Introduction
Morphea is a fibrosing skin disease with a spectrum of presentations ranging from localized indurated plaques to circumferential involvement of limbs that impairs mobility.1 Eosinophilic fasciitis (EF) is often considered a severe form of morphea.1 Treatment of severe sclerosing diseases, such as generalized deep morphea (GDM) and EF, can be challenging. Methotrexate and prednisone are therapeutic mainstays but are not effective in all cases.1 A recent retrospective study showed that ruxolitinib, a Janus kinase (JAK)1/2 inhibitor, was effective in treating sclerodermatous cutaneous graft-versus-host disease.2 Another series found the benefit of JAK inhibitors in the treatment of hypereosinophilic syndrome (HES) with cutaneous involvement.3 Based on these data and mechanistic studies of sclerosis from animal models, we hypothesized that JAK inhibitors, such as tofacitinib, would be effective for GDM and EF. Here we describe the use of tofacitinib in 2 patients, one with GDM and the other with EF.
Case reports
Case 1
A 53-year-old woman presented with diffusely indurated skin. Her medical history included Guillain-Barre syndrome (treated 5 years prior with intravenous immunoglobulin without residual functional deficit) and stage 1 ductal breast carcinoma (treated 2 years prior with lumpectomy and radiation). One year after radiation to the left breast, skin lesions developed in her breast and then became widespread, leading to restricted range-of-motion (ROM) of several joints (Table I). Her mother had rheumatoid arthritis. Medications included prednisone (40 mg daily), omeprazole, and letrozole. Review of systems was negative and absent for Raynaud syndrome or dysphagia.
Table I.
Active range of motion of wrist and knee joints before and after treatment with tofacitinib as measured by physical therapist
| Joint | Patient 1 (GDM) |
Patient 2 (EF) |
||
|---|---|---|---|---|
| ROM, pretreatment | ROM, after 14 months treatment | ROM, pretreatment | ROM, after 12 months treatment | |
| Right wrist (extension) | 63 | 65 | 15 | 72 |
| Right wrist (flexion) | 39 | 50 | 33 | 68 |
| Left wrist (extension) | 42 | 65 | 34 | 68 |
| Left wrist (flexion) | 25 | 40 | 45 | 70 |
| Right knee (flexion) | 118 | 125 | 87 | 118 |
| Left knee (flexion) | 118 | 135 | 85 | 114 |
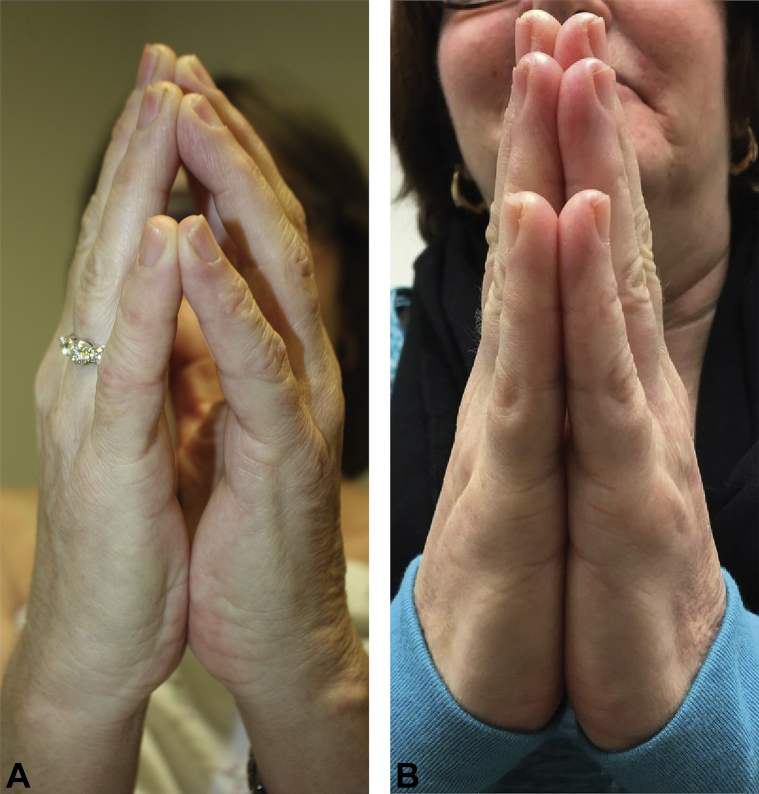
Physical examination revealed marked induration of the previously irradiated breast and generalized induration involving the torso, arms, and legs; prayer sign was positive (Fig 1, A). There were no mat telangiectasias, sclerodactyly, nailfold capillary changes, dyspigmentation, or decreased oral aperture. Biopsy of a truncal plaque showed thickened collagen bundles and a perivascular and interstitial lymphocytic infiltrate. Laboratory evaluation, including antinuclear antibodies and antitopoisomerase I antibodies, was unremarkable.
Fig 1.
A, Before treatment with tofacitinib, patient with GDM was unable to appose her hands (positive prayer sign). B, After 8 months of treatment with tofacitinib, she could fully extend her digits.
Treatment with prednisone, 40 mg daily, for 3 months had not halted disease progression; therefore, tofacitinib, 5 mg twice daily, and extracorporeal photopheresis (ECP) twice weekly were started (and prednisone was tapered to 10 mg daily). Tofacitinib was briefly increased up to 10 mg twice daily before again decreasing to 5 mg twice daily (for insurance reasons). Methotrexate (up to 10 mg weekly) was added 3 months later but discontinued after 5 months because of thrombocytopenia, together with ECP (heparin, which is given with ECP, may cause thrombocytopenia). After more than 1 year of treatment with tofacitinib, 5 mg twice daily, and prednisone, 10 mg daily (including shorter periods of treatment with methotrexate and ECP, as above), her cutaneous induration improved markedly and continues to do so. There has been improvement in ROM of many joints (Table I) and resolution of the prayer sign (Fig 1, B).
Case 2
A 66-year-old woman presented with edema and pain of the lower extremities that spread to her upper extremities and trunk over 6 months. Her medical history included hypertension, hyperlipidemia, gastroesophageal reflux disease, depression, osteoarthritis, and hip replacement. Her medications included alendronate, aspirin, atorvastatin, furosemide, amitriptyline, duloxetine, and omeprazole. Family history was unremarkable. Review of systems was negative and absent for Raynaud syndrome or dysphagia.
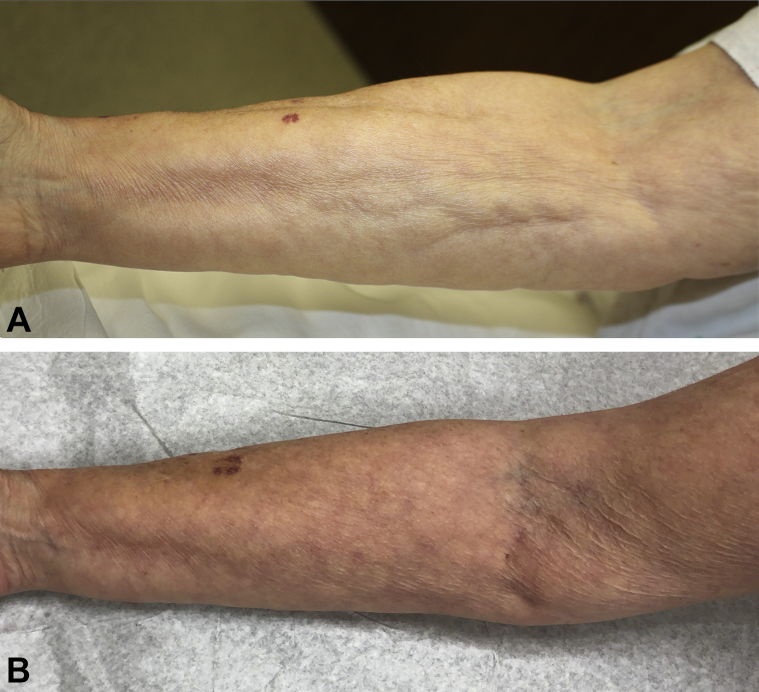
Physical examination revealed diffuse, marked induration and pseudocellulite appearance of the skin involving the torso, arms, and legs (Fig 2, A). There were no mat telangiectasias, sclerodactyly, nailfold capillary changes, dyspigmentation, or decreased oral aperture. There was restricted ROM in multiple joints markedly affecting her mobility and ability to carry out activities of daily living (Table I). Her white blood cell count was 20,500 cells/μL with 47 % eosinophils, and the results of an extensive laboratory evaluation, including serum protein electrophoresis, immunofixation electrophoresis, and flow cytometry, were normal. Biopsy of the right thigh found fascial thickening and dermal fibrosis.
Fig 2.
A, Before treatment with tofacitinib, pseudocellulite appearance and linear depressions (groove sign) of the right arm were prominent in patient with EF. B, After 19 months of treatment with tofacitinib and methotrexate, pseudocellulite appearance and linear depressions are resolved.
Treatment with prednisone, 60 mg daily for 6 months, had been only minimally beneficial; therefore, tofacitinib, 5 mg twice daily, and methotrexate, 10 mg weekly, were started. Prednisone, 15 mg daily, was tapered and discontinued over 3 months. After more than 1 year of treatment with tofacitinib, 5 mg twice daily, and methotrexate, 10 mg weekly, her cutaneous induration improved markedly and continues to do so (Fig 2, B). There has been improvement in both ROM of multiple joints (Table I) and strength.
Discussion
Methotrexate with or without corticosteroids is the mainstay of therapy for GDM,1, 4 and corticosteroids with or without methotrexate are the mainstay of therapy for EF,1 and efficacy of these treatments is variable.5 Targeted therapies, based on pathogenic factors involved in sclerosis, have not been tested in GDM or EF.
Lymphocyte-derived interleukin (IL)-4 and transforming growth factor-β (TGF-β) may be important in morphea pathogenesis by causing overproduction of collagen and other extracellular matrix proteins by fibroblasts.5 JAK inhibition blocks IL-4 signaling, and animal models have suggested that TGF-β–induced fibrosis is JAK2 dependent.6, 7 JAK inhibition has also recently been found to be effective in the treatment of sclerodermatous cutaneous graft-versus-host disease,2 another disorder characterized by severe cutaneous sclerosis. We previously showed that JAK inhibitor monotherapy is effective for HES with skin involvement.3 In lymphocytic-variant HES, pathogenic IL-5 secretion by a clonal lymphocyte population drives eosinophil proliferation and activation.8 Although no T-cell clone is detectable in EF, serum IL-5 levels can be elevated.9 Because IL-5 secretion and activity are blocked by JAK inhibitors,10 we hypothesized that JAK inhibitors would also be effective in EF.
In 2 patients with GDM and EF, tofacitinib with or without low-dose methotrexate both halted disease progression and reversed prior pathology, even when prednisone failed to show response. Treatment with tofacitinib, 5 mg twice daily, led to significant improvement in disease over 8 to 12 months, with efficacy evident as early as 2 months. JAK inhibition, alone or in combination with methotrexate, may be a therapeutic option for severe forms of morphea and other sclerosing skin disorders. A clinical trial of tofacitinib in patients with diffuse cutaneous systemic sclerosis is in progress (NCT03274076).
Footnotes
Authors Kim and Charos contributed equally to this work.
Funding sources: Dr King received funding support from The Ranjini and Ajay Poddar Resource Fund for Dermatologic Diseases Research.
Conflicts of interest: Dr King has served on advisory boards or is a consultant for Aclaris Therapeutics Inc, Celgene, Eli Lilly and Company, Concert Pharmaceuticals Inc, Pfizer Inc, and Roivant Sciences Inc. The rest of the authors have no conflicts to declare.
References
- 1.Mertens J.S., Seyger M.M.B., Thurlings R.M. Morphea and eosinophilic fasciitis: an update. Am J Clin Dermatol. 2017;18:491–512. doi: 10.1007/s40257-017-0269-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hurabielle C., Sicre de Fontbrune F., Moins-Teisserenc H. Efficacy and tolerance of ruxolitinib in refractory sclerodermatous chronic graft-versus-host-disease. Br J Dermatol. 2017 doi: 10.1111/bjd.15593. [DOI] [PubMed] [Google Scholar]
- 3.King B., Lee A.I., Choi J. Treatment of hypereosinophilic syndrome with cutaneous involvement with the JAK inhibitors tofacitinib and ruxolitinib. J Invest Dermatol. 2017;137:951–954. doi: 10.1016/j.jid.2016.10.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Li S.C., Torok K.S., Pope E. Development of consensus treatment plans for juvenile localized scleroderma: a roadmap toward comparative effectiveness studies in juvenile localized scleroderma. Arthritis Care Res (Hoboken) 2012;64:1175–1185. doi: 10.1002/acr.21687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Badea I., Taylor M., Rosenberg A., Foldvari M. Pathogenesis and therapeutic approaches for improved topical treatment in localized scleroderma and systemic sclerosis. Rheumatology (Oxford) 2009;48:213–221. doi: 10.1093/rheumatology/ken405. [DOI] [PubMed] [Google Scholar]
- 6.Dees C., Tomcik M., Palumbo-Zerr K. JAK-2 as a novel mediator of the profibrotic effects of transforming growth factor β in systemic sclerosis. Arthritis Rheum. 2012;64:3006–3015. doi: 10.1002/art.34500. [DOI] [PubMed] [Google Scholar]
- 7.Furumoto Y., Gadina M. The arrival of JAK inhibitors: advancing the treatment of immune and hematologic disorders. BioDrugs. 2013;27:431–438. doi: 10.1007/s40259-013-0040-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Roufosse F., Cogan E., Goldman M. Lymphocytic variant hypereosinophilic syndromes. Immunol Allergy Clin North Am. 2007;27:389–413. doi: 10.1016/j.iac.2007.07.002. [DOI] [PubMed] [Google Scholar]
- 9.Viallard J.F., Taupin J.L., Ranchin V. Analysis of leukemia inhibitory factor, type 1 and type 2 cytokine production in patients with eosinophilic fasciitis. J Rheumatol. 2001;28:75–80. [PubMed] [Google Scholar]
- 10.Parampalli Yajnanarayana S., Stübig T., Cornez I. JAK1/2 inhibition impairs T cell function in vitro and in patients with myeloproliferative neoplasms. Br J Haematol. 2015;169:824–833. doi: 10.1111/bjh.13373. [DOI] [PubMed] [Google Scholar]