Abstract
BACKGROUND:
Disorders in the metabolism of iron in the direction of iron overload are observed not only in primary hemochromatosis but also in some chronic liver diseases other aetiology. Elevation of serum iron, ferritin and transferrin saturation is reported in nonalcoholic fatty liver disease and alcohol, chronic hepatitis C and liver cirrhosis.
AIM:
Aim of the study was to evaluate and compare the frequency of the iron serum markers in patients with various chronic liver diseases.
MATERIAL AND METHODS:
The study included a total of 246 persons -186 patients with chronic liver disease without cirrhosis (-115 men, women -71; average age of 50.41 ± 12.85, from 23 to 77 years) and 60 healthy controls (-30 men, women -30, middle-aged 50.50 ± 11.31, from 29 to 83 years). Medical history, physical examination and demographic data including height, weight, laboratory and instrumental studies were performed.
RESULTS:
The highest incidence of elevated serum iron, transferrin saturation and ferritin and decreased serum hepcidin found in cases of alcoholic liver disease (ALD), nonalcoholic fatty liver disease (NAFLD) and chronic hepatitis C (CHC).
CONCLUSION:
Finally, analysis of the changes in serum markers of iron metabolism shows that the difference between healthy and sick with liver disease is primarily due to changes in alcoholic and nonalcoholic fatty liver disease, particularly steatohepatitis, and chronic hepatitis C.
Keywords: Steatosis, Serum ferritin, Serum iron, Transferrin saturation, Chronic liver disease
Introduction
The liver is an important organ for the homeostasis of the iron. Reticuloendothelial macrophages and hepatocytes are the main depots more than iron. In the liver is performed the synthesis of the hormone hepcidin and thus it became the central authority for the regulation of the body’s reserves of iron [1] [2] [3].
Disorders in the metabolism of iron in the direction of iron overload are observed not only in primary hemochromatosis but also in some chronic liver disease with another aetiology [4] [5] [6] [7]. Increases in serum levels of iron, ferritin, and transferrin saturation are reported in nonalcoholic fatty liver disease and alcoholic fatty liver disease, chronic hepatitis C and liver cirrhosis [8] [9] [10] [11] [12] [13].
In recent years, there accumulated a lot of new data, some of them contradictory, about the clinical significance of serum parameters of iron metabolism as surrogate markers of siderosis and severity of liver disease [8] [10] [14] [15] [16] [17]. There remain open questions regarding the clinical significance of serum parameters of iron metabolism and hepcidin in various chronic liver diseases and the role of some genetic factors and environmental factors for organic liver damage during overload syndrome iron.
The study aimed to evaluate and compare the frequency of the iron serum markers in patients with various chronic liver diseases.
To implement the above objective we set ourselves the following tasks:
1. To evaluate the change of serum parameters of iron metabolism in patients with the chronic liver disease compared to healthy subjects
2. To carry out comparative serum markers of iron metabolism in various chronic liver diseases
Material and Methods
Investigated persons
The study included a total of 246 persons -186 patients with chronic liver diseases without cirrhosis (- 115 men, women - 71; average age of 50.41 ± 12.85, from 23 to 77 years) and 60 healthy controls (men -30 women -30 middle-aged 50.50 ± 11.31, from 29 to 83 years) in seven groups:
Group I - 38 patients with primary fatty liver disease, non-alcoholic (male 20, female 18, mean age of 12.45 ± 53.13, from 30 to 76 years) -nonalcoholic steatosis (n = 22) and non-alcoholic steatohepatitis (n = 16). The diagnosis was based on standard criteria, ultrasound evidence of steatosis and histologically verified in 22 cases and ruled out other etiologies.
Group II - 45 patients with alcoholic liver disease (31 men, 14 women; mean age of 10.80 ± 53.42 from 30 to °73) - alcoholic fatty liver (n = 13) and alcoholic steatohepatitis (n = 32) with the intake of absolute alcohol over 40 g/day, histologically confirmed in 39 of them. The diagnosis was based on standard criteria and excluded other causes of liver damage.
Group III - 38 patients with chronic hepatitis C (21 men, 17 women; mean age of 13.61 ± 52.66 from 27 to 77 years). All patients with active viral replication and the diagnosis were histologically verified in 29 of them.
Group IV - 35 patients with chronic hepatitis B (29 men, 6 women, middle-aged 14.40 ± 44.15 from 23 to 74 years) with evidence of viral replication and histologically confirmed the diagnosis in 32 of them.
Group V -13 patients with chronic hepatitis D (7 men, women 6; middle-aged 45.08 ± 9.57, from 30 to 60 d), serologically demonstrated chronic infection in any histologically verified only in 3 of them.
Group VI -17 patients with autoimmune liver disease (male 10, female 7; middle-aged 47.59 ± 11.52, from 26 to 62 years) - primary biliary cirrhosis (n=7) and chronic autoimmune hepatitis (n=10). In all patients, the diagnosis was confirmed histologically and immunologically.
All included patients with chronic liver diseases no significant comorbidities, interfering with the syndrome of iron overload.
Group VII - 60 healthy volunteers participating in clinical trials. All respondents have not a history, physical, laboratory, serological (test for HIV, HBV, HCV and acute HAV), ECG and ultrasound evidence of past or present diseases of the liver and biliary system.
Written informed consent for research is taken by all persons involved in the study.
Methods
History, physical examination and demographic data incl. Height, weight, BMI, abdominal circumference. Estimates for daily consumption of absolute alcohol are based on recalculation in one standard alcoholic unit, which equates to about 10-12 g of absolute alcohol.
They were investigated following standard and specific to the disease laboratory parameters, incl. Immunological and virological parameters.
Conventional ultrasound of abdomen and pelvis was performed in all respondents, and other pictorial or research tools-necessarily. Percutaneous or surgical liver biopsy with histologic evaluation of grades of steatosis and activity and stage of fibrosis was conducted at a total of 142 studied patients with chronic liver diseases judged by the criteria of Brunt, METAVIR and other standard histological criteria (Table 1).
Table 1.
Frequency of histological parameters in patients with different groups chronic liver diseases
| Histological parameters | NAFLD | Ald | Chc (n=29) | Chb (n=32) | Chb Hdv (n=3) | Chronic autoimmune hepatitis (n=17) | Total (n=142) | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Nas (n=6) | Nash (n=16) | Total (n=22) | As (n=7) | Ash (n=32) | Total (n=39) | ||||||
| Steatosis | |||||||||||
| Steatosis (-) (n) | 0 | 0 | 0 | 0 | 2 | 2 | 16 | 25 | 2 | 14 | 59 |
| Steatosis (+),(n) | 6 | 16 | 22 | 7 | 30 | 37 | 13 | 7 | 1 | 3 | 83 |
| 1 degree, (n) | 1 | - | 1 | - | 3 | 3 | 10 | 5 | 1 | 1 | 21 |
| 2 degree (n) | 5 | 12 | 17 | 7 | 19 | 26 | 3 | 2 | 0 | 2 | 50 |
| 3 degree (n) | - | 4 | 4 | - | 8 | 8 | - | - | - | - | 12 |
| Activity | |||||||||||
| Activity (-),(n) | 6 | - | 6 | 7 | 13 | 20 | 0 | 0 | 0 | 1 | 27 |
| Activity (+),(n) | 0 | 16 | 16 | 0 | 19 | 19 | 29 | 32 | 3 | 16 | 115 |
| 1 degree, (n) | - | - | 0 | - | 2 | 2 | 5 | 13 | 0 | 5 | 25 |
| 2 degree, (n) | - | 10 | 10 | - | 11 | 11 | 16 | 13 | 3 | 11 | 64 |
| 3 degree, (n) | - | 6 | 6 | - | 6 | 6 | 8 | 6 | 0 | 0 | 26 |
| Fibrosis | |||||||||||
| Fibrosis (-),(n) | 6 | 1 | 7 | 7 | 1 | 8 | 0 | 6 | 0 | 8 | 29 |
| Fibrosis (+),(n) | 0 | 15 | 15 | 0 | 31 | 31 | 29 | 26 | 3 | 9 | 113 |
| 1 stage (n) | - | 8 | 8 | - | - | 13 | 14 | 19 | 3 | 9 | 67 |
| 2 stage (n) | - | 7 | 7 | - | - | 12 | 11 | 7 | 0 | 0 | 37 |
| 3 stage (n) | - | - | 0 | - | - | 6 | 4 | 0 | 0 | 0 | 10 |
| 3 stage (n) | - | - | 0 | - | - | 0 | 0 | 0 | 0 | 0 | 0 |
All patients received the standard treatment for the disease.
The credibility of the results evaluated by the following statistical analysis: parametric -t-test for paired differences to assess the changes in dynamics; -Mann-Whitney nonparametric comparison of averages for non-Gaussian distribution of data; ANOVA, correlation analysis: Pearson and Spearman, χ2-square test and other (SPSS v.13). The results evaluated statistically significant at a threshold level of significance p > 0.05.
Results
The highest incidence of elevated serum iron, transferrin saturation and ferritin and decreased serum hepcidin was found in cases with AFLD, NAFLD and CHC. Increased serum iron was found in 53% of NAFLD in 64% - at AFLD, 42% - at CHC but only 6% in CHB, 8% for CHB+HDV infection and 6% in autoimmune liver diseases. Increased serum ferritin was reported in 42% for NAFLD in 49% -in AFLD and 29%-CHC, but only 3% on CHB and 0% in CHB+HDV infection and autoimmune liver diseases. Increased transferrin saturation was established in NAFLD in 18%, 36% - at AFLD, 29% - at CHC, 14% - on CHB and 8% - at CHB+HDV infection. Decreased serum hepcidin was reported in 47% at AFLD, 60% - at NAFLD, 53% - at CHC, 26% - on CHB, 8% - at CHB+HDV infection and 6% - at autoimmune liver diseases. Share distribution in the different groups chronic liver diseases with simultaneous increase of 2 or 3 of serum parameters of iron metabolism is shown in Table 2.
Table 2.
Values of iron metabolism in different groups chronic liver diseases
| Values of iron metabolism | NAFLD (n=38) | AFLD (n=45) | CHC (n=38) | CHB (n=35) | CHB, HDV (n=13) | Autoimmune liver diseases (n=17) | Total (n=186) |
|---|---|---|---|---|---|---|---|
| ↑ iron + ↑ ferritin (n, %) |
16 (42%) | 22 (49%) | 10 (26%) | 1 (3%) | 0 (0%) | 0 (0%) | 49 (26%) |
| ↑ iron + ↑ transferrin saturation (n, %) |
6 (16%) | 16 (36%) | 9 (24%) | 2 (6%) | 1 (8%) | 0 (0%) | 34 (18%) |
| ↑ ferritin + ↑ transferrin saturation (n, %) |
4 (11%) | 14 (31%) | 5 (13%) | 1 (3%) | 0 (0%) | 0 (0%) | 24 (13%) |
| ↑ iron + ↑ ferritin + ↑ transferrin saturation (n, %) |
4 (11%) | 14 (31%) | 4 (11%) | 1 (3%) | 0 (0%) | 0 (0%) | 23 (12%) |
The most common abnormal iron metabolism found in the group with AFLD, followed by that of CHC and NAFLD. In all these cases there was reduced and serum hepcidin.
Serum iron was significantly higher in patients with NAFLD compared to controls (Z = -2,2494, P = 0.013) and CHB (Z = -2,357, P = 0.018), and that at AFLD-compared to controls (Z = -4,151, P = 0.0001), CHB (Z = -2,125, P = 0.034), CHB+HDV (Z = -4.002, P = 0.0001) and autoimmune liver diseases (Z = -2,864, P = 0.004) (Figure 1).
Figure 1.

Serum iron (mmol/l) in different groups
Significantly higher values were in CHC compared to controls (Z = -2,173, P = 0.030) and CHB (Z = -2,205, P = 0.027). Values of iron binding capacity were significantly higher in patients with NAFLD compared to controls (Z = -5,565, P = 0.0001) and CHB (Z = -4,736, P = 0.0001) at AFLD - compared to controls (Z = -7,844, P = 0.0001), CHC (Z = -3,442, P = 0.001), CHB (Z = -6,578, P = 0.0001) and autoimmune liver diseases (Z = -2,036, P = 0.042) (Figure 2).
Figure 2.

Values of iron binding capacity in different groups
Significantly higher values were in CHC compared to controls (Z = -4,825, P = 0.0001) and CHB (Z = -4,319, P = 0.0001), and in CHB, compared to autoimmune liver diseases (Z = -3,921, P = 0.0001) as well as in autoimmune liver diseases compared to controls (Z = -5,475, P = 0.0001). Ferritin values were significantly higher in: NAFLD compared with controls (Z = -1,965, P = 0.05), CHB (Z = -3,832, P = 0.0001), autoimmune liver diseases (Z = -3,561, P = 0.0001); AFLD when compared to controls (Z = -2,953, P = 0.003), CHB (Z = -3,895, P = 0.0001), and the autoimmune liver diseases (Z = -3,740, P = 0.0001), p = 0.0001); CHC when compared to the CHB (Z = -2,892, P = 0.004), and the autoimmune liver diseases (Z = -2,942, P = 0.003); CHB relative to controls (Z = -3,683, P = 0.0001) and autoimmune liver diseases (Z = -2,088, P = 0.037) (Figure 3).
Figure 3.

Values of serum ferritin in different groups
The values of transferrin saturation were significantly higher in: NAFLD compared with autoimmune liver diseases (Z = -2,343, P = 0.019); AFLD compared with autoimmune liver diseases (Z = -3,032, P = 0.002); CHC compared with autoimmune liver diseases (Z = -3,035, P = 0.002); CHB compared with autoimmune liver diseases (Z = -3,409, P = 0.001); autoimmune liver diseases and compared to controls (Z = -3,450, P = 0.001) (Figure 4).
Figure 4.

Values of transferrin saturation in different groups
Serum levels of hepcidin were significantly lower in patients with NAFLD, AFLD and CHC in comparison to controls (respectively Z = -2,326, P = 0.02, Z = -3,312, Z = -2,559, P = 0.001 and P = 0.01), CHB (respectively Z = -2,122, P = 0.034, Z = -2,991, P = 0.003 and Z = -2,295, P = 0.023) and autoimmune liver diseases (respectively Z = -2,277, P = 0.023, Z = -2.840, P = 0.005 and Z = -2,295, P = 0.022), but no statistically significant difference in the comparison between them (Figure 5).
Figure 5.

Values of serum hepcidin in different groups
In all groups chronic liver diseases, serum iron, ferritin, transferrin saturation and hepcidin showed correlation (r = 0.470-0.875, P = 0.003-0.0001). Increased serum iron, ferritin, transferrin saturation and serum decreased hepcidin, and combinations thereof, found more often in metabolic steatohepatitis or alcoholic aetiology when comparing the cases with steatosis (Table 3).
Table 3.
Values of iron metabolism in NAFLD and AFLD
| Values of iron metabolism | NAFLD | AFLD | ||||
|---|---|---|---|---|---|---|
| NAS (n=22) | NASH (n=16) | Total (n=38) | AS (n=13) | ASH (n=32) | Total (n=45) | |
| ↑ Serum Iron (n, %) | 5 (23 %) | 15 (94%) | 20 (53 %) | 3 (23%) | 26 (81%) | 29 (64%) |
| ↑ Serum ferritin (n, %) | 2 (9 %) | 14 (87%) | 16 (42 %) | 1 (8%) | 21 (65%) | 22 (48.9%) |
| ↑ transferrin saturation (n, %) | 2 (9 %) | 5 (31%) | 7 (18%) | 1 (8%) | 15 (47%) | 16 (36%) |
| ↑ Serum Iron + ↑ Serum ferritin (n, %) |
2 (9 %) | 14 (87%) | 16 (42%) | 1 (8%) | 21 (66%) | 22 49%) |
| ↑ ↑ Serum Iron + ↑ transferrin saturation (n, %) |
1 (4%) | 5 (31%) | 6 (16%) | 1 (8%) | 5 (47%) | 16 (36% |
| ↑ Serum ferritin + ↑ transferrin saturation (n, %) |
0 (0%) | 4 (25%) | 4 (11%) | 0 (0%) | 14 (44%) | 14 (31%) |
| ↑ ↑ Serum Iron + ↑ Serum ferritin + ↑ transferrin saturation (n, %) |
0 (0%) | 4 (25%) | 4 (10%) | 0 (0%) | 14 (44%) | 14 (31%) |
| ↓ hepcidin (n, %) | 5 (23 %) | 13 (81%) | 18 (47%) | 3 (23%) | 24 (75%) | 27 (60%) |
Mean serum iron, transferrin and transferrin saturation in patients with nonalcoholic and alcoholic steatohepatitis showed significantly higher values compared with steatosis (respectively Z = -4.155, P = 0.0001, Z = -3.474, P = 0.001, Z = -4.110, P = 0.0001 and Z = -3.628, P = 0.0001 (Figures 6, 7, 8).
Figure 6.

Values of serum iron in NAFLD and AFLD
Figure 7.

Values of iron binding capacity in NAFLD and AFLD
Figure 8.

Values of serum ferritin in NAFLD and AFLD
Serum hepcidin was significantly lower in patients with NASH and ASH compared in groups with NAFLD and AFLD, respectively (respectively Z=-3.252, p=0.001 and Z=2.304, p=0.021) (Figure 9).
Figure 9.

Values of transferrin saturation in NAFLD and AFLD
Figure 10.
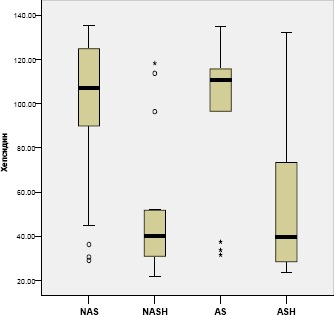
Values of serum hepcidin in NAFLD and AFLD
Discussion
The results of our study show that chronic liver diseases detect abnormalities in all serum parameters of iron metabolism-elevated serum iron, ferritin and transferrin saturation, as well as reduced hepcidin compared with healthy individuals. Considered changes in parameters of iron metabolism separately, the most frequent and approximately equally are variations in serum iron and hepcidin, followed by those of ferritin and transferrin saturation.
Deviations from the normal range in healthy persons are identified only in serum hepcidin, which was reduced in 20% of cases. If we use standard laboratory parameters, the frequency of the syndrome of iron overload is estimated at 22% based on transferrin saturation above 45%.
Approximately at this frequency is the combined increase in serum levels of the ferritin and the iron (26%), as well as the simultaneous increase in the iron and transferrin saturation (18%).
Two times lower is the frequency of the simultaneously elevated transferrin saturation and the ferritin -13%, as well as the simultaneous increases of the three serum parameters -iron, transferrin saturation and ferritin to -12%. This frequency does not change significantly if these combinations are added and reduced hepcidin.
This applies both to frequency deviations and for their serum levels. At most this is true for serum hepcidin. Changes in these groups are more apparent, in comparison with those of patients with chronic hepatitis B and D, chronic autoimmune hepatitis and primary biliary cirrhosis. In all cases of combined changes in standard serum markers of iron metabolism and all groups HLD is presented and reduced hepcidin. We did not find a difference in the serum levels of the examined parameters of iron metabolism in patients with nonalcoholic and alcoholic fatty liver disease. In patients with chronic hepatitis B, alone or in combination with chronic hepatitis D, as well as autoimmune diseases a change in serum markers of iron metabolism is reported only in individual cases.
Our data shows that unlike healthy, in which serum iron, ferritin and IBC are higher for men than for women, in case of a liver disease gender difference in the level of serum iron and IBC disappears. Deviations of the serum indicators of iron metabolism were more pronounced with age (predominantly in HH C) and increased BMI. Relationship with BMI was mainly at the expense of patients with NAFLD and HH C. Only in patients with NAFLD all indicators were more obviously deviated in obesity. This can be explained by the relationship of these diseases with insulin resistance [17] [18].
For the first time, we found different correlations between serum parameters of iron metabolism in healthy patients and those with the disease. While in healthy individuals only have a positive relationship between the level of iron and IBC and feedback between serum hepcidin and transferrin saturation, then in HLD as a whole and individual subgroups of liver disease serum levels of iron, IBC, ferritin, transferrin saturation and hepcidin showed a strong correlation.
In HLD is proved a link between indicators reflecting liver damage and liver function and serum parameters of iron metabolism. We found a correlation between serum markers of iron metabolism and liver enzymes AST, ALT and GGT, as the abnormalities were higher in groups with diverted indicators of iron metabolism in comparison to the group with normal levels. Further-more serum markers of iron metabolism correlated with total bilirubin and direct bilirubin, prothrombin time, and platelet count. These results confirm established by other authors interconnections between the parameters of the syndrome of iron overload and liver enzymes and liver function [19] [20] Also, we have established a link with IgA. The advantage of our study is that we proved the greatest relationship of these indicators with the global assessment of severity of liver disease-index weight of liver damage. Serum iron, IBC, ferritin and transferrin saturation increases with TLD and values of hepcidin-reduces. We proved and their relationship with HDL-cholesterol, triglycerides and glucose. All correlations are determined primarily by changes in patients with NAFLD and ALD. Also, in patients with NAFLD and ALD, we found another correlation between AF and albumin. The relationship between KKLI-indicator to assess the severity of alcoholic liver disease and the serum parameters of iron metabolism is determined by patients with alcoholic steatohepatitis. In patients with proven HH C, we proved a connection mainly with indicators for severity of liver damage-total and direct bilirubin, albumin and prothrombin index.
In our study, we examined various liver diseases, and we examined the overall changes in iron metabolism in the control group of healthy persons and in between them. In NAFLD is often increased serum iron (53%), followed by a decrease in serum hepcidin (47%) and elevated ferritin (42%), and about 2 times less frequently elevated transferrin saturation (18%). The combined standard deviation of serum markers of iron metabolism is most often the simultaneous increase in serum iron and ferritin-42%, followed by increases in serum iron and HT-16%. In 11% had elevated serum iron, ferritin and HT. Our results show that the incidence of the syndrome of over-saturation with iron, as measured according to the standard serum markers is 18% based on the increased NT and 11%-based on changes in any serum markers. The higher incidence of abnormal serum iron and ferritin corresponds to the hepcidin, but can be explained by non-specific changes related to the underlying disease.
In ALD increased serum iron (64%) together with reduced serum hepcidin (60%) are the most common abnormalities. Ferritin was promoted to ½ of the cases (49%). And in this condition as in NAFLD transferrin saturation above 45% is the rare deviation-36%. The simultaneous increase in serum iron and ferritin was 48%, but the combined tolerances of increased serum iron and LT, as well as increased iron, transferrin and HT, are the most common in alcoholic aetiology, respectively in 36% and 31%. The incidence of the syndrome of over-saturation with iron was 36% by elevated HT and 31%-based on changes in any serum markers. This frequency is the highest in comparison with all other chronic liver diseases, and it is about 2 to 3 times greater in comparison with patients with NAFLD. Here, as in NAFLD incidence of abnormal serum iron corresponds to the hepcidin, but about half of these cases can certainly be considered an overload of iron based on changes in the standard serum markers of iron metabolism. It is more pronounced changes in hepcidin to meet in earlier changes in the liver before a heavy load of iron.
Interest is the comparison of the results in patients with steatosis and steatohepatitis inside NAFLD groups and ALD and between them. We found more pronounced deviations in all serum markers of iron metabolism in both non-alcoholic and alcoholic steatohepatitis when compared to cases with steatosis, and no difference between patients with NAD and ALD, and between those with NASH and ASH. The values of serum iron parameters of metabolism in patients with nonalcoholic steatosis and alcohol were close to the controls and other groups HLD, but transferrin saturation in them is higher. The values of the parameters of iron metabolism in alcoholic and nonalcoholic steatohepatitis are highest.
Increased serum iron, ferritin, transferrin saturation and decreased serum hepcidin, as well as a combination between them, we found more often in steatohepatitis with alcohol or metabolic aetiology compared to cases with steatosis. Thus the frequency of the overload of iron in NASH increases to 31% (on NT) and 25% (all indices), and in ASH-respectively 47% and 44%. The increase in serum iron and ferritin and reduction of hepcidin were found in over two-thirds of those surveyed with NASH and ASH.
In HH C deflected (low), the most are serum hepcidin (53%), followed by increased serum iron (42%). Elevated transferrin saturation and ferritin are detected equally-29%. Differences in the rate of change of the different parameters are less in comparison to the alcoholic and nonalcoholic fatty liver disease. Almost 2 times less frequent is the combination of increased serum iron and increased ferritin. On the other hand at the same time, an increase in the iron and HT (24%), ferritin and HT (13%) and the deviation in all the indicators (11%) are close to that of NAFLD. The frequency of iron overload by elevated HT is 29% and 11%-13%-based on the above latter two combinations.
Unlike HH C in HH B increased serum iron is found only in 2 of 35 patients (6%) and in the same proportion (1 out of 13 patients-8%)-for chronic HDV infection, and the ferritin-in 1 patient (3%), and none of the patients with HHD. At higher levels, we found elevated transferrin saturation (14%) and decreased serum hepcidin (26%). However, this frequency corresponds to that in healthy individuals. Variation in these indicators we found in only 1 case (8%) with accompanying HDV infection. A combination of increased iron with increased HT we reported in 2 patients with HH B (respectively 1 patient with HHD), and when one of them combined with the rest of the parameters. Accordingly to the above changes in 2 patients with HH B and 1-with HHD maintained overload with iron according to the increased transferrin saturation, and in one of the patients with HH B-and according to the combination of the remaining markers.
In autoimmune diseases, increased serum iron we found in only 1 out of 17 tested cases (6%) diagnosed with PBC. In another case, we found reduced serum hepcidin (6%). Data for immune-mediated liver diseases are extremely small. Similar to our observation, it is reported a very low incidence of biliary cirrhosis [16] [21].
In conclusion, the analysis of data from our study shows changes in every serum marker of iron metabolism, including reducing of serum hepcidin in nonalcoholic and alcoholic fatty liver disease, and chronic hepatitis C in comparison with healthy persons, and other chronic liver diseases associated with HBV, HDV or autoimmune pathogenesis. It is proved a strong correlation between them. The syndrome of iron overload correlates mainly with liver enzymes, liver function tests, and metabolic parameters. None of the surrogate serum markers of iron overload in chronic liver disease does reflect fully and accurately haemosiderosis. However, we recommend the evaluation to be carried out by transferrin saturation above 45%, alone, especially NAFLD or together with the increased transferrin-in alcoholic liver disease and chronic hepatitis C. Thus, with the highest probability is confirmed iron overload.
Footnotes
Funding: This research did not receive any financial support
Competing Interests: The authors have declared that no competing interests exist
References
- 1.Day CP, James OF. Steatohepatitis: a tale of two “hits”? Gastroenterology. 1998;11:842–5. doi: 10.1016/s0016-5085(98)70599-2. https://doi.org/10.1016/S0016-5085(98)70599-2. [DOI] [PubMed] [Google Scholar]
- 2.Deguti MM, Sipahi AM, Gayotto LC, et al. Lack of evidence for the pathogenic role of iron and HFE gene mutations in Brazilian patients with nonalcoholic steatohepatitis. Braz J Med Biol Res. 2003;36:739–45. doi: 10.1590/s0100-879x2003000600009. https://doi.org/10.1590/S0100-879X2003000600009 PMid:12792703. [DOI] [PubMed] [Google Scholar]
- 3.Deugnier Y, Brissot P, Loréal O. Iron and the liver: update 2008. J Hepatol. 2008;48(Suppl 1):S113–23. doi: 10.1016/j.jhep.2008.01.014. https://doi.org/10.1016/j.jhep.2008.01.014 PMid:18304682. [DOI] [PubMed] [Google Scholar]
- 4.Deugnier Y, Turlin B. Pathology of hepatic iron overload. World J Gastroenterol. 2007;13:4755–4760. doi: 10.3748/wjg.v13.i35.4755. https://doi.org/10.3748/wjg.v13.i35.4755 PMid:17729397 PMCid: PMC4611197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Di Bisceglie AM, Bonkovsky HL, et al. Iron reduction as an adjuvant to interferon therapy in patients with chronic hepatitis C who have previously not responded to interferon: a multicenter, prospective, randomized, controlled trial. Hepatology. 2000;32:135–138. doi: 10.1053/jhep.2000.8700. https://doi.org/10.1053/jhep.2000.8700 PMid:10869301. [DOI] [PubMed] [Google Scholar]
- 6.Dostalikova-Cimburova M, Kratka K, Stransky J, et al. Iron overload and HFE gene mutations in Czech patients with chronic liver diseases. Dis Markers. 2012;32(1):65–72. doi: 10.3233/DMA-2012-0861. https://doi.org/10.1155/2012/790464 PMid:22297603 PMCid: PMC3826488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Duseja A, Das A, Das R, et al. The clinicopathological profile of Indian patients with nonalcoholic fatty liver disease (NAFLD) is different from that in the West. Dig Dis Sci. 2007;52:2368–74. doi: 10.1007/s10620-006-9136-y. https://doi.org/10.1007/s10620-006-9136-y PMid:17420951. [DOI] [PubMed] [Google Scholar]
- 8.Ekstedt M, Franzen LE, Mathiesen UL, et al. Long-term follow-up of patients with NAFLD and elevated liver enzymes. Hepatology. 2006;44:865–73. doi: 10.1002/hep.21327. https://doi.org/10.1002/hep.21327 PMid:17006923. [DOI] [PubMed] [Google Scholar]
- 9.Fernandez-Real JM, Lopez-Bermejo A, Ricart W. Cross-talk between iron metabolism and diabetes. Diabetes. 2002;51:2348–54. doi: 10.2337/diabetes.51.8.2348. https://doi.org/10.2337/diabetes.51.8.2348 PMid:12145144. [DOI] [PubMed] [Google Scholar]
- 10.Fracanzani AL, Conte D, Fraquelli M, et al. Increased cancer risk in a cohort of 230 patients with hereditary hemochromatosis in comparison to matched control patients with non-iron-related chronic liver disease. Hepatology. 2001;33:647–51. doi: 10.1053/jhep.2001.22506. https://doi.org/10.1053/jhep.2001.22506 PMid:11230745. [DOI] [PubMed] [Google Scholar]
- 11.Fujita N, Miyachi H, Tanaka H, et al. Iron overload is associated with hepatic oxidative Damage to DNA in nonalcoholic steatohepatitis. Cancer Epidemiol Biomarkers Prev. 2009;18:424–32. doi: 10.1158/1055-9965.EPI-08-0725. https://doi.org/10.1158/1055-9965.EPI-08-0725 PMid:19190144. [DOI] [PubMed] [Google Scholar]
- 12.Harrison-Findik DD, Schafer DE, Klein, et al. Alcohol metabolism -mediated oxidative stress downregulates hepcidin transcription and leads to increased duodenal iron transporter expression. J Biol Chem. 2006;281:22974–82. doi: 10.1074/jbc.M602098200. https://doi.org/10.1074/jbc.M602098200 PMid:16737972. [DOI] [PubMed] [Google Scholar]
- 13.Ioannou GN, Dominitz JA, Weiss NS, et al. Racial differences in the relationship between hepatitis C infection and iron stores. Hepatology. 2003;37:795–801. doi: 10.1053/jhep.2003.50147. https://doi.org/10.1053/jhep.2003.50147 PMid:12668972. [DOI] [PubMed] [Google Scholar]
- 14.Kemna EH, Kartikasari AE, van Tits LJ, et al. Regulation of hepcidin: Insights from biochemical analyses on human serum samples. Blood Cells Mol Dis. 2008;40:339–346. doi: 10.1016/j.bcmd.2007.10.002. https://doi.org/10.1016/j.bcmd.2007.10.002 PMid:18023212. [DOI] [PubMed] [Google Scholar]
- 15.Kohgo Y, Ohtake T, Ikuta K, Suzuki Y, Torimoto Y, Kato J. Dysregulation of systemic iron metabolism in alcoholic liver diseases. J Gastroenterol Hepatol. 2008;23(Suppl 1):S78–81. doi: 10.1111/j.1440-1746.2007.05290.x. https://doi.org/10.1111/j.1440-1746.2007.05290.x PMid:18336670. [DOI] [PubMed] [Google Scholar]
- 16.Krastev Z. Liver damage score-a new index for evaluation of the severity of chronic liver diseases. Hepatogastroenterology. 1998;45(19):160–9. PMid:9496507. [PubMed] [Google Scholar]
- 17.Nelson JE, Bhattacharya R, Lindor KD, et al. HFE C282Y mutations are associated with advanced hepatic fibrosis in Caucasians with nonalcoholic steatohepatitis. Hepatology. 2007;46:723–9. doi: 10.1002/hep.21742. https://doi.org/10.1002/hep.21742 PMid:17680648. [DOI] [PubMed] [Google Scholar]
- 18.Nelson JE, Wilson L, Brunt EM, Yeh MM, et al. Relationship between the pattern of hepatic iron deposition and histological severity in nonalcoholic fatty liver disease. Hepatology. 2011;53(2):448–57. doi: 10.1002/hep.24038. https://doi.org/10.1002/hep.24038 PMid:21274866 PMCid: PMC3058264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Olynyk JK, Reddy KR, Di Bisceglie AM, et al. Hepatic iron concentration as a predictor of response to interferon-alfa therapy in chronic hepatitis C. Gastroenterology. 1995;108:1104–9. doi: 10.1016/0016-5085(95)90209-0. https://doi.org/10.1016/0016-5085(95)90209-0. [DOI] [PubMed] [Google Scholar]
- 20.Price L, Kowdley KV. The role of iron in the pathophysiology and treatment of chronic hepatitis C. Can J Gastroenterol. 2009;23(12):822–8. doi: 10.1155/2009/290383. https://doi.org/10.1155/2009/290383 PMid:20011735 PMCid: PMC2805519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Recalcati S, Invernizzi P, Arosio P, et al. New functions for an iron storage protein: the role of ferritin in immunity and autoimmunity. J Autoimmun. 2008;30:84–9. doi: 10.1016/j.jaut.2007.11.003. https://doi.org/10.1016/j.jaut.2007.11.003 PMid:18191543. [DOI] [PubMed] [Google Scholar]


