Abstract
It has been reported that microRNA-142 (miR-142) is a tumor suppressor gene. The present study primarily investigated whether the overexpression of miR-142 was able to inhibit the proliferation, apoptosis and expression of apoptosis-associated proteins in osteosarcoma (OS) cells. Different concentrations of miR-142 were transfected into the OS MG-63 cell line using Lipofectamine 2000. The cell lines were divided into three groups: Normal group (non-transfected group), miR-142 transfected group, and negative group, which were transfected with random miR-142 fragment. The proliferation of cells was detected by MTT assay. The expression of miR-142 was detected by reverse transcription-quantitative polymerase chain reaction (RT-qPCR). DAPI staining was performed to investigate the influence of miR-142 on the morphology of MG-63c ells. The apoptotic cell percentages were determined by flow cytometry with Annexin V-fluorescein isothiocyanate/propidium iodide double staining. Expression of tumor suppressors, phosphatase and tensin homolog (PTEN) and Retinoblastoma-associated protein (Rb), and apoptosis-associated proteins were evaluated by western blotting. RT-qPCR indicated a higher expression of miR-142 in the transfected group (miR-142 was transfected into the MG-63 cell line) compared with that in the normal (non-transfected group) and negative control groups. The proliferation of miR-142 transfected cells was significantly lower compared with that in the normal and negative groups. Furthermore, an increased apoptosis rate accompanied by a statistically significant upregulation of PTEN, Rb phosphorylation, cleaved caspase-3 and cytochrome c protein levels were detected in the transfected group, indicating an internal apoptosis pathway was involved in this process. Furthermore, no significant changes were identified between the normal and negative groups (P>0.05). The present study demonstrated that miR-142 overexpression by liposomal transfection resulted in an inhibitory effect on MG-63 cell proliferation. The underlying mechanisms may relate to the upregulation of tumor suppressor and activation of caspase signaling pathway, which may provide a novel horizon in short nucleotide drugs on the management of OS.
Keywords: osteosarcoma, microRNA-142, proliferation, apoptosis
Introduction
Osteosarcoma is a bone cancer, which means it can easily spread to other organs or tissues in the body. Osteosarcoma is the most common malignant bone tumor among children, adolescents and young adults and occurs most frequently in children and young adults between the ages of 10 and 20 years and often during adolescence. With a higher prevalence in boys compared with girls (1). The incidence rate and 95% confidence intervals for osteosarcoma is 4.0 (3.5–4.6) from 0–14 years of age, and 5.0 (4.6–5.6) for 0–19 years of age per year per million persons (1). The surgical procedures for OS primarily involve extensive resection or amputation, and rendering patients disabled, thus having a major impact on the quality of life of patients. Therefore, early detection, diagnosis and treatment are important (2). MicroRNAs (miRNAs), which are a class of 21~25 nucleotides of non-coding single-stranded small RNAs, have been demonstrated to serve as oncogenes or tumor suppressors in a variety of tumor types, particularly OS (3).
Previous studies have demonstrated that miR-142 is downregulated as a tumor suppressor gene in a variety of cancer tissues and cell lines. Overexpression of miR-142-3p may cause cell cycle arrest and exert its effect by targeting cell division cycle 25C (CDC25C) (4). In non-small cell lung cancer, miRNA-142 exerts tumor suppressor effects and induces apoptosis by targeting high mobility group box 1 (5,6). In addition, miR-142 targets a number of different genes and inhibits cancer cell proliferation, including targeting ASH1 like histone lysine methyltransferase (ASH1L) and mixed-lineage leukemia 1 (MLL1), and inhibiting cell proliferation in thyroid follicular carcinoma (7), and in colorectal cancer (8), breast cancer (9,10). Similar findings have been reported in previous studies on OS. miR-142 has been demonstrated to inhibit the proliferation of OS cells by targeting high mobility group AT-hook 1 (11), and subsequent studies have revealed that miR-142 expression is reduced in OS tissues and cell lines (12). miR-142 inhibits cell proliferation and cell invasion by upregulating E-cadherin, inhibiting matrix metalloproteinase-2 and −9, and affecting Ras-related C3 botulinum toxin substrate 1 (Rac1) expression, and arrests the cell cycle in the S phase (13). These results demonstrate that overexpression of miR-142 in OS cell lines inhibits cell proliferation.
Whether miR-142 may be useful in OS gene therapy remains to be elucidated. The present study aimed to investigate the effect of miR-142 on the apoptosis of OS cell lines and explore the specific pathways underlying miR-142-induced OS cell apoptosis. The findings in this study may lay the foundation for subsequent studies into the therapeutic significance of miR-142 in OS.
Materials and methods
Materials
Human OS cell lines hFOB1.19, MG-63, U-2OS and Saos-2 cells were purchased from the Cell Resource Center of Shanghai Institutes for Biological Sciences (Shanghai, China). MTT, DMSO, apoptosis detection kits and DAPI were purchased from Sigma-Aldrich (Merck KGaA, Darmstadt, Germany), the miRNA-142 analogue was purchased from Shanghai GenePharma Co., Ltd., Shanghai, China with the following sequence: Fluorescein (FAM)-5′-CAUAAAGUAGAAAGCACUACU-3′. Negative control (NC) miRNA was synthesized and the sequence was FAM-5′-UUCUCCGAACGUGUCACGU-3′. diethyl pyrocarbonate was dissolved in water and diluted into 10 µM stock for cell transfection, which was added to the complete culture medium when required. RPMI-1640 medium was obtained from Gibco (Thermo Fisher Scientific, Inc., Waltham, MA, USA), fetal bovine serum (FBS) was purchased from (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA). The Cytoplasmic and Mitochondrial Protein Extraction kit was obtained from Sangon Biotech Co., Ltd. (Shanghai, China). The remaining reagents were of analytical grade or molecular reagent grade. All primary and secondary antibodies are listed in Table I.
Table I.
The antibodies used for western blot analysis.
| Antibody name | Catalog number | Dilutions | Supplier |
|---|---|---|---|
| β-actin | ab5694 | 1:1,000 | Abcam |
| Caspase-3 | ab13847 | 1:500 | Abcam |
| Cleaved Caspase-3 | ab136812 | 1:500 | Abcam |
| Anti-cytochrome c | ab13575 | 1:5,000 | Abcam |
| p-Rb | ab184796 | 1:10,000 | Abcam |
| P-TEN | ab32199 | 1:10,000 | Abcam |
| Rb | ab181616 | 1:10,000 | Abcam |
| Goat anti-rabbit IgG | ab6721 | 1:1,000 | Abcam |
Cell culture
Immortalized human fetal osteoblastic cell line hFOB1.19 or OS cells (MG-63, U-2OS and Saos-2) were routinely passaged. All cells were separately cultured in RPMI-1640 medium with 10% FBS at 37°C in a humidified atmosphere with 5% CO2.
Reverse transcription-quantitative polymerase chain reaction (RT-qPCR) assay for the expression of miR-142 and phosphatase and tensin homolog (PTEN) gene
Cells in the logarithmic growth phase were collected to detect the expression levels of miR-142 in hFOB1.19 cells or OS cells. Cells in the logarithmic growth phase were digested with 0.25% trypsin and washed three times with PBS (pH 7.2). The final concentration of the cells was adjusted to 8×104 cells/ml in RPMI-1640 medium containing 10% FBS and inoculated into 6-well plates (2×105 cells/well).
Transfection
Cells were transfected with 20 nM miR-142 20 nM, miR-NC and control (adding the completed medium without mircoRNA) using Lipofectamine™ 2000 (Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA). Following transfection, the cells were incubated for 48 h and then collected to determine the expression of miR-142. Additionally, for quantitate the expression of PTEN gene, cells in the logarithmic growth phase were obtained, re-suspended and then seeded in 24-well plates at a density of 5×104 cells/well. Cells were randomly divided into 5 groups: Control group, treated with complete RPMI-1640 medium, miR-NC group and 3 miRNA-142 treatment groups, with final concentrations of 20, 40 and 80 nM.
RT-qPCR
Then, these cells were obtained to measure the expression of PTEN gene following miR-142 transfection. Total RNA was extracted using TRIzol reagent (Life Technologies; Thermo Fisher Scientific, Inc.), and then was used as the template for reverse transcription. cDNA was extracted from mRNA by TaKaRa PrimeScript™ II 1st strand cDNA Synthesis kit (Clontech Laboratories, Inc., Mountainview, CA, USA). The TaqMan real-time PCR kit (Applied Biosystems; Thermo Fisher Scientific, Inc.) and the Eco real-time PCR system (Illumina, Inc., San Diego, CA, USA) were used. GAPDH was used as an internal reference. The following primers were used: Human miR-142-3p sequence: FAM-5′-UUUCUACUUUAUG-3′. (PN4427975, assay ID: 000434) and U18 (PN4427975, assay ID: 001204; both from Life Technologies; Thermo Fisher Scientific, Inc.); PTEN forward, 5′-AAAGCTGGAAAGGGACGAAC-3′ and reverse, 5′-CAGGTAACGGCTGAGGGAAC-3′; GAPDH forward, 5′-GAAGGTGAAGGTCGGAGTC-3′ and reverse, 5′-GAAGATGGTGATGGGATTTC-3′. After the amplification reaction, The thermocycling conditions were as follows: 35 cycles, 30 sec at 95°C, 30 sec at 56°C and extension for 10 min at 72°C. The 2−ΔΔCq method was used for data analysis (14).
MTT analysis for the inhibitory effect of miR-142 on proliferation
Following transfection with different concentrations of miR-142 (0, 20, 40, 80 nM) and control, MG-63, U-2OS and Saos-2 cell lines were trypsinized, re-suspended and seeded in 96-well plates at a density of 1×104 cells/well. Cells were incubated at 37°C for 48 h, and then 10 µl 5 mg/ml MTT reagent was added to each well, the cells were incubated for another 4 h at 37°C. Then, the supernatant was discarded and 200 µl of DMSO was added to each well. Finally, samples were measured on a multi-well spectrophotometer, to measure the absorbance at 570 nm (OD value) using a microplate reader. Cell inhibition rate (%)=(1-ODtest/ODcontrol) ×100.
Fluorescence microscopy assay for morphological observation of apoptotic cells
When MG-63 cells reached the logarithmic growth phase, cells were treated with miR-NC and different concentrations of miR-142 (20, 40 or 80 nM) for 48 h at 37°C, and then fixed with cold 100% methanol and were incubated for 5 min at room temperature, washed three times with PBS. Stained with DAPI for 5 min at room temperature, washed once with PBS. One side of the cells was inverted onto a slide glass pre-incubated with a fluorescent anti-quencher (Guangzhou RiboBio Co., Ltd., Guangzhou, China) and observed under an inverted fluorescence microscope.
Flow cytometric analysis of the cell cycle and apoptosis rate
The OS cell lines treated with control anddifferent concentrations of miR-142 for 48 h at 37°C were collected and treated according to the Annexin V/propidium iodide (PI) apoptosis detection kit (BD Biosciences, Franklin Lakes, NJ, USA). Cells were washed twice with PBS and then re-suspended with 1X binding buffer to adjust the cell density to 1×108 cells/ml. Following the addition of Annexin V and PI, the mixture was incubated at room temperature for 15 min, and then transferred to a flow cytometer tube. To avoid blockage of the flow cytometer channel by cell masses, cells were passed through a 200-nm screen mesh filter prior to analysis using the flow cytometer. The blank control and single-stained cells were used to set gate and color compensation in flow cytometry. A total of 1×104 cells were analyzed each time. The experiment was repeated for three times, and presented as the mean ± SD.
Western blot assay for the detection of apoptosis- or cell cycle-associated protein expression
After washing twice by cold PBS, MG-63 cells treated with control, miR-NC and different concentrations of miR-142 (20, 40 or 80 nM) for 48 h were collected. The cytosol and mitochondrial protein was obtained using the Cytoplasmic and Mitochondrial Protein Extraction kit. Protein concentration was determined by BCA assay. Each sample (40 µg/lane) was then electrophoresed on 10% SDS-PAGE and transferred to a polyvinylidene fluoride membrane. The membrane was blocked with 5% skim milk for 1 h at room temperature, then incubated with apoptosis-associated and cycle-specific antibodies at 4°C overnight. Subsequently, the membrane was incubated in milk with HRP-conjugated secondary antibodies at room temperature for 1 h and washed three times with Tris buffered saline with Tween-20. All antibodies used are listed in Table I. Membranes were developed in a dark chamber following the addition of enhanced chemiluminescence reagent (Beijing Leagene Biotechnology Co., Ltd.; cat. no., PW0111). Densitometric analysis of blotting bands was performed using ImageJ software (version 1.41; National Institutes of Health, Bethesda, MD, USA) and normalized to β-actin levels.
Statistical analysis
All statistical analysis was performed using IBM SPSS software version 20 (IBM Corp., Armonk, NY, USA). Data are expressed as the mean ± standard deviation. Statistical analysis was performed using one-way analysis of variance followed by the Dunnett's test for multiple group comparisons. P<0.05 was considered to indicate a statistically significant difference.
Results
miR-142 suppresses the proliferation of OS cells
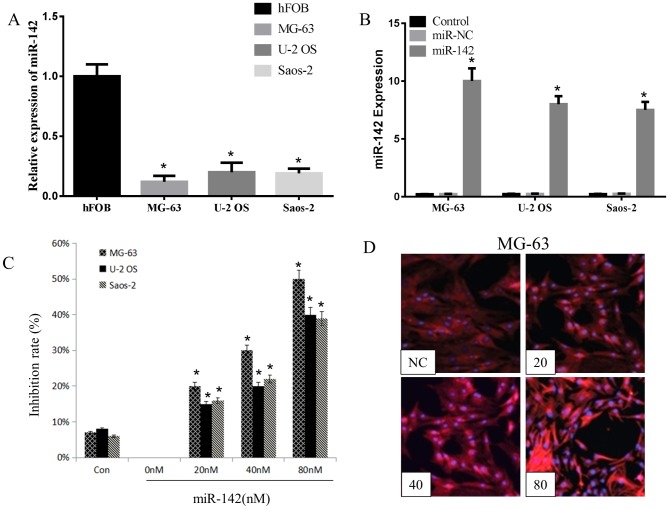
miR-142 has been demonstrated to be downregulated in a variety of cancer tissues and cell lines, including OS cell lines (14). The present study detected the expression levels of miR-142 in hFOB1.19 cells or OS cells (MG-63, U-2OS and Saos-2 cells) using RT-qPCR, and observed that miR-142 expression was significantly reduced in the three OS cell lines, particularly in the MG-63 cell line, compared with that in hFOB1.19 cells (Fig. 1A). This suggests that the overexpression of miR-142-3p serves a tumor suppressor effect.
Figure 1.
miR-142 suppressed the proliferation of OS cells. (A) The expression levels of miR-142 in hFOB1.19 cells or OS cells (MG-63, U-2OS and Saos-2 cells) using RT-qRCR. *P<0.05, compared with hFOB group. (B) The expression levels of miR-142 in three OS cells after miR-142 or random miR-142 fragment was transfected. *P<0.05, compared with miR-NC group. (C) MTT detection of the proliferation inhibitory effect of miR-142 analogue on three kinds of OS cell lines (n=3) post 48 h transfection. *P<0.05, compared with control group. (D) The red fluorescence signal intensity of MG-63 cells in different concentrations of miR-142 groups or the NC group post 24 h transfection; Original magnification, 200×. miR, microRNA; NC, negative control.
miR-142 or random miR-142 fragment was transfected into MG-63, U-2OS and Saos-2 cells using Lipofectamine 2000 to investigate the inhibitory effect of miR-142-3 on OS cell proliferation. As indicated in Fig. 1B, a significantly higher expression of miR-142 was detected in transfected groups compared with the miR-NC groups, particularly in MG-63 cells. Wang and Youle (15) reported that miR-142 serves a vital role in suppressing the proliferation of colon cancer cells. In the MTT assay of the present study, miR-142 significantly inhibited the growth of all three OS cell lines post 48 h transfection. At 80 nM, the inhibitory effect of miR-142 on MG-63 cells was 50±6% (Fig. 1C). The inhibitory rate was observed to be concentration-dependent in all three cell lines. In addition, the NC group exhibited a certain degree of inhibition, indicating that the transfection caused toxicity to the cells. Of the three OS cell lines; MG-63 was most sensitive to miR-142 (Fig. 1C). In the present study, compared with all other OS cell lines, the MG-63 cell line exhibited the lowest and highest expression of miR-142 prior to, and following transfection with miR-142, respectively. Thus, MG-63 cells were selected for apoptosis and western analysis. The fluorescence intensity of Cy3 in MG-63 cells was detected by fluorescence microscopy following miR-142 transfection. As presented in Fig. 1D, with increasing miR-142 concentrations, the distribution and intensity of red fluorescence also increased. The fluorescence intensity of the control group was similar to that of the 20 nM group.
miR-142 promotes the apoptosis of MG-63 cells
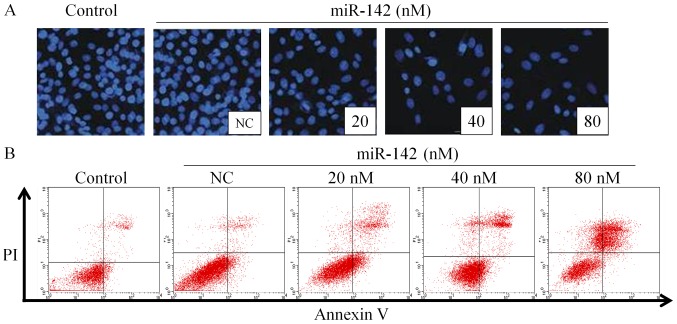
Post 48 h transfection with different concentrations of miR-142, the nuclei of MG-63 cells was stained with DAPI, as shown in Fig. 2A. As the miR-142 concentration increased, the percentage of apoptotic cells increased gradually (Fig. 2B), which is similar to a previous study whereby miR-142 was observed to regulate apoptosis in cancer cells (16). This indicates that miR-142 exhibited strong apoptosis-inducing effects on MG-63 cells.
Figure 2.
Effects of miR-142 on apoptosis. (A) The results of nuclear DAPI staining. Magnification, ×200. (B) Annexin V/PI double staining in MG-63 cells following transfection with different concentrations of miR-142 for 48 h. miR, microRNA; NC, negative control; PI, propidium iodide.
MG-63 cells were harvested post 48 h transfection and the percentage of apoptotic cells was detected by Annexin V/PI double staining. The results in Table II revealed that the population of normal cells significantly decreased in miR-142-treated groups compared with the NC group (P<0.05). Furthermore, the population of early apoptotic cells was significantly increased in miR-142-transfected groups (P<0.05), particularly in the 40 nM group. The percentage of apoptotic cells overall, including early apoptosis and late apoptosis, was significantly increased post miR-142 transfection (80 nM) when compared with the NC group (3.28±0.72 vs. 52.91±8.79%; P<0.05). In addition, miR-142 treatment induced the formation of necrotic cells, particularly in the 80 nM group, which exhibited a significant increase when compared with the NC group (1.28±0.01 vs. 10.02±5.25%; P<0.05). These results confirmed that miR-142 induced MG-63 cell apoptosis in a dose-dependent manner. Compared with the blank control group, the proportion of early apoptotic cells in the NC group was 3.24±0.51%, which difference was significant (P<0.05), indicating that Lipofectamine 2000 was mildly toxic to cells.
Table II.
The effect of miR-142 on the percentage of apoptotic osteosarcoma cells at different stages (n=3).
| Percentage of cells, (%) | |||||
|---|---|---|---|---|---|
| miR-142 | |||||
| Groups | Blank control | Negative control | 20 NM | 40 nM | 80 nM |
| Normal cells | 97.01±9.32 | 94.02±8.11 | 56.92±4.11a | 44.36±8.21a | 36.45±7.91a |
| Early apoptosis | 1.24±0.01 | 3.24±0.51b | 23.12±1.21a | 36.02±1.11a | 7.89±2.31a |
| Late apoptosis | 0.04±0.01 | 0.04±0.21 | 14.02±3.64a | 13.81±7.41a | 45.02±6.48a |
| Necrotic cells | 0.98±0.01 | 1.28±0.01 | 6.21±4.11 | 9.52±6.33 | 10.02±5.25a |
P<0.05, vs. the negative control group
P<0.05, vs. the blank control group. miR, microRNA.
miR-142 upregulates the expression of apoptotic-associated proteins
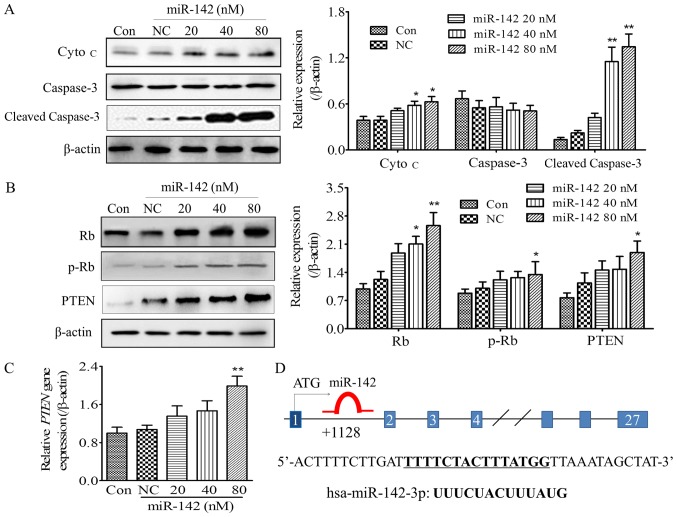
It has reported that miR-142 aids in mediating apoptosis signaling in OS (13,17–19). In order to further explore the specific mechanism underlying miR-142-induced OS cell apoptosis, caspase-3 and cytochrome c (Cyto c) expression was detected. At 48 h post transfection with different concentrations of miR-142-3p (20, 40 and 80 nM), the MG-63 cells were lysed and apoptosis-associated protein expression was detected. The results indicated that caspase-3, a key molecule that represents activation of apoptosis, was significantly activated with increasing miR-142 concentrations (40 and 80 nM), as indicated by a significant increase in cleaved caspase-3 expression (Fig. 3A). It has been proposed that activated caspases are involved in a feedback mechanism, which disrupts mitochondrial function, thereby amplifying the release of Cyto c (15). To further differentiate whether apoptosis was an endogenous or exogenous pathway, and to determine the effect of miR-142 on caspase activation and Cyto c release, Cyto c expression was detected by western blot analysis. As shown in Fig. 3A, the expression of cytosol Cyto c was significantly increased following miR-142 transfection (40 and 80 nM) when compared with the NC group (P<0.05). It demonstrated that Cyto c, which represents the endogenous pathway, was released from the mitochondria into the cytoplasm under the action of miR-142, indicating the primary pathway underlying miR-142-induced OS cell apoptosis may be the endogenous mitochondrial pathway.
Figure 3.
Effect of miR-142 on apoptosis-associated signaling molecules and tumor suppressors, Rb and PTEN. (A) The protein expression of cytosol Cyto c and caspase-3 cleaved. (B) The expression of PTEN and Rb and the phosphorylation status of p-Rb in MG-63 cells post 48 h transfection with different concentrations of miR-142. Left panel: Representative images; Right panel: Quantification of the gray scale of the bands in different groups using ImageJ software. (C) The gene expression of PTEN measured by reverse transcription-quantitative polymerase chain reaction. (D) The potential binding pattern of mir-142 to the rb gene. *P<0.05, **P<0.01, compared with Con and NC groups. miR, microRNA; NC, negative control; p-, phosphorylated-, PTEN, phosphatase and tensin homolog; Rb, Retinoblastoma-associated protein; Cyto c, cytochrome c; Con, blank control.
miR-142 increases PTEN expression and Retinoblastoma-associated protein (Rb) phosphorylation
Zheng et al indicated that miR-142, acts as a tumor suppressor in the OS cell lines, arresting cell cycle progression in the S phase (13). Phosphorylation of Rb was originally considered to drive cell cycle progression, and influence cell proliferation and apoptosis (16). The present study investigated the influence of miR-142 upregulation of the tumor suppressor PTEN and Rb phosphorylation on miR-142-regulated OS cell apoptosis. The results demonstrated that miR-142-3p upregulated PTEN gene and protein expression in a dose-dependent manner, with a significant difference observed at a concentration of 80 nM (P<0.05 when compared with the blank control or the NC group; Fig. 3B and C). Furthermore, phosphorylation of Rb increased significantly with 80 nM miR-142 compared with the control group, while the Rb expression also increased significantly, suggesting that the upregulation of Rb serves an important role in miR-142-induced apoptosis.
Discussion
miRNAs have been identified to serve as oncogenes or tumor supressors in a variety of tumors (17–19), also in OS (20). miR-142 expression is significantly dysregulated in numerous diseases, particularly in cancer (21). The target genes of miR-142 in tumor research have been confirmed, including: CDC25C (4), Rac1 (13), high mobility group protein A1 (HMGA1) (11), high mobility group box-1 protein (HMGB1) (5), Wiskott-Aldrich syndrome like, Integrin αV, and additional cytoskeletal elements (9), suppressor of cytokine signaling 6 (SOCS6) (22), ASH1L and MLL1 (7), ATP binding cassette subfamily G member 2 (Junior blood group), and leucine rich repeat containing G protein-coupled receptor 5 (8), CD133 (23), interleukin 6 signal transducer (24) and glucocorticoid receptor α (25). A previous study reported that miR-142 expression is reduced in a variety of cancer cells, such as breast cancer stem cells (10) and pancreatic cancer cells (26). Overexpression of miR-142 exhibits a tumor suppressor effect (11). Therefore, it is considered to be an anti-oncomirs, despite other studies reporting that miR-142 may serve an oncomir role in certain tumors like human T-cell acute lymphoblastic leukemia (T-ALL) (25). In the current study, the expression levels of miR-142 were detected in hFOB1.19 cells or OS cells (MG-63, U-2OS and Saos-2 cells), whereby a lower expression of miR-142 was observed in OS cells, particularly in the MG-63 cell line. The subsequent experiments in the present study all suggested that miR-142 serves a tumor suppressor role in OS.
miR-142, as a marker of prognosis in a variety of cancer types, including lung cancer (27), colon cancer (28) and leukemia (29), may affect cell proliferation, and induce apoptosis. Here, miR-142-overexpressed cells were obtained by liposomal transfection and miR-142 was observed to inhibit the growth of three types of OS cell lines post 48 h transfection in a dose-dependent manner. Previous studies have reported that the transfection of miR-142-3p for 48 h induces apoptosis in lung cancer cell lines NCI-H23 (5) and glioma-related murine M2 macrophage (30). In the present study, to investigate whether miR-142-3p was able to induce MG-63 cell apoptosis, fluorescence microscopy assays and flow cytometric analysis were performed. The results indicated that miR-142 enhanced the population of early apoptotic cells, promoted MG-63 cell apoptosis in a dose-dependent manner and induced the formation of necrotic cells.
The associations between miR-142-mediated proliferation and apoptosis signaling have been previously reported in OS. As previous study reported, miR-142 can mediates cancer cell proliferation and apoptosis in OS (11,13,31,32). Ding et al (31) reported that the expression level of miR-142 was significantly lower in OS tissues and cells due to hypermethylation, suggesting that miR-142 served an important role in the inhibition of the proliferation, and invasiveness of OS cell lines induced by demethylation agents. Xu et al (11) has identified that overexpression of miR-142-3p in OS cells suppressed cell proliferation, migration and invasion by directly targeting HMGA1. Zheng et al (13) reported that the expression of miR-142 was significantly reduced, but Rac1 was increased in OS tissues and cell lines, as miR-142 directly targeted to the 3′-untranslated region (UTR) of the Rac1 gene, which then regulates Rac1 expression at the transcriptional and translational levels. Liu et al (32) confirmed that miR-142-3p inhibits OS cell growth and promotes apoptosis by directly binding to the HMGB1 3′-UTR, negatively regulating its expression, and thus decreasing HMGB1 expression. However, to the best of our knowledge, there are no reports on the interaction between miR-142-3p and the upregulation of tumor suppressors (Rb and PTEN) or activation of the caspase signaling pathway. Therefore, the roles of miR-142 in proliferation and apoptosis of OS cells were further elucidated by investigating the expression of apoptosis-associated proteins and PTEN, to decipher the molecular pathway underlying the effects of miR-142-3p on OS.
The present study determined the expression of PTEN and caspase-3, and the phosphorylation levels of Rb following miR-142 transfection. PTEN, a tumor suppressor gene with active phosphatase, promotes cell apoptosis through the PI3K/Akt signaling pathway (33). It was reported that miRNAs enhance tumor formation and metastasis by directly targeting the 3′-UTR of the PTEN gene, and promote the proliferation of human cancer cells through direct suppression of PTEN expression (34). In the present study, miR-142 overexpression resulted in upregulation of PTEN gene and protein expression, thus suppressing cell proliferation. In the caspase family, caspase-3 is the most critical downstream protease in the caspase cascade. Upregulating the expression of caspase-3 promotes the apoptosis of tumor cells. In the current study, the detection of the apoptosis of tumor cells following the upregulation of caspase-3 expression was not only verified by annexin V/PI double staining, but also demonstrated by the expression of apoptosis-associated proteins at a molecular level. In the present study, miR-142-3p significantly upregulated the protein expression of cleaved caspase-3, suggesting it activated the caspase signaling pathway and regulated caspase-3-induced apoptosis. Schwartzbauer and Robbins (35) reported that the activity of caspase-3 and cell apoptosis rate were reduced following PTEN inhibition by PTEN cDNA in rat primary cardiomyocytes, which indicates that PTEN is able to increase caspase-3 expression and further promotes the occurrence of apoptosis. Similarly, in the present study, miR-142 transfection significantly upregulated PTEN gene and protein expression, leading to the activation of caspase-3. These data suggest that PTEN may influence the level of apoptosis of OS cells through adjusting the expression of caspase-3.
Rb is a tumor suppressor in OS cell lines (36). Notably, phosphorylation of Rb, which is an important gene in cell cycle regulation, is considered to drive cell cycle progression, and influence cell proliferation and apoptosis (16). Therefore, the influence of miR-142 on Rb activation was detected in the present study, and the western blotting results revealed that miR-142 significantly promoted the phosphorylation of Rb in MG-63 cells, which may be associated with miR-142-induced OS cell apoptosis. In patients with OS, the mutation of the Rb gene inhibits Rb from activating its downstream signaling molecules, thus causing a dysregulation of the cell cycle, and subsequently, uncontrolled proliferation. The Saos-2 cell line is resistant to methotrexate, a therapeutic drug for OS, due to lack of an active Rb gene (37), compared with Saos-2 cells, the activity of RB1 in MG-63 cells is unaffected. This may be the reason why the proliferation-inhibitory activity against MG-63 was superior to that of SaOS-2 following miR-142 transfection. In the present study, miR-142 was suggested to induce MG-63 cell apoptosis by regulating Rb activity.
Apoptosis pathways may be divided into endogenous and exogenous pathways. The former is also known as the mitochondrial pathway, primarily because it involves the release of mitochondrial-associated proteins, including Cyto c and a change in the mitochondrial membrane potential. The present study reported that miR-142 significantly activated caspase-3. The activated caspase may feedback, disrupting mitochondrial function, and amplifying further Cyto c release (38). To further differentiate whether apoptosis was an endogenous or exogenous pathway, the cytosol and mitochondrial proteins were obtained using Cytoplasmic and Mitochondrial Protein Extraction kits, respectively. As the results indicated, significantly increased protein expression of cytosol Cyto c in the miR-142-treated groups (40 and 80 nM) was observed, suggesting that miR-142 induces apoptosis through the mitochondrial pathway, due to miR-142 enhancing the release of Cyto c from mitochondria to the cytosol. Furthermore, Rb and PTEN expression was significantly activated, as well as Cyto c, indicating that multiple signaling pathways were involved with crosstalk occurring between themselves. miR-142 has been revealed to be aberrantly expressed in AML, and may be used as a novel diagnostic marker (29). Furthermore, expression of miR-142 could be considered a prognostic indicator in ESCC (39). Therefore, further in vivo studies on the targets and effects of miRNAs should be performed to provide a basis for early diagnosis or prognosis of OS using the abnormal expression of miRNAs as biomarkers. In our future studies, the influence of PTEN upregulation, Rb phosphorylation and caspase signaling pathway activation on miR-142-regulated cell apoptosis requires further investigation, with comparisons using multiple OS cell lines, including Saos-2 and U-2OS cells.
In summary, it was confirmed that the overexpression miR-142 significantly inhibited the proliferation and induced the apoptosis of MG-63 cells, thus suppressing OS. These observations may be associated with the upregulation of tumor suppressors, Rb and PTEN, and activation of the caspase signaling pathway. These results have laid a foundation for the application of miR-142 in the prevention and treatment of OS.
Acknowledgements
Not applicable.
Glossary
Abbreviations
- Cyto c
cytochrome c
- PTEN
phosphatase and tensin homolog
- HMGB1
high mobility group box-1 protein
- HMGA1
high mobility group protein A1
- MLL1
myeloid/lymphoid or mixed-lineage leukemia 1
- MMP-2
matrix metalloproteinase-2
- Rac1
ras-related C3 botulinum toxin substrate 1
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Authors' contributions
YG performed the cell culture, qPCR and transfection experiments. QZ was responsible for fluorescent staining and flow cytometry, ZY perform the western blot analysis, SL collected data and statistics. JL designed the study and wrote the manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Ottaviani G, Jaffe N. The epidemiology of osteosarcoma. Cancer Treat Res. 2009;152:3–13. doi: 10.1007/978-1-4419-0284-9_1. [DOI] [PubMed] [Google Scholar]
- 2.Isakoff MS, Bielack SS, Meltzer P, Gorlick R. Osteosarcoma: Current treatment and a collaborative pathway to success. J Clin Oncol. 2015;33:3029–3035. doi: 10.1200/JCO.2014.59.4895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Berindan-Neagoe I, Monroig PC, Pasculli B, Calin GA. MicroRNAome genome: A treasure for cancer diagnosis and therapy. CA Cancer J Clin. 2014;64:311–336. doi: 10.3322/caac.21244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cao XC, Yu Y, Hou LK, Sun XH, Ge J, Zhang B, Wang X. miR-142-3p inhibits cancer cell proliferation by targeting CDC25C. Cell Prolif. 2016;49:58–68. doi: 10.1111/cpr.12235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Xiao P, Liu WL. MiR-142-3p functions as a potential tumor suppressor directly targeting HMGB1 in non-small-cell lung carcinoma. Int J Clin Exp Pathol. 2015;8:10800–10807. [PMC free article] [PubMed] [Google Scholar]
- 6.Su YH, Zhou Z, Yang KP, Wang XG, Zhu Y, Fa XE. MIR-142-5p and miR-9 may be involved in squamous lung cancer by regulating cell cycle related genes. Eur Rev Med Pharmacol Sci. 2013;17:3213–3220. [PubMed] [Google Scholar]
- 7.Colamaio M, Puca F, Ragozzino E, Gemei M, Decaussin-Petrucci M, Aiello C, Bastos AU, Federico A, Chiappetta G, Del Vecchio L, et al. miR-142-3p down-regulation contributes to thyroid follicular tumorigenesis by targeting ASH1L and MLL1. J Clin Endocrinol Metab. 2015;100:E59–E69. doi: 10.1210/jc.2014-2280. [DOI] [PubMed] [Google Scholar]
- 8.Shen WW, Zeng Z, Zhu WX, Fu GH. MiR-142-3p functions as a tumor suppressor by targeting CD133, ABCG2, and Lgr5 in colon cancer cells. J Mol Med (Berl) 2013;91:989–1000. doi: 10.1007/s00109-013-1037-x. [DOI] [PubMed] [Google Scholar]
- 9.Schwickert A, Weghake E, Brüggemann K, Engbers A, Brinkmann BF, Kemper B, Seggewiß J, Stock C, Ebnet K, Kiesel L, et al. microRNA miR-142-3p inhibits breast cancer cell invasiveness by synchronous targeting of WASL, integrin Alpha V, and additional cytoskeletal elements. PLoS One. 2015;10:e0143993. doi: 10.1371/journal.pone.0143993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Isobe T, Hisamori S, Hogan DJ, Zabala M, Hendrickson DG, Dalerba P, Cai S, Scheeren F, Kuo AH, Sikandar SS, et al. miR-142 regulates the tumorigenicity of human breast cancer stem cells through the canonical WNT signaling pathway. Elife. 2014;3 doi: 10.7554/eLife.01977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Xu G, Wang J, Jia Y, Shen F, Han W, Kang Y. MiR-142-3p functions as a potential tumor suppressor in human osteosarcoma by targeting HMGA1. Cell Physiol Biochem. 2014;33:1329–1339. doi: 10.1159/000358700. [DOI] [PubMed] [Google Scholar]
- 12.Namløs HM, Meza-Zepeda LA, Barøy T, Østensen IH, Kresse SH, Kuijjer ML, Serra M, Bürger H, Cleton-Jansen AM, Myklebost O. Modulation of the osteosarcoma expression phenotype by microRNAs. PLoS One. 2012;7:e48086. doi: 10.1371/journal.pone.0048086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zheng Z, Ding M, Ni J, Song D, Huang J, Wang J. MiR-142 acts as a tumor suppressor in osteosarcoma cell lines by targeting Rac1. Oncol Rep. 2015;33:1291–1299. doi: 10.3892/or.2014.3687. [DOI] [PubMed] [Google Scholar]
- 14.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 15.Wang C, Youle RJ. The role of mitochondria in apoptosis*. Annu Rev Genet. 2009;43:95–118. doi: 10.1146/annurev-genet-102108-134850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Young AP, Longmore GD. Differential regulation of apoptotic genes by Rb in human versus mouse cells. Oncogene. 2004;23:2587–2599. doi: 10.1038/sj.onc.1207330. [DOI] [PubMed] [Google Scholar]
- 17.Kent OA, Mendell JT. A small piece in the cancer puzzle: microRNAs as tumor suppressors and oncogenes. Oncogene. 2006;25:6188–6196. doi: 10.1038/sj.onc.1209913. [DOI] [PubMed] [Google Scholar]
- 18.Zhang B, Pan X, Cobb GP, Anderson TA. microRNAs as oncogenes and tumor suppressors. Dev Biol. 2007;302:1–12. doi: 10.1016/j.ydbio.2006.08.028. [DOI] [PubMed] [Google Scholar]
- 19.Lee YS, Dutta A. MicroRNAs: Small but potent oncogenes or tumor suppressors. Curr Opin Investig Drugs. 2006;7:560–564. [PubMed] [Google Scholar]
- 20.Sampson VB, Yoo S, Kumar A, Vetter NS, Kolb EA. Micrornas and potential targets in osteosarcoma: Review. Front Pediatr. 2015;3:69. doi: 10.3389/fped.2015.00069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Shrestha A, Mukhametshina RT, Taghizadeh S, Vásquez-Pacheco E, Cabrera-Fuentes H, Rizvanov A, Mari B, Carraro G, Bellusci S. MicroRNA-142 is a multifaceted regulator in organogenesis, homeostasis, and disease. Dev Dyn. 2017;246:285–290. doi: 10.1002/dvdy.24477. [DOI] [PubMed] [Google Scholar]
- 22.Qi X, Li J, Zhou C, Lv C, Tian M. Mir-142-3p suppresses socs6 expression and promotes cell proliferation in nasopharyngeal carcinoma. Cell Physiol Biochem. 2015;36:1743–1752. doi: 10.1159/000430147. [DOI] [PubMed] [Google Scholar]
- 23.Chai S, Tong M, Ng KY, Kwan PS, Chan YP, Fung TM, Lee TK, Wong N, Xie D, Yuan YF, et al. Regulatory role of miR-142-3p on the functional hepatic cancer stem cell marker CD133. Oncotarget. 2014;5:5725–5735. doi: 10.18632/oncotarget.2167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sonda N, Simonato F, Peranzoni E, Calì B, Bortoluzzi S, Bisognin A, Wang E, Marincola FM, Naldini L, Gentner B, et al. miR-142-3p prevents macrophage differentiation during cancer-induced myelopoiesis. Immunity. 2013;38:1236–1249. doi: 10.1016/j.immuni.2013.06.004. [DOI] [PubMed] [Google Scholar]
- 25.Lv M, Zhang X, Jia H, Li D, Zhang B, Zhang H, Hong M, Jiang T, Jiang Q, Lu J, et al. An oncogenic role of miR-142-3p in human T-cell acute lymphoblastic leukemia (T-ALL) by targeting glucocorticoid receptor-α and cAMP/PKA pathways. Leukemia. 2012;26:769–777. doi: 10.1038/leu.2011.273. [DOI] [PubMed] [Google Scholar]
- 26.Lu Y, Ji N, Wei W, Sun W, Gong X, Wang X. MiR-142 modulates human pancreatic cancer proliferation and invasion by targeting hypoxia-inducible factor 1 (HIF-1α) in the tumor microenvironments. Biol Open. 2017;6:252–259. doi: 10.1242/bio.021774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kaduthanam S, Gade S, Meister M, Brase JC, Johannes M, Dienemann H, Warth A, Schnabel PA, Herth FJ, Sültmann H, et al. Serum miR-142-3p is associated with early relapse in operable lung adenocarcinoma patients. Lung Cancer. 2013;80:223–227. doi: 10.1016/j.lungcan.2013.01.013. [DOI] [PubMed] [Google Scholar]
- 28.Ghanbari R, Mosakhani N, Asadi J, Nouraee N, Mowla SJ, Yazdani Y, Mohamadkhani A, Poustchi H, Knuutila S, Malekzadeh R. Downregulation of plasma mir-142-3p and mir-26a-5p in patients with colorectal carcinoma. Iran J Cancer Prev. 2015;8:e2329. doi: 10.17795/ijcp2329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wang F, Wang XS, Yang GH, Zhai PF, Xiao Z, Xia LY, Chen LR, Wang Y, Wang XZ, Bi LX, et al. miR-29a and miR-142-3p downregulation and diagnostic implication in human acute myeloid leukemia. Mol Biol Rep. 2012;39:2713–2722. doi: 10.1007/s11033-011-1026-5. [DOI] [PubMed] [Google Scholar]
- 30.Xu S, Wei J, Wang F, Kong LY, Ling XY, Nduom E, Gabrusiewicz K, Doucette T, Yang Y, Yaghi NK, et al. Effect of miR-142-3p on the M2 macrophage and therapeutic efficacy against murine glioblastoma. J Natl Cancer Inst. 2014;106 doi: 10.1093/jnci/dju162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ding M, Hu J, Ni J, Zheng Z, Song D, Wang J. Demethylation of microRNA-142 induced by demethylation agents plays a suppressive role in osteosarcoma cells. Oncol Lett. 2015;9:2261–2267. doi: 10.3892/ol.2015.3036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Liu K, Huang J, Ni J, Song D, Ding M, Wang J, Huang X, Li W. MALAT1 promotes osteosarcoma development by regulation of HMGB1 via miR-142-3p and miR-129-5p. Cell Cycle. 2017;16:578–587. doi: 10.1080/15384101.2017.1288324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lu XX, Cao LY, Chen X, Xiao J, Zou Y, Chen Q. PTEN inhibits cell proliferation, promotes cell apoptosis, and induces cell cycle arrest via downregulating the pi3K/akt/htertpathway in lung adenocarcinoma a549 Cells. Biomed Res Int. 2016;2016:2476842. doi: 10.1155/2016/2476842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bermúdez BM, Goulielmaki E, Papakonstanti EA. Focus on PTEN regulation. Front Oncol. 2015;5:166. doi: 10.3389/fonc.2015.00166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Schwartzbauer G, Robbins J. The tumor suppressor gene PTEN can regulate cardiac hypertrophy and survival. J Biol Chem. 2001;276:35786–35793. doi: 10.1074/jbc.M102479200. [DOI] [PubMed] [Google Scholar]
- 36.Hellwinkel OJ, Müller J, Pollmann A, Kabisch H. Osteosarcoma cell lines display variable individual reactions on wildtype p53 and Rb tumour-suppressor transgenes. J Gene Med. 2005;7:407–419. doi: 10.1002/jgm.684. [DOI] [PubMed] [Google Scholar]
- 37.Iida K, Nobori T, Matsumine A, Isaka A, Seto M, Shiraishi T, Uchida A. Effect of retinoblastoma tumor suppressor gene expression on chemosensitivity of human osteosarcoma cell lines. Oncol Rep. 2003;10:1961–1965. [PubMed] [Google Scholar]
- 38.Zhu Y, Li M, Wang X, Jin H, Liu S, Xu J, Chen Q. Caspase cleavage of cytochrome c1 disrupts mitochondrial function and enhances cytochrome c release. Cell Res. 2012;22:127–141. doi: 10.1038/cr.2011.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lin RJ, Xiao DW, Liao LD, Chen T, Xie ZF, Huang WZ, Wang WS, Jiang TF, Wu BL, Li EM, Xu LY. MiR-142-3p as a potential prognostic biomarker for esophageal squamous cell carcinoma. J Surg Oncol. 2012;105:175–182. doi: 10.1002/jso.22066. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.