Abstract
Background
To compare the abundance of vitreous proteins between the patients with proliferative diabetic retinopathy (PDR) and idiopathic macular hole (IMH).
Methods
In this study, we performed mass spectrometry-based label-free quantitative proteomics analysis of vitreous samples from type 2 diabetic patients with PDR (n = 9) and IMH subjects (n = 9) and identified the abundance of 610 proteins.
Results
Out of 610 proteins, 64 proteins (Group A) were unique to PDR patients, while 212 proteins (Group B) could be identified in IMH vitreous only. Among the other 334 proteins that could be detected in both PDR and IMH eyes, 62 proteins differed significantly (p < 0.05, fold change > 2), which included 52 proteins (Group C) and 10 proteins (Group D) over- and under-expressed in PDR vitreous compared with the control. All proteins in these four groups were counted as significant proteins in our study.
Conclusions
We identified and quantified 610 proteins in total, which included 338 significant proteins in our study. Protein distribution analysis demonstrated a clear separation of protein expression in PDR and IMH. The protein function analysis illustrated that immunity and transport related proteins might be associated with PDR.
Keywords: Proliferative diabetic retinopathy, Quantitative proteomics, Bioinformatics
Background
Diabetic retinopathy (DR), a pathological condition in which damage occurs to the retina due to diabetes mellitus, is the most common microvascular complication of diabetes [1]. This disease can be further classified into two types: nonproliferative diabetic retinopathy (NPDR) and proliferative diabetic retinopathy (PDR). Classification of NPDR is based on clinical findings manifested by visible features, including microaneurysms, retinal hemorrhages, intraretinal microvascular abnormalities (IRMA), and venous caliber changes, while PDR is characterized by the hallmark feature of pathologic preretinal neovascularization [2]. With the increasing global prevalence of diabetes, DR is the major cause of vision loss and blindness among working-age adults in developed countries [3]. Current treatments for diabetic eye disease, which include laser photocoagulation, intravitreous injections of anti-VEGF and steroid agents and vitreoretinal surgery, mainly focus on advanced diseases such as PDR [4]. However, because of side effects and individual differences, there is an urgent need for new therapeutic options for PDR [5–7]. Therefore, a better understanding of the pathological mechanism is required for the development of new treatment options.
Research on PDR is limited because no current model can completely replicate the full pathophysiology of neuronal and vascular changes that occur at each stage of DR [8]. The physiologic and pathologic conditions of the retina are reflected in the protein composition of the vitreous due to their close anatomical and biological relationship, which can be sampled as part of routine surgical procedures [9, 10]. In this regard, vitreous fluid obtained through vitrectomy is currently used to indirectly examine the condition of the retina. Mass spectrometry (MS) has been widely applied to study biomolecules and one rapidly developing field is the global analysis of proteins, proteomics [11]. MS-based proteomics is of great value to study different ocular diseases such as cataracts [12], age-related macular degeneration (AMD) [13, 14] and DR [15, 16]. Despite the great efforts that have been devoted to DR vitreous protein identification [17, 18], we are still far from a comprehensive understanding of the complex molecular pathomechanisms. Quantitative proteomics is an analytical chemistry technique for determining the amount of proteins, which can contribute to better insight into the underlying pathogenesis [19].
In this study, the abundance of vitreous proteins was compared between the PDR and IMH subjects through a label-free quantitative method. We also analyzed protein distribution and then conducted bioinformatic analyses, which included protein function, related disease, gene ontology (GO), protein-protein interaction (PPI) and the Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway.
Methods
Subject enrollment and sample collection
This prospective observational study was conducted at the First Affiliated Hospital of Soochow University according to the principles of the Declaration of Helsinki and the approved Human Discarded Specimen Research Protocols from the respective institutional review boards. The study was approved by the institutional ethics committee of the First Affiliated Hospital of Soochow University and signed informed consent was obtained from every participant before being included into the study. A total of 18 subjects who received vitrectomy were enrolled, 9 were type 2 diabetic patients with PDR, and 9 served as controls with IMH. The exclusion criteria were as follows: (1) acute or chronic infection, (2) vitreous hemorrhage or vitreous opacities, (3) a history of ocular surgery or laser photocoagulation, (4) other ocular diseases, (5) systemic diseases other than diabetes, and (6) use of antimetabolites or immunosuppressants.
The vitreous samples were collected by a syringe prior to the infusion procedure of the 3-port pars plana vitrectomy and then transferred into a centrifugal tube and stored at − 80 °C.
Sample preparation
Vitreous samples were dissolved in reducing solution (6 M urea, 2 M thiourea; Sigma, St. Louis, MO) and then centrifuged at 12000 g for 45 min at 4 °C to collect the supernatant. Afterwards, the protein concentrations were measured by bicinchoninic acid (BCA) protein assay kit (Pierce, Thermo Scientific, Waltham, MA USA). For LC-MS analysis, 100 μg of protein from each sample was taken and then reduced with 10 mM dithiothreitol (Sigma, St. Louis, MO) at 37 °C for 2.5 h, alkylated with 50 mM iodoacetamide (Sigma, St. Louis, MO) at room temperature for 40 min followed by trypsin digestion with sequencing grade modified trypsin (Promega, Madison, WI, USA) using a 1:50 enzyme/protein ratio at 37 °C overnight. Tryptic peptides were purified with C18 microspin columns (Nest Group, Southborough, MA, USA).
MS based label-free quantification
The purified peptide samples were loaded onto Orbitrap Elite hybrid mass spectrometer (Thermo Scientific) coupled to EASY-nLC II system (Thermo Scientific) using the Xcalibur version 2.7.0 SP1 (Thermo Scientific). The MS analysis was conducted in data-dependent acquisition where one high resolution (120000) FTMS full scan (m/z 300–1700) was followed by top20 CID-MS2 scans in ion trap (energy 35). Only the precursor ions with over 500 ion counts were allowed for MSn. Charge state rejection was enabled as well as dynamic exclusion which was fixed at 30 s for the selected ions.
The MS1 intensities of peptides for label-free quantification were acquired by the Progenesis LC-MS software (v 4.1, Nonlinear Dynamics Limited, Tyne, UK). For protein identification, the MS2-scan data obtained from Progenesis LC - MS were searched against the human component of the UniProtKB database using the SEQUEST search engine in Proteome Discoverer software (version 1.4, Thermo Scientific). The results were filtered to a maximum false discovery rate (FDR) of 0.05. Afterwards, spectral counts for each protein were extracted from the search results of the SEQUEST database and used to quantify protein abundance differences [20].
Data processing
Student’s t test (p < 0.05) and fold change (fold change > 2) were calculated on protein abundance changes between PDR and IMH vitreous.
Analysis of protein distribution contained boxplot, correlation analysis and principal component analysis (PCA) which were produced using R version 3.4.1 and the hierarchical clustering analysis (HCA) of the identified proteins being constructed by Cluster 3.0.
DAVID bioinformatics Resources (Available online: https://david.ncifcrf.gov/) was employed to study significant proteins about protein functions, related disease and GO, which included biological processes, cellular components and molecular function.
PPI and KEGG pathway were analyzed through the String database (Available online: http://www.string-db.org) and produced by Cytoscape.
Results
Proteome
To analyze the differences in vitreous proteomics between PDR and IMH patients, we enrolled 9 subjects in each group. The difference of the protein concentration was not statistically significant in PDR vitreous and control eyes (5.23 ± 0.86 vs. 5.12 ± 0.56 μg/μl, p = 0.75). The proteins were then digested into peptides and analyzed by LC-MS/MS-based label-free quantitation.
We identified and quantified the abundance of 610 vitreous proteins. Among these, 64 proteins (Group A) could be identified only in PDR patients while 212 proteins (Group B) were unique to IMH vitreous. Among the other 334 proteins that could be detected in both PDR and IMH eyes, 62 proteins differed significantly (p < 0.05, fold change > 2), which included 52 (Group C) and 10 (Group D) that were up- and down- regulated, respectively, in PDR vitreous compared with the control. All proteins in these four groups were counted as significant proteins in our study.
Protein distribution analysis
Boxplot
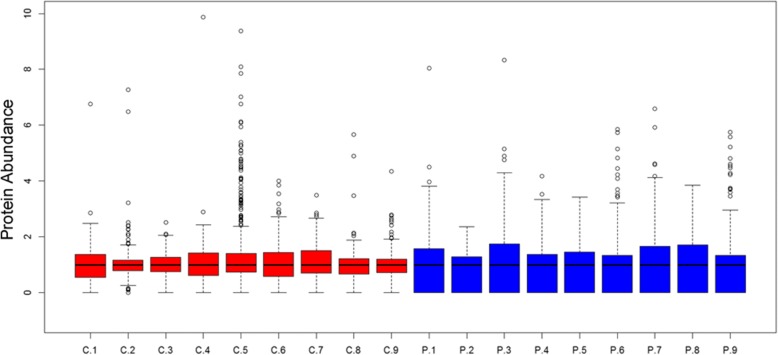
We made a boxplot pertaining to all of the proteins in the 18 samples. As shown in Fig. 1, the proteins in PDR samples had a wider distribution than those in IMH vitreous as the interquartile ranges and the distances between the upper limit and the lower limit were longer. In addition, the higher number of outliers in the control samples reflected more proteins with abnormal abundance than those in the PDR group. The number 5 sample in the control group (C5) had the most outliers that declared unreliability to some extent.
Fig. 1.
A boxplot produced by R version 3.4.1 about all proteins in each sample. C1 is short for the number 1 sample in the control group while P1 represents the number 1 sample in the proliferative diabetic retinopathy group. The results demonstrate a wider protein distribution in PDR samples than those in IMH vitreous. Furthers, more proteins in the control group have abnormal abundance and C5 is the most unreliable.
Correlation analysis
Correlation analysis was conducted to confirm the distribution of our identified proteins. The value of correlation coefficient ranges from − 1 to 1. A positive value shows positive correlation, while a negative value is opposite. When the absolute value of correlation coefficient approaches 1, a stronger correlative extent exists, and when it approaches 0, a weaker correlative extent results. It is reflected in Fig. 2 that IMH samples indeed shared weaker correlations than those among PDR samples. In addition, C5 had the least correlation compared to the other IMH samples, which was in support of the conclusion from the boxplot.
Fig. 2.
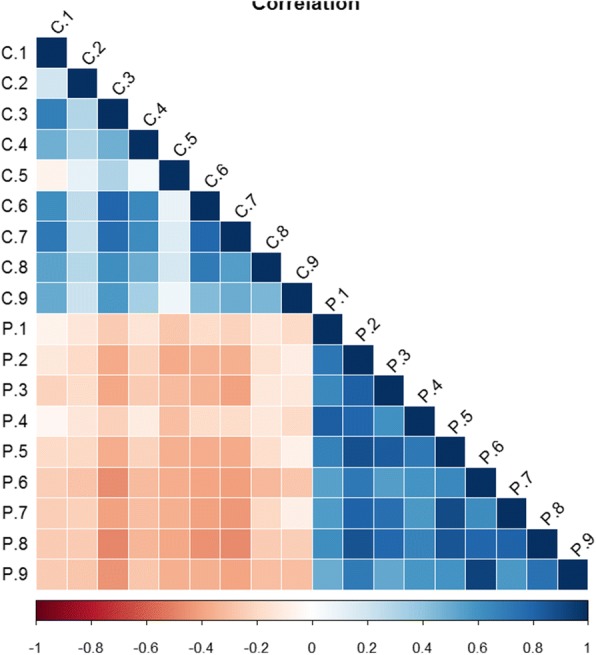
Correlation analysis of all proteins in every sample conducted by R version 3.4.1. The IMH samples have weaker correlation than PDR ones and C5 have the least correlation with the other IMH samples
Principal component analysis
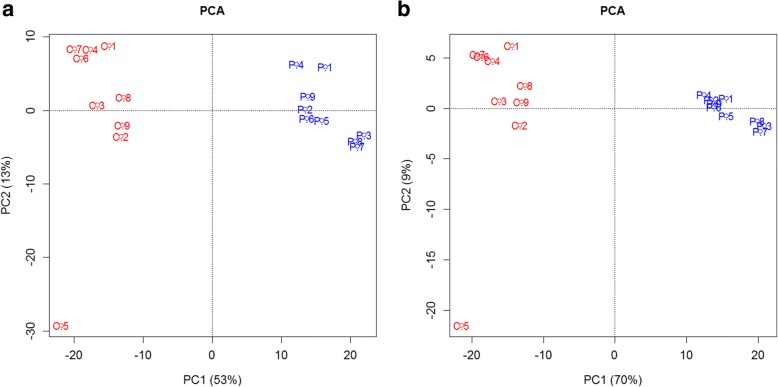
We conducted principal component analysis on all detected proteins and significant proteins. The separation of the PDR group and the control group demonstrated their difference in protein expression. C5, which contained the most proteins with extreme abundance, was certainly an isolated one in both analyses, as shown in Fig. 3. Moreover, when taking only significant proteins into account, the dots in the same group became closer to each other which stated less difference among the samples. The principal component 1, which indicated that the differences between the two groups can explain the overall analysis results, varied from 53 to 70%, thus illustrating a reduction in confounding factors.
Fig. 3.
Principal component analysis (PCA) carried out by R version 3.4.1. a PCA of all proteins. b PCA of significant proteins. A clear difference in protein expression of the PDR group and the control group is displayed and C5 is proved to be unreliable again. The difference among the samples in one group becomes less when significant proteins been taken into account
Hierarchical clustering analysis
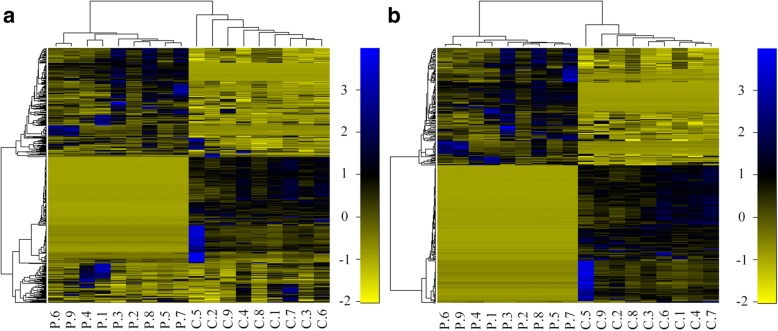
To assess the qualitative and quantitative differences between the PDR and the IMH vitreous proteome, we performed hierarchical clustering of the 18 vitreous samples based on their protein content. The color gradient showed the fold change between the two groups. The quantitative proteomes (n = 610) of the PDR and the IMH samples differed and resulted in clear separation of the two disease states (Fig. 4a). When hierarchical clustering was performed on significant proteins only (n = 62), the separation became more apparent (Fig. 4b).
Fig. 4.
Hierarchical clustering analysis (HCA) by R version 3.4.1. a HCA of all proteins. b HCA of significant proteins. The color gradient illustrates the fold change of protein abundance between the two groups. A clear separation of the protein expression in the two disease states is shown and the separation becomes more apparent when significant proteins been taken into account
Bioinformatics results
We combined Group A and Group C into Group 1 (n = 116) and Group B and Group D into Group 2 (n = 222) and carried out bioinformatic analyses such as protein function, related disease, and GO on these two groups. When comparing the two groups, we defined the fold change, which had a significant change of over 2.
Protein function
We compared protein functions between the two groups. In Group 1, 111 proteins out of 116 were classified into 50 categories, while in Group 2, 214 proteins were categorized for 64 protein functions.
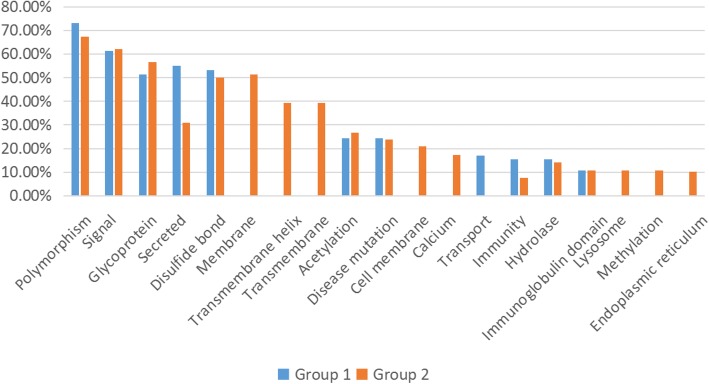
Some elementary physiological functions, which included polymorphism, signal, glycoprotein and disulfide bond, were of high abundance in both groups, yet they were related to no less than 50% proteins in each group. However, although secreted was associated with over 50% proteins in Group 1, the related proteins in Group 2 was only 30.84%.
Moreover, though immunity was related to 15.32% proteins in Group 1, only 7.48% proteins in Group 2 were relevant.
Among the functions that were related to over 10% of proteins (Fig. 5), transport was identified only in Group 1, while more functions were unique to Group 2 such as membrane, transmembrane helix, transmembrane, cell membrane, calcium, transport, lysosome, methylation and endoplasmic reticulum.
Fig. 5.
Protein functions detected by DAVID bioinformatics Resources relates to more than 10% proteins in two groups. Some elementary physiological functions are of high level in both groups. Furthers, immunity is associated significantly with more proteins in Group 1 than Group 2. Meanwhile, transport is unique to Group 1
Related disease
Related diseases of the significant proteins (Group 1 and Group 2) were analyzed. In Group 1, 87 proteins out of 116 were classified, and in Group 2, 164 out of 222 were identified.
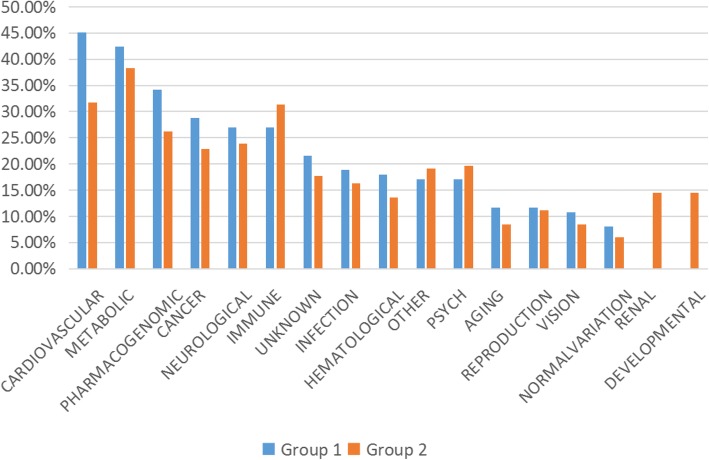
Disease classification was conducted on both groups (Fig. 6). The top 6 disease categories were cardiovascular, metabolic, pharmacogenomic, cancer, neurological and immune. Apart from immune, the other 5 classifications were associated with more proteins in Group 1 than Group 2, yet the fold changes were lower than 2.
Fig. 6.
Related disease classification analyzed by DAVID bioinformatics Resources. It revealed that the top 6 disease categories were cardiovascular, metabolic, pharmacogenomic, cancer, neurological and immune
Vision-related proteins were the key points of this study. As shown in Table 1, there was no overlap on the proteins associated with vision from the two groups. In Group 1, the relevant proteins included: apolipoprotein F (APOF), carbonic anhydrase 1 (CA1), catalase (CAT), collagen type I alpha 1 chain (COL1A1), complement C2 (C2), complement component 4 binding protein alpha (C4BPA), complement factor D (CFD), complement factor H related 3 (CFHR3), complement factor H (CFH), fibrinogen alpha chain (FGA), paraoxonase 1 (PON1) and vitronectin (VTN). These proteins should be carefully analyzed.
Table 1.
Vision-related proteins in two groups
| Group 1 | Group 2 | ||
|---|---|---|---|
| ID | Name | ID | Name |
| Q13790 | apolipoprotein F (APOF) | P35625 | TIMP metallopeptidase inhibitor 3 (TIMP3) |
| P00915 | carbonic anhydrase 1 (CA1) | Q96JP9 | cadherin related family member 1 (CDHR1) |
| P04040 | Catalase (CAT) | P00488 | coagulation factor XIII A chain (F13A1) |
| P02452 | collagen type I alpha 1 chain (COL1A1) | P02458 | collagen type II alpha 1 chain (COL2A1) |
| P06681 | complement C2 (C2) | P05813 | crystallin beta A1 (CRYBA1) |
| P04003 | complement component 4 binding protein alpha (C4BPA) | P07099 | epoxide hydrolase 1 (EPHX1) |
| P00746 | complement factor D (CFD) | P14210 | hepatocyte growth factor (HGF) |
| A0A087WYK9 | complement factor H related 3 (CFHR3) | P11215 | integrin subunit alpha M (ITGAM) |
| P08603 | complement factor H (CFH) | D3DSM0 | integrin subunit beta 2 (ITGB2) |
| P02671 | fibrinogen alpha chain (FGA) | P05362 | intercellular adhesion molecule 1 (ICAM1) |
| P27169 | paraoxonase 1 (PON1) | P30492 | major histocompatibility complex, class I, B (HLA-B) |
| P04004 | Vitronectin (VTN) | Q5Y7A7 | major histocompatibility complex, class II, DR beta 1 (HLA-DRB1) |
| P35579 | myosin heavy chain 9 (MYH9) | ||
| K7EKW2 | olfactomedin 2 (OLFM2) | ||
| Q6UX71 | plexin domain containing 2 (PLXDC2) | ||
| O15537 | retinoschisin 1 (RS1) | ||
| G8JLJ2 | superoxide dismutase 2, mitochondrial (SOD2) | ||
| C9JXZ5 | vesicle associated membrane protein 8 (VAMP8) | ||
The two categories that were unique to Group 2 were renal and developmental.
GO
GO is a major bioinformatics initiative to unify the representation of gene and gene product attributes across all species [21], which include three categories: biological processes, cellular components and molecular function. Significant proteins were analyzed to get a functional overview. Among all of the significant proteins, 115 in Group 1 and 214 in Group 2 could be recognized by DAVID Bioinformatics Resources through an identifier named UNIPROT ACCESSION.
Biological processes
In Group 1, 102 proteins were identified for biological processes, and a total of 79 processes were identified while in Group 2, the number of related proteins was 204 which were associated with 99 biological processes. We made a comparison of biological processes that involved over 5% of the proteins in each group (Fig. 7a).
Fig. 7.
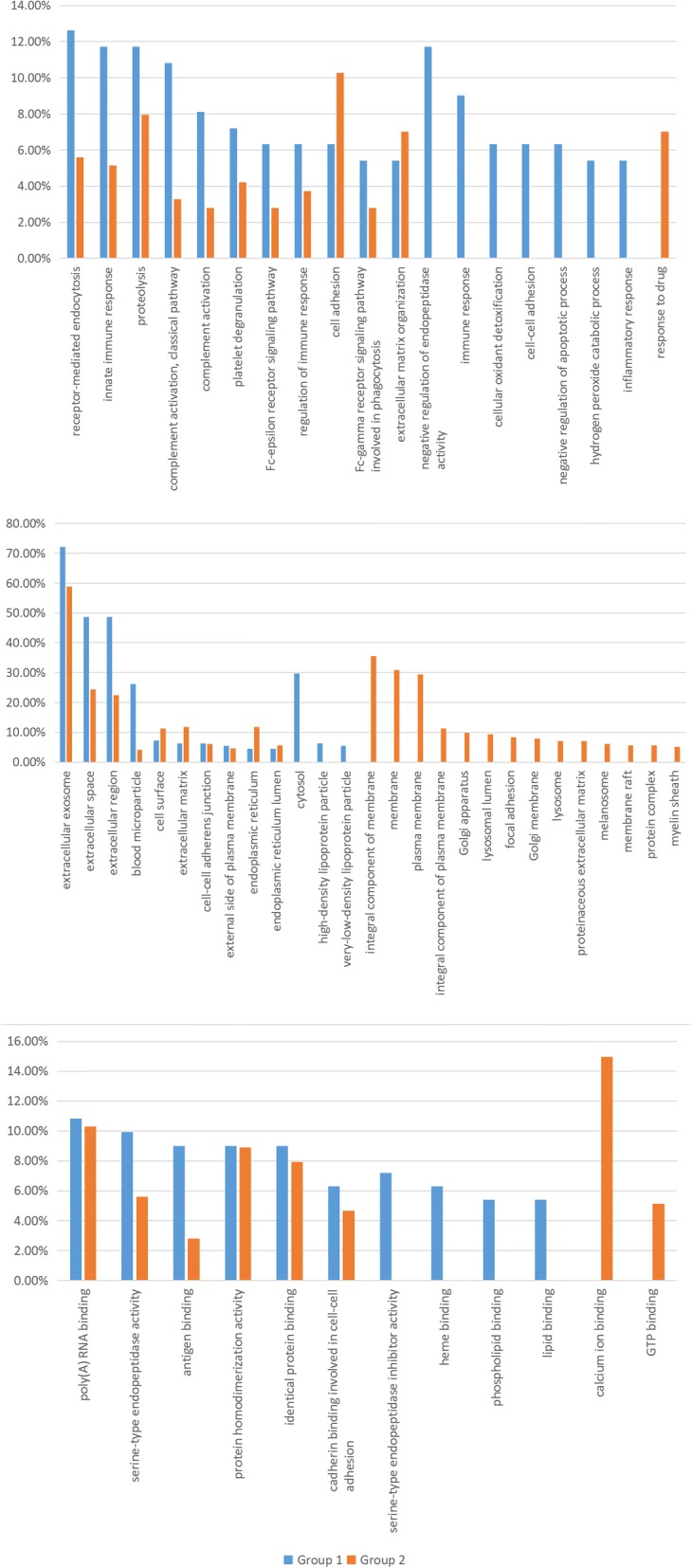
Gene Ontology which involves more than 5% significant proteins from the two groups. A. 5 biological processes relates significantly to more proteins in Group 1, and 7 biological processes are unique to Group 1. B. Three cellular components relate significantly to more proteins in Group 1, and 3 were unique to Group 1. C. Only 1 molecular functions relate significant to more proteins in Group 1, while 4 were unique to Group 1
As shown in Fig. 7a, a total of 5 biological processes were related to significantly more proteins (fold change > 2) in Group 1, including receptor-mediated endocytosis, innate immune response, complement activation, classical pathway, complement activation, and Fc-epsilon receptor signaling pathway.
Moreover, 7 biological processes were unique to Group 1, which included negative regulation of endopeptidase activity, immune response, cellular oxidant detoxification, cell-cell adhesion, negative regulation of apoptotic process, hydrogen peroxide catabolic process and inflammatory response. In addition, one biological process could be identified in Group 2 only.
Cellular component
A total of 105 proteins related to 29 different cellular components in Group 1 could be detected, and 209 proteins from Group 2 were relevant to 59 cellular components. The cellular components that were involved with over 5% proteins were compared. (Fig. 7b).
Extracellular exosome was involved with the most proteins in both groups, and their difference was not significant (fold change < 2). Three cellular components were related to significantly more proteins in Group 1: extracellular space, extracellular region and blood microparticle. Furthermore, 1 cellular component had significantly more proteins in Group 2 which was endoplasmic reticulum.
Lastly, 3 cellular components could be identified in Group 1 only: cytosol, high-density lipoprotein particle and very-low-density lipoprotein particle, while 14 cellular components were unique to Group 2.
Molecular functioning
In Group 1, 34 different molecular functions were identified, involving 99 proteins in total, while in group 2, 200 proteins were detected that were related to 46 molecular functions. Similar analysis was conducted on molecular functions, which concerned approximately 5% proteins.
Apart from molecular functions that could be detected only in Group 1 or 2, the remaining 6 were all associated with more proteins in Group 1 than 2. However, only 1 out of them owned a fold change over 2: antigen binding.
In addition, the four molecular functions that were unique to Group 1 involved serine-type endopeptidase inhibitor activity, heme binding, phospholipid binding and lipid binding. Furthermore, the two unique to Group 2 were calcium ion binding and GTP binding.
Protein–protein interaction analysis
The research pertaining to protein–protein interaction was conducted only on Group 1 using the String online database and Cytoscape software [22].
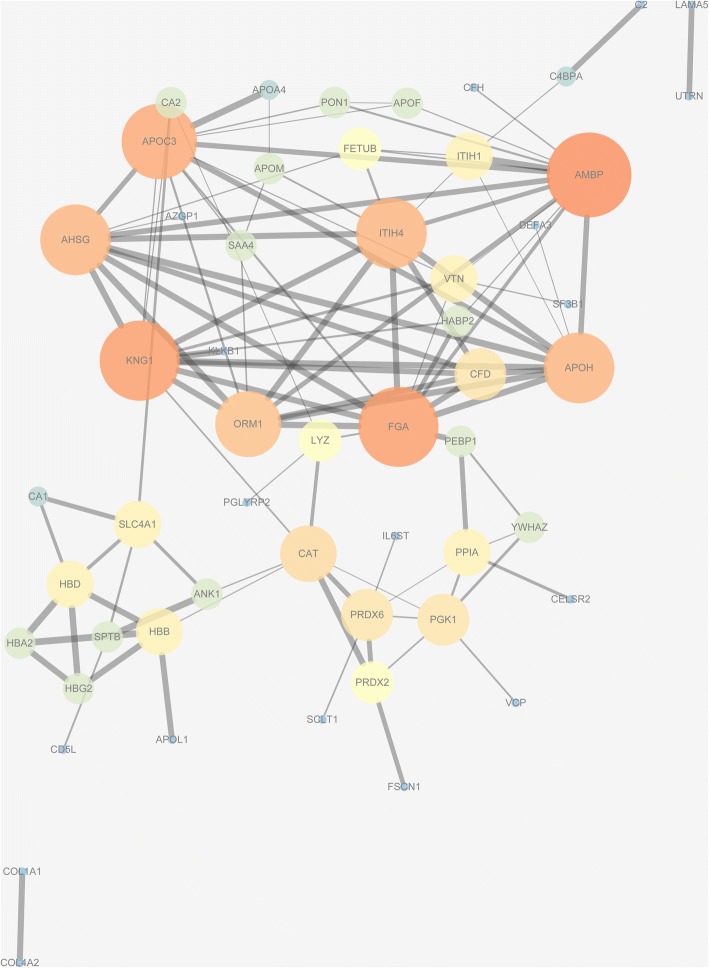
Through the String database, 69 proteins out of 116 were filtered into the PPI network complex, containing 69 nodes and 107 edges, and the remaining 47 did not fall into the PPI network. We processed these statistics using the Cytoscape software and produced an image (Fig. 8). The style of the figure was generated from statistics, to be specific, the size and color were influenced by the degree and the combined score dictated the edge size. It was designed so that low value led to small sizes and dark colors. As shown in the image, the most significant 8 proteins were Protein AMBP (AMBP), Apolipoprotein C-III (APOC3), Kininogen-1 (KNG1), Fibrinogen alpha chain (FGA), Inter-alpha-trypsin inhibitor heavy chain H4 (ITIH4), Alpha-2-HS-glycoprotein (AHSG), Beta-2-glycoprotein 1 (apolipoprotein H) (APOH) and Alpha-1-acid glycoprotein 1 (orosomucoid 1) (ORM1).
Fig. 8.
Protein–protein interaction analysis conducted by the String database and Cytoscape software. The most significant 8 proteins related to PDR is displayed
KEGG pathway
When analyzing Group 1 using the String database, it was concluded that the only involved KEGG pathway were complement and coagulation cascades, which referred to 7 proteins: KNG1, FGA, C2, C4b-binding protein alpha chain (C4BPA), CFD, CFH, and kallikrein B1 (KLKB1). Among them, CFH was an isolated one which had no interaction with other proteins. In total, we obtained 7 nodes and 5 edges and presented the data to the Cytoscape software and then generated the figure style from the statistics as mentioned above (Fig. 9). As shown in Fig. 9, CFD and C4BPA interact only with each other. KLKB1 strongly interplayed with KNG1, which interacted with FGA and CFD; FGA and CFD also interacted.
Fig. 9.
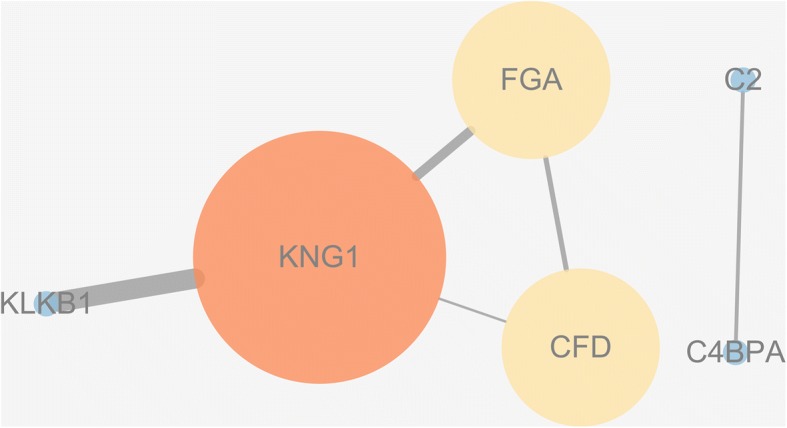
The exclusively involved KEGG pathway and 7 proteins detected by the String database. Complement factor H is an isolated one which has no interaction with other proteins. Complement factor D (CFD) and C4b-binding protein alpha chain interacts mutually and exclusively. Furthers, kallikrein B1 interplays highly with Kininogen-1 and Kininogen-1 has interaction with Fibrinogen alpha chain (FGA) and CFD. Meanwhile, FGA and CFD also interacts
Discussion
Hitherto, there have been several proteomics studies conducted on DR using serum [23–25], tear fluid [26–28], aqueous humor [29] and vitreous [30, 31] from patients. However, only a few have quantitatively analyzed PDR [32]. Loukovaara S et al. analyzed 138 vitreous humor samples from patients with NPDR or PDR which had been the most extensive diabetic vitreous proteome analysis so far [20]. Gao BB et al. studied characterization of the vitreous proteome in diabetes without diabetic retinopathy and PDR [33]. However, these two studies both compared PDR and patients who were in a diabetic condition, and thus could not reveal the comprehensive understanding of the underlying molecular pathogenesis of PDR.
García-Ramírez M et al. compared proteomic analysis on vitreous humor from type 1 diabetic patients with PDR and from non-diabetic patients with IMH using fluorescence-based difference gel electrophoresis (DIGE) [32]. Our study was carried out on type 2 diabetic patients with PDR and on IMH subjects through MS-based label-free quantitative proteomics analysis. In this study, the proteomic analysis of PDR vitreous was optimized in the following ways. First, strict exclusion criteria were applied to protect our result from interference, such as infection, hemorrhage or opacities. Second, our proteomic analysis was quantitative, which could obtain the exact abundance of the proteins detected in the samples so that the difference of protein expression in these two diseases could be quantified. An analysis of protein distribution was carried out so that we could gain a brief understanding of protein content, sample reliability as well as a clear separation of the two disease states. Detailed bioinformatic analysis was conducted, which included protein function, related disease, biological processes, molecular functioning, cellular components and the KEGG pathway. Therefore, many inspirations about the target protein function, biomarkers, physiological and pathological changes and the related pathways were revealed through this study.
To our knowledge, this research was the first quantitative proteomics analysis to study the protein distribution which could obtain a brief understanding of protein content and sample reliability. A clear separation of the protein expression in the two diseases was observed. When only taking significant proteins into account, the separation became more apparent and the difference among the samples in one group became less because confounding factors were reduced. Besides, more proteins in the control group had abnormal abundance and C5 was the most unreliable one, the reason of which could be that IMH was another disease condition which might be the result of a variety of unknown factors. Due to its complexity, we just counted IMH samples as controls but did not analyze the related proteins or statistics. Moreover, although there was a weaker correlation among IMH samples, a wider protein distribution in PDR samples was detected which illustrated that PDR was a complex disease which influenced a great deal of protein expression.
A comprehensive bioinformatics research was carried out which provided us with a better understanding of the pathomechanism of PDR as well as the molecular signaling pathway, thus could be of great value to the development of therapeutic method. Apolipoproteins have been reported involving with the pathology of PDR [34, 35], here we identified a total of 6 apolipoproteins, 5 were the first time to be found related to PDR while the remaining one, apolipoprotein A-IV (APOA4) was found reduced in the PDR patients compared with the macular hole patients [36] which had conflicts with our result. Besides, several researches related to PDR have been carried out on vision related proteins, such as CA1 [37], CAT [38], CFH [39], PON1 [40] and VTN [41]. However, no researches have been conducted on APOF, COL1A1, C2, C4BPA, CFD and CFHR3, FGA. Furthermore, KNG1 has never been studied in the pathology of PDR. It initiates the kallikrein-kinin system which is related to the blood pressure systems and inflammation, the latter one is known to cause dilation of blood vessels and increase vascular permeability.
However, there are some limitations in our study. First, the IMH patients were enrolled as controls. Although IMH has no protopathy, it does not equal to normal condition. Because any pathological state may lead to changes in protein profile, normal eyes are the best options for the control group, whereas it is against the ethic. Second, further validation study like dot blot, western blot, enzyme-linked immunosorbent assays or immunohistochemistry should have been conducted in order to confirm some proteins of particular interest. Furthermore, the role of a protein in pathogenesis could have been tested by injection of purified protein into an animal model or by using a pharmaceutical invention.
Conclusions
In conclusion, we identified and quantified 610 proteins in total, which included 338 significant proteins in our study. Protein distribution analysis demonstrated a clear separation of protein expression in PDR and IMH. The protein function analysis illustrated that immunity and transport related proteins might be associated with PDR. In addition, 12 proteins were identified that were related to vision diseases, some of which have already been studied, but more studies should be carried out to analyze the relation between these proteins and PDR as well as the underlying mechanisms. Meanwhile, GO annotation indicated that the proteins in the vitreous may reflected the physiological and pathological changes in retinal lesion. Also, PPI analysis in our study identified the most significant 8 proteins related to PDR. Furthermore, pathway research indicated the complement and coagulation cascades might be the most important pathway and KNG1 is the key protein in the pathology of PDR.
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (30972712, 81671641, and 81700847), the Natural Science Foundation of Jiangsu Province (Grants No. BK20151208 and BK20170365), Soochow Scholar Project of Soochow University (R5122001), and Jiangsu Province’s Outstanding Medical Academic Leader program (CXTDA2017039).
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AHSG
Alpha-2-HS-glycoprotein
- AMD
Age-related macular degeneration
- APOA4
apolipoprotein A-IV
- APOC3
Apolipoprotein C-III
- APOF
Apolipoprotein
- APOH
Apolipoprotein H
- BCA
Bicinchoninic acid
- C2
Complement C2
- C4BPA
C4b-binding protein alpha chain
- C4BPA
Complement component 4 binding protein alpha
- CA1
Carbonic anhydrase 1
- CAT
Catalase
- CFD
Complement factor D
- CFH
Complement factor H
- CFHR3
Complement factor H related 3
- COL1A1
Collagen type I alpha 1 chain
- DIGE
Difference gel electrophoresis
- DR
Diabetic retinopathy
- FGA
Fibrinogen alpha chain
- GO
Gene ontology
- HCA
Hierarchical clustering analysis
- IMH
Idiopathic macular hole
- IRMA
Intraretinal microvascular abnormalities
- ITIH4
Inter-alpha-trypsin inhibitor heavy chain H4
- KEGG
Kyoto Encyclopedia of Genes and Genomes
- KLKB1
Kallikrein B1
- KNG1
Kininogen-1
- MS
Mass spectrometry
- NPDR
Nonproliferative diabetic retinopathy
- ORM1
Orosomucoid 1
- PC
Principal component analysis
- PDR
Proliferative diabetic retinopathy
- PON1
Paraoxonase 1
- PPI
Protein-protein interaction
- VTN
Vitronectin
Authors’ contributions
JL and QL made substantial contributions to conception and design of this study and drafted the manuscript; PL revised it critically for important intellectual content and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All authors have given the final approval of the version to be published.
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study was approved by the institutional ethics committee of the First Affiliated Hospital of Soochow University and signed informed consent was obtained from every participant before being included into the study.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Footnotes
Jianqing Li and Qianyi Lu contributed equally to this work.
References
- 1.Antonetti DA, Klein R, Gardner TW. Diabetic retinopathy. N Engl J Med. 2012;366(13):1227–1239. doi: 10.1056/NEJMra1005073. [DOI] [PubMed] [Google Scholar]
- 2.Stitt AW, Curtis TM, Chen M, Medina RJ, McKay GJ, Jenkins A, Gardiner TA, Lyons TJ, Hammes HP, Simo R, et al. The progress in understanding and treatment of diabetic retinopathy. Prog Retin Eye Res. 2016;51:156–186. doi: 10.1016/j.preteyeres.2015.08.001. [DOI] [PubMed] [Google Scholar]
- 3.Liu Y, Swearingen R. Diabetic Eye Screening: Knowledge and Perspectives from Providers and Patients. Curr Diab Rep. 2017;17(10):94. doi: 10.1007/s11892-017-0911-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Duh EJ, Sun JK, Stitt AW. Diabetic retinopathy: current understanding, mechanisms, and treatment strategies. JCI insight. 2017;2(14):e93751. [DOI] [PMC free article] [PubMed]
- 5.Schatz P, Aldayel A, Taskintuna I, Abdelkader E, Mura M. Serous retinal detachment after panretinal photocoagulation for proliferative diabetic retinopathy: a case report. J Med Case Rep. 2017;11(1):265. doi: 10.1186/s13256-017-1424-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li X, Zarbin MA, Bhagat N. Anti-Vascular Endothelial Growth Factor Injections: The New Standard of Care in Proliferative Diabetic Retinopathy? Dev Ophthalmol. 2017;60:131–142. doi: 10.1159/000459699. [DOI] [PubMed] [Google Scholar]
- 7.Sato T, Tsuboi K, Nakashima H, Emi K. Characteristics of cases with postoperative vitreous hemorrhage after 25-gauge vitrectomy for repair of proliferative diabetic retinopathy. Graefe’s Arch Clin Exp Ophthalmol. 2017;255(4):665–671. doi: 10.1007/s00417-016-3522-8. [DOI] [PubMed] [Google Scholar]
- 8.Olivares AM, Althoff K, Chen GF, Wu S, Morrisson MA, DeAngelis MM, Haider N. Animal Models of Diabetic Retinopathy. Curr Diab Rep. 2017;17(10):93. doi: 10.1007/s11892-017-0913-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cehofski LJ, Honore B, Vorum H. A Review: Proteomics in Retinal Artery Occlusion, Retinal Vein Occlusion, Diabetic Retinopathy and Acquired Macular Disorders. Int J Mol Sci. 2017;18:907. 10.3390/ijms18050907. [DOI] [PMC free article] [PubMed]
- 10.Angi M, Kalirai H, Coupland SE, Damato BE, Semeraro F, Romano MR. Proteomic analyses of the vitreous humour. Mediators Inflamm. 2012;2012:148039. doi: 10.1155/2012/148039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Matthiesen R, Bunkenborg J. Introduction to mass spectrometry-based proteomics. Methods Mol Biol. 2013;1007:1–45. doi: 10.1007/978-1-62703-392-3_1. [DOI] [PubMed] [Google Scholar]
- 12.Kyselova Z. Mass spectrometry-based proteomics approaches applied in cataract research. Mass Spectrom Rev. 2011;30(6):1173–1184. doi: 10.1002/mas.20317. [DOI] [PubMed] [Google Scholar]
- 13.Koss MJ, Hoffmann J, Nguyen N, Pfister M, Mischak H, Mullen W, Husi H, Rejdak R, Koch F, Jankowski J, et al. Proteomics of vitreous humor of patients with exudative age-related macular degeneration. PloS One. 2014;9(5):e96895. doi: 10.1371/journal.pone.0096895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kim TW, Kang JW, Ahn J, Lee EK, Cho KC, Han BN, Hong NY, Park J, Kim KP. Proteomic analysis of the aqueous humor in age-related macular degeneration (AMD) patients. J Proteome Res. 2012;11(8):4034–4043. doi: 10.1021/pr300080s. [DOI] [PubMed] [Google Scholar]
- 15.Csosz E, Deak E, Kallo G, Csutak A, Tozser J. Diabetic retinopathy: Proteomic approaches to help the differential diagnosis and to understand the underlying molecular mechanisms. J Proteomics. 2017;150:351–358. doi: 10.1016/j.jprot.2016.06.034. [DOI] [PubMed] [Google Scholar]
- 16.Kim HJ, Kim PK, Yoo HS, Kim CW. Comparison of tear proteins between healthy and early diabetic retinopathy patients. Clin Biochem. 2012;45(1–2):60–67. doi: 10.1016/j.clinbiochem.2011.10.006. [DOI] [PubMed] [Google Scholar]
- 17.Balaiya S, Zhou Z, Chalam KV. Characterization of Vitreous and Aqueous Proteome in Humans With Proliferative Diabetic Retinopathy and Its Clinical Correlation. Proteomics Insights. 2017;8:1178641816686078. doi: 10.1177/1178641816686078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Simo-Servat O, Hernandez C, Simo R. Usefulness of the vitreous fluid analysis in the translational research of diabetic retinopathy. Mediators Inflamm. 2012;2012:872978. doi: 10.1155/2012/872978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Nikolov M, Schmidt C, Urlaub H. Quantitative mass spectrometry-based proteomics: an overview. Methods Mol Biol. 2012;893:85–100. doi: 10.1007/978-1-61779-885-6_7. [DOI] [PubMed] [Google Scholar]
- 20.Loukovaara S, Nurkkala H, Tamene F, Gucciardo E, Liu X, Repo P, Lehti K, Varjosalo M. Quantitative Proteomics Analysis of Vitreous Humor from Diabetic Retinopathy Patients. J Proteome Res. 2015;14(12):5131–5143. doi: 10.1021/acs.jproteome.5b00900. [DOI] [PubMed] [Google Scholar]
- 21.Gene Ontology Consortium The Gene Ontology project in 2008. Nucleic Acids Res. 2008;36(Database issue):D440–D444. doi: 10.1093/nar/gkm883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Guo Y, Bao Y, Ma M, Yang W. Identification of Key Candidate Genes and Pathways in Colorectal Cancer by Integrated Bioinformatical Analysis. Int J Mol Sci. 2017;18(4) [DOI] [PMC free article] [PubMed]
- 23.Gopalakrishnan V, Purushothaman P, Bhaskar A. Proteomic analysis of plasma proteins in diabetic retinopathy patients by two dimensional electrophoresis and MALDI-Tof-MS. J Diabetes Complicat. 2015;29(7):928–936. doi: 10.1016/j.jdiacomp.2015.05.021. [DOI] [PubMed] [Google Scholar]
- 24.Lu CH, Lin ST, Chou HC, Lee YR, Chan HL. Proteomic analysis of retinopathy-related plasma biomarkers in diabetic patients. Arch Biochem Biophys. 2013;529(2):146–156. doi: 10.1016/j.abb.2012.11.004. [DOI] [PubMed] [Google Scholar]
- 25.Liu YP, Hu SW, Wu ZF, Mei LX, Lang P, Lu XH. Proteomic analysis of human serum from diabetic retinopathy. Int J Ophthalmol. 2011;4(6):616–622. doi: 10.3980/j.issn.2222-3959.2011.06.08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Torok Z, Peto T, Csosz E, Tukacs E, Molnar AM, Berta A, Tozser J, Hajdu A, Nagy V, Domokos B, et al. Combined Methods for Diabetic Retinopathy Screening, Using Retina Photographs and Tear Fluid Proteomics Biomarkers. J Diabetes Res. 2015;2015:623619. doi: 10.1155/2015/623619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Csosz E, Boross P, Csutak A, Berta A, Toth F, Poliska S, Torok Z, Tozser J. Quantitative analysis of proteins in the tear fluid of patients with diabetic retinopathy. J Proteomics. 2012;75(7):2196–2204. doi: 10.1016/j.jprot.2012.01.019. [DOI] [PubMed] [Google Scholar]
- 28.Torok Z, Peto T, Csosz E, Tukacs E, Molnar A, Maros-Szabo Z, Berta A, Tozser J, Hajdu A, Nagy V, et al. Tear fluid proteomics multimarkers for diabetic retinopathy screening. BMC Ophthalmol. 2013;13(1):40. doi: 10.1186/1471-2415-13-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chiang SY, Tsai ML, Wang CY, Chen A, Chou YC, Hsia CW, Wu YF, Chen HM, Huang TH, Chen PH, et al. Proteomic analysis and identification of aqueous humor proteins with a pathophysiological role in diabetic retinopathy. J Proteomics. 2012;75(10):2950–2959. doi: 10.1016/j.jprot.2011.12.006. [DOI] [PubMed] [Google Scholar]
- 30.Wang H, Feng L, Hu J, Xie C, Wang F. Differentiating vitreous proteomes in proliferative diabetic retinopathy using high-performance liquid chromatography coupled to tandem mass spectrometry. Exp Eye Res. 2013;108:110–119. doi: 10.1016/j.exer.2012.11.023. [DOI] [PubMed] [Google Scholar]
- 31.Kim T, Kim SJ, Kim K, Kang UB, Lee C, Park KS, Yu HG, Kim Y. Profiling of vitreous proteomes from proliferative diabetic retinopathy and nondiabetic patients. Proteomics. 2007;7(22):4203–4215. doi: 10.1002/pmic.200700745. [DOI] [PubMed] [Google Scholar]
- 32.Garcia-Ramirez M, Canals F, Hernandez C, Colome N, Ferrer C, Carrasco E, Garcia-Arumi J, Simo R. Proteomic analysis of human vitreous fluid by fluorescence-based difference gel electrophoresis (DIGE): a new strategy for identifying potential candidates in the pathogenesis of proliferative diabetic retinopathy. Diabetologia. 2007;50(6):1294–1303. doi: 10.1007/s00125-007-0627-y. [DOI] [PubMed] [Google Scholar]
- 33.Gao BB, Chen X, Timothy N, Aiello LP, Feener EP. Characterization of the vitreous proteome in diabetes without diabetic retinopathy and diabetes with proliferative diabetic retinopathy. J Proteome Res. 2008;7(6):2516–2525. doi: 10.1021/pr800112g. [DOI] [PubMed] [Google Scholar]
- 34.Ankit BS, Mathur G, Agrawal RP, Mathur KC. Stronger relationship of serum apolipoprotein A-1 and B with diabetic retinopathy than traditional lipids. Indian J Endocrinol Metab. 2017;21(1):102–105. doi: 10.4103/2230-8210.196030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hu A, Luo Y, Li T, Guo X, Ding X, Zhu X, Wang X, Tang S. Low serum apolipoprotein A1/B ratio is associated with proliferative diabetic retinopathy in type 2 diabetes. Graefe’s Arch Clin Exp Ophthalmol. 2012;250(7):957–962. doi: 10.1007/s00417-011-1855-x. [DOI] [PubMed] [Google Scholar]
- 36.Kim SJ, Kim S, Park J, Lee HK, Park KS, Yu HG, Kim Y. Differential expression of vitreous proteins in proliferative diabetic retinopathy. Curr Eye Res. 2006;31(3):231–240. doi: 10.1080/02713680600557030. [DOI] [PubMed] [Google Scholar]
- 37.Gao BB, Clermont A, Rook S, Fonda SJ, Srinivasan VJ, Wojtkowski M, Fujimoto JG, Avery RL, Arrigg PG, Bursell SE, et al. Extracellular carbonic anhydrase mediates hemorrhagic retinal and cerebral vascular permeability through prekallikrein activation. Nat Med. 2007;13(2):181–188. doi: 10.1038/nm1534. [DOI] [PubMed] [Google Scholar]
- 38.Sozmen EY, Sozmen B, Delen Y, Onat T. Catalase/superoxide dismutase (SOD) and catalase/paraoxonase (PON) ratios may implicate poor glycemic control. Arch Med Res. 2001;32(4):283–287. doi: 10.1016/S0188-4409(01)00285-5. [DOI] [PubMed] [Google Scholar]
- 39.Toni M, Hermida J, Toledo E, Goni MJ, Diez Goni N. Role of CFH and ARMS2 polymorphisms in retinopathy and coronary artery disease in type 1 diabetes. An Sist Sanit Navar. 2012;35(3):425–432. doi: 10.4321/S1137-66272012000300008. [DOI] [PubMed] [Google Scholar]
- 40.Nowak M, Wielkoszynski T, Marek B, Kos-Kudla B, Swietochowska E, Sieminska L, Karpe J, Kajdaniuk D, Glogowska-Szelag J, Nowak K. Antioxidant potential, paraoxonase 1, ceruloplasmin activity and C-reactive protein concentration in diabetic retinopathy. Clin Exp Med. 2010;10(3):185–192. doi: 10.1007/s10238-009-0084-7. [DOI] [PubMed] [Google Scholar]
- 41.Casaroli Marano RP, Preissner KT, Vilaro S. Fibronectin, laminin, vitronectin and their receptors at newly-formed capillaries in proliferative diabetic retinopathy. Exp Eye Res. 1995;60(1):5–17. doi: 10.1016/S0014-4835(05)80079-X. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.