shortened in patients with IST at faster HR when compared with baseline. The PR interval shortening was similar to what was seen in patients following isoproterenol infusion. The present findings support an extrinsic mechanism for IST consistent with a state of increased sympatho‐excitation, resulting in a cephalic shift in sinus node activation and enhanced atrioventricular nodal conduction. The absence of PR lengthening strongly argues against increased automaticity as the primary underlying mechanism, as the latter would be expected to be associated with PR lengthening as seen with focal AT. To the best of our knowledge, this is the first article to describe the effects of changes in HR on atrioventricular conduction in patients with IST.
Previous reports have suggested that IST could be caused by primary sinus node abnormalities.7, 8 Morillo and colleagues assessed intrinsic HR using pharmacological interventions in 6 patients with IST. Compared with age‐ and sex‐matched controls, the authors reported a higher intrinsic HR in all patients with IST. Enhanced sinus node automaticity was thought to be potentiated by β‐adrenergic receptor hypersensitivity and impaired response to vagal stimulation.9, 10 Chiale and colleagues reported the presence of β‐adrenergic receptor auto‐antibodies in 52% of patients with IST. These auto‐antibodies are capable of stimulating β‐receptor‐dependent production of cAMP, which in turn could lead to increases in HR.11 Still and colleagues hypothesized another mechanism involving an impairment of the acetylcholine‐ and adenosine‐mediated pathways.12 In 18 patients with IST, the authors demonstrated impairment of the negative chronotropic response commonly seen following adenosine administration. Lastly, Baruscotti and his team recently reported a gain‐of‐function mutation in the gene encoding the pacemaker HCN4 channel, leading to increased sensitivity to adrenergic input and cAMP‐dependent activation.13
Unlike the previous studies, our results strongly suggest an extrinsic mechanism in patients with IST. If the rate increase was caused by increased automaticity, the P‐wave amplitude should remain the same since the site of activation has not changed. An increase in P‐wave amplitude with faster HR suggests a cephalic shift on the CT where sinus nodal cells have a higher rate of automaticity.4 While increased automaticity along the entire CT with preserved cephalic shift is a plausible hypothesis, our PR interval findings argue against it. Indeed, focal increases in automaticity, regardless of location along the CT, should result in PR lengthening because of decrement in the atrioventricular node and a likely reflex decrease in sympathetic activity secondary to the increase in cardiac output and input to the arterial baroreceptors.5 In our study, we saw PR shortening instead of PR lengthening in every patient with IST. In contrast, most patients with AT had PR lengthening.
We do not think the absence of PR lengthening in the IST Group is because of the concomitant presence of enhanced atrioventricular nodal conduction. Our limited EPS findings revealed no differences in atrioventricular conduction among the 3 groups. Furthermore, rapid atrial pacing resulted in PR lengthening in the IST Group, suggesting the presence of normal atrioventricular conduction. While we did not measure changes in intra‐atrial activation time associated with the changes in earliest activation site, a cephalic shift along the CT should result in an increase in intra‐atrial conduction time, which renders our findings of PR shortening in the IST Group even more relevant.
In summary, our findings of increased P‐wave amplitude and PR shortening at faster HRs in the IST group favor an extrinsic mechanism consistent with a state of sympatho‐excitation rather than an intrinsic mechanism.
Limitations
First, we did not image the CT in patients with IST and therefore could not comment on the HR relative to CT location among the groups (ie, whether at any given location along the CT, the HR was similar or higher in the IST Group compared with the Isuprel Group). We did not think the deployment of a mapping system was justifiable in the IST Group in the absence of arrhythmia induction. Furthermore, a large control group would have been required to assess the normal variability in HR along the CT. Second, we did not measure blood levels of catecholamines or perform autonomic testing to support or refute our conclusion that the mechanism of IST is likely extrinsic. While such information would have been helpful, this was not the purpose of the study. Third, we used isoproterenol for cardiac stimulation. Other methods of endogenous sympathetic excitation such as mental stress and cold pressor test might have yielded different results. Indeed, mental stress has been shown to increase HR, blood pressure, and peripheral sympathetic activity,14 whereas isoproterenol infusion results in increases in HR but reduction in blood pressure because of its peripheral β‐mediated vasodilation. Fourth, the P‐wave and PR measurements were obtained from only 1 lead, namely, lead II. This lead was selected because the tracings with HR >120 bpm in patients with IST were frequently obtained from the event monitor where only 1 inferior lead was available (lead II). Fifth, the number of subjects enrolled in each group was relatively small and therefore additional validation in a larger cohort of subjects is needed. However, the differences in each group were statistically significant and consistent among the subjects.
Clinical Implications
Our results could be helpful in the evaluation of patients presenting with palpitations and ECG findings of tachycardia consistent with a sinus mechanism, where the differential diagnosis includes IST versus focal AT originating from the high CT. The presence of an increase in P‐wave amplitude in the inferior leads and PR shortening during tachycardia when compared with lower HR strongly favors IST as the underlying mechanism. Conversely, the absence of an increase in P‐wave amplitude in the inferior leads and the presence of PR lengthening during tachycardia strongly favor AT originating from the CT as the underlying mechanism. The sensitivity and specificity of these findings remain to be determined.
Sources of Funding
This work was supported in part by funds provided from the Mildred and Marv Conney Chair in Cardiology.
Disclosures
None.
(J Am Heart Assoc. 2018;7:e008528 DOI: 10.1161/JAHA.118.008528.)29674334
References
- 1. Sheldon RS, Grubb BP II, Olshansky B, Shen WK, Calkins H, Brignole M, Raj SR, Krahn AD, Morillo CA, Stewart JM, Sutton R, Sandroni P, Friday KJ, Hachul DT, Cohen MI, Lau DH, Mayuga KA, Moak JP, Sandhu RK, Kanjwal K. 2015 Heart Rhythm Society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm. 2015;12:e41–e63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Olshansky B, Sullivan RM. Inappropriate sinus tachycardia. J Am Coll Cardiol. 2013;61:793–801. [DOI] [PubMed] [Google Scholar]
- 3. Pellegrini CN, Scheinman MM. Epidemiology and definition of inappropriate sinus tachycardia. J Interv Card Electrophysiol. 2016;46:29–32. DOI: 10.1007/s10840‐015‐0039‐8. [DOI] [PubMed] [Google Scholar]
- 4. Lee RJ, Shinbane JS. Inappropriate sinus tachycardia. Diagnosis and treatment. Cardiol Clin. 1997;15:599–605. [DOI] [PubMed] [Google Scholar]
- 5. Segerson NM, Wasmund SL, Daccarett M, Fabela ML, Hammond CH, Stoddard G, Smith ML, Hamdan MH. The acute effect of atrioventricular pacing on sympathetic nerve activity in patients with normal and depressed left ventricular function. Am J Physiol Heart Circ Physiol. 2008;295:H1076–H1080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Saoudi N, Cosio F, Waldo A, Chen SA, Iesaka Y, Lesh M, Saksena S, Salerno J, Schoels W. A classification of atrial flutter and regular atrial tachycardia according to electrophysiological mechanisms and anatomical bases; a Statement from a Joint Expert Group from the Working Group of Arrhythmias of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Eur Heart J. 2001;22:1162–1182. [DOI] [PubMed] [Google Scholar]
- 7. Bauernfeind RA, Amat YLF, Dhingra RC, Kehoe R, Wyndham C, Rosen KM. Chronic nonparoxysmal sinus tachycardia in otherwise healthy persons. Ann Intern Med. 1979;91:702–710. [DOI] [PubMed] [Google Scholar]
- 8. Morillo CA, Klein GJ, Thakur RK, Li H, Zardini M, Yee R. Mechanism of ‘inappropriate’ sinus tachycardia. Role of sympathovagal balance. Circulation. 1994;90:873–877. [DOI] [PubMed] [Google Scholar]
- 9. Leon H, Guzman JC, Kuusela T, Dillenburg R, Kamath M, Morillo CA. Impaired baroreflex gain in patients with inappropriate sinus tachycardia. J Cardiovasc Electrophysiol. 2005;16:64–68. [DOI] [PubMed] [Google Scholar]
- 10. Castellanos A, Moleiro F, Chakko S, Acosta H, Huikuri H, Mitrani RD, Myerburg RJ. Heart rate variability in inappropriate sinus tachycardia. Am J Cardiol. 1998;82:531–534. [DOI] [PubMed] [Google Scholar]
- 11. Chiale PA, Garro HA, Schmidberg J, Sanchez RA, Acunzo RS, Lago M, Levy G, Levin M. Inappropriate sinus tachycardia may be related to an immunologic disorder involving cardiac beta adrenergic receptors. Heart Rhythm. 2006;3:1182–1186. [DOI] [PubMed] [Google Scholar]
- 12. Still AM, Huikuri HV, Airaksinen KE, Koistinen MJ, Kettunen R, Hartikainen J, Mitrani RD, Castellanos A, Myerburg RJ, Raatikainen MJ. Impaired negative chronotropic response to adenosine in patients with inappropriate sinus tachycardia. J Cardiovasc Electrophysiol. 2002;13:557–562. [DOI] [PubMed] [Google Scholar]
- 13. Baruscotti M, Bucchi A, Milanesi R, Paina M, Barbuti A, Gnecchi‐Ruscone T, Bianco E, Vitali‐Serdoz L, Cappato R, DiFrancesco D. A gain‐of‐function mutation in the cardiac pacemaker HCN4 channel increasing cAMP sensitivity is associated with familial inappropriate sinus tachycardia. Eur Heart J. 2017;38:280–288. [DOI] [PubMed] [Google Scholar]
- 14. Wallin BG, Esler M, Dorward P, Eisenhofer G, Ferrier C, Westerman R, Jennings G. Simultaneous measurements of cardiac noradrenaline spillover and sympathetic outflow to skeletal muscle in humans. J Physiol. 1992;453:45–58. [DOI] [PMC free article] [PubMed] [Google Scholar]


