Abstract
Survival rates of children with acute lymphoblastic leukemia have improved since the incorporation of asparaginase in the treatment protocol, but the medication has potential serious complications, including vascular thrombosis. Here, we describe the case of a 13-year-old boy with pre-T-cell acute lymphoblastic leukemia whose treatment course was complicated by perforated jejunitis requiring resection of a portion of his small bowel. Pathologic assessment showed transmural ischemia, mesenteric venous and arterial thrombi, and scattered cytomegalovirus inclusion bodies. Pediatric mesenteric ischemia is rare, and its consideration in patients treated with asparaginase is discussed.
Keywords: Acute lymphoblastic leukemia, Pediatric, Asparaginase, Complications, Acute mesenteric ischemia, Computed tomography
Introduction
Acute lymphoblastic leukemia (ALL) is the most common cancer diagnosed in children, with an incidence of approximately 3.9% among individuals younger than 19 years and a mortality rate of 0.3 per 100,000 in the United States [1]. Reported toxicities in patients undergoing chemotherapy for ALL are high [2], and gastrointestinal (GI) complications such as diarrhea and neutropenic colitis are particularly common [3]. The long-term survival of patients with ALL has improved substantially since the introduction of asparaginase therapy [4]. However, this therapy can be associated with many serious toxicities, including an increased propensity for thrombosis [5]. We present a case of an adolescent with pre-T-cell ALL who was found to have perforated jejunitis during induction chemotherapy. Pathologic assessment of the resected bowel demonstrated changes associated with transmural ischemia, as well as focal mesenteric venous and arterial thrombi and scattered cytomegalovirus (CMV) inclusion bodies. This case illustrates the complex pathophysiology of jejunitis in a child undergoing induction chemotherapy for ALL and allows for a discussion about both mesenteric ischemia and CMV enteritis in children.
Case report
A 13-year-old boy with a recent diagnosis of pre-T-cell ALL presented with severe worsening abdominal pain that began 2 weeks after initiation of chemotherapy. The boy was undergoing induction chemotherapy per the standard arm of Children's Oncology Group study AALL1231 that includes dexamethasone, daunorubicin, vincristine, and intensified pegaspargase (given on days 4 and 18). On day 14 of induction, the patient developed epigastric abdominal pain and was found to have lost 5 kg since his initial ALL diagnosis. Abdominal pain and nutritional status initially improved with optimization of proton pump inhibition and initiation of nasogastric feeds. However, on day 25 of induction, the patient presented to the emergency department with increasing epigastric pain and multiple episodes of nonbilious, nonbloody emesis.
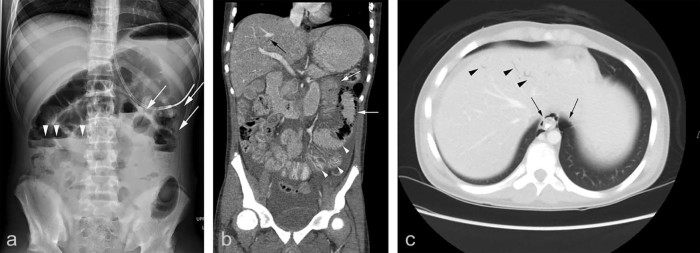
On examination, the patient was afebrile and normotensive. He had left periumbilical tenderness, without peritoneal signs. Significant laboratory results included an absolute neutrophil count of 3100 cells/µL, elevated serum lactate, and worsening metabolic acidosis. Abdominal radiograph (Fig. 1A) revealed multiple air-fluid levels, suggestive of ileus or early bowel obstruction, as well as probable pneumatosis in the left midabdomen. Contrast-enhanced computed tomography (CT) performed in standard portal venous phase confirmed ileus and pneumatosis intestinalis in the jejunum, with associated bowel wall thickening and segments of mucosal hypoenhancement and hyperenhancement (Fig. 1B). Simple-appearing ascites was seen. There was portal venous gas, along with small amounts of ectopic air dissecting along the mesenteric root into the esophageal or periesophageal region of the lower posterior mediastinum (Fig. 1C). Mesenteric vasculature was normal, without obvious CT evidence of arterial or venous thrombosis, although CT angiography was not performed.
Fig. 1.
Abdominal imaging acquired on day 14 of induction chemotherapy after onset of abdominal pain showing jejunal pneumatosis and dissection of gas through the mesentery into the retroperitoneum and the portal venous system. (A) The upright anterior-posterior abdominal radiograph shows pneumatosis in the left upper quadrant (white arrows), as well as air-fluid levels in mildly dilated loops of bowel (white arrowheads). Contrast-enhanced computed tomography images in (B) the coronal plane with soft tissue algorithm and in (C) the axial plane with lung algorithm shows pneumatosis involving an abnormally dilated segment of jejunum (white arrows in B) and abnormally enhancing mucosa in the jejunum (white arrowheads in B). Abnormal retroperitoneal air is present at the diaphragmatic hiatus (black arrows in C), and portal venous gas is also observed (black arrow in B, black arrowheads in C).
Two days after admission (day 26 of induction), the patient developed rebound tenderness, and the abdominal radiographs revealed pneumoperitoneum. A jejunal perforation was found during emergent and exploratory laparotomy. Two separate 30- to 35-cm segments of jejunum with gangrenous necrosis were resected, and the abdomen was left open. Re-exploration and washout of the abdomen were performed 2 days later, along with end-to-end anastomoses of 2 of the 3 jejunal segments and creation of mucus fistulae.
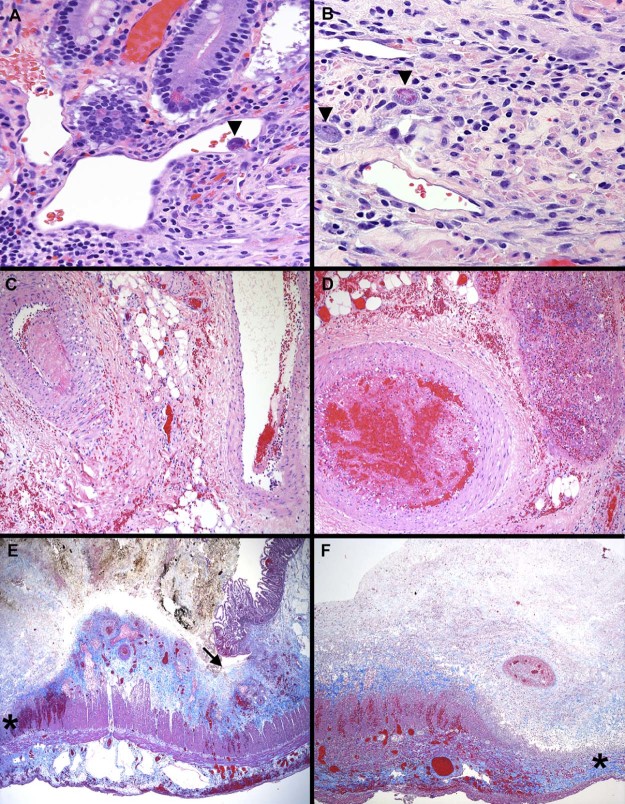
The resected bowel segments (Fig. 2) showed multiple foci of geographic transmural or mucosal necrosis with bile-stained bacteria-rich fibrinopurulent exudates borne between areas of residual mucosa. Several submucosal vessels near the ulcers were thrombosed. Few scattered CMV inclusions were identified in mucosal and submucosal endothelial and stromal cells. Occasional partially occlusive fibrin thrombi were seen within few mesenteric vessels; large feeding arteries were patent.
Fig. 2.
Cytomegalovirus inclusions (arrowheads) were identified in the endothelial cells of the mucosa (A) and in the submucosal stromal cells (B) (hematoxylin and eosin, 200×). Mesenteric arteries (C, D, left) have fibrin thrombi at varying stages of organization, adherent to the walls, and are variably occluding the lumens. A strand of fibrin clings to the vein wall (C, right). Fibrin and red and white blood cells obstruct a mesenteric vein (D, right) (hematoxylin and eosin, 100×). Low-power images show a small segment of residual mucosa adjacent to an ulcer bed (arrow) that is covered by bile-stained fibrinous exudate, resembling a pseudomembrane (E). There is partial necrosis of the muscularis propria (*) and a fibrotic serosa that contains dilated lymphatic spaces (E). Diffuse ulceration is associated with full-thickness necrosis (*, F) (Masson trichrome, 40×).
The patient was treated with postoperative broad-spectrum antibiotics for non-neutropenic enterocolitis and ganciclovir (6 weeks) for CMV colitis. Jejunostomy takedown was uncomplicated, and the patient was discharged from the hospital tolerating full enteral feeds. The end of induction assessment for minimal residual disease (<0.01%) was negative for residual leukemia in the bone marrow.
Discussion
We present a rare case of non-neutropenic jejunal necrosis and perforation, in the setting of immunosuppression by chemotherapy, including pegaspargase. We believe the etiology of this patient's intestinal issues was multifactorial with contributions from pegaspargase-related hypercoagulability resulting in mesenteric ischemia and concurrent CMV.
The overall survival rates of children with ALL have improved substantially since the incorporation of asparaginase therapy into treatment protocols [6], [7]. However, this therapy's known toxicities include altered coagulation and fibrinolysis, which increase a patient's risk of thrombosis and bleeding, especially during induction. The overall incidence of thrombotic complications in children with ALL is 5.2%, and usually, these complications are intracranial thrombotic events or catheter-related thrombi [5], [8]. To the best of our knowledge, mesenteric thrombotic events have not been ascribed to pegaspargase therapy in published literature. However, the temporal association of the medication with the small intestine ischemia and perforation in this case may implicate pegaspargase-induced hypercoagulability in its development.
A hypercoagulable state can lead to mesenteric arterial thrombosis or thromboembolism or mesenteric venous thrombosis. The published descriptions of mesenteric ischemia in children are quite limited. In both adults and children, contrast-enhanced CT of the abdomen and pelvis is appropriate [9], [10], [11], [12], [13]. Although dual-phase (arterial and venous) scanning protocols are commonly used in adults, repeat CT scanning of pediatric patients is typically avoided to reduce exposure to ionizing radiation [14]. In a review of 24 adults with acute mesenteric ischemia (AMI) who underwent single venous-phase CT, sensitivity and specificity for detection of mesenteric ischemia were 0.79 and 0.98, respectively [10]. The contrast-enhanced CT findings of AMI can vary, depending on the stage of disease. If the etiology is thromboembolism or thrombosis, filling defects may be seen in mesenteric vessels, although these were not discretely seen in our patient's CT findings. Bowel wall enhancement can be present with vasodilatory effects or can be absent with vasoconstriction and does not reflect ischemic severity [13]. Bowel wall thickening and dilation of bowel loops are common with ileus, but are nonspecific findings, as with this patient. With mucosal necrosis, intraluminal gas dissects into the wall, causing pneumatosis and possibly subsequent pneumoperitoneum and ascites [9], [10], [12], [13]. Notably, the CT findings associated with inflammatory or infectious arteritis overlap with those of ischemic enteritis. In the absence of discernible thrombi or emboli, CT has a low diagnostic utility for distinguishing the cause of intestinal abnormalities. Likewise, the clinical presentation of AMI is also nonspecific, as are the associated laboratory findings, such as elevated lactate and metabolic acidosis [13], [15]. Therefore, diagnostic imaging serves as the most practical and most rapidly acquired means for diagnosis in these cases, with the greatest impact on outcome due to preinfarction diagnosis [16].
The likely contribution of CMV to this patient's disease cannot be overlooked. Primary CMV infection is rarely the cause of jejunal perforation [17], [18], [19]. Based on reported clinical experiences with adult patients, the colon and the upper GI tract are the typical sites of involvement [18], [20], [21]. However, primary CMV infection has been associated with small bowel perforation in both adults and children [17], [18], [19], [22]. Latent CMV reactivates locally at sites of inflammation [23] and has been associated with vascular angiogenesis and endothelial damage that triggers thrombosis [24], [25]. In the rare reports of jejunal perforation in the setting of CMV infection, mesenteric thrombi or ulcerations can be seen [17], [18], [19].
The presentation of CMV-related GI infection can vary. Although CMV manifests typically in immunosuppressed individuals, vascular damage and thrombosis have also been reported in immunocompetent patients with acute CMV [20], [21], [26], [27]. In a review of 38 pediatric patients with symptomatic and histologically confirmed active CMV GI disease (median age of 5 months, ranging from 3 days to 12 years), 18% of the patients presented with clinical signs of peritonitis from perforation [22]. All 38 patients had impaired immunity, and 63% were malnourished. Additionally, in 10 of the 38 patients, a preceding local inflammatory insult was documented, such as surgery or necrotizing enterocolitis. Inflammatory polyps, ulceration, and mucosal sloughing are typical, in addition to perforation in the small bowel. Although our patient was not neutropenic, he had recent diarrhea preceding his jejunal disease. It is conceivable that in our patient, a local inflammatory cascade resulting from asparaginase-related ischemia triggered reactivation of latent CMV in the jejunum.
The etiology of perforated jejunitis in our patient was most likely multifactorial. In retrospect, had the index of suspicion for mesenteric ischemia been high, given the recent use of asparaginase and the associated risk of thrombotic complications, a dedicated CT angiography might have been performed to search for filling defects in the peripheral mesenteric vessels. Additionally, consideration of mesenteric ischemia as a contributing factor to jejunitis may have lowered the threshold for exploratory laparotomy.
References
- 1.Howlader N., Noone A.M., Krapcho M., Miller D., Bishop K., Kosary C.L. National Cancer Institute; Bethesda, MD: 1975-2014. SEER cancer statistics review.https://seer.cancer.gov/csr/1975_2014/ editors. based on November 2016 SEER data submission, posted to the SEER web site, April 2017. [Google Scholar]
- 2.Schmiegelow K., Müller K., Mogensen S.S., Mogensen P.R., Wolthers B.O., Stoltze U.K. Non-infectious chemotherapy-associated acute toxicities during childhood acute lymphoblastic leukemia therapy. F1000Res. 2017;6:444. doi: 10.12688/f1000research.10768.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Board PS and PCE Gastrointestinal Complications (PDQ®)–Health Professional Version. 2018. http://www.cancer.gov/about-cancer/treatment/side-effects/constipation/GI-complications-hp-pdq [accessed 03.18]
- 4.Egler R.A., Ahuja S.P., Matloub Y. L-Asparaginase in the treatment of patients with acute lymphoblastic leukemia. J Pharmacol Pharmacother. 2016;7(2):62–71. doi: 10.4103/0976-500X.184769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hijiya N., Van Der Sluis I.M. Asparaginase-associated toxicity in children with acute lymphoblastic leukemia. Leuk Lymphoma. 2016;57(4):748–757. doi: 10.3109/10428194.2015.1101098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Muller H., Boos J. Use of L-asparaginase in childhood ALL. Crit Rev Oncol Hematol. 1998;28:97–113. doi: 10.1016/s1040-8428(98)00015-8. [DOI] [PubMed] [Google Scholar]
- 7.Riccardi R., Holcenberg J., Glaubiger D., Wood J., Poplack D. L-asparaginase pharmacokinetics and asparagine levels in cerebrospinal fluid of rhesus monkeys and humans. Cancer Res. 1981;41:4554–4558. [PubMed] [Google Scholar]
- 8.Caruso V., Iacoviello L., Di Castelnuovo A., Storti S., Mariani G., de Gaetano G. Thrombotic complications in childhood acute lymphoblastic leukemia: a meta-analysis of 17 prospective studies comprising 1752 pediatric patients. Blood. 2006;108(7):2216–2222. doi: 10.1182/blood-2006-04-015511. [DOI] [PubMed] [Google Scholar]
- 9.Wiesner W., Khurana B., Ji H., Rox P. CT of acute bowel ischemia. Radiology. 2003;226:635–650. doi: 10.1148/radiol.2263011540. [DOI] [PubMed] [Google Scholar]
- 10.Wiesner W., Hauser A., Steinbrich W. Accuracy of multidetector row computed tomography for the diagnosis of acute bowel ischemia in a non-selected study population. Eur Radiol. 2004;14(12):2347–2356. doi: 10.1007/s00330-004-2462-6. [DOI] [PubMed] [Google Scholar]
- 11.Blachar A., Barnes S., Adam S.Z., Levy G., Weinstein I., Precel R. Radiologists' performance in the diagnosis of acute intestinal ischemia, using MDCT and specific CT findings, using a variety of CT protocols. Emerg Radiol. 2011;18(5):385–394. doi: 10.1007/s10140-011-0965-4. [DOI] [PubMed] [Google Scholar]
- 12.Angelelli G., Scardapane A., Memeo M., Stabile Ianora A.A., Rotondo A. Acute bowel ischemia: CT findings. Eur J Radiol. 2004;50(1):37–47. doi: 10.1016/j.ejrad.2003.11.013. [DOI] [PubMed] [Google Scholar]
- 13.Sandstrom C.K., Ingraham C.R., Monroe E.J., Johnson G.E. Beyond decreased bowel enhancement: acute abnormalities of the mesenteric and portal vasculature. Abdom Imaging. 2015;40(8):2977–2992. doi: 10.1007/s00261-015-0498-5. [DOI] [PubMed] [Google Scholar]
- 14.Miglioretti D.L., Johnson E., Williams A., Greenlee R.T., Weinmann S., Solberg L.I. The use of computed tomography in pediatrics and the associated radiation exposure and estimated cancer risk. JAMA Pediatr. 2013;167(8):700–707. doi: 10.1001/jamapediatrics.2013.311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Evennett N., Petrov M., Mittal A., Windsor J. Systematic review and pooled estimates for the diagnostic accuracy of serological markers for intestinal ischemia. World J Surg. 2009;33(7):1374–1383. doi: 10.1007/s00268-009-0074-7. [DOI] [PubMed] [Google Scholar]
- 16.Brandt L., Boley S. AGA technical review on intestinal ischemia. Gastroenterology. 2000;118:954–968. doi: 10.1016/s0016-5085(00)70183-1. [DOI] [PubMed] [Google Scholar]
- 17.Nabeshima K., Sakaguchi E., Inoue S., Eizuru Y., Minamishima Y., Koono M. Jejunal perforation associated with cytomegalovirus infection in a patient with adult T-cell leukemia-lymphoma. Acta Pathol Jpn. 1992;42(4):267–271. doi: 10.1111/j.1440-1827.1992.tb02540.x. [DOI] [PubMed] [Google Scholar]
- 18.Jun Y.J., Sim J., Ahn H.I., Han H., Kim H., Yi K. Cytomegalovirus enteritis with jejunal perforation in a patient with endometrial adenocarcinoma. World J Clin Cases. 2013;1(7):220–223. doi: 10.12998/wjcc.v1.i7.220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.DeRiso A.J., Kemeny M., Torres R., Oliver J. Multiple jejunal perforations secondary to cytomegalovirus in a patient with acquired immune deficiency syndrome. Dig Dis Sci. 1989;34(4):623–629. doi: 10.1007/BF01536342. [DOI] [PubMed] [Google Scholar]
- 20.Hinnant K., Rotterdam H., Bell E., Tapper M. Cytomegalovirus infection of the alimentary tract: a clinicopathological correlation. Am J Gastroenterol. 1986;81(10):944–950. [PubMed] [Google Scholar]
- 21.Goodgame R. Gastrointestinal cytomegalovirus disease. Ann Intern Med. 1993;119(9):924–935. doi: 10.7326/0003-4819-119-9-199311010-00010. [DOI] [PubMed] [Google Scholar]
- 22.Arnold M., Itzikowitz R., Young B., Machoki S.M., Hsiao N.Y., Pillay K. Surgical manifestations of gastrointestinal cytomegalovirus infection in children: clinical audit and literature review. J Pediatr Surg. 2015;50(11):1874–1879. doi: 10.1016/j.jpedsurg.2015.06.018. [DOI] [PubMed] [Google Scholar]
- 23.Soderberg-Naucler C., Fish K., Nelson J. Reactivation of latent human cytomegalovirus by allogeneic stimulation of blood cells from healthy donors. Cell. 1997;91(1):119–126. doi: 10.1016/s0092-8674(01)80014-3. [DOI] [PubMed] [Google Scholar]
- 24.Rahbar A., Soderberg-Naucler C. Human cytomegalovirus infection of endothelial cells triggers platelet adhesion and aggregation. J Virol. 2005;79(4):2211–2220. doi: 10.1128/JVI.79.4.2211-2220.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Caposio P., Orloff S.L., Streblow D.N. The role of cytomegalovirus in angiogenesis. Virus Res. 2011;157(2):204–211. doi: 10.1016/j.virusres.2010.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Drew W.L. Laboratory diagnosis of cytomegalovirus infection and disease in immunocompromised patients. Curr Opin Infect Dis. 2007;20(4):408–411. doi: 10.1097/QCO.0b013e32821f6010. [DOI] [PubMed] [Google Scholar]
- 27.Bang S., Park Y., Kang B., Park M.C., Hwang M.H., Kim H.K. CMV enteritis causing ileal perforation in underlying lupus enteritis. Clin Rheumatol. 2004;23(1):69–72. doi: 10.1007/s10067-003-0825-z. [DOI] [PubMed] [Google Scholar]