Abstract
Brugada syndrome (BrS) is a cardiac disease caused by an inherited ion channelopathy associated with a propensity to develop ventricular fibrillation. Implantable cardioverter defibrillator implantation is recommended in BrS, based on the clinical presentation in the presence of diagnostic ECG criteria. Implantable cardioverter defibrillator implantation is not always indicated or sufficient in BrS, and is associated with a high device complication rate. Pharmacological therapy aimed at rebalancing the membrane action potential can prevent arrhythmogenesis in BrS. Quinidine, a class 1A antiarrhythmic drug with significant Ito blocking properties, is the most extensively used drug for the prevention of arrhythmias in BrS. The present review provides contemporary data gathered on all drugs effective in the therapy of BrS, and on ineffective or contraindicated antiarrhythmic drugs.
Keywords: Brugada syndrome,; arrhythmia,; pharmacology,; quinidine,; disopyramide,; isoproterenol,; denopamine,; orciprenaline,; cilostazol,; bepridil,; quinine
Brugada syndrome (BrS) is a cardiac disease caused by an inherited ion channelopathy. It was first described by the Brugada brothers in 1992[1] and is associated with a propensity to develop ventricular fibrillation (VF). Brugada syndrome is characterised by prominent J waves appearing as an ST segment elevation in the right precordial leads. In the latest guidelines, diagnosis of BrS constitutes ST elevation with type 1 morphology (coved) ≥2 mm in one or more leads among the right precordial leads V1 and V2, occurring either spontaneously or after intravenous administration of class I antiarrhythmic drugs.[2–4] A recent expert consensus differs in the definition and recommends that when a type 1 ST segment elevation is unmasked using a sodium channel blocker, diagnosis of BrS should require that the patient also presents with one of the following: documented VF or polymorphic ventricular tachycardia (VT), syncope of probable arrhythmic cause, a family history of sudden cardiac death (SCD) at <45 years of age with negative autopsy, coved-type ECGs in family members, or nocturnal agonal respiration.[5]
Historically, BrS was considered to be inherited in an autosomal dominant inheritance with incomplete penetrance. There is growing evidence that presenting with BrS and the susceptibility to VF and SCD may not be due to a single mutation (classic Mendelian view) but rather to inheritance of multiple BrS susceptibility variants (oligogenic) acting in concert through one or more mechanistic pathways.[5] Multiple mutations have been associated with the Brugada phenotype. These mutations cause either a decrease in the inward sodium or calcium current or an increase in the transient outward potassium channel current (Ito). Both result in an outward shift in the net transmembrane current active at the end of phase one of the right ventricular epicardial action potential (where Ito is most prominent).[6–8]
The guidelines recommend ICD implantation in patients with BrS who have survived a cardiac arrest, or have documented spontaneous sustained VT (class I).[3,4] ICD implantation should also be considered in patients with BrS who have had a syncopal episode suspected to be caused by VT or VF (class IIa). In asymptomatic BrS individuals who have inducible sustained VF during programmed ventricular stimulation with two or three extrastimuli at two sites, ICD implantation is controversial and is a class IIb indication. ICDs are not recommended (class III) in reflex-mediated syncope or in asymptomatic patients.[3,4] This recommendation is based on the low yearly event rate of 0.5 % found in asymptomatic Brugada patients,[9,10] coupled with the high complication rate reported for ICDs in BrS.[11–13] Aggregate rates of inappropriate shocks and lead failure have been reported to be as high as 37 and 29 %, respectively, at 10 years, including one death as a result of inappropriate ICD discharge resulting from lead failure.[14,15] A near-fatal VF in BrS despite an implanted ICD has been reported, suggesting that lone therapy of ICD in cardiac arrest survivors with BrS is not without risks.[16] The death of a BrS patient diagnosed after syncope (with an ajmaline provocation test), due to incessant VT and VF that developed during lead extraction procedure, has also been reported.[17] In children and adolescents with BrS, high rates of inappropriate shocks and device-related complications were reported at 20 and 14 %, respectively.[18] The option of subcutaneous ICD (S-ICD) for young patients with inherited arrhythmic syndromes who do not need pacing therapy is being increasingly used. A recent study found high rates of sensing screening failure in patients with BrS, due to high T wave voltages.[19] Long-term clinical data are lacking at present on the utility of S-ICD in BrS.
A pharmacological therapy approach aimed at rebalancing the epicardial action potential in the right ventricle and normalising the action potential dome can prevent arrhythmogenesis in BrS, unlike a device therapy approach, which addresses only the symptoms of BrS without preventing the arrhythmias from occurring. Drug therapy in BrS has several utilities: first, in the acute management of arrhythmic storm; second, in prevention of arrhythmic events in patients with implanted ICD who require many shocks; and third, as an alternative to ICD implantation when the latter is contraindicated, not feasible (infants and young children), unaffordable, or refused by the patient. This review provides contemporary data gathered on all drugs effective in the therapy of BrS as well as ineffective or contraindicated antiarrhythmic drugs that should be avoided.
Class IA Antiarrhythmic Drugs
Quinidine
Quinidine’s beneficial effect in preventing arrhythmic events in BrS is mainly attributed to its significant Ito blocking property. It was shown that quinidine is effective in normalising the epicardial action potential dome and the ST segment and preventing phase-two re-entry and polymorphic VT in experimental models of BrS.[6,20,21] The anticholinergic effect of quinidine may also contribute to its antiarrhythmic effect.[8]
Electrophysiologically Guided Quinidine Therapy
The first case showing the efficacy of oral quinidine in preventing inducible VF during electrophysiological study (EPS) and recurrent arrhythmias was reported in 1981 by Belhassen et al.[22] in a young male with recurrent storms of idiopathic VF (IVF). This patient is still arrhythmia-free on quinidine treatment and with no implanted ICD after a follow-up of 39 years. Two of the five patients with IVF successfully treated with EP-guided class 1A antiarrhythmic drugs (AD) were later diagnosed as suffering from BrS.[23] The first prospective series of 25 BrS patients treated with EP-guided therapy were also reported by Belhassen et al.[24] Quinidine prevented VF inducibility in 22 (88 %) patients and no arrhythmic events were observed during follow-up in EP-quinidine responders treated by the medication. The latest study reporting the 33-year experience of the same group involved 96 patients with BrS (10 cardiac arrest survivors, 27 who presented with syncope and 59 who were asymptomatic). VF was induced in 66 (68.8 %) patients using an aggressive protocol of programmed ventricular stimulation (inducibility rates of 100 %, 74 % and 61 % in patients with cardiac arrest, syncope and no symptoms, respectively). All but six of the 66 patients with inducible VF underwent EPS on quinidine (n=54), disopyramide (n=2) or both (n=4). Two different formulations of quinidine were used during EP-guided therapy: quinidine bisulphate (QBS) (750–2000 mg daily) and hydroquinidine chlorhydrate (HQ) (600–900 mg daily). Fifty-four (90 %) patients were EP responders to more than one AD with similar efficacy rates (≈90 %) in all patient groups. After a mean follow-up of 113.3 ± 71.5 months, 92 patients were alive, whereas four had died from non-cardiac causes. No arrhythmic event occurred during class 1A AD therapy in any of the EP drug responders and in patients with no baseline inducible VF. Arrhythmic events occurred in only two cardiac arrest survivors treated with ICD alone but did not recur on quinidine. All cases of recurrent syncope (n=12) were attributed to a vasovagal (n=10) or nonarrhythmic mechanism (n=2). Class 1A AD resulted in a 38 % incidence of side-effects (mainly diarrhoea) that resolved after drug discontinuation. Sixty per cent of patients were compliant with the medication by the end of follow up.[25] An illustrative example of the efficacy of quinidine in preventing VF induction is shown in Figures 1 and 2.
Figure 1: Twelve-lead ECG of a Man Aged 38 Years Presenting with Syncope.
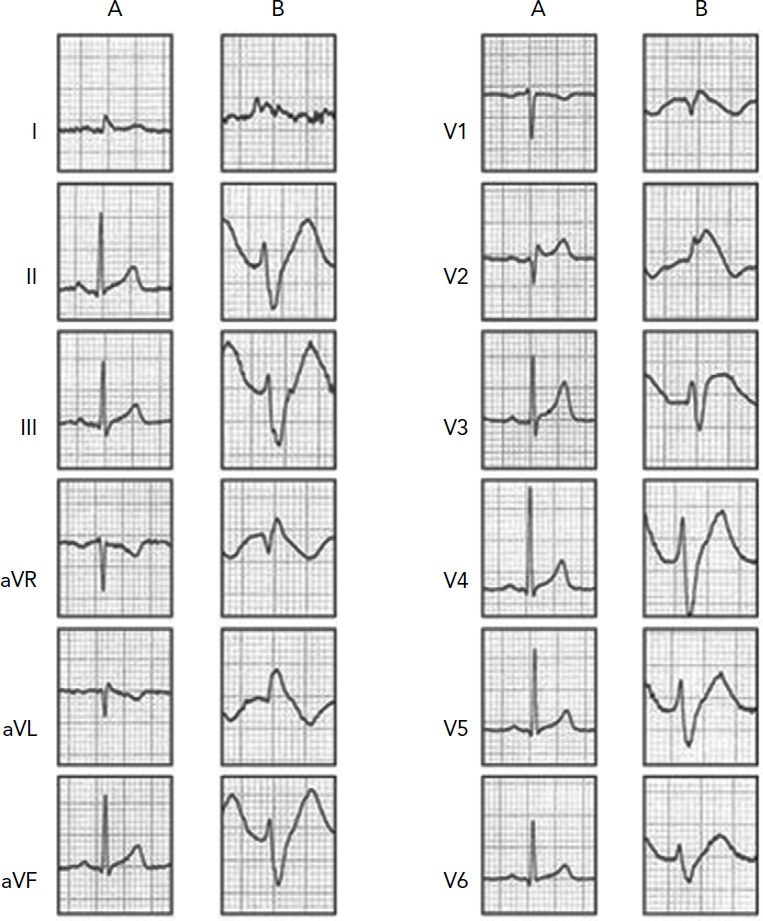
Syncope occurred when the patient had stopped at a red light while driving. Echocardiogram was normal. A: Baseline ECG was unremarkable except for the presence of 1 mm saddle ST-elevation in V2; B: During a flecainide challenge test, type 1 Brugada-ECG was unmasked.
Figure 2: Twelve-lead ECG of a Man Aged 38 Years Undergoing Programmed Ventricular Stimulation.
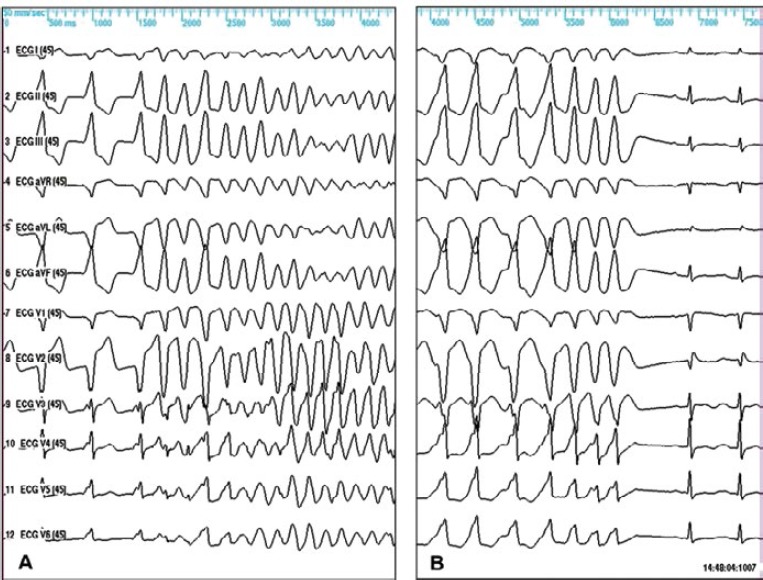
A: Baseline programmed ventricular stimulation (PVS). Induction of sustained polymorphic VT using double extrastimuli from the right ventricular outflow tract (RVOT); B: Repeated PVS on quinidine bisulfate (1,500 mg/day, 2.78 mg/l) demonstrated no inducible arrhythmias using triple extrastimuli delivered from the RV apex and the RVOT, with repetition (n=5) of triple extrastimulation at the shortest coupling intervals. The patient has remained asymptomatic on quinidine therapy during follow-up without implantation of an ICD.
Based on these results, Belhassen et al.[25] suggested that EP-guided therapy may be an excellent alternative to ICD therapy in selected patients who are committed to a life-long drug therapy and exhibit good tolerance to the medication.
Hermida et al.[26] reported data of 31 asymptomatic BrS patients with inducible VF at baseline. They used HQ 600 mg daily in all their patients but two in whom they used 900 mg and found prevention of VT/VF inducibility in 76 % of their inducible patients. Syncope occurred in two of the 21 patients who received long-term (17 ± 13 months) HQ therapy. One syncope associated with QT interval prolongation and one unexplained syncope associated with probable noncompliance.[26] Bouzeman et al.[27] reported the results from two French centres, on 44 asymptomatic BrS patients. In 34 (77 %) of these patients, 600 mg HQ daily effectively prevented VF induction during a follow-up period of 3–6.2 years. VF occurred only in one patient in whom HQ was discontinued due to intolerance. Among the 10 other patients (22 %) who remained inducible and received ICD, none received appropriate therapy during a mean follow-up of 2–7.7 years. The overall annual rate of arrhythmic events was 1.04 %. One-third of patients experienced device-related complications.[27] The higher rates of prevention of VF inducibility reported by Belhassen might be explained by the higher doses of HQ were used in their studies[24,25,28] (900 mg daily versus 600 mg daily).[26,27]
It is noteworthy that in the current guidelines “quinidine should be considered (class IIa) in patients who qualify for an ICD but present a contraindication or refuse it, and in patients requiring treatment for supraventricular arrhythmias”.[4]
Empiric Quinidine Therapy for Arrhythmic Storm
Electric storms of VF represent the more malignant form of ventricular arrhythmias in patients with BrS, accounting for up to 10 % of the untreated patient population[29] and up to 38 % of arrhythmic events in ICD patients.[30] Isoproterenol infusion has been shown to be very effective in the acute management of arrhythmic storms in the setting of BrS.[31] However, it can be administered only intravenously during a short period of time, and the arrhythmia frequently recurs upon drug discontinuation. Quinidine has been shown in multiple studies to successfully control arrhythmic storms as a single agent or in combination with isoproterenol.[29,32–42] Anguera[43] reported the effectiveness of quinidine in 29 BrS patients treated for secondary prevention due to arrhythmic storm (31 %) or frequent ICD shocks (69 %). Ten patients received QBS (mean dose 591 ± 239 mg/day), and 19 patients HQ (mean dose 697 ± 318 mg/day). After a mean period of 60 ± 41 months under quinidine treatment, 19 patients (66 %) remained free of appropriate ICD discharges. A significant reduction in total number of shocks and median number of shocks per patient was observed, from 203 to 41 shocks and from six shocks per patient IQR (4 to 9) to no shocks per patient IQR (0 to 2.5), p<0.0001, respectively. A total of 10 patients (34 %) experienced at least one recurrent shock (in four patients, shocks were related to a reduction in the dose of quinidine due to side-effects [n=2] or to temporary discontinuation of treatment by the patient [n=2]). During quinidine therapy, QT intervals corrected for heart rate increased by a mean of <10 % (413 ± 18 to 442 ± 35; p=0.001) without episodes of torsade de pointes. Side-effects appeared in five patients (17 %). The effectiveness of quinidine in controlling VF storm has also been described in children[44,45] and pregnant women.[46] The use of low-dose quinidine (<600 mg) in four patients with arrhythmic storms was reported by Márquez et al.[47]; quinidine prevented the recurrence of arrhythmic events in all patients.[47]
In the current guidelines, quinidine should be considered (class IIa indication) in BrS patients presenting with electrical storms and in patients implanted with an ICD who are experiencing repeated appropriate shocks.[4]
Empiric Quinidine Therapy for Asymptomatic Patients
The prophylactic use of empirical quinidine for asymptomatic patients with type 1 Brugada-ECG was first suggested by Viskin et al.[48] Preliminary results in 19 patients who received empiric quinidine therapy were reported.[49] No arrhythmic events occurred, and five patients (26 %) discontinued therapy due to side-effects. The administration of quinidine without further risk stratification to all asymptomatic patients with a type 1 Brugada-ECG is controversial. We routinely give quinidine to asymptomatic patients only in those who have inducible ventricular arrhythmias at EP testing.[25] The recently published QUIDAM study: Hydroquinidine Therapy for the Management of Brugada Syndrome Patients at High Arrhythmic Risk[50] is a prospective multicentre randomised (HQ versus placebo) double-blinded study with two 18-month crossover phases in high-risk patients with BrS and implanted with an ICD. Of the 50 patients enrolled, only 26 (52 %) fully completed both phases. Thirty-four (68 %) patients presented HQ-related side-effects, mainly gastrointestinal, which led to discontinuation of the therapy in 13 (26 %). During the 36-month follow-up period, two arrhythmic events occurred under placebo therapy (one appropriate ICD shock [0.97 % event per year] and one self-terminating VF). In addition, one inappropriate ICD shock occurred under placebo therapy. No arrhythmic events were reported under HQ therapy. The authors concluded that although HQ seems to be effective in preventing life-threatening arrhythmic events, it could not be an alternative for ICD implantation due to its frequent side-effects limiting compliance to the drug. This study, although well structured, was underpowered to prove the efficacy of HQ due to the small number of patients enrolled and even smaller number of patients who completed both phases. In addition, the very high incidence of side-effects reported was inconsistent with previous reports.[25,27] In his editorial comment on the paper by Andorin et al., Belhassen[51] pointed to the study’s patient population being mainly at intermediate and low arrhythmic risk, which explains the difficulty of assessing the efficacy of quinidine therapy, even more so with a small cohort population. In addition, he postulated that the high rate of side effects (mainly diarrhoea) requiring drug discontinuation could have been related to patients’ reluctance to continue the trial while they felt fully protected by the implanted ICDs.
Due to the low incidence of arrhythmic events in asymptomatic BrS patients, a large-scale double-blinded trial is needed for further validation of the efficacy of HQ in asymptomatic BrS patients. A prospective registry of empiric quinidine for asymptomatic Brugada syndrome has been established.[48]
Low-dose Quinidine
To decrease the frequency of side-effects associated with quinidine, administration of lower doses of the drug have been attempted. Márquez et al.[47] reported a small series of six BrS patients, in whom doses of quinidine or hydroquinidine (<600 mg) prevented the recurrence of arrhythmic events in all patients without side-effects during a median follow-up of four years. In the literature review, 14 additional patients treated with <600 mg of quinidine were found. Quinidine was well tolerated and associated with acute and long-term arrhythmia control in 85 % of cases. In four patients who stopped taking the medication, recurrent arrhythmias occurred, which were successfully controlled after treatment was reinitiated.[47] Hasegawa reported the normalisation of J waves and coved-type ST-segment elevation with VF suppression in a patient who presented with a VF storm after daily administration of 300 mg quinidine and a follow-up of 20 months.[52] These data suggest that low doses of quinidine, which are generally well tolerated, may be useful in controlling arrhythmias. Further studies of larger scale are needed to validate the efficacy and safety of low-dose HQ.
Limited Worldwide Commercial Availability of the Drug
A major limitation for the wide use of quinidine in the world is its lack of accessibility.[53,54] Viskin et al.[54] asked a total of 273 physicians from 131 countries regarding the availability of quinidine in their countries. Quinidine was readily available in 19 countries (14 %), not accessible in 99 countries (76 %), and available only through specific regulatory processes that require 4 to 90 days for completion in 13 countries (10 %). Viskin et al.[54] were able to gather information concerning 22 patients who had serious arrhythmias probably related (10 cases) or possibly related (12 cases) to the absence of quinidine, including two fatalities possibly attributable to the unavailability of quinidine.
Disopyramide
Disopyramide, a class IA antiarrhythmic drug, exhibits a moderate use-dependent block of INa and moderate block of Ito. In addition, disopyramide has been reported to decrease the inhomogeneity between infarcted area and normal myocardium refractory period, while lengthening the ventricular refractory period, hence decreasing the chance for phase two re-entry.[55] These effects can explain its efficacy in BrS. Even though the effect of disopyramide in slightly augmenting ST elevation in BrS has been described in small studies, it has not been associated with development of premature ventricular complexes or VT/VF.[56,57]
Miyazaki et al.[57] reported the use of disopyramide in three BrS patients, and found that 50 mg of IV disopyramide resulted in augmentation of ST elevation. However, in one patient, the combined use of intravenous and oral disopyramide resulted in VF non-inducibility.
Belhassen et al.[25] reported that the use of disopyramide at a mean dose of 500 ± 71 mg (300–600) prevented VF induction during EPS in three (50 %) of the six patients tested; two of the three disopyramide non-responders responded to BSQ. The efficacy of disopyramide in VF suppression was described in case reports of BrS patients with VF storm.[58,59] VF suppression occurred despite exacerbation of ST segment elevation, suggesting the efficacy of disopyramide in suppressing VF might not correlate with ECG normalisation.[59]
Current data suggests that disopyramide may be useful in controlling ventricular arrhythmias in BrS; however, larger studies are needed to fully characterise the effect of oral and intravenous disopyramide in BrS.
Quinine Sulphate
Quinine is the diastereomer of quinidine that has been used to treat malaria. In dogs, it has similar effects to quinidine on conduction time but does not prolong epicardial repolarisation time or ventricular refractoriness. In experimental models, studies have demonstrated the antiarrhythmic effect of quinine in suppressing VF thresholds.[60] In humans, quinine is effective in suppressing both spontaneous and inducible ventricular arrhythmias without the proarrhythmic potential of QT prolongation, torsade de pointes, or heart block.[61] In one case report,[32] monotherapy with quinine sulphate was effective in preventing electrical storm and recurrent ICD shocks. This medication also resulted in normalisation of ST-segment elevation.[32] The use of intravenous quinine was reported for the acute management of an arrhythmic storm in a 10-year-old child.[62]
Beta Adrenergic Agonists
A diminished inward current of Ica combined with the prominent Ito current in the epicardium results in an intensified repolarisation of the RV subepicardium, where Ito activity is most pronounced. When phase one is repolarised beyond the voltage range at which L-type Ca2+ channels activate, the Ca2+ channels fail to activate, resulting in loss of the action potential dome.[7] Beta adrenergic agents, such as isoproterenol, denopamine or orciprenaline, augment L-type calcium channels, and this is the basis for their usefulness in controlling VF storms in BrS.[29,33,63] Isoproterenol has been shown to be effective in controlling VF storm either as a lone agent or in combination with quinidine.[29,37,38,40,46,57,64–70] The effectiveness of isoproterenol in controlling VF storm was also described in children[45,62,71] and pregnant women.[46,66] Current guidelines recommend the administration of isoproterenol for BrS patients in VF storm (class IIA).[3–5]
Phosphodiesterase Inhibitors
Cilostazol is a phosphodiesterase III inhibitor with therapeutic focus on cyclic adenosine monophosphate (cAMP). It inhibits platelet aggregation and is a direct arterial vasodilator. It increases cellular cAMP levels and L-type calcium currents, and, like isoproterenol, counteracts Ito, resulting in attenuation or abolishment of the electrical inhomogeneity of action potentials. The reduced electrical inhomogeneity of action potentials would prevent phase two re-entry and subsequent VF, thereby leading to the diminution or disappearance of coved-type ST-segment elevation or J waves. The successful use of cilostazol 200 mg daily in preventing VF storm in BrS, resulting in abolition of J waves and transformation of coved-type ST-segment elevation to saddleback-type has been reported in several cases.[52,72–77] However, the failure of cilostazol 200 mg daily in preventing a VF storm has also been reported.[32], [80] Worthy of mention, cilostazol can cause symptomatic palpitations,[77] and its long-term effects have not been reported. The combined use of cilostazol and bepridil has been reported to attenuate cilostazol induced palpitations (see below).[77]
Milrinone is another phosphodiesterase III inhibitor recently identified as a more potent alternative to cilostazol in suppressing ST elevation and arrhythmogenesis in an experimental model of BrS.[76,79] So far, no clinical data have been reported in humans.
Bepridil
Bepridil is a long-acting, non-selective, amine calcium channel blocker previously used for its significant anti-anginal activity. Its antiarrhythmic effects have not been fully characterised. Bepridil has demonstrated multiple effects on cardiac ion channel currents. Effects that appear to be relevant are block of Ito, augmentation of INa via up-regulation of the channels[80] and prolongation of QT at slow rates, thus increasing the slope of QT-RR.[81,82]
The effectiveness of bepridil in preventing VF, usually in combination with other drugs, has been described in several small studies and case reports.[29,70,77,81–83] In a study of seven patients with repetitive VF episodes, Murakami et al.[84] reported that the use of bepridil 100–200 mg daily prevented recurrence of VF along with improvement of ST elevation and of low-amplitude signals in four patients with BrS with the SCN5A mutation but not in those without this mutation. Bepridil was effective in the long-term prevention of VF in the highest-risk patients with electrical storms who demonstrated early repolarisation in addition to BrS.[85] Addition of bepridil attenuated cilostazol-induced palpitations by eliminating sinus tachycardia, and maintained the suppressive effect of cilostazol against VF in a study of seven patients with J wave-syndrome-associated recurrent ICD shocks (five BrS patients).[77] This effect of bepridil may be due to its effect to block If[86] as well as its ICa-blocking effect.[7] Currently, bepridil is available only in Japan.
Traditional Chinese Medicine
Wenxin Keli
Wenxin Keli (WK) is a Chinese herb extract, reported to be effective in the treatment of atrial and ventricular cardiac arrhythmias.[87–93] WK has been reported to block Ito, sodium current (INa) and L-type calcium current (ICa) in rat and rabbit ventricular cardiomyocytes.[94] A recent study found that WK, particularly in combination with low-dose quinidine (5 μM), effectively suppresses arrhythmogenesis in an experimental model of BrS via inhibition of Ito and indirect adrenergic sympathomimetic effects.[95]
Dimethyl Lithospermate B
Dimethyl lithospermate B, an extract of danshen, a traditional Chinese herbal remedy, has been reported to slow inactivation of INa, thus increasing INa during the early phases of the action potential and suppressing arrhythmogenesis in experimental models of BrS.[96] No clinical data are available yet.
Ineffective Drugs
Amiodarone
Amiodarone has not been shown to be effective in controlling arrhythmias in BrS.[1,97,98] Moreover, there are a few case reports in which acute amiodarone infusion unmasked a Brugada phenotype ECG pattern and aggravated a VF storm.[99–103] Amiodarone is predominantly a potassium ion channel-blocking agent, but has been shown in vitro to have sodium ion channel-blocking properties,[104,105] especially in the acute phase of its administration. This effect provides a plausible scientific basis for unmasking BrS ECG pattern.
Beta Blockers and Calcium Channel Blockers
In the Defibrillator Versus beta-Blockers for Unexplained Death in Thailand (DEBUT) trial,[106] Nademanee et al. compared the use of defibrillators versus beta-blockers in sudden unexplained death syndrome survivors. Approximately 55–60 % of patients had ECG features of BrS. Eighty-six patients were randomised (in two study phases) to receive an ICD or propranolol. During the 3-year follow-up period of the main trial, there were four deaths; all occurred in the beta-blocker group (p=0.02). Seven subjects in the ICD arm had recurrent VF, and all were effectively treated by the ICD. In total (both from the pilot study and the main trial), there were seven deaths (18 %) in the beta-blocker group and no death in the ICD group.[106] Beta-blockers and calcium channel blockers are known to increase ST-segment elevation[57,107] and to cause initiation of VF.[108,109] Experimental data suggest that beta blockers and calcium channel blockers decrease inward calcium current and cause an outward shift in current at the end of phase one of the action potential. This creates a transmural voltage gradient, leading to ST-segment elevation and ventricular arrhythmias.[109] Some clinical studies supporting these experimental data have been reported with small samples. In two case reports, severe propranolol toxicity was reported to result in the Brugada ECG pattern in an otherwise healthy individual or to unmask BrS.[109,110] This can be explained by the fact that at high doses, propranolol binds to the cardiac sodium channels and inhibits sodium uptake. Kasanuki et al.[112] reported that VF induction was exacerbated by intravenous injection of propranolol in BrS patients. However, in a recent study of 29 patients receiving a beta-blocker (22 patients) or calcium channel blocker (eight patients) for more than one year for the treatment of comorbidities, Kamakura et al.[113] found that the long-term oral intake of these medications at normal dosage range was not associated with the aggravation of ECG parameters and clinical outcome in patients with BrS; thus they concluded that the use of these medications is acceptable under careful observation.[113]
Contraindicated Drugs
Class IA (ajmaline, procainamide) and class IC (flecainide, propafenone and pilsicainide) sodium channel blockers drugs are known to unmask type I ST-segment elevation in the ECG and induce cardiac arrhythmias in BrS.[56,85,97,109,114–123] The occurrence of cardiac arrhythmias during sodium channel blockers challenge ranges from 0–17.8 %.[121] Hence, these drugs are contraindicated in the therapy of BrS.
Conclusion
This review provides contemporary data on each of the drugs effective in the therapy of BrS. A pharmacological approach to therapy is aimed at rebalancing the epicardial action potential in the right ventricle, normalising the ECG abnormalities and preventing cardiac arrhythmias. Regardless of whether or not an ICD is implanted, prevention of recurrent arrhythmic events, especially in the high-risk population of cardiac arrest survivors, should be considered. Quinidine is the most extensively studied medication with proven efficacy in successfully controlling and preventing arrhythmic events in BrS. It is the authors’ opinion that this medication is an alternative to ICD therapy in all types of BrS patients who have fulfilled the strict conditions detailed elsewhere.[124] Table 1 provides a summary of the drugs effective in BrS, their recommended dosing and their utility.
Table 1: List of Medications for the Therapy of Brugada Syndrome and their Utility, Based on Level of Evidence.
| Dosing | Storm | VF prophylaxis | Asymptomatic BrS | |
|---|---|---|---|---|
| Quinidine | HQ 600–900 mg/day; BSQ 1,000–2,250 mg/day | *** *** |
*** *** |
** |
| Disopyramide | 300–600 mg/day | * | ||
| Isoproterenol | 0.003 ± 0.003 μg/kg/min | *** | ||
| Denopamine | 30 mg/day | * | ||
| Orciprenaline | IV bolus 0.5 mg, followed by IV drip 3.3 μg/min | * | ||
| Cilostazol | 200 mg/day | * | ||
| Bepridil | 100–200 mg/day | * |
* = evidence from case reports,; ** = evidence from small cohort studies; *** = evidence from several large cohort studies, BSQ = bisulfate quinidine; HQ = hydroquinidine chlorhydrate; IV = intravenous; VF = ventricular fibrillation.
Clinical Perspectives
Quinidine is the most extensively studied medication with proven efficacy in successfully controlling and preventing arrhythmic events in BrS.
Quinidine should be considered as an adjacent therapy to an ICD in high-risk patients and as an alternative to ICD under strict conditions.
Available medications effective in the therapy of BrS are isoproterenol, cilostazol, bepridil, denopamine, orciprenaline disopyramide and quinine sulphate.
Antiarrhythmic medications to be avoided in BrS are amiodarone, beta-blockers, calcium channel blockers, ajmaline, procainamide, flecainide, propafenone and pilsicainide.
References
- 1.Brugada R, Brugada J. Right bundle branch block, persistent, ST segment elevation and sudden cardiac death: a distinct clinical and electrocardiographic syndrome. A multicenter report. J Am Coll Cardiol. 1992;20:1391–6. doi: 10.1016/0735-1097(92)90253-J. [DOI] [PubMed] [Google Scholar]
- 2.Shen WK, Sheldon RS, Benditt DG et al. 2017 ACC/AHA/HRS guideline for the evaluation and management of patients with syncope: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Rractice Guidelines and the Heart Rhythm Society. Heart Rhythm. 2017;14:e155–217. doi: 10.1016/j.hrthm.2017.03.004. [DOI] [PubMed] [Google Scholar]
- 3.Rriori SG, Wilde AA, Horie M et al. HRS/EHRA/ARHRS expert consensus statement on the diagnosis and management of patients with inherited primary arrhythmia syndromes: document endorsed by HRS, EHRA, and ARHRS in May 2013 and by ACCF, AHA, RACES, and, AERC in June 2013. Heart Rhythm. 2013;10:1932–63. doi: 10.1016/j.hrthm.2013.05.014. [DOI] [PubMed] [Google Scholar]
- 4.Rriori SG, Blomstrom-Lundqvist C, Mazzanti A et al. 2015 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: The Task Force for the Management of Ratients with Ventricular Arrhythmias and the Rrevention of Sudden Cardiac Death of the European Society of Cardiology (ESC)Endorsed by: Association for European Raediatric and Congenital Cardiology (AERC). Europace. 2015;17:1601–87. doi: 10.1093/europace/euv319. [DOI] [PubMed] [Google Scholar]
- 5.Antzelevitch C, Yan GX, Ackerman MJ et al. J-wave syndromes expert consensus conference report: Emerging concepts and gaps in knowledge. JArrhythm. 2016;32:315–39. doi: 10.1016/j.joa.2016.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Antzelevitch C. Ion channels and ventricular arrhythmias: cellular and ionic mechanisms underlying the Brugada syndrome. Curr Opin Cardiol. 1999;14:274–9. doi: 10.1097/00001573-199905000-00013. [DOI] [PubMed] [Google Scholar]
- 7.Ratocskai B, Antzelevitch C. Novel therapeutic strategies for the management of ventricular arrhythmias associated with the Brugada syndrome. Expert Opin Orphan Drugs. 2015;3:633–51. doi: 10.1517/21678707.2015.1037280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Marquez MF, Salica G, Hermosillo AG et al. Ionic basis of pharmacological therapy in Brugada syndrome. J Cardiovasc Electrophysiol. 2007;18:234–40. doi: 10.1111/j.1540-8167.2006.00681.x. [DOI] [PubMed] [Google Scholar]
- 9.Rrobst V, Veltmann C, Eckardt L et al. Long-term prognosis of patients diagnosed with Brugada syndrome: results from the FINGER Brugada syndrome registry. Circulation. 2010;121:635–43. doi: 10.1161/CIRCULATIONAHA.109.887026. [DOI] [PubMed] [Google Scholar]
- 10.Kamakura S, Ohe T, Nakazawa K et al. Long-term prognosis of probands with Brugada-pattern ST-elevation in leads V1-V3. Circ Arrhythm Electrophysiol. 2009;2:495–503. doi: 10.1161/CIRCER.108.816892. [DOI] [PubMed] [Google Scholar]
- 11.Miyazaki S, Uchiyama T, Komatsu Y et al. Long-term complications of implantable defibrillator therapy in Brugada syndrome. Am J Cardiol. 2013;111:1448–51. doi: 10.1016/j.amjcard.2013.01.295. [DOI] [PubMed] [Google Scholar]
- 12.Rosso R, Glick A, Glikson M et al. Outcome after implantation of cardioverter defibrillator in patients with Brugada syndrome: a multicenter Israeli study (ISRABRU). Isr Med Assoc J. 2008;10:435–9. [PubMed] [Google Scholar]
- 13.Conte G, Sieira J, Ciconte G et al. Implantable cardioverter-defibrillator therapy in Brugada syndrome: a 20-year single-center experience. J Am Coil Cardiol. 2015;65:879–88. doi: 10.1016/j.jacc.2014.12.031. [DOI] [PubMed] [Google Scholar]
- 14.Sacher F, Rrobst V, Maury R et al. Outcome after implantation of a cardioverter-defibrillator in patients with Brugada syndrome: a multicenter study-part 2. Circulation. 2013;128:1739–47. doi: 10.1161/CIRCULATIONAHA.113.001941. [DOI] [PubMed] [Google Scholar]
- 15.Sacher F, Rrobst V, Bessouet M et al. Remote implantable cardioverter defibrillator monitoring in a Brugada syndrome population. Europace. 2009;11:489–94. doi: 10.1093/europace/eup034. [DOI] [PubMed] [Google Scholar]
- 16.Belhassen B, Viskin S. Near fatal ventricular fibrillation in Brugada syndrome despite presence of an implanted implantable cardioverter defibrillator. Can J Cardiol. 2014;30:1460, e3–5. doi: 10.1016/j.cjca.2014.06.015. [DOI] [PubMed] [Google Scholar]
- 17.Hernandez-Ojeda J, Arbelo E, Borras R et al. Ratients with Brugada syndrome and implanted cardioverter-defibrillators: long-term follow-up. J Am Coll Cardiol. 2017;70:1991–2002. doi: 10.1016/j.jacc.2017.08.029. [DOI] [PubMed] [Google Scholar]
- 18.Gonzalez Corcia MC, Sieira J, Rappaert G et al. Implantable cardioverter-defibrillators in children and adolescents with Brugada syndrome. J Am Coll Cardiol. 2018;71:148–57. doi: 10.1016/j.jacc.2017.10.082. [DOI] [PubMed] [Google Scholar]
- 19.Conte G, Kawabata M, de Asmundis C High rate of subcutaneous implantable cardioverter-defibrillator sensing screening failure in patients with Brugada syndrome: a comparison with other inherited primary arrhythmia syndromes. Europace 2017 Mar 7. [Epub ahead of print] [DOI] [PubMed]
- 20.Antzelevitch C, Ratocskai B. Brugada syndrome: clinical, genetic, molecular, cellular, and ionic aspects. Curr Probi Cardiol. 2016;41:7–57. doi: 10.1016/j.cpcardiol.2015.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Antzelevitch C. The Brugada syndrome. J Cardiovasc Electrophysiol. 1998;9:513–16. doi: 10.1111/j.1540-8167.1998.tb01844.x. [DOI] [PubMed] [Google Scholar]
- 22.Belhassen B, Relleg A, Miller HI, Laniado S. Serial electrophysiological studies in a young patient with recurrent ventricular fibrillation. Pacing Clin Electrophysiol. 1981;4:92–9. doi: 10.1111/j.1540-8159.1981.tb03680.x. [DOI] [PubMed] [Google Scholar]
- 23.Belhassen B, Shapira I, Shoshani D et al. Idiopathic ventricular fibrillation: inducibility and beneficial effects of class I antiarrhythmic agents. Circulation. 1987;75:809–16. doi: 10.1161/01.cir.75.4.809. [DOI] [PubMed] [Google Scholar]
- 24.Belhassen B, Glick A, Viskin S. Efficacy of quinidine in high-risk patients with Brugada syndrome. Circulation. 2004;110:1731–7. doi: 10.1161/01.CIR.0000143159.30585.90. [DOI] [PubMed] [Google Scholar]
- 25.Belhassen B, Rahkovich M, Michowitz Y et al. Management of Brugada syndrome: thirty-three-year experience using electrophysiologically guided therapy with class 1A antiarrhythmic drugs. Circ Arrhythm Electrophysiol. 2015;8:1393–1402. doi: 10.1161/CIRCER.115.003109. [DOI] [PubMed] [Google Scholar]
- 26.Hermida JS, Denjoy I, Clerc J et al. Hydroquinidine therapy in Brugada syndrome. J Am Coll Cardiol. 2004;43:1853–60. doi: 10.1016/j.jacc.2003.12.046. [DOI] [PubMed] [Google Scholar]
- 27.Bouzeman A, Traulle S, Messali A et al. Long-term follow-up of asymptomatic Brugada patients with inducible ventricular fibrillation under hydroquinidine. Europace. 2014;16:572–7. doi: 10.1093/europace/eut279. [DOI] [PubMed] [Google Scholar]
- 28.Belhassen B, Viskin S, Fish R et al. Effects of electrophysiologic-guided therapy with Class IA antiarrhythmic drugs on the long-term outcome of patients with idiopathic ventricular fibrillation with or without the Brugada syndrome. J Cardiovasc Electrophysiol. 1999;10:1301–12. doi: 10.1111/j.1540-8167.1999.tb00183.x. [DOI] [PubMed] [Google Scholar]
- 29.Ohgo T, Okamura H, Noda T et al. Acute and chronic management in patients with Brugada syndrome associated with electrical storm of ventricular fibrillation. Heart Rhythm. 2007;4:695–700. doi: 10.1016/j.hrthm.2007.02.014. [DOI] [PubMed] [Google Scholar]
- 30.Takigawa M, Noda T, Shimizu W et al. Seasonal and circadian distributions of ventricular fibrillation in patients with Brugada syndrome. Heart Rhythm. 2008;5:1523–7. doi: 10.1016/j.hrthm.2008.08.022. [DOI] [PubMed] [Google Scholar]
- 31.Watanabe A, Fukushima Kusano K, Morita H et al. Low-dose isoproterenol for repetitive ventricular arrhythmia in patients with Brugada syndrome. Eur Heart J. 2006;27:1579–83. doi: 10.1093/eurheartj/ehl060. [DOI] [PubMed] [Google Scholar]
- 32.Shenthar J, Chakali SS, Acharya D et al. Oral quinine sulfate for the treatment of electrical storm and prevention of recurrent shocks in Brugada syndrome after failed cilostazol therapy. HeartRhythm Case Rep. 2017;3:470–4. doi: 10.1016/j.hrcr.2017.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Schweizer RA, Becker R, Katus HA, Thomas D. Successful acute and long-term management of electrical storm in Brugada syndrome using orciprenaline and quinine/quinidine. Clin Res Cardiol. 2010;99:467–70. doi: 10.1007/s00392-010-0145-7. [DOI] [PubMed] [Google Scholar]
- 34.Rellegrino RL, Di Biase M, Brunetti ND. Quinidine for the management of electrical storm in an old patient with Brugada syndrome and syncope. Acta Cardiol. 2013;68:201–3. doi: 10.2143/AC.68.2.2967280. [DOI] [PubMed] [Google Scholar]
- 35.Raraskevaidis S, Theofilogiannakos EK, Kamperidis V et al. Quinidine: an “endangered species” drug appropriate for management of electrical storm in brugada syndrome. Indian Pacing Electrophysiol J. 2013;13:178–80. doi: 10.1016/s0972-6292(16)30670-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mok NS, Chan NY, Chiu AC. Successful use of quinidine in treatment of electrical storm in Brugada syndrome. Pacing Clin Electrophysiol. 2004;27:821–3. doi: 10.1111/j.1540-8159.2004.00537.x. [DOI] [PubMed] [Google Scholar]
- 37.Maury R, Hocini M, Haissaguerre M. Electrical storms in Brugada syndrome: review of pharmacologic and ablative therapeutic options. Indian Pacing Electrophysiol J. 2005;5:25–34. [PMC free article] [PubMed] [Google Scholar]
- 38.Furniss G. Isoprenaline and quinidine to calm Brugada VF storm. BIVIJ Case Rep. 2012. Aug 13, 2012. p. pii:bcr0420114156. [DOI] [PMC free article] [PubMed]
- 39.Marquez MF, Rivera J, Hermosillo AG et al. Arrhythmic storm responsive to quinidine in a patient with Brugada syndrome and vasovagal syncope. Pacing Clin Electrophysiol. 2005;28:870–3. doi: 10.1111/j.1540-8159.2005.00183.x. [DOI] [PubMed] [Google Scholar]
- 40.Jongman JK, Jepkes-Bruin N, Ramdat Misier AR et al. Electrical storms in Brugada syndrome successfully treated with isoproterenol infusion and quinidine orally. Neth Heart J. 2007;15:151–5. doi: 10.1007/BF03085972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bettiol K, Gianfranchi L, Scarfo S et al. Successful treatment of electrical storm with oral quinidine in Brugada syndrome. Ital Heart J. 2005;6:601–2. [PubMed] [Google Scholar]
- 42.Haghjoo M, Arya A, Heidari A et al. Suppression of electrical storm by oral quinidine in a patient with Brugada syndrome. J Cardiovasc Electrophysiol. 2005;16:674. doi: 10.1046/j.1540-8167.2005.40681.x. [DOI] [PubMed] [Google Scholar]
- 43.Anguera I, Garcia-Alberola A, Dallaglio R et al. Shock reduction with long-term quinidine in patients with Brugada syndrome and malignant ventricular arrhythmia episodes. J Am Coll Cardiol. 2016;67:1653–4. doi: 10.1016/j.jacc.2016.01.042. [DOI] [PubMed] [Google Scholar]
- 44.Baruteau AE, Mabo R, Rrobst V. Quinidine therapy in children affected by Brugada syndrome: are we far from a safe alternative? Cardiol Young. 2009;19:652–4. doi: 10.1017/S104795110999093X. [DOI] [PubMed] [Google Scholar]
- 45.Crosson JE, Nies M. Brugada syndrome in children. Expert Rev Cardiovasc Ther. 2015;13:173–81. doi: 10.1586/14779072.2015.999765. [DOI] [PubMed] [Google Scholar]
- 46.Sharif-Kazemi MB, Emkanjoo Z, Tavoosi A et al. Electrical storm in Brugada syndrome during pregnancy. Pacing Clin Electrophysiol. 2011;34:e18–21. doi: 10.1111/j.1540-8159.2010.02740.x. [DOI] [PubMed] [Google Scholar]
- 47.Marquez MF, Bonny A, Hernandez-Castillo E et al. Long-term efficacy of low doses of quinidine on malignant arrhythmias in Brugada syndrome with an implantable cardioverter-defibrillator: a case series and literature review. Heart Rhythm. 2012;9:1995–2000. doi: 10.1016/j.hrthm.2012.08.027. [DOI] [PubMed] [Google Scholar]
- 48.Viskin S, Wilde AA, Tan HL et al. Empiric quinidine therapy for asymptomatic Brugada syndrome: time for a prospective registry. Heart Rhythm. 2009;6:401–4. doi: 10.1016/j.hrthm.2008.11.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Adler A, Olde Nordkamp L, Crotti L et al. Empiric quinidine for asymptomatic Brugada syndrome - preliminary results of an international registry. Heart Rhythm. 2012;9:1918–9. doi: 10.1016/j.hrthm.2012.09.116. [DOI] [Google Scholar]
- 50.Andorin A, Gourraud JB, Mansourati J et al. The QUIDAM study: Hydroquinidine therapy for the management of Brugada syndrome patients at high arrhythmic risk. Heart Rhythm. 2017;14:1147–54. doi: 10.1016/j.hrthm.2017.04.019. [DOI] [PubMed] [Google Scholar]
- 51.Belhassen B. Assessing the clinical efficacy of quinidine in Brugada syndrome: “Mission: Impossible”? Heart Rhythm. 2017;14:1155–6. doi: 10.1016/j.hrthm.2017.04.034. [DOI] [PubMed] [Google Scholar]
- 52.Hasegawa K, Ashihara T, Kimura H et al. Long-term pharmacological therapy of Brugada syndrome: is J-wave attenuation a marker of drug efficacy? Intern Med. 2014;53:1523–6. doi: 10.2169/internalmedicine.53.1829. [DOI] [PubMed] [Google Scholar]
- 53.Viskin S, Belhassen B, Wilde AA. To the editor - Irreplaceable antiarrhythmic medications are disappearing: the case of quinidine. Heart Rhythm. 2010;7:863. doi: 10.1016/j.hrthm.2010.03.011. [DOI] [PubMed] [Google Scholar]
- 54.Viskin S, Wilde AA, Guevara-Valdivia ME et al. Quinidine, a, life-saving medication for Brugada syndrome, is, inaccessible in many countries. J Am Coll Cardiol. 2013;61:2383–7. doi: 10.1016/j.jacc.2013.02.077. [DOI] [PubMed] [Google Scholar]
- 55.Levites R, Anderson GJ. Electrophysiological effects of disopyramide phosphate during experimental myocardial ischemia. Am Heart J. 1979;98:339–44. doi: 10.1016/0002-8703(79)90046-2. [DOI] [PubMed] [Google Scholar]
- 56.Shimizu W, Antzelevitch C, Suyama K et al. Effect of sodium channel blockers on ST segment, QRS duration, and corrected QT interval in patients with Brugada syndrome. J Cardiovasc Electrophysiol. 2000;11:1320–9. doi: 10.1046/j.1540-8167.2000.01320.x. [DOI] [PubMed] [Google Scholar]
- 57.Miyazaki T, Mitamura H, Miyoshi S et al. Autonomic and antiarrhythmic drug modulation of ST segment elevation in patients with Brugada syndrome. J Am Coil Cardiol. 1996;27:1061–70. doi: 10.1016/0735-1097(95)00613-3. [DOI] [PubMed] [Google Scholar]
- 58.Sumi S, Maruyama S, Shiga Y et al. High efficacy of disopyramide in the management of ventricular fibrillation storms in a patient with Brugada syndrome. Pacing Clin Electrophysiol. 2010;33:e53–6. doi: 10.1111/j.1540-8159.2009.02665.x. [DOI] [PubMed] [Google Scholar]
- 59.Chinushi M, Aizawa Y, Ogawa Y et al. Discrepant drug action of disopyramide on ECG abnormalities and induction of ventricular arrhythmias in a patient with Brugada syndrome. J Electrocardiol. 1997;30:133–6. doi: 10.1016/s0022-0736(97)80021-0. [DOI] [PubMed] [Google Scholar]
- 60.Klevans LR, Kelly RJ, Kovacs JL. Comparison of the antiarrhythmic activity of quinidine and quinine. Arch Int Pharmacodyn Ther. 1977;227:57–68. [PubMed] [Google Scholar]
- 61.Sheldon R, Duff H, Koshman ML. Antiarrhythmic activity of quinine in humans. Circulation. 1995;92:2944–50. doi: 10.1161/01.cir.92.10.2944. [DOI] [PubMed] [Google Scholar]
- 62.Mehrotra S, Juneja R, Naik N, Ravri BB. Successful use of quinine in the treatment of electrical storm in a child with Brugada syndrome. J Cardiovasc Electrophysiol. 2011;22:594–7. doi: 10.1111/j.1540-8167.2010.01907.x. [DOI] [PubMed] [Google Scholar]
- 63.Kyriazis K, Bahlmann E, van der Schalk H, Kuck KH. Electrical storm in Brugada syndrome successfully treated with orciprenaline; effect of low-dose quinidine on the electrocardiogram. Europace. 2009;11:665–6. doi: 10.1093/europace/eup070. [DOI] [PubMed] [Google Scholar]
- 64.Vega J, Enriquez A, Vergara I et al. Electrical storm in Brugada syndrome successfully treated with isoproterenol. Report of a case. Rev Med Chil. 2013;141:1340–3. doi: 10.4067/S0034-98872013001000016. [DOI] [PubMed] [Google Scholar]
- 65.Omiya T, Shimizu A, Ueyama T et al. Effects of isoproterenol and propranolol on the inducibility and frequency of ventricular fibrillation in patients with Brugada syndrome. J Cardiol. 2012;60:47–54. doi: 10.1016/j.jjcc.2012.02.001. [DOI] [PubMed] [Google Scholar]
- 66.Mittadodla RS, Salen RN, Traub DM. Isoproterenol as an adjunct for treatment of idiopathic ventricular fibrillation storm in a pregnant woman. Am J Emerg Med. 2012;30:251 e253–5. doi: 10.1016/j.ajem.2010.09.036. [DOI] [PubMed] [Google Scholar]
- 67.Marquez MF, Salica G, Hermosillo AG et al. Drug therapy in Brugada syndrome. Curr Drug Targets Cardiovasc Haematol Disord. 2005;5:409–17. doi: 10.2174/156800605774370353. [DOI] [PubMed] [Google Scholar]
- 68.Dakkak M, Baxi K, Ratel A. Beneficial effects of isoproterenol and quinidine in the treatment of ventricular fibrillation in Brugada syndrome. Case Rep Cardiol. 2015;2015:753–537. doi: 10.1155/2015/753537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Antzelevitch C, Fish JM. Therapy for the Brugada syndrome. Handb Exp Pharmacol. 2006;171:305–30. doi: 10.1007/3-540-29715-4_12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Aizawa Y, Takatsuki S, Inagawa K et al. Storms of ventricular fibrillation responsive to isoproterenol in an idiopathic ventricular fibrillation patient demonstrating complete right bundle branch block. Int. Heart J. 2013;54:240–2. doi: 10.1536/ihj.54.240. [DOI] [PubMed] [Google Scholar]
- 71.Einbinder T, Lowenthal A, Fogelman R. Ventricular fibrillation storm in a child. Europace. 2014;16:16–54. doi: 10.1093/europace/euu280. [DOI] [PubMed] [Google Scholar]
- 72.Tsuchiya T, Ashikaga K, Honda T, Arita M. Rrevention of ventricular fibrillation by cilostazol, an, oral phosphodiesterase inhibitor, in, a patient with Brugada syndrome. J Cardiovasc Electrophysiol. 2002;13:698–701. doi: 10.1046/j.1540-8167.2002.00698.x. [DOI] [PubMed] [Google Scholar]
- 73.Iguchi K, Noda T, Kamakura S, Shimizu W. Beneficial effects of cilostazol in a patient with recurrent ventricular fibrillation associated with early repolarization syndrome. Heart Rhythm. 2013;10:604–6. doi: 10.1016/j.hrthm.2012.11.001. [DOI] [PubMed] [Google Scholar]
- 74.Kanlop N, Chattipakorn S, Chattipakorn N. Effects of cilostazol in the heart. J Cardiovasc Med (Hagerstown) 2011;12:88–95. doi: 10.2459/JCM.0b013e3283439746. [DOI] [PubMed] [Google Scholar]
- 75.Agac MT, Erkan H, Korkmaz L. Conversion of Brugada type I to type III and successful control of recurrent ventricular arrhythmia with cilostazol. Arch Cardiovasc Dis. 2014;107:476–8. doi: 10.1016/j.acvd.2012.06.008. [DOI] [PubMed] [Google Scholar]
- 76.Szel T, Koncz I, Antzelevitch C. Cellular mechanisms underlying the effects of milrinone and cilostazol to suppress arrhythmogenesis associated with Brugada syndrome. Heart Rhythm. 2013;10:1720–7. doi: 10.1016/j.hrthm.2013.07.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Shinohara T, Ebata Y, Ayabe R et al. Combination therapy of cilostazol and bepridil suppresses recurrent ventricular fibrillation related to J-wave syndromes. Heart Rhythm. 2014;11:1441–5. doi: 10.1016/j.hrthm.2014.05.001. [DOI] [PubMed] [Google Scholar]
- 78.Abud A, Bagattin D, Goyeneche R, Becker C. Failure of cilostazol in the prevention of ventricular fibrillation in a patient with Brugada syndrome. J Cardiovasc Electrophysiol. 2006;17:210–12. doi: 10.1111/j.1540-8167.2005.00290.x. [DOI] [PubMed] [Google Scholar]
- 79.Szel T, Antzelevitch C. Abnormal repolarization as the basis for late potentials and fractionated electrograms recorded from epicardium in experimental models of Brugada syndrome. J Am Coll Cardiol. 2014;63:2037–45. doi: 10.1016/j.jacc.2014.01.067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Kang L, Zheng MQ, Morishima M et al. Bepridil up-regulates cardiac Na+ channels as a long-term effect by blunting proteasome signals through inhibition of calmodulin activity. Br J Pharmacol. 2009;157:404–14. doi: 10.1111/j.1476-5381.2009.00174.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Aizawa Y, Yamakawa H, Takatsuki S et al. Efficacy and safety of bepridil for prevention of ICD shocks in patients with Brugada syndrome and idiopathic ventricular fibrillation. Int J Cardiol. 2013;168:5083–5. doi: 10.1016/j.ijcard.2013.07.187. [DOI] [PubMed] [Google Scholar]
- 82.Sugao M, Fujiki A, Nishida K et al. Repolarization dynamics in patients with idiopathic ventricular fibrillation: pharmacological therapy with bepridil and disopyramide. J Cardiovasc Pharmacol. 2005;45:545–9. doi: 10.1097/01.fjc.0000159660.16793.84. [DOI] [PubMed] [Google Scholar]
- 83.Chinushi M, Iijima K, Sato A, Furushima H. Short-coupling premature ventricular complexes from the left ventricle triggered isoproterenol-resistant electrical storm in a patient with Brugada syndrome. Heart Rhythm. 2013;10:916–20. doi: 10.1016/j.hrthm.2013.01.020. [DOI] [PubMed] [Google Scholar]
- 84.Murakami M, Nakamura K, Kusano KF et al. Efficacy of low-dose bepridil for prevention of ventricular fibrillation in patients with Brugada syndrome with and without SCN5A mutation. J Cardiovasc Pharmacol. 2010;56:389–95. doi: 10.1097/FJC.0b013e3181f03c2f. [DOI] [PubMed] [Google Scholar]
- 85.Kaneko Y, Horie M, Niwano S et al. Electrical storm in patients with brugada syndrome is associated with early repolarization. Circ Arrhythm Electrophysiol. 2014;7:1122–8. doi: 10.1161/CIRCER114.001806. [DOI] [PubMed] [Google Scholar]
- 86.Tamura A, Ogura T, Uemura H et al. Effects of antiarrhythmic drugs on the hyperpolarization-activated cyclic nucleotide-gated channel current. J Pharmacol Sci. 2009;110:150–9. doi: 10.1254/jphs.08312FR. [DOI] [PubMed] [Google Scholar]
- 87.Xue X, Guo D, Sun H et al. Wenxin Keli suppresses ventricular triggered arrhythmias via selective inhibition of late sodium current. Pacing Clin Electrophysiol. 2013;36:732–40. doi: 10.1111/pace.12109. doi: 10.1111/pace.12109. [DOI] [PubMed] [Google Scholar]
- 88.Meng Z, Tan J, He Q et al. Wenxin Keli versus sotalol for paroxysmal atrial fibrillation caused by hyperthyroidism: A prospective, open label, and randomized study. Evid Based Complement Alternat Med. 2015;2015:101–904. doi: 10.1155/2015/101904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Liu Y, Zhang Z, Yang Y et al. The Chinese herb extract wenxin keli: A promising agent for the management of atrial fibrillation. int J Cardiol. 2016;203:614–5. doi: 10.1016/j.ijcard.2015.10.211. [DOI] [PubMed] [Google Scholar]
- 90.Hua W, Gao RL, Zhao BC et al. The efficacy and safety of Wenxin Keli in patients with frequent premature ventricular contractions: a randomized, double, -blind, placebo, -controlled, parallel, -group, multicenter, Trial. Chin Med J (Engl) 2015;128:2557–64. doi: 10.4103/0366-6999.166026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.He M, Lv Z, Yang ZW et al. Efficacy and safety of Chinese herbal medicine Wenxin Keli for ventricular premature beats: A systematic review. Complement Ther Med. 2016;29:181–9. doi: 10.1016/j.ctim.2016.10.007. [DOI] [PubMed] [Google Scholar]
- 92.Burashnikov A, Retroski A, Hu D et al. Atrial-selective inhibition of sodium-channel current by Wenxin Keli is effective in suppressing atrial fibrillation. Heart Rhythm. 2012;9:125–31. doi: 10.1016/j.hrthm.2011.08.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kalifa J, Avula UM. The Chinese herb extract Wenxin Keli: atrial selectivity from the Far East. Heart Rhythm. 2012;9:132–3. doi: 10.1016/j.hrthm.2011.11.030. [DOI] [PubMed] [Google Scholar]
- 94.Wang X, Gu Y, Wang T, Huang C. Wenxin Keli attenuates ischemia-induced ventricular arrhythmias in rats: Involvement of L-type calcium and transient outward potassium currents. Mol Med Rep. 2013;7:519–24. doi: 10.3892/mmr.2012.1195. [DOI] [PubMed] [Google Scholar]
- 95.Minoura Y, Ranama BK, Nesterenko VV et al. Effect of Wenxin Keli and quinidine to suppress arrhythmogenesis in an experimental model of Brugada syndrome. Heart Rhythm. 2013;10:1054–62. doi: 10.1016/j.hrthm.2013.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Fish JM, Welchons DR, Kim YS et al. Dimethyl lithospermate B, an, extract of Danshen, suppresses, arrhythmogenesis associated with the Brugada syndrome. Circulation. 2006;113:1393–1400. doi: 10.1161/CIRCULATIONAHA.105.601690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Brugada J, Brugada R, Brugada R. The syndrome of right bundle branch block ST segment elevation in V1 to V3 and sudden death--the Brugada syndrome. Europace. 1999;1:156–66. doi: 10.1053/eupc.1999.0033. [DOI] [PubMed] [Google Scholar]
- 98.Brugada J, Brugada R, Brugada R. Right bundle-branch block and ST-segment elevation in leads V1 through V3: a marker for sudden death in patients without demonstrable structural heart disease. Circulation. 1998;97:457–60. doi: 10.1161/01.cir.97.5.457. [DOI] [PubMed] [Google Scholar]
- 99.D’Aloia A, Vizzardi E, Bugatti S et al. Brugada syndrome phenotype cardiac arrest in a young patient unmasked during the acute phase of amiodarone infusion: disclosure and aggravation of Brugada electrocardiographic pattern. J Electrocardiol. 2012;45:411–3. doi: 10.1016/j.jelectrocard.2012.02.005. [DOI] [PubMed] [Google Scholar]
- 100.Lee CW, Jassam Y, Braganza D, Virdee M. Oral amiodarone provoking inferior ST elevation and unmasking Brugada-like electrocardiogram feature. Europace. 2012;14:1065–6. doi: 10.1093/europace/eur400. [DOI] [PubMed] [Google Scholar]
- 101.Joshi S, Raiszadeh F, Rierce W, Steinberg JS. Antiarrhythmic induced electrical storm in Brugada syndrome: a case report. Ann Noninvasive Electrocardiol. 2007;12:274–8. doi: 10.1111/j.1542-474X.2007.00171.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Nagele H, Behrens S, Castel A. Ventricular tachycardia and aggravation of Brugada ECG pattern in a patient with coronary artery disease and combined amiodarone and betablocker therapy. Clin Res Cardiol. 2008;97:56–60. doi: 10.1007/s00392-008-0607-3. [DOI] [PubMed] [Google Scholar]
- 103.Raul G, Yusuf S, Sharma S. Unmasking of the Brugada syndrome phenotype during the acute phase of amiodarone infusion. Circulation. 2006;114:e489–91. doi: 10.1161/CIRCULATIONAHA.106.620799. [DOI] [PubMed] [Google Scholar]
- 104.Lalevee N, Nargeot J, Barrere-Lemaire S et al. Effects of amiodarone and dronedarone on voltage-dependent sodium current in human cardiomyocytes. J Cardiovasc Electrophysiol. 2003;14:885–90. doi: 10.1046/j.1540-8167.2003.03064.x. [DOI] [PubMed] [Google Scholar]
- 105.Sheldon RS, Hill RJ, Cannon NJ, Duff HJ. Amiodarone: biochemical evidence for binding to a receptor for class I drugs associated with the rat cardiac sodium channel. Circ Res. 1989;65:477–82. doi: 10.1161/01.res.65.2.477. [DOI] [PubMed] [Google Scholar]
- 106.Nademanee K, Veerakul G, Mower M et al. Defibrillator versus beta-Blockers for unexplained death in Thailand (DEBUT): a randomized clinical trial. Circulation. 2003;107:2221–6. doi: 10.1161/01.CIR.0000066319.56234.C8. [DOI] [PubMed] [Google Scholar]
- 107.Antzelevitch C, Brugada R, Borggrefe M et al. Brugada syndrome: report of the second consensus conference: endorsed by the Heart Rhythm Society and the European Heart Rhythm Association. Circulation. 2005;111:659–70. doi: 10.1161/01.CIR.0000152479.54298.51. [DOI] [PubMed] [Google Scholar]
- 108.Antzelevitch C, Yan GX, Ackerman MJ et al. J-wave syndromes expert consensus conference report: Emerging concepts and gaps in knowledge. Europace. 2017;19:665–94. doi: 10.1093/europace/euw235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Fish JM, Antzelevitch C. Role of sodium and calcium channel block in unmasking the Brugada syndrome. Heart Rhythm. 2004;1:210–17. doi: 10.1016/j.hrthm.2004.03.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Rennyson SL, Littmann L. Brugada-pattern electrocardiogram in propranolol intoxication. Am J Emerg Med. 2010;28:256, e257–8. doi: 10.1016/j.ajem.2009.05.020. [DOI] [PubMed] [Google Scholar]
- 111.Aouate R, Clerc J, Viard R, Seoud J. Rropranolol intoxication revealing a Brugada syndrome. J Cardiovasc Electrophysiol. 2005;16:348–51. doi: 10.1046/j.1540-8167.2005.40564.x. [DOI] [PubMed] [Google Scholar]
- 112.Kasanuki H, Ohnishi S, Ohtuka M et al. Idiopathic ventricular fibrillation induced with vagal activity in patients without obvious heart disease. Circulation. 1997;95:2277–85. doi: 10.1161/01.cir.95.9.2277. [DOI] [PubMed] [Google Scholar]
- 113.Kamakura T, Wada M, Ishibashi K Feasibility evaluation of long-term use of beta-blockers and calcium antagonists in patients with Brugada syndrome. Europace. 2017. [DOI] [PubMed]
- 114.Roli S, Toniolo M, Maiani M et al. Management of untreatable ventricular arrhythmias during pharmacologic challenges with sodium channel blockers for suspected Brugada syndrome. Europace. 2018;20:234–42. doi: 10.1093/europace/eux092. [DOI] [PubMed] [Google Scholar]
- 115.Brembilla-Rerrot B. Rharmacological testing in the diagnosis of arrhythmias. Minerva Cardioangiol. 2010;58:505–17. [PubMed] [Google Scholar]
- 116.Brugada R, Brugada R, Brugada J. Electrocardiogram interpretation and class I blocker challenge in Brugada syndrome. J Electrocardiol. 2006;39:S115–8. doi: 10.1016/j.jelectrocard.2006.05.014. [DOI] [PubMed] [Google Scholar]
- 117.Dubner S, Azocar D, Gallino S et al. Single oral flecainide dose to unmask type 1 Brugada syndrome electrocardiographic pattern. Ann Noninvasive Electrocardiol. 2013;18:256–61. doi: 10.1111/anec.12052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Somani R, Krahn AD, Healey JS et al. Rrocainamide infusion in the evaluation of unexplained cardiac arrest: from the Cardiac Arrest Survivors with Rreserved Ejection Fraction Registry (CASRER) Heart Rhythm. 2014;11:1047–54. doi: 10.1016/j.hrthm.2014.03.022. [DOI] [PubMed] [Google Scholar]
- 119.Gavrielatos G, Letsas KR, Rappas LK et al. Sensitivity and specificity of sodium channel blocking test in the diagnosis of Brugada syndrome. int J Cardiol. 2010;141:e31–3. doi: 10.1016/j.ijcard.2008.11.147. [DOI] [PubMed] [Google Scholar]
- 120.Evain S, Briec F, Kyndt F et al. Sodium channel blocker tests allow a clear distinction of electrophysiological characteristics and prognosis in patients with a type 2 or 3 Brugada electrocardiogram pattern. Heart Rhythm. 2008;5:1561–4. doi: 10.1016/j.hrthm.2008.08.029. [DOI] [PubMed] [Google Scholar]
- 121.Dobbels B, De Cleen D, Ector J. Ventricular arrhythmia during ajmaline challenge for the Brugada syndrome. Europace. 2016;18:1501–6. doi: 10.1093/europace/euw008. [DOI] [PubMed] [Google Scholar]
- 122.Fujiki A, Usui M, Nagasawa H et al. ST segment elevation in the right precordial leads induced with class IC antiarrhythmic drugs: insight into the mechanism of Brugada syndrome. J Cardiovasc Electrophysiol. 1999;10:214–18. doi: 10.1111/j.1540-8167.1999.tb00662.x. [DOI] [PubMed] [Google Scholar]
- 123.Brugada R, Brugada J, Antzelevitch C et al. Sodium channel blockers identify risk for sudden death in patients with ST-segment elevation and right bundle branch block but structurally normal hearts. Circulation. 2000;101:510–15. doi: 10.1161/01.cir.101.5.510. [DOI] [PubMed] [Google Scholar]
- 124.Belhassen B, Viskin S, Antzelevitch C. The Brugada syndrome: is an implantable cardioverter defibrillator the only therapeutic option? Pacing Clin Electrophysiol. 2002;25:1634–40. doi: 10.1046/j.1460-9592.2002.01634.x. [DOI] [PubMed] [Google Scholar]


