Abstract
Hippocampal damage in adult humans impairs episodic and semantic memory, whereas hippocampal damage early in life impairs episodic memory but leaves semantic learning relatively preserved. We have previously shown a similar behavioral dissociation in nonhuman primates. Hippocampal lesion in adult monkeys prevents allocentric spatial relational learning, whereas spatial learning persists following neonatal lesion. Here, we quantified the number of cells expressing the immediate-early gene c-fos, a marker of neuronal activity, to characterize the functional organization of the medial temporal lobe memory system following neonatal hippocampal lesion. Ninety minutes before brain collection, three control and four adult monkeys with bilateral neonatal hippocampal lesions explored a novel environment in order to activate brain structures involved in spatial learning. Three other adult monkeys with neonatal hippocampal lesions remained in their housing quarters. In unlesioned monkeys, we found high levels of c-fos expression in the intermediate and caudal regions of the entorhinal cortex, and in the perirhinal, parahippocampal and retrosplenial cortices. In lesioned monkeys, spatial exploration induced an increase in c-fos expression in the intermediate field of the entorhinal cortex, the perirhinal, parahippocampal and retrosplenial cortices, but not in the caudal entorhinal cortex. These findings suggest that different regions of the medial temporal lobe memory system may require different types of interaction with the hippocampus in support of memory. The caudal perirhinal cortex, the parahippocampal cortex and the retrosplenial cortex may contribute to spatial learning in absence of functional hippocampal circuits, whereas the caudal entorhinal cortex may require hippocampal output to support spatial learning.
Keywords: hippocampus, entorhinal, perirhinal, parahippocampal, cingulate, retrosplenial
Introduction
Hippocampal damage in adult humans impairs episodic and semantic memory (Milner et al. 1998). In contrast, while individuals with early hippocampal damage exhibit strong deficits in episodic memory, semantic learning abilities are relatively preserved (Vargha-Khadem et al. 1997). Interestingly, experimental studies of hippocampus-dependent spatial memory in monkeys have produced comparable behavioral results (Banta Lavenex et al. 2006; Lavenex et al. 2007). Such similarities highlight the benefit of using nonhuman primates for identifying the neurobiological substrates underlying memory preservation following early brain damage. Indeed, since the initial description of hippocampal place cells in rats (O’Keefe and Dostrovsky 1971), the central role of the hippocampus in spatial memory has been demonstrated in rodents (Morris et al. 1982; Morris 2007), monkeys (Banta Lavenex et al. 2006; Banta Lavenex and Lavenex 2009) and humans (Abrahams et al. 1997; Banta Lavenex et al. 2014; Bartsch et al. 2011; Bohbot et al. 2004; Holdstock et al. 2000; Morris et al. 1996; Parslow et al. 2005; Parslow et al. 2004). In recent years, however, a functional and neuroanatomical distinction has been proposed between detailed, episode-specific spatial representations of experienced environments (analogous to episodic memory) and schematic, episode-free representations of the topography of the environment (analogous to semantic memory) (Moscovitch et al. 2006; Moscovitch et al. 2005). In a series of previous studies employing the same experimental design, we assessed the effects of hippocampal lesions on allocentric spatial relational learning tasks in which participants had the opportunity to learn spatial information over multiple repeated trials, thus enabling the creation of schematic, episode-free spatial representations. These studies revealed allocentric spatial memory deficits in a human patient with bilateral damage of the hippocampus acquired in adulthood (Banta Lavenex et al. 2014), and in adult monkeys with experimentally-induced hippocampal lesions (Banta Lavenex et al. 2006). In contrast, we found that allocentric spatial relational learning over repeated trials was preserved in monkeys that received neonatal hippocampal lesions (Lavenex et al. 2007). In accordance with our findings, a recent study by Glavis-Bloom and colleagues revealed that adult monkeys that received neonatal hippocampal lesions can learn to discriminate preferred from non-preferred food locations, but they are impaired, as compared to controls, at using trial-unique information during spatial and visual working memory tasks (Glavis-Bloom et al. 2013). In sum, experimental evidence suggests that in monkeys, adult hippocampal lesions impair episode-free spatial learning, whereas the ability to learn episode-free spatial information may persist following early hippocampal lesion.
The hippocampus is considered to be the central structure of a brain network essential for the acquisition, maintenance and retrieval of all aspects of the allocentric, spatial relational coding of space (Banta Lavenex and Lavenex 2009; Banta Lavenex et al. 2014; Nadel 1991; Nadel and Hardt 2004; O’Keefe and Nadel 1978). However, together with the hippocampus, the entorhinal, parahippocampal, parietal, posterior cingulate and retrosplenial cortices form a network of brain regions which contribute to spatial learning, memory and navigation (Aguirre and D’Esposito 1999; Bohbot et al. 2015; Burgess et al. 2002; Howard et al. 2014). We hypothesized, therefore, that cortical regions normally involved in the processing of spatial information, and which are characterized by direct connections with the hippocampal formation (including the entorhinal cortex (Amaral and Lavenex 2007)), may undergo a functional reorganization to enable the acquisition of these memories following neonatal hippocampal damage (Lavenex et al. 2007).
In the current study, we aimed to provide experimental evidence of the functional organization of the medial temporal lobe memory system following neonatal hippocampal damage in monkeys. We quantified the number of cells expressing the protein coded by the immediate-early gene c-fos, a reliable marker of neuronal activity (Guzowski et al. 2001), to characterize the patterns of neural activity within the different layers of distinct regions of the entorhinal, perirhinal, parahippocampal, posterior cingulate and retrosplenial cortices, induced by the exploration of a novel environment.
Materials and methods
Animals
Eleven macaque monkeys (Macaca mulatta) were used for this study. Monkeys were naturally born from multiparous mothers and raised at the California National Primate Research Center (CNPRC). All experimental procedures were approved by the Institutional Animal Care and Use Committee of the University of California, Davis, and were conducted in accordance with the National Institutes of Health guidelines for the use of animals in research.
Eight experimentally lesioned animals (5F, 3M; 9.1 to 9.4 years of age at the end of the experiment) were part of a longitudinal study on the effects of neonatal damage to the amygdala or hippocampus on the development of social behavior. Comprehensive rearing history and subject selection of the neonatal-lesioned animals was described previously (Bauman et al. 2004b, a; Bauman et al. 2006). Three monkeys (3F; 9.2 to 9.5 years) served as unoperated control monkeys. They were maternally reared in 2,000 m2 outdoor enclosures and lived in large social groups until their inclusion in this study, approximately three months before brain acquisition, when they were moved into indoor cages.
Lesion surgeries
Selective, bilateral lesions of the hippocampus were performed 12–16 days after birth. Detailed descriptions of these procedures can be found in (Bauman et al. 2004b, a). Briefly, all monkeys underwent presurgical magnetic resonance imaging in order to define the exact lesion coordinates for each individual monkey. All surgical procedures were performed aseptically at the CNPRC, with the support of veterinarians specialized in primate medicine. Monkeys were first anesthetized with ketamine hydrochloride (15 mg/kg, i.m.) and medetomidine (25–50 μg/kg, i.m.). A stable level of anesthesia was maintained throughout surgery with a combination of isoflurane (1.0%, inhalation; varied as needed to maintain an adequate level of anesthesia) and intravenous infusion of fentanyl (7–10 μg/kg/hr, i.v.). Monkeys were ventilated, and their vital signs were monitored throughout surgery. Simultaneous bilateral ibotenic acid (10 mg/ml in 0.1 M PBS; Biosearch Technologies, Novato, CA) injections were placed into the hippocampus using 10 μl Hamilton syringes (26 gauge beveled needles) at a rate of 0.2 μl/min. Lesion extent has been previously described in (Lavenex et al. 2007) and (Chareyron et al. 2016). In addition, two weeks before brain acquisition, each monkey received up to five neuroanatomical tracer injections following protocols previously described (Lavenex et al. 2002, 2004).
Immediate early-gene expression and exploratory behavior
A behavioral procedure was performed to induce immediate-early gene expression in brain regions involved in spatial learning. In rats, novel environments have been shown to trigger exploratory behavior, and induce a pattern of expression of immediate-early genes similar to that induced by a spatial working memory task in the radial arm maze (Albasser et al. 2007). Control monkeys (n=3; ControlExplo) and half of the hippocampus-lesioned monkeys chosen randomly (n=4; LesionExplo) were exposed to a novel environment individually for 15 minutes (Fig. 1), and returned to their home cage for 90 minutes prior to perfusion. The novel exploration cage was 4.27 m long, 1.52 wide and 2.13 m high. The front and back panels were made of solid stainless steel panels, whereas the two sides and the ceiling were made of stainless steel wire mesh. Three 25 cm deep perches made of PVC plastic bars were placed at different heights (0.91 m, 1.22 m, 1.52 m) against the back stainless steel panel. Attached to the left side wire mesh wall was a wire mesh chute with a remote controlled door, through which monkeys entered and exited the room when prompted by the experimenter. Another perch made of PVC plastic bars was placed in front of the door to the chute. The black lines in Fig. 1 represent stainless steel bars forming the structural frame of the cage. They also served to delimitate the virtual zones used to score the monkeys’ locomotor activity while exploring the environment. Specifically, an observer standing 2 meters away from the right side of the cage observed the monkey through the wire mesh door. She used the Observer 5.0 software package (Noldus, Wageningen, NL) and our standard behavioral ethogram (Supplementary Table 1), in order to analyze each monkey’s behavior during the individual exploration session. In addition, she noted the position of the monkey every 15 seconds based on a coordinate system including 18 possible locations within the room (left, center, right; front or back; floor, perch, ceiling).
Fig. 1.
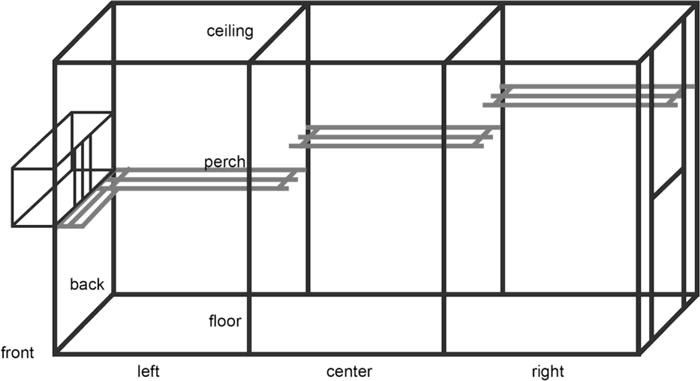
Schematic representation of the novel environment, in which monkeys spent 15 minutes in order to trigger the activation of brain regions involved in spatial learning. The cage was 4.27 m long, 1.52 wide and 2.13 m high. The front and back panels were made of solid stainless steel panels, whereas the two sides and the ceiling were made of stainless steel wire mesh. Three 25 cm deep perches (in grey) made of PVC plastic bars were placed at different heights (0.91 m, 1.22 m, 1.52 m) against the back stainless steel panel. Attached to the left side wire mesh wall was a wire mesh chute with a remote controlled door, through which monkeys entered and exited the cage when prompted by the experimenter. The black lines represent stainless steel bars forming the structural frame of the room. They also served to delimitate the virtual zones used to score the monkeys’ locomotor activity while exploring the environment (see main text for details).
The other four hippocampus-lesioned monkeys were taken out of their home cage and returned to it within a one-minute interval (LesionCage). This procedure was repeated after a 15-minute interval, after which the animals remained in their home cage for an additional 90 minutes prior to perfusion. This procedure was aimed at controlling for the non-specific activation of gene expression triggered by the transfer of the animals by the technical staff in and out of their home cages. The 90-minute delay between behavioral induction and perfusion was chosen based on prior research showing that this is an optimal delay for the detection of c-fos proteins (Rouiller 1997; Shires and Aggleton 2008). Because one of the LesionCage monkeys exhibited high-levels of non-specific c-fos expression, it was removed from subsequent analyses of c-fos expression, but was included in the volumetric analyses of brain regions.
Brain acquisition
All monkeys were deeply anesthetized with an intravenous injection of sodium pentobarbital (50 mg/kg; Fatal-Plus, Vortech Pharmaceuticals, Dearborn, MI) and perfused transcardially with 1% and then 4% paraformaldehyde in 0.1 M phosphate buffer (PB; pH 7.4) following protocols previously described (Lavenex et al. 2009). Coronal sections were cut using a freezing, sliding microtome (Microm HM 450, Microm International, Germany) in eight series at 30 μm.
Nissl staining with thionin
One series of sections were collected in 10% formaldehyde solution in 0.1 M PB (pH 7.4) and postfixed at 4°C for 4 weeks prior to Nissl staining with thionin. All other series were collected in tissue collection solution (TCS) and kept at −70°C until further processing (see below for c-fos immunohistochemistry). The procedure for Nissl-stained sections followed our standard laboratory protocol (Lavenex et al. 2009). Briefly, sections were taken from the 10% formaldehyde solution, thoroughly washed, mounted on gelatin-coated slides, and air-dried overnight at 37°C. Sections were then defatted 2 × 2 hours in a mixture of chloroform/ethanol (1:1, vol.), partially rehydrated and air-dried overnight at 37°C. Sections were then fully rehydrated and stained 20 seconds in a 0.25% thionin solution (Fisher Scientific, Waltham, MA, USA; T-409), dehydrated, and coverslipped with DePeX (VWR chemicals, Radnor, PA, USA; 361254D).
c-fos immunohistochemistry
We used standard immunohistochemical procedures to detect the expression of the protein c-fos in neurons that were activated during the exploration of the novel environment (Fig. 2). One series of sections (960 μm apart) was taken from TCS and rinsed for 3 × 5 min in 0.1 M phosphate-buffered saline containing 0.2% Triton X-100 (PBST, pH 7.4; Sigma-Aldrich, St Louis, MO, USA; 9002-93-1), incubated for 25 min in 0.3% H2O2 in PBST, rinsed for 4 × 5 min in 0.1 M PBST and incubated for 4 h in a blocking solution comprised of 3% normal goat serum (VWR chemicals, Radnor, PA, USA; CHMCS26) in 0.1 M PBST at room temperature. Sections were then incubated for 48 h in primary antiserum (rabbit polyclonal antibody anti-c-fos PC38; Calbiochem, San Diego, CA, USA; at 1:20,000 in 0.1 M PBST) at 4°C, rinsed for 5 × 5 min at room temperature in 0.1 M PBST, incubated for 2 h in secondary antiserum (biotinylated goat anti-rabbit IgG; Vector Laboratories, Burlingame, CA, USA; BA-1000; at 1:2,000 in 0.1 M PBST), rinsed for 5 × 5 min in 0.1 M PBST, incubated for 1.5 h in avidin-biotin complex (HRP-Basic IHC kit, ImmunoBioScience Corp., Mukilteo, WA; 8106) in 0.1 M PBST at room temperature, rinsed for 3 × 5 min in 0.1 M PBST, rinsed for 2 × 5 min in 0.01 M Tris buffer and incubated for 90 sec in a 0.05% diaminobenzidine (Sigma-Aldrich, St Louis, MO, USA, D5905) ⁄ 0.04% H2O2 solution. Sections were then rinsed, mounted onto gelatin-coated slides, defatted and coverslipped with DePeX (VWR chemicals, Radnor, PA, USA; 361254D).
Fig. 2.
Photomicrograph of c-fos positive cells, as they could be observed during the quantitative analyses, using a 20 X objective (N.A. 0.5) on a Nikon Eclipse 80i microscope (Nikon Instruments, Melville, NY, USA) linked to PC-based StereoInvestigator 11.0 (MBF Bioscience, Williston, VT, USA). Scale bar: 25 μm.
Data analysis
c-fos labeled cell counts
c-fos positive cells were plotted using a 20 X objective (N.A. 0.5) on a Nikon Eclipse 80i microscope (Nikon Instruments, Melville, NY, USA) linked to PC-based StereoInvestigator 11.0 (MBF Bioscience, Williston, VT, USA). The number of c-fos positive cells was estimated in the right or in the left hemisphere only (choice of side was made pseudorandomly, with half of the estimates made on the right hemisphere), in the entire entorhinal, perirhinal, parahippocampal, posterior cingulate, and retrosplenial cortices (i.e., cortical areas thought to be normally involved in the processing of spatial information, and which have direct connections with the hippocampal formation). We counted every c-fos labeled cell within each layer of a particular structure of interest defined from adjacent Nissl-stained sections, in every section processed for the detection of c-fos expression (960 μm apart).
Definition of cortical areas
Nissl-stained sections adjacent to c-fos labeled sections were used to delineate the regions of interest, including individual layers. We used Nissl-stain-based descriptions of the cytoarchitectonic organization of the entorhinal cortex (Amaral et al. 1987), perirhinal and parahippocampal cortices (Suzuki and Amaral 2003), and posterior cingulate and retrosplenial cortices (Kobayashi and Amaral 2000). For the entorhinal cortex, we followed the description of Amaral et al. (1987) and defined Eo as the olfactory field of the entorhinal cortex, Er as the rostral entorhinal cortex, Ei as the intermediate entorhinal cortex, El as the lateral entorhinal cortex (which included areas Elr and Elc), and Ecaudal as the caudal portion of the entorhinal cortex which included the fields Ec (caudal entorhinal cortex) and Ecl (caudal limiting field) as originally defined by Amaral et al. (1987). For the perirhinal cortex, we followed the description of Amaral et al. (1987) and Suzuki and Amaral (2003) and considered areas 35, 36d, 36r (which included areas 36rm and 36rl) and 36c (which included areas 36cm and 36cl). For the parahippocampal cortex, we followed the description of Suzuki and Amaral (2003) and considered areas TH and TF, which included areas TFm and TFl. For the posterior cingulate cortex, we followed the description of Kobayashi and Amaral (2000) and defined area 23, which comprises areas 23e, 23i and 23v. For the retrosplenial cortex, we followed the description of Kobayashi and Amaral (2000) and included areas 29, which comprises areas 29m and 29l, and area 30, which comprises areas 30 and 30v. We estimated the volume of each brain region using manual tracing performed on Nissl-stained coronal sections. We multiplied the areal measurements, derived from StereoInvestigator 11.0 (MBF Bioscience, Williston, VT, USA), by the section cutting thickness (30 μm) and the inverse sampling fraction to calculate the volumes.
Cortical unfolded maps
Two-dimensional unfolded maps representing the distribution of c-fos positive neurons were generated using Matlab R2012b (MathWorks, Inc., Natick, MA, USA). The limit between layer III and layer IV (or V, if IV is not present) was used as a reference to divide the cortex in 960 μm wide columns, in which c-fos positive neurons were counted, per layer, in order to build unfolded maps. The rhinal sulcus in the temporal lobe and the callosal sulcus in the cingulate gyrus were used as recognizable landmarks to align the sections along the mediolateral axis. Detailed description of the procedures to construct unfolded maps can be found in (Suzuki and Amaral 1996).
Statistical analysis
The same experimenter (L.J.C.) performed all neuron plotting and data analyses. Statistical analyses were confirmed by one of the senior authors (P.L.). We performed analyses of variance (ANOVAs) with the experimental groups (ControlExplo, LesionExplo, LesionCage) as factors on the numbers of c-fos positive cells in different brain regions, as these data were normally distributed. Post-hoc analyses were performed with the Fisher-LSD test when the ANOVA F ratio was significant and thus controlling for Type I error rate (Carmer and Swanson 1973). Significance level was set at P < 0.05 for all analyses. Since the lesion status was obvious, only the exploring vs non-exploring lesioned groups could be coded for blind analysis; the code was broken after completion of the analyses.
Results
Volume of brain regions
The volume of the hippocampal formation (Fig. 3), including the dentate gyrus, the hippocampal fields (CA3, CA2, CA1), the subiculum, presubiculum and parasubiculum (but excluding the entorhinal cortex), differed between control and lesioned animals (t(9) = 13.436 ; P < 0.001). As planned by the experimental design, it was larger in control monkeys than in lesioned monkeys, but importantly, it did not differ between LesionExplo and LesionCage monkeys (t(6) = 0.362 ; P = 0.730). The same differences were observed when considering each subfield separately (Fig. 3): dentate gyrus (t(9) = 6.484 ; P < 0.001), CA3 (t(9) = 11.787 ; P < 0.001), CA2 (t(9) = 9.378 ; P < 0.001), CA1 (t(9) = 10.129 ; P < 0.001), subiculum (t(9) = 19.481 ; P < 0.001), presubiculum (t(9) = 4.698 ; P = 0.001), parasubiculum (t(9) = 2.332 ; P = 0.045). There was no difference between groups in the volume of the entorhinal cortex (t(9) = 0.333 ; P = 0.746), posterior cingulate cortex (t(9) = 0.162 ; P = 0.875), or retrosplenial cortex (t(9) = 0.663 ; P = 0.524). In contrast, the volume of the perirhinal cortex was larger in lesioned monkeys than in controls (t(9) = 2.828 ; P = 0.02). Although the parahippocampal cortex was obviously damaged in some hippocampus-lesioned monkeys, the extent of this extraneous damage varied between animals, and the difference between groups in the volume of the parahippocampal cortex was not statistically significant (t(9) = 1.454 ; P = 0.18).
Fig. 3.
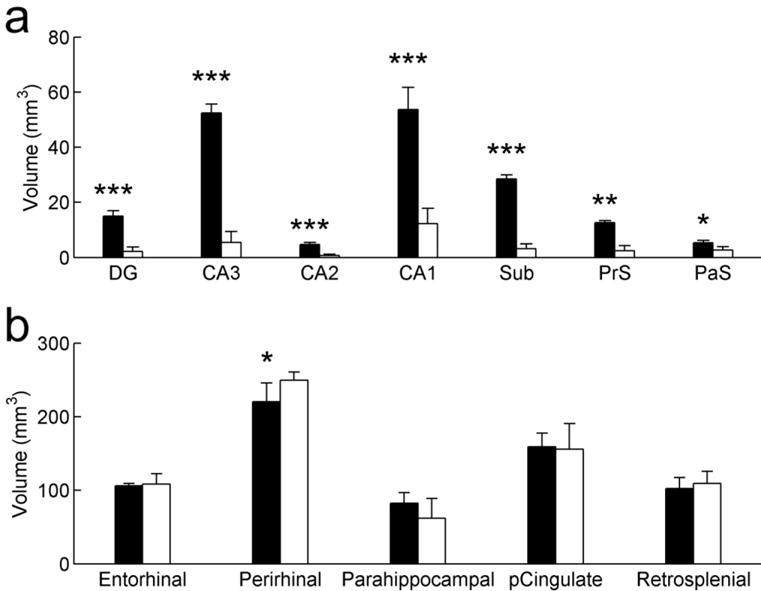
Volumes of the different regions of the hippocampal formation (a) and cortical areas (b) analyzed in the current study. Control monkeys in black; Hippocampal-lesioned monkeys in white. Average ± SD; *: P < 0.05; **: P < 0.01; ***: P < 0.001.
Exploratory behavior
We used the Observer 5.0 software package (Noldus, Wageningen, NL) and our standard behavioral ethogram (Bliss-Moreau et al. 2017) (Supplementary Table 1), in order to analyze each monkey’s behavior during the individual exploration session. Quantitative analyses of exploratory behavior demonstrated that both groups of animals were motivated to explore the novel environment. Although there was a tendency for hippocampus-lesioned monkeys to be more active than controls, as shown previously (Lavenex et al. 2007), this trend was not statistically significant. During the 15-minute exploration session, control monkeys locomoted for 9.0 ± 3.7 minutes, whereas lesioned monkeys locomoted for 10.1 ± 1.9 minutes (t(5) = 0.532, P = 0.617). Control monkeys crossed the borders between virtual zones subdividing the novel environment 21.7 ± 6.7 times, whereas lesioned monkeys crossed these borders 34.3 ± 13.3 times (t(5) = 1.483, P = 0.198). There was no difference between groups in the positions occupied by monkeys within the environment. We did not find any group differences in the frequency of exploration of the cage (combined manual and oral exploration: t(5) = 0.441, P = 0.677), or in stereotypic behaviors (t(5) = 1.265, P = 0.262).
Number of c-fos positive cells
Entorhinal cortex
The number of c-fos positive cells counted across all layers and subdivisions of the entorhinal cortex differed between groups (Fig. 4a, 5; F(2,7) = 5.257, P = 0.040). It was higher in ControlExplo than in LesionCage monkeys (P = 0.015), but the difference between ControlExplo and LesionExplo monkeys did not reach the predefined level of statistical significance (P = 0.061). The number of c-fos positive cells did not differ between LesionExplo and LesionCage monkeys (P = 0.280). However, a more detailed analysis of c-fos expression in the different layers of distinct subdivisions of the entorhinal cortex revealed different patterns of activation (Fig. 4a, 5; Supplementary Table 2).
Fig. 4.
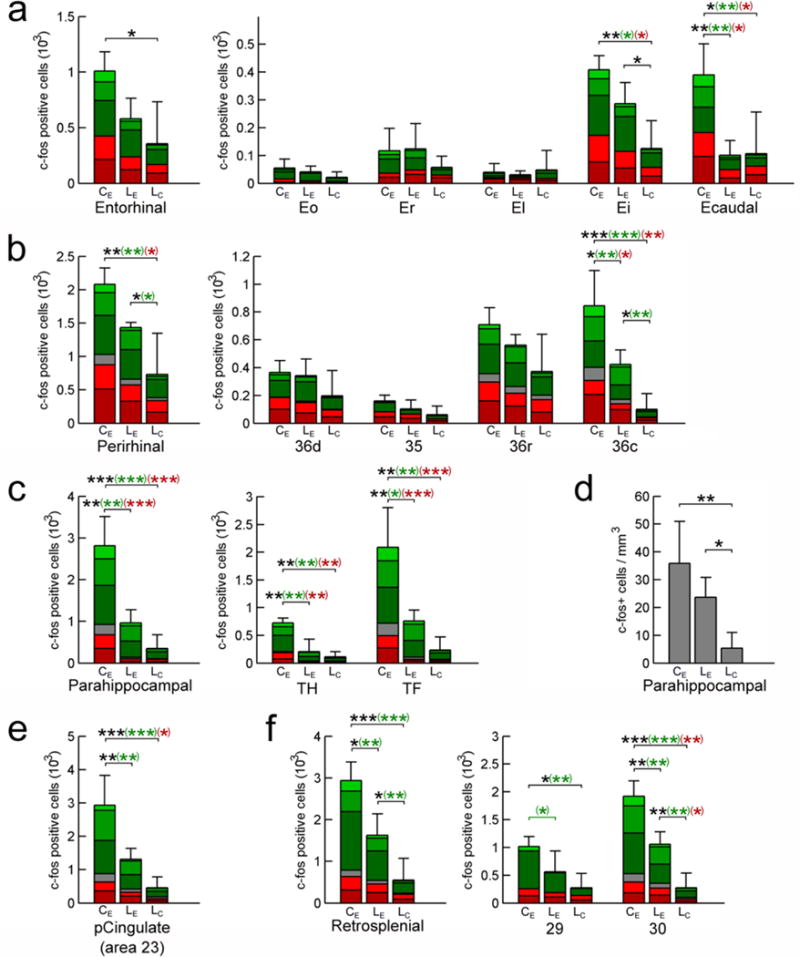
Number of c-fos positive cells: entorhinal cortex (a), perirhinal cortex (b), parahippocampal cortex (c), posterior cingulate cortex (e), and retrosplenial cortex (f). Panel (d): density of c-fos positive cells in the parahippocampal cortex (across regions and layers). ControlExplo (CE), LesionExplo (LE) and LesionCage (LC) monkeys. Stacked bars represent the number of c-fos positive cells in the six cortical layers (light green: layer I; green: layer II; dark green: layer III; gray: layer IV (not present in entorhinal cortex); light red: layer V; dark red: layer VI). Average ± SD; *: P < 0.05; **: P < 0.01; ***: P < 0.001; in black: all layers; in green: superficial layers (I, II, III); in red: deep layers (V, VI).
Fig. 5.
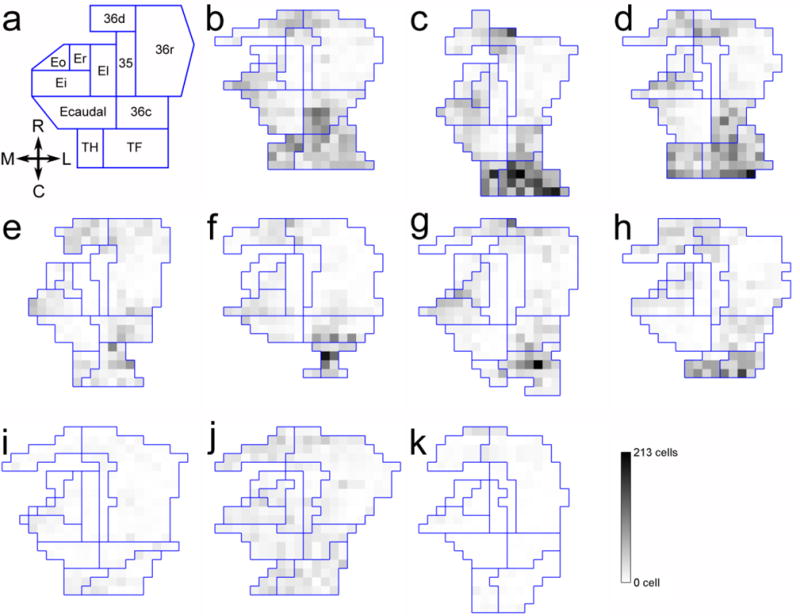
Two-dimensional unfolded map representations of the number of c-fos positive cells in the entorhinal cortex (areas Eo, Er, El, Ei, Ecaudal), perirhinal cortex (areas 35, 36d, 36r, 36c) and parahippocampal cortex (TH, TF). a: Unfolded map showing the different subdivisions considered in the study. b–d. ControlExplo monkeys. e–h: LesionExplo monkeys. i–k: LesionCage monkeys.
In areas Eo, Er and El, there were no differences in the number of c-fos positive cells between groups when considering all cortical layers, the superficial layers or the deep layers.
In area Ei, there was a difference in the number of c-fos positive cells between groups when considering all cortical layers. There were more c-fos positive cells in ControlExplo and LesionExplo monkeys than in LesionCage monkeys, whereas there was no difference between ControlExplo and LesionExplo monkeys. There were also group differences in the number of c-fos positive cells in the superficial layers of Ei. There were more c-fos positive cells in ControlExplo than in LesionCage monkeys, but the differences between LesionExplo monkeys and either ControlExplo or LesionCage monkeys did not reach significance. In the deep layers, there were more c-fos positive cells in ControlExplo than in LesionCage monkeys, whereas there was no difference between LesionExplo and either ControlExplo or LesionCage monkeys. Altogether, these findings indicate that the exploration of a novel environment was associated with neuronal activation in area Ei of both ControlExplo and LesionExplo monkeys.
In Ecaudal, there was a difference in the number of c-fos positive cells between groups when considering all cortical layers. There were more c-fos positive cells in ControlExplo monkeys than in LesionExplo or LesionCage monkeys, and importantly, there was no difference between LesionExplo and LesionCage monkeys. Similar results were obtained when the group comparisons considered the number of c-fos positive cells located in the superficial (I, II, III) or deep (V, VI) layers. These findings indicate that the exploration of a novel environment was not associated with an increase in neuronal activation in the caudal portion of the entorhinal cortex in hippocampus-lesioned monkeys.
Perirhinal cortex
The number of c-fos positive cells across all layers and subdivisions of the perirhinal cortex differed between groups (Fig. 4b, 5; F(2,7) = 10.582; P = 0.008). It was higher in ControlExplo than in LesionCage monkeys (P = 0.002); it was also higher in LesionExplo than in LesionCage monkeys (P = 0.038), whereas the difference between ControlExplo and LesionExplo monkeys had a probability value of P = 0.050. However, similar to what was observed in the entorhinal cortex, a detailed analysis of c-fos expression in different layers of distinct subdivisions of the perirhinal cortex revealed different patterns of activation (Fig. 4b, 5; Supplementary Table 3).
In areas 35, 36d and 36r, there were no differences in the number of c-fos positive cells between groups when considering all cortical layers, the superficial layers, or the deep layers.
In contrast, in area 36c, there were group differences in the number of c-fos positive cells across all cortical layers. There were more c-fos positive cells in ControlExplo monkeys than in LesionExplo and LesionCage monkeys. There were also more c-fos positive cells in LesionExplo than in LesionCage monkeys. Similarly, in the superficial layers, there were more c-fos positive cells in ControlExplo monkeys than in LesionExplo and LesionCage monkeys. There were also more c-fos positive cells in LesionExplo than in LesionCage monkeys. In layer IV, there were more c-fos positive cells in ControlExplo monkeys than in LesionExplo and LesionCage monkeys, but no difference between LesionExplo and LesionCage monkeys. In the deep layers, there were more c-fos positive cells in ControlExplo monkeys than in LesionExplo and LesionCage monkeys, and no difference between LesionExplo and LesionCage monkeys. In sum, although the exploration of a novel environment was associated with an increase in c-fos positive cells in area 36c of LesionExplo monkeys, c-fos expression was not as high as that observed in ControlExplo monkeys. Interestingly, the c-fos expression observed in LesionExplo monkeys appeared more prominent in the superficial layers, since the difference with the LesionCage monkeys was statistically significant in the superficial layers but not in the deep layers (Supplementary Table 3).
Parahippocampal cortex
The number of c-fos positive cells across all layers and subdivisions of the parahippocampal cortex differed between groups (Fig. 4c, 5; F(2,7) = 23.813; P = 0.001). It was higher in ControlExplo than in LesionExplo (P = 0.001) and LesionCage (P < 0.001) monkeys; it did not differ between LesionExplo and LesionCage monkeys (P = 0.121). A detailed analysis of c-fos expression revealed the same patterns of activation across the different layers of areas TH and TF (Fig. 4c, 5; Supplementary Table 4).
However, it is important to note that the parahippocampal cortex was inadvertently damaged in some lesioned monkeys. And, although the volume of the parahippocampal cortex did not differ statistically between groups, we performed an analysis of the density of c-fos positive cells in the parahippocampal cortex. We found that the density of c-fos positive cells across all layers and subdivisions of the parahippocampal cortex differed between groups (Fig. 4d; Supplementary Table 4; F(2,7) = 7.492; P = 0.018). It was higher in ControlExplo than in LesionCage (P = 0.006) monkeys; it was also higher in LesionExplo than in LesionCage (P = 0.043) monkeys. In contrast, it did not differ between ControlExplo and LesionExplo monkeys (P = 0.146). The density analysis is interesting and important to consider, since it suggests that the exploration of a novel environment may have induced greater c-fos expression in the parahippocampal cortex of LesionExplo monkeys if this region had not been damaged by the lesion procedure.
Posterior cingulate cortex
The number of c-fos positive cells across all layers of area 23 differed between groups (Fig. 4e, 6; F(2,7) = 15.983; P = 0.002). It was higher in ControlExplo than in LesionExplo and LesionCage monkeys; it did not differ between LesionExplo and LesionCage monkeys (Supplementary Table 4). Similar differences between groups were observed when the analyses considered the number of c-fos positive cells located in the superficial layers or in layer IV. In the deep layers, there were more c-fos positive cells in ControlExplo monkeys than in LesionCage monkeys, but it did not differ between LesionExplo and LesionCage monkeys, or between LesionExplo and ControlExplo monkeys.
Fig. 6.
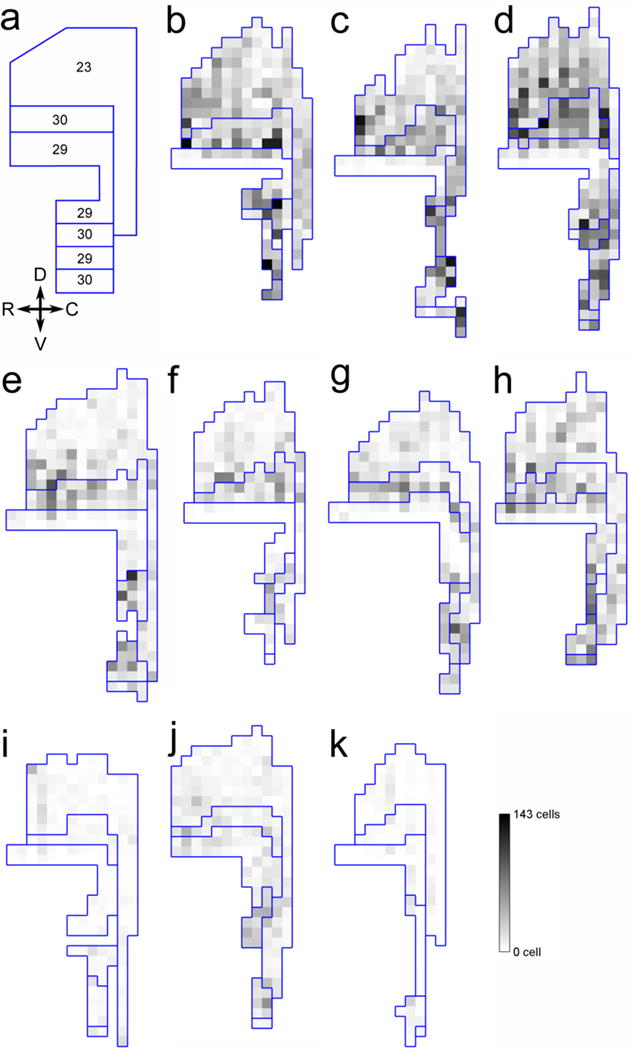
Two-dimensional unfolded map representations of the number of c-fos positive cells in the posterior cingulate cortex (area 23) and retrosplenial cortex (areas 29, 30). a: Unfolded map showing the different subdivisions considered in the study. b–d. ControlExplo monkeys. e–h: LesionExplo monkeys. i–k: LesionCage monkeys.
Retrosplenial cortex
The number of c-fos positive cells across all layers and subdivisions of the retrosplenial cortex differed between groups (Fig. 4f, 6; F(2,7) = 17.299; P = 0.002). It was higher in ControlExplo than in LesionCage monkeys (P = 0.001); it was also higher in ControlExplo than in LesionExplo monkeys (P = 0.011), as well as in LesionExplo monkeys as compared to LesionCage monkeys (P = 0.026). Moreover, similar to what was observed in the entorhinal and perirhinal cortices, a detailed analysis of c-fos expression in different layers of areas 29 and 30 revealed different patterns of activation (Fig. 4f, 6; Supplementary Table 4).
In area 29, there was a difference in the number of c-fos positive cells between groups when considering all cortical layers. It was higher in ControlExplo than in LesionCage monkeys; it did not differ between LesionExplo and ControlExplo or LesionCage monkeys. Similarly, there were group differences in the number of c-fos positive cells in the superficial layers (molecular layer + external pyramidal layer). There were more c-fos positive cells in ControlExplo monkeys than in LesionExplo and LesionCage monkeys. The number of c-fos positive cells did not differ between LesionExplo and LesionCage monkeys. In contrast, there were no group differences in the number of c-fos positive cells in the deep layers of area 29 (internal pyramidal layer + polymorphic layer).
In area 30, there was a difference in the number of c-fos positive cells between groups when considering all cortical layers. It was higher in ControlExplo than in LesionCage monkeys; it was also higher in ControlExplo than in LesionExplo monkeys, as well as in LesionExplo monkeys as compared to LesionCage monkeys. Similarly, there were group differences in the number of c-fos positive cells in the superficial layers. It was higher in ControlExplo than in LesionCage monkeys; it was also higher in ControlExplo than in LesionExplo monkeys, as well as in LesionExplo monkeys as compared to LesionCage monkeys. There were also group differences in the number of c-fos positive cells in layer IV. It was higher in ControlExplo than in LesionCage monkeys; it was also higher in ControlExplo than in LesionExplo monkeys, as well as in LesionExplo monkeys as compared to LesionCage monkeys. In addition, there were group differences in the number of c-fos positive cells in the deep layers. It was higher in ControlExplo than in LesionCage monkeys; it was also higher in LesionExplo than in LesionCage monkeys. In contrast, it did not differ between LesionExplo and ControlExplo monkeys.
In sum, although the exploration of a novel environment led to an increased activation of neurons in the retrosplenial cortex of LesionExplo monkeys, in particular in area 30, the number of c-fos positive cells was not as high as that observed in ControlExplo monkeys. Interestingly, the c-fos expression observed in LesionExplo monkeys appeared more prominent in the superficial layers.
Discussion
We have quantified the expression of the immediate-early gene c-fos, a marker of neuronal activity, to characterize the functional organization of the medial temporal lobe memory system following neonatal hippocampal lesions in monkeys. We focused our analysis on the cortical regions normally involved in the processing of spatial information (Aguirre and D’Esposito 1999; Bohbot et al. 2015; Burgess et al. 2002; Howard et al. 2014), which are also characterized by direct connections with the hippocampal formation (including the entorhinal cortex (Amaral and Lavenex 2007)). Following the exploration of a novel environment, we found c-fos expression in the intermediate and caudal fields of the entorhinal cortex, the caudal perirhinal, parahippocampal and retrosplenial cortices of control monkeys. In lesioned monkeys, the exploration of a novel environment was associated with an increase in c-fos expression, as compared to lesioned monkeys that did not explore, in the intermediate field of the entorhinal cortex, the caudal perirhinal, parahippocampal and retrosplenial cortices, but, strikingly, not in the caudal entorhinal cortex. As suggested by its neuroanatomical organization, different regions of the medial temporal lobe may require different types of interaction with the hippocampus in support of memory. In this discussion, we consider our results in light of the known structural organization of the medial temporal memory system, and the putative functions assigned to these different cortical areas.
Entorhinal cortex
The entorhinal cortex is considered to be the main gateway for bi-directional interaction between the neocortex and the hippocampal formation (Amaral and Lavenex 2007; Lavenex and Amaral 2000). However, if one follows the flow of information from the neocortex to the hippocampus, the perirhinal and parahippocampal cortices represent the first stage in the integration of multimodal polysensory information within the medial temporal lobe memory system. The perirhinal cortex projects to the rostral portion of the entorhinal cortex, which is thought to be more importantly involved in object information processing (Chrobak and Amaral 2007; Davachi 2006; Lavenex et al. 2002; Suzuki 2010; Suzuki and Amaral 1994b). The parahippocampal cortex projects to the caudal two thirds of the entorhinal cortex, which is thought to be more importantly involved in spatial information processing (Banta Lavenex and Lavenex 2009; Chrobak and Amaral 2007; Davachi 2006; Lavenex et al. 2002; Suzuki and Amaral 1994b; Suzuki 2010). Accordingly, we did not find many c-fos positive cells in areas Eo, Er or El of ControlExplo or LesionExplo monkeys following the exploration of a novel environment that did not contain objects or prominent proximal landmarks. This finding is consistent with a number of studies on the role of the rodent perirhinal and lateral entorhinal cortices (which corresponds to the rostral entorhinal cortex in monkeys) (Aggleton 2014; Knierim et al. 2014).
We also found that spatial exploration was not associated with an increase in c-fos expression in the caudal entorhinal cortex of hippocampus-lesioned monkeys, as compared to lesioned monkeys that did not explore, which suggests that the caudal entorhinal cortex may not be fully functional in the absence of functional hippocampal circuits. This finding is consistent with studies showing that the grid cells found in the medial entorhinal cortex of the rat (which corresponds to the caudal portion of the entorhinal cortex in monkeys) require an excitatory drive from the hippocampus in order to maintain their normal firing pattern (Bonnevie et al. 2013). In rats, hippocampal output is necessary for the emergence of grid cell patterns, and in the absence of a hippocampal signal, the grid cells become head directional. Monosynaptic inputs from the presubiculum and parasubiculum are thought to convey the directional information to principal neurons in all layers of the medial entorhinal cortex (Canto et al. 2012). Although some parts of the parasubiculum were somewhat preserved in hippocampus-lesioned monkeys in the current study, both the presubiculum and the parasubiculum were significantly lesioned. Such damage may have affected the directional output driving the head-direction activity of entorhinal cortex neurons, and thus contributed to the lack of increase in c-fos expression in the caudal portion of the entorhinal cortex of hippocampus-lesioned monkeys following spatial exploration.
Inadvertent damage to the parahippocampal cortex may also explain that spatial exploration was not associated with an increase in c-fos expression in the caudal entorhinal cortex of hippocampus-lesioned monkeys. However, we found that more neurons in area Ei, the intermediate division of the monkey entorhinal cortex, exhibited activation following spatial exploration in lesioned monkeys, albeit to a lesser extent than in ControlExplo monkeys. This activity is consistent with the described anatomical connections between the parahippocampal cortex and area Ei (Suzuki and Amaral 1994b), and contrasts with the finding that the caudal portion of the entorhinal cortex (including areas Ec and Ecl), which shares similar interconnections with the parahippocampal cortex, did not exhibit a similar level of activation. One possible explanation lies in the fact that area Ei is also highly interconnected with the caudal portion of the perirhinal cortex, area 36c, whereas areas Ec and Ecl are not. We found that many neurons in area 36c, in particular in the superficial layers, were activated following spatial exploration in both control and hippocampus-lesioned monkeys (see below).
Finally, despite differences in handling and testing experience between control and hippocampus-lesioned monkeys, a differential response to stress is unlikely to explain our current findings. Indeed, the projections from the amygdala to the entorhinal cortex are directed toward the rostral subdivisions (Pitkänen et al. 2002), in which we did not observe a differential c-fos expression between ControlExplo and LesionExplo monkeys. In contrast, the caudal portion of the entorhinal cortex, in which we observed differential c-fos expression between groups, does not receive significant projections from the amygdala.
In sum, the patterns of activation observed in the different layers of distinct regions of the entorhinal cortex suggest that a serial unidirectional model of information processing via two main pathways segregating object versus spatial information within the entorhinal cortex may be too simplistic. The entorhinal cortex should not be considered as a mere entryway, since hippocampal output may be necessary for the normal processing of spatial information in the caudal portion of the entorhinal cortex.
Perirhinal cortex
In hippocampus-lesioned monkeys, the exploration of a novel environment induced an increase in the number of c-fos positive cells in the caudal perirhinal cortex, in particular in the superficial layers (Fig. 4b; LesionExplo vs LesionCage). However, the level of c-fos expression in LesionExplo monkeys remained lower than in ControlExplo monkeys. The major cortical inputs to the perirhinal cortex arise from unimodal visual areas TE and rostral TEO, and from area TF of the parahippocampal cortex (Suzuki and Amaral 1994a). Work carried out in rats (Ennaceur et al. 1996; Olarte-Sanchez et al. 2015; Warburton and Brown 2015), monkeys (Murray and Richmond 2001), and humans (Clarke and Tyler 2014) has shown that the perirhinal cortex is implicated in object recognition memory. However, neuroanatomical work in monkeys has shown that the caudal perirhinal cortex constitutes an important interface between the perirhinal and parahippocampal cortices (Lavenex et al. 2004). Whereas the parahippocampal cortex projects to area 36c of the perirhinal cortex, the projections from the perirhinal cortex to the parahippocampal cortex are weak (Suzuki and Amaral 1994a). Based on the laminar organization of their interconnections, the perirhinal cortex is thought to be at a higher level than the parahippocampal cortex in the hierarchy of associational cortices supporting memory (Lavenex et al. 2004). Interestingly, as demonstrated in rats with late hippocampal lesion, the perirhinal cortex can function independently of the hippocampus in object recognition memory tasks (Kinnavane et al. 2014). Our data suggest that following neonatal hippocampal lesion, the caudal perirhinal cortex may be similarly involved in the processing of some basic spatial information, potentially provided by the projections from the parahippocampal cortex.
Detailed comparison of the levels of c-fos expression in the superficial and deep layers of area 36c in ControlExplo and LesionExplo monkeys revealed different patterns of activation. An increase in c-fos expression appeared limited to the superficial layers of area 36c in LesionExplo monkeys, as compared to LesionCage monkeys. However, as compared to ControlExplo monkeys, the number of c-fos positive neurons was lower in both the deep and superficial layers of area 36c in LesionExplo monkeys. Importantly, neurons located in the superficial layers of the perirhinal cortex project to the entorhinal cortex, whereas perirhinal neurons located in both superficial and deep layers receive feedback projections originating mainly from neurons located in layer V of the entorhinal cortex. Thus, although neurons of area 36c were activated following spatial exploration in LesionExplo monkeys, the lack of activity in the caudal entorhinal cortex in lesioned animals may have resulted in less c-fos activation in the perirhinal cortex of monkeys LesionExplo, as compared to ControlExplo, monkeys. This may thus suggest that hippocampal output relayed via the entorhinal cortex may normally contribute to information processing (and thus c-fos activation) in the caudal perirhinal cortex.
Parahippocampal cortex
In contrast to what we expected, we did not find a significant increase in the total number of c-fos positive cells in the parahippocampal cortex following spatial exploration in LesionExplo as compared to LesionCage monkeys. As described in the Results, the parahippocampal cortex was inadvertently damaged in hippocampus-lesioned monkeys. Although an obvious interpretation of this finding is that the absence of an increase in c-fos expression in the parahippocampal cortex was due to the lesion-induced damage, density measures revealed unambiguously that parahippocampal neurons were activated following the exploration of a novel environment in both ControlExplo and LesionExplo monkeys. Importantly, we found an increase in the density of c-fos positive cells in LesionExplo monkeys as compared to LesionCage monkeys, which suggests a significant and specific activation of the parahippocampal cortex in monkeys with early hippocampal lesions.
The parahippocampal cortex receives its strongest inputs from visuo-spatial areas in the dorsal stream, dorsolateral prefrontal cortex and retrosplenial cortex, in addition to unimodal inputs (Suzuki and Amaral 1994a). The parahippocampal cortex is thought to be involved in viewpoint-dependent spatial representations (Banta Lavenex and Lavenex 2009; Epstein 2008; Epstein and Vass 2014) and encoding of spatial context (Davachi 2006). In particular, the parahippocampal cortex might be directly involved with the processing of egocentric representations of space (Banta Lavenex and Lavenex 2009), and could enable the relatively imprecise learning of one but not two locations, as suggested by studies of spatial memory performance of a human amnesic patient (Banta Lavenex et al. 2014) and monkeys with bilateral hippocampal damage acquired in adulthood (Angeli et al. 1993). Accordingly, the level of c-fos expression in ControlExplo and LesionExplo monkeys is consistent with the putative role of the parahippocampal cortex in spatial learning and memory.
Alternatively, or perhaps additionally, the absence of hippocampal output, which was evidenced by a lack of increased activation in the caudal entorhinal cortex of LesionExplo monkeys (as compared to LesionCage monkeys), may have also engendered reduced neuronal activation in the parahippocampal cortex. This interpretation is consistent with the described topographical organization of medial temporal lobe structures. Indeed, the connections between the parahippocampal cortex and the entorhinal cortex are highly reciprocal, whereas the reciprocity between the perirhinal cortex and the entorhinal cortex is variable (Suzuki and Amaral 1994b). Similarly, the interconnections between the parahippocampal cortex and the neocortex are highly reciprocal, whereas the interconnections between the perirhinal cortex and the neocortex are asymmetric (Lavenex et al. 2002). As discussed previously (Lavenex et al. 2002), the perirhinal and parahippocampal cortices differ in their patterns of neocortical inputs and, thus, in the kind of information reaching these two cortices. It is possible that different types of information processed by these two cortical areas necessitate different types of feedback projections from the hippocampus. Spatial information, reaching preferentially the parahippocampal cortex, may require tight reciprocal connections, whereas visual object information reaching preferentially the perirhinal cortex may not (Kinnavane et al. 2014).
Posterior cingulate cortex
Spatial exploration did not induce a significant increase in the number of c-fos positive cells in the posterior cingulate cortex of hippocampus-lesioned monkeys. In these animals, the number of c-fos positive cells was particularly low in the superficial layers, as compared to ControlExplo monkeys, whereas there was no significant difference in the number of c-fos positive cells in the deep layers between ControlExplo and LesionExplo monkeys. In monkeys, the perirhinal cortex sends very sparse projections to the posterior cingulate cortex whereas the parahippocampal cortex gives rise to light to moderate projections (Lavenex et al. 2002). The efferent projections of the perirhinal and parahippocampal cortices are primarily directed to the superficial layers of the neocortex and are characteristic of projections of the feedback type (Lavenex et al. 2002). Thus, in LesionExplo monkeys, the reduced activation of the perirhinal and parahippocampal cortices may have contributed to the reduced, non-significant increase of activation of the posterior cingulate cortex following spatial exploration.
Retrosplenial cortex
Spatial exploration induced an increase in the number of c-fos positive cells in the retrosplenial cortex of hippocampus-lesioned monkeys. Nevertheless, this activation remained lower than that observed in ControlExplo monkeys. The differential pattern of activation was particularly visible in area 30, where different levels of expression between LesionExplo and LesionCage were significant for both deep and superficial layers, whereas the difference between LesionExplo and ControlExplo was only significant in the superficial layers, and across all layers, but not for the deep layers analyzed separately.
A number of studies have demonstrated the role of the retrosplenial cortex in spatial learning, memory and navigation in rats, monkeys and humans (Aggleton 2014; Auger et al. 2015; Epstein 2008; Epstein and Vass 2014; Vann et al. 2009). Germane to the current findings, a previous study in rats has shown that adult hippocampal lesion induces a decrease of c-fos expression in the retrosplenial cortex (Albasser, 2007). Moreover, using microPET technology on the same monkeys analyzed in the present study, we previously reported a decrease in basal metabolism in the retrosplenial cortex following neonatal hippocampal lesion (Machado et al., 2008). These behavioral and functional findings are consistent with the fact that the heaviest cortical projections to the retrosplenial cortex originate in the hippocampal formation, including the entorhinal cortex, subiculum, presubiculum and parasubiculum (Kobayashi and Amaral 2003). Neurons located in layers V and VI of the caudal entorhinal cortex send dense projections to the retrosplenial cortex, predominantly to layers I and II. The parahippocampal cortex also gives rise to moderate to heavy projections to the retrosplenial cortex, whereas the perirhinal cortex does not (Lavenex et al. 2002). Since the subiculum, presubiculum and parasubiculum were consistently damaged in our hippocampus-lesioned monkeys, and we did not observe any activation in the caudal entorhinal cortex in LesionExplo monkeys, it is not surprising to find a relatively decreased activation of the retrosplenial cortex in lesioned monkeys following spatial exploration, as compared to control monkeys. With respect to the observed c-fos labeling in the superficial layers of the retrosplenial cortex of hippocampus-lesioned monkeys following spatial exploration, it is likely that other inputs from the parahippocampal and parietal cortices, structures implicated in spatial information processing, may have contributed to this activation.
MTL memory system following neonatal hippocampal lesion
In the current study, we found a number of region- and layer-specific changes in neuronal activation following spatial exploration in hippocampus-lesioned monkeys. These findings indicate that several cortical regions which make up the medial temporal lobe memory network may be activated by, and therefore contribute to, spatial learning in absence of a functional hippocampus. Interestingly, the caudal entorhinal cortex, which is homologous to the medial entorhinal cortex in rats and thought to be essential for spatial information processing, did not exhibit an increase in neuronal activation following spatial exploration in hippocampus-lesioned monkeys. As discussed previously, these findings indicate that with respect to the processing of at least some types of spatial information, the entorhinal cortex may not function properly without a functioning hippocampus.
We found an increase in the volume of the perirhinal cortex in hippocampus-lesioned monkeys, which may suggest some structural and/or functional compensation mechanisms, the nature of which remains to be determined. However, we did not find a “smoking gun”, i.e., one or more regions in which activation was increased following exploration in monkeys with neonatal hippocampal lesion as compared to control monkeys, and which could account for the preserved context-free allocentric spatial relational memory capacities observed previously in these monkeys (Lavenex et al. 2007). One possibility may be that the exploration of a novel environment, which has been shown in rats to induce a pattern of c-fos expression similar to that induced by a spatial working memory task in the radial maze (Albasser et al. 2007), does not activate the brain structures involved in spatial learning in the same manner as our previously used spatial learning task with repeated trials (Lavenex et al. 2007). Nonetheless, we found significant yet reduced levels of neuronal activation following a single 15-min spatial exploration session in hippocampus-lesioned monkeys, as compared to unlesioned monkeys, in area Ei of the entorhinal cortex, in area 36c of the perirhinal cortex, in area TF of the parahippocampal cortex, and in area 30 of the retrosplenial cortex. Furthermore, we found an increase in the overall volume of the perirhinal cortex in hippocampus-lesioned as compared to unlesioned monkeys. These findings suggest that these regions may contribute to spatial information processing in the absence of a functioning hippocampus.
Conclusion
Our findings have important implications for the interpretation of studies reporting the effects of selective brain lesions, particularly when the lesion takes place in developing organisms. A lesion that, upon non-invasive MRI or even post-mortem histological examination, may appear restricted to a given brain area, may provoke functional changes in interconnected brain regions that are sensitive to deafferentation and which may exhibit covert functional pathology. Despite the overall hierarchy characterizing the medial temporal lobe memory system, the complex structural and functional organization of its afferent/efferent and feedforward/feedback connections makes it difficult to dissociate the functional contribution of any individual region considered in isolation. A more general network-based approach will thus likely be necessary to provide a comprehensive view of the functional organization of the medial temporal lobe memory system.
Supplementary Material
Table 1.
Behavioral Ethogram (Bliss-Moreau, 2017)
| Behavior | Description |
|---|---|
| States | |
| Close Social Interaction | |
| Extended Aggression | Animals bite, slap, chase, or grab at each other |
| Extended Contact | Animals are in physical contact |
| Extended Groom | Examination, picking, or licking of another animal’s fur or body |
| Extended Mount | Any instance of mounting |
| Extended Play | Continuous rough and tumble play and/or play threats, including playful chase |
| Proximity Zone | Focal animal is within the marked rectangle in front of the stimulus cage |
| Proximity | Focal animal is directly in front of the constrained animal |
| Non-Social States | |
| Nonsocial Activity | Animal remains out of all social states and Is locomotive with head up, actively engaged in the environment |
| Nonsocial Stationary | Animal remains out of all social states and Is non-locomotive with head up, actively engaged in the environment |
| Nonsocial Inactivity | Animal remains out of all social states with head down, not engaged in environment, often staring off into space |
| Extended Stereotypy | Focal animal is engaged in repetitive self-directed or motor stereotypic behavior |
| Events | |
| Communicative Signaling | |
| Affiliative | |
| Approach | Intentional movement within arm’s reach of another animal |
| Accept Approach | Animal remains within arm’s reach after the other animal approaches |
| Anogenital Exploration | Oral, visual, olfactory, or manual exploration of the other animals anogenital area |
| Contact | Physical contact between animals that is not aggressive |
| Coo | Clear, soft sounds, moderate in pitch and intensity; usually sounds like “whoooooo” |
| Follow | Intentional follow of another animal |
| Groom | Examination, picking, or licking of another animal’s fur or body |
| Grunt | Deep, muffled, low-intensity vocalization |
| Girney | Quiet, nasal whine, usually emitted during affiliative encounters |
| Lipsmack | Rapid lip movements with pursed or puckered lips, usually accompanied by smacking sounds |
| Huddle | Physical contact that involves one animal ventrally touching another animal |
| Jaw-Thrust | Rapid lipsmack and grimace and/or brow raise usually seen while the animal walks by or approaches and then leaves quickly |
| Joint Threat | Both animals threaten observer in unison |
| Mount | Mount that includes all of the following components: appropriate positioning of partner, hands on back, double foot clasp |
| Mount Attempt | Any component of a mouth that is attempted through the metal grille |
| Present Groom | Intentional, exaggerated presentation of a part of body to another animal |
| Present Neck | Presentation of neck to another animal |
| Present Rump | Rigid posture with rump and tail elevated and oriented toward another individual |
| Threat-Solicitation | Animal recruits the other animal in threatening the observer or another animal |
| Agonistic/“Aggression” | |
| Aggression | Grabbing, slapping, and biting of another animal |
| Aggressive Grunt | Low-pitched guttural sound, accompanied by a threat |
| Alarm Bark | Short, sharp sound |
| Cage Shake | Vigorous shaking of cage, or body slam against bars |
| Crooktail | Tail held up stiff in a “?” shape |
| Displacement | Physical movement in which an animal “takes the place” of another animal |
| Muzzle | Contact |
| Threat | Contains one or more of the following components: open mouth stare, head bobbing, ear flaps, bark vocalizations, or lunges |
| Submission/“Fear” | |
| Avoid | Animal leaves the area when the other animal comes near or is about to approach |
| Grimace | Exaggerated movement of lips such that lips are pulled back with teeth showing |
| Flee | Rapid, intentional movement away from another animal |
| Freeze | Stiff body posture without any movement for more than three seconds |
| Scream | High-pitched vocalization, with extreme high intensity; sounds like “eeeeeeeeee” |
| Exploration | |
| Manual | Exploration of the cage or environment with the hands |
| Oral | Exploration of the cage or environment with the mouth |
| Stress-Related | |
| Scratch | Scratches own body |
| Self-Groom | Use of hands to pick through or lick a fur or non-fur body part |
| Self-Shake | Vigorous shaking of the body |
| Tooth Grind | Repetitive audible rubbing of upper and lower teeth |
| Yawn | Yawn |
| Other | |
| Self Sex | Anogenital exploration of self |
| Stereotypic | |
| Self-Directed | Repetitive behavior acted on self, including: repetitive swaying back and forth, covering hand over eye or eye pokes, unusual holding of body part or limb, biting at oneself, self-strumming |
| Whole-Body | Repetitive motor behavior including: back flipping, hopping, twirling, swinging, or undirected movement with the same path repeated |
| Heat Twist | Animal twists neck in a dramatic display |
Note that a number of codes were not scored in the current study, since they represent social interaction with other monkeys.
Significance Statement.
We characterized the functional organization of the medial temporal lobe memory system following neonatal hippocampal lesions in nonhuman primates. After exploration of a novel environment, c-fos expression, a marker of neuronal activity, was found in the intermediate and caudal regions of the entorhinal cortex, the caudal perirhinal cortex, and the parahippocampal and retrosplenial cortices of control monkeys. In lesioned monkeys, spatial exploration induced increased c-fos expression in the intermediate field of the entorhinal cortex, the caudal perirhinal cortex, and the parahippocampal and retrosplenial cortices. These findings suggest that the caudal perirhinal, parahippocampal and retrosplenial cortices may contribute to spatial learning in the absence of functional hippocampal circuits, whereas the caudal entorhinal cortex may require hippocampal output to support spatial learning.
Acknowledgments
This research was supported by grants from the Swiss National Science Foundation (P00A-106701, PP00P3-124536, 310030_143956), the US National Institutes of Health (NIH; MH041479; NS16980); and conducted, in part, at the California National Primate Research Center (OD011107)
References
- Abrahams S, Pickering A, Polkey CE, Morris RG. Spatial memory deficits in patients with unilateral damage to the right hippocampal formation. Neuropsychologia. 1997;35(1):11–24. doi: 10.1016/s0028-3932(96)00051-6. [DOI] [PubMed] [Google Scholar]
- Aggleton JP. Looking beyond the hippocampus: old and new neurological targets for understanding memory disorders. Proc Biol Sci. 2014;281(1786) doi: 10.1098/rspb.2014.0565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aguirre GK, D’Esposito M. Topographical disorientation: a synthesis and taxonomy. Brain. 1999;122( Pt 9):1613–1628. doi: 10.1093/brain/122.9.1613. [DOI] [PubMed] [Google Scholar]
- Albasser MM, Poirier GL, Warburton EC, Aggleton JP. Hippocampal lesions halve immediate-early gene protein counts in retrosplenial cortex: distal dysfunctions in a spatial memory system. Eur J Neurosci. 2007;26(5):1254–1266. doi: 10.1111/j.1460-9568.2007.05753.x. [DOI] [PubMed] [Google Scholar]
- Amaral DG, Insausti R, Cowan WM. The entorhinal cortex of the monkey: I. Cytoarchitectonic organization. J Comp Neurol. 1987;264(3):326–355. doi: 10.1002/cne.902640305. [DOI] [PubMed] [Google Scholar]
- Amaral DG, Lavenex P. Hippocampal neuroanatomy. In: Amaral DG, Andersen P, Bliss T, Morris RGM, O’Keefe J, editors. The Hippocampus Book. Oxford University Press; Oxford: 2007. pp. 37–114. [Google Scholar]
- Angeli SJ, Murray EA, Mishkin M. Hippocampectomized monkeys can remember one place but not two. Neuropsychologia. 1993;31(10):1021–1030. doi: 10.1016/0028-3932(93)90030-4. [DOI] [PubMed] [Google Scholar]
- Auger SD, Zeidman P, Maguire EA. A central role for the retrosplenial cortex in de novo environmental learning. Elife. 2015;4 doi: 10.7554/eLife.09031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banta Lavenex P, Amaral DG, Lavenex P. Hippocampal lesion prevents spatial relational learning in adult macaque monkeys. J Neurosci. 2006;26(17):4546–4558. doi: 10.1523/JNEUROSCI.5412-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Banta Lavenex P, Lavenex P. Spatial memory and the monkey hippocampus: Not all space is created equal. Hippocampus. 2009;19(1):8–19. doi: 10.1002/hipo.20485. [DOI] [PubMed] [Google Scholar]
- Banta Lavenex PA, Colombo F, Ribordy Lambert F, Lavenex P. The human hippocampus beyond the cognitive map: Evidence from a densely amnesic patient. Front Hum Neurosci. 2014:1–18. doi: 10.3389/fnhum.2014.00711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartsch T, Dohring J, Rohr A, Jansen O, Deuschl G. CA1 neurons in the human hippocampus are critical for autobiographical memory, mental time travel, and autonoetic consciousness. Proc Natl Acad Sci U S A. 2011;108(42):17562–17567. doi: 10.1073/pnas.1110266108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bauman MD, Lavenex P, Mason WA, Capitanio JP, Amaral DG. The development of mother-infant interactions after neonatal amygdala lesions in rhesus monkeys. J Neurosci. 2004a;24(3):711–721. doi: 10.1523/JNEUROSCI.3263-03.2004. doi: 10.1523/JNEUROSCI.3263-03.200424/3/711 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bauman MD, Lavenex P, Mason WA, Capitanio JP, Amaral DG. The development of social behavior following neonatal amygdala lesions in rhesus monkeys. J Cogn Neurosci. 2004b;16(8):1388–1411. doi: 10.1162/0898929042304741. [DOI] [PubMed] [Google Scholar]
- Bauman MD, Toscano JE, Mason WA, Lavenex P, Amaral DG. The expression of social dominance following neonatal lesions of the amygdala or hippocampus in rhesus monkeys (Macaca mulatta) Behav Neurosci. 2006;120(4):749–760. doi: 10.1037/0735-7044.120.4.749. doi: 2006-09890-001 [pii] 10.1037/0735-7044.120.4.749. [DOI] [PubMed] [Google Scholar]
- Bliss-Moreau E, Moadab G, Santistevan A, Amaral DG. The effects of neonatal amygdala or hippocampus lesions on adult social behavior. Behav Brain Res. 2017;322(Pt A):123–137. doi: 10.1016/j.bbr.2016.11.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohbot VD, Allen JJ, Dagher A, Dumoulin SO, Evans AC, Petrides M, Kalina M, Stepankova K, Nadel L. Role of the parahippocampal cortex in memory for the configuration but not the identity of objects: converging evidence from patients with selective thermal lesions and fMRI. Front Hum Neurosci. 2015;9:431. doi: 10.3389/fnhum.2015.00431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohbot VD, Iaria G, Petrides M. Hippocampal function and spatial memory: evidence from functional neuroimaging in healthy participants and performance of patients with medial temporal lobe resections. Neuropsychology. 2004;18(3):418–425. doi: 10.1037/0894-4105.18.3.418. [DOI] [PubMed] [Google Scholar]
- Bonnevie T, Dunn B, Fyhn M, Hafting T, Derdikman D, Kubie JL, Roudi Y, Moser EI, Moser MB. Grid cells require excitatory drive from the hippocampus. Nat Neurosci. 2013;16(3):309–317. doi: 10.1038/nn.3311. [DOI] [PubMed] [Google Scholar]
- Burgess N, Maguire EA, O’Keefe J. The human hippocampus and spatial and episodic memory. Neuron. 2002;35(4):625–641. doi: 10.1016/s0896-6273(02)00830-9. [DOI] [PubMed] [Google Scholar]
- Canto CB, Koganezawa N, Beed P, Moser EI, Witter MP. All layers of medial entorhinal cortex receive presubicular and parasubicular inputs. J Neurosci. 2012;32(49):17620–17631. doi: 10.1523/JNEUROSCI.3526-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carmer SG, Swanson MR. Evaluation of 10 Pairwise Multiple Comparison Procedures by Monte-Carlo Methods. Journal of the American Statistical Association. 1973;68(341):66–74. doi: 10.2307/2284140. [DOI] [Google Scholar]
- Chareyron LJ, Amaral DG, Lavenex P. Selective lesion of the hippocampus increases the differentiation of immature neurons in the monkey amygdala. Proc Natl Acad Sci U S A. 2016;113(50):14420–14425. doi: 10.1073/pnas.1604288113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chrobak JJ, Amaral DG. Entorhinal cortex of the monkey: VII. intrinsic connections. J Comp Neurol. 2007;500(4):612–633. doi: 10.1002/cne.21200. [DOI] [PubMed] [Google Scholar]
- Clarke A, Tyler LK. Object-specific semantic coding in human perirhinal cortex. J Neurosci. 2014;34(14):4766–4775. doi: 10.1523/JNEUROSCI.2828-13.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davachi L. Item, context and relational episodic encoding in humans. Curr Opin Neurobiol. 2006;16(6):693–700. doi: 10.1016/j.conb.2006.10.012. [DOI] [PubMed] [Google Scholar]
- Ennaceur A, Neave N, Aggleton JP. Neurotoxic lesions of the perirhinal cortex do not mimic the behavioural effects of fornix transection in the rat. Behav Brain Res. 1996;80(1–2):9–25. doi: 10.1016/0166-4328(96)00006-x. [DOI] [PubMed] [Google Scholar]
- Epstein RA. Parahippocampal and retrosplenial contributions to human spatial navigation. Trends Cogn Sci. 2008;12(10):388–396. doi: 10.1016/j.tics.2008.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Epstein RA, Vass LK. Neural systems for landmark-based wayfinding in humans. Philos Trans R Soc Lond B Biol Sci. 2014;369(1635):20120533. doi: 10.1098/rstb.2012.0533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glavis-Bloom C, Alvarado MC, Bachevalier J. Neonatal hippocampal damage impairs specific food/place associations in adult macaques. Behav Neurosci. 2013;127(1):9–22. doi: 10.1037/a0031498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guzowski JF, Setlow B, Wagner EK, McGaugh JL. Experience-dependent gene expression in the rat hippocampus after spatial learning: A comparison of the immediate-early genes Arc, c-fos, and zif268. Journal of Neuroscience. 2001;21(14):5089–5098. doi: 10.1523/JNEUROSCI.21-14-05089.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holdstock JS, Gutnikov SA, Gaffan D, Mayes AR. Perceptual and mnemonic matching-to-sample in humans: contributions of the hippocampus, perirhinal and other medial temporal lobe cortices. Cortex. 2000;36(3):301–322. doi: 10.1016/s0010-9452(08)70843-8. [DOI] [PubMed] [Google Scholar]
- Howard LR, Javadi AH, Yu Y, Mill RD, Morrison LC, Knight R, Loftus MM, Staskute L, Spiers HJ. The hippocampus and entorhinal cortex encode the path and Euclidean distances to goals during navigation. Curr Biol. 2014;24(12):1331–1340. doi: 10.1016/j.cub.2014.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kinnavane L, Amin E, Horne M, Aggleton JP. Mapping parahippocampal systems for recognition and recency memory in the absence of the rat hippocampus. Eur J Neurosci. 2014;40(12):3720–3734. doi: 10.1111/ejn.12740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knierim JJ, Neunuebel JP, Deshmukh SS. Functional correlates of the lateral and medial entorhinal cortex: objects, path integration and local-global reference frames. Philos Trans R Soc Lond B Biol Sci. 2014;369(1635):20130369. doi: 10.1098/rstb.2013.0369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi Y, Amaral DG. Macaque monkey retrosplenial cortex: I. three-dimensional and cytoarchitectonic organization. J Comp Neurol. 2000;426(3):339–365. doi: 10.1002/1096-9861(20001023)426:3<339::aid-cne1>3.0.co;2-8. [DOI] [PubMed] [Google Scholar]
- Kobayashi Y, Amaral DG. Macaque monkey retrosplenial cortex: II. Cortical afferents. J Comp Neurol. 2003;466(1):48–79. doi: 10.1002/cne.10883. [DOI] [PubMed] [Google Scholar]
- Lavenex P, Amaral DG. Hippocampal-neocortical interaction: a hierarchy of associativity. Hippocampus. 2000;10(4):420–430. doi: 10.1002/1098-1063(2000)10:4<420::AID-HIPO8>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- Lavenex P, Lavenex PB, Amaral DG. Spatial relational learning persists following neonatal hippocampal lesions in macaque monkeys. Nat Neurosci. 2007;10(2):234–239. doi: 10.1038/nn1820. doi: nn1820 [pii] 10.1038/nn1820. [DOI] [PubMed] [Google Scholar]
- Lavenex P, Lavenex PB, Bennett JL, Amaral DG. Postmortem changes in the neuroanatomical characteristics of the primate brain: Hippocampal formation. J Comp Neurol. 2009;512(1):27–51. doi: 10.1002/cne.21906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lavenex P, Suzuki WA, Amaral DG. Perirhinal and parahippocampal cortices of the macaque monkey: projections to the neocortex. J Comp Neurol. 2002;447(4):394–420. doi: 10.1002/cne.10243. [DOI] [PubMed] [Google Scholar]
- Lavenex P, Suzuki WA, Amaral DG. Perirhinal and parahippocampal cortices of the macaque monkey: Intrinsic projections and interconnections. Journal of Comparative Neurology. 2004;472(3):371–394. doi: 10.1002/cne.20079. [DOI] [PubMed] [Google Scholar]
- Milner B, Squire LR, Kandel ER. Cognitive neuroscience and the study of memory. Neuron. 1998;20(3):445–468. doi: 10.1016/s0896-6273(00)80987-3. [DOI] [PubMed] [Google Scholar]
- Morris RG, Garrud P, Rawlins JN, O’Keefe J. Place navigation impaired in rats with hippocampal lesions. Nature. 1982;297(5868):681–683. doi: 10.1038/297681a0. [DOI] [PubMed] [Google Scholar]
- Morris RG, Pickering A, Abrahams S, Feigenbaum JD. Space and the hippocampal formation in humans. Brain research bulletin. 1996;40(5–6):487–490. doi: 10.1016/0361-9230(96)00146-3. [DOI] [PubMed] [Google Scholar]
- Morris RGM. Theories of hippocampal function. The Hippocampus Book. 2007:581–713. [Google Scholar]
- Moscovitch M, Nadel L, Winocur G, Gilboa A, Rosenbaum RS. The cognitive neuroscience of remote episodic, semantic and spatial memory. Curr Opin Neurobiol. 2006;16(2):179–190. doi: 10.1016/j.conb.2006.03.013. [DOI] [PubMed] [Google Scholar]
- Moscovitch M, Rosenbaum RS, Gilboa A, Addis DR, Westmacott R, Grady C, McAndrews MP, Levine B, Black S, Winocur G, Nadel L. Functional neuroanatomy of remote episodic, semantic and spatial memory: a unified account based on multiple trace theory. J Anat. 2005;207(1):35–66. doi: 10.1111/j.1469-7580.2005.00421.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray EA, Richmond BJ. Role of perirhinal cortex in object perception, memory, and associations. Curr Opin Neurobiol. 2001;11(2):188–193. doi: 10.1016/s0959-4388(00)00195-1. [DOI] [PubMed] [Google Scholar]
- Nadel L. The hippocampus and space revisited. Hippocampus. 1991;1(3):221–229. doi: 10.1002/hipo.450010302. [DOI] [PubMed] [Google Scholar]
- Nadel L, Hardt O. The spatial brain. Neuropsychology. 2004;18(3):473–476. doi: 10.1037/0894-4105.18.3.473. [DOI] [PubMed] [Google Scholar]
- O’Keefe J, Dostrovsky J. The hippocampus as a spatial map. Preliminary evidence from unit activity in the freely-moving rat. Brain Res. 1971;34(1):171–175. doi: 10.1016/0006-8993(71)90358-1. [DOI] [PubMed] [Google Scholar]
- O’Keefe J, Nadel L. The Hippocampus as a Cognitive Map. Clarendon Press; Oxford: 1978. [Google Scholar]
- Olarte-Sanchez CM, Amin E, Warburton EC, Aggleton JP. Perirhinal cortex lesions impair tests of object recognition memory but spare novelty detection. Eur J Neurosci. 2015;42(12):3117–3127. doi: 10.1111/ejn.13106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parslow DM, Morris RG, Fleminger S, Rahman Q, Abrahams S, Recce M. Allocentric spatial memory in humans with hippocampal lesions. Acta psychologica. 2005;118(1–2):123–147. doi: 10.1016/j.actpsy.2004.10.006. [DOI] [PubMed] [Google Scholar]
- Parslow DM, Rose D, Brooks B, Fleminger S, Gray JA, Giampietro V, Brammer MJ, Williams S, Gasston D, Andrew C, Vythelingum GN, Ioannou G, Simmons A, Morris RG. Allocentric spatial memory activation of the hippocampal formation measured with fMRI. Neuropsychology. 2004;18(3):450–461. doi: 10.1037/0894-4105.18.3.450. [DOI] [PubMed] [Google Scholar]
- Pitkänen A, Kelly JL, Amaral DG. Projections from the lateral, basal, and accessory basal nuclei of the amygdala to the entorhinal cortex in the macaque monkey. Hippocampus. 2002;12(2):186–205. doi: 10.1002/hipo.1099. [DOI] [PubMed] [Google Scholar]
- Rouiller EM. Acoustical Signal Processing in the Central Auditory System. Plemun Press; New York: 1997. Mapping activity in the adutiory pathway with c-fos; pp. 33–48. [Google Scholar]
- Shires KL, Aggleton JP. Mapping immediate-early gene activity in the rat after place learning in a water-maze: the importance of matched control conditions. Eur J Neurosci. 2008;28(5):982–996. doi: 10.1111/j.1460-9568.2008.06402.x. [DOI] [PubMed] [Google Scholar]
- Suzuki WA. Untangling memory from perception in the medial temporal lobe. Trends Cogn Sci. 2010;14(5):195–200. doi: 10.1016/j.tics.2010.02.002. [DOI] [PubMed] [Google Scholar]
- Suzuki WA, Amaral DG. Perirhinal and parahippocampal cortices of the macaque monkey: cortical afferents. J Comp Neurol. 1994a;350:497–533. doi: 10.1002/cne.903500402. [DOI] [PubMed] [Google Scholar]
- Suzuki WA, Amaral DG. Topographic organization of the reciprocal connections between the monkey entorhinal cortex and the perirhinal and parahippocampal cortices. J Neurosci. 1994b;14(3 Pt 2):1856–1877. doi: 10.1523/JNEUROSCI.14-03-01856.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki WA, Amaral DG. The construction of straight-line unfolded two-dimensional density maps of neuroanatomical projections in the monkey cerebral cortex. Neuroscience protocols. 1996;96:7–19. [Google Scholar]
- Suzuki WA, Amaral DG. Where are the perirhinal and parahippocampal cortices? A historical overview of the nomenclature and boundaries applied to the primate medial temporal lobe. Neuroscience. 2003;120:893–906. doi: 10.1016/s0306-4522(03)00281-1. [DOI] [PubMed] [Google Scholar]
- Vann SD, Aggleton JP, Maguire EA. What does the retrosplenial cortex do? Nat Rev Neurosci. 2009;10(11):792–802. doi: 10.1038/nrn2733. [DOI] [PubMed] [Google Scholar]
- Vargha-Khadem F, Gadian DG, Watkins KE, Connelly A, VanPaesschen W, Mishkin M. Differential effects of early hippocampal pathology on episodic and semantic memory. Science. 1997;277(5324):376–380. doi: 10.1126/science.277.5324.376. [DOI] [PubMed] [Google Scholar]
- Warburton EC, Brown MW. Neural circuitry for rat recognition memory. Behav Brain Res. 2015;285:131–139. doi: 10.1016/j.bbr.2014.09.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


