Abstract
Background
There is a growing use of procalcitonin (PCT) to facilitate the diagnosis and management of severe sepsis. We investigated the impact of one to two PCT determinations on ICU day 1 on health-care utilization and cost in a large research database.
Methods
A retrospective, propensity score-matched multivariable analysis was performed on the Premier Healthcare Database for patients admitted to the ICU with one to two PCT evaluations on day 1 of ICU admission vs patients who did not have PCT testing.
Results
A total of 33,569 PCT-managed patients were compared with 98,543 propensity score-matched non-PCT patients. In multivariable regression analysis, PCT utilization was associated with significantly decreased total length of stay (11.6 days [95% CI, 11.4 to 11.7] vs 12.7 days [95% CI, 12.6 to 12.8]; 95% CI for difference, 1 to 1.3; P < .001) and ICU length of stay (5.1 days [95% CI, 5.1 to 5.2] vs 5.3 days [95% CI, 5.3 to 5.4]; 95% CI for difference, 0.1 to 0.3; P < .03), and lower hospital costs ($30,454 [95% CI, 29,968 to 31,033] vs $33,213 [95% CI, 32,964 to 33,556); 95% CI for difference, 2,159 to 3,321; P < .001). There was significantly less total antibiotic exposure (16.2 days [95% CI, 16.1 to 16.5] vs 16.9 days [95% CI, 16.8 to 17.1]; 95% CI for difference, –0.9 to 0.4; P = .006) in PCT-managed patients. Patients in the PCT group were more likely to be discharged to home (44.1% [95% CI, 43.7 to 44.6] vs 41.3% [95% CI, 41 to 41.6]; 95% CI for difference, 2.3 to 3.3; P = .006). Mortality was not different in an analysis including the 96% of patients who had an independent measure of mortality risk available (19.1% [95% CI, 18.7 to 19.4] vs 19.1% [95% CI, 18.9 to 19.3]; 95% CI for difference, –0.5 to 0.4; P = .93).
Conclusions
Use of PCT testing on the first day of ICU admission was associated with significantly lower hospital and ICU lengths of stay, as well as decreased total, ICU, and pharmacy cost of care. Further elucidation of clinical outcomes requires additional data.
Key Words: antibiotic use, cost of care, intensive care unit, procalcitonin, sepsis
Abbreviations: FDA, US Food and Drug Administration; LOS, length of stay; PCT, procalcitonin
FOR EDITORIAL COMMENT SEE PAGE 6
More than $20 billion was spent on sepsis care in 2011, making it the most expensive condition managed in US hospitals.1 Sepsis is increasingly common, and despite advances in antimicrobial and supportive therapy, it continues to be associated with a high risk for morbidity and mortality.2 The current consensus sepsis definition has been found to be both nonspecific and nonsensitive.3 Lack of a “gold standard” diagnostic test for sepsis has resulted in diagnostic dilemmas that may delay appropriate treatment and lead to poor outcomes.3, 4, 5 Unnecessary antibiotic use, as a consequence of sepsis overdiagnosis, is associated with increased length of stay (LOS), drug-related toxicities, Clostridium difficile infection, antimicrobial resistance, and health-care costs.6 Optimizing outcomes and minimizing resource utilization are contingent on timely and accurate discrimination between sepsis and mirroring conditions.
Procalcitonin (PCT), a precursor of calcitonin, has good specificity for a bacterial cause of suspected infection7, 8 and has been investigated for the diagnosis of bacterial infection, prognosis of sepsis, and management of antibiotic therapy.9, 10, 11, 12, 13, 14, 15 PCT guidance has been shown to reduce duration of antibiotic use in sepsis with no beneficial or detrimental impact on survival.10 Despite the emergence of PCT evaluation as a diagnostic criterion for sepsis in the 2012 Surviving Sepsis Campaign Guidelines, PCT testing has not been uniformly adopted, in part because of cost considerations.16 However, a Dutch study showed that PCT-based algorithms lowered hospital spending by €3503 per patient, saving the Netherlands health system an estimated €46 million annually.9
To provide current real-world information on health-care utilization and cost, we evaluated the impact of PCT testing performed on the first day of ICU admission in critically ill adult patients with suspected sepsis. An enhanced administrative database was used that included discharges from >550 US hospitals.
Patients and Methods
This study was a retrospective trial of patients aged ≥18 years admitted to an ICU with an admitting or discharge diagnosis code consistent with suspected or documented sepsis, septicemia, systemic inflammatory response syndrome, or shock (e-Table 1) between January 1, 2011, and May 31, 2014. Patients were grouped into those who did or did not receive 1 or 2 PCT determinations around the time of ICU admission. The initial PCT testing may occur in the ED or on the floor, but the PCT-tested patients were excluded if they received PCT determinations >2 days before or after ICU admission. The reason for PCT testing and the result were not specified in the administrative database that was used for the study. The database does not contain data to define the physician specialty who ordered the test or the patient characteristics that led to testing. The study design and methods were reviewed and approved by the Rush University Medical Center Office of Research Affairs per policy RA-IRB-118.
The data source was the Premier Healthcare Database, a large US hospital-based, service-level, all-payer administrative database. Utilization, cost, and outcome data were directly available in the database. Total antibiotic exposure was calculated as the total number of selected systemic antibiotics (e-Table 2) on a given day summed over the entire course of administration such that three antibiotics administered daily over 7 days would equal 21 days of exposure.
Because patients were not randomly assigned to PCT and non-PCT cohorts, differences in patient characteristics and severity may lead to bias in the estimated effect of PCT. To address this issue, 1:3 propensity score matching was implemented by using nearest neighbor matching to account for differences in the comparison groups.17 The analysis was limited to patients with a PCT determination within 1 day of ICU admission. Despite this approach, there were some control subjects with very short ICU stays who were discharged before the time that the PCT determination was run on the matched case. These control subjects were dropped because the shorter stays could not be attributable to a “treatment effect” (relating to whether a PCT determination was or was not obtained). Propensity scores were determined by using a logistic regression with receipt of PCT testing as the dependent variable and the following covariates: patient demographic characteristics (age categories, sex, race, ethnicity, health-care coverage type, and year of hospitalization), hospital characteristics (urbanicity, teaching status, bed size, and region), and patient’s clinical characteristics (admission type, admission source, number of types of antibiotics received on the first ICU day, dialysis on or before the first ICU day, ventilator use on or before the first ICU day, use of vasopressors or inotropes on the first ICU day, previous hospitalization within 30 days, and presence of the 10 most frequent admission diagnoses). After propensity score matching, the balancing properties of the matching covariates were examined through a review of the standardized differences between the PCT group and the non-PCT group. Standardized differences <0.1 were considered negligible.18, 19 Descriptive statistics of the outcomes were generated, and bivariate analysis was implemented on the propensity score-matched sample. The analytic and data processing plan are depicted in e-Figure 1.
Data measured on a continuous scale were summarized by using means; 95% CIs of the mean for the outcomes are also reported. Categorical data are expressed as counts and percentage of patients within each category. Bivariate analysis was performed to compare variables between those patients receiving PCT testing and those who did not. A t test was used to evaluate continuous variables, and χ2 tests were used for categorical variables. P values <.05 were considered statistically significant.
Multivariable regression models were used to assess the association between PCT test and outcomes in the propensity score-matched sample. Control variables included the following: patient demographic characteristics (age categories, sex, race, ethnicity, health-care coverage type, and year of hospitalization), hospital characteristics (urbanicity, teaching status, bed size, and region), and patient’s clinical characteristics (admission type, admission source, number of different types of antibiotics received on the first ICU day, dialysis on or before the first ICU day, ventilator use on or before the first ICU day, use of vasopressors or inotropes on the first ICU day, previous hospitalization within 30 days, and presence of the 10 most frequent discharge diagnoses such as septicemia, other lung diseases, pneumonia with organism unspecified, heart failure, acute myocardial infarction, replacement and graft complication, other surgical complications that were not elsewhere classified, other bacterial pneumonia, acute renal failure, chronic bronchitis, pneumonitis due to solids and liquids, cardiac dysrhythmias, other forms of chronic ischemic heart disease, diabetes mellitus, and complications of medical care that were not elsewhere classified).
A supplemental analysis included the CareScience Mortality Risk Model score, a predictive value derived from clinical and demographic characteristics and available for those not transferred to another inpatient facility.20 This score is calculated based on diagnosis codes and is available in the Premier Healthcare Database. Because we conducted a retrospective observational study using a hospital administrative database, the information needed to calculate The Acute Physiology and Chronic Health Evaluation Score (APACHE) or Simplified Acute Physiology Score (SAPS) (eg, vital signs, laboratory values) was not necessarily available, and these severity-of-illness scores could therefore not be determined. The missing values of the CareScience Mortality Risk Model score were due to the fact that some patients were transferred to other acute care facilities, and their mortality was not known. Because the basic assumption for imputation is that the missing values should be missing at random, the imputation may bring in bias for the group of patients transferred to another facility.
Generalized estimating equation models were used to model LOS, costs, and days of antibiotic therapy. These models with an exchangeable correlation structure generated robust variance estimates by adjusting for the within-hospital correlation. Logistic regression was used to model discharge status. The recycled prediction method21 was used to calculate the adjusted outcome values. Recycled prediction method is a way to calculate the predicted outcomes based on regression estimates. First, all patients were assumed to receive PCT, and the adjusted outcomes were predicted based on the regression coefficients, holding all covariates at their actual values. Second, predication was made assuming all patients were in the non-PCT cohort. The mean differences in the predicted values for the two hypothetical groups (PCT and non-PCT) were examined and the 95% CIs calculated by using the bootstrapping method. All statistical analyses were performed by using SAS version 9.2 (SAS Institute, Inc).
Results
This study evaluated 15,041,827 patients in the Premier Healthcare Database universe of patients; 730,088 had a potential sepsis, systemic inflammatory response syndrome, septicemia, or shock-related diagnosis on admission or discharge and were cared for in the ICU (Fig 1). This cohort was further stratified according to whether one or two PCT levels were obtained within the first day of ICU admission (n = 34,989) vs the non-PCT-managed cohort of ICU patients (n = 671,473). To reduce potential bias, a 1:3 propensity matching was performed. After matching, there were 98,543 non-PCT-managed patients at 570 hospitals and 33,569 PCT-managed patients at 286 hospitals. The data cleaning step to correct for the patients with extremely short stays affected 3% of control subjects. In most of these cases, the net effect was that <2% of cases had two or one rather than three matched control subjects. In 1% of case subjects, all three control subjects were affected; these cases were dropped. Even in this small number of affected patients, the numerical effect cannot be large because of the very short stays involved.
Figure 1.
Graphic illustration of the study flow from the Premier Healthcare Database universe of patients sorted according to ICU stay and selected sepsis, systemic inflammatory response syndrome, septicemia, and shock diagnoses and further sorted according to the use of PCT during the admission. Subsequent sorting used PCT testing on day 1 of ICU admission and a propensity score to match the study populations. PCT = procalcitonin.
There was no difference between the groups in the percentage of admissions from rural and urban hospitals, teaching and nonteaching hospitals, and bed size of the included hospitals (e-Table 3). The study populations were well matched for age, sex, race, ethnicity, health-care insurance status, and admission type (Table 1). The admission diagnosis, when present, was also similar between the two groups. No significant differences were noted in the number of systemic antibiotics administered to patients on the first day of their ICU stay or in the use of mechanical ventilatory support on or before the first day of ICU stay. There was no statistically significant difference in the use of vasopressors and/or inotropic medications on the first day of ICU stay for the two groups of patients.
Table 1.
Matched Patient Characteristics
| Variable | Any PCT Test | No PCT Test | PCT vs No PCT, P Value | Standardized Differences |
|---|---|---|---|---|
| No. of discharges | 33,569 | 98,543 | … | … |
| Age, y | .424 | |||
| Mean | 66.29 | 66.37 | … | 0.005 |
| SD | 16.19 | 16.15 | … | … |
| Variable | No. of Discharges (%) | No. of Discharges (%) | P Value | Standardized Differences |
|---|---|---|---|---|
| Sex | .666 | |||
| Female | 16,649 (49.6) | 48,599 (49.3) | … | 0.006 |
| Male | 16,917 (50.4) | 49,934 (50.7) | … | 0.006 |
| Unknown | 3 (0) | 10 (0) | … | 0.001 |
| Race | .34 | |||
| Black | 3,813 (11.4) | 11,048 (11.2) | … | 0.005 |
| Hispanic | 40 (0.1) | 130 (0.1) | … | 0.004 |
| Other | 5,666 (16.9) | 17,025 (17.3) | … | 0.011 |
| White | 24,050 (71.6) | 70,340 (71.4) | … | 0.006 |
| Ethnicity | .045 | |||
| Hispanic | 1,478 (4.4) | 4,134 (4.2) | … | 0.010 |
| Non-Hispanic | 28,452 (84.8) | 83,334 (84.6) | … | 0.005 |
| Unknown | 3,639 (10.8) | 11,075 (11.2) | … | 0.013 |
| Health-care coverage type | > .999 | |||
| Commercial | 5,448 (16.2) | 16,030 (16.3) | … | 0.001 |
| Medicaid | 3,302 (9.8) | 9,540 (9.7) | … | 0.005 |
| Medicare | 22,027 (65.6) | 64,976 (65.9) | … | 0.007 |
| Charity/indigent/self-pay | 1,728 (5.1) | 4,935 (5.0) | … | 0.006 |
| Other | 1,064 (3.2) | 3,062 (3.1) | … | 0.004 |
| Admission type | ||||
| Elective | 1,614 (4.8) | 4,735 (4.8) | .996 | 0.000 |
| Emergency | 27,912 (83.1) | 81,894 (83.1) | … | 0.001 |
| Other/unknown | 27 (0.1) | 81 (0.1) | … | 0.001 |
| Trauma center/urgent | 4,016 (12.0) | 11,833 (12.0) | … | 0.001 |
| Discharge status | ||||
| Expired | 6,469 (19.3) | 17,948 (18.2) | < .001 | 0.027 |
| Home | 14,715 (43.8) | 40,789 (41.4) | … | 0.049 |
| Other/unknown | 336 (1.0) | 935 (0.9) | … | 0.005 |
| Transferred to another health-care facility | 12,049 (35.9) | 38,871 (39.4) | … | 0.073 |
| Admission diagnosis | ||||
| No admission diagnosis | 5,965 (17.8) | 16,508 (16.8) | < .001 | 0.027 |
| Septicemia | 4,432 (13.2) | 12,859 (13.0) | .471 | 0.005 |
| General symptoms | 3,876 (11.5) | 11,537 (11.7) | .427 | 0.005 |
| Symptoms involving respiratory system and other chest symptoms | 3,670 (10.9) | 10,832 (11.0) | .763 | 0.002 |
| Pneumonia, organism unspecified | 2,680 (8.0) | 8,071 (8.2) | .231 | 0.008 |
| Other lung diseases | 2,262 (6.7) | 6,669 (6.8) | .854 | 0.001 |
| Other symptoms involving abdomen and pelvis | 771 (2.3) | 2,307 (2.3) | .642 | 0.003 |
| Cardiac dysrhythmias | 552 (1.6) | 1,552 (1.6) | .38 | 0.006 |
| Heart failure | 541 (1.6) | 1,623 (1.6) | .659 | 0.003 |
| Gastrointestinal hemorrhage | 362 (1.1) | 1,049 (1.1) | .831 | 0.001 |
| Gastrointestinal system symptoms | 414 (1.2) | 1,220 (1.2) | .946 | 0.000 |
| Certain adverse effects not elsewhere classified | 491 (1.5) | 1,373 (1.4) | .352 | 0.006 |
| Hypotension | 462 (1.4) | 1,283 (1.3) | .303 | 0.006 |
| Acute myocardial infarction | 299 (0.9) | 884 (0.9) | .915 | 0.001 |
| Other urinary tract disorder | 424 (1.3) | 1,227 (1.2) | .798 | 0.002 |
| No. of different types of antibiotics received on the first ICU day | .895 | |||
| 0 | 2,956 (8.8) | 8,602 (8.7) | … | 0.003 |
| 1 | 5,726 (17.1) | 16,868 (17.1) | … | 0.002 |
| ≥2 | 24,887 (74.1) | 73,073 (74.2) | … | 0.002 |
| Any dialysis on or before ICU day 1 | 980 (2.9) | 2,850 (2.9) | .797 | 0.002 |
| Any ventilator use on or before ICU day 1 | 11,094 (33.0) | 32,733 (33.2) | .571 | 0.004 |
| Any use of vasopressors or inotropes on ICU day 1 | 10,425 (31.1) | 30,461 (30.9) | .622 | 0.003 |
PCT = procalcitonin.
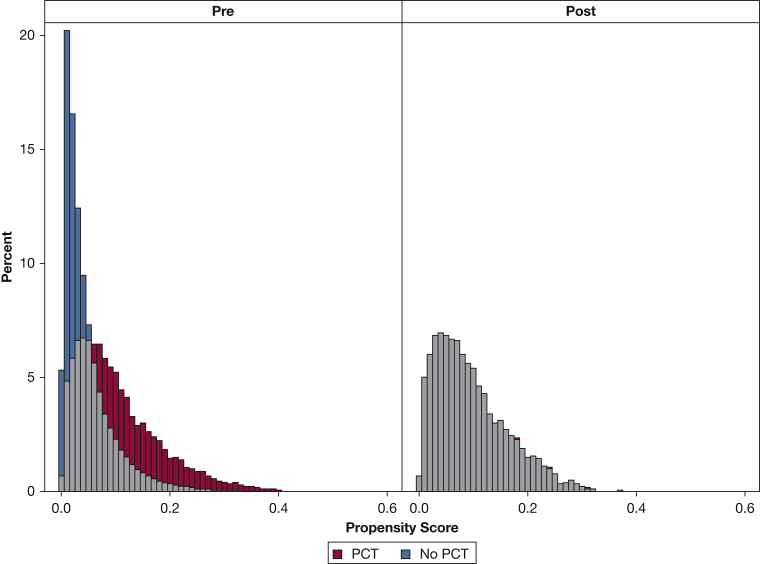
Figure 2 displays the success of the propensity score adjustment in the matched outcomes analysis without regression adjustment. In unadjusted analysis, PCT-guided care was associated with a significantly shorter hospital and ICU LOS (Table 2). PCT-guided care was also associated with significantly decreased total hospital, room and board, pharmacy, antibiotic, and laboratory costs. Antibiotic exposure was lower in the PCT-managed patients. More PCT-managed patients were discharged home, but inpatient mortality was slightly higher in this group. The PCT group was also less likely to be transferred to acute care, skilled nursing, intermediate care, or long-term care facilities.
Figure 2.
Graphic demonstration of assessment of the success of propensity score matching between the PCT-guided population and the non-PCT-guided study population. See Figure 1 legend for expansion of abbreviation.
Table 2.
Matched Outcomes Without Regression Adjustment
| Variable | 1 to 2 PCT Assessments Within 1 Day of ICU Admission (n = 33,569 discharges) |
No PCT (n = 98,543 discharges) |
Difference |
PCT vs No PCT, P Value | |||
|---|---|---|---|---|---|---|---|
| Mean | 95% CI | Mean | 95% CI | Mean | 95% CI | ||
| LOS, d | 10.5 | 10.4 to 10.6 | 12.8 | 12.7 to 12.9 | −2.3 | −2.4 to −2.2 | < .001 |
| ICU LOS, d | 4.8 | 4.8 to 4.9 | 5.6 | 5.5 to 5.6 | −0.7 | −0.8 to −0.7 | < .001 |
| Total cost, $ | 25,513 | 25,163 to 25,864 | 33,164 | 32,898 to 33,429 | −7,650 | −8,090 to −7,211 | < .001 |
| ICU cost, $ | 16,814 | 16,560 to 17,069 | 21,630 | 21,429 to 21,832 | −4,816 | −5,140 to −4,492 | < .001 |
| Pharmacy cost, $ | 3,866 | 3,694 to 4,038 | 4,589 | 4,494 to 4,684 | −723 | −920 to −527 | < .001 |
| Antibiotic cost, $ | 832 | 768 to 897 | 936 | 911 to 961 | −103 | −173 to −34 | < .001 |
| Laboratory cost, $ | 1457 | 1,442 to 1,472 | 1,710 | 1,697 to 1,724 | −253 | −274 to −233 | < .001 |
| Room and board cost, $ | 12,212 | 12,053 to 12,370 | 15,379 | 15,254 to 15,503 | −3,167 | −3,369 to −2,965 | < .001 |
| Total antibiotic exposurea | 14.9 | 14.7 to 15.0 | 17.3 | 17.2 to 17.4 | −2.4 | −2.6 to −2.3 | < .001 |
| Inpatient mortality | 19.3% | 18.8 to 19.7 | 18.2% | 18.0 to 18.5 | 1.1% | 0.6 to 1.5 | < .001 |
| Home | 43.8% | 43.3 to 44.4 | 41.4% | 41.1 to 41.7 | 2.4% | 1.8 to 3.1 | < .001 |
| Hospice | 6.3% | 6.1 to 6.6 | 6.4% | 6.2 to 6.6 | −0.1% | −0.4 to 0.2 | .617 |
| Other | 1.7% | 1.6 to 1.9 | 1.6% | 1.5 to 1.7 | 0.1% | 0 to 0.3 | .122 |
| Transfer to SNF/ICF/LTC | 22.7% | 22.2 to 23.1 | 25.2% | 24.9 to 25.4 | −2.5% | −3.0 to −2.0 | < .001 |
| Transfer to acute care | 6.2% | 5.9 to 6.4 | 7.2% | 7.0 to 7.4 | −1.0% | −1.3 to −0.7 | < .001 |
ICF = intermediate nursing facility; LOS = length of stay; LTC = long-term care facility; SNF = skilled nursing facility. See Table 1 legend for expansion of other abbreviation.
Total antibiotic exposure comprises both total number of systemic antibiotics administered and duration of administration.
Multivariable regression adjustment was performed to further address residual imbalances between patient and hospital characteristics. This analysis generally substantiated and refined the findings of the unadjusted matched analysis (Table 3). PCT-managed patients averaged 1.2 fewer days in the hospital (11.6 vs 12.7 days [95% CI for difference, 1.0 to 1.3]), 0.2 fewer ICU days (5.1 vs 5.2 days [95% CI for difference, 0.1 to 0.3]), and 0.7 day less of antibiotic exposure (16.2 vs 16.9 antibiotic days [95% CI for difference, 0.4 to 0.9]). In the PCT-managed group, total hospital costs were $2,759 less ($30,454 vs $33,213 [95% CI for difference, 2,156 to 3,321]), ICU costs were $1,310 less ($20,155 vs $21,465 [95% CI for difference, 847 to 1,702]), and pharmacy costs were $331 less ($4,238 vs $4,568 [95% CI for difference, 99 to 488]). Laboratory costs of the PCT-managed patients were $81 greater ($1,807 vs $1,726 [95% CI for difference, 51 to 114]). PCT-managed ICU patients were more commonly discharged home (44.1% vs 41.3% [95% for difference, 2.3 to 3.3]), but inpatient mortality was 0.7% greater (19% vs 18.3% [95% CI for difference, 0.3 to 1.2]).
Table 3.
Matched, Regression Adjusted Outcomes (N = 132,112)
| Variable | PCT |
No PCT |
Difference |
P Value | |||
|---|---|---|---|---|---|---|---|
| Mean of Adjusted Value | 95% CI | Mean of Adjusted Value | 95% CI | Mean of Adjusted Value | 95% CI | ||
| Total LOS | 11.6 | 11.4 to 11.7 | 12.7 | 12.6 to 12.8 | −1.2 | −1.3 to −1.0 | < .001 |
| ICU LOS | 5.1 | 5.1 to 5.2 | 5.3 | 5.3 to 5.4 | −0.2 | −0.3 to −0.1 | .031 |
| Total cost, $ | 30,454 | 29,968 to 31,033 | 33,213 | 32,964 to 33,556 | −2,759 | −3,321 to −2,156 | < .001 |
| ICU cost, $ | 20,155 | 20,625 to 19,798 | 21,465 | 21,270 to 21,710 | −1,310 | −1,702 to −847 | < .001 |
| Pharmacy cost, $ | 4,238 | 4,119 to 4,453 | 4,568 | 4,480 to 4,678 | −331 | −488 to −99 | .002 |
| Antibiotic cost, $ | 882 | 854 to 948 | 952 | 931 to 980 | −70 | −105 to 4 | .074 |
| Laboratory cost, $ | 1,807 | 1,778 to 1,839 | 1,726 | 1,710 to 1,744 | 81 | 51 to 114 | .002 |
| Total antibiotic exposurea | 16.2 | 16.1 to 16.5 | 16.9 | 16.8 to 17.1 | −0.7 | −0.9 to −0.4 | .006 |
| Discharged to home | 44.1% | 43.7 to 44.6 | 41.3 | 41.0 to 41.6 | 2.8 | 2.3 to 3.3 | .012 |
| Discharged to hospice | 6.3% | 6.0 to 6.5 | 6.4 | 6.3 to 6.6 | −0.2 | −0.4 to 0.0 | < .001 |
| Discharged to other | 1.8% | 1.6 to 1.9 | 1.6 | 1.5 to 1.7 | 0.1 | 0.0 to 0.3 | .779 |
| Transfer to SNF/ICF/LTC | 22.6% | 22.2 to 23.0 | 25.2 | 24.9 to 25.5 | −2.6 | −3.1 to −2.2 | < .001 |
| Transfer to acute care | 6.3% | 6.0 to 6.5 | 7.2 | 7.0 to 7.3 | −0.9 | −1.2 to −0.6 | < .001 |
| Inpatient mortalityb | 19.0% | 18.6 to 19.4 | 18.3 | 18.0 to 18.5 | 0.7 | 0.3 to 1.2 | .001 |
| Inpatient mortalityc | 19.1% | 18.7 to 19.4 | 19.1 | 18.9 to 19.3 | −0.0 | −0.5 to 0.4 | .93 |
95% CIs were calculated using bootstrap method with 500 replications. See Table 1 and 2 legends for expansion of abbreviations.
Total antibiotic exposure comprises both total number of systemic antibiotics administered and duration of administration.
All subjects.
Analysis incorporating the CareScience Mortality Risk Model score; N = 127,809.
A sensitivity analysis limited to survivors was performed and yielded effectively identical results for utilization and cost as the all-patient analysis. An additional analysis added the CareScience Mortality Risk Model score (available for the 127,809 [96%] patients not transferred to another inpatient facility) to the previous multivariable regression. In this analysis, the observed mortality difference was no longer present (19.1% vs 19.1% [95% CI for difference, −0.5 to 0.4]).
Discussion
PCT testing on the first day of ICU care has received US Food and Drug Administration (FDA) clearance as a tool to assist in the identification of patients with a high likelihood of sepsis. A growing number of hospitals have adopted PCT testing to aid in the diagnosis and management of patients with potential sepsis. This study evaluated the large Premier Healthcare Database, which contained data from >619 million patient encounters (inpatient and outpatient) or approximately one in every five discharges in the nation over nearly 3.5 years to assess the impact of first ICU day PCT testing on health-care utilization and cost of care in adults with potential sepsis. Although this sample was not random, preliminary comparisons between patient and hospital characteristics for the hospitals that submit data to Premier and those of the probability sample of hospitals and patients selected for the National Hospital Discharge Survey suggest that the patient populations were similar regarding patient age, sex, LOS, mortality, primary discharge diagnosis, and primary procedure groups. The Premier Healthcare Database seems to reflect the landscape of US hospitals, which are primarily smaller nonteaching hospitals, but it also includes larger and academic medical centers, which comprise 5% and 7%, respectively, of the nation’s 5,627 hospitals in the 2016 American Hospital Association statistics.22 A comparison was made of the hospitals that comprise the Premier Hospital Database vs the American Hospital Association Database (e-Table 4); the results support the generalizability of the findings in this article because the study population has similarities to the overall composition of US hospitals.
To date, there is no approved “septic test” that identifies patients with sepsis with a sufficiently high likelihood to make it clinically useful.23 PCT has been used, predominantly in Europe, to assist in identifying patients with respiratory infections who would benefit from antibiotic treatment.6, 8, 23, 24, 25, 26, 27 Studies have also suggested a potential benefit of using PCT testing to help with ICU prognosis and antibiotic stewardship related to decreasing antibiotic exposure.10, 24, 25, 26, 27, 28, 29 Neither of these uses has gained FDA approval.
The present study showed that the use of PCT testing on the first day of ICU care was associated with significantly lower hospital and ICU LOS. There was also a significant difference in the total hospital, ICU, and pharmacy costs when day 1 PCT testing in the ICU was used in critically ill adult patients. The PCT-managed patients had less total antibiotic exposure but higher laboratory costs. The all-patient analysis revealed a statistically significant, but only slightly higher (0.7%), risk of mortality that was not observed in an enhanced risk-adjusted analysis that included 96% of patients. This outcome is consistent with numerous prospective studies involving thousands of patients that have found no difference in mortality or other clinical outcomes using PCT guidance.24, 25, 26, 28, 30
The interpretation of the latter findings is not readily apparent. One would certainly expect that more rapid identification and management of patients with sepsis would have also translated into improved survival in addition to improved health-care utilization and cost of care.16, 23, 31 Unfortunately, we cannot control for differences in severity of illness or chronic health conditions between the two cohorts, which may have affected survival. Similarly, we lacked information on subsequent use of vasopressors, ventilator support, renal replacement therapy, and other aspects of support that may influence outcome of the patient with sepsis.16, 23, 31, 32, 33, 34, 35 Another important factor that affects overall survival is the limitation and/or withdrawal of care, which can have a significant effect on overall outcome and does not necessarily reflect the adequacy of sepsis management but rather patient/family preferences or desires regarding end-of-life care.16
The present study is important in the validation of PCT testing’s ability to favorably affect the outcome of critically ill patients when used according to the FDA-cleared guideline. The study population was large and extremely diverse. The use of PCT was evaluated over nearly a 3.5-year period and in a variety of clinical settings, including academic and nonacademic institutions. The cost savings were real and consequential, exceeding the potential increased costs of laboratory testing associated with PCT testing on ICU admission.
The mechanisms resulting in the improved health-care utilization and cost of care are not immediately evident from this administrative database. It seems likely that PCT is being used both to help rule-out and rule-in sepsis on ICU admission. The former would directly save resources, whereas the latter would translate into earlier administration of appropriate antibiotic therapy and source control (ie, key components of sepsis management).16, 23 In addition, earlier recognition of the patient with sepsis may allow for earlier use of the appropriate resuscitation and management protocols (eg, the Surviving Sepsis Campaign Management Guideline), which have also been associated with improved outcome for patients with sepsis.16, 23, 31, 32, 33, 34, 35 Improved management using early effective appropriate antibiotics, source control, and fluid/vasopressor administration would be expected to lead to earlier patient improvement, reduced LOS, and decreased health-care utilization and cost.16, 31, 35 These outcomes were noted in this large patient population from a diverse group of hospitals and patient care settings.
The suspected timely and improved sepsis management in the PCT-managed patients was associated with decreased hospital, ICU, and pharmacy costs. These patients also required less antibiotic treatment as evidenced by decreased antibiotic exposure, which may have future implications on the ever-growing problem of bacterial resistance and development of multidrug-resistant organisms. Because patients with PCT testing after the first day of ICU care were not included in this database, it is unlikely that PCT was used to guide early antibiotic discontinuation. The higher laboratory costs (<$100) related to the use of PCT in the management of these critically ill patients may reflect the additional cost of PCT testing.
The major limitations of the present study include the retrospective nature of the analysis and the lack of clinical and certain other data. The identification of PCT testing was based on a text string search in the hospital billing records. Because hospitals recorded the cost of laboratory tests in various ways, we might not have captured all PCT tests in the hospitals. Second, as in all retrospective observational studies, the PCT and non-PCT cohorts were not randomly assigned. Therefore, there is a potential bias due to the difference in patient severity and unmeasured confounders. Although propensity score matching and multivariable analysis were implemented to control for the patient’s severity as much as possible, unobservable characteristics may still exist that were not controlled for in the analysis. The database lacked specific information as to whether there were hospital protocols governing the use of PCT in patient management or ICU admission. Similarly, there were no data regarding confirmatory culture results, the presence and site of actual infection, the adequacy of initial empiric antibiotic therapy, and/or source control measures, which may all affect outcome.16, 23, 32, 33, 34 There were also no data on subsequent need for vasopressor/inotropic therapy, ventilator support, renal replacement therapy, or the severity of illness, comorbidities, or limitations on aggressive care between the two groups, which could have affected treatment decisions and outcomes. All of these factors may influence health-care utilization and outcomes in severe sepsis and septic shock. Not all hospitals had onsite PCT testing available, and we do not have information protocols or decision processes that determined which patients would undergo PCT testing.
Conclusions
This evaluation of adult patients from US hospitals in the Premier Healthcare Database suggests that use of PCT testing on ICU admission was associated with a significant decrease in hospital and ICU LOS, less systemic antibiotic exposure, a slight increase in laboratory costs, and decreased hospital, ICU, and pharmacy costs. The significance and mechanisms surrounding the observed clinical outcomes warrant additional evaluation.
Acknowledgments
Author contributions: Data acquisition and analysis were performed by Z. C., C. L., and S. B. R. Z. C. had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis, including and especially any adverse effects. Data discussion, manuscript preparation, manuscript review was performed by all authors.
Financial/nonfinancial disclosures: The authors have reported to CHEST the following: R. A. B. has received advisory board fees from bioMérieux USA, Roche Diagnostics, and ThermoFisher Scientific; he has also been the recipient of research support and speaking honoraria from bioMérieux USA. Z. C., C. L., and S. B. R. are employees of Premier Research Services, which received funding from bioMérieux USA to conduct this study. S. A. B. is an employee of bioMérieux USA. None declared (S. S. K.).
Role of sponsors: bioMérieux USA provided financial support for the data acquisition and analysis performed by Premier Services, Inc. Although the funding for the project was provided by bioMérieux USA, they did not influence or control data analysis or the content of this manuscript.
Additional information: The e-Figure and e-Tables can be found in the Supplemental Materials section of the online article.
Footnotes
FUNDING/SUPPORT: This study was funded by bioMérieux USA.
Supplementary Data
References
- 1.Torio CM, Andrews RM. National Inpatient Hospital Costs: The Most Expensive Conditions by Payer, 2011. HCUP Statistical Brief #160. Rockville, MD: Agency for Healthcare Research and Quality; August 2013. [PubMed]
- 2.Vincent J.L., Rello J., Marshall J. International study of the prevalence and outcomes of infection in intensive care units. JAMA. 2009;302(21):2323–2329. doi: 10.1001/jama.2009.1754. [DOI] [PubMed] [Google Scholar]
- 3.Kaukonen K.M., Bailey M., Pilcher D. Systemic inflammatory response syndrome criteria in defining severe sepsis. N Engl J Med. 2015;372(17):1629–1638. doi: 10.1056/NEJMoa1415236. [DOI] [PubMed] [Google Scholar]
- 4.Kumar A., Roberts D., Wood K.E. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med. 2006;34(6):1589–1596. doi: 10.1097/01.CCM.0000217961.75225.E9. [DOI] [PubMed] [Google Scholar]
- 5.Zilberberg M.D., Shorr A.F., Micek S.T. Multi-drug resistance, inappropriate initial antibiotic therapy and mortality in Gram-negative severe sepsis and septic shock: a retrospective cohort study. Crit Care. 2014;18(6):596. doi: 10.1186/s13054-014-0596-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Schuetz P., Chiappa V., Briel M., Greenwald J.L. Procalcitonin algorithms for antibiotic therapy decisions: a systematic review of randomized controlled trials and recommendations for clinical algorithms. Arch Intern Med. 2011;171(15):1322–1331. doi: 10.1001/archinternmed.2011.318. [DOI] [PubMed] [Google Scholar]
- 7.Assicot M., Gendrel D., Carsin H. High serum procalcitonin concentrations in patients with sepsis and infection. Lancet. 1993;341(8844):515–518. doi: 10.1016/0140-6736(93)90277-N. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Simon L., Gauvin F., Amre D.K. Serum procalcitonin and C-reactive protein levels as markers of bacterial infection: a systematic review and meta-analysis. Clin Infect Dis. 2004;39(2):206–217. doi: 10.1086/421997. [DOI] [PubMed] [Google Scholar]
- 9.Kip M.M., Kusters R., Izerman M.J., Steuten L.M. A PCT algorithm for discontinuation of antibiotic therapy is a cost-effective way to reduce antibiotic exposure in adult intensive care patients with sepsis. J Med Econ. 2015;18(11):944–953. doi: 10.3111/13696998.2015.1064934. [DOI] [PubMed] [Google Scholar]
- 10.Bouadma L., Luyt C.E., Tubach F. Use of procalcitonin to reduce patients' exposure to antibiotics in intensive care units (PRORATA trial): a multicentre randomised controlled trial. Lancet. 2010;375(9713):463–474. doi: 10.1016/S0140-6736(09)61879-1. [DOI] [PubMed] [Google Scholar]
- 11.Hochreiter M., Kohler T., Schweiger A.M. Procalcitonin to guide duration of antibiotic therapy in intensive care patients: a randomized prospective controlled trial. Crit Care. 2009;13(3):R83. doi: 10.1186/cc7903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nobre V., Harbarth S., Graf J.D. Use of procalcitonin to shorten antibiotic treatment duration in septic patients: a randomized trial. Am J Respir Crit Care Med. 2008;177(5):498–505. doi: 10.1164/rccm.200708-1238OC. [DOI] [PubMed] [Google Scholar]
- 13.Christ-Crain M., Jaccard-Stolz D., Bingisser R. Effect of procalcitonin-guided treatment on antibiotic use and outcome in lower respiratory tract infections: cluster-randomised, single-blinded intervention trial. Lancet. 2004;363(9409):600–607. doi: 10.1016/S0140-6736(04)15591-8. [DOI] [PubMed] [Google Scholar]
- 14.Shehabi Y., Sterba M., Garrett P.M. Procalcitonin algorithm in critically ill adults with undifferentiated infection or suspected sepsis. A randomized controlled trial. Am J Respir Crit Care Med. 2014;190(10):1102–1110. doi: 10.1164/rccm.201408-1483OC. [DOI] [PubMed] [Google Scholar]
- 15.Schuetz P., Christ-Crain M., Thomann R. Effect of procalcitonin-based guidelines vs standard guidelines on antibiotic use in lower respiratory tract infections: the ProHOSP randomized controlled trial. JAMA. 2009;302(10):1059–1066. doi: 10.1001/jama.2009.1297. [DOI] [PubMed] [Google Scholar]
- 16.Dellinger R.P., Levy M.M., Rhodes A. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med. 2013;41(2):580–637. doi: 10.1097/CCM.0b013e31827e83af. [DOI] [PubMed] [Google Scholar]
- 17.Rosenbaum P.R., Rubin D.B. The central role of the propensity score in observational studies for causal effects. Biometrika. 1983;70(1):41–55. [Google Scholar]
- 18.Austin P.C. Balance diagnostics for comparing the distribution of baseline covariates between treatment groups in propensity-score matched samples. Stat Med. 2009;28(25):3083–3107. doi: 10.1002/sim.3697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Austin P.C. Using the standardized difference to compare the prevalence of a binary variable between two groups in observational research. Communications in Statistics Simulation Computation. 2009;38(6):1228–1234. [Google Scholar]
- 20.Kroch EA, Duan M. CareScience Risk Assessment Model—Hospital Performance Measurement, Presentations from a November 2008 meeting to discuss issues related to mortality measures. http://archive.ahrq.gov/professionals/quality-patient-safety/quality-resources/tools/mortality/KrochRisk.html. Accessed January 15, 2016.
- 21.Li Z, Mahendra G. Using “Recycled Predictions” for Computing Marginal Effects, Statistics and Data Analysis, SAS Global Forum 2010, Paper 272-2010. http://support.sas.com/resources/papers/proceedings10/272-2010.pdf. Accessed January 12, 2016.
- 22.American Hospital Association Website. https://www.ahadataviewer.com/quickreport/A_Summary_View_Of_United_States_Hospitals_Preview.pdf. Accessed March 18, 2016.
- 23.Cohen J., Vincent J.L., Adhikari N.K. Sepsis roadmap for future research. Lancet Infect Dis. 2015;15(5):581–614. doi: 10.1016/S1473-3099(15)70112-X. [DOI] [PubMed] [Google Scholar]
- 24.Heyland D.K., Johnson A.P., Reynolds S.C., Muscedere J. Procalcitonin for reduced antibiotic exposure in the critical care setting: a systematic review and an economic evaluation. Crit Care Med. 2011;39(7):1792–1799. doi: 10.1097/CCM.0b013e31821201a5. [DOI] [PubMed] [Google Scholar]
- 25.Kopterides P., Siempos, Tsangaris I. Procalcitonin-guided algorithms of antibiotic therapy in the intensive care unit: a systematic review and meta-analysis of randomized controlled trials. Crit Care Med. 2010;38(11):2229–2241. doi: 10.1097/CCM.0b013e3181f17bf9. [DOI] [PubMed] [Google Scholar]
- 26.Schuetz P., Briel M., Mueller B. Clinical outcomes associated with procalcitonin algorithms to guide antibiotic therapy in respiratory tract infections. JAMA. 2013;309(7):717–718. doi: 10.1001/jama.2013.697. [DOI] [PubMed] [Google Scholar]
- 27.Westwood M., Ramackers B., Whiting P. Procalcitonin testing to guide antibiotic therapy for the treatment of sepsis in intensive care settings and for suspected bacterial infection in emergency department settings: a systematic review and cost-effectiveness analysis. Health Technol Assess. 2015;19(96):1–236. doi: 10.3310/hta19960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Soni N.J., Samson D.J., Galaydick J.L. Procalcitonin-guided antibiotic therapy: a systematic review and meta-analysis. J Hosp Med. 2013;8(9):530–540. doi: 10.1002/jhm.2067. [DOI] [PubMed] [Google Scholar]
- 29.Schuetz P., Balk R., Briel M. Economic evaluation of procalcitonin-guided antibiotic therapy in acute respiratory infections: a US health system perspective. Clin Chem Lab Med. 2015;53(4):583–592. doi: 10.1515/cclm-2014-1015. [DOI] [PubMed] [Google Scholar]
- 30.Prkno A., Wacker C., Brunkhorst F.M., Schlattmann P. Procalcitonin-guided therapy in intensive care unit patients with severe sepsis and septic shock—a systematic review and meta-analysis. Crit Care. 2013;17(6):R291. doi: 10.1186/cc13157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Waechter J., Kumar A., Lapinsky S.E. Interaction between fluids and vasoactive agents on mortality in septic shock: a multicenter, observational study. Crit Care Med. 2014;42(10):2158–2168. doi: 10.1097/CCM.0000000000000520. [DOI] [PubMed] [Google Scholar]
- 32.Mouncey P.R., Osborn T.M., Power G.S. Trial of early, goal-directed resuscitation for septic shock. N Engl J Med. 2015;372(14):1301–1311. doi: 10.1056/NEJMoa1500896. [DOI] [PubMed] [Google Scholar]
- 33.The ARISE Investigators and the ANZICS Clinical Trials Group Goal-directed resuscitation for patients with early septic shock. N Engl J Med. 2014;371(16):1496–1506. doi: 10.1056/NEJMoa1404380. [DOI] [PubMed] [Google Scholar]
- 34.The ProCESS Investigators A randomized trial of protocol-based care for early septic shock. N Engl J Med. 2014;370(18):1683–1693. doi: 10.1056/NEJMoa1401602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Levy M.M., Rhodes A., Phillips G.S. Surviving sepsis campaign: association between performance metrics and outcomes in a 7.5 year study. Crit Care Med. 2015;43(1):3–12. doi: 10.1097/CCM.0000000000000723. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.