Abstract
The percentage of children in the United States with obesity has more than tripled since the 1970s. Despite considerable work in this area, there has been limited success in the long-term treatment and prevention of childhood obesity. In order for intervention approaches to be successful, it will be crucial to study individual variation and the underlying mechanisms that impact human ingestive behavior. Research in my laboratory has focused on identifying behavioral phenotypes for childhood obesity which may be used to develop targeted intervention approaches that are tailored to a child’s individual predisposition. This work has included studying select appetitive traits among weight-discordant siblings and unrelated children to elucidate potential differences in children’s ability to regulate short-term energy intake and susceptibility to eating in response to food cues in the environment. I am greatly honored to be the recipient of the 2017 Alan N. Epstein Research Award from the Society for the Study of Ingestive Behavior (SSIB). I want to acknowledge and express my sincere gratitude to my mentors and colleagues who have supported me and provided guidance throughout my research career and to my research team and students for their hard work on our studies.
Keywords: children, obesity, ingestive behavior, behavioral phenotypes, caloric compensation, eating in the absence of hunger
Introduction
I was profoundly grateful to be the recipient of the 2017 Alan N. Epstein Research Award which was presented to me at the annual meeting of the Society for the Study of Ingestive Behavior (SSIB) in Montreal, Canada. I am greatly honored to have received this prestigious award and am appreciative of being recognized by SSIB in this way. Dr. Alan Epstein served as SSIB’s 4th president and was a distinguished researcher and Professor of Behavioral Neuroscience at the University of Pennsylvania studying the neuroendocrine mechanisms of ingestive behavior, the regulation of food intake, water intake, salt intake, and suckling and the ontogeny of these behaviors (Figure 1) [1–5]. In an article written in memory of Alan, Dr. Elliott Stellar wrote: “[Alan] had a brilliant mind” and “[…] was a recognized world authority […]” in his area of science. I am therefore humbled by this prestigious award knowing what a leading authority Alan was in ingestive behavior research.
Figure 1.

Dr. Alan N. Epstein
With permission from Stellar E. Alan N. Epstein. Appetite, 18, 89–91, © 1992 by Elsevier.
This award also carries a special meaning for me because, although I never had the opportunity to personally meet Alan, I know he was an inspiration, mentor, and great colleague and friend to several of my own mentors who played instrumental roles in my training and research career. As a graduate student in the Department of Nutritional Sciences at The Pennsylvania State University, I had the great honor to train with Dr. Barbara Rolls, one of the leading scientists in human ingestive behavior research. Back when Barbara was an undergraduate student at the University of Pennsylvania, she enrolled in an introductory biology class taught by Alan and later worked closely with him on several research projects in his laboratory. Alan mentored and inspired Barbara to pursue a career in ingestive behavior research and welcomed her “[…] into the inner circle of ingestive behavior at Penn […]” [6].
As a postdoctoral fellow, I was fortunate to have worked with Dr. Albert (Mickey) Stunkard, who was a Professor of Psychiatry and the founder of the Center for Weight and Eating Disorders at Penn, and Dr. Robert Berkowitz who serves as the Senior Medical Director of the Center to this day. Their conversations with Alan on infant feeding styles inspired Bob and Mickey to pursue a longitudinal study of growth and development which I later worked on when I first came to Penn as a postdoctoral fellow in 2004. To this day, Bob has very fond memories of Alan and “[…] knew him to be an enthusiastic, passionate scientist and so encouraging of other colleagues.”
Having trained with these mentors who were inspired by Alan to either pursue a career in ingestive behavior research or initiate research projects which I later got to work on, this award takes on a very special meaning for me.
Behavioral Phenotyping for Obesity
My interest in behavioral phenotyping for obesity was inspired by my former graduate mentor Dr. Barbara Rolls. In 2003, Barbara was invited to give a talk at a workshop on “Phenotyping Obesity for Human Genetic Studies,” which was organized by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). Barbara invited me to help her prepare for her talk on “Eating Behavior as a Phenotype for Obesity in Adults.” This was the first time I learned about behavioral phenotyping for obesity and I quickly developed an interest in the topic as I immersed myself into the literature.
In 2004, I extended my knowledge of behavioral phenotypes for obesity to pediatric populations as I joined the Center for Weight and Eating Disorders at Penn as a postdoctoral fellow to work on a high-risk study for human obesity under the mentorship of Dr. Myles Faith. I worked on a prospective, longitudinal study (‘Infant Growth Study’) which aimed to determine risk factors for excess weight gain in infants born at high or low risk for obesity based on their mothers’ pre-pregnancy weight status [7, 8]. Between-group differences in children’s weight- and adiposity-related outcomes as well as behavioral outcomes related to food ingestion emerged early in life and persisted over time. By age 6 years, 30% of the high-risk children, compared to only 3% of the low-risk children, developed overweight or obesity [9]. At 3 months of age, infants born at high-risk for obesity showed a more vigorous sucking behavior, as indicated by a significantly greater number of sucks (920 ± 559 vs. 620 ± 293, P < 0.01) and greater sucking rate (0.75 ± 0.25 vs. 0.59 ± 0.26 sucks/second, P < 0.01), than infants born at low-risk for obesity, which in turn predicted weight and adiposity outcomes at 1 year [8]. I joined the study when youth were 12 years of age. One of the first projects I worked on was a laboratory-based assessment of youth’s self-selected meal during which they had free access to 15 foods and 7 beverages which ranged in energy density (kcal/g). Subjects could freely choose the types and amounts of foods and beverages they wanted to consume. This study showed that high-risk youth self-selected a meal which was 30% more energy-dense than that of low-risk youth; a difference which remained statistically significant when controlling for subjects’ body mass index (BMI) z-score (Figure 2) [10].
Figure 2.
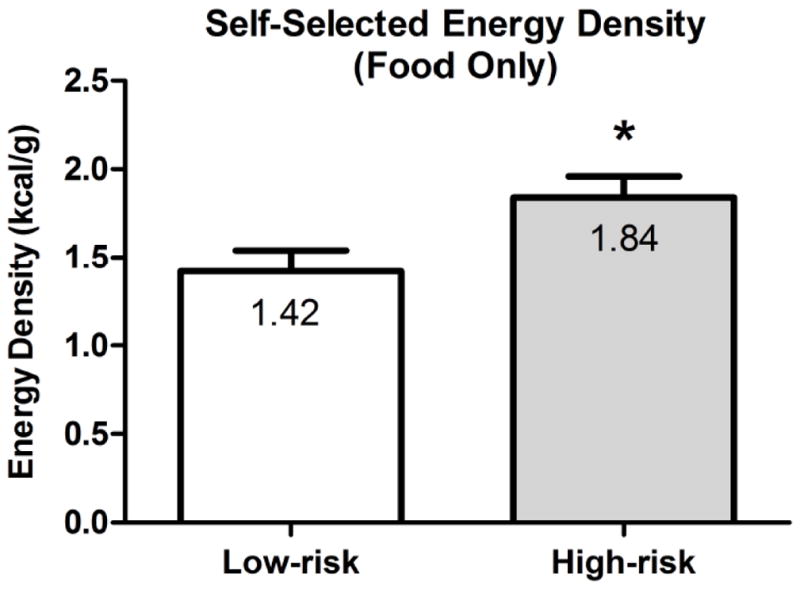
Mean (± SEM) energy density (kcal/g; foods only) at a self-selected, single buffet meal for youth born at low risk (LR; N = 15) or high-risk (HR; N = 15) for obesity
Adapted and reprinted from Kral TV, Stunkard AJ, Berkowitz RI, Stettler N, Stallings VA, Kabay A, Faith MS. Energy density at a buffet-style lunch differs for adolescents born at high or low risk of obesity. Eating Behaviors, 10, 209–214, © 2009 by Elsevier, with permission from Elsevier.
Through my work on the Infant Growth Study, I developed a deeper understanding of the familial influences contributing to the development of childhood obesity and was intrigued by the behavioral differences between the two risk groups which emerged early and persisted over time. Today, it has been well established that parental obesity is one of the strongest risk factors for childhood obesity [11, 12], which points to the combination of shared genes and factors in the early home environment affecting energy balance in children. It has also been proposed that genetic predispositions to obesity may in part exert their effects through appetitive traits [13]. Several appetitive traits have been identified that put children at increased risk for overeating and excess weight gain. Children with a genetic predisposition to these traits are believed to show a behavioral susceptibility to overeating in environments where highly palatable, energy-dense foods are easily accessible. Some of these traits represent food-seeking appetitive traits, such as sensitivity to reward [14], the relative reinforcing value of food [15], or eating in the absence of hunger (EAH) [16], while other traits, such as satiation or satiety, relate more to the self-regulation of energy intake. It is possible that behavioral phenotypes for childhood obesity may represent a strengthening of food-seeking appetitive traits and a simultaneous weakening of self-regulatory systems, such as satiety. Over the course of my postdoctoral training, I became very interested in two of these appetitive traits, namely the study of satiety and EAH.
Studying Appetitive Traits using a Discordant Sibling Design
In the 1960s, Drs. Albert Stunkard and Stanley Schachter, through their seminal work on lean-obese differences, were among the first to propose that persons with obesity showed an impaired sensitivity to internal satiety cues, when compared to persons with normal weight [17, 18]. The satiety value of a food is typically assessed in the laboratory by having subjects consume the food as a compulsory first course (preload) and then assess its effects on energy intake at a subsequent main course (test meal), which is consumed ad libitum [19]. In the context of energy intake regulation, caloric compensation then refers to adjustments in energy intake at the test meal in response to changes in the energy density of a preload. The percentage compensation index (%COMPX), which represents a linear transformation of the difference in intake across the two preload conditions, can be used to assess individual differences in satiety. EAH, on the other hand refers to children’s susceptibility to eating when satiated in response to the presence of palatable snack foods [16, 20]. It is typically assessed by using the ‘free access procedure’ in which children consume a meal to fullness and then, after a short delay, are given access to a variety of snack foods, which they consume over a period of ~15 minutes [21]. EAH is then defined as the number of calories consumed from the snacks. Important conceptual differences exist between these two appetitive traits. While caloric compensation 1) is assessed during a meal, 2) is based on feedback from internal cues, and 3) measures children’s responsiveness to differences in the energy density of a preload, EAH 1) is assessed after a meal when hunger is not present and 2) measures children’s responsiveness to external cues.
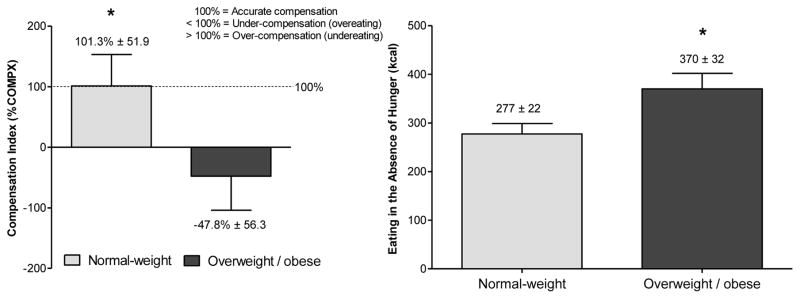
In 2008, I received a Mentored Research Scientist Development Award (K01DK078601) from the NIDDK which enabled me to receive further training in behavioral genetics with the support of an expert team of mentors consisting of Drs. Myles Faith, David Allison, Leann Birch, and Virginia Stallings. Using an unmeasured genotype approach, we compared 47 same-sex sibling pairs (55% full siblings), ages 5 to 12 years, who were discordant in their weight status in caloric compensation ability and EAH to elucidate potential differences in siblings’ ability to regulate short-term energy intake [22]. The satiety protocol was designed to create ideal conditions for optimal caloric compensation to occur. Specifically, we 1) opted for a semi-solid preload (pudding) as previous studies indicated that liquid foods elicit a weaker compensatory response in energy intake than do solid or semi-solid foods [23]; 2) chose the preload size to represent ~30% of children’s no-preload (baseline) meal energy intake [24]; 3) tailored both the preload and test meal size to subjects’ age group to account for different energy requirements among older and younger children; and 4) chose a fairly short, 25-minute time interval between the preload and the test meal [25]. In this study, subjects were served the same dinner, but, 25 minutes before dinner, they either consumed or did not consume in full one of two preloads that varied in energy density (0.57 or 0.97 kcal/g). On the day when no preload was consumed, EAH was assessed after dinner. The results showed that siblings with overweight or obesity under-compensated and therefore overate after the high energy-dense preload, whereas siblings with normal weight showed accurate caloric compensation (Figure 3). Further, siblings with overweight or obesity consumed 34% more calories (93 kcal) in the absence of hunger compared to siblings with normal weight. We also found that within-pair resemblances in both appetitive traits were stronger for full siblings (%COMPX: r = 0.36; EAH: r = 0.37) than half siblings (%COMPX: r = 0.02; EAH: r = 0.16), which points to genetic influences underlying these traits.
Figure 3.
Caloric compensation index (%COMPX) and number of calories consumed in the absence of hunger (mean ± SEM) for weight-discordant siblings (N = 47 sibling pairs)
The American Journal of Clinical Nutrition, 2012, 96, 574–583, American Society for Nutrition
The findings from this study suggest that an impaired ability to regulate short-term energy intake, which includes adhering less to internal cues of hunger and fullness and eating when satiated, may represent a behavioral phenotype for childhood obesity. If so, this behavioral phenotype may help predict children’s response to an intervention. Therefore, we may be able to use it to develop targeted (precision) prevention and treatment approaches that are tailored to a child’s individual predisposition with respect to these appetitive traits.
Research Gaps and Current Work
While our understanding of pediatric ingestive behavior has greatly increased thanks to decades of creative and carefully designed experiments, new opportunities exist to build on and extend this prior research in important ways. First, many prior studies, including our own, assessed child eating phenotypes in isolation, studying one phenotype at a time. It will be important for future research to use a more integrated approach to study multiple eating phenotypes concurrently but at the same time take into consideration familial and home environmental influences that may impact children’s ingestive behavior. This may enable us to identify clusters of eating phenotypes, the development of which may be influenced by factors in children’s early home environment. Second, many prior studies, including our own, assessed behavioral phenotypes only during a single meal. Studying behavioral phenotypes across multiple meals and assessing children’s energy intakes at those meals in the context of their daily energy needs may enable us to determine the extent to which individual differences in energy intake regulation may predict longer-term (e.g., 24-hr) energy intake and weight-related outcomes over time. Lastly, another research gap and important goal for future research focuses on identifying at-risk children who exhibit obesogenic eating phenotypes before they develop obesity. If successful, identifying these phenotypes early has the potential to move primary prevention strategies in a new direction.
My laboratory’s current research focuses on using an integrative approach to concurrently study select eating behaviors under states of hunger and satiety and to assess the impact of short-term appetite and intake regulation on longer-term weight outcomes in children with normal weight (NW) and overweight/obesity (OB) who are at high risk (HR) or low risk (LR) for obesity based on their mothers’ current weight status. It is important to determine whether HR-NW children are similar in their eating behaviors to LR-NW children or if they have already adopted behaviors that resemble more closely those of HR-OB children, which, if so, may put them on the path to excess weight gain. In this current research, caloric compensation is being evaluated in the context of children’s overall appetite control by comparing their satiety quotient and postprandial ratings of appetite after consuming fixed amounts of foods (preloads) and ad libitum (free-feeding) meals. This research also aims to determine if these eating traits predict 1-year changes in children’s BMI z-score, waist circumference, and percent body fat. Additionally, we are assessing, for the first time, if EAH extends to healthier low energy-dense snacks (fruit). Lastly, our research aims to determine the relationship between children’s home food environment (i.e., availability of low and high energy-dense foods in the home), eating phenotypes, and 1-year changes in weight-related outcomes. The knowledge gleaned from this research can be used to design personalized behavioral interventions for children who are at greatest risk for obesity that target the individual components of the behavioral phenotype.
Highlights.
There has been limited success in the long-term treatment of childhood obesity;
My lab has focused on identifying behavioral phenotypes for childhood obesity;
Targeted interventions are needed which are tailored to a child’s predisposition;
Precision prevention and treatment approaches offer new targets for intervention;
I am honored to have received the 2017 Alan N. Epstein Research Award from SSIB.
Acknowledgments
In closing, I would like to acknowledge and thank the many individuals who have supported me and provided guidance throughout my academic and research career. First and foremost, I want to thank my family and partner for their love and encouragement at every step of my education and career. I have also been very fortunate to have had the unwavering support and guidance of many formal and informal mentors, in particular, Drs. Barbara Rolls, Myles Faith, Michael Lowe, Thomas Wadden, David Allison, Virginia Stallings, Leann Birch, and Albert (Mickey) Stunkard. Barbara Rolls, especially, introduced me to and trained me in all aspects of the study of human ingestive behavior. Most importantly though, she also instilled in me scientific curiosity, fostered critical thinking, and spent many hours training me in the art of effectively communicating science. I also want to thank the many talented and dedicated clinical research coordinators and students who have worked in my lab over the past few years and have been instrumental in the overall success of our studies. I further want to thank the Epstein family for the establishment and generous endowment of this award in honor of Dr. Alan Epstein, the individual(s) who nominated me for this award, and the SSIB nomination committee for selecting me as this year’s award recipient. Last but not least, I would like to thank the funding agencies who provided the financial support for these and other studies: NIH NIDDK (R01DK101480, R03DK091492, K01DK078601, K01DK078601-02S2), NICHD (R21HD091330), the Egg Nutrition Center, The Obesity Society, Penn Center for Health Incentives and Behavioral Economics, and the Penn Biobehavioral Research Center.
Abbreviations
- EAH
eating in the absence of hunger
- %COMPX
percentage compensation index
- ED
energy density
- BMI
body mass index
- HR
high risk
- LR
low risk
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Bryant RW, Epstein AN, Fitzsimons JT, Fluharty SJ. Arousal of a specific and persistent sodium appetite in the rat with continuous intracerebroventricular infusion of angiotensin II. J Physiol. 1980;301:365–382. doi: 10.1113/jphysiol.1980.sp013211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Epstein AN. Reciprocal changes in feeding behavior produced by intrahypothalamic chemical injections. Am J Physiol. 1960;199:969–974. doi: 10.1152/ajplegacy.1960.199.6.969. [DOI] [PubMed] [Google Scholar]
- 3.Epstein AN. Mineralocorticoids and cerebral angiotensin may act together to produce sodium appetite. Peptides. 1982;3(3):493–494. doi: 10.1016/0196-9781(82)90113-9. [DOI] [PubMed] [Google Scholar]
- 4.Epstein AN, Fitzsimons JT, Rolls BJ. Drinking induced by injection of angiotensin into the rain of the rat. J Physiol. 1970;210(2):457–474. doi: 10.1113/jphysiol.1970.sp009220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Teitelbaum P, Epstein AN. The lateral hypothalamic syndrome: recovery of feeding and drinking after lateral hypothalamic lesions. Psychol Rev. 1962;69:74–90. doi: 10.1037/h0039285. [DOI] [PubMed] [Google Scholar]
- 6.Rolls BJ. Creativity needs some serendipity: Reflections on a career in ingestive behavior. Physiol Behav. 2016;162:186–195. doi: 10.1016/j.physbeh.2016.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Stunkard AJ, Berkowitz RI, Schoeller D, Maislin G, Stallings VA. Predictors of body size in the first 2 y of life: a high-risk study of human obesity. Int J Obes Relat Metab Disord. 2004;28(4):503–513. doi: 10.1038/sj.ijo.0802517. [DOI] [PubMed] [Google Scholar]
- 8.Stunkard AJ, Berkowitz RI, Stallings VA, Schoeller DA. Energy intake, not energy output, is a determinant of body size in infants. Am J Clin Nutr. 1999;69(3):524–530. doi: 10.1093/ajcn/69.3.524. [DOI] [PubMed] [Google Scholar]
- 9.Berkowitz RI, Stallings VA, Maislin G, Stunkard AJ. Growth of children at high risk of obesity during the first 6 y of life: implications for prevention. Am J Clin Nutr. 2005;81(1):140–146. doi: 10.1093/ajcn/81.1.140. [DOI] [PubMed] [Google Scholar]
- 10.Kral TV, Stunkard AJ, Berkowitz RI, Stettler N, Stallings VA, Kabay A, Faith MS. Energy density at a buffet-style lunch differs for adolescents born at high and low risk of obesity. Eat Behav. 2009;10(4):209–214. doi: 10.1016/j.eatbeh.2009.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Whitaker RC, Wright JA, Pepe MS, Seidel KD, Dietz WH. Predicting obesity in young adulthood from childhood and parental obesity. N Engl J Med. 1997;337(13):869–873. doi: 10.1056/NEJM199709253371301. [DOI] [PubMed] [Google Scholar]
- 12.Strauss RS, Knight J. Influence of the home environment on the development of obesity in children. Pediatrics. 1999;103(6):e85. doi: 10.1542/peds.103.6.e85. [DOI] [PubMed] [Google Scholar]
- 13.Konttinen H, Llewellyn C, Wardle J, Silventoinen K, Joensuu A, Mannisto S, Salomaa V, Jousilahti P, Kaprio J, Perola M, et al. Appetitive traits as behavioural pathways in genetic susceptibility to obesity: a population-based cross-sectional study. Sci Rep. 2015;5:14726. doi: 10.1038/srep14726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rollins BY, Loken E, Savage JS, Birch LL. Measurement of food reinforcement in preschool children. Associations with food intake, BMI, and reward sensitivity. Appetite. 2014;72:21–27. doi: 10.1016/j.appet.2013.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Temple JL, Legierski CM, Giacomelli AM, Salvy SJ, Epstein LH. Overweight children find food more reinforcing and consume more energy than do nonoverweight children. Am J Clin Nutr. 2008;87(5):1121–1127. doi: 10.1093/ajcn/87.5.1121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fisher JO, Cai G, Jaramillo SJ, Cole SA, Comuzzie AG, Butte NF. Heritability of Hyperphagic Eating Behavior and Appetite-Related Hormones among Hispanic Children. Obesity (Silver Spring) 2007;15(6):1484–1495. doi: 10.1038/oby.2007.177. [DOI] [PubMed] [Google Scholar]
- 17.Stunkard AJ, Fox S. The relationship of gastric motility and hunger. A summary of the evidence. Psychosom Med. 1971;33(2):123–134. doi: 10.1097/00006842-197103000-00004. [DOI] [PubMed] [Google Scholar]
- 18.Schachter S, Goldman R, Gordon A. Effects of fear, food deprivation, and obesity on eating. J Pers Soc Psychol. 1968;10(2):91–97. doi: 10.1037/h0026284. [DOI] [PubMed] [Google Scholar]
- 19.Blundell J, de Graaf C, Hulshof T, Jebb S, Livingstone B, Lluch A, Mela D, Salah S, Schuring E, van der Knaap H, et al. Appetite control: methodological aspects of the evaluation of foods. Obes Rev. 2010;11(3):251–270. doi: 10.1111/j.1467-789X.2010.00714.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cutting TM, Fisher JO, Grimm-Thomas K, Birch LL. Like mother, like daughter: familial patterns of overweight are mediated by mothers’ dietary disinhibition. Am J Clin Nutr. 1999;69(4):608–613. doi: 10.1093/ajcn/69.4.608. [DOI] [PubMed] [Google Scholar]
- 21.Birch LL, Fisher JO, Davison KK. Learning to overeat: maternal use of restrictive feeding practices promotes girls’ eating in the absence of hunger. Am J Clin Nutr. 2003;78(2):215–220. doi: 10.1093/ajcn/78.2.215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kral TV, Allison DB, Birch LL, Stallings VA, Moore RH, Faith MS. Caloric compensation and eating in the absence of hunger in 5- to 12-y-old weight-discordant siblings. Am J Clin Nutr. 2012;96(3):574–583. doi: 10.3945/ajcn.112.037952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Almiron-Roig E, Palla L, Guest K, Ricchiuti C, Vint N, Jebb SA, Drewnowski A. Factors that determine energy compensation: a systematic review of preload studies. Nutr Rev. 2013;71(7):458–473. doi: 10.1111/nure.12048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Roe LS, Thorwart ML, Pelkman CL, Rolls BJ. A meta-analysis of factors predicting energy compensation in preloading studies. FASEB Journal. 1999;13:A871. [Google Scholar]
- 25.Rolls BJ, Kim S, McNelis AL, Fischman MW, Foltin RW, Moran TH. Time course of effects of preloads high in fat or carbohydrate on food intake and hunger ratings in humans. Am J Physiol. 1991;260(4 Pt 2):R756–763. doi: 10.1152/ajpregu.1991.260.4.R756. [DOI] [PubMed] [Google Scholar]