ABSTRACT
Carbapenem-resistant Klebsiella pneumoniae causes important health care-associated infections worldwide. An outbreak of sequence type 11 (ST11) OXA-48-producing K. pneumoniae (OXA-48-Kp) isolates occurred in Tzaneio Hospital in 2012 and was contained until 2014, when OXA-48-Kp reemerged. The present study involved 19 bloodstream infection (BSI) OXA-48-Kp isolates recovered from 19 intensive care unit (ICU) patients hospitalized between August 2014 and July 2016. MICs were determined by broth microdilution. Beta-lactamase genes were detected by PCR. All isolates were typed by pulsed-field gel electrophoresis/multilocus sequence typing (PFGE/MLST), and 10 representative isolates were typed by next-generation sequencing (NGS). Of the 19 study patients, 9 had previous hospitalizations, and 10 carried OXA-48-Kp prior to BSI isolation; median time from ICU admission to BSI was 29 days. Four OXA-48-Kp isolates belonged to PFGE profile A (ST147) and were pandrug resistant (PDR), while 15 isolates exhibited PFGE profile B (ST101) and were extensively drug resistant. Genes detected via NGS resistome analysis accounted for most of the resistance phenotypes, except for tigecycline and fosfomycin. Insertional inactivation of mgrB (distinct per clone) conferred colistin resistance in all 19 isolates. NGS single nucleotide polymorphism (SNP) analysis validated the clonal relatedness of the ST147 and ST101 strains and revealed the possible presence of two index ST147 strains and the microevolution of ST101 strains. Distinct, but highly related, IncL OXA-48-encoding plasmid lineages were identified; plasmids of the ST147 strains were identical with the plasmid of ST11 OXA-48-Kp which caused the 2012 outbreak. In conclusion, biclonal circulation of OXA-48-Kp and, alarmingly, emergence of a PDR clone are reported. These observations, along with the challenging phenotypic detection of OXA-48 producers and the high reported transmissibility of blaOXA-48, necessitate intensive efforts to prevent their further spread.
KEYWORDS: carbapenemases, NGS, SNPs, biclonal circulation, microevolution
INTRODUCTION
Klebsiella pneumoniae is an important cause of community-acquired and health care-associated infections (1). The emergence and dissemination of carbapenem-resistant K. pneumoniae (CRKP) strains has posed an urgent threat to public health on a global scale (2). In fact, CRKP strains are characterized by multiantibiotic resistance profiles that involve most beta-lactams, including carbapenems, along with other non-beta-lactam antibiotics (3), limiting the therapeutic options and affecting the clinical outcome of infections, with high mortality rates reported in some categories of patients (4).
Carbapenem resistance in K. pneumoniae is mainly due to the acquisition of carbapenemase genes associated with mobile genetic elements such as plasmids, transposons, and mobile gene cassettes carried on integrons (5, 6). Among the various acquired carbapenemases that can be found in CRKP strains, OXA-48 is one of the most prevalent. Strains of CRKP producing OXA-48 are highly endemic in Turkey, North Africa, India, and the Middle East (7, 8), and OXA-48 is currently the most prevalent carbapenemase among CRKP strains in several European countries, including France, Spain, Belgium, and Malta (9). In two recent reports from Spain and France, the prevalence of OXA-48-like among carbapenemase-producing K. pneumoniae isolates was 85.2% and 87.2%, respectively (10, 11).
In Europe, Greece has been among the first countries reporting the emergence of CRKP since 2002 (12) and thenceforth has been the one with the highest proportions of instances (13). The Greek CRKP epidemic was initially caused by strains producing VIM-1-type carbapenemases (14), which have subsequently been outnumbered by KPC-producing strains, which emerged since 2007 (15). More recently, double-carbapenemase-bearing strains that harbored both KPC and VIM-1 genes (16, 17) and, eventually, OXA-48 producers have also been reported (18). Currently, both KPC-2 and VIM-1 enzymes are considered endemic in Greece (19–22), whereas OXA-48 carbapenemase-producing K. pneumoniae (OXA-48-Kp) strains are still sporadic (23).
Hospital settings are high-risk environments for the spread of CRKP infections, which often result in severe outbreaks. Nosocomial spread is most prominent in intensive care units (ICUs) due to the critically ill population that receives prolonged treatment with last-line antibiotics, which enhance the selection of resistant microbiota (24). In the ICU of our hospital, a tertiary care hospital of Piraeus, Greece, from 2010 to 2015 the incidence of bacteremia due to CRKP grew from 7.96 in 2010 to 12.72 per 1,000 patient days in 2015. The incidence refers to all bloodstream infections (BSIs) caused by CRKP, i.e., K. pneumoniae strains producing KPC, VIM, both KPC and VIM (KPC+VIM), and OXA-48, with the predominant carbapenemase type being KPC. In 2011 to 2012, however, an outbreak of sequence type 11 (ST11) OXA-48 K. pneumoniae was reported in the ICU of our hospital (18), which was contained after the implementation of appropriate infection control measures. In June 2014 we noticed the reintroduction of OXA-48 producers in the same setting when we isolated the first OXA-48-Kp from a rectal screening sample; however, the first OXA-48-Kp BSI episode occurred in August 2014. Analytically, in 2010 we had 35 K. pneumoniae BSI episodes (19 KPC, 13 VIM, and 3 KPC+VIM), followed by 42 BSI episodes in 2011 (35 KPC, 4 VIM, 2 KPC+VIM, and 1 OXA-48), 42 BSI episodes in 2012 (33 KPC, 1 VIM, 3 KPC+VIM, and 5 OXA-48), 42 BSI episodes in 2013 after the control of the OXA-48 outbreak (28 KPC and 14 VIM), 45 BSI episodes in 2014 (27 KPC, 12 VIM, 1 KPC+VIM, and 5 OXA-48), and 66 BSI episodes in 2015 (41 KPC, 7 VIM, 5 KPC+VIM, and 13 OXA-48). BSI episodes caused by OXA-48-Kp represented 2.4% in 2011, 11.9% in 2012, 11.1% in 2014, and 19.1% in 2015.
The aim of this study is to analyze the mode of spread and the characteristics of epidemic OXA-48-Kp strains responsible for bloodstream infections in ICU patients that emerged in our setting in June 2014 and have been circulating thereafter. We assessed the strains' relationships via clonal lineage profiling and evaluated the presence of relevant resistance determinants and epidemic OXA-48-encoding IncL plasmid by next-generation sequencing (NGS) analysis. Additionally, we investigated the molecular epidemiology of these strains in order to better understand the clinical evolution and eventually contain their spread.
RESULTS
Description of the OXA-48-Kp occurrence in the ICU.
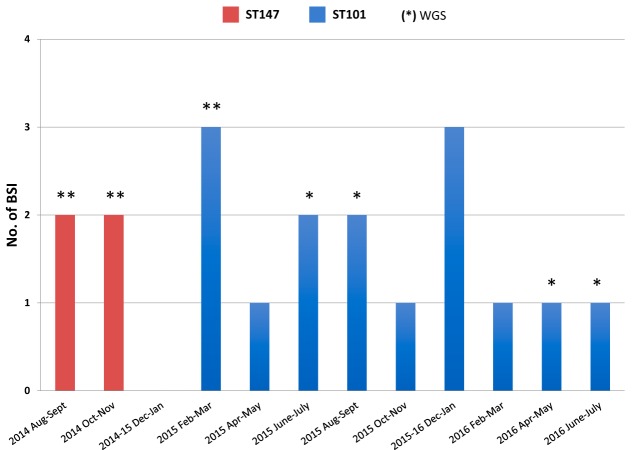
The first case of bacteremia caused by OXA-48-Kp was observed in the ICU in August 2014. Thereafter, 18 additional cases were observed until July 2016 (Fig. 1). The characteristics of the 19 patients that developed OXA-48-Kp bacteremia are summarized in Table 1. Of these patients, 5 patients were transferred to the ICU after prior admission to surgical wards, while another 4 were directly transferred to the ICU from other hospitals, and the remaining 10 were directly admitted to the ICU. Bacteremia was diagnosed from 0 to 151 days following admission to the ICU, with a median time to bloodstream isolation of 29 days. The overall patient mortality rate from BSI was 26.3% at 7 days, 36.8% at 25 days, 47.4% at 80 days, 52.6% at 120 days, and 57.9% at 330 days. All patients were screened for rectal carriage of CRKP, and previous colonization by OXA-48-Kp was documented in 10 patients.
FIG 1.
Evolution of the outbreak caused by OXA-48-producing K. pneumoniae (OXA-48-Kp) in Tzaneio Hospital. Red columns, numbers of new BSI cases caused by ST147 OXA-48-Kp; blue columns, numbers of new BSI cases caused by ST101 OXA-48-Kp. The number of isolates selected for WGS typing is indicated by asterisks.
TABLE 1.
Demographic and clinical data of the 19 study patients that yielded the 19 BSI isolates
| Pt no.a | Blood isolate no. | Sexb | Age range (yr) | ICU admission date (mo/yr) | Time from ICU admission to OXA-48 Kp BSI (days) | CRKP screening result upon ICU admission | Date of previous OXA-48 Kp carriage prior to BSI (mo/yr) | Previous hospitalization | Patient transferc | Outcome |
|---|---|---|---|---|---|---|---|---|---|---|
| P1 | K1 | F | 80–89 | 5/14 | 100 | Negative | None | Yes | Intrahospital | Death |
| P2 | K2 | M | 70–79 | 8/14 | 14 | Negative | None | Yes | Intrahospital | Death |
| P3 | K3 | M | 40–49 | 9/14 | 31 | Negative | 9/14 | Yes | Intrahospital | Death |
| P4 | K4 | F | 70–79 | 9/14 | 29 | Negative | None | Yes | Intrahospital | Exit |
| P5 | K5 | F | 60–69 | 1/15 | 23 | Negative | 1/15 | No | Death | |
| P6 | K6 | F | 60–69 | 1/15 | 151 | Negative | 4/15 | No | Death | |
| P7 | K7 | F | 60–69 | 1/15 | 16 | Negative | None | No | Exit | |
| P8 | K8 | F | 70–79 | 2/15 | 90 | Positive | None | Yes | Intrahospital | Death |
| P9 | K9 | M | 60–69 | 3/15 | 12 | Negative | None | No | Death | |
| P10 | K10 | M | 60–69 | 6/15 | 29 | Positive | None | Yes | Intrahospital | Exit |
| P11 | K11 | F | 30–39 | 9/15 | 22 | Positive | 9/15 | Yes | Intrahospital | Exit |
| P12 | K12 | F | 40–49 | 9/15 | 75 | Negative | 9/15 | No | Exit | |
| P13 | K13 | M | 80–89 | 9/15 | 0 | Positive | None | Yes | Intrahospital | Death |
| P14 | K14 | M | 50–59 | 11/15 | 15 | Negative | None | No | Death | |
| P15 | K15 | F | 60–59 | 12/15 | 14 | Negative | 12/15 | No | Death | |
| P16 | K16 | F | 50–59 | 1/16 | 13 | Negative | 1/16 | No | Exit | |
| P17 | K17 | M | 80–89 | 2/16 | 20 | Negative | 2/16 | No | Death | |
| P18 | K18 | M | 20–29 | 3/16 | 29 | Negative | 3/16 | No | Exit | |
| P19 | K19 | F | 60–69 | 7/16 | 22 | Positive | 7/16 | Yes | Intrahospital | Exit |
Pt, patient.
M, male; F, female.
All intrahospital transfers refer to patients that had undergone surgery and were hospitalized in different surgical units of our hospital prior to their admission in ICU.
All 19 isolates were nonsusceptible to carbapenems. Four isolates were pandrug resistant (PDR) (25), while the remaining ones exhibited an extensively drug-resistant (XDR) phenotype, with various resistance profiles (Table 2). Carbapenemase production was confirmed by a modified Hodge test (MHT); a combined disk test (CDT), containing meropenem, phenylboronic acid, and EDTA (26), was negative for both Ambler class A and B carbapenemases. PCR amplification and sequencing verified that all isolates harbored the blaOXA-48-like carbapenemase gene.
TABLE 2.
Characteristics and resistance phenotypes of the 19 BSI isolates
| Isolate no. | Isolation date (month/yr) | Phenotypea | PFGE | ST | MIC (μg/ml)b |
||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| WGS | COL | TIGE | AK | CN | CIP | AMC | CAZ | CTX | FEP | TZP | SXT | IMI | ERT | MEM | FOSF | CHLOR | |||||
| K1 | 8/14 | PDR | A | 147 | Yes | >8 | 4 | >32 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 8 | >4 | 32 | >64 | >128 |
| K2 | 8/14 | PDR | A | 147 | Yes | >8 | 4 | >32 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 8 | >4 | 32 | 64 | >128 |
| K3 | 10/14 | PDR | A | 147 | Yes | >8 | 4 | >32 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 16 | >4 | 32 | 64 | >128 |
| K4 | 10/14 | PDR | A | 147 | Yes | 8 | 4 | >32 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 8 | >4 | >32 | >64 | >128 |
| K5 | 2/15 | XDR | B | 101 | Yes | >8 | 0.5 | 8 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 8 | >4 | 8 | 16 | 16 |
| K6 | 6/15 | XDR | B | 101 | No | >8 | 4 | 16 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 8 | >4 | 16 | 32 | 32 |
| K7 | 2/15 | XDR | B | 101 | No | >8 | 1 | 8 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 8 | >4 | 32 | ≤8 | 16 |
| K8 | 5/15 | XDR | B | 101 | No | >8 | 4 | 8 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 4 | >4 | 16 | ≤8 | >128 |
| K9 | 3/15 | XDR | B | 101 | Yes | >8 | 0.5 | 8 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 8 | >4 | 16 | 32 | 16 |
| K10 | 7/15 | XDR | B | 101 | Yes | >8 | 0.5 | 8 | 1 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 8 | >4 | 32 | 32 | 32 |
| K11 | 9/15 | XDR | B | 101 | No | >8 | 0.5 | 8 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 8 | >4 | 16 | 16 | 16 |
| K12 | 11/15 | XDR | B | 101 | No | >8 | 0.5 | 16 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 32 | >4 | >32 | 16 | 16 |
| K13 | 9/15 | XDR | B | 101 | Yes | >8 | 4 | 8 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 4 | >4 | 16 | 32 | 32 |
| K14 | 12/15 | XDR | B | 101 | No | >8 | 0.5 | 16 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 16 | >4 | 32 | 16 | 16 |
| K15 | 12/15 | XDR | B | 101 | No | >8 | 0.5 | 16 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 4 | >4 | 16 | ≤8 | 16 |
| K16 | 1/16 | XDR | B | 101 | No | >8 | 4 | 16 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 16 | >4 | 32 | 16 | >128 |
| K17 | 2/16 | XDR | B | 101 | No | >8 | 4 | 16 | >8 | >4 | >32 | >16 | >4 | >8 | >64 | >8 | 8 | >4 | 32 | 16 | >128 |
| K18 | 4/16 | XDR | B | 101 | Yes | >8 | 4 | 16 | >8 | >4 | >32 | 1 | >4 | 4 | >64 | >8 | 16 | >4 | >32 | 32 | >128 |
| K19 | 7/16 | XDR | B | 101 | Yes | >8 | 4 | 8 | >8 | >4 | >32 | 1 | 4 | 8 | >64 | >8 | 8 | >4 | >32 | 64 | >256 |
PDR, pandrug resistant; XDR, extensively drug resistant.
COL, colistin; TIGE, tigecycline; AK, amikacin; CN, gentamicin; CIP, ciprofloxacin; AMC, amoxicillin-clavulanate; CAZ, ceftazidime; CTX, cefotaxime; FEP, cefepime; TZP, piperacillin-tazobactam; SXT, trimethoprim-sulfamethoxazole; IMI, imipenem; ERT, ertapenem; MEM, meropenem; FOSF, fosfomycin; CHLOR, chloramphenicol.
Analysis by pulsed-field gel electrophoresis (PFGE) yielded two different profiles, A and B, which were identified as ST147 and ST101, respectively, by multilocus sequence typing (MLST). The ST147 isolates were responsible for the first four cases, which occurred between August and October 2014, while the ST101 isolates contributed all the subsequent cases (Fig. 1). The ST147 isolates exhibited a PDR phenotype, while the ST101 isolates exhibited an XDR phenotype (Table 2).
NGS analysis.
To further investigate the outbreak, NGS was performed on all four ST147 isolates and on six ST101 isolates representative of different times of isolation and different resistance profiles (Fig. 1 and Table 2).
NGS of the ST147 isolates.
Single nucleotide polymorphisms (SNPs) showed that three ST147 strains (K1, K3, and K4) were closely related (2 to 16 SNPs) while one (K2) was more divergent (91 to 97 SNPs). These data suggested that the first phase of the outbreak was contributed by two ST147 strains, one of which (K1) was responsible for two additional cases (K3 and K4). Investigation of CRISPR regions further supported this hypothesis since K1, K3, and K4, but not K2, shared identical CRISPR contents (in terms of type and number of spacers and direct repeat regions within confirmed CRISPRs). In detail, in each strain two confirmed CRISPR arrays were identified, of which one was common to all ST147 members (CRISPR region 1), while the other showed marked differences between K1, K3, K4 (CRISPR region 2), and K2 (CRISPR region 3) in terms of spacer sequences (see Table S1 in the supplemental material).
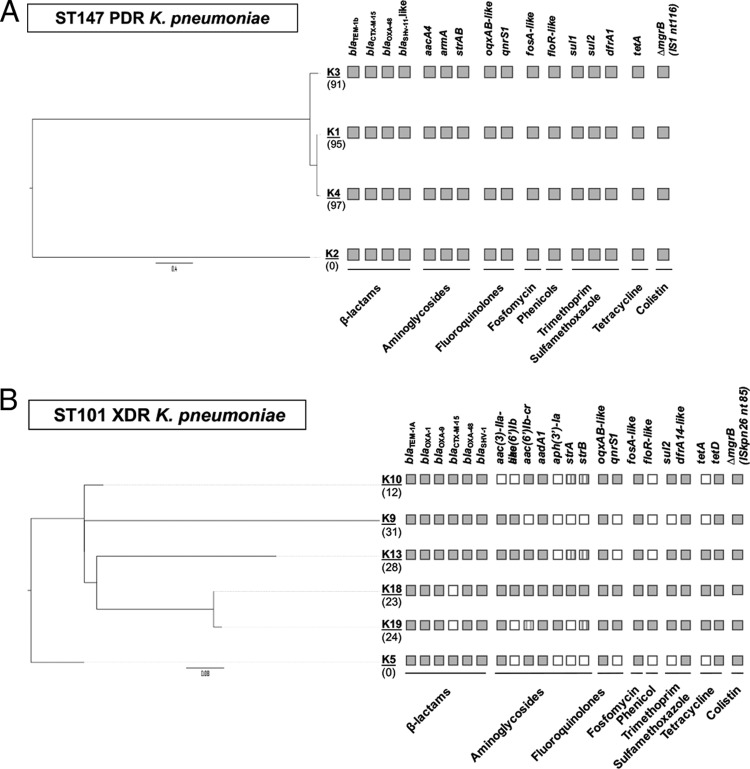
Analysis of the resistome of the ST147 isolates could account for most of their resistance traits, including resistance to chloramphenicol (floR), aminoglycosides (aacA4, armA, strA, and strB), quinolones (qnrS1, oqxAB, parC encoding the S-to-I change at position 80 [parCS80I], and gyrAS83I), trimethoprim-sulfamethoxazole (dfrA1, sul1, and sul2), colistin (insertional inactivation of mgrB by an IS1 element at nucleotide 116), and all beta-lactams (blaOXA-48, blaCTX-M-15, blaTEM-1b, and blaSHV-11) (Fig. 2A).
FIG 2.
Resistance genes and genetic relatedness of ST147 (top) and ST101 (bottom) OXA-48-producing K. pneumoniae strains. Isolates K2 and K5 were used as references for the core-genome-based phylogeny of ST147 and ST101 outbreak lineages, respectively. The distance scale bars are shown below the trees and are in units of the number of nucleotide substitutions per site. The SNP differences (n), with respect to the reference genomes (K2 and K5), are shown in brackets under each isolate. Presence or absence of specific genes is indicated by filled or blank squares, respectively; the presence of allelic variants is indicated by striped squares.
Interestingly, the blaCTX-M-15 gene was found to be located on large chromosomal contigs (855 kb) in both ST147 sublineages. Whole-genome alignments performed against the K. pneumoniae reference strain NTUH-K2044 (GenBank accession number AP006725.1) revealed that the chromosomal integration of blaCTX-M-15 occurred in all ST147 strains at the same position, resulting in the disruption of a gene coding for a cytochrome o ubiquinol oxidase subunit (K. pneumoniae NTUH-K2044 locus tag KP1_1262), of which 5′ and 3′ fragments were located onto two different contigs. The mobilization of blaCTX-M-15 was likely mediated by ISEcp1 since its remnants and inverted repeat ends were detected within the disrupted gene and were flanked by a 5-bp target site duplication (5′-AACCAA-3′) generated by the transposition event.
These data suggested that the beta-lactamase gene integration had occurred before divergence of the two sublineages.
Regarding fosfomycin and tigecycline resistance, analysis of genes known to be involved in the development of a resistance phenotype (Table S2) revealed several genetic alterations compared to the genes from the susceptible K. pneumoniae reference strain NTUH-K2044 (GenBank accession number AP006725.1). With respect to fosfomycin resistance, all isolates carried the same fosA allelic variant (fosA variant belonging to ST147 [fosAST147]), and two of them (K1 and K4) also carried an insertion (G at position 1138) causing a frameshift in glpT (coding for one of the main transport systems responsible for fosfomycin uptake), likely explaining the higher fosfomycin resistance level observed in these isolates (Table 2). It should be noted here that of the four patients carrying fosfomycin-resistant strains, only the first one (patient 1 [P1]) received fosfomycin for 2 days prior to the OXA-48-Kp BSI. Concerning tigecycline resistance, only acrE among the screened genes showed a missense mutation compared to the sequence of the reference strain NTUH-K2044 (Table S2). However, the lack of an isogenic tigecycline-susceptible strain and the presence of the same mutation in a number of publicly available K. pneumoniae sequences with unknown tigecycline resistance status do not allow a direct correlation of the tigecycline resistance phenotype/genotype.
NGS of the ST101 isolates.
An SNP-based phylogenetic analysis revealed that all ST101 strains were highly genetically related, exhibiting a limited number of SNPs, ranging from 1 to 39. The most divergent tested strain was K9, a strain that exhibits similarities with K10, which likely evolved from K9 as a distinct sublineage (Fig. 2B). Interestingly, although P9 was hospitalized in the ICU during the same period as P6 and P8, OXA-48-Kp colonization was never identified. However, P9 developed an OXA-48-Kp catheter-related urinary tract infection 2 days prior to the BSI isolation (data not shown). The transfer of the patient to the isolation ward may have prevented the spread of this sublineage. Another divergent strain was K13, which was supposed to have been acquired during the hospitalization of patient P13 in the surgical ward and which is the only study OXA-48-Kp isolate from a patient previously hospitalized in this department; no apparent epidemiological link was found between patient P13 and the remaining OXA-48-Kp-carrying patients. We should note that OXA-48-Kp was not recovered from this patient during his hospitalization in the surgical ward or from any other patient in this ward. In contrast to ST147, all ST101 strains belonged to a phylogenetically more homogeneous population and most probably evolved throughout the outbreak period from a common index case.
The ST101 isolates exhibited a rather heterogeneous, acquired resistome profile (Fig. 2B) yet one still concordant with the XDR phenotype. All ST101 isolates were nonsusceptible to ciprofloxacin (presence of oqxAB-like, qnrS1, parCS80I, and gyrAS83Y-D87N), trimethoprim-sulfamethoxazole (sul2 and dfrA14-like), aminoglycosides (aac- and aad-type modifying enzymes), and colistin (insertional inactivation of mgrB by ISKpn26 at nucleotide 85). Four out of six isolates tested were nonsusceptible to all beta-lactams (for blaOXA-48, blaOXA-1, blaOXA-9, blaCTX-M-15, blaTEM-1A, and blaSHV-1), while the remaining two isolates were susceptible to ceftazidime and did not carry any extended-spectrum beta-lactamase (ESBL) gene (Fig. 2B).
Analysis of genes possibly involved in tigecycline and fosfomycin resistance revealed that all ST101 strains exhibited several genetic alterations, including missense (ramR, acrEF, marR, soxS, murA, glpT, and uhpBC) and nonsense (romA) mutations or insertional inactivation by an insertion element ([IS] acrR) (Table S2), and carried the same allelic variant of fosA (ST101 fosA [fosAST101]), which differed from the one found in ST147 isolates.
However, the aforementioned alterations were detected in all ST101 strains, regardless of the fosfomycin/tigecycline resistance pattern (Table 2), and a clear genotype-phenotype correlation could not be assessed.
NGS analysis of OXA-48-encoding plasmids.
Analysis of plasmid replicons revealed various types of plasmid incompatibility groups, among which only the IncR and IncL types were constantly detected in all strains of both ST147 and ST101 OXA-48-Kp isolates (Fig. S1). Sequence analysis revealed that in all strains the blaOXA-48 gene was embedded in a Tn1999.2 composite transposon, in turn invariably associated with a 64-kb IncL plasmid, corresponding to the elective plasmid lineage responsible for blaOXA-48 dissemination worldwide (27).
A sequence comparison with the prototype IncL OXA-48-encoding plasmid pOXA-48 (GenBank accession number NC_019154) showed that two plasmid sublineages were present, differing in 125 and 165 SNPs and associated with ST147 and ST101 OXA-48-Kp strains, respectively. Of note, this comparison revealed that 125 of 165 SNPs detected in the plasmid sublineage of ST101 were identical to those found in the cognate sublineages of ST147, and most of these polymorphisms (n = 102) were located within a specific region (from nucleotide 20514 to 25629 of pOXA-48) that might have diverged more rapidly since it included genes not relevant for plasmid stability and maintenance processes. A direct comparison of the blaOXA-48-carrying plasmids from the two STs revealed that they differed in 40 SNPs, located in a specific region of ST101 plasmids (corresponding to nucleotide 9779 to 13128 of pOXA-48). Altogether these findings suggested the existence of two distinct, but highly related, IncL plasmid sublineages in ST147 and ST101 isolates, likely derived from a common ancestor.
Since during 2012 an outbreak of ST11 OXA-48-Kp occurred in the same ICU (18) and since, similarly to the present case, the presence of an OXA-48-encoding IncL plasmid was reported, whole-genome sequencing (WGS) was performed on plasmid DNA obtained from two selected ST11 OXA-48-Kp isolates (i.e., the first and the last collected OXA-48-Kp isolates causing BSI episodes, namely, K1 and K11 in Voulgari et al. [18]) in order to evaluate the possible transmission routes of such epidemic plasmids.
Sequence analysis revealed that in both plasmids from ST11, the blaOXA-48 gene was carried on a Tn1999.2 element, as observed for the 2014–2016 OXA-48-Kp. Intriguingly, a finer sequence comparison showed that plasmids from ST11 and ST147 were identical at the nucleotide level and shared the same number of SNP differences (n = 40), in contrast to plasmids from ST101, which were more phylogenetically divergent.
A BLAST search of plasmid regions carrying the most informative SNPs identified in ST147 and ST11 plasmids (i.e., regions including the previously identified 102 SNP differences) revealed a perfect match only with IncL OXA-48-encoding plasmids isolated from ST147 or ST101 K. pneumoniae strains involved in a nosocomial outbreak that occurred in Germany during 2013 to 2014 (28). The IncL plasmid sublineage detected in Greek ST147 isolates was found to be highly related (differing in three SNPs only) to the cognate lineage detected in ST147 OXA-48-Kp from Germany, thus pointing out a close genomic relatedness of such plasmids circulating in this K. pneumoniae high-risk clone.
DISCUSSION
Since the first report of VIM-1-producing K. pneumoniae (12), the prevalence of CRKP in Greek hospitals has increased drastically, and currently KPC and VIM carbapenemases are considered endemic in Greece. Although OXA-48 enzymes have been reported in sporadic cases, we estimate that the real prevalence of OXA-48-Kp in our country is currently unknown because of the scarce surveillance data available from other Greek health care settings.
In fact, a previous outbreak of OXA-48-Kp belonging to the distinct lineage ST11 emerged in 2011 and terminated in 2012 in the ICU of our tertiary hospital (18). Despite the occurrence of multiple simultaneous CRKP epidemics in our ICU, the present study selected for analysis only archived BSI OXA-48-producing K. pneumoniae isolates recovered from 2014 to 2016. To our knowledge, none of the patients had traveled abroad to high-endemicity settings, but nine patients had been previously hospitalized in different wards of the hospital or in other health care facilities. The current outbreak was linked to the clonal dissemination of K. pneumoniae strains that belonged consecutively to ST147 and ST101, both of which have been previously associated with OXA-48 and other carbapenemase producers (29–32). A pan-resistant ST147 KPC-2-producing K. pneumoniae strain has been described recently (33) but, to our knowledge, this is the first report of a PDR ST147 OXA-48-Kp isolate.
ST147 is a successful clone with worldwide spread, associated with KPC-2-producing K. pneumoniae (33), with NDM- and CTX-M coproducers in the United States (34), and with OXA-48-Kp (29, 35), while ST101 has been associated with CTX-M-producing K. pneumoniae in Hungary (36), with KPC-2 in Spain (37) and Italy (38), and with OXA-48 in Europe (8, 31, 32, 35, 39, 40) and Africa (31), suggesting intercontinental spread.
In our study, the ST147 strains were identified in four patients (P1 to P4) who had undergone surgery prior to their transfer to the ICU. It is noteworthy that patient P1 was transferred to a different ICU for 19 days when our ICU was closed for disinfection. At readmission to our ICU, she carried a KPC-producing organism and 2 weeks later had been transferred to the isolation ward, where bacteremia caused by the K1 OXA-48-Kp was later diagnosed. The stay of patient P1 in the ICU or in the isolation ward never coincided with that of patients P2 to P4, suggesting the presence of different sources for the epidemic (see Fig. S2 in the supplemental material). Although the stay of P2 to P4 in the ICU overlapped for long periods of time, NGS data indicated that K2 was the most divergent strain, suggesting the simultaneous circulation of two different subclones in our setting, transmitted via an unknown environmental source. Analyses of core-genome SNPs and of conserved CRISPR regions prompted us to identify genetically diverse sublineages of ST147. The analysis of CRISPR loci has recently been proven to be a useful epidemiological tool for investigating the genome diversity of closely related strains of Clostridium difficile (41), and present results showed that the same approach could provide important insight into the genome microevolution of K. pneumoniae as well.
The ST101 clone appeared later, in patient P5, who was colonized after a stay of 8 days in the ICU (Table 1 and Fig. S2). Unlike ST147, the more limited number of core-genome SNP differences suggested a clonal expansion for ST101 isolates.
Sequencing data demonstrated that ST147 and ST101 isolates carried two distinct OXA-48-encoding IncL plasmid lineages, exhibiting several SNP differences from each other and differing from the prototype pOXA-48 plasmid (27). Noteworthy, sequence comparison performed on OXA-48-encoding plasmids from ST147 and ST101 strains (2014 to 2016 outbreak) and from two representative ST11 strains (2011 to 2012 outbreak) highlighted that the ST11 plasmids were identical on the nucleotide level to the cognate elements found in the ST147 clone.
Altogether these findings suggest the following. (i) Distinct, but closely related, IncL OXA-48-encoding plasmid sublineages were associated with the epidemic clones of ST11/ST147 and ST101. (ii) While increasing diversity was observed in isolates from both ST147 and ST101, with several chromosomal polymorphisms being identified over time likely due to the circulation of different strains belonging to the same ST, neither of the two distinct IncL plasmid sublineages had undergone a microevolution throughout the outbreak period, showing no nucleotide differences between older and most recent plasmids within the corresponding ST (i.e., ST11/ST147 and ST101). (iii) In contrast to findings of other reports (28, 29, 42), sequence data did not suggest a direct transfer of the OXA-48-encoding IncL plasmid from ST147 to ST101 during the outbreak because of the high number of SNP differences (n = 40) uniquely detected in the ST101 plasmid sublineage but not in the ST147 counterpart. (iv) The plasmid backbone was overall identical in the IncL plasmids of the ST147 and ST101 strains, differing only in a region carrying 40 SNPs. Present findings suggested that microevolution of these peculiar IncL sublineages may have selectively occurred among the ST11, ST147, and, to a lesser extent, ST101 clones. The fact that IncL OXA-48-encoding plasmids closely related to those analyzed here were described from ST11 and ST147 K. pneumoniae strains from a recently described outbreak in Germany (28) further supports this hypothesis. (v) Considering that the same OXA-48-encoding plasmid was found to be harbored by ST11 and ST147 strains, it is plausible that such a plasmid remained undetected and still circulated after the (supposed) resolution of the 2012 outbreak until late 2014, when it reemerged in the ST147 lineage and was then responsible for a subsequent epidemic in the same setting.
Additionally, all of the tested strains also shared an IncR-type plasmid, which is probably responsible for the wide dissemination of multiple resistance genes in K. pneumoniae (29), underlining the danger of pan-resistance dissemination. Within the ST147 clone, for example, the IncR plasmid was supposed to be the source platform from which the ISEcp1 transposition unit had moved, mediating the chromosomal integration of blaCTX-M-15. These results were consistent with the data recently reported by a study on microevolution of ST147 OXA-48-Kp spreading in a hospital setting in Germany (28). In that case, indeed, an IncR-type multidrug-resistant (MDR) plasmid was responsible for the dissemination of blaCTX-M-15 along with other determinants conferring resistance to quinolones and aminoglycosides.
Analysis of the overall plasmid content of ST147 and ST101 strains revealed a different trend in the acquisition and loss of such elements and resistance determinants. The plasmidome of ST147 strains appeared almost stable in terms of detected replicons (Fig. S1A), and any variations were observed in the global resistome (Fig. 2A), unlike cognate ST147 strains from Germany (28). Conversely, a more heterogeneous genetic background was observed in the ST101 clone over time due to the presence of a novel plasmid replicon (IncN-type) in most recent strains that, on the other hand, tended to lose some resistance determinants [i.e., aac(6′)-lb, strA, and blaCTX-M-15], thus leading to some phenotypic changes (Table 2).
All four study ST147 strains harbored the beta-lactamase genes blaCTX-M-15, blaTEM1-b, and blaSHV-11, along with blaOXA-48. In our study, opposed to other findings (29, 35) and in accordance with recent reports (28), all of the aforementioned strains were resistant to carbapenems. Additionally, they all presented the same insertion sequence (IS1) at nucleotide 116 in mgrB, a negative regulator of phoPQ, suggesting that acquired colistin resistance is mediated by the same mechanism described previously for KPC-producing K. pneumoniae strains (43). In accordance with the phenotype, the resistance genes for phenicol, co-trimoxazole, and aminoglycosides were expressed by all of the strains. Among the aminoglycoside genes, our ST147 strains harbored armA, as described previously in an OXA-48-Kp strain of the same lineage (23), validating the circulation of this clone in our country.
Similar to ST147 strains, ST101 study strains displayed an mgrB insertional inactivation (ISKpn26 at nucleotide 85) that conferred resistance to colistin. With respect to aminoglycoside resistance, all study strains, including K10 which was susceptible to both amikacin and gentamicin, bore various genes encoding aminoglycoside-modifying enzymes (AME).
With respect to fosfomycin and tigecycline, in both ST147 and ST101 lineages several genetic alterations were identified in genes known to be involved in resistance (Table S2); however, the observed resistance phenotypes were not clearly explained by WGS, and further studies are therefore needed.
The challenging phenotypic detection of OXA-48 carbapenemases and the rapid horizontal transfer of OXA-48-encoding plasmids favor hospital outbreaks linked to patient transfer (29) and draw attention to the need for continuous and meticulous surveillance, as well as timely investigation.
The unraveling of the resistance phenotypes via resistome analysis along with characterization of the population genetic structure revealed a double origin of the epidemic described here, suggesting the importance of molecular diagnostics for tracing outbreak isolates. Further molecular studies are needed in order to better clarify the evolution and transmission trajectories of plasmids, determine the virulence factors and the potential rearrangements in chromosomal regions, and decipher currently unknown resistance mechanisms.
MATERIALS AND METHODS
A total of 19 K. pneumoniae isolates producing the OXA-48 carbapenemase were recovered from blood cultures of 19 patients hospitalized in the general ICU of Tzaneio tertiary hospital in Piraeus, Attica, Greece, in the period from August 2014 to July 2016. Some of the patients had undergone surgery and thus were hospitalized in surgical wards prior to their admission to the ICU. Considering the endemicity of CRKP in our country, all patients were screened for rectal carriage of CRKP upon ICU admission (26). Except for one patient (P13), rectal screening results were negative, and CRKP colonization and/or infection occurred only after the patients' admission to the ICU (Table 1). Patient P13 was screened OXA-48-Kp positive upon ICU admission; however, OXA-48-Kp had never been recovered from any clinical samples during hospitalization in the surgical unit. All 19 study isolates were further sent to reference laboratories at the University of Athens and Siena for molecular testing. Among them, 10 were considered representative of the outbreak and were further studied by NGS.
Phenotypic analysis.
Identification and initial susceptibility testing were performed by a Vitek 2 automated system (bioMérieux, Marcy l'Etoile, France). Carbapenemase production was evaluated by a modified Hodge test (MHT), and carbapenemase differentiation was assessed by the aforementioned CDT (26). Susceptibility testing and determination of MIC values were also carried out by reference broth microdilution using lyophilized custom plates (Sensititre; Alere Technologies, GmbH, Jena, Germany) and by reference agar dilution for fosfomycin susceptibility testing, according to CLSI guidelines (44) (Table 2). Results were interpreted according to the European Committee on Antimicrobial Susceptibility Testing (EUCAST) clinical breakpoints (www.eucast.org). Escherichia coli ATCC 25922 and K. pneumoniae KP-6884 (mcr-1.2 positive) (43) were used as control strains for antimicrobial susceptibility testing.
Molecular analysis.
Detection of blaOXA-48 genes was performed by PCR (18). Genotyping of the K. pneumoniae isolates was carried out by PFGE after digestion of genomic DNA with XbaI (45), and results were interpreted as described previously (46). MLST was performed (47), and sequence type (ST) was assigned according to the K. pneumoniae MLST database (Institut Pasteur Klebsiella pneumoniae MLST Database [http://bigsdb.pasteur.fr/klebsiella/klebsiella.html]).
Whole-genome sequencing and analysis.
Bacterial DNA was extracted from 10 selected K. pneumoniae outbreak isolates (four ST147 and six ST101) using the phenol-chloroform method (48). Plasmid DNA was extracted using a commercial kit (PureYield plasmid miniprep kit; Promega, Madison, WI, USA) from two selected ST11 K. pneumoniae isolates (i.e., the first and the last collected OXA-48-Kp isolate causing BSI) involved in a previous outbreak that occurred during 2012 in the same ICU (18). WGS was performed using an Illumina MiSeq platform (Illumina, Inc., San Diego, CA, USA), following a 2- by 300-bp paired-end approach, and raw reads were assembled using SPAdes (49). Each genome assembly was filtered from contigs shorter than 300 bp (i.e., the sequencing read length). Resistome analysis was carried out through ResFinder (http://www.genomicepidemiology.org), using genome assemblies as input, and by manual inspection for (i) identification of genes associated with colistin resistance (i.e., mgrB, pmrAB, and crrAB) and (ii) determination of allelic variants not correctly identified by ResFinder. Plasmid replicon analysis was performed through PlasmidFinder (http://www.genomicepidemiology.org) using genome assemblies as input. Clonal relatedness was evaluated using CSI Phylogeny (http://www.genomicepidemiology.org) with raw read data sets and default parameters, except for the minimum distance between SNPs that was set to 0. After any plasmid and repeated sequences were filtered out according to coverage values, the genome assembly of the oldest ST147 and ST101 isolates (i.e., K1 and K5) was used as a reference in the clonal analysis of the corresponding outbreak clone. Evaluation of SNPs occurring in OXA-48-encoding plasmids from outbreak isolates was performed using raw reads and the prototype IncL pOXA-48 plasmid (GenBank accession number NC_019154) as a reference.
The K. pneumoniae NTUH-K2044 strain (GenBank accession number AP006725.1) was used as a reference to evaluate the status of genes known to be involved in fosfomycin and tigecycline resistance. Search and analysis of clustered regularly interspaced short palindromic repeats (CRISPR) were carried out using the CRISPR web server, using default parameters and genome assemblies as input (50); only confirmed CRISPR regions were considered for the comparative analysis. Whole-genome alignments were performed using Mauve (51) and BLAST (http://blast.ncbi.nlm.nih.gov/Blast.cgi).
Archived isolates and corresponding patient data that were not previously published were used for the manuscript.
Accession number(s).
Draft genomes of sequenced ST147 and ST101 K. pneumoniae isolates have been deposited in the NCBI WGS database and are available under BioProject accession number PRJNA419295.
Supplementary Material
Footnotes
Supplemental material for this article may be found at https://doi.org/10.1128/AAC.02457-17.
REFERENCES
- 1.Chung PY. 2016. The emerging problems of Klebsiella pneumoniae infections: carbapenem resistance and biofilm formation. FEMS Microbiol Lett 363:fnw219. doi: 10.1093/femsle/fnw219. [DOI] [PubMed] [Google Scholar]
- 2.Zowawi HM, Forde BM, Alfaresi M, Alzarouni A, Farahat Y, Chong TM, Yin WF, Chan KG, Li J, Schembri MA, Beatson S, Paterson DL. 2015. Stepwise evolution of pandrug-resistance in Klebsiella pneumoniae. Sci Rep 5:15082. doi: 10.1038/srep15082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jeon JH, Lee JH, Lee JJ, Park KS, Karim AM, Lee CR, Jeong BC, Lee SH. 2015. Structural basis for carbapenem-hydrolyzing mechanisms of carbapenemases conferring antibiotic resistance. Int J Mol Sci 16:9654–9692. doi: 10.3390/ijms16059654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Munoz-Price LS, Poirel L, Bonomo RA, Schwaber MJ, Daikos GL, Cormican M, Cornaglia G, Garau J, Gniadkowski M, Hayden MK, Kumarasamy K, Livermore DM, Maya JJ, Nordmann P, Patel JB, Paterson DL, Pitout J, Villegas MV, Wang H, Woodford N, Quinn JP. 2013. Clinical epidemiology of the global expansion of Klebsiella pneumoniae carbapenemases. Lancet Infect Dis 13:785–796. doi: 10.1016/S1473-3099(13)70190-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Queenan AM, Bush K. 2007. Carbapenemases: the versatile β-lactamases. Clin Microbiol Rev 20:440–458. doi: 10.1128/CMR.00001-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nordmann P, Naas T, Poirel L. 2011. Global spread of carbapenemase-producing Enterobacteriaceae. Emerg Infect Dis 17:1791–1798. doi: 10.3201/eid1710.110655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nordmann P, Poirel L. 2014. The difficult-to-control spread of carbapenemase producers among Enterobacteriaceae worldwide. Clin Microbiol Infect 20:821–830. doi: 10.1111/1469-0691.12719. [DOI] [PubMed] [Google Scholar]
- 8.Potron A, Poirel L, Rondinaud E, Nordmann P. 2013. Intercontinental spread of OXA-48 beta-lactamase producing Enterobacteriaceae over a 11-year period, 2001 to 2011. Euro Surveill 18:20549. doi: 10.2807/1560-7917.ES2013.18.31.20549. [DOI] [PubMed] [Google Scholar]
- 9.Grundmann H, Glasner C, Albiger B, Aanensen DM, Tomlinson CT, Andraseviæ AT, Cantón R, Carmeli Y, Friedrich AW, Giske CG, Glupczynski Y, Gniadkowski M, Livermore DM, Nordmann P, Poirel L, Rossolini GM, Seifert H, Vatopoulos A, Walsh T, Woodford N, Monnet DL, European Survey of Carbapenemase-Producing Enterobacteriaceae (EuSCAPE) Working Group . 2017. Occurrence of carbapenemase-producing Klebsiella pneumoniae and Escherichia coli in the European survey of carbapenemase-producing Enterobacteriaceae (EuSCAPE): a prospective, multinational study. Lancet Infect Dis 17:153–163. doi: 10.1016/S1473-3099(16)30257-2. [DOI] [PubMed] [Google Scholar]
- 10.Palacios-Baena ZR, Oteo J, Conejo C, Larrosa MN, Bou G, Fernández-Martínez M, González-López JJ, Pintado V, Martínez-Martínez L, Merino M, Pomar V, Mora-Rillo M, Rivera MA, Oliver A, Ruiz-Carrascoso G, Ruiz-Garbajosa P, Zamorano L, Bautista V, Ortega A, Morales I, Pascual A, Campos J, Rodríguez-Baño J, GEIH-GEMARA (SEIMC) and REIPI Group for CPE . 2016. Comprehensive clinical and epidemiological assessment of colonisation and infection due to carbapenemase-producing Enterobacteriaceae in Spain. J Infect 72:152–160. doi: 10.1016/j.jinf.2015.10.008. [DOI] [PubMed] [Google Scholar]
- 11.Dortet L, Cuzon G, Ponties V, Nordmann P. 2017. Trends in carbapenemase-producing Enterobacteriaceae, France, 2012 to 2014. Euro Surveill 22:30461. doi: 10.2807/1560-7917.ES.2017.22.6.30461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Giakkoupi P, Xanthaki A, Kanelopoulou M, Vlahaki A, Miriagou V, Kontou S, Papafraggas E, Malamou-Lada H, Tzouvelekis LS, Legakis NJ, Vatopoulos AC. 2003. VIM-1 metallo-β-lactamase-producing Klebsiella pneumoniae strains in Greek hospitals. J Clin Microbiol 41:3893–3896. doi: 10.1128/JCM.41.8.3893-3896.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.European Centre for Disease Prevention and Control. 2017. Antimicrobial resistance surveillance in Europe 2016. Annual report of the European Antimicrobial Resistance Surveillance Network (EARS-Net). European Centre for Disease Prevention and Control, Stockholm, Sweden: https://ecdc.europa.eu/sites/portal/files/documents/AMR-surveillance-Europe-2016.pdf. [Google Scholar]
- 14.Psichogiou M, Tassios PT, Avlamis A, Stefanou I, Kosmidis C, Platsouka E, Paniara O, Xanthaki A, Toutouza M, Daikos GL, Tzouvelekis LS. 2008. Ongoing epidemic of blaVIM-1-positive Klebsiella pneumoniae in Athens, Greece: a prospective survey. J Antimicrob Chemother 61:59–63. doi: 10.1093/jac/dkm443. [DOI] [PubMed] [Google Scholar]
- 15.Tsakris A, Kristo I, Poulou A, Markou F, Ikonomidis A, Pournaras S. 2008. First occurrence of KPC-2-possessing Klebsiella pneumoniae in a Greek hospital and recommendation for detection with boronic acid disc tests. J Antimicrob Chemother 62:1257–1260. doi: 10.1093/jac/dkn364. [DOI] [PubMed] [Google Scholar]
- 16.Pournaras S, Poulou A, Voulgari E, Vrioni G, Kristo I, Tsakris A. 2010. Detection of the new metallo-beta-lactamase VIM-19 along with KPC-2, CMY-2 and CTX-M-15 in Klebsiella pneumoniae. J Antimicrob Chemother 65:1604–1607. doi: 10.1093/jac/dkq190. [DOI] [PubMed] [Google Scholar]
- 17.Woodford N, Turton JF, Livermore DM. 2011. Multiresistant Gram-negative bacteria: the role of high-risk clones in the dissemination of antibiotic resistance. FEMS Microbiol Rev 35:736–755. doi: 10.1111/j.1574-6976.2011.00268.x. [DOI] [PubMed] [Google Scholar]
- 18.Voulgari E, Zarkotou O, Ranellou K, Karageorgopoulos DE, Vrioni G, Mamali V, Themeli-Digalaki K, Tsakris A. 2013. Outbreak of OXA-48 carbapenemase-producing Klebsiella pneumoniae in Greece involving an ST11 clone. J Antimicrob Chemother 68:84–88. doi: 10.1093/jac/dks356. [DOI] [PubMed] [Google Scholar]
- 19.Karampatakis T, Antachopoulos C, Iosifidis E, Tsakris A, Roilides E. 2016. Molecular epidemiology of carbapenem-resistant Klebsiella pneumoniae in Greece. Future Microbiol 11:809–823. doi: 10.2217/fmb-2016-0042. [DOI] [PubMed] [Google Scholar]
- 20.Tokatlidou D, Tsivitanidou M, Pournaras S, Ikonomidis A, Tsakris A, Sofianou D. 2008. Outbreak caused by a multidrug-resistant Klebsiella pneumoniae clone carrying blaVIM-12 in a university hospital. J Clin Microbiol 46:1005–1008. doi: 10.1128/JCM.01573-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pournaras S, Protonotariou E, Voulgari E, Kristo I, Dimitroulia E, Vitti D, Tsalidou M, Maniatis AN, Tsakris A, Sofianou D. 2009. Clonal spread of KPC-2 carbapenemase-producing Klebsiella pneumoniae strains in Greece. J Antimicrob Chemother 64:348–352. doi: 10.1093/jac/dkp207. [DOI] [PubMed] [Google Scholar]
- 22.Kontopoulou K, Protonotariou E, Vasilakos K, Kriti M, Koteli A, Antoniadou E, Sofianou D. 2010. Hospital outbreak caused by Klebsiella pneumoniae producing KPC-2 beta-lactamase resistant to colistin. J Hosp Infect 76:70–73. doi: 10.1016/j.jhin.2010.03.021. [DOI] [PubMed] [Google Scholar]
- 23.Galani I, Anagnostoulis G, Chatzikonstantinou M, Petrikkos G, Souli M. 2016. Emergence of Klebsiella pneumoniae co-producing OXA-48, CTX-M-15, and ArmA in Greece. Clin Microbiol Infect 22:898–899. doi: 10.1016/j.cmi.2016.08.002. [DOI] [PubMed] [Google Scholar]
- 24.Tacconelli E, Cataldo MA, Dancer SJ, De Angelis G, Falcone M, Frank U, Kahlmeter G, Pan A, Petrosillo N, Rodríguez-Baño J, Singh N, Venditti M, Yokoe DS, Cookson B, European Society of Clinical Microbiology . 2014. ESCMID guidelines for the management of the infection control measures to reduce transmission of multidrug-resistant Gram-negative bacteria in hospitalized patients. Clin Microbiol Infect 20(Suppl 1):S1–S55. doi: 10.1111/1469-0691.12427. [DOI] [PubMed] [Google Scholar]
- 25.Magiorakos AP, Srinivasan A, Carey RB, Carmeli Y, Falagas ME, Giske CG, Harbarth S, Hindler JF, Kahlmeter G, Olsson-Liljequist B, Paterson DL, Rice LB, Stelling J, Struelens MJ, Vatopoulos A, Weber JT, Monnet DL. 2012. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect 18:268–281. doi: 10.1111/j.1469-0691.2011.03570.x. [DOI] [PubMed] [Google Scholar]
- 26.Tsakris A, Themeli-Digalaki K, Poulou A, Vrioni G, Voulgari E, Koumaki V, Agodi A, Pournaras S, Sofianou D. 2011. Comparative evaluation of combined-disk tests using different boronic acid compounds for detection of Klebsiella pneumoniae carbapenemase-producing Enterobacteriaceae clinical isolates. J Clin Microbiol 49:2804–2809. doi: 10.1128/JCM.00666-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Poirel L, Bonnin RA, Nordmann P. 2012. Genetic features of the widespread plasmid coding for the carbapenemase OXA-48. Antimicrob Agents Chemother 56:559–562. doi: 10.1128/AAC.05289-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zautner AE, Bunk B, Pfeifer Y, Spröer C, Reichard U, Eiffert H, Scheithauer S, Groß U, Overmann J, Bohne W. 2017. Monitoring microevolution of OXA-48-producing Klebsiella pneumoniae ST147 in a hospital setting by SMRT sequencing. J Antimicrob Chemother 72:2737–2744. doi: 10.1093/jac/dkx216. [DOI] [PubMed] [Google Scholar]
- 29.Guo L, An J, Ma Y, Ye L, Luo Y, Tao C, Yang J. 2016. Nosocomial outbreak of OXA-48-producing Klebsiella pneumoniae in a Chinese hospital: clonal transmission of ST147 and ST383. PLoS One 11:e0160754. doi: 10.1371/journal.pone.0160754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lascols C, Peirano G, Hackel M, Laupland KB, Pitout JD. 2013. Surveillance and molecular epidemiology of Klebsiella pneumoniae isolates that produce carbapenemases: first report of OXA-48-like enzymes in North America. Antimicrob Agents Chemother 57:130–136. doi: 10.1128/AAC.01686-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Loucif L, Kassah Laouar A, Saidi M, Messala A, Chelaghma W, Rolain JM. 2016. Outbreak of OXA-48-producing Klebsiella pneumoniae involving an ST101 clone in Batna University Hospital, Algeria. Antimicrob Agents Chemother 60:7494–7497. doi: 10.1128/AAC.00871-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Skálová A, Chudejová K, Rotová V, Medvecky M, Študentová V, Chudáèková E, Lavicka P, Bergerova T, Jakubu V, Zemlickova H, Papagiannitsis CC, Hrabák J. 2017. Molecular characterization of OXA-48-like-producing Enterobacteriaceae in the Czech Republic and evidence for horizontal transfer of pOXA-48-like plasmids. Antimicrob Agents Chemother 61:e01889-16. doi: 10.1128/AAC.01889-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bathoorn E, Tsioutis C, da Silva Voorham JM, Scoulica EV, Ioannidou E, Zhou K, Rossen JW, Gikas A, Friedrich AW, Grundmann H. 2016. Emergence of pan-resistance in KPC-2 carbapenemase-producing Klebsiella pneumoniae in Crete, Greece: a close call. J Antimicrob Chemother 71:1207–1212. doi: 10.1093/jac/dkv467. [DOI] [PubMed] [Google Scholar]
- 34.Lee CS, Vasoo S, Hu F, Patel R, Doi Y. 2014. Klebsiella pneumoniae ST147 coproducing NDM-7 carbapenemase and RmtF 16S rRNA methyltransferase in Minnesota. J Clin Microbiol 52:4109-4110. doi: 10.1128/JCM.01404-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Liapis E, Pantel A, Robert J, Nicolas-Chanoine MH, Cavalié L, van der Mee-Marquet N, de Champs C, Aissa N, Eloy C, Blanc V, Guyeux C, Hocquet D, Lavigne JP, Bertrand X, ONERBA . 2014. Molecular epidemiology of OXA-48-producing Klebsiella pneumoniae in France. Clin Microbiol Infect 20:O1121–O1123. doi: 10.1111/1469-0691.12727. [DOI] [PubMed] [Google Scholar]
- 36.Melegh S, Schneider G, Horváth M, Jakab F, Emõdy L, Tigyi Z. 2015. Identification and characterization of CTX-M-15 producing Klebsiella pneumoniae clone ST101 in a Hungarian university teaching hospital. Acta Microbiol Immunol Hung 62:233–245. doi: 10.1556/030.62.2015.3.2. [DOI] [PubMed] [Google Scholar]
- 37.Oteo J, Pérez-Vázquez M, Bautista V, Ortega A, Zamarrón P, Saez D, Fernández-Romero S, Lara N, Ramiro R, Aracil B, Campos J, Spanish Antibiotic Resistance Surveillance Program Collaborating Group . 2016. The spread of KPC-producing Enterobacteriaceae in Spain: WGS analysis of the emerging high-risk clones of Klebsiella pneumoniae ST11/KPC-2, ST101/KPC-2 and ST512/KPC-3. J Antimicrob Chemother 71:3392–3399. doi: 10.1093/jac/dkw321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Del Franco M, Paone L, Novati R, Giacomazzi CG, Bagattini M, Galotto C, Montanera PG, Triassi M, Zarrilli R. 2015. Molecular epidemiology of carbapenem resistant Enterobacteriaceae in Valle d'Aosta region, Italy, shows the emergence of KPC-2 producing Klebsiella pneumoniae clonal complex 101 (ST101 and ST1789). BMC Microbiol 15:260. doi: 10.1186/s12866-015-0597-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Czobor I, Novais Â, Rodrigues C, Chifiriuc MC, Mihãescu G, Lazãr V, Peixe L. 2016. Efficient transmission of IncFIIY and IncL plasmids and Klebsiella pneumoniae ST101 clone producing OXA-48, NDM-1 or OXA-181 in Bucharest hospitals. Int J Antimicrob Agents 48:223–224. doi: 10.1016/j.ijantimicag.2016.05.004. [DOI] [PubMed] [Google Scholar]
- 40.Cubero M, Cuervo G, Dominguez MÁ, Tubau F, Martí S, Sevillano E, Gallego L, Ayats J, Peña C, Pujol M, Liñares J, Ardanuy C. 2015. Carbapenem-resistant and carbapenem-susceptible isogenic isolates of Klebsiella pneumoniae ST101 causing infection in a tertiary hospital. BMC Microbiol 15:177. doi: 10.1186/s12866-015-0510-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Andersen JM, Shoup M, Robinson C, Britton R, Olsen KE, Barrangou R. 2016. CRISPR diversity and microevolution in Clostridium difficile. Genome Biol Evol 8:2841–2855. doi: 10.1093/gbe/evw203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Dautzenberg MJ, Ossewaarde JM, de Kraker ME, van der Zee A, van Burgh S, de Greeff SC, Bijlmer HA, Grundmann H, Cohen Stuart JW, Fluit AC, Troelstra A, Bonten MJ. 2014. Successful control of a hospital-wide outbreak of OXA-48 producing Enterobacteriaceae in the Netherlands, 2009 to 2011. Euro Surveill 19:20723. doi: 10.2807/1560-7917.ES2014.19.9.20723. [DOI] [PubMed] [Google Scholar]
- 43.Di Pilato V, Arena F, Tascini C, Cannatelli A, De Angelis LH, Fortunato S, Giani T, Menichetti F, Rossolini GM. 2016. mcr-1.2, a new mcr variant carried on a transferable plasmid from a colistin-resistant KPC carbapenemase-producing Klebsiella pneumoniae strain of sequence type 512. Antimicrob Agents Chemother 60:5612–5615. doi: 10.1128/AAC.01075-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Clinical and Laboratory Standards Institute. 2015. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; approved standard-tenth edition. CLSI document M07-A10. Clinical and Laboratory Standards Institute, Wayne, PA. [Google Scholar]
- 45.Giani T, Pini B, Arena F, Conte V, Bracco S, Migliavacca R, Collective the AMCLI-CRE Survey Participants, Pantosti A, Pagani L, Luzzaro F, Rossolini GM. 2013. Epidemic diffusion of KPC carbapenemase-producing Klebsiella pneumoniae in Italy: results of the first countrywide survey, 15 May to 30 June 2011. Euro Surveill 18:20489. doi: 10.2807/ese.18.22.20489-en. [DOI] [PubMed] [Google Scholar]
- 46.Van Belkum A, Tassios PT, Dijkshoorn L, Haeggman S, Cookson B, Fry NK, Fussing V, Green J, Feil E, Gerner-Smidt P, Brisse S, Struelens M, European Society of Clinical Microbiology and Infectious Diseases (ESCMID) Study Group on Epidemiological Markers (ESGEM) . 2007. Guidelines for the validation and application of typing methods for use in bacterial epidemiology. Clin Microbiol Infect 13(Suppl 3):S1–S46. doi: 10.1111/j.1469-0691.2007.01732.x. [DOI] [PubMed] [Google Scholar]
- 47.Diancourt L, Passet V, Verhoef J, Grimont PA, Brisse S. 2005. Multilocus sequence typing of Klebsiella pneumoniae nosocomial isolates. J Clin Microbiol 43:4178–4182. doi: 10.1128/JCM.43.8.4178-4182.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sambrook J, Russell DW. 2001. Molecular cloning: a laboratory manual, 3rd ed Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY. [Google Scholar]
- 49.Bankevich A, Nurk S, Antipov D, Gurevich AA, Dvorkin M, Kulikov AS, Lesin VM, Nikolenko SI, Pham S, Prjibelski AD, Pyshkin AV, Sirotkin AV, Vyahhi N, Tesler G, Alekseyev MA, Pevzner PA. 2012. SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J Comp Biol 19:455–477. doi: 10.1089/cmb.2012.0021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Grissa I, Vergnaud G, Pourcel C. 2007. CRISPRFinder: a web tool to identify clustered regularly interspaced short palindromic repeats. Nucleic Acids Res 35:W52–W57. doi: 10.1093/nar/gkm360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Darling AE, Mau B, Perna NT. 2010. progressiveMauve: multiple genome alignment with gene gain, loss and rearrangement. PLoS One 5:e11147 doi: 10.1371/journal.pone.0011147. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.