Abstract
Objective. The aim of this meta-analysis was to evaluate the effectiveness of oral Chinese herbal medicine (OCHM) combined with pharmacotherapy for menopausal depression. Methods. The electronic databases were searched from their inception to December 25, 2016, comprising PubMed, Embase, Cochrane Central Register of Controlled Trials, Chinese National Knowledge Infrastructure (CNKI), Wanfang database, and Chinese Biomedical (CBM) database. Randomized controlled trials investigating the effectiveness of OCHM combined with pharmacotherapy for the people with menopausal depression were eligible. Risk of bias was evaluated according to the Cochrane handbook. Meta-analyses were performed to pool the effect size. Heterogeneity and publication bias were also examined. Results. Twenty-two RCTs with 1770 participants were included in the review. None of the studies used placebo as the control and the risk of bias was high in blinding the participants and personnel. Overall, the meta-analysis demonstrated that adjuvant therapy of OCHM was effective in reducing the Hamilton Rating Scale for Depression (HAMD) scores compared to pharmacotherapy (MD = −3.75; 95% CI = −5.22, −2.29; P < 0.00001). The meta-analysis also suggested that OCHM adjuvant therapy for menopausal depression was superior to pharmacotherapy in terms of response rate of reducing HAMD scores (RR = 1.17; 95% CI = 1.10, 1.25; I2 = 55%). Conclusions. OCHM may provide additional effectiveness to pharmacotherapy for the people with menopausal depression. RCTs including the placebo control were required to further determine the additional efficacy of OCHM for menopausal depression.
1. Introduction
Depression is the most common mental disorder in menopausal women, with a prevalence ranging from 26% to 41.8% [1–5]. Menopause represents a significant transition in the life of a woman, and it has been considered as a specific window of vulnerability to depression [6, 7]. Women with menopausal depression are related to decreased quality of life, increased cardiovascular disease, and metabolic syndrome [8–11].
Antidepressants are considered the most common pharmacotherapy specifically for menopausal depression. However, the effect of antidepressants is unsatisfactory clinically, and the long-term use leads to considerable adverse effects. For example, selective serotonin reuptake inhibitors (SSRIs) are associated with sexual dysfunction, weight gain, and sleep disturbance [12–14]. Hormone therapy (HT) is widely used to alleviate menopause-related symptoms, and it has also been used in the treatment of menopausal depression [15, 16]. However, its effectiveness was inclusive [17]. Therefore, alternative therapies with more benefits and fewer harms were in continuous demand.
Traditional Chinese medicine (TCM) is one of the oldest medicine systems in the world and has been widely used as a form of complementary and alternative medicine [18, 19]. In China, oral Chinese herbal medicine (OCHM) is commonly used in combination with pharmacotherapy for menopausal depression and the associated clinical studies have been conducted [20, 21]. However, most of the clinical studies were of insufficient sample size and of varied quality of methodological design. Systematic reviews on OCHM for depression [22, 23] and menopausal-related symptoms have already been published [24]. However, previous systematic reviews did not specifically evaluate the effectiveness of OCHM as an adjunctive therapy to pharmacotherapy for menopausal depression.
This systematic review was motivated by the large number of published clinical trials on OCHM combined with pharmacotherapy and the unresolved problems of pharmacotherapy. Our objective was to evaluate the effectiveness of OCHM combined with pharmacotherapy for menopausal depression.
2. Materials and Methods
2.1. Search Strategy
We conducted and reported the systematic review according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement guidelines [25]. The electronic databases were searched from their inception to December 25, 2016, comprising PubMed, EMBASE, Cochrane Central Register of Controlled Trials, Chinese National Knowledge Infrastructure (CNKI), Wan Fang Database, Chinese Scientific Journal Database (VIP), and Chinese Biomedical (CBM) database. The combination of search terms was presence of menopause, depression, traditional Chinese medicine, and randomized controlled trial (RCT). There was no limit to the language or publication status. The search strategy is attached in the Appendix.
2.2. Eligibility Criteria
Types of Studies. Only randomized controlled trials were eligible.
Types of Participants. Menopausal women aged 40 to 60 years with depressive symptoms were eligible. Depression was diagnosed according to the Classification and Diagnosis of Mental Diseases (CCMD-3) (Psychiatry Branch of Chinese Medical Association) [26], the Diagnostic and Statistical Manual (DSM-IV) (American Psychiatric Association) [27, 28], and the International Classification of Disease (ICD-10, WHO) (World Health Organization) [29].
Types of Intervention and Control. Studies that compared OCHM plus pharmacotherapy to the same pharmacotherapy were included. All forms of OCHM (i.e., decoctions, tables, capsules, pills, and powders) were included. The pharmacotherapy included conventional antidepressants (paroxetine hydrochloride, fluoxetine hydrochloride, deanxit, and venlafaxine), HT (estradiol valerate tablets and tibolone), and a combination of antidepressants and HT. The treatment duration had to last for three weeks at least.
Types of Outcome Measurements. The primary outcome was the total scores of Hamilton Rating Scale for Depression (HAMD). Clinically, HAMD scores have been commonly used to assess the severity of depression [30]. The secondary outcome was the response rate by reducing the scores of HAMD, Kupperman Menopausal Index (KMI), and adverse events (AEs).
2.3. Data Extraction and Quality Assessment
Titles and abstracts of all retrieved studies were screened, and then full texts were reviewed for eligibility by two authors (Jiju Wang and Jian Liu) independently. The data was collected by using the predefined data extraction form, including the journal title, first author, year of publication, size of study, baseline characteristics of women (e.g., age, course of disease), methodological design, intervention strategy, treatment duration, and outcomes. The dataset was validated by the third reviewer (Xiaouyn Wang).
Two reviewers independently evaluated the risk of bias by using the Cochrane Collaboration's tool [31]. Each domain of the bias was classified as a “low risk”, “unclear”, or “high risk”.
2.4. Data Synthesis
Meta-analysis was performed using Review Manager (the Cochrane Collaboration) software, Version 5.3.0. For dichotomous data, relative risk ratio (RR) with 95 % confidence intervals (CIs) was used to present the therapeutic effect; for continuous data, mean difference (MD) with 95 % CIs was used. The chi-square test and the Higgins I2 test were used to assess heterogeneity. If heterogeneity was low (I2 < 50% or P > 0.1), the fixed effects model was used. If heterogeneity was high (I2 > 50% or P < 0.1), the random effect model was used and further analyses such as subgroup or sensitivity analysis were planned to clarify the source of heterogeneity.
Publication bias was visualized by the funnel chart. Begg's correlation test and Egger's intercept test were used to quantify the publication bias, with a significant level at 0.05.
3. Results
3.1. Study Selection
Databases search found 1076 articles. After duplicates were removed, 761 articles were screened by reading the title/abstract and 628 ineligible articles were removed. After reading the full texts of the remaining 133 articles, 22 trials [32–53] were finally included in the systematic review. The flowchart summarizes the screening process (Figure 1).
Figure 1.
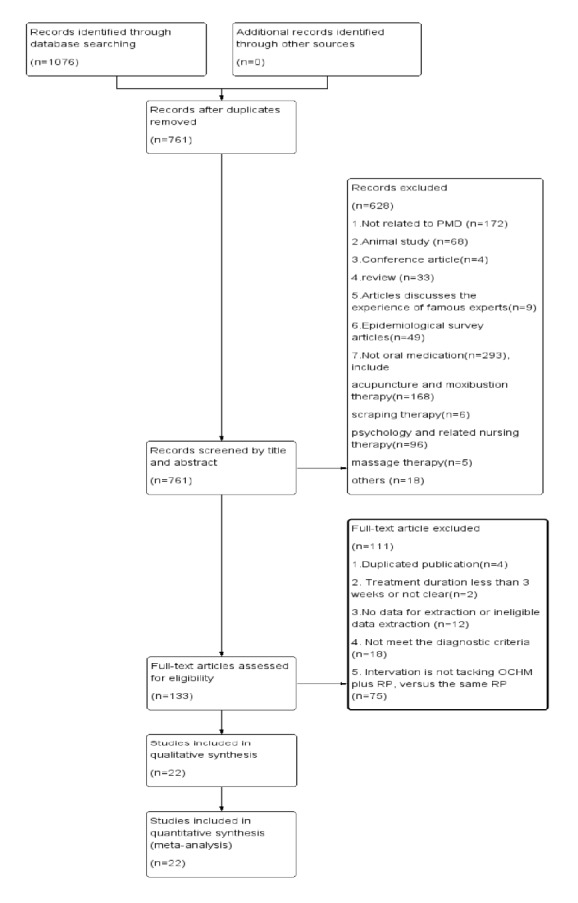
Flowchart of study selection.
3.2. Study Characteristics
All included trials were conducted in China and they were published ranging from 2005 to 2016. The total sample size was 1777, including 901 women in the experimental group and 876 women in the control group. The participants ranged between 40 and 60 years. The most common diagnostic instrument was CCMD-3, and the combination of CCMD-3 and HAMD score was usually used as the inclusion criterion of RCTs. For outcome measurements, eighteen trials reported HAMD score [32–34, 36–40, 42–46, 49–53], eighteen calculated response rate by reducing HAMD scores [33, 35–39, 41–49, 51–53], and three trials reported KMI score [39, 42, 45]. AEs were reported in 10 trails [32, 37–39, 41, 43, 45, 46, 51]. The basic characteristics of the included studies are summarized in Table 1.
Table 1.
Basic characteristics of the included studies.
| Reference | Sample size(I/C) | Age(I/C) | Duration of disease(I/C) | Diagnosis standard | Intervention group | Control group | Treatment duration | Outcome |
|---|---|---|---|---|---|---|---|---|
| Chen H, 2012 | 68/51 | 42-59 (50.20 ± 3.94) /43-58 (50.78 ± 4.08) |
1.76 ± 0.99 y /1.91 ± 1.25 y |
Age; CCMD-3; HAMD |
OCHM + antidepressant | antidepressant | 8w | ①②④ |
|
| ||||||||
| Chu YH, 2005 | 30/30 | 41-60 (47.86 ± 4.42) /41-60 (48.3 ± 4.06) | NS | Age; CCMD-3; HAMD |
OCHM + HT | HT | 6w | ①② |
|
| ||||||||
| Guo LH, 2016 | 43/43 | 40-55/42-55 | (1.8 ± 0.6) y/(1.6 ± 0.8) y | Age; CCMD-3; HAMD |
OCHM + antidepressant | antidepressant | 6w | ② |
|
| ||||||||
| Jiang LX, 2016 | 98/98 | 41-60 (51.18 ± 4.52)/46-54(48.85 ± 3.27) | (4.8 ± 1.7) y/(4.2 ± 1.0) y | Age; CCMD-3; HAMD |
OCHM + antidepressant | antidepressant | 8w | ①②③④ |
|
| ||||||||
| Li QY, 2009 | 34/34 | 41-56 (46.8 ± 4.1) | NS | Age; ICD-10; HAMD |
OCHM + pharmacotherapy | pharmacotherapy | 4w | ①②④ |
|
| ||||||||
| Liu F, 2014 | 50/50 | 44-56 (49.3 ± 2.4) /43-57 (48.4 ± 2.3) |
(2.3 ± 0.5) y/(2.1 ± 0.4)y | Age; ICD-10 |
OCHM + antidepressant | antidepressant | 8w | ①② |
|
| ||||||||
| Liu R, 2007 | 30/30 | (54.32 ± 3.29) /(54.0 ± 4.62) |
(11.32 ± 6.25) m/(12.12 ± 4.58)m | Age; CCMD-3; HAMD |
OCHM + antidepressant | antidepressant | 8w | ①②④ |
|
| ||||||||
| Ni JP, 2014 | 37/33 | (52 ± 4)/(52 ± 4) | (9 ± 5) m/ (9 ± 5) m |
Age; CCMD-3; HAMD |
OCHM + antidepressant | antidepressant | 4w | ②④ |
|
| ||||||||
| Qu LX, 2010 | 30/30 | 45-55(51.6 ± 3.2) /45-55(50.8 ± 2.8) |
(10.5±2.7)m /(11.1±5.2)m |
Age; CCMD-3; HAMD |
OCHM + HT + antidepressant | HT + antidepressant | 8w | ①②④ |
|
| ||||||||
| Shi LR, 2016 | 45/45 | 41-58(46.4 ± 4.2) /42-57(44.8 ± 3.3) |
(11.7 ± 4.5)m /(10.6 ± 6.3)m |
Age; CCMD-3; HAMD |
OCHM + HT + antidepressant | HT + antidepressant | 12w | ①② |
|
| ||||||||
| Sun GC, 2015 | 40/40 | 42.2 ± 2.60 /42.2 ± 2.60 |
(1.92 ± 1.06) y /NS | Age; ICD-10; HAMD |
OCHM + antidepressant | antidepressant | 4w | ① |
|
| ||||||||
| Sun YH, 2013 | 38/38 | 43-48(44.76 ± 2.24) /42-49(45.02 ± 3.15) |
(2.38 ± 1.24) y /(2.54 ± 1.56)y |
Age; CCMD-3; HAMD |
OCHM + HT + antidepressant | HT + antidepressant | 4w | ② |
|
| ||||||||
| Wang CG, 2015 | 46/46 | 44-53(46.93 ± 6.8) /44-53(47.52 ± 7.2) |
NS | Age; CCMD-3 |
OCHM + antidepressant | antidepressant | 6w | ①④ |
|
| ||||||||
| Wang SF, 2015 | 40/40 | 43-50(46.45 ± 6.15) /42-49(45.82 ± 5.45) |
(2.75 ± 0.35)y /(2.85 ± 0.43)y |
Age; CCMD-3; HAMD |
OCHM + HT + antidepressant | HT + antidepressant | 3w | ① |
|
| ||||||||
| Wang XL, 2011 | 60/60 | 45-55(51.21 ± 3.17) /45-55(50.86 ± 3.41) |
NS | Age; CCMD-3; HAMD |
OCHM + HT + antidepressant | HT + antidepressant | 8w | ① |
|
| ||||||||
| Wang Y, 2015 | 30/30 | 45.20 ± 2.90 /44.80 ± 3.10 |
NS | Age; ICD-10; HAMD |
OCHM + antidepressant | antidepressant | 8w | ①②③④ |
|
| ||||||||
| Zhang CH, 2013 | 40/40 | 43-56(46.80 ± 4.10) /44-55(45.40 ± 3.2) |
(11.50±4.30)m /(10.90±6.50)m |
Age; CCMD-3; HAMD |
OCHM + HT + antidepressant | HT + antidepressant | 3w | ①② |
|
| ||||||||
| Zhang CH, 2011 | 30/30 | 45-55(43.50 ± 12.43) /45-55(42.25 ±11.38) |
NS | Age; CCMD-3; HAMD |
OCHM + antidepressant | antidepressant | 6w | ①② |
|
| ||||||||
| Zhang GQ, 2009 | 38/34 | 45-56(51.63 ± 3.68) /45-56(51.95 ± 3.86) |
(9.44 ± 4.69)m /(8.88 ± 4.65) m |
Age; CCMD-3; HAMD |
OCHM + antidepressant | antidepressant | 4w | ①②④ |
|
| ||||||||
| Zhang HZ, 2014 | 20/20 | 45-53 /46-52 |
(6-24)m /(5-22)m |
Age; CCMD-3 |
OCHM + HT | HT | 12w | ①②③ |
|
| ||||||||
| Zhang SJ, 2012 | 27/27 | 43-54(46.5 ± 6.3) /44-55(45.3 ± 7.2) |
(3.2 ± 2.8)m /(4.1 ± 3.2)m |
Age; DSM-IV |
OCHM + antidepressant | antidepressant | 4w | ② |
|
| ||||||||
| Zhang XH, 2014 | 27/27 | (47 ± 6.8) /(46.0 ± 6.8) |
NS | Age; CCMD-3; HAMD |
OCHM + antidepressant | antidepressant | 8w | ①② |
CCMD-3: Criteria for Classification and Diagnosis of Mental Diseases; DSM-IV: Diagnostic and Statistical Manual; ICD-10: International Classification of Disease; m = month; y = year; NS: not stated.OCHM: oral Chinese herbal medicine; HT: hormone therapy; w = week; ①: Hamilton Rating Scale for Depression (HAMD) score; ②: response rate; ③: Kupperman Menopausal Index (KMI) score; ④: adverse events (AEs).
Four forms of OCHM were investigated, consisting of decoctions, capsule, granule, and powder. Twenty-two formulas of OCHM were found. Bupleuri Radix (Chinese name: Chaihu) and Paeoniae Radix Alba (Chinese name: Baishao) were the herbs with top frequency. Details of OCHM in the included studies are summarized in Table 2. Control group included three categories of pharmacologic agents, antidepressants in 13 trials [32, 37–41, 44–49, 52], HT in 2 trials [42, 53], and the combination of antidepressants and HT in 6 trials [33–36, 50, 51]. One trial did not specify the pharmacotherapy [43]. The treatment duration ranged from three to 12 weeks.
Table 2.
Chinese herbal medicine of the included studies.
| Study | Formula | a Herbal ingredients | Preparation | Dosage | Frequency |
|---|---|---|---|---|---|
| Chen H, 2012 | Jie Yu Jing Xin Ke Li | zhenzhumu, huaixiaomai, shoudihuang, shanzhuyu, tusizi, suanzaoren, fuling, chaihu, baishao, meiguihua, danggui, nvzhenzi | granule | 1 bag | bid |
|
| |||||
| Chu YH, 2005 | Xiao Yao Jie Yu Tang | Chaihu, danggui, baishao, baizhu, fuling, weijiang, bohe, zhi gan cao, xian ling pi, nvzhenzi, shengmaiya, chaomaiya | decoction | 1 pack decocted twice | bid |
|
| |||||
| Guo LH, 2016 | Bai He Di Huang Tang | Baihe, sheng di huang, long gu, muli, danggui, he huan pi, chaihu, ye jiao teng, fushen, yujin, zhi mu | decoction | 1 pack decocted twice | bid |
|
| |||||
| Jiang LX, 2016 | Wu Ling Jiao Nang | wulingjun | capsule | 3 tablets | tid |
|
| |||||
| Li QY, 2009 | Jie Yu Tang | Dangshen, chuanxiong, danggui, danshen, xiangfu, he huan pi, gualou, yujin | decoction | 1 pack decocted twice | bid |
|
| |||||
| Liu F, 2014 | Suan Zao Ren Tang | Suanzaoren, chuanxiong, fuling, wuweizi, zhi mu, gan cao | decoction | 1 pack decocted twice | bid |
|
| |||||
| Liu R, 2007 | Xue Fu Zhu Yu Jiao Nang | Taoren, honghua, chishao, chuanxiong, zhiqiao, chaihu, jiegeng, danggui, dihuang, niuxi, gan cao | capsule | 6 tablets | bid |
|
| |||||
| Ni JP, 2014 | Shen Song Yang Xin Jiao Nang | Renshen, maidong, shanzhuyu, danshen, suanzaoren, sangjishen, chishao, tu bie chong, gansong, huanglian, wuweizi, long gu | capsule | 4 tablets | tid |
|
| |||||
| Qu LX, 2010 | Jie Yu Zi Shen Tang | Chaihu, xiangfu, baishao, baizhu, yujin, gan cao, suanzaoren, fuling, shichangpu, shoudihuang, shanyurou, shanyao | decoction | 1 pack decocted twice | bid |
|
| |||||
| Shi LR, 2016 | Zi Ni Zi Gan Yang Shen Tang | Fuling, gan cao, baishao, chaihu, chuanxiong | decoction | 1 pack decocted twice | bid |
|
| |||||
| Sun GC, 2015 | Jia Wei Xiao Yao San | Chaihu, baizhu, bohe, danggui, fuling, baishao, gan cao, shengjiang, zhizi, danpi | granule | 1 bag | tid |
|
| |||||
| Sun YH, 2013 | Zi Ni Shu Gan Jie Yu Tang | Chaihu, xiangfu, chuanxiong, chishao, yujin, chenpi, sheng di huang, shanzhuyu, danpi, yuanzhi, gan cao | decoction | 1 pack decocted twice | bid |
|
| |||||
| Wang CG, 2015 | Zi Ni An Shen Jie Yu Tang | Huangqi, huangqin, chaihu, muxiang, zhiqiao, gan cao, sharen, peilan, dangshen, fushen, danggui, baizhu, chuanxiong, yujin, suanzaoren, ye jiao teng, long yan rou | decoction | 1 pack decocted twice | bid |
|
| |||||
| Wang SF, 2015 | Zi Ni Shu Gan Jie Yu Tang | Chaihu, xiangfu, chuanxiong, baishao, yujin, chenpi, zhi mu, shengdi, shanzhuyu, danpi, suanzaoren, gan cao | decoction | 1 pack decocted twice | bid |
|
| |||||
| Wang XL, 2011 | Si Hua Jie Yu Tang | He huan hua, xuanhua, meiguihua, baimeihua, suanzaoren, bai zi ren, fuxiaomai, fushen, ye jiao teng, tiandong, maidong, wuweizi, gan cao, dazao | decoction | 1 pack decocted twice | bid |
|
| |||||
| Wang Y, 2015 | Zi Ni Bai He Di Huang Tang | Baihe, shengdi, maidong, wuweizi, he huan pi, ye jiao teng, fuling, yuanzhi, shichangpu, yujin, chuanxiong, gan cao | decoction | 1 pack decocted twice | bid |
|
| |||||
| Zhang CH, 2013 | Wu Ling Jiao Nang | wulingjun | capsule | 3 tablets | tid |
|
| |||||
| Zhang CH, 2011 | Zi Ni Zi Yin Bu Shen Tang | Chaihu, xiangfu, chuanxiong, baishao, yujin, shoudihuang, fuling, shanzhuyu, suanzaoren, gan cao | decoction | 1 pack decocted twice | bid |
|
| |||||
| Zhang GQ, 2009 | Zi Ni Zao Ren Bu Xue Tang |
Suanzaoren, fuling, chuanxiong, zhi mu, gan cao, huangqi, danggui, shanzhuyu, shoudihuang, danshen, chaihu, xiangfu, yujin, shichangpu | decoction | 1 pack decocted twice | bid |
|
| |||||
| Zhang HZ, 2014 | Si Er Wu He Fang And Gan Mai Da Zao Tang | Danggui, baishao, danshen, shoudihuang, xianmao, xian ling pi, fu pen zi, tusizi, wuweizi, che qian zi, gouqi, yujin, huanglian, rougui, xiaomai, gan cao, dazao | decoction | 1 pack decocted twice | bid |
|
| |||||
| Zhang SJ, 2012 | Zi Ni Bai He Di Huang Tang | Baihe, shengdi, danshen, chuanxiong, juhua | decoction | 1 pack decocted twice | bid |
|
| |||||
| Zhang XH, 2014 | Kun Tai Jiao Nang | Shoudihuang, huanglian, baishao, huangqin, ejiao, fuling | capsule | 4 tablets | tid |
a The herbal ingredients are presented as Chinese pinyin. Bid: twice per day; tid: three times per day.
3.3. Risk of Bias
The risk of bias was summarized in Figures 2 and 3. For random sequence generation, six trials used random number tables [32, 39, 42, 45, 46, 52], and 13 trials [33–35, 37, 38, 40, 41, 43, 47–50, 53] did not provide details of randomization. Three trials considered the order of visits as random, which were of high risks in selection bias [36, 44, 51]. None of the trials reported allocation concealment. Binding of participants, personnel, and outcome assessors was not applied in any studies. None of the studies had attribution bias. The risk of bias in selective reporting was unclear as none of the studies published their protocols.
Figure 2.
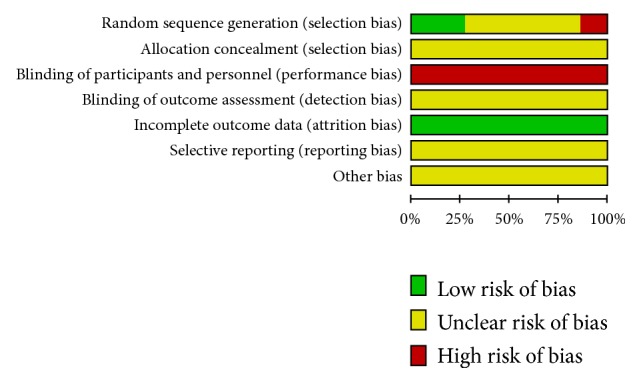
Risk of bias across included studies.
Figure 3.
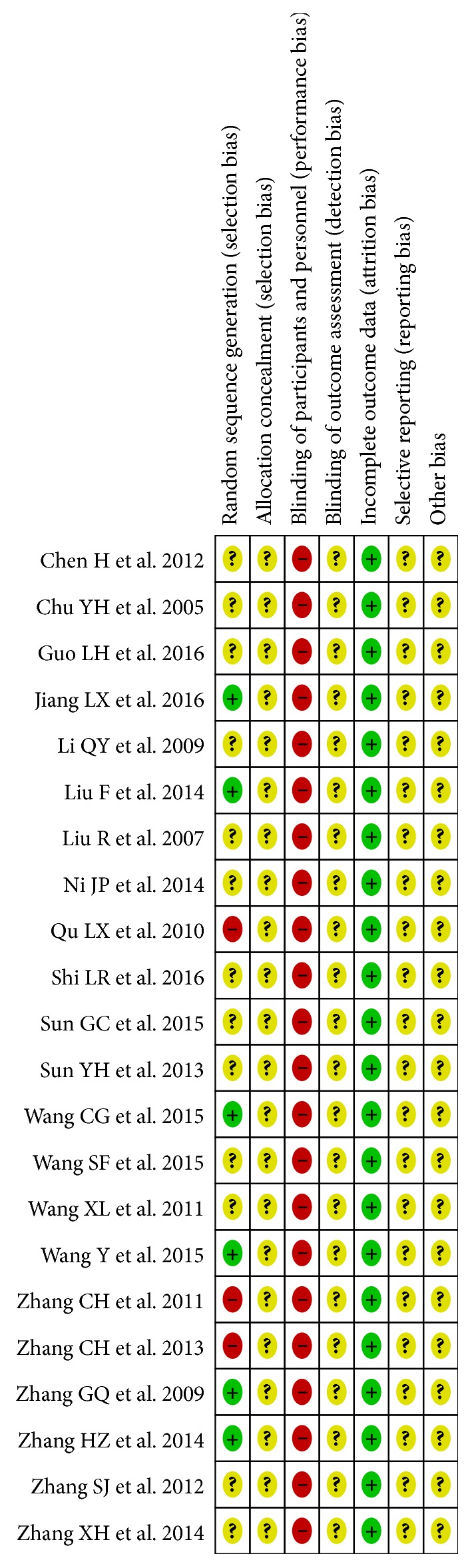
Risk of bias of individual studies. +: low risk of bias; ?: unclear risk of bias; −: high risk of bias.
3.4. Effect of Intervention
3.4.1. Hamilton Rating Scale for Depression (HAMD) Scores
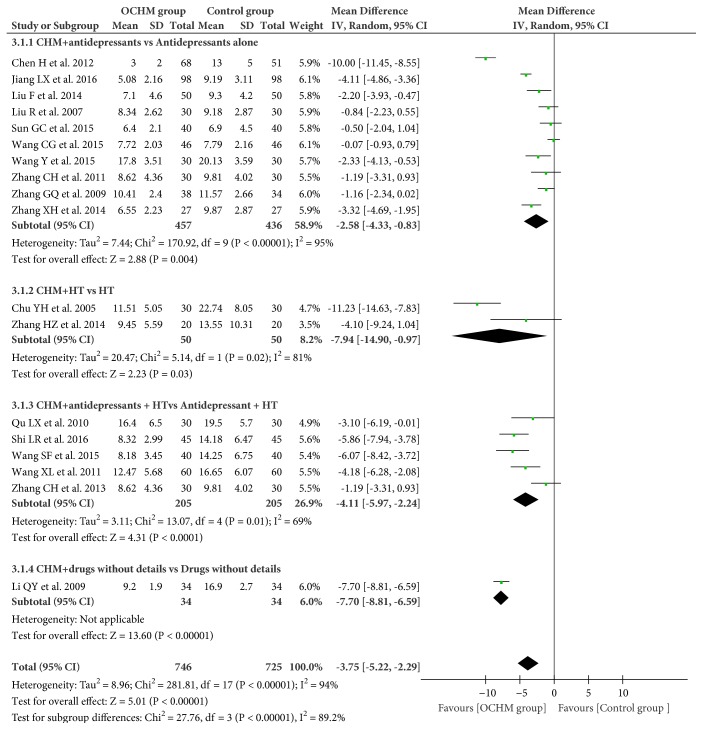
Eighteen RCTs (n = 1417 participants) used HAMD scores to measure the effect of OCHM for menopausal depression [32–34, 36–40, 42–46, 49–53]. Overall, the adjuvant use of OCHM therapy in pharmacotherapy was superior to pharmacotherapy alone (mean difference (MD) = −3.75; 95% CI = −5.22, −2.29; I2 = 94%; random model) (Figure 4).
Figure 4.
Forest plot of HAMD scores. HAMD: Hamilton Rating Scale for Depression; CHM: Chinese herbal medicine; HT: hormone therapy.
As the meta-analysis was heterogeneous, subgroup analysis in terms of different categories of pharmacologic agents was performed (Figure 4). The subgroup analysis showed that adjuvant therapy of OCHM for menopausal depression was more effective than antidepressants alone (MD = −2.58; 95% CI = −4.33, −0.83; I2 = 95%) [32, 37–40, 44–46, 49, 52]. It was superior to HT alone (MD = −7.94; 95% CI = −14.90, −0.97; I2 = 81%) [42, 53] and antidepressants combined with HT (MD = −4.11; 95% CI = −5.97, −2.24; I2 = 69%; random model) [33, 34, 36, 50, 51]. Another study [43] did not specify the pharmacotherapy.
3.4.2. Response Rate
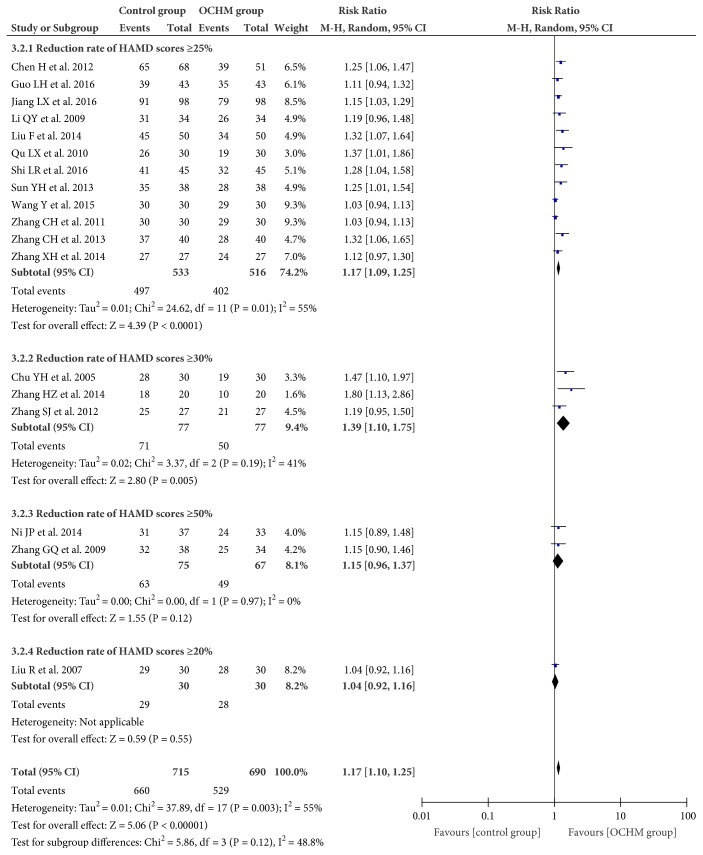
Eighteen trials (n = 1189 participant) applied response rate of reducing HAMD scores in outcome measurement [33, 35–39, 41–49, 51–53]. The overall meta-analysis showed OCHM adjuvant therapy for menopausal depression was superior to pharmacotherapy (RR = 1.17; 95% CI = 1.10, 1.25; I2 = 55%; random model) (Figure 5).
Figure 5.
Forest plot of response rate.
3.4.3. Kupperman Menopausal Index (KMI) Scores
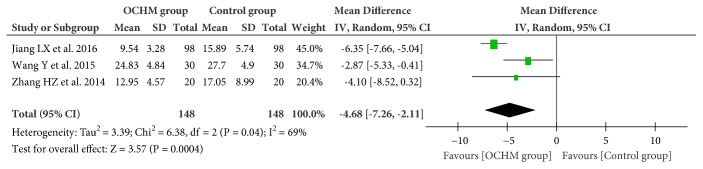
Three RCTs [39, 42, 45] measured the effectiveness of OCHM for menopausal depression by using KMI scores. Meta-analysis showed OCHM combined with pharmacotherapy was more effective than pharmacotherapy alone (MD = −4.68; 95% CI = −7.26, −2.11; I2 = 69%; random model) (Figure 6).
Figure 6.
Forest plot of Kupperman menopausal index scores.
3.4.4. Adverse Events (AEs)
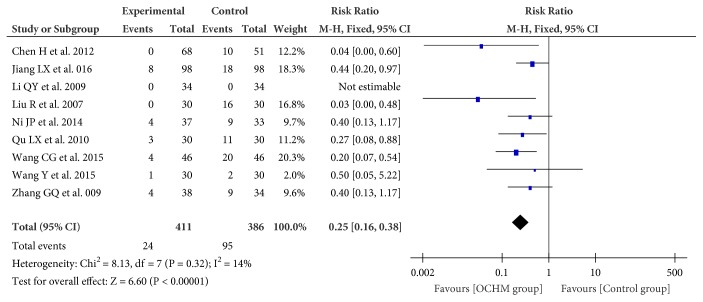
AEs were monitored in nine studies (n = 797 participants) [32, 37–39, 41, 43, 45, 46, 51]. Meta-analysis showed that the incident of adverse events in the group of OCHM plus pharmacotherapy was less than the pharmacotherapy (RR = 0.25; 95% CI = 0.16, 0.38; I2 = 14%; fixed model) (Figure 7).
Figure 7.
Forest plot of adverse event.
3.5. Publication Bias
The funnel plots of HAMD scores and the response rate were asymmetrical (Figures 8 and 9). The regression analysis of Begg's rank correlation test and Egger's intercept test was statistically significant (P > 0.05), suggesting the presence of publication bias. The detection of publication bias was not available for other outcomes as the included studies were less than 10.
Figure 8.
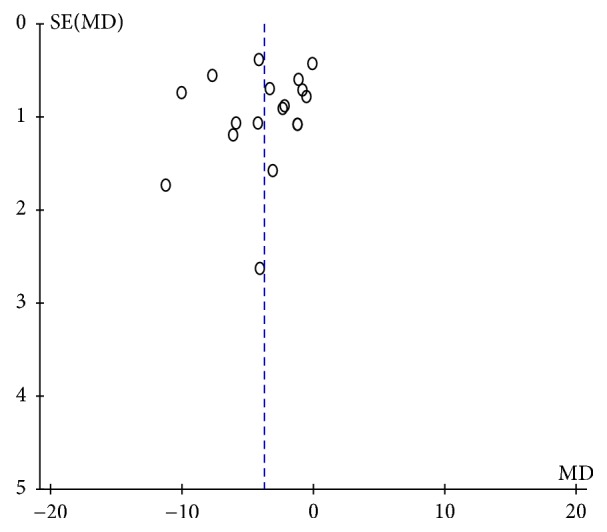
Funnel plot of HAMD scores.
Figure 9.
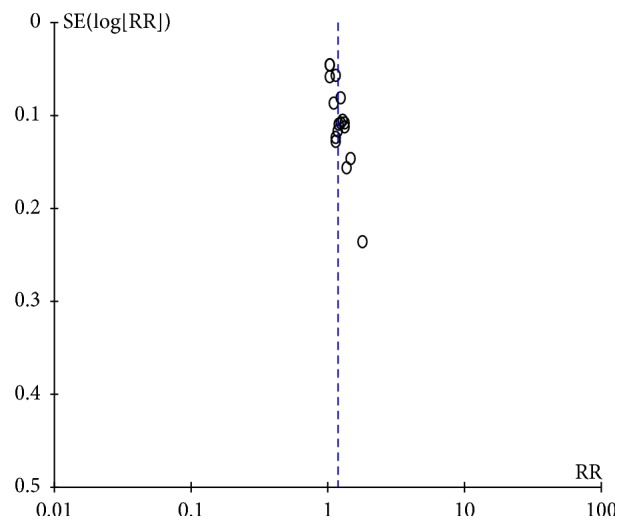
Funnel plot of response rate.
4. Discussion
4.1. Statement of Principal Findings
In the present study, we reviewed 22 RCTs involving a total of 1777 participants and assessed the add-on effects and safety of OCHM to pharmacotherapy in women with menopausal depression. Meta-analysis showed that the combination of OCHM and pharmacotherapy was more effective in improving menopausal depression. In addition, the incidence of AEs in the participants treated with OCHM adjunctive therapy was less than those without OCHM adjunctive therapy. However, the risk of bias of the included RCTs may affect the evidence certainty; particularly most studies did not blind the participants or personnel.
4.2. Possible Explanations for the Evidence
OCHM can increase the expression level of estrogen receptors in hypothalamic pituitary ovarian (HPO) axis [54, 55]. OCHM can also alleviate hippocampal neuron damage, inhibit early apoptosis of neurons, and increase the content of monoamine transmitters in brain tissue through the CREB-BDNF signaling pathway [56]. Furthermore, OCHM can regulate the hypothalamus pituitary adrenal (HPA) axis in menopausal depression rats and improve the behavior of model rats [57]. Bupleuri Radix (Chinese name: Chaihu) and Paeoniae Radix Alba (Chinese name: Baishao) were the herbs with top frequency. Bupleurum-saikoside, the main active ingredient of Bupleuri Radix, improved depression by regulating the monoamine neurotransmitters and BDNF in the brain [58]. Paeoniae Radix Alba improved depression by increasing the single amine neurotransmitter and adjusting the dysfunction of HPA axis [59].
4.3. Limitations and Implications of the Research
There were several limitations in this study. Firstly, the quality of the included trials was generally poor. The risks of selection bias and reporting bias were unclear most of the time. And the risk of performance bias in almost all the RCTs was high. These biases may affect the results of meta-analysis. Secondly, the heterogeneity was observed across meta-analyses and it was not resolved by subgroup analysis. This can also reduce the evidence certainty. Thirdly, none of the RCTs included considered placebo as the control. Hence the current studies were unable to conclude the efficacy of OCHM. Although it is difficult to successfully produce a placebo to CHM because of its special characteristics such as the appearance, smell, and taste, recent progress of placebo making can be applied. For example, when researchers explored the preparation method of placebo to Moron Dan, they found that the flavor characteristics and disintegration of the placebo made of soybean powder 100g, starch 100g, carbon black pigment 1g, and honey 70g were similar to the experimental drug [60]. Some researchers suggested that encrypted capsules could be also used as a placebo [61].
5. Conclusion
Adjuvant therapy of OCHM provided additional benefits to pharmacotherapy in the people with menopausal depression. More RCTs with a rigorous design, particularly applying placebo as the control as well as blinding the participants and personnel, are needed to confirm the efficacy of OCHM for menopausal depression.
Acknowledgments
This study was supported by the funding from Traditional Chinese Medicine Bureau of Guangdong Province (no. 2015KT1164 and no. 20171111) and the grant from Natural Science Foundation of Guangdong Province (no. 2017A030313728).
Appendix
Search strategy included the following:
#1 Menopause [Mesh]
#2 ((((((((((Perimenopausal [ti.ab]) OR menopausal [ti.ab]) OR menopause [ti.ab]) OR perimenopause [ti.ab]) OR premenopause [ti.ab]) OR postmenopause [ti.ab]) OR menopausal transition[ti.ab]) OR premenopausal [ti.ab]) OR postmenopausal [ti.ab]) OR climacterium [ti.ab]) OR climacteric [ti.ab]
#3 #1 OR #2
#4 Depression [Mesh]
#5 (((((((((Depression [ti.ab]) OR Depressions [ti.ab]) OR Depressive Symptoms [ti.ab]) OR Depressive Symptom [ti.ab]) OR Symptom, Depressive [ti.ab]) OR Symptoms, Depressive [ti.ab]) OR Emotional Depression [ti.ab]) OR Depression, Emotional [ti.ab]) OR Depressions, Emotional [ti.ab]) OR Emotional Depressions [ti.ab]
#6 #4 OR #5
#7 Medicine, Chinese Traditional [Mesh]
#8 ((((((Medicine, Chinese Traditional [ti.ab]) OR Traditional Chinese Medicine [ti.ab]) OR Chinese Medicine, Traditional [ti.ab]) OR Chung I Hsueh [ti.ab]) OR Hsueh, Chung I [ti.ab]) OR Zhong Yi Xue [ti.ab]) OR Chinese Traditional Medicine [ti.ab]) OR Traditional Medicine, Chinese [ti.ab]
#9 #7 OR #8
#10 ((randomized controlled trial [Publication Type]) OR randomized [ti.ab]) OR placebo [ti.ab]
#11 #3 AND #6 AND #9 AND #10
Conflicts of Interest
The authors declare that there are no conflicts of interest regarding the publication of this article.
Authors' Contributions
The study was conceived and designed by Xiaoyun Wang. Jiju Wang and Jian Liu independently screened literature, extracted data, and evaluated study quality. Xiaoyun Wang validated the data. Jiju Wang conducted data synthesis and analysis. Xiaoyun Wang, Xiaojia Ni, Jian Liu, and Guangning Nie finalized the results. Yuyan Zeng, Xiaojing Cao, and Xiaoyu Li participated in the discussion of data interpretation. Wang Jiju drafted the manuscript and Xiaojia Ni revised it.
References
- 1.Avis N. E., Stellato R., Crawford S., et al. Is there a menopausal syndrome? Menopausal status and symptoms across racial/ethnic groups. Social Science & Medicine. 2001;52(3):345–356. doi: 10.1016/S0277-9536(00)00147-7. [DOI] [PubMed] [Google Scholar]
- 2.Bosworth H. B., Bastian L. A., Kuchibhatla M. N., et al. Depressive symptoms, menopausal status, and climacteric symptoms in women at midlife. Psychosomatic Medicine. 2001;63(4):603–608. doi: 10.1097/00006842-200107000-00013. [DOI] [PubMed] [Google Scholar]
- 3.Dennerstein L., Dudley E. C., Hopper J. L., et al. A prospective population-based study of menopausal symptoms. Obstet Gynecol. 2000;96(3):351–358. doi: 10.1016/s0029-7844(00)00930-3. [DOI] [PubMed] [Google Scholar]
- 4.de Novaes Soares C. Depression During the Perimenopause. Archives of General Psychiatry. 58(3):306–306. doi: 10.1001/archpsyc.58.3.306. [DOI] [PubMed] [Google Scholar]
- 5.Timur S., Şahin N. H. The prevalence of depression symptoms and influencing factors among perimenopausal and postmenopausal women. Menopause. 2010;17(3):545–551. doi: 10.1097/gme.0b013e3181cf8997. [DOI] [PubMed] [Google Scholar]
- 6.Freeman E. W. Associations of depression with the transition to menopause. Menopause. 2010;17(4):823–827. doi: 10.1097/gme.0b013e3181db9f8b. [DOI] [PubMed] [Google Scholar]
- 7.Soares C. N. Depression during the menopausal transition: Window of vulnerability or continuum of risk? Menopause. 2008;15(2):207–209. doi: 10.1097/gme.0b013e31816381e9. [DOI] [PubMed] [Google Scholar]
- 8.Goldbacher E. M., Bromberger J., Matthews K. A. Lifetime history of major depression predicts the development of the metabolic syndrome in middle-aged women. Psychosomatic Medicine. 2009;71(3):266–272. doi: 10.1097/PSY.0b013e318197a4d5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Janssen I., Powell L. H., Matthews K. A., et al. Depressive symptoms are related to progression of coronary calcium in midlife women: The Study of Women's Health Across the Nation (SWAN) Heart Study. American Heart Journal. 2011;161(6):1186–e1. doi: 10.1016/j.ahj.2011.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mantani A., Yamashita H., Fujikawa T., Yamawaki S. Higher incidence of hysterectomy and oophorectomy in women suffering from clinical depression: Retrospective chart review. Psychiatry and Clinical Neurosciences. 2010;64(1):95–98. doi: 10.1111/j.1440-1819.2009.02044.x. [DOI] [PubMed] [Google Scholar]
- 11.Wariso B. A., Guerrieri G. M., Thompson K., et al. Depression during the menopause transition: impact on quality of life, social adjustment, and disability. Archives of Women's Mental Health. 2017;20(2):273–282. doi: 10.1007/s00737-016-0701-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ferguson J. M. SSRI Antidepressant Medications. The Primary Care Companion for CNS Disorders. 2001;03(01):22–27. doi: 10.4088/PCC.v03n0105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dording C. M., Mischoulon D., Petersen T. J., et al. The pharmacologic management of SSRI-induced side effects: a survey of psychiatrists. Annals of Clinical Psychiatry. 2002;14(3):143–147. doi: 10.1023/a:1021137118956. [DOI] [PubMed] [Google Scholar]
- 14.Reichenpfader U., Gartlehner G., Morgan L. C., et al. Sexual dysfunction associated with second-generation antidepressants in patients with major depressive disorder: Results from a systematic review with network meta-analysis. Drug Safety. 2014;37(1):19–31. doi: 10.1007/s40264-013-0129-4. [DOI] [PubMed] [Google Scholar]
- 15.Gordon J. L., Girdler S. S. Hormone Replacement Therapy in the Treatment of Perimenopausal Depression. Current Psychiatry Reports. 2014;16(12) doi: 10.1007/s11920-014-0517-1. [DOI] [PubMed] [Google Scholar]
- 16.de Villiers T. J., Hall J. E., Pinkerton J. V., et al. Revised Global Consensus Statement on Menopausal Hormone Therapy. Climacteric. 2016;19(4):313–315. doi: 10.1080/13697137.2016.1196047. [DOI] [PubMed] [Google Scholar]
- 17.Toffol E., Heikinheimo O., Partonen T. Hormone therapy and mood in perimenopausal and postmenopausal women: a narrative review. Menopause. 2014 doi: 10.1097/GME.0000000000000323. [DOI] [PubMed] [Google Scholar]
- 18.Chen F.-P., Chen T.-J., Kung Y.-Y., Chen Y.-C., Chou L.-F., Hwang S.-J. Use frequency of traditional Chinese medicine in Taiwan. BMC Health Services Research. 2007;7, article 26 doi: 10.1186/1472-6963-7-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Xue C. C. L., Zhang A. L., Lin V., da Costa C., Story D. F. Complementary and alternative medicine use in Australia: a national population-based survey. The Journal of Alternative and Complementary Medicine. 2007;13(6):643–650. doi: 10.1089/acm.2006.6355. [DOI] [PubMed] [Google Scholar]
- 20.Borrelli F., Ernst E. Alternative and complementary therapies for the menopause. Maturitas. 2010;66(4):333–343. doi: 10.1016/j.maturitas.2010.05.010. [DOI] [PubMed] [Google Scholar]
- 21.Koike K., Ohno S., Takahashi N., et al. Efficacy of the herbal medicine Unkei-to as an adjunctive treatment to hormone replacement therapy for postmenopausal women with depressive symptoms. Clinical Neuropharmacology. 2004;27(4):157–162. doi: 10.1097/01.wnf.0000138634.34498.05. [DOI] [PubMed] [Google Scholar]
- 22.Jun J. H., Choi T.-Y., Lee J. A., Yun K.-J., Lee M. S. Herbal medicine (Gan Mai da Zao decoction) for depression: A systematic review and meta-analysis of randomized controlled trials. Maturitas. 2014;79(4):370–380. doi: 10.1016/j.maturitas.2014.08.008. [DOI] [PubMed] [Google Scholar]
- 23.Yeung W.-F., Chung K.-F., Ng K.-Y., Yu Y.-M., Ziea E. T.-C., Ng B. F.-L. A systematic review on the efficacy, safety and types of Chinese herbal medicine for depression. Journal of Psychiatric Research. 2014;57(7):165–175. doi: 10.1016/j.jpsychires.2014.05.016. [DOI] [PubMed] [Google Scholar]
- 24.Franco O. H., Chowdhury R., Troup J., et al. Use of plant-based therapies and menopausal symptoms: A systematic review and meta-analysis. Journal of the American Medical Association. 2016;315(23):2554–2563. doi: 10.1001/jama.2016.8012. [DOI] [PubMed] [Google Scholar]
- 25.Moher D., Liberati A., Tetzlaff J. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Journal of Clinical Epidemiology. 2009;62(10):1006–1012. doi: 10.1016/j.jclinepi.2009.06.005. [DOI] [PubMed] [Google Scholar]
- 26.Association PBoCM. Category and diagnostic criteria of mental disorder of china. 3rd. Shandong Science and Technology Press; 2001. [Google Scholar]
- 27.American psychiatric association. Diagnostic and statistical manual of mental disorders. 4th. Hutchison book press; 2003. [Google Scholar]
- 28.Category and Diagnostic Criteria of Mental Disorder of China. 3rd. Jinan, China: Psychiatry Branch of Chinese Medical Association; 2001. [Google Scholar]
- 29.Organization WH. The tenth revision of the intermational classification of diseases and related health problems. 10th. Geneva: World health organization; 1992. http://www.Who.Int/classifications/icd/en/ [Google Scholar]
- 30.Zimmerman M., Martinez J. H., Young D., Chelminski I., Dalrymple K. Severity classification on the hamilton depression rating scale. Journal of Affective Disorders. 2013;150(2):384–388. doi: 10.1016/j.jad.2013.04.028. [DOI] [PubMed] [Google Scholar]
- 31.Julian P. T. Higgins and Sally Green. Cochrane handbook for systematic reviews of interventions. 2011;(Version 5.1.0) The Cochrane Collaboration.
- 32.Wang C. G., Mei J., Zhang Y., et al. Cases with permenopausal depression treated by traditional Chinese medicine combined with paroxetine. Henan Traditional Chinese Medicine. 2015;35(3):578–579. [Google Scholar]
- 33.Shi L. R. Observation on the efficacy of integrated traditional Chinese medicine and western medicine in the treatment of perimenopausal depression. China’s Naturopathy. 2016;24(1):63–64. [Google Scholar]
- 34.Wang S. F. in women with integrated traditional Chinese and western medicine. no. 8. Women's Health Research; 2015. Treatment of perimenopausal depression; p. p. 51. [Google Scholar]
- 35.Sun Y. H. Treatment of female perimenopausal depression by combination of traditional Chinese medicine and western medicine. Health must-Read magazine. 2013;12(6):p. 378. [Google Scholar]
- 36.Zhang C. H., Xu S. W. Clinical observation on 40 cases of perienopausal depression treated by integrated traditional Chinese medicine and western medicine. Henan Journal of Traditional Chinese Medicine and Pharmacy. 2013;36(9):1534–1535. [Google Scholar]
- 37.Tang F., Zheng Y. R. Analysis on safety reserve of slope with two strength reduction factor. Journal of Chongqing Jiaotong University (Natural Science) 2007;26(4):95–100. [Google Scholar]
- 38.Chen H., Yao Q. Y., Lu J. M., et al. Clinical observation on integrated treatment of perimenopausal depression by traditional Chinese medicine and western medicine. Chinese Journal of Traditional Medical Science and Technology. 2012;18(5):429–430. [Google Scholar]
- 39.Jiang L. X., Chen Y. C., Yang S. C. Clinical evaluation on efficacy of Wuling Capsules combined with mirtazapine in perimenopausal women with depression. Drug Evaluation Research. 2016;39(5):832–835. [Google Scholar]
- 40.Sun G. C., Chang J. H. Clinical observation on the effect of Jiawei Xiaoyao powder used in perimenopausal women with depression. World Latest Medicine Information. 2015;15(42):p. 109. [Google Scholar]
- 41.Ni J. P. A case-control study of Shensong Yangxin capsules combined with fluoxetine in treatment of climacteric depression. Zhejiang Journal of Traditional Chinese Medicine. 2014;49(11):p. 856. [Google Scholar]
- 42.Zhang H. Z., Shan S. l., Zhao C. Clinical observation on treatment of perimenopausal depression by co-prescriptio n of Four-Two-Five and Ganmai Dazao decoction. Journal of Basic Chinese Medicine. 2014;20(9):1256–1257. [Google Scholar]
- 43.Li Q. Y., Wang X. H., Song Y. H., et al. A case-control study on climacteric depression treated with traditional Chinese medicine and western medicine. World Chinese Medicine. 2009;4(2):80–81. [Google Scholar]
- 44.Zhang C. H., Yao W. B. Clinical observation on the therapeutic effect of paroxetine combined with Wuling capsules in treatment of perimenopausal women with depression; 2011; pp. 545–546. [Google Scholar]
- 45.Wang Y., Kang M. X., Ma J., et al. A case-control study of duloxetine plus modified Baihe Dihuang decoction in the treatment of perimenopausal depression. Journal of Clinical Psychosomatic Diseases. 2015;21(5):44–46. [Google Scholar]
- 46.Zhang G. Q., Zhang G. P., Ai C. M., et al. Clinical observation on climacteric depression treated by Zaoren Buxue decoction combined with fluoxetine. Beijing Journal of Traditional Chinese Medicine. 2009;28(11):873–874. [Google Scholar]
- 47.Zhang S. J. Clinical research on the treatment of climacteric depression by modified Baihe Dihuang decoction combined with western medicine. China Journal of Chinese medicine. 2012;27(171):1049–1050. [Google Scholar]
- 48.Guo L. H., Yao H. Q., Kang Z. The clinical efficacy and influence on neuroendocrine system of therapies using Baihe Dihuang decoction in climacteric patients with depression. Guiding Journal of Traditional Chinese Medicine and Pharmacology. 2016;22(8):70–72. [Google Scholar]
- 49.Zhang X. H., Kang P., Chen G. J. Observation of the curative effects in treatment of climacteric depression by using a combination of mirtazapine and kuntai capsules. Chinese Journal of Women and Child Health Research. 2014;25(5):874–875. [Google Scholar]
- 50.Wang X. L., Ma H. Z., Zi X. H., et al. Observation on Clinical Efficacy on Sihua Jieyu decoction as adjuvant therapy in treatment of climacteric depression. Journal of China Traditional Chinese Medicine Information. 2011;3(17):99–100. [Google Scholar]
- 51.Qu L. X., Dai Q. J., Ke J., et al. Clinical observation on efficacy of Jieyu Zishen decoction in treating climacteric depression. Journal of Sichuan of Traditional Chinese Medicine. 2010;28(4):93–94. [Google Scholar]
- 52.Liu F. Clinical observation on efficacy of modified Suanzaoren decoction combined with deanxit in treatment of climacteric depression. Chinese Archives of Traditional Chinese Medicine. 2014;32(12):3044–3046. [Google Scholar]
- 53.Chu Y. H. Combined Chinese medicine and western medicine in the treatment of perimenopause depression. Shandong University of Traditional Chinese Medicine: 2005. [Google Scholar]
- 54.Jiang D. Z., Feng Q. X., Gao Y. T., et al. Effect of Zishui Qinggan Decoction on hypothalamus ER-amRNA expression and 5-HT content in rats with Depression in peri-menopausal period. Tianjin Journal of Traditional Chinese Medicine. 2008;25(2):170–173. [Google Scholar]
- 55.Li P. F. Molecular Mechanisms Study of Total Flavonoids of Cynomoriumon Climacteric Syndrome Rats Models. Shool of Pharmacy: Henan College of Traditional Chinese Medicine: Chinese Medicine,, Shool of Pharmacy: Henan College of Traditional; 2015. [Google Scholar]
- 56.Wang R., Liu J. C., J C. The neuroprtective effects of puerarin on mouse model of perimenopausal depression and possible mechanism. Chinese Journal of Neuroanatomy. 2017;33(2):190–196. [Google Scholar]
- 57.Dong L., Jiang L., Meng W., et al. Effect of Bushen Jieyu Qinxin Decoction on the Behavior and Secretion of Hypothalamus-Pituitary-Adrenal Axis of the Perimenopausal Depression Model in Rat. Chinese Journal of Experimental Traditional Medical Formulae. 2008;14(5):63–65. [Google Scholar]
- 58.Ge H. Y. Studies on the antidepressant effect and mechanisms of saikosaponina. Jilin University: 2010.. [Google Scholar]
- 59.Zhang Y. C., Huang S. J. Mechanism analysis for the antidepressant effect of Peony. Journal of Traditional Chinese Medicine. 2013;6(10):795–798. [Google Scholar]
- 60.Tang Xu., Dong B. L. Q., Gao R., Guan S. J. Exploration into the Preparation of Placebos Used in Chinese Medicinal Clinical Trial. Chinese Journal of Integrated Traditional and Western Medicine. 2009;29(7):656–658. [PubMed] [Google Scholar]
- 61.Huang Q. H., Xiao J., Sun Y. M., et al. Preparation Method and Quality Evaluation of Chinese Medicine Compound Placeboes. Journal of Traditional Chinese Medicine. 2015;56(15):1294–1297. [Google Scholar]