Abstract
Background
Patterns of ganglion cell complex (GCC) loss detected by optical coherence tomography provide an objective measure of optic nerve injury. These patterns aid in early diagnosis and localization of chiasmal lesions.
Methods
Twenty-three patients with chiasmal compression seen between 2010 and 2015 were imaged with the Cirrus high-definition optical coherence tomography macular cube 512 × 128, retinal nerve fiber layer (RNFL) scan protocols and automated (30-2 Humphrey) visual fields (VFs). Age-matched controls were included for comparison. Generalized estimating equations were performed comparing RNFL and GCC thicknesses between patients and their controls. Effect size (d) was calculated to assess the magnitude of difference between patients and controls. The average GCC and RNFL thicknesses also were correlated with VF mean deviation (MD). Pre operative average GCC thickness was correlated to post operative VF MD.
Results
Patterns of GCC thinning corresponded to VF defects. The average GCC thickness was 67 ± 9 μm in patients and 86 ± 5 μm in controls (P < 0.001). The effect size was the greatest for GCC thickness (d = 2.72). The mean deviation was better correlated with GCC thickness (r2 =0.25) than RNFL thicknesses (r2 =0.15). Postoperatively, VF MD improved in 7 of 8 patients with persistent nasal GCC thinning. Six patients had no VF defect and showed statistically significant loss of GCC compared with controls (P = 0.001).
Conclusions
Distinct patterns of GCC loss were identified in patients with chiasmal compression. Binasal GCC loss was typical and could be seen with minimal or no detectable VF loss. Thinning of the GCC may be detected before loss of the RNFL in some patients. After decompression, the majority of patients showed improvement in VF despite persistent GCC loss. Patients with less GCC loss before decompression had better postoperative VFs. Therefore, GCC analysis may be an objective method to diagnose and follow patients with chiasmal lesions.
Several methods for predicting improvement in visual function after decompression of the anterior visual pathway have been studied, including duration of symptoms, age, preoperative visual acuity, appearance of optic disc pallor, and photopic negative response (1,2). Detection of optic nerve injury has been facilitated by funduscopic appearance of the retinal nerve fiber layer (RNFL) (3) and computerized enhancement of fundus photographs using digital processing techniques (4). With development of optical coherence tomography (OCT), more objective measurements of optic nerve damage have become available (5,6). Several reports have shown the potential utility of RNFL thickness, macular thickness, and inner retinal thickness measurements in assessing compressive lesions of the anterior visual pathway (7–12). Analysis of the ganglion cell layer using manual segmentation of OCT has been described (13). More recently, high-definition OCT (HD-OCT) algorithms have been developed to enable automated evaluation of the macular ganglion cell complex (GCC) (14).
The goal of our study was to determine if GCC analysis may be a more sensitive method to detect anterior visual pathway compression by comparing average GCC thickness to RNFL thickness and VF loss. In addition, specific patterns of GCC thinning were observed and compared with patterns of VF loss.
METHODS
Subjects and Study Design
A case–control retrospective study was carried out in 23 patients and 23 age-matched healthy controls. Patients eligible for this study had a history of chiasmal compression secondary to brain tumors documented on brain magnetic resonance imaging or computed tomography. Patients were evaluated by the neuro-ophthalmology service at the New England Eye Center, Tufts Medical Center in Boston, Massachusetts, between January 2010 and September 2015. A set of age-matched controls also was established for quantitative analysis. Medical records were reviewed to exclude patients with evidence of significant anterior segment, retinal or other optic nerve diseases. This study was approved by the Tufts Medical Center Institutional Review Board and was conducted in accordance with the ethical standards stated in the 1964 Declaration of Helsinki and was Health Insurance Portability and Accountability Act compliant.
Neuro-Ophthalmological Evaluation
All subjects had a complete neuro-ophthalmological evaluation, including assessment of the best-corrected visual acuity (Snellen optotypes), color discrimination (American Optical Hardy-Rand-Rittler), fundus examination, VFs, and HD-OCT. All patients underwent examination before surgical intervention. Those who required surgical decompression had repeat testing postoperatively. Automated VF testing of the central 30° (HFA750i, SITA-FAST 30-2; Carl Zeiss, Dublin, CA) was performed on all patients and controls, and the mean deviation (MD) was used for statistical analysis.
HD-OCT was performed in a darkened room using Food and Drug Administration-approved equipment (Cirrus 4000/5000; Carl Zeiss). Each subject had both eyes scanned 3 times using 2 standard acquisition protocols: macular cube (512 × 128 line scans) and optic disc cube (200 × 200 line scans) (15). The quality of the obtained images was assessed by evaluation of the signal strength (a value from 0 to 10 in arbitrary units) automatically provided by the system. Only scans with signal strength above 6 units were included in the analysis. For the RNFL analysis, the optic disc cube (200 × 200 line scan) was used. Subsequently, a recognition algorithm detected the inner (vitreoretinal interface) and outer (inner plexiform layer) borders of the RNFL, from a 1.73-mm diameter circle extracted from the optic nerve cube scan and centered on the optic nerve head. The distance between the 2 lines was measured as RNFL thickness at specific locations around the optic nerve (temporal, superior, nasal, inferior) and globally. For GCC analysis, the macular 512 × 128 line scan cube centered on the fovea was used. A different recognition algorithm was applied to detect the outer border of the RNFL and the inner plexiform layer. GCC analysis evaluates the thickness of the ganglion cell plus the inner plexiform layers. GCC thickness was measured at different locations in the macula, around the foveal center (superotemporal, superior, superonasal, inferonasal, inferior, inferotermporal) and globally. Global RNFL and GCC thickness values, and those at each specific location, expressed in micrometers (μm), were used for analysis.
Anterior Visual Pathway Compression Descriptive Analysis
Patients with chiasmal compression were identified with neuroimaging of the brain. In each group, the pattern of VF defects was compared with the pattern of GCC thinning by 6 different sectors. VF defects also were compared with thinning of the RNFL by quadrants. In 8 cases, 1-year postoperative follow-up information was available, and comparisons were made to baseline data.
Statistical Analysis
Patients with evidence of chiasmal compression on neuro-imaging were compared with age-matched controls. Average and individual sector/quadrant GCC and RNFL thickness measurements were compared with controls via generalized estimating equation to account for inter eye dependency. Subgroup analysis also was conducted for patients with bitemporal hemianopia or absence of defects on VF testing. Effect size (Cohen’s d) was calculated to estimate the strength or consistency in the difference between patients and controls (where “d” is defined as the difference in the means between patients and controls, divided by the standard deviation of the data). Average GCC and RNFL thicknesses were compared with MD on VF examinations in all patients. In patients who underwent surgical decompression, average GCC thickness preoperatively was compared with MD on VF examinations postoperatively. Microsoft Excel (version 14.3.9; Microsoft, Redmond, WA), Graph-Pad Prism (version 6.0; GraphPad Software Inc, San Diego, CA), and IBM SPSS Statistics (version 23; IBM, Armonk, NY) were used for statistical analysis.
RESULTS
Subject Characteristics
A total of 23 patients (8 women, 15 men, 45 eyes) were enrolled in the study with a mean age of 52 ± 16 years. Visual acuity ranged from 20/20 to counting fingers. One eye was excluded due to prior damage during surgery to remove a pineal tumor. Imaging confirmed brain tumors with chiasmal compression in all 23 patients. Twenty-three healthy, age-matched control subjects (15 women, 8 men, 45 eyes) with a mean age of 51.9 ± 15.8 years were enrolled for statistical analysis.
Anterior Visual Pathway Compression and Visual Field Descriptive Analysis
Pituitary adenoma was present in 15 patients, tuberculum sella meningiomas in 2, germ cell tumor in 1, astrocytic hamartoma in 2, mucocele in 1, Rathke cyst in 1, and craniopharyngioma in 1. Six patients had bitemporal hemianopia, 2 junctional scotoma, 1 superior homonymous quadrantanopia, 1 altitudinal defect, 3 homonymous hemianopia, 2 nasal defects, 1 unilateral nasal hemianopia, and 1 inferotemporal field loss, and 6 patients did not have field defects.
Optical Coherence Tomography Analysis
Forty-five eyes from 23 patients with chiasmal compressive lesions and 45 eyes from age-matched controls were available for quantitative analysis. Average GCC thickness was 67 ± 9 μm in patients with chiasmal compression and 86 ± 5 μm in controls (P < 0.001). Average GCC thinning and all individual sectors were statistically significant compared with controls (Table 1). In the subset of 6 patients with bitemporal field loss, the average GCC and all sectors besides the inferior temporal sector thickness was significantly thinner than in controls (Table 1). Within this subgroup, the lack of significant inferotemporal thinning (P = 0.195) correlated to the preserved nasal field. GCC thinning remained relatively unchanged in all patients before and after decompression (63.99 μm before resection and 61.51 μm after resection). Of the 6 patients without VF defects, significant GCC loss (P = 0.001) and significant RNFL loss (P = 0.001) were observed. The effect size was stronger in GCC thinning than in RNFL thinning (1.82 and 1.64).
TABLE 1.
GCC loss in patients with compressive lesions of the anterior visual pathway
| Average RNFL | Average GCC | Superior Nasal GCC | Inferior Nasal GCC | Inferior GCC | Interior Temporal GCC | Superior Temporal GCC | Superior GCC | |
|---|---|---|---|---|---|---|---|---|
| All chiasmal compression patients | ||||||||
| Avg | 75 | 67 | 63 | 62 | 67 | 72 | 69 | 66 |
| STDV | 14 | 9 | 11 | 10 | 9 | 11 | 11 | 10 |
| Effect size | −1.452 | −2.718 | −2.150 | −2.247 | −1.631 | −1.044 | −1.422 | −2.034 |
| P value | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 |
| Bitemporal Hemianopia Subgroup | ||||||||
| Avg | 80 | 67 | 59 | 57 | 67 | 77 | 74 | 65 |
| STDV | 16 | 7 | 9 | 9 | 7 | 8 | 7 | 9 |
| Effect size | −0.901 | −2.215 | −2.727 | −2.792 | −1.890 | −0.600 | −1.099 | −2.318 |
| P value | 0.088 | <0.001 | <0.001 | <0.049 | <0.001 | 0.195 | 0.012 | <0.001 |
| No VF defect subgroup | ||||||||
| Avg | 82 | 72 | 72 | 69 | 70 | 75 | 73 | 72 |
| STDV | 11 | 8 | 9 | 9 | 8 | 9 | 8 | 8 |
| Effect size | −1.647 | −1.822 | −1.701 | −1.896 | −1.765 | −1.317 | −1.447 | −2.009 |
| P value | 0.001 | 0.001 | 0.001 | <0.001 | 0.001 | 0.010 | 0.003 | <0.001 |
Bold indicates the greater effect size of the average GCC compared to average RNFL in all patients as well as subgroups.
Avg, average; GCC, ganglion cell complex; GCL, ganglion cell layer; RNFL, retinal nerve fiber layer; STDV, standard deviation; VF, visual field.
Average RNFL thickness was 75 ± 14 μm in patients with chiasmal compression and 91 ± 8 μm in controls (P < 0.001) (Table 1). The average RNFL thickness was not statistically significant between patients and controls in the bitemporal hemianopia subgroup (P = 0.08).
The effect size was calculated to evaluate the strength of the difference in various parameters between patients and controls. The effect size was largest for GCC thickness (d = 2.72) followed by RNFL thickness (d = 1.45). Within the bitemporal VF defect subgroup, the effect size was stronger for nasal GCC thinning (2.81) than for nasal RNFL thinning (−1.05). Within the subgroup without VF defects, the effect size was greatest for average GCC thinning (1.82) followed by RNFL thinning (1.65).
Patterns of Ganglion Cell Complex Loss
Binasal GCC thinning associated with bitemporal hemianopia was seen in 6 patients. In some, there was binasal GCC loss without RNFL thinning on OCT. An additional pattern was diffuse GCC thinning associated with a junctional scotoma. In the 6 cases without VF loss, there was more significant GCC than RNFL thinning. Persistent GCC thinning with improvement in VF was noted in 7 of 8 patients who underwent surgical decompression (after decompression, 93% of eyes had nasal GCC loss postoperatively and 87% experienced VF improvement). One patient showed superior GCC thinning with corresponding inferior altitudinal defects bilaterally on VF examination.
Visual Field Correlation
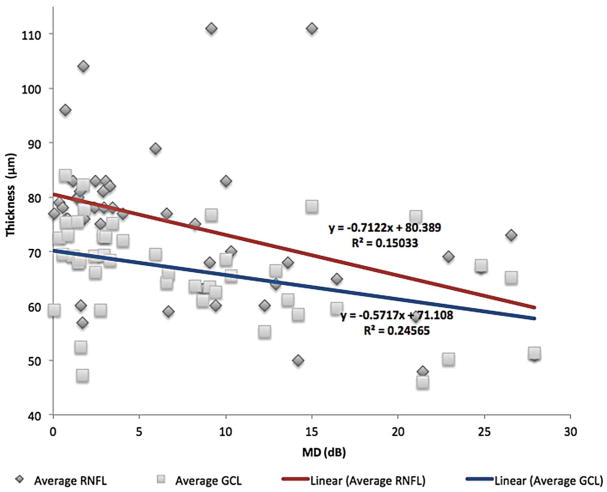
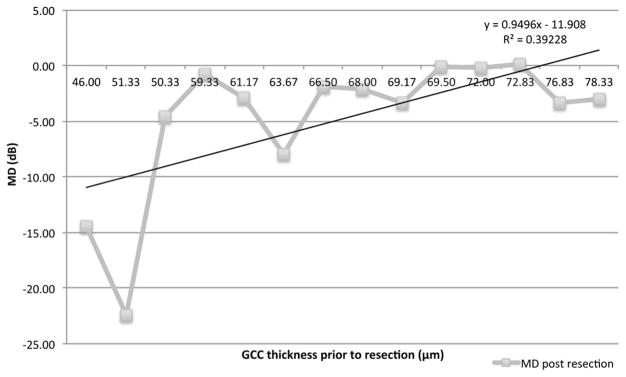
VF MD was correlated to average GCC and RNFL thicknesses in all patients (Fig. 1). The GCC thickness was most strongly correlated with MD (r2 = 0.25) followed by RNFL thicknesses (r2 = 0.15). In patients who underwent decompression, postoperative VF MD was correlated with preoperative GCC thickness (r2 = 0.39), showing those with greater preoperative GCC thickness had better postoperative visual fields (VFs) (Fig. 2).
FIG. 1.
The VF mean deviation was more strongly correlated to GCC thickness than to RNFL thickness (0.25 compared with 0.15). GCL, ganglion cell layer; RNFL, retinal nerve fiber layer; VF, visual field.
FIG. 2.
In patients who underwent decompression, postoperative visual field mean deviation (MD) was correlated with preoperative ganglion cell complex (GCC) thickness (r2 = 0.39), showing that those with greater preoperative GCC thickness had better postoperative visual fields.
DISCUSSION
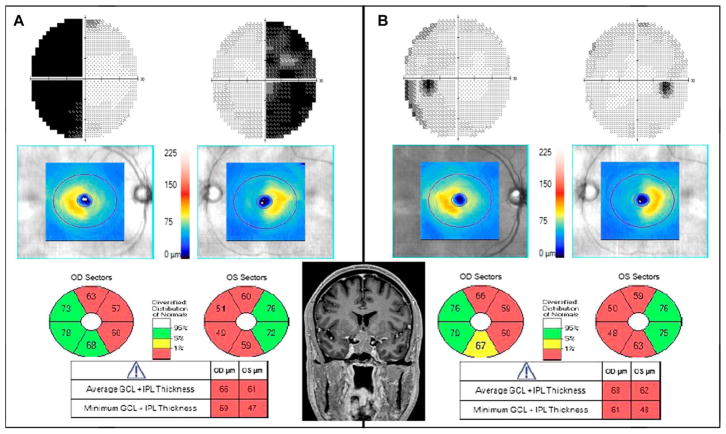
We observed various patterns of GCC loss, which corresponded to VF changes, including junctional, binasal (Fig. 3), and homonymous GCC loss. We also saw distinctive binasal loss of GCC in patients with mild or no VF defects. This indicates that GCC analysis has the ability to detect early or mild chiasmal compression and aid in localization.
FIG. 3.
A 61 year-old man with visual acuity of 20/20 in the right eye, 20/25 in the left eye, normal color vision, and pupillary reactions had progressive bitemporal VF loss over 8 months. MRI revealed a pituitary adenoma. A. Initial examination findings show GCC thinning binasally on OCT in addition to a bitemporal hemianopia. B. One year after surgery, VFs have markedly improved, yet there is persistent binasal GCC thinning. GCC is GCL + IPL. GCC, ganglion cell complex; GCL, ganglion cell layer; IPL, inner plexiform layer; MRI, magnetic resonance imaging; OCT, optical coherence tomography; OD, right eye; OS, left eye; VF, visual field.
Furthermore, we found GCC thinning in patients with normal RNFL thickness. The relative insensitivity of RNFL measurement also was suggested in a previous report of normal RNFL in eyes with VF defects from chiasmal compression (7). Overall, when comparing RNFL to GCC in patients with chiasmal compression, the average nasal RNFL and GCC thinning were statistically significant, yet the effect size was much greater for GCC thinning (Table 1). Analysis of those with no VF defects showed statistically significant RNFL and GCC thinning with greater effect size of GCC thinning.
These results add another dimension and further refine previous studies of OCT in patients with chiasmal compression. Monteiro et al (9) described hemianopic full-thickness macular thinning in patients with chiasmal compression. Others reported inner retinal thickness (combined RNFL, ganglion cell layer, and inner plexiform layer) thinning with similar results (7,8), and a more recent study showed preferential ganglion cell loss in the nasal hemirentina in patients with pituitary tumors (16).
VF changes were shown to precede ganglion cell loss in another series of cases with chiasmal compression (11). However, our results indicate that GCC thinning can occur before VF loss, and improvement in VF occurs in some cases to normal despite persistent GCC thinning. This may be the result of a lack of sensitivity in VF testing and the ability of the remaining axons and ganglion cells to compensate for the loss until a critical threshold is reached, at which point VF defects become detectable on examination.
OCT analysis may be useful for the prognosis of VF recovery from surgical or medical decompression of the chiasm. Some reports indicate that postoperative improvement in VF occurs with preserved RNFL or preserved inner retinal thickness (RNFL, ganglion cell layer, and inner plexiform layer) (7–9,17) An additional report described nasal thinning of the inner retinal layers postoperatively (8). In our study, 93% of eyes had nasal GCC loss postoperatively and 87% experienced VF improvement postoperatively. Our results suggest that patients with VF defects and relatively preserved GCC thickness will have better postoperative VF recovery.
Patterns of GCC loss seen on OCT can be useful for localizing compressive lesions of the anterior visual pathway, specifically in cases of chiasmal compression. Observation of GCC thinning on OCT may be a useful method for early detection of visual pathway injury before thinning of the RNFL and, in some cases, before VF loss. In particular, OCT is objective, whereas VF testing is subjective and may not reflect early or mild axonal damage. Early surgical decompression before significant RNFL and GCC thinning may lead to better postoperative visual function. Recovery of VF, despite persistent GCC thinning after tumor resection, suggests that damage to the anterior visual pathway from brain tumors may be more significant than was previously thought.
Acknowledgments
C. E. Mendoza-Santiesteban receives research support from The Dysautonomia Foundation, Inc. and The Massachusetts Lions Clubs/Research to Prevent Blindness Challenge Grant. He has served as a consultant/advisory board member for EISAI/H3, Roland Consult, and Carl Zeiss Meditec. T. R. Hedges receives research support from The Massachusetts Lions Clubs/Research to Prevent Blindness Challenge Grant. This work was supported in part by a Research to Prevent Blindness Challenge Grant to the New England Eye Center/Department of Ophthalmology, Tufts University School of Medicine, National Institutes of Health (NIH) contracts RO1-EY11289-24, R01-EY13178-10, R01-EY013516-07, Air Force Office of Scientific Research FA9550-07-1-0101 and FA9550-07-1-0014. The sponsors had no role in the design or conduct of this research. The project described was supported by the National Center for Advancing Translational Sciences, National Institutes of Health, award number UL1TR001064. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Footnotes
The other authors report no conflict of interest.
STATEMENT OF AUTHORSHIP
Category 1: a. Conception and design: Marisa Gobuty, Thomas R. Hedges III, Joseph Ho, Natalie Erlich-Malona, Laurel N. Vuong, Geetha K. Athappilly, Carlos E. Mendoza-Santiesteban; b. Acquisition of data: Marisa Gobuty, Thomas R. Hedges III, Joseph Ho, Natalie Erlich-Malona, Laurel N. Vuong, Geetha K. Athappilly, Carlos E. Mendoza-Santiesteban; c. Analysis and interpretation of data: Marisa Gobuty, Thomas R. Hedges III, Joseph Ho, Laurel N. Vuong, Carlos E. Mendoza-Santiesteban. Category 2: a. Drafting the manuscript: Marisa Gobuty, Thomas R. Hedges III, Joseph Ho, Laurel N. Vuong, Carlos E. Mendoza-Santiesteban; b. Revising it for intellectual content: Marisa Gobuty, Thomas R. Hedges III, Joseph Ho, Natalie Erlich-Malona, Laurel N. Vuong, Geetha K. Athappilly, Carlos E. Mendoza-Santiesteban. Category 3: a. Final approval of the completed manuscript: Marisa Gobuty, Thomas R. Hedges III, Joseph Ho, Natalie Erlich-Malona, Laurel N. Vuong, Geetha K. Athappilly, Carlos E. Mendoza-Santiesteban.
References
- 1.Cohen A, Cooper P, Kupersmith M, Kupersmith M, Flamm E, Ransohoff J. Visual recovery after transsphenoidal removal of pituitary adenomas. Neurosurgery. 1985;17:446–452. doi: 10.1227/00006123-198509000-00008. [DOI] [PubMed] [Google Scholar]
- 2.Moon C, Hwang S, Kim B, Ohn Y, Park T. Visual prognostic value of optical coherence tomography and photopic negative response in chiasmal compression. Invest Ophthalmol Vis Sci. 2011;52:8527–8533. doi: 10.1167/iovs.11-8034. [DOI] [PubMed] [Google Scholar]
- 3.Hoyt W. Fundoscopic changes in the retinal nervefibre layer in chronic and acute optic neuropathies. Trans Ophthal Soc U K. 1976;96:368–371. [PubMed] [Google Scholar]
- 4.Peli E, Hedges T, Schwartz B. Computerized enhancement of retinal nerve fiber layer. Acta Ophthalmol (Copenh) 1986;64:113–122. doi: 10.1111/j.1755-3768.1986.tb06885.x. [DOI] [PubMed] [Google Scholar]
- 5.Frohman E, Fujimoto J, Frohman T, Calabresi P, Cutter G, Balcer L. Optical coherence tomography: a window into the mechanisms of multiple sclerosis. Nat Clin Pract Neurol. 2008;4:664–675. doi: 10.1038/ncpneuro0950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Schuman J, Hee M, Arya A, Pedut-Kloizman T, Puliafito CA, Fujimoto JF, Swanson EA. Optical coherence tomography: a new tool for glaucoma diagnosis. Curr Opin Ophthalmol. 1995;6:89–95. doi: 10.1097/00055735-199504000-00014. [DOI] [PubMed] [Google Scholar]
- 7.Danesh-Meyer H, Papchenko T, Savino P, Law A, Evans J, Gamble G. In vivo retinal nerve fiber layer thickness measured by optical coherence tomography predicts visual recovery after surgery for parachiasmal tumors. Invest Ophthalmol Vis Sci. 2008;49:1879–1885. doi: 10.1167/iovs.07-1127. [DOI] [PubMed] [Google Scholar]
- 8.Ohkubo S, Higashide T, Takeda H, Murotani E, Hayashi Y, Sugiyama K. Relationship between macular ganglion cell complex parameters and visual field parameters after tumor resection in chiasmal compressio. Jpn J Ophthalmol. 2012;56:68–75. doi: 10.1007/s10384-011-0093-4. [DOI] [PubMed] [Google Scholar]
- 9.Monteiro M, Hokazono K, Fernandes DB, Costa-Cunha LV, Sousa RM, Raza AS, Wang DL, Hood DC. Evaluation of inner retinal layers in eyes with temporal hemianopic visual loss from chiasmal compression using optical coherence tomography. Invest Ophthalmol Vis Sci. 2014;55:3328–3336. doi: 10.1167/iovs.14-14118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Monteiro M, Costa-Cunha L, Cunha LV, Cunha LP, Malta R. Correlation between macular and retinal nerve fibre layer Fourier-domain OCT measurements and visual field loss in chiasmal compression. Eye (Lond) 2010;24:1382–1390. doi: 10.1038/eye.2010.48. [DOI] [PubMed] [Google Scholar]
- 11.Moon CH, Hwang SC, Ohn Y, Park T. The time course of visual field recovery and changes of retinal ganglion cells after optic chiasmal decompression. Invest Ophthalmol Vis Sci. 2011;52:7966–7973. doi: 10.1167/iovs.11-7450. [DOI] [PubMed] [Google Scholar]
- 12.Monteiro M, Cunha L, Costa-Cunha L, Maia O, Oyamada M. Relationship between optical coherence tomography, pattern electroretinogram and automated perimetry in eyes with temporal hemianopia from chiasmal compression. Invest Ophthalmol Vis Sci. 2009;50:3535–3541. doi: 10.1167/iovs.08-3093. [DOI] [PubMed] [Google Scholar]
- 13.Kardon R. Role of the macular optical coherence tomography scan in neuro-ophthalmology. J Neuroophthalmol. 2011;31:353–361. doi: 10.1097/WNO.0b013e318238b9cb. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wang M, Hood D, Cho J, Ghadiali Q, De Moraes CG, Zhang X, Ritch R, Liebmann JM. Measurement of local retinal ganglion cell layer thickness in patients with glaucoma using frequency-domain optical coherence tomography. Arch Ophthalmol. 2009;127:875–881. doi: 10.1001/archophthalmol.2009.145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ho J, Sull AC, Vuong LN, Chen Y, Liu J, Fujimoto JG, Schuman JS, Duker JS. Assessment of artifacts and reproducibility across spectral- and time-domain optical coherence tomography devices. Ophthalmology. 2009;116:1960–1970. doi: 10.1016/j.ophtha.2009.03.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jeong AR, Kim E, Kim NR. Preferential ganglion cell loss in the nasal hemiretina in patients with pituitary tumor. J Neuroophthalmol. 2016;36:152–155. doi: 10.1097/WNO.0000000000000331. [DOI] [PubMed] [Google Scholar]
- 17.Jacob M, Raverot G, Jouanneau E, Borson-Chazot F, Perrin G, Rabillous M, Tilikete C, Bernard M, Vighetto A. Predicting visual outcome after treatment of pituitary adenomas with optical coherence tomography. Am J Ophthalmol. 2009;147:64–70. doi: 10.1016/j.ajo.2008.07.016. [DOI] [PubMed] [Google Scholar]