Abstract
Triglycerides are the conventional tool to measure VLDLs, whereas LDL cholesterol (LDL-C) is the conventional tool to measure LDLs. Multiple epidemiological studies, including a series of genetically based analyses, have demonstrated that cardiovascular risk is related to triglycerides independently of LDL-C, and this has led to a series of new therapeutic agents designed specifically to reduce plasma triglycerides. The triglyceride hypothesis posits that increased levels of triglycerides increase cardiovascular risk and decreasing plasma triglycerides decreases cardiovascular risk. In this work, we will examine the validity of the triglyceride hypothesis by detailing the biological complexities associated with hypertriglyceridemia, the genetic epidemiological evidence in favor of hypertriglyceridemia, the evidence from the fibrate randomized clinical trials relating triglycerides and clinical outcomes, and the completeness of the evidence from the initial studies of novel mutations and the therapeutic agents based on these mutations that lower triglycerides. Because of the multiple metabolic links between VLDL and LDL, we will try to demonstrate that measuring triglycerides and LDL-C alone are inadequate to document the lipoprotein profile. We will try to demonstrate that apoB must be measured, as well as triglycerides and cholesterol, to have an accurate estimate of lipoprotein status.
Keywords: triglycerides, apolipoprotein B, very low density lipoprotein, low density lipoprotein, apolipoprotein CIII, apolipoprotein CIII inhibitor, angiopoietin-like 3 protein
Multiple lines of epidemiological evidence, including most recently, Mendelian randomization studies (1–3), tie plasma triglycerides to the risk of atherosclerotic CVD. Conventionally, cholesterol and triglycerides have been regarded as markers that represent separate and distinct lipoprotein classes: triglycerides are the principal lipid constituent of VLDL particles and elevated triglycerides, therefore, identify elevated VLDL levels; whereas cholesterol is the principal lipid constituent of LDL particles and elevated LDL cholesterol (LDL-C), therefore, identifies elevated LDL levels. In this paradigm, the lipid component of a lipoprotein particle is accepted as fully informative of the atherogenic risk associated with the lipoprotein particle. Accordingly, based on the epidemiological evidence, novel therapeutic agents with major effects on triglyceride concentrations, but with little effect on LDL-C, are being developed. The operating assumption is that if LDL-C is not affected, clinical risk and benefit must relate to differences in plasma triglycerides, not to differences in LDL.
However, hypertriglyceridemia may reflect increased numbers of one or more classes of triglyceride-rich lipoprotein particles, which have different relations to cardiovascular risk. Most VLDL apoB particles are small enough to enter the arterial wall. By contrast, most chylomicron particles are too large to enter the arterial wall and, therefore, the cholesterol and triglyceride they contain are no threat to the integrity of the arterial wall. Although VLDL particles do, in general, account for most of the mass of triglycerides in plasma, the mass of triglyceride per VLDL particle is not uniform. Accordingly, hypertriglyceridemia due to VLDL may be the consequence of an increased mass of triglyceride per VLDL particle, an increase in the number of VLDL particles, or both. Moreover, VLDL particles are the metabolic precursors of LDL particles and the concentration of LDL particles in plasma depends upon the production rate of VLDL particles and the proportion that are converted to LDL particles, as well as the clearance rate of LDL particles (4). All of these can vary independently and substantially. In addition, the composition of VLDL and LDL particles can change independently of their production rates: cholesterol ester transfer protein (CETP)-mediated exchanges and transfers of triglyceride and cholesterol ester between chylomicrons and VLDL particles, on the one hand, and LDL and HDL particles, on the other, produce VLDL, LDL, and HDL particles of variable size with variable masses of cholesterol and triglyceride. Higher plasma triglycerides lead to cholesterol-enriched VLDL particles and cholesterol-diminished LDL and HDL particles (4), whereas lower triglycerides may be associated with cholesterol-enriched apoB particles. This variance in composition means that VLDL particle concentration cannot be reliably deduced from the concentration of triglyceride in plasma and LDL particle concentration cannot be reliably deduced from the concentration of LDL-C in plasma (4).
This discordance between lipid composition and particle number is important because there is considerable evidence that the atherogenic risk associated with the apoB lipoproteins relates more closely to the number of apoB particles compared with the mass of cholesterol within them (5). Moreover, with the exception of type III hyperlipoproteinemia, while the proportion of VLDL apoB versus LDL apoB particles is greater in patients with hypertriglyceridemia than in others, LDL particles continue to account for the majority of apoB particles (6). This means that clinical benefit from genetic mutations and pharmacological interventions that lower triglycerides might be based, at least in part, on their concurrent effects on the number of LDL particles, effects that might not be obvious from measurement of LDL-C. Furthermore, we posit that VLDL apoB is at least as accurate a marker of the risk due to VLDL particles as plasma triglycerides or VLDL cholesterol (VLDL-C).
To date, all the pharmacological agents that lower cardiovascular event rates (statins, statins plus ezetimibe, statins plus PCSK9 inhibitors, and statins plus anacetrapib) lower plasma apoB. Moreover, their clinical benefit relates more closely to the decrease in apoB than to the decrease in LDL-C or non-HDL cholesterol (HDL-C) (7, 8). Indeed, in the case of statins plus CETP inhibitors, the evidence from Mendelian randomization is that clinical benefit relates to the decrease in apoB, not to the increase in HDL-C or the decrease in LDL-C (9). This means that changes in apoB particle number and their relation to changes in risk need to be documented before it can be determined whether triglycerides are an independent risk factor for CVD and whether pharmacological agents that lower triglycerides are likely to succeed clinically and, if they do, why.
Accordingly, this report will review the metabolic interrelationships between the triglyceride- and cholesterol-rich apoB lipoproteins, assess the evidence from the genetic mutations and Mendelian randomization analyses that triglycerides are an independent risk factor for CVD, review the evidence from randomized clinical trials that lowering triglycerides reduces clinical events, and, finally, evaluate the adequacy of the evidence from the new series of triglyceride-lowering agents as to their total effect on the apoB lipoprotein profile. Our conclusion will be that measurement of lipids alone does not provide a complete portrait of lipoprotein status. Measuring apoB is also essential.
METABOLIC RELATIONSHIPS BETWEEN THE TRIGLYCERIDE- AND CHOLESTEROL-RICH apoB LIPOPROTEINS
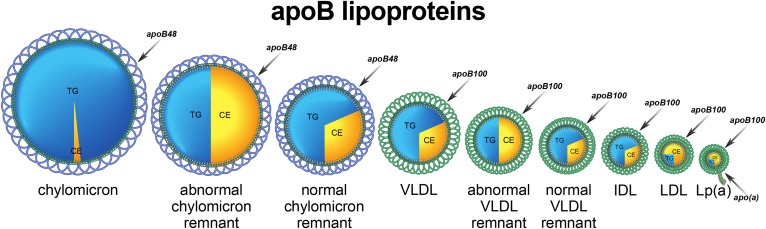
Trapping of apoB particles within the arterial wall is fundamental to the initiation, expansion, and maturation of the atherosclerotic process. The relative importance of the different apoB lipoproteins to this process is determined by their number, their size, and their propensity to bind to constituents of the subintimal spaces within the arterial wall (10) (Fig. 1).
Fig. 1.
apoB lipoprotein particles.
Chylomicron particles
Chylomicron particles each contain one molecule of apoB48 and are present in plasma either as intact or remnant particles (5). Within minutes of entering the systemic circulation, a chylomicron particle binds to multiple molecules of LPL on the capillary endothelium of adipose tissue, skeletal muscle, and the heart. Massive amounts of triglyceride are hydrolyzed in just moments and the torrents of fatty acids released are taken up by the adjacent adipocytes and reformed into triglycerides, a metabolic transformation, which would be almost instantaneous if cytotoxic levels of fatty acids are to be avoided. Moreover, as the chylomicron triglycerides are being hydrolyzed, the central core of the particle partially collapses and phospholipid transfer protein (PLTP) facilitates the shedding of excess surface phospholipids from the chylomicrons. The combination of massive core triglyceride hydrolysis and extensive loss of surface phospholipid generates the smaller chylomicron particles, which are released from the surface of the endothelium in adipocytes and myocytes. The smaller remnant apoB48 particle, which is the normal product of the first step in chylomicron metabolism and contains only a fraction of the original triglyceride, but all the original cholesterol, is removed by the liver. The processes that regulate the removal of LDL particles, the LDL pathway, have been worked out in exquisite detail. This contrasts with the processes responsible for the removal of the other apoB lipoproteins, which appear to be much more complex and remain incompletely understood.
Intact chylomicron particles are too large to pose any significant threat to the arterial wall even when plasma triglycerides are massively elevated. By contrast, when they lodge within the capillaries of the pancreas, they can provoke an acute inflammatory pancreatitis. Normal remnant apoB48 particles are small enough to enter the arterial wall and do contain substantial amounts of cholesterol. However, in normal individuals, even at peak postprandial levels, there are at least 10 times more VLDL apoB100 particles than apoB48 particles (11–13). Nevertheless, each of these particles contains large amounts of cholesterol and contributes to atherogenic risk.
VLDL particles
VLDL particles are triglyceride-rich apoB100 particles that transport fatty acids from the liver to myocytes and adipocytes (4). With few exceptions, most of the triglycerides in plasma are present in VLDL particles, but the mass of triglyceride per VLDL particle and, therefore, the size of VLDL particles varies substantially within and between individuals. There are multiple reasons for this variance in composition. First, composition of VLDL particles secreted by the liver can vary, with the larger more buoyant VLDL1 particles containing more triglyceride than the smaller less buoyant VLDL2 particles (14–16). Second, because the rates at which each of these particles is secreted may vary, the overall rate of triglyceride secretion by the liver will vary relative to the rate of secretion of VLDL particles. Third, as VLDL particles undergo hydrolysis by LPL in the adipose tissue and muscle, their triglyceride content will be diminished and their composition, therefore, will change. Fourth, CETP-mediated core lipid exchange will tend to increase the cholesterol content while lowering the triglyceride content of VLDL particles.
Furthermore, the metabolic fate of VLDL particles is more complex than chylomicron particles. The smaller VLDL particles that are produced after hydrolysis and removal of some of their triglyceride by peripheral cells may either be cleared from the plasma compartment by the liver or converted to IDL and then LDL particles. Why one outcome occurs rather than the other is not understood and represents a critical deficit in our understanding of apoB metabolism and atherogenic risk. In most species, only a small portion of VLDL particles are converted to LDL particles. Only the species in which conversion of VLDL to LDL is a major metabolic outcome of VLDL metabolism are susceptible to atherogenesis because only these species have large numbers of LDL particles. Humans belong to this unfortunate minority. Many of the newer agents that will be reviewed below appear to act at this step. This makes it essential to determine their relative effects on VLDL and LDL particle number.
Abnormal remnant particles
Abnormal apoB48 and apoB100 remnant lipoprotein particles may also be present. A dramatic delay in clearance of partially hydrolyzed VLDL and chylomicron particles by the liver exaggerates core lipid exchanges, which result in increased numbers of cholesterol-enriched remnant particles (17, 18). In classical type III hyperlipoproteinemia, abnormal apoB48 remnant particles plus abnormal VLDL apoB100 remnant particles are present in concentrations 30–50 times or more the concentration of normal remnant particles (11, 12, 19). This disorder produces high risk of CVD in those who are affected and can be diagnosed based on total cholesterol, triglyceride, and apoB (20).
LDL particles
Classically, apoB particles with a density of 1.006–1.019 g/l were labeled as IDL particles, whereas those with a density of 1.019–1.063 g/l were labeled as LDL particles. However, this distinction is not made clinically and all apoB particles with a density between 1.006 and 1.063 g/l are considered LDL particles. Thus LDL-C includes all the cholesterol within what was considered IDL as well as all the cholesterol within what was considered LDL. The evidence to date indicates that the mass of cholesterol within the LDL particle does not significantly change the atherogenic potential of the particle: smaller cholesterol-poor particles are as atherogenic as larger more cholesterol-rich LDL particles (21). However, for any given total cholesterol concentration, a higher concentration of smaller cholesterol-poor LDL particles requires more LDL particles to transport the cholesterol in the circulation; this, in turn, leads to higher CVD risk than would be evident from the LDL-C. Interestingly, while the core lipid content of LDL particles does not appear to alter their atherogenic risk, the addition of apoCIII to the particle may increase its atherogenic potential (22), although concurrent presence of apoE may diminish this risk (23).
Lipoprotein (a)
Lipoprotein (a) particles are LDL particles to which a molecule of apo(a) has been attached. There is strong evidence that the risk of CVD relates, in part, to the level of lipoprotein (a), a relation that is independent of LDL-C or apoB (24, 25).
VARIABILITY OF THE COMPOSITION OF VLDL PARTICLES AND OF THE RATIO OF VLDL apoB TO LDL apoB
That the relation of VLDL apoB to LDL apoB in patients with hypertriglyceridemia and others is variable is not widely appreciated. With normal triglyceride levels, VLDL particles average 10 ± 0.04% of total apoB particles; with mild hypertriglyceridemia (triglycerides 1.5–3.0 mmol/l), they average 16 ± 0.06% of total apoB particles; but with moderate to marked hypertriglyceridemia (>3.00 mmol/l), they average 26 ± 0.09% of total apoB (6). These results agree with those reported by others (14–16). Thus, the proportion of total apoB particles that are VLDL particles varies importantly as triglyceride levels increase. Nevertheless, the number of LDL particles always exceeds the number of VLDL particles. That said, as VLDL particle number increases, so also does atherogenic risk.
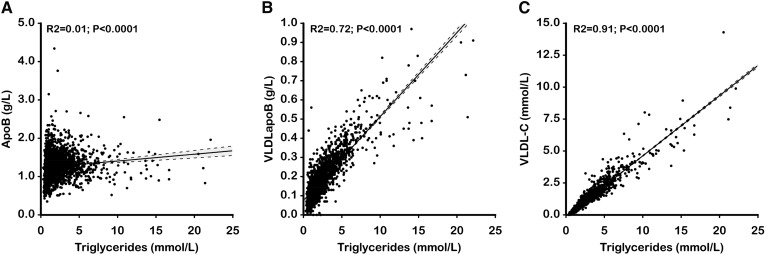
Nevertheless, because the triglyceride content of VLDL apoB particles is so variable, plasma apoB cannot be inferred from plasma triglycerides. This is evident from Fig. 2A, which plots triglycerides against apoB in 1,635 subjects, of whom 407 were normal, 33 were type IIa, and 735 were hypertriglyceridemic (26). There is no significant correlation between the two markers. Figure 2B demonstrates a reasonable correlation between VLDL apoB and plasma triglycerides. Nevertheless, total apoB cannot be reliably inferred from plasma triglycerides. This is relevant because multiple cross-sectional studies have shown that cardiovascular risk was greater in patients who were hypertriglyceridemic, but with elevated apoB, than in patients who were hypertriglyceridemic, but with normal apoB (27–32). This feature was also evident in the Quebec Heart Study, which was a prospective epidemiological study (33). Finally, the difference in LDL particle number and, therefore, total apoB also largely explains why cardiovascular risk is greater in hypertriglyceridemic subjects with familial combined hyperlipidemia than in hypertriglyceridemic subjects with familial hypertriglyceridemia (34). By contrast, as evident in Fig. 2C, there is a relatively strong correlation between plasma triglycerides and VLDL-C. This means that the relative cardiovascular risk associated with these two markers would be difficult, if not impossible, to distinguish one from the other.
Fig. 2.
Correlations between total apoB levels (A), VLDL apoB levels (B), VLDL-C levels (C), and plasma triglyceride levels in 2,023 patients (736 with IIa phenotype, 371 with IIb phenotype, 509 with IV phenotype, and 407 with normal lipid profile). The dashed lines indicate the 90% confidence intervals for the regression lines.
MENDELIAN RANDOMIZATION ANALYSES OF TRIGLYCERIDE AS A RISK FACTOR FOR CVD
Many conventional epidemiological analyses have demonstrated a positive relation between plasma triglycerides and coronary heart disease (CHD) risk. However, when adjusted for HDL-C or non-HDL-C, these relations were no longer significant. This should not be surprising given the high intercorrelations among the variables. Nevertheless, it creates uncomfortable uncertainty as to which relation may be causal and which may not. Mendelian randomization has been applied to resolve this uncertainty. Mendelian randomization tests to determine whether genetic variants that modify plasma levels of factors that may be related to a disease of interest modify the frequency of that disease. The core strength of such analyses is that the genetic assortment of alleles for the trait of interest is random and, therefore, should be independent of all other genetic and environmental factors. Inferring a causal relation by Mendelian randomization is reasonably straightforward if the genetic variant is related to only a single product. Any change in risk is presumed to be due only to a change in exposure to that factor. However, if a genetic variant were to affect multiple traits in different pathways (so-called horizontal pleiotropy), attributing causality to any one of these differences in exposure would be problematic. Alternatively, a genetic variant might result in multiple changes in factors within the same pathway (so-called vertical pleiotropy). Thus, causality can be assigned only to the pathway, but, without further information, not to any of the specific participants (35). This restriction applies to Mendelian randomization of the relation of triglycerides to cardiovascular risk.
Exome sequencing identifies rare LDLR and apoAV alleles conferring risk for myocardial infarction (36)
This study used exome sequencing to compare early onset myocardial infarction cases with older individuals without a history of myocardial infarction. Two genes associated with higher risk were identified, which were more frequent in cases than controls. As expected, rare loss-of-function variants in the LDLR that were associated with elevated LDL-C were more frequent in cases than controls. More novel was the observation that rare loss-of-function variants in apoAV associated with higher triglycerides were also more frequent in cases than controls. These apoAV mutants were associated with higher HDL-C and lower LDL-C. However, no data on apoB were reported in this study.
Triglyceride-mediated pathways and coronary disease: collaborative analysis of 101 studies (37)
apoAV is a 366 amino acid peptide, which associates with VLDL particles and has multiple effects on triglyceride metabolism, including an increase in LPL-driven triglyceride lipolysis and a reduction in triglyceride secretion without an effect on apoB secretion. The level of LDL-C was not affected by the −1131T>C (rs662799) promoter polymorphism of the C allele. The higher triglyceride (16%) per C allele was greater than the decrease in HDL-C (3.5%) and the increase in apoB (3.2%). However, the polymorphisms of the C allele were associated with an increase in VLDL particle size as well as VLDL particle number, and the relative importance of triglyceride versus VLDL particle number on risk was not assessed. Moreover, the positive effect of triglyceride on risk was lost after adjustment for non-HDL-C and HDL-C. The authors noted that these results are consistent with those of the Emerging Risk Factor Collaboration, which demonstrated that the relation between triglycerides and cardiovascular risk was positive in a univariate analysis, but not in a multivariate analysis (38). They concluded that their findings do not demonstrate that triglycerides per se increase cardiovascular risk, but rather that risk is increased by triglyceride-mediated pathways.
Mendelian randomization of blood lipids for coronary heart disease (39)
This study identified multiple independent SNPs for LDL-C, triglycerides, and HDL-C. These were incorporated into two allelic scores: an unrestricted score, which included all the independent SNPs, and a restricted score, which included only those alleles associated with one trait but not the other two. The unrestricted allele score is biologically more inclusive and, therefore, potentially more powerful. The restricted score is biologically more specific, but potentially less powerful. Combining prevalent and incident CHD, the odds ratio (OR) for LDL-C was significant in both restricted and unrestricted analyses, 1.92 (1.68, 2.19; P = 4.8 × 10−22) and 1.76 (1.58, 2.01; P = 2.0 × 10−21), respectively. The OR for triglyceride was significant in the unrestricted analysis, 1.62 (1.24, 2.11; P = 3.7 × 10−4) and unchanged numerically, but just significant in the restricted analysis, 1.61 (1.00–2.59; P = 0.05). The OR for HDL-C was significant in the unrestricted, but not the restricted, analysis. Multiple other analyses demonstrate the same relative relations to cardiovascular risk. The authors concluded that LDL-C was causal for cardiovascular risk; triglycerides were also likely causal, but HDL-C was unlikely to be causal. Once again, the conclusions regarding triglycerides and cardiovascular risk were provisional. Moreover, apoB was not included in the analysis.
Genetically elevated nonfasting triglycerides and calculated remnant cholesterol as causal risk factors for myocardial infarction (1)
Two genetic variants in the core promotor and coding regions of apoAV in individuals with the lowest (1%) and highest triglycerides (2%) were identified in the Copenhagen City Heart Study (CCHS) and their effect on risk determined in the Copenhagen General Population Study (CGPS), the CHHS, and the Copenhagen Ischemic Heart Disease Study (CIHDS). The hazard ratio (HR) for doubling of triglycerides was 1.57 (1.32–2.68) and was similar to a genetic OR of 1.94 (1.40–1.85). The absolute allele-related differences in triglycerides were greater than for apoB or HDL-C and correcting for apoB did not reduce the risk associated with triglycerides. Accordingly, they argue that the observed differences in risk should relate to triglycerides and not to HDL-C or apoB. This is not certain however. If cardiovascular risk is more closely related to apoB than triglycerides, any unit change in risk will be associated with a smaller change in apoB than in triglycerides. Moreover, apoB was higher in cases than controls in the CIHS, equal in CGPS, but substantially lower in cases than controls in the CIHS. The inconsistencies are disturbing given the mass of evidence that apoB is higher in those with CVD compared with those without CVD.
DOES LOWERING PLASMA TRIGLYCERIDES WITH FIBRATES REDUCE CARDIOVASCULAR RISK? EVIDENCE FROM THE FIBRATE RANDOMIZED CLINICAL TRIALS
We will now briefly review the seven fibrate randomized clinical trials and contrast the consistency of the triglyceride-lowering effect with the inconsistency of the clinical results.
The World Health Organization (WHO) trial tested whether clofibrate would reduce coronary events (40). Ischemic events were reduced significantly, but total mortality was increased. Triglycerides were not measured at baseline and were measured in only one of three sites during follow up. Therefore, this trial presents no evidence with regard to triglyceride. Cardiovascular benefit was reported to parallel the absolute reduction in total cholesterol.
The Coronary Drug Project tested whether clofibrate reduced coronary risk (41). Total cholesterol was reduced by 6.5%, while triglycerides were reduced by 22.3%. Cardiovascular risk was reduced by 9%, which was not significant. The outcome corresponds reasonably well to that predicted by the Cholesterol Treatment Trialists (CTT) (42).
The Helsinki Heart Study trial was designed to test the clinical benefit of elevating HDL-C and reducing non-HDL-C using gemfibrozil in markedly hypercholesterolemic patients (non-HDL-C ≥200 mg/dl) (43). CHD events were reduced by 34%; HDL-C was increased by 14%, although this diminished to about 10% over the study period. Total cholesterol was reduced by 10%, non-HDL-C by 14%, and triglycerides by 43%. The decrease in non-HDL-C would have been approximately 34 mg/dl, which would predict a 20% benefit, as estimated from the results of the REVEAL trial (44).
The Bezafibrate Infarction Prevention (BIP) study was a secondary prevention designed to reduce CVD risk by raising HDL-C and reducing triglycerides with bezafibrate therapy (45). Based on changes in statistical design that were made during the trial (switching from a one-sided to a two-sided t-test), this trial was underpowered with only a 60–85% chance of detecting a 20–25% event reduction. There was a 7.3% nonsignificant decrease in the primary endpoint. This was associated with a 21% decrease in triglycerides and an 18% increase in HDL-C, but only a 10 mg/dl decrease in LDL-C and a 16 mg/dl decrease in non-HDL-C. Based on the CTT results, neither the decrease in LDL-C (42) nor the decrease in non-HDL-C (44) would be predicted to produce a larger outcome than was observed.
The Veteran Affairs High-Density Lipoprotein Cholesterol Intervention (VA-HIT) trial was a secondary prevention trial designed to treat those with low HDL-C, but with a normal LDL-C (46). At 1 year, HDL-C was 6% higher and triglycerides 31% lower; total cholesterol was 4% less with no change in LDL-C. VLDL-C would have decreased from 32 to 23 mg/dl, a decrease of 9 mg/dl. Non-HDL-C would also have decreased by 9 mg/dl. The clinical improvement that was recorded was much greater than predicted by CTT (44). Interestingly, the benefit was the same in those with triglycerides above and below 151 mg/dl, whereas it was greater in those with HDL-C <31.5 versus those with HDL-C ≥31.5.
The Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) study was a trial in patients with type 2 diabetes mellitus that were tested to determine whether fenofibrate reduced cardiovascular risk (47). Overall, there was a nonsignificant trend to benefit with an 11% reduction [HR 0.89 (0.75–1.05)] in the combined primary endpoint. Total cholesterol was reduced by 11%, LDL-C by 12%, triglycerides by 29%, and HDL-C increased by 5%. apoB was reduced by 13.6% at 4 months, 7.5% overall at close, and 15.7% in those not taking any other lipid-lowering agent. Women made up 37.3% of the cohort and had higher levels of total cholesterol, non-HDL-C, LDL-C, and apoB, but not triglycerides. LDL-C and apoB were more substantially reduced in women than in men, although differences attenuated over the course of the study. apoB was reduced from 0.99 at onset in women to about 0.8. For women, the HR on Rx was 0.80 (P < 0.04), whereas it was 0.92 (P = NS) for men. Notably there was no sex interaction. The benefit in the women and the overall benefit are consistent with the changes in apoB (7).
The Action to Control Cardiovascular Risk in Diabetes (ACCORD)-Lipid trial was designed to test whether the combination of simvastatin-fenofibrate was more effective than statin alone in patients with type 2 diabetes mellitus (48). Triglycerides were reduced by 21% and HDL-C was increased by 6.5%, whereas LDL-C did not differ between the two treatment groups. The primary outcome was not significantly reduced in the statin-fenofibrate compared with the fenofibrate group (HR 0.92, 0.79–1.08; P = 0.32).
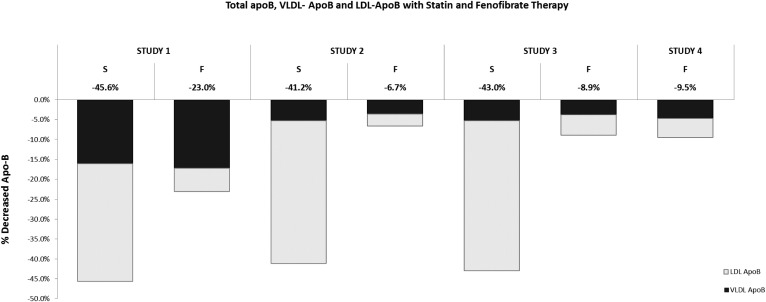
In summary, the clinical benefit of fibrate therapy was variable; ischemic events were reduced significantly in only three of the seven trials. However, in the six fibrate trials in which they were measured, there was a robust lowering of triglycerides. Thus, the triglyceride response to fibrates was consistent, whereas the clinical response was not. Moreover, with the exception of the VA-HIT trial, the degree of benefit was consistent with the benefit that would be predicted from the absolute reductions in LDL-C or non-HDL-C or apoB. These results argue against the triglyceride hypothesis: namely that when triglycerides are elevated, cardiovascular risk is elevated and that lowering triglycerides will, in itself and by itself, reduce cardiovascular risk and produce clinical benefit. On the other hand, this failure of the triglyceride hypothesis suggests an alternative one: namely, the apoB hypothesis, which states that the benefit of fibrate therapy is primarily due to lowering of the apoB atherogenic lipoproteins, which is reflected in lowering of plasma apoB. Figure 3 compares the overall effects of statins and fibrates on total plasma apoB, VLDL apoB, and LDL apoB (14–16, 49). Statins substantially lower both VLDL and LDL apoB. Hence, they substantially lower plasma total apoB. By contrast, fibrates substantially lower VLDL apoB, but only modestly reduce LDL apoB. The effect of statins and fibrates on VLDL apoB is similar, but their effects on LDL apoB are not. Fibrates are used in hypertriglyceridemic patients in whom absolute levels of VLDL apoB tend to be higher than normal. The net effect, therefore, is a modest to moderate lowering of total plasma apoB. The apoB hypothesis would account for why benefit from statin therapy is robust, whereas benefit from fibrate therapy is marginal and inconsistent because only if VLDL apoB accounts for a significant proportion of total apoB will there be a sufficient reduction in apoB to translate into clinical benefit.
Fig. 3.
Decreases in total apoB are illustrated from statin and fibrate arms of Study 1 (14), Study 2 (15), and Study 3 (16), and the fibrate treatment arm of Study 4 (49). The proportion of the decrease in total apoB due to the decrease in VLDL apoB is indicated in black, whereas the decrease in total apoB due to the decrease in LDL apoB is indicated in white. S, statin; F, fibrate.
NOVEL THERAPEUTIC AGENTS TO REDUCE CARDIOVASCULAR RISK
A series of novel and potentially important therapeutic agents, whose primary impact is to lower triglycerides, are in the process of clinical evaluation. These include inhibitors of apoCIII and angiopoietin-like 3 protein (ANGPTL3). The potential utility of such agents is buttressed by evidence that genetic inactivating variants have been associated with positive impacts on plasma triglycerides and, in some cases, cardiovascular risk. Their design and analysis has been based primarily on the simple triglyceride hypothesis. We will focus on the available information as to their impact on VLDL apoB, LDL apoB, and total apoB.
apoCIII
apoCIII is a 79 amino acid glycoprotein, which is synthesized in the liver and is associated with chylomicrons and VLDL, LDL, and HDL particles. apoCIII may influence the metabolism of apoB particles at multiple steps. apoCIII is a potent inhibitor of LPL activity, but may also inhibit hepatic lipase activity and hepatic uptake and clearance of triglyceride-rich remnant lipoprotein particles (50). Additionally, apoCIII may stimulate VLDL synthesis and secretion (51). Furthermore, a positive relation between plasma apoCIII and coronary risk has been demonstrated (22, 52), as well as between LDL-apoCIII and HDL-CIII and CVD risk (22). Indeed, LDL-apoCIII may account for much of the excess CVD risk associated with apoCIII and CVD (22). Further extending the complexity associated with apoCIII is the observation that the atherogenic risk of a VLDL apoB-CIII particle or a LDL apoB-CIII particle is inversely related to the apoE content of the particle (23). The evidence suggesting that reduction of apoCIII levels in plasma may reduce cardiovascular risk is based on the associated effects of spontaneous apoCIII mutations that produce apoCIII deficiency and the effects of pharmacological inhibitors of apoCIII.
apoCIII mutations producing apoCIII deficiency
Exome sequencing of 3,734 people by the TG and HDL Working Group of the Exome Sequencing Project of the National Heart, Lung, and Blood Institute identified rare reduction-in-function apoCIII mutations, which were associated with lower levels of triglyceride and lower levels of cardiovascular risk (53). apoCIII carriers also had lower levels of LDL-C than noncarriers. The correlation between apoCIII and plasma triglycerides was the strongest, by far, of any lipid marker, stronger than the correlation between apoCIII and VLDL particle number. There were, however, positive correlations between apoCIII and large VLDL particles, small LDL particles, and apoB with negative correlations to large LDL particles and large HDL particles, all pointing to the impact of apoCIII on LDL and HDL lipoprotein particles, perhaps secondarily, as well as its primary impact on VLDL particles. The impact on the apoB lipoprotein profile, therefore, was broad, not limited.
This interpretation is consistent with the results of the companion report by Jørgensen et al. (54) in the same issue of the New England Journal of Medicine, which demonstrated that loss of function mutations in apoCIII were associated with lower levels of triglycerides and cardiovascular risk in 75,725 subjects in two Danish population studies. Triglycerides in heterozygotes were reduced by 44%. apoB, however, was also reduced by 16%. Given that these were life-long exposures, the decrease in apoB could account for much of the reduction in cardiovascular risk. Taken together, these results indicate that reduction in risk with the apoCIII mutations could be due to reduction of apoB particle number as well as triglyceride. VLDL- and LDL-apoB were not measured directly.
Further evidence that apoCIII affects LDL as well as triglyceride levels comes from the observations of Pollin et al. (55) that a null mutation of apoCIII in Old Order Amish individuals was associated with lower levels of LDL-C, lower levels of fasting and postprandial triglycerides, higher HDL-C, and a lower prevalence of coronary calcification in carriers compared with noncarriers. apoB was not measured. By contrast, four homoygotes with a loss-of-function mutation of apoCIII in a population of Pakistani individuals had lower plasma triglyceride, higher HDL-C, but similar LDL-C (56).
apoCIII inhibitor studies
ISIS 30481 (Volanesorsen) is a second-generation antisense inhibitor of apoCIII synthesis. Administration of this agent to three patients with familial hyperchylomicronemia syndrome resulted in major decreases in plasma triglycerides, which were associated with substantial decreases in plasma apoCIII and plasma apoE and increases in plasma HDL-C (57). That the agent was effective in individuals who lack LPL argues for yet another major mechanism of action affecting triglyceride-rich lipoprotein particles. Data on apoB100 were not reported.
A larger exploratory study of the effects of ISIS 30481 in patients with moderate to marked, but not severe, hypertriglyceridemia, some of whom were on fibrate therapy, was undertaken by the same group (49). apoCIII levels dropped substantially in a dose-dependent manner in both groups and this was associated with a concordant drop in plasma triglycerides, but an increase in HDL-C. As well, LDL-C increased in a dose-dependent fashion. There was a dose-dependent decrease in apoB48. Data on apoB were reported in the supplement and separated based on whether ISIS 304801 was administered as monotherapy or with fibrate therapy. With monotherapy, there were generally increases in LDL-C. With combination therapy, LDL-C increased at the higher dose, but not at the lower dose. There were decreases in VLDL-C and VLDL apoCIII with both monotherapy and combination therapy. Total apoB was not reduced with monotherapy. With fibrate monotherapy, apoB was reduced by approximately 8%, but this change was not significant. Combination therapy did not produce a significant reduction in total apoB. apoB48 was reduced significantly by 20–60% with monotherapy and combination therapy. The absolute reduction in apoB48 particle number varied between 0.2 and 0.6 mg/dl. With monotherapy, there was a dose-dependent reduction of VLDL apoB, which was maximal at approximately 60%, representing an absolute decrease of approximately 12 mg/dl. By contrast with the decrease in VLDL apoB, there was a progressive increase in LDL apoB from just over 20% with the lowest dose to just over 45% with the highest monotherapy dose. The results with combination therapy with fibrates were inconsistent.
A study of ISIS 30481 in patients with type 2 diabetes and hypertriglyceridemia confirmed marked decreases in triglycerides and VLDL-C, as well as increases in HDL-C, but no change in LDL-C. Plasma apoB was lowered, but not significantly (58). Based on total apoB, the net effect of ISIS 30481 on cardiovascular risk appears to be neutral. Further work is necessary to clarify the extent to which the changes in lipid composition are independent of any changes in particle number, particularly in VLDL apoB. Given that apoB was reduced in subjects with mutations of apoCIII (54), the possibility that pharmacological interventions do not fully recapitulate all the biological mechanisms needs to be considered.
ANGPTL3
ANGPTL3 is an endogenous inhibitor of LPL. The DiscovEHR study examined the associations between loss-of-function variants and coronary artery risk in a large sampling of Americans (59). Carriers of loss-of-function variants had 27% lower triglyceride levels, 9% lower LDL-C levels, and 4% lower HDL-C levels. Carriers also had a 41% lower OR of coronary artery disease. A monoclonal antibody to ANGPTL3 reduced atherosclerotic lesion size in mice; while in human volunteers, triglycerides were reduced 76% at day 4 and directly measured LDL-C was reduced by 23.3% at day 15 and HDL-C by 18.4% at day 15. No data on apoB were presented. Graham et al. (60) demonstrated that antisense oligonucleotides that target Angptl3 mRNA produced dose-dependent decreases in ANGPTL3 protein, plasma triglycerides, LDL-C, non-HDL-C, and apoB, as well as reducing atherosclerosis in a mouse experimental model. Furthermore, in nine patients with homozygous familial hypercholesterolemia and, therefore, without functional LDL receptors, Gaudet et al. (61) confirmed that ANGPTL3 could produce a substantial reduction in LDL and apoB, an effect that in these patients must be mediated by a mechanism other than enhanced LDL receptor clearance. In this regard, the observations by Xu et al. (62), that RNAi-mediated silencing of ANGPTL3 in mouse livers resulted in reduced apoB secretion as well as increased uptake are of particular interest. Finally, familial combined hypolipidemia, which is characterized by profound reductions in plasma triglycerides and LDL-C, has been associated with nonsense mutations in ANGPTL3 (56, 63). From the data available, it seems likely that impairment of ANGPTL3 function affects LDL as well as VLDL levels and, therefore, benefit is unlikely to be mediated entirely by changes in triglycerides and could relate more closely to concurrent reduction in apoB.
SUMMARY
In summary, multiple metabolic links intricately interconnect VLDL and LDL metabolism, but neither plasma triglycerides nor LDL-C directly reflect VLDL and LDL particle number. Thus, changes in cardiovascular risk associated with changes in plasma triglycerides in the absence of changes in LDL-C are not interpretable without simultaneous measures of apoB particle number. The commonly made assumption that if LDL-C is not affected, clinical benefit must relate to differences in plasma triglycerides is not valid. Even if LDL-C is altered, this may not reflect the full quantitative impact of the intervention on LDL apoB. Indeed, there is no evidence yet that the benefits of lipid-lowering in general cannot be adequately accounted for by lowering of total apoB. It is unfortunate that apoB was not measured in so many of these studies. However, the absence of apoB data does not mean that apoB does not matter. As this review demonstrates, without apoB, the clinical significance of changes in lipids cannot be reliably determined.
Footnotes
Abbreviations:
- ANGPTL3
- angiopoietin-like 3 protein
- CETP
- cholesterol ester transfer protein
- CHD
- coronary heart disease
- CTT
- Cholesterol Treatment Trialists
- HDL-C
- HDL cholesterol
- HR
- hazard ratio
- LDL-C
- LDL cholesterol
- OR
- odds ratio
- VLDL-C
- VLDL cholesterol
A.D.S., J.D., J.T.W, and P.R.L. report that they have no conflicts of interest. P.C. has received funding in the last 5 years from the Canadian Institutes for Health Research, Agriculture and Agri-Food Canada (Growing Forward program supported by the Dairy Farmers of Canada, Canola Council of Canada, Flax Council of Canada, Dow Agrosciences, Dairy Research Institute, Dairy Australia, Danone Institute, Merck, Pfizer, Atrium Innovations, and Kaneka Corporation. S.S.M. is listed as a co-inventor on a pending patent filed by Johns Hopkins University for an algorithm to estimate LDL cholesterol; has served on scientific advisory boards for Quest Diagnostics, Sanofi/Regeneron, Amgen, and Akcea Therapeutics; and has received research support from the PJ Schafer Cardiovascular Research Fund, the David and June Trone Family Foundation, the American Heart Association, the Aetna Foundation, CASCADE FH, the Maryland Innovation Initiative, Google, and Apple. W.C.C. is self-employed and is in private practice at the Lipoprotein and Metabolic Disorders Institute, PLLC, and is also an employee of LabCorp and is a consultant and lecturer for Amarin, Amgen, and Kiowa. G.T. is a consultant for Ionis Pharma; on the Advisory boards of Ionis Pharma, Amgen, and Servier Canada; a member of the Speaker’s Bureau of Amgen, Sanofi, Servier Canada, and Boerhinger Ingelheim; and has received a research grant from Ionis Pharma.
REFERENCES
- 1.Jørgensen A. B., Frikke-Schmidt R., West A. S., Grande P., Nordestgaard B. G., and Tybjaerg-Hansen A.. 2013. Genetically elevated non-fasting triglycerides and calculated remnant cholesterol as causal risk factors for myocardial infarction. Eur. Heart J. 34: 1826–1833. [DOI] [PubMed] [Google Scholar]
- 2.Varbo A., Benn M., Tybjaerg-Hansen A., Jørgensen A. B., Frikke-Schmidt R., and Nordestgaard B. G.. 2013. Remnant cholesterol as a causal risk factor for ischemic heart disease. J. Am. Coll. Cardiol. 61: 427–436. [DOI] [PubMed] [Google Scholar]
- 3.Do R., Willer C. J., Schmidt E. M., Sengupta S., Gao C., Peloso G. M., Gustafsson S., Kanoni S., Ganna A., Chen J., et al. . 2013. Common variants associated with plasma triglycerides and risk for coronary artery disease. Nat. Genet. 45: 1345–1352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.De Graaf J., Couture P., and Sniderman A.. 2015. ApoB in Clinical Care. Springer, Houten, The Netherlands. [Google Scholar]
- 5.Cantey E. P., and Wilkins J. T.. 2018. Discordance between lipoprotein particle number and cholesterol content: an update. Curr. Opin. Endocrinol. Diabetes Obes. 25: 130–136. [DOI] [PubMed] [Google Scholar]
- 6.Sniderman A. D., Tremblay A., De Graaf J., and Couture P.. 2012. Phenotypes of hypertriglyceridemia caused by excess very-low-density lipoprotein. J. Clin. Lipidol. 6: 427–433. [DOI] [PubMed] [Google Scholar]
- 7.Thanassoulis G., Williams K., Ye K., Brook R., Couture P., Lawler P. R., De Graaf J., Furberg C. D., and Sniderman A.. 2014. Relations of change in plasma levels of LDL-C, non-HDL-C and apoB with risk reduction from statin therapy: a meta-analysis of randomized trials. J. Am. Heart Assoc. 3: e000759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Robinson J. G., Wang S., and Jacobson T. A.. 2012. Meta-analysis of comparison of effectiveness of lowering apolipoprotein B versus low-density lipoprotein cholesterol and nonhigh-density lipoprotein cholesterol for cardiovascular risk reduction in randomized trials. Am. J. Cardiol. 110: 1468–1476. [DOI] [PubMed] [Google Scholar]
- 9.Ference B. A., Kastelein J. J. P., Ginsberg H. N., Chapman M. J., Nicholls S. J., Ray K. K., Packard C. J., Laufs U., Brook R. D., Oliver-Williams C., et al. . 2017. Association of genetic variants related to CETP inhibitors and statins with lipoprotein levels and cardiovascular risk. JAMA. 318: 947–956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Borén J., and Williams K. J.. 2016. The central role of arterial retention of cholesterol-rich apolipoprotein-B-containing lipoproteins in the pathogenesis of atherosclerosis: a triumph of simplicity. Curr. Opin. Lipidol. 27: 473–483. [DOI] [PubMed] [Google Scholar]
- 11.Xiao C., Dash S., Morgantini C., and Lewis G. F.. 2016. Intravenous glucose acutely stimulates intestinal lipoprotein secretion in healthy humans. Arterioscler. Thromb. Vasc. Biol. 36: 1457–1463. [DOI] [PubMed] [Google Scholar]
- 12.Mori K., Ishida T., Yasuda T., Monguchi T., Sasaki M., Kondo K., Hasokawa M., Nakajima H., Haraguchi Y., Sun L., et al. . 2013. Fasting serum concentration of apolipoprotein B48 represents residual risks in patients with new-onset and chronic coronary artery disease. Clin. Chim. Acta. 421: 51–56. [DOI] [PubMed] [Google Scholar]
- 13.Park C. Y., Park J-Y., Choi J., Kim D. J., Park K. S., Yoon K-H., Lee M-K., and Park S. W.. 2016. Increased postprandial apolipoprotein B-48 level after a test meal in diabetic patients: a multicenter, cross-sectional study. Metabolism. 65: 843–851. [DOI] [PubMed] [Google Scholar]
- 14.Hogue J-C., Lamarche B., Deshaies Y., Tremblay A. J., Bergeron J., Gagné C., and Couture P.. 2008. Differential effect of fenofibrate and atorvastatin on in vivo kinetics of apolipoproteins B-100 and B-48 in subjects with type 2 diabetes mellitus with marked hypertriglyceridemia. Metabolism. 57: 246–254. [DOI] [PubMed] [Google Scholar]
- 15.Bilz S., Wagner S., Schmitz M., Bedynek A., Keller U., and Demant T.. 2004. Effects of atorvastatin versus fenofibrate on apoB-100 and apoA-I kinetics in mixed hyperlipidemia. J. Lipid Res. 45: 174–185. [DOI] [PubMed] [Google Scholar]
- 16.Watts G. F., Barrett P. H. R., Ji J., Serone A. P., Chan D. C., Croft K. D., Loehrer F., and Johnson A. G.. 2003. Differential regulation of lipoprotein kinetics by atorvastatin and fenofibrate in subjects with the metabolic syndrome. Diabetes. 52: 803–811. [DOI] [PubMed] [Google Scholar]
- 17.Hopkins P. N., Brinton E. A., and Nanjee M. N.. 2014. Hyperlipoproteinemia type 3: the forgotten phenotype. Curr. Atheroscler. Rep. 16: 440. [DOI] [PubMed] [Google Scholar]
- 18.Marais D. 2015. Dysbetalipoproteinemia: an extreme disorder of remnant metabolism. Curr. Opin. Lipidol. 26: 292–297. [DOI] [PubMed] [Google Scholar]
- 19.Drouin-Chartier J. P., Hogue J. C., Tremblay A. J., Bergeron J., Lamarche B., and Couture P.. 2017. The elevation of plasma concentrations of apoB-48-containing lipoproteins in familial hypercholesterolemia is independent of PCSK9 levels. Lipids Health Dis. 16: 119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sniderman A., Tremblay A., Bergeron J., Gagné C., and Couture P.. 2007. Diagnosis of type III hyperlipoproteinemia from plasma total cholesterol, triglyceride, and apolipoprotein B. J. Clin. Lipidol. 1: 256–263. [DOI] [PubMed] [Google Scholar]
- 21.Mora S., Szklo M., Otvos J. D., Greenland P., Psaty B. M., Goff D. C., O’Leary D. H., Saad M. F., Tsai M. Y., and Sharrett A. R.. 2007. LDL particle subclasses, LDL particle size, and carotid atherosclerosis in the Multi-Ethnic Study of Atherosclerosis (MESA). Atherosclerosis. 192: 211–217. [DOI] [PubMed] [Google Scholar]
- 22.Mendivil C. O., Rimm E. B., Furtado J., Chiuve S. E., and Sacks F. M.. 2011. Low-density lipoproteins containing apolipoprotein C-III and the risk of coronary heart disease. Circulation. 124: 2065–2072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mendivil C. O., Rimm E. B., Furtado J., and Sacks F. M.. 2013. Apolipoprotein E in VLDL and LDL with apolipoprotein C-III is associated with a lower risk of coronary heart disease. J. Am. Heart Assoc. 2: e000130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tsimikas S. 2017. A test in context: lipoprotein(a): diagnosis, prognosis, controversies, and emerging Therapies. J. Am. Coll. Cardiol. 69: 692–711. [DOI] [PubMed] [Google Scholar]
- 25.Afshar M., and Thanassoulis G.. 2017. Lipoprotein(a): new insights from modern genomics. Curr. Opin. Lipidol. 28: 170–176. [DOI] [PubMed] [Google Scholar]
- 26.Sniderman A. D., Tremblay A. J., De Graaf J., and Couture P.. 2014. Calculation of LDL apoB. Atherosclerosis. 234: 373–376. [DOI] [PubMed] [Google Scholar]
- 27.Durrington P. N., Bolton C. H., and Hartog M.. 1978. Serum and lipoprotein apolipoprotein B levels in normal subjects and patients with hyperlipoproteinaemia. Clin. Chim. Acta. 82: 151–160. [DOI] [PubMed] [Google Scholar]
- 28.Sniderman A., Vu H., and Cianflone K.. 1991. Effect of moderate hypertriglyceridemia on the relation of plasma total and LDL apo B levels. Atherosclerosis. 89: 109–116. [DOI] [PubMed] [Google Scholar]
- 29.Sniderman A. D., Wolfson C., Teng B., Franklin F. A., Bachorik P. S., and Kwiterovich P. O.. 1982. Association of hyperapobetalipoproteinemia with endogenous hypertriglyceridemia and atherosclerosis. Ann. Intern. Med. 97: 833–839. [DOI] [PubMed] [Google Scholar]
- 30.Durrington P. N., Hunt L., Ishola M., Kane J., and Stephens W. P.. 1986. Serum apolipoproteins AI and B and lipoproteins in middle aged men with and without previous myocardial infarction. Br. Heart J. 56: 206–212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Barbir M., Wile D., Trayner I., Aber V. R., and Thompson G. R.. 1988. High prevalence of hypertriglyceridaemia and apolipoprotein abnormalities in coronary artery disease. Br. Heart J. 60: 397–403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kukita H., Hamada M., Hiwada K., and Kokubu T.. 1985. Clinical significance of measurements of serum apolipoprotein A-I, A-II and B in hypertriglyceridemic male patients with and without coronary artery disease. Atherosclerosis. 55: 143–149. [DOI] [PubMed] [Google Scholar]
- 33.Lamarche B., Després J. P., Moorjani S., Cantin B., Dagenais G. R., and Lupien P. J.. 1995. Prevalence of dyslipidemic phenotypes in ischemic heart disease (prospective results from the Québec Cardiovascular Study). Am. J. Cardiol. 75: 1189–1195. [DOI] [PubMed] [Google Scholar]
- 34.Brunzell J. D., Schrott H. G., Motulsky A. G., and Bierman E. L.. 1976. Myocardial infarction in the familial forms of hypertriglyceridemia. Metabolism. 25: 313–320. [DOI] [PubMed] [Google Scholar]
- 35.Holmes M. V., Ala-Korpela M., and Smith G. D.. 2017. Mendelian randomization in cardiometabolic disease: challenges in evaluating causality. Nat. Rev. Cardiol. 14: 577–590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Do R., Stitziel N. O., Won H. H., Jørgensen A. B., Duga S., Angelica Merlini P., Kiezun A., Farrall M., Goel A., Zuk O., et al. . 2015. Exome sequencing identifies rare LDLR and APOA5 alleles conferring risk for myocardial infarction. Nature. 518: 102–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Triglyceride Coronary Disease Genetics Consortium and Emerging Risk Factors Collaboration, Sarwar N., Sandhu M. S., Ricketts S. L., Butterworth A. S., Di Angelantonio E., Boekholdt S. M., Ouwehand W., Watkins H., Samani N. J., et al. . 2010. Triglyceride-mediated pathways and coronary disease: collaborative analysis of 101 studies. Lancet. 375: 1634–1639. [Erratum. 2010. Lancet. 376: 90.] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Emerging Risk Factors Collaboration, Di Angelantonio E., Sarwar N., Perry P., Kaptoge S., Ray K. K., Thompson A., Wood A. M., Lewington S., Sattar N., et al. . 2009. Major lipids, apolipoproteins, and risk of vascular disease. JAMA. 302: 1993–2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Holmes M. V., Asselbergs F. W., Palmer T. M., Drenos F., Lanktree M. B., Nelson C. P., Dale C. E., Padmanabhan S., Finan C., Swerdlow D. I., et al. . 2015. Mendelian randomization of blood lipids for coronary heart disease. Eur. Heart J. 36: 539–550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.1978. A co-operative trial in the primary prevention of ischaemic heart disease using clofibrate. Report from the Committee of Principal Investigators. Br. Heart J. 40: 1069–1118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.1975. Clofibrate and niacin in coronary heart disease. JAMA. 231: 360–381. [PubMed] [Google Scholar]
- 42.Cholesterol Treatment Trialists’ (CTT) Collaboration, Fulcher J., O’Connell R., Voysey M., Emberson J., Blackwell L., Mihaylova B., Simes J., Collins R., Kirby A., et al. . 2015. Efficacy and safety of LDL-lowering therapy among men and women: meta-analysis of individual data from 174,000 participants in 27 randomised trials. Lancet. 385: 1397–1405. [DOI] [PubMed] [Google Scholar]
- 43.Frick M. H., Elo O., Haapa K., Heinonen O. P., Heinsalmi P., Helo P., Huttunen J. K., Kaitaniemi P., Koskinen P., and Manninen V.. 1987. Helsinki Heart Study: primary-prevention trial with gemfibrozil in middle-aged men with dyslipidemia. Safety of treatment, changes in risk factors, and incidence of coronary heart disease. N. Engl. J. Med. 317: 1237–1245. [DOI] [PubMed] [Google Scholar]
- 44.HPS3/TIMI55–REVEAL Collaborative Group, Bowman L., Hopewell J. C., Chen F., Wallendszus K., Stevens W., Collins R., Wiviott S. D., Cannon C. P., Braunwald E., et al. . 2017. Effects of anacetrapib in patients with atherosclerotic vascular disease. N. Engl. J. Med. 377: 1217–1227. [DOI] [PubMed] [Google Scholar]
- 45. Bezafibrate Infarction Prevention (BIP) Study. 2000. Secondary prevention by raising HDL cholesterol and reducing triglycerides in patients with coronary artery disease. Circulation. 102: 21–27. [DOI] [PubMed] [Google Scholar]
- 46.Rubins H. B., Robins S. J., Collins D., Fye C. L., Anderson J. W., Elam M. B., Faas F. H., Linares E., Schaefer E. J., Schectman G., et al. . 1999. Gemfibrozil for the secondary prevention of coronary heart disease in men with low levels of high-density lipoprotein cholesterol. Veterans Affairs High-Density Lipoprotein Cholesterol Intervention Trial Study Group. N. Engl. J. Med. 341: 410–418. [DOI] [PubMed] [Google Scholar]
- 47.Keech A., Simes R. J., Barter P., Best J., Scott R., Taskinen M. R., Forder P., Pillai A., Davis T., Glasziou P., et al. ; FIELD Study Investigators. 2005. Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomised controlled trial. Lancet. 366: 1849–1861. [DOI] [PubMed] [Google Scholar]
- 48.ACCORD Study Group, Ginsberg H. N., Elam M. B., Lovato L. C., Crouse J. R., Leiter L. A., Linz P., Friedewald W. T., Buse J. B., Gerstein H. C., et al. . 2010. Effects of combination lipid therapy in type 2 diabetes mellitus. N. Engl. J. Med. 362: 1563–1574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Gaudet D., Alexander V. J., Baker B. F., Brisson D., Tremblay K., Singleton W., Geary R. S., Hughes S. G., Viney N. J., Graham M. J., et al. . 2015. Antisense inhibition of apolipoprotein C-III in patients with hypertriglyceridemia. N. Engl. J. Med. 373: 438–447. [DOI] [PubMed] [Google Scholar]
- 50.Huff M. W., and Hegele R. A.. 2013. Apolipoprotein C-III: going back to the future for a lipid drug target. Circ. Res. 112: 1405–1408. [DOI] [PubMed] [Google Scholar]
- 51.Yao Z. 2012. Human apolipoprotein C-III - a new intrahepatic protein factor promoting assembly and secretion of very low density lipoproteins. Cardiovasc. Hematol. Disord. Drug Targets. 12: 133–140. [DOI] [PubMed] [Google Scholar]
- 52.Sacks F. M., Alaupovic P., Moyé L. A., Cole T. G., Sussex B., Stampfer M. J., Pfeffer M. A., and Braunwald E.. 2000. VLDL, apolipoproteins B, CIII, and E, and risk of recurrent coronary events in the Cholesterol and Recurrent Events (CARE) trial. Circulation. 102: 1886–1892. [DOI] [PubMed] [Google Scholar]
- 53.TG and HDL Working Group of the Exome Sequencing Project, National Heart, Lung, and Blood Institute, Crosby J., Peloso G. M., Auer P. L., Crosslin D. R., Stitziel N. O., Lange L. A., Lu Y., Tang Z. Z., Zhang H., et al. 2014. Loss-of-function mutations in APOC3, triglycerides, and coronary disease. N. Engl. J. Med. 371: 22–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Jørgensen A. B., Frikke-Schmidt R., Nordestgaard B. G., and Tybjaerg-Hansen A.. 2014. Loss-of-function mutations in APOC3 and risk of ischemic vascular disease. N. Engl. J. Med. 371: 32–41. [DOI] [PubMed] [Google Scholar]
- 55.Pollin T. I., Damcott C. M., Shen H., Ott S. H., Shelton J., Horenstein R. B., Post W., McLenithan J. C., Bielak L. F., Peyser P. A., et al. . 2008. A null mutation in human APOC3 confers a favorable plasma lipid profile and apparent cardioprotection. Science. 322: 1702–1705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Saleheen D., Natarajan P., Armean I. M., Zhao W., Rasheed A., Khetarpal S. A., Won H-H., Karczewski K. J., O’Donnell-Luria A. H., Samocha K. E., et al. . 2017. Human knockouts and phenotypic analysis in a cohort with a high rate of consanguinity. Nature. 544: 235–239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Gaudet D., Brisson D., Tremblay K., Alexander V. J., Singleton W., Hughes S. G., Geary R. S., Baker B. F., Graham M. J., Crooke R. M., et al. . 2014. Targeting APOC3 in the familial chylomicronemia syndrome. N. Engl. J. Med. 371: 2200–2206. [DOI] [PubMed] [Google Scholar]
- 58.Digenio A., Dunbar R. L., Alexander V. J., Hompesch M., Morrow L., Lee R. G., Graham M. J., Hughes S. G., Yu R., Singleton W., et al. . 2016. Antisense-mediated lowering of plasma apolipoprotein C-III by volanesorsen improves dyslipidemia and insulin sensitivity in type 2 diabetes. Diabetes Care. 39: 1408–1415. [DOI] [PubMed] [Google Scholar]
- 59.Dewey F. E., Gusarova V., Dunbar R. L., O’Dushlaine C., Schurmann C., Gottesman O., McCarthy S., Van Hout C. V., Bruse S., Dansky H. M., et al. . 2017. Genetic and pharmacologic inactivation of ANGPTL3 and cardiovascular disease. N. Engl. J. Med. 377: 211–221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Graham M. J., Lee R. G., Brandt T. A., Tai L. J., Fu W., Peralta R., Yu R., Hurh E., Paz E., McEvoy B. W., et al. . 2017. Cardiovascular and metabolic effects of ANGPTL3 antisense oligonucleotides. N. Engl. J. Med. 377: 222–232. [DOI] [PubMed] [Google Scholar]
- 61.Gaudet D., Gipe D. A., Pordy R., Ahmad Z., Cuchel M., Shah P. K., Chyu K-Y., Sasiela W. J., Chan K-C., Brisson D., et al. . 2017. ANGPTL3 inhibition in homozygous familial hypercholesterolemia. N. Engl. J. Med. 377: 296–297. [DOI] [PubMed] [Google Scholar]
- 62.Xu Y-X., Redon V., Yu H., Querbes W., Pirruccello J., Liebow A., Deik A., Trindade K., Wang X., Musunuru K., et al. . 2018. Role of angiopoietin-like 3 (ANGPTL3) in regulating plasma level of low-density lipoprotein cholesterol. Atherosclerosis. 268: 196–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Musunuru K., Pirruccello J. P., Do R., Peloso G. M., Guiducci C., Sougnez C., Garimella K. V., Fisher S., Abreu J., Barry A. J., et al. . 2010. Exome sequencing, ANGPTL3 mutations, and familial combined hypolipidemia. N. Engl. J. Med. 363: 2220–2227. [DOI] [PMC free article] [PubMed] [Google Scholar]