Perinatal brain injury (PBI) is one of the most important causes of lifelong deficits in cognition, behavior, social interaction and motor skills, as well as epilepsy. PBI encompasses, but is not limited to, hypoxic-ischemic encephalopathy (HIE), intraventricular hemorrhage, periventricular leukomalacia and encephalopathy of prematurity. PBI affects both preterm and term neonates and is a reflection of diverse and complex etiologies and initiating insults, including prenatal genetic and environmental contributions, sentinel obstetrical and perinatal events, and drug exposures (Figure 1). Additionally, consequences of other illnesses and events such as neonatal sepsis, cardiac defects and infantile traumatic brain injury can result in central nervous system (CNS) damage that contributes to PBI. Thus, a large majority of infants who suffer PBI experience a complex blend of CNS insults that impact multiple molecular, cellular, microstructural and ultrastructural pathophysiological mechanisms and result in cumulative deficits.
Figure 1.
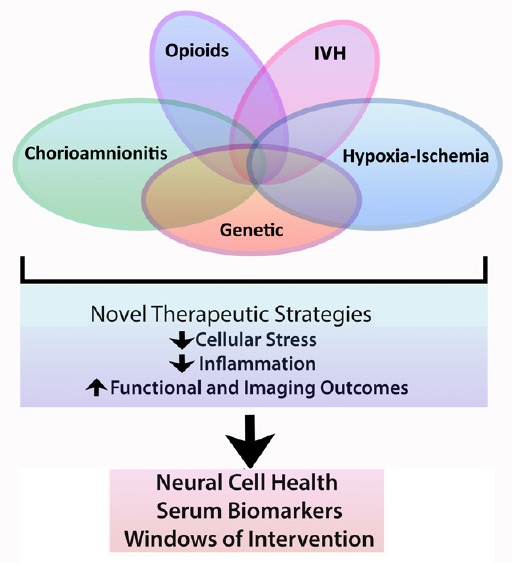
Perinatal brain injury is often multi-factorial.
IVH: Intraventricular hemorrhage.
The scope and challenges of diagnosing, treating and managing PBI throughout the lifespan are rapidly evolving, especially as mortality following neonatal insults decreases and the burden of chronic disability increases. Emerging preclinical, clinical and epidemiological evidence supports the concept that the pathophysiology of CNS injury following PBI reflects a combination of multiple insults including: 1) inflammation from prenatal infection and/or hypoxia-ischemia (HI); 2) individualized risk from genetic/congenital disorders; 3) acquired prenatal exposures to drugs, and toxins; 4) gut microbiota and nutritional status; and 5) postnatal stresses, such as sepsis and surgery. Interestingly, while the incidence of classic HIE is decreasing, rates of prematurity are not, with the opioid crisis likely contributing profoundly to surging numbers of preterm birth (National Center for Health Statistics). Accompanying this change in etiology of PBI, the distribution of children who develop cerebral palsy (CP) and neuropsychiatric conditions following early CNS insult is also shifting, with more children in the USA experiencing more severe motor involvement (Durkin, 2016).
PBI remains an important public health issue with few safe, effective treatments for a broad segment of neonates. Currently, half of HIE neonates fail to recover with therapeutic hypothermia, and its implementation in economically disadvantaged regions is limited. While clinical trials have shown progress for extremely preterm infants, neonates with severe intraventricular hemorrhage (IVH) continue to exhibit poor long-term outcomes. Additionally, the nature of how infection and placental inflammation catalyze PBI in preterm infants is incompletely understood and therapies are currently non-existent. Further, the clinical, pathophysiologic and mechanistic overlap between HIE, IVH and chorioamnionitis leading to PBI is under-recognized. Indeed, despite greater use of sophisticated perinatal imaging and genetic evaluations, many at risk neonates often suffer CNS injury from overlapping, incompletely identified, and poorly understood etiologies. With a greater appreciation of complex, intersecting mechanistic pathophysiology and shifting outcomes, appropriate treatment of infants with PBI necessitates that physicians and scientists alike alter their approaches to address the increasingly complicated medical, social, educational and societal needs of this highly vulnerable population. This changing landscape also demands clinicians and scientists respond by re-conceptualizing and transforming their approach to neurorepair and therapeutic strategies for PBI.
To minimize deficits and optimize outcomes for infants and children with PBI, new therapies are required to restore sensorimotor function, vision, hearing, behavior and cognition, and to limit epilepsy. Few novel therapies have directly addressed these cumulative, complex and compound deficits, particularly the functional pillars of cognition and behavior with motor impairment, however clinical trials such as PENUT (NCT#01378273; http://www.clinicaltrials.gov) and HEAL (NCT#02811263; http://www.clinicaltrials.gov) show promise. When one considers the intricacy of injury patterns, it becomes clear that an exceedingly large number of infants with PBI will not benefit from the current highly-specific clinical trials that target very select patient populations. To address the challenges and needs of neonates today, clinical practice and research directions need to shift focus and redefine strategies for neurorepair to include the broad, overlapping eitiologies and patterns of damage. Through 1) identifying liquid and neuroimaging biomarkers that define common endpoints of damage and repair, and 2) testing for broader use, emerging neuroreparative strategies using insights informed by molecular mechanisms, a much larger, at risk, neonatal cohort can be targeted (Lee et al., 2017; Graham et al., 2018). Indeed, contrary to prior dogma suggesting the developing brain exhibits a static response to early insults, neural repair involving neurons, glia and immune cells occurs both spontaneously in the developing CNS, and in response to specific therapies (Mazur et al., 2010; Jantzie et al., 2013; Robinson et al., 2018). Recent investigations on myelin plasticity and regeneration support novel approaches to neurorepair following PBI and reveal innovative avenues for functional recovery. By the very nature of ongoing neurodevelopment, and the incomplete and constantly evolving scaffold of an injured developing nervous system, the immature CNS differs from the mature brain, offering distinct and unique mechanisms of injury and opportunities for repair. Specifically, CNS development is characterized by regulated developmental expression of multiple neurotransmitter systems (i.e., glutamate, GABAergic and glycine), maturational trajectories of ion transporters (i.e., NKCC1 and KCC2), cell maturation throughout gestation (i.e., the oligodendroglial lineage and subplate neurons), and circumscribed elaboration of the neural immune system throughout pregnancy and the perinatal period. As a result, each neural cell type, as well as each receptor, transporter, and signal transduction pathway has its own maturational trajectory, and thus vulnerability to injury that requires repair after PBI. For these reasons, therapies that are appropriate for adults with CNS injury may be ineffective in infants, and vice versa.
An example of a partially successful therapy for PBI is hypothermia. Hypothermia has been used successfully to mitigate HIE in term infants in locations with advanced medical care. Despite this, there are remaining challenges. While hypothermia has multiple beneficial mechanisms of action including decreasing cell metabolism, oxidative stress and neural cell death, hypothermia only improves outcomes in a subset of neonates. Specifically, hypothermia is less effective in neonates with severe injury, has tenuous impact on infants subject to in utero infection, and is unproven for neonates not meeting strict inclusion criteria based on gestational age and degree of encephalopathy.
Erythropoietin (EPO) is another example of a therapy currently in large clinical trials, including those devoted to preterm infants with brain injury (i.e., PENUT NCT#01378273; http://www.clinicaltrials.gov) and term infants with encephalopathy (i.e., HEAL NCT#02811263; http://www.clinicaltrials.gov). With relatively abundant EPO receptor expression in the developing CNS, EPO's beneficial effects in the developing CNS are unique. EPO is putatively effective due to its action on multiple neural cell types (i.e., the subplate, developing oligodendrocytes and neurons). Moreover, EPO acts through diverse reparative mechanisms, including enhancing multiple stages of oligodendroglial lineage development, reducing calpain-mediated neural cell degeneration, and modulating circuit formation. However, EPO trials and studies have not been without challenges. Notably, while early trials have shown some promise, doses in those early studies were likely lower than needed for optimal repair. Repeated, high dosing has been emphasized in current trials and advanced preclinical work.
Given the lessons learned from hypothermia and EPO intervention trials, emerging preclinical data supports the use of multiple agents used in combination to achieve synergistic benefits. The multifaceted etiologies, cumulative insults, and composite neural cell lineages in need of repair that are all inherent in PBI make it the epitome of a disorder with a network of disease-relevant targets. Multiple therapeutic targets, addressed simultaneously or sequentially by treatments with overlapping mechanisms of action, may lead to superior efficacy. The concept that a cocktail of agents may be more effective than a single agent is gaining traction in neonatology and has shifted the focus of neurorepair (Gonzalez and Ferriero, 2008). General focus of current research aims to combine proven therapies like hypothermia with pharmacological interventions, and to test multiple pharmacotherapies together. This strategy capitalizes on overlapping intricate mechanisms of injury and repair that may respond to such combinatorial treatments. Indeed, as PBI causes diffuse CNS injury and deficits in multiple domains, repair may be widespread across multiple circuits and networks, and responsive to different therapeutic agents. In this context, selected use of well-characterized cell-based therapies, exosomes and nanoparticles will likely be beneficial (Kannan et al., 2012; Jantzie et al., 2018), in combination with more established interventions. Indeed, the use of nanoparticles targeted to microglia in the developing CNS may be a strategy that directly addresses the tremendous overlap and interdependence between neural and immune systems. In preclinical studies, mesenchymal stem cells were more effective when combined with EPO than when used in isolation (Min et al., 2013). Thus, as new treatments are translated from the bench into clinical trials, consideration should be given to adding to existing regimens.
As we move forward and consider new combinations for use with neonates with PBI from overlapping etiologies, EPO plus melatonin is a promising combination for both safety and efficacy. Both agents have been safely used in isolation in numerous NICU populations. Several mechanisms of action for each agent suggest that they may provide unique, highly effective synergy for repair (Jantzie and et al., 2018). For example, melatonin in addition to EPO may provide additional defense against oxidative and high-energy organelle stress, which is likely to be particularly important for cells with high bioenergetics demand such as oligodendrocytes with myelin and ependymal cells with motile cilia. In addition to reducing oxidative stress, both EPO and melatonin also reduce neuroinflammation. They both are also known to reduce micro-environmental toxins such as excess calpain activity. Finally, and specific to the developing CNS, they both support the genesis and maturation of multiple neural cells types over most of the perinatal period. Thus, the cocktail incorporating EPO plus melatonin capitalizes on overlapping complex mechanisms of injury and repair that may be particularly responsive to combinatorial therapies.
In conclusion, the cumulative effect of PBI results from a cascading impact on neurodevelopment and presents unique challenges to repair that are distinct from the mature CNS. While myelin, synapses and circuits are plastic and modifiable throughout the lifespan, there remain many distinctive hurdles to repair of the developing brain. Notably, individual predisposition and response to injury within the context of the constantly evolving, incompletely formed, damaged platform of the developing CNS demands individualized and precision medicine-diagnostic and trial design approaches over “one size fits all” strategies. Clinicians and researchers alike must adjust and expand their definition of neonatal repair and recovery to include the complex, overlapping etiologies of PBI and establish broad, cost-effective combination therapies of drugs, agents, biologic or cell-based therapeutics that address emerging issues in the treatment of PBI.
Footnotes
Copyright license agreement: The Copyright License Agreement has been signed by all authors before publication.
Plagiarism check: Checked twice by iThenticate.
Peer review: Externally peer reviewed.
Open peer reviewer: Isaac G. Onyango, Gencia Biotechnology, USA.
References
- Durkin MS. Socio-economic disparities and functional limitations of children with cerebral palsy. Dev Med Child Neurol. 2016;58:115. doi: 10.1111/dmcn.12821. [DOI] [PubMed] [Google Scholar]
- Gonzalez FF, Ferriero DM. Therapeutics for neonatal brain injury. Pharmacol Ther. 2008;120:43–53. doi: 10.1016/j.pharmthera.2008.07.003. [DOI] [PubMed] [Google Scholar]
- Graham EM, Everett AD, Delpech JC, Northington FJ. Blood biomarkers for evaluation of perinatal encephalopathy: state of the art. Curr Opin Pediatr. 2018;30:199–203. doi: 10.1097/MOP.0000000000000591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jantzie LL, Oppong AY, Conteh FS, Yellowhair TR, Kim J, Fink G, Wolin AR, Northington FJ, Robinson S. Repetitive neonatal erythropoietin and melatonin combinatorial treatment provides sustained repair of function deficits in a rat model of cerebral pals. Front Neurol. 2018;9:233. doi: 10.3389/fneur.2018.00233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jantzie LL, Miller RH, Robinson S. Erythropoietin signaling promotes oligodendrocyte development following prenatal systemic hypoxic-ischemic brain injury. Pediatr Res. 2013;74:658–667. doi: 10.1038/pr.2013.155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jantzie LL, Scafidi J, Robinson S. Stem cells and cell-based therapies for cerebral palsy: a call for rigor. Pediatr Res. 2018;83:345–355. doi: 10.1038/pr.2017.233. [DOI] [PubMed] [Google Scholar]
- Kannan S, Dai H, Navath RS, Balakrishnan B, Jyoti A, Janisse J, Romero R, Kannan RM. Dendrimer-based postnatal therapy for neuroinflammation and cerebral palsy in a rabbit model. Sci Transl Med. 2012;4:130ra146. doi: 10.1126/scitranslmed.3003162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee JK, Massaro AN, Northington FJ. The search continues: neuroprotection for all neonates with hypoxic-ischemic encephalopathy. J Thorac Dis. 2017;9:3553–3536. doi: 10.21037/jtd.2017.09.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- National Center for Health Statistics, final natality data. Retrieved on 2/15/18 from www.marchofdimes.org/peristats .
- Mazur M, Miller RH, Robinson S. Postnatal erythropoietin treatment mitigates neural cell loss after systemic prenatal hypoxic-ischemic injury. J Neurosurg Pediatr. 2010;6:206–221. doi: 10.3171/2010.5.PEDS1032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Min K, Song J, Kang JY, Ko J, Ryu JS, Kang MS, Jang SJ, Kim SH, Oh D, Kim MK, Kim SS, Kim M. Umbilical cord blood therapy potentiated with erythropoietin for children with cerebral palsy: a double-blind, randomized, placebo-controlled trial. Stem Cells. 2013;31:581–591. doi: 10.1002/stem.1304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson S, Corbett CJ, Winer JL, Chan LAS, Maxwell JR, Anstine CV, Yellowhair TR, Andrews NA, Yang Y, Sillerud LO, Jantzie LL. Neonatal erythropoietin mitigates impaired gait, social interaction and diffusion tensor imaging abnormalities in a rat model of prenatal brain injury. Exp Neurol. 2018;302:1–13. doi: 10.1016/j.expneurol.2017.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]


