Abstract
Sudan dyes are phenyl-azoic derivatives widely used in industry. Classified as carcinogenic and are strictly forbidden in foodstuffs; however, some unscrupulous businessmen adopted it for coloring foodstuffs. Here, a simple and effective fluorescence (FL) assay platform has been developed for the detection of Sudan I–IV based on CsPbBr3 perovskite quantum dots (QDs). It was found that the fluorescent emission of CsPbBr3 QDs can be effectively quenched by Sudan I–IV. Under the optimized conditions, the FL quenching efficiency of CsPbBr3 QDs was quantitatively correlated to the logarithmic concentrations of Sudan I–IV over the ranges of 100–10,000, 0.1–1000, 0.1–2000 and 0.4–1000 ng mL−1 for Sudan I–IV, and the corresponding limits of detection were 3.33, 0.03, 0.03 and 0.04 ng mL−1 (at 3σ/slope), respectively. CsPbX3 QDs (X = Cl, Br, and I or mixed halide systems Cl/Br and Br/I) was utilized as sensor in FL assay, which have unique optical properties of high FL quantum yields (up to 90%), narrow half peak width (26 nm) and tunable FL emissions spectra (410–700 nm). Meanwhile, the practical use of this assay platform for Sudan I–IV detection in chilli powder samples was also demonstrated, which indicated the potential in practical applications.
Electronic supplementary material
The online version of this article (10.1007/s13197-018-3167-1) contains supplementary material, which is available to authorized users.
Keywords: Chili powder, Sudan I–IV, CsPbBr3 QDs, FL assay platform, detection
Introduction
Sudan dyes, including Sudan I, II, III and IV, are a class of lipophilic azo dyes with an orange-red appearance, which are commonly used as colorants in plastics, textiles, waxes, printing inks, cosmetics and so on. But using Sudan dyes as food additive is strictly forbidden, as they may induce some forms of cancer and have been classed as category 3 carcinogens by International Agency for Research on Cancer (IARC) (Stiborová et al. 2002). However, because of their low cost and bright color, they are still utilized illegally in food products (Shan et al. 2012). Therefore, it is crucial to develop a rapid method to detect Sudan dyes in foodstuffs for guaranteeing the safety of consumers. To date, a variety of analytical methods for Sudan dyes in foodstuffs have been proposed, mainly including high performance liquid chromatography (HPLC) (Sricharoen et al. 2017), mass spectrometry (Mazzotti et al. 2008; Sciuto et al. 2017), raman spectroscopy (Cheung et al. 2010; Anibal et al. 2012; Ou et al. 2017), chemiluminescence flow injection analysis (Liu et al. 2007), molecular imprinting method (Xu et al. 2014), electroanalytical technique (Ensafi et al. 2012), enzyme-linked immune method (Haustein et al. 2012; Wang et al. 2012) and fluorescence method. Among them, fluorescence method has attracted great interests due to the simple instruments and easy operations.
In fluorescent assay, the fluorophore, as an indispensable part of the fluorescent sensor, determines the performance and application. Commonly, organic fluorophores and quantum dots (QDs) are used as fluorophores. However, most organic fluorophores excited at short wavelengths have photobleaching and limited photostability (Kim et al. 2011; Toutchkine et al. 2003), which restricts their use in some fields. Recently, QDs have also been widely studied and applied in many fields as an potentially superior alternative to organic fluorophores (Louie 2010; Yao et al. 2014; Zhou et al. 2015), due to their unique merits of being easy to synthesize, high resistance to photobleaching (Alivisatos 1996) and good photostability (Dubertret et al. 2002; Wu et al. 2003; Medintz et al. 2005). Unfortunately, the FL quantum yields of most traditional QDs are not high (Singh et al. 2010; Zeng et al. 2012). Furthermore, FL emission spectra of most traditional QDs are not tunable or only be tuned via size-tuning. While the size-tuning route is limited in many fields, it is hard to achieve a batch-to-batch consistent fluorescence efficiency or an exact size (Look and Manthuruthil 1976; Murray et al. 2002; Ritter and Lyding 2009). Therefore, it is meaningful to find stable QDs with high FL quantum yields and tunable emissions.
It was reported that CsPbX3 QDs were novel optoelectronic materials with unique optical properties (Akkerman et al. 2015; Protesescu et al. 2015; Wei et al. 2016). Firstly, the quantum yields of CsPbX3 QDs were high up to 90%, which were critical for high sensitivity. Secondly, the narrow line-widths of the FL emission spectra were just 12–42 nm, which were beneficial to selectivity detection. Of particular importance, the FL emission spectra were readily tunable over the entire visible region of 410–700 nm via quantum size-effects and compositional modulations. With high quantum yields, narrow line-widths and tunable FL emission feature, CsPbX3 QDs are expected to be capable of functioning as a powerful fluorophore to build inner filter effect (IFE) based fluorescent assay platform. It might provide sensitive and selective assay platform for many analytes, whose UV–Vis absorption spectra located in the region of 410–700 nm, based on IFE. However, to the best of our knowledge, its application in fluorescence detection has not been reported to date.
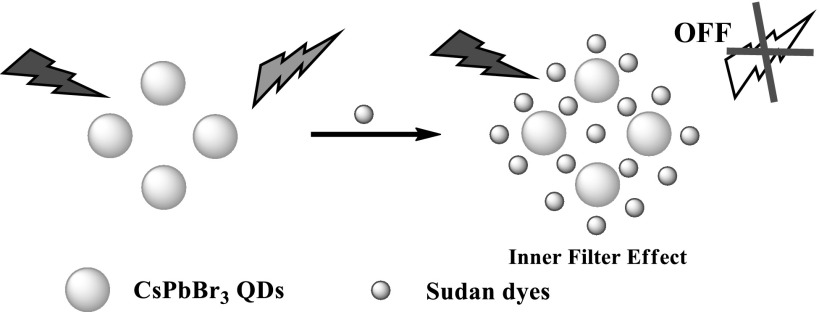
Herein, CsPbBr3 QDs have been synthesized and firstly utilized for the detection of Sudan I–IV (Chemical structures are shown in Fig. S1) based on IFE. As illustrated in Scheme 1, CsPbBr3 QDs exhibited bright green fluorescence under the UV excitation, and upon addition of Sudan I–IV, the fluorescence could be efficiently quenched by Sudan I–IV as the absorption band of Sudan I–IV largely overlapped with the fluorescence spectrum of CsPbBr3 QDs. Based on this mechanism, a simple and rapid, highly selective and sensitive sensing method has been developed for the detection of Sudan I–IV. Furthermore, the assay platform was successfully employed in the detection of Sudan I–IV in chilli powder samples, which demonstrated its potential for practical applications.
Scheme 1.
Schematic illustration of the Sudan dyes detection mechanism using the CsPbBr3 QDs
Materials and methods
Materials
Cesium carbonate (Cs2CO3) (Sigma-Aldrich, 99.9%), octadecene (ODE) (Sigma-Aldrich, 90%), oleic acid (OA) (Sigma-Aldrich, 90%), oleylamine (OLA) (Sigma-Aldrich, 80–90%), Lead (II) bromide (PbBr2) (Sigma-Aldrich, 98%), 1-phenylazo-2-naphthalenol (Sudan I), 1-[(2,4-dimethylphenyl)azo]-2-naphthalenol (Sudan II), 1-[4-(-(phenylazo)phenyl]azo]-2-naphthalenol (Sudan III) and o-tolylazo-otolylazobetanaphthalenol (Sudan IV) were all obtained from Sigma (Shanghai, China, > 93.0%). Other reagents were purchased from Sinopharm Chemical Reagent Co., Ltd. (China), and used directly without further purification. The chili powder was purchased from local supermarket (Changsha, Hunan, China), colour: red, size: 30–50 mesh, moisture: < 10%.
Apparatus
A Hitachi F20 transmission electron microscope (Tokyo, Japan) was used to characterize the morphology and size distribution of CsPbX3 QDs. Samples were prepared by dropping washed and diluted (in n-hexane) QDs solutions onto carbon-coated 200 mesh copper grids with subsequent solvent evaporation. And a FT-IR spectrophotometer (Nicolet Instrument Co., USA) was used to obtain fourier transform infrared (FT-IR) spectra between 4000 and 700 cm−1. The X-ray diffraction (XRD) patterns of CsPbX3 QDs were obtained using a D8 Advance (Bruker) X-ray diffractometer with Cu Kα radiation. Chromatographic analysis was performed using a Shimadzu liquid chromatograph LC-20A (Kyoto, Japan), equipped with binary solvent delivery units (LC-20AD), UV–Vis detector (SPD-20A). Separations were conducted with a Zorbax SB-C18 column (4.6 mm × 150 mm, 3.5 μm particle size, Kyoto, Japan), the column temperature was thermostated at 30 °C and the sample injection volume was 10 μL. An UV-2450 spectrophotometer (Shimadzu Co., Japan) was employed to record the UV–visible absorption spectra, the QDs solutions were prepared by diluting the crude QDs solutions in n-hexane (50 μL in 1 mL), in 1 cm path length quartz cuvettes with airtight screw caps. An F-7000 fluorescence spectrophotometer (Hitachi Co., Japan) was used to record all the fluorescence spectra and measure the fluorescence intensity under the excitation of 400 nm continuous-wave, the QDs solutions were prepared by diluting the crude QDs solutions in n-hexane (50 μL in 1 mL), in 4 mL covered quartz cuvette. Fluorescence test conditions: the excitation wavelength was 400 nm, temperature was 25 °C, incubation time was 4 min, the excitation slit width was 5 nm and the emission slit width was 5 nm.
Synthesis of CsPbBr3 QDs
The preparation of CsPbBr3 QDs was carried out as follows (Protesescu et al. 2015): typically, PbBr2 (0.376 mmol), ODE (10 mL), OA (1.0 mL) and OLAM (1.0 mL) were loaded in a 25 mL 3-neck flask and dried under N2 for 1 h at 120 °C. Then, the temperature was raised to 155 °C and 0.8 mL of Cs-oleate (previously synthesized: Cs2CO3 (0.4 g) in 15 mL of ODE and 1.75 mL of OA at 150 °C under N2) was quickly injected. 3–5 s later, the reaction mixture was quickly cooled down in an ice bath. Then, the QDs were purified by high speed centrifugation (at 12,000 rpm for 20 min). Finally, CsPbBr3 QDs were re-dispersed in 10 mL n-hexane forming long-term stable solutions.
Fluorescence quenching experiments with analytes
Fluorescence titrimetric method was used in this work. Firstly, different concentrations of Sudan I–IV were prepared, and the concentration of Sudan I–IV was 0–0.6 mg mL−1. Then 20 μL Sudan I–IV was added into 4 mL covered quartz cuvette containing 2 mL CsPbBr3 QDs solutions from low concentration to high concentration successively, the QDs solutions were prepared by diluting the crude QDs solutions in n-hexane (50 μL in 1 mL). After mixed evenly, the fluorescence spectra were obtained on an F-7000 fluorescence spectrophotometer. Test conditions: the excitation wavelength was 400 nm, temperature was 25 °C, incubation time was 4 min, the excitation slit width was 5 nm and the emission slit width was 5 nm.
Real sample assays
Chilli powder samples were used to demonstrate the feasibility of CsPbBr3 QDs for the detection of Sudan I–IV from complicated matrices. The samples were prepared with a standard addition method: appropriate amounts of Sudan I–IV standard solution were added into the chili powder commercially purchased from a local supermarket (Changsha, China). Then 1.0 g prepared sample was extracted ultrasonically by 30 mL n-hexane for 30 min and centrifugal sedimentation was carried out for 10 min at 10,000 rpm. At last, the extraction liquid was collected in a sealed flask for detection. The concentrations of Sudan I–IV in each samples were determined using the proposed method and were also analyzed using the National Standard of the People’s Republic of China (GB/T 19681-2005) to test the accuracy of the proposed method.
Results and discussion
Characterization of CsPbBr3 QDs
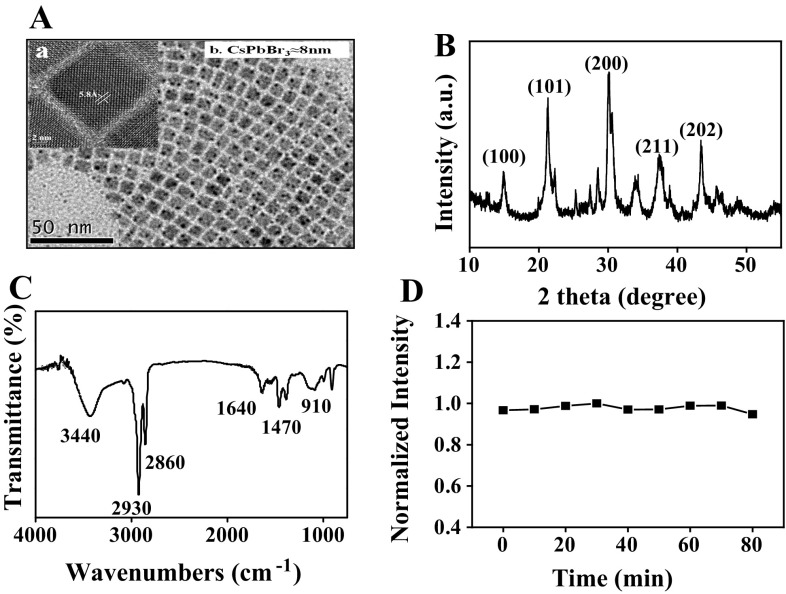
The morphology of CsPbBr3 QDs was characterized by TEM. As shown in Fig. 1a, the TEM image reveals that the CsPbBr3 QDs have the average diameters of about 8 nm and well dispersed in n-hexane solution. The HRTEM of CsPbBr3 QDs shows the lattice spacing of 0.58 nm, which agrees well with the (100) facet of CsPbBr3 QDs. The structure of CsPbBr3 QDs was characterized by XRD and FT-IR. The XRD pattern (Fig. 1b) and the HRTEM of CsPbBr3 QDs showed that CsPbBr3 QDs crystallized in the cubic phase. As shown in Fig. 1c, two peaks at 3440 and 1640 cm−1 are observed, which could be attributed to the O–H(–COOH) stretching and C=C bending vibrations, respectively. It implied that the oleic acid was successfully capped on the CsPbBr3 QDs surface. The absorption spectrum of CsPbBr3 QDs is shown in Fig. S2A. The fluorescence spectra of CsPbBr3 QDs under different excitation wavelength are shown in Fig. S2B, it can be seen that the CsPbBr3 QDs show excitation-independent fluorescence behavior. Consider the fluorescence intensity and the difficult to eliminate the background interference in complex samples due to the shorter wavelength excitation, we chosen 400 nm as the excitation light for further application. The optical stability of CsPbBr3 QDs was characterized by fluorescence spectra. As shown in Fig. 1d, the FL intensity shows negligible change (< 5%) even after continuous excitation with a 150W Xe lamp (400 nm) for 80 min, indicating that the CsPbBr3 QDs show excellent photostability.
Fig. 1.
a Typical TEM image, b XRD pattern and c FT-IR spectra of CsPbBr3 QDs; inset: the HRTEM image of single CsPbBr3 QDs. d Normalized FL intensity of the CsPbBr3 QDs under continuous excitation with a 150 W Xe lamp for 80 min. Test conditions: the excitation wavelength was 400 nm, temperature was 25 °C, incubation time was 4 min, the excitation slit width was 5 nm and the emission slit width was 5 nm
Detection mechanism
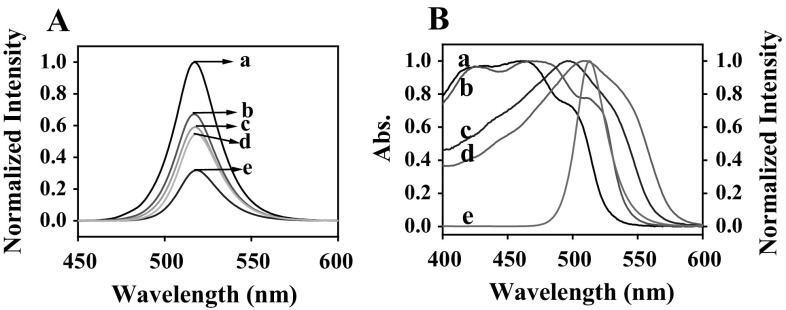
The FL response of CsPbBr3 QDs to the Sudan I–IV and the quenching mechanism were discussed. As shown in Fig. 2a, upon addition of Sudan I–IV, the emission of CsPbBr3 QDs is efficiently quenched and no shift of the wavelength is observed. Under the excitation of 400 nm, the CsPbBr3 QDs exhibited strong fluorescence emission around 515 nm, with a narrow half peak width of just 26 nm, which was greatly overlapped with the absorption spectra of Sudan I–IV (As illustrated in Fig. 2b). It has been reported that an efficient spectral overlap between the absorption band of the absorber and the emission band of the fluorophore would result FRET (Chen et al. 2014; Medintz et al. 2003; Sapsford et al. 2006) or IFE (Dong et al. 2012; Shao et al. 2005; Xiao et al. 2004). Compared with FRET, IFE did not require any covalent linking (or interaction) between the receptor and the fluorophore. As shown in Fig. 2a, the emission intensity of CsPbBr3 QDs remarkably decreased by adding increasing concentrations of Sudan I–IV without changes of the spectral feature (emission position and full width half maximum). It indicated that the observed decrease in fluorescence intensity of the CsPbBr3 QDs was attributed to the Sudan I–IV directly quenching the CsPbBr3 QDs fluorescence, i.e. fluorescence may be quenched by the so-called inner filter effect (Zhou et al. 2013). To further confirm the mechanism, UV–Vis absorption spectra of Sudan I–IV solution in the absence and presence CsPbBr3 QDs were recorded and shown in Fig. S3. No changes in the absorption bands position of Sudan I–IV are observed after adding CsPbBr3 QDs, which indicates that no complexes were formed. Therefore, no interaction between Sudan I–IV and CsPbBr3 QDs occurred. Therefore, we inferred the fluorescence quenching mechanism was IFE.
Fig. 2.
a Normalized FL intensity of the CsPbBr3 QDs (a) in the absence and presence of 1 µg mL−1 Sudan I (b), Sudan II (c), Sudan III (e), Sudan IV (d), respectively. b Emission spectrum of CsPbBr3 QDs (e) and absorption spectra of Sudan I (a), Sudan II (b), Sudan III (c), Sudan IV (d), respectively. Test conditions: the excitation wavelength was 400 nm, temperature was 25 °C, incubation time was 4 min, the excitation slit width was 5 nm and the emission slit width was 5 nm
Optimization of the fluorescent assay platform for Sudan I–IV
In order to optimize the detection conditions for Sudan I–IV by using CsPbBr3 QDs as the fluorescence probe, several factors including solvent, temperature, and reaction time were investigated. Firstly, we tested the fluorescence intensities of CsPbBr3 QDs in different solvents, such as n-hexane, methylbenzene acetone, petroleum ether and dimethyl sulfoxide (DMSO) and the results were shown in Fig. S4A. The results suggested that the fluorescence intensities of CsPbBr3 QDs in n-hexane were higher than in other solvents, which is probably due to better dispersion of oleic-capped CsPbBr3 QDs in n-hexane than that in other organic solvent. So we choose n-hexane as detection solvent. Next, we optimized the detection temperature and choose Sudan I as example. As shown in Fig. S4B, it obviously conveyed that the most suitable temperature for this sensing system is 25 °C. Finally, the effect of different incubation times ranging from 0 to 10 min on quenching efficiency ((F0 − F)/F0) was investigated. As shown in Fig. S4C, the ((F0 − F)/F0 achieved a stable value at 3, 3, 4, 3 min for Sudan I–IV, respectively. So the incubation time is selected as 4 min.
Detection of Sudan dyes under the optimized condition
Under the optimized conditions, the fluorescence spectra of CsPbBr3 QDs in the presence of different concentrations of Sudan I–IV were recorded. As shown in Fig. 3, the fluorescence intensity of the CsPbBr3 QDs gradually decreased with the concentration of Sudan I–IV. There were good correlations between the quenching efficiency ((F0 − F)/F0) and the logarithmic concentration of Sudan I–IV (insets of Fig. 3) in the range of 100–10,000, 0.1–1000, 0.1–2000 and 0.4–1000 ng mL−1 with the correlation coefficients (R2) of 0.9936, 0.9886, 0.9922 and 0.9876, and the regression equations were (F0 − F)/F0= − 0.51446 + 0.28859logCSudan I, (F0 − F)/F0= 0.12856 + 0.08735logCSudan II, (F0 − F)/F0= 0.23584 + 0.14911logCSudan III, and (F0 − F)/F0 = 0.05772 + 0.12813logCSudan IV, the limit of detection (LOD) was 3.33, 0.03, 0.03 and 0.04 ng mL−1 (3σ/slope), respectively. It is obvious that the fluorescence quenching efficiency of Sudan II–IV is higher than that of Sudan I, which is probably due to the bigger overlap extent between fluorescence spectrum of CsPbBr3 QDs and absorption spectra of Sudan II–IV than that of Sudan I (Fig. 2b). The good linear relationships and the low LODs implied that the CsPbBr3 QDs have promising application in the detection of Sudan dyes. For comparison, an overview of recently reported analytical methods for Sudan dyes detection was shown in Table S1. It is obvious that the proposed fluorescence assay platform exhibits superior sensitivity, rapid response and a wider linear response range for Sudan dyes detection.
Fig. 3.
The normalized fluorescence emission intensity of the CsPbBr3 QDs in the presence of different concentrations of a Sudan I (from 1 to 15: 0, 100, 300, 500, 700, 1000, 2000, 3000, 4000, 5000, 6000, 7000, 8000, 9000 and 10,000 ng mL−1), b Sudan II (from 1 to 15: 0, 0.1, 0.5,1, 5, 10, 50, 100, 200, 300, 400, 500, 600, 800 and 1000 ng mL−1), c Sudan III (from 1 to 18: 0, 0.1, 0.5, 1, 5, 10, 50, 100, 200, 300, 400, 500, 600, 700, 800, 900, 1000 and 2000 ng mL−1), d Sudan IV (from 1 to 16: 0, 0.4, 0.9, 5, 10, 50, 100, 200, 300, 400, 500, 600, 700, 800, 900 and 1000 ng mL−1). F0 and F are the fluorescence intensity of the CsPbBr3 QDs in the absence and presence of Sudan I–IV, respectively. The inset shows the linear relationship between the quenching efficiency ((F0 − F)/F0) and the logarithmic concentration of Sudan I–IV. Test conditions: the excitation wavelength was 400 nm, temperature was 25 °C, incubation time was 4 min, the excitation slit width was 5 nm and the emission slit width was 5 nm
Selectivity for detection of Sudan I–IV
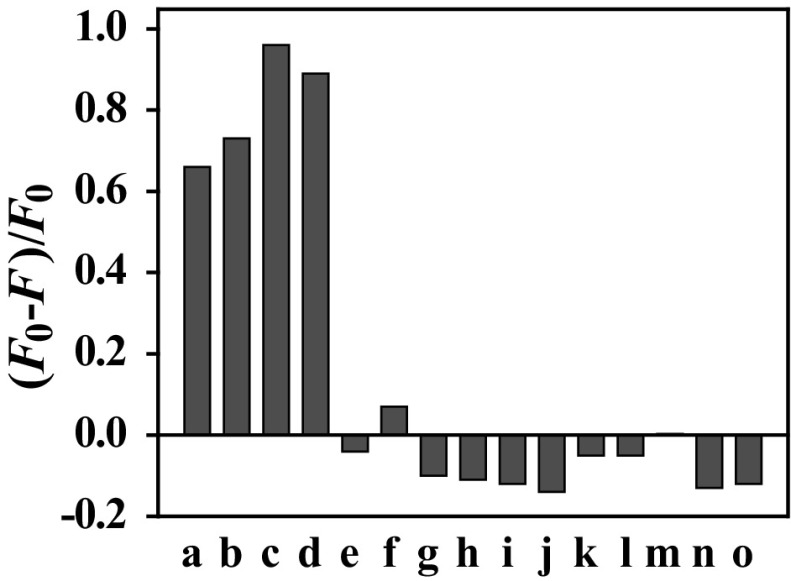
To interstate the selectivity, the interferences of l-Cys, l-Lys, l-Glu, ascorbic acid, fructose, glucose, sucrose, urea, β-cyclodextrin, sodium citrate and sodium benzoate were carried out under optimum conditions. As shown in Fig. 4, there are little changes of fluorescence intensity of CsPbBr3 QDs even the interfering substance are saturated in this detection system, which could be explained by the two reasons: first, the interferences from some hydrophilic chemicals such as salts are hardly dissolved in the non-polar solvent (n-hexane). Second, the CsPbBr3 QDs have narrow fluorescence emission spectrum with half peak width of 26 nm, which is difficult to be effected by other chemical. So, the developed platform here showed high selectivity toward Sudan dyes detection.
Fig. 4.
The quenching efficiencies of the Sudan I–IV and other compounds. [The concentration of (a–d) Sudan I–IV are 10 μg mL−1; the concentrations of (e) l-Cys, (f) l-Lys, (g) l-Glu, (h) ascorbic acid, (i) fructose, (j) glucose, (k) sucrose, (l) urea, (m) β-cyclodextrin, (n) sodium citrate, (o) sodium benzoate are all saturated solution]. Test conditions: the excitation wavelength was 400 nm, temperature was 25 °C, incubation time was 4 min, the excitation slit width was 5 nm and the emission slit width was 5 nm
Real sample analysis
Because Sudan I–IV are phenyl-azoic derivatives with an orange-red appearance, so some unscrupulous businessmen adopted it for coloring foodstuffs, such as chili powder. However, owning to their azoic structure, Sudan I–IV has evident toxic effects to human healthiness. Thus, determinations of Sudan I–IV in chili powder are required for the assurance of consumer healthiness. Thus, the developed method was employed to determine Sudan dyes in the chili powder samples. The results are given in Table 1, the recoveries of Sudan I–IV were obtained, which ranged from 97.0 to 103.0% and the relative standard deviations (RSD, n = 3) were less than 2.9%. To assess the accuracy of the proposed fluorescence assay platform, the obtained results were compared with those obtained via the National Standard of the People’s Republic of China (GB/T 19681-2005) (Table 1). These results indicating that the proposed sensor can be successfully applied for the detection of Sudan dyes in real samples.
Table 1.
Results of Sudan dyes determination from chili powder using the proposed method (P) and high performance liquid chromatography GB/T 19681-2005 (G)
| Sudan dyes | Found value (μg mL−1) | Added value (μg mL−1) | Found value (μg mL−1) | Recovery (%) | RSD (n = 3%) | |||
|---|---|---|---|---|---|---|---|---|
| P | G | P | G | P | G | |||
| I | 0.00 | 0.50 | 0.49 | 0.40 | 98.00 | 80.00 | 1.3 | 1.8 |
| II | 0.00 | 0.50 | 0.51 | 0.44 | 103.00 | 88.00 | 1.9 | 1.6 |
| III | 0.00 | 0.50 | 0.49 | 0.42 | 98.00 | 84.00 | 2.9 | 2.5 |
| IV | 0.00 | 0.50 | 0.50 | 0.43 | 100.00 | 86.00 | 1.7 | 1.7 |
Conclusion
In conclusion, we have successfully developed a fluorescent assay platform based on CsPbBr3 QDs, which provided an efficient approach for the detection of Sudan I–IV with good selectivity and sensitivity. This fluorescent assay platform has many advantages, such as simplicity, rapidity, low-cost, high sensitivity and high selectivity. Furthermore, the assay platform has been successfully employed in the detection of Sudan I–IV in chilli powder samples with satisfactory results. This work has opened a gate for the use of CsPbX3 (X = Cl, Br, and I or mixed halide systems Cl/Br and Br/I) in fluorescence detection and also gives new insight into monitoring the quality of chilli powder.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21475043 and 21675051), and The Foundation of the Science and Technology Department of Hunan Province (2016SK2020).
Footnotes
Electronic supplementary material
The online version of this article (10.1007/s13197-018-3167-1) contains supplementary material, which is available to authorized users.
References
- Akkerman QA, D’Innocenzo V, Accornero S, Scarpellini A, Petrozza A, Prato M, Manna L. Tuning the optical properties of cesium lead halide perovskite nanocrystals by anion exchange reactions. J Am Chem Soc. 2015;137:10276–10281. doi: 10.1021/jacs.5b05602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alivisatos AP. Semiconductor clusters, nanocrystals, and quantum dots. Science. 1996;271:933–937. doi: 10.1126/science.271.5251.933. [DOI] [Google Scholar]
- Anibal CVD, Marsal LF, Callao MP, Ruisánchez I. Surface Enhanced Raman Spectroscopy (SERS) and multivariate analysis as a screening tool for detecting Sudan I dye in culinary spices. Spectrocheim Acta A. 2012;87:135–141. doi: 10.1016/j.saa.2011.11.027. [DOI] [PubMed] [Google Scholar]
- Chen NY, Li HF, Gao ZF, Qu F, Li NB, Luo HQ. Utilizing polyethyleneimine-capped silver nanoclusters as a new fluorescence probe for Sudan I–IV sensing in ethanol based on fluorescence resonance energy transfer. Sens Actuat B Chem. 2014;193:730–736. doi: 10.1016/j.snb.2013.12.020. [DOI] [Google Scholar]
- Cheung W, Shadi IT, Xu Y, Goodacre R. Quantitative analysis of the banned food dye Sudan-1 using surface enhanced raman scattering with multivariate chemometrics. J Phys Chem C. 2010;114:7285–7290. doi: 10.1021/jp908892n. [DOI] [Google Scholar]
- Dong Y, Wang R, Li G, Chen C, Chi Y, Chen G. Polyamine-functionalized carbon quantum dots as fluorescent probes for selective and sensitive detection of copper ions. Anal Chem. 2012;84:6220–6224. doi: 10.1021/ac3012126. [DOI] [PubMed] [Google Scholar]
- Dubertret B, Skourides P, Norris DJ, Noireaux V, Brivanlou AH, Libchaber A. In vivo imaging of quantum dots encapsulated in phospholipid micelles. Science. 2002;298:1759–1762. doi: 10.1126/science.1077194. [DOI] [PubMed] [Google Scholar]
- Ensafi AA, Rezaei B, Amini M, Heydari-Bafrooei E. A novel sensitive DNA–biosensor for detection of a carcinogen, Sudan II, using electrochemically treated pencil graphite electrode by voltammetric methods. Talanta. 2012;88:244–251. doi: 10.1016/j.talanta.2011.10.038. [DOI] [PubMed] [Google Scholar]
- Haustein HD, Tebrügge G, Rohlfs W, Kneer R. Local heat transfer coefficient measurement through a visibly-transparent heater under jet-impingement cooling. Int J Heat Mass Transf. 2012;55:6410–6424. doi: 10.1016/j.ijheatmasstransfer.2012.06.029. [DOI] [Google Scholar]
- Kim HN, Ren WX, Kim JS, Yoon J. ChemInform abstract: fluorescent and colorimetric sensors for detection of lead, cadmium, and mercury ions. Chem Soc Rev. 2011;41:3210–3244. doi: 10.1039/C1CS15245A. [DOI] [PubMed] [Google Scholar]
- Liu Y, Song Z, Dong F, Zhang L. Flow injection chemiluminescence determination of Sudan I in hot chilli sauce. J Agric Food Chem. 2007;55:614–617. doi: 10.1021/jf063332h. [DOI] [PubMed] [Google Scholar]
- Look DC, Manthuruthil JC. Electron and hole conductivity in CuInS2. J Phys Chem Solids. 1976;37:173–180. doi: 10.1016/0022-3697(76)90157-8. [DOI] [Google Scholar]
- Louie A. Multimodality imaging probes: design and challenges. Chem Rev. 2010;110:3146–3195. doi: 10.1021/cr9003538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazzotti F, Di Donna L, Maiuolo L, Napoli A, Salerno R, Sajjad A, Sindona G. Assay of the set of all Sudan azodye (I, II, III, IV, and para-red) contaminating agents by liquid chromatography–tandem mass spectrometry and isotope dilution methodology. J Agric Food Chem. 2008;56:63–67. doi: 10.1021/jf072286y. [DOI] [PubMed] [Google Scholar]
- Medintz IL, Clapp AR, Mattoussi H, Goldman ER, Fisher B, Mauro JM. Self-assembled nanoscale biosensors based on quantum dot FRET donors. Nat Mater. 2003;2:630–638. doi: 10.1038/nmat961. [DOI] [PubMed] [Google Scholar]
- Medintz IL, Uyeda HT, Goldman ER, Mattoussi H. Quantum dot bioconjugates for imaging, labelling and sensing. Nat Mater. 2005;4:435–446. doi: 10.1038/nmat1390. [DOI] [PubMed] [Google Scholar]
- Murray CB, Norris DJ, Bawendi MG. Synthesis and characterization of nearly monodisperse CdE (E = sulfur, selenium, tellurium) semiconductor nanocrystallites. J Am Chem Soc. 2002;115:8706–8715. doi: 10.1021/ja00072a025. [DOI] [Google Scholar]
- Ou Y, Pei L, Lai K, Huang Y, Rasco BA, Wang X, Fan Y. Rapid analysis of multiple sudan dyes in chili flakes using surface-enhanced raman spectroscopy coupled with Au–Ag core-shell nanospheres. Food Anal Methods. 2017;10:1–10. doi: 10.1007/s12161-016-0618-z. [DOI] [Google Scholar]
- Protesescu L, Yakunin S, Bodnarchuk MI, Krieg F, Caputo R, Hendon CH, Yang RX, Walsh A, Kovalenko MV. Nanocrystals of cesium lead halide perovskites (CsPbX3, X = Cl, Br, and I): novel optoelectronic materials showing bright emission with wide color gamut. Nano Lett. 2015;15:3692–3696. doi: 10.1021/nl5048779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ritter KA, Lyding JW. The influence of edge structure on the electronic properties of graphene quantum dots and nanoribbons. Nat Mater. 2009;8:235–242. doi: 10.1038/nmat2378. [DOI] [PubMed] [Google Scholar]
- Sapsford KE, Lorenzo B, Medintz IL. Materials for fluorescence resonance energy transfer analysis: beyond traditional donor-acceptor combinations. Angew Chem Int Ed. 2006;45:4562–4589. doi: 10.1002/anie.200503873. [DOI] [PubMed] [Google Scholar]
- Sciuto S, Esposito G, Dell’Atti L, Guglielmetti C, Acutis PL, Martucci F. Rapid screening technique to Identify Sudan Dyes (I to IV) in adulterated tomato sauce, chilli powder, and palm oil by innovative high-resolution mass spectrometry. J Food Prot. 2017;80:640–644. doi: 10.4315/0362-028X.JFP-16-313. [DOI] [PubMed] [Google Scholar]
- Shan WC, Xi JZ, Sun J, Zhang YJ, Wang JP. Production of the monoclonal antibody against Sudan 4 for multi-immunoassay of Sudan dyes in egg. Food Control. 2012;27:146–152. doi: 10.1016/j.foodcont.2012.03.017. [DOI] [Google Scholar]
- Shao N, Zhang Y, Cheung S, Yang R, Chan W, Mo T, Li K, Liu F. Copper ion-selective fluorescent sensor based on the inner filter effect using a spiropyran derivative. Anal Chem. 2005;77:7294–72303. doi: 10.1021/ac051010r. [DOI] [PubMed] [Google Scholar]
- Singh SC, Mishra SK, Srivastava RK, Gopal R. Optical properties of selenium quantum dots produced with laser irradiation of water suspended Se nanoparticles. J Phys Chem C. 2010;114:17374–17384. doi: 10.1021/jp105037w. [DOI] [Google Scholar]
- Sricharoen P, Limchoowong N, Techawongstien S, Chanthai S. New approach applying a pet fish air pump in liquid-phase microextraction for the determination of Sudan dyes in food samples by HPLC. J Sep Sci. 2017;40:3848–3856. doi: 10.1002/jssc.201700642. [DOI] [PubMed] [Google Scholar]
- Stiborová M, Martínek V, Rýdlová H, Hodek P, Frei E. Sudan I is a potential carcinogen for humans: evidence for its metabolic activation and detoxication by human recombinant cytochrome P450 1A1 and liver microsomes. Cancer Res. 2002;62:5678–5684. [PubMed] [Google Scholar]
- Toutchkine A, Kraynov V, Hahn K. Solvent-sensitive dyes to report protein conformational changes in living cells. J Am Chem Soc. 2003;125:4132–4145. doi: 10.1021/ja0290882. [DOI] [PubMed] [Google Scholar]
- Wang J, Wei K, Li H, Li QX, Li J, Xu T. A sensitive and selective enzyme-linked immunosorbent assay for the analysis of para red in foods. Analyst. 2012;137:2136–2142. doi: 10.1039/c2an35127g. [DOI] [PubMed] [Google Scholar]
- Wei S, Yang Y, Kang X, Wang L, Huang L, Pan D. Room-temperature and gram-scale synthesis of CsPbX3 (X = Cl, Br, I) perovskite nanocrystals with 50–85% photoluminescence quantum yields. Chem Commun. 2016;52:7265–7268. doi: 10.1039/C6CC01500J. [DOI] [PubMed] [Google Scholar]
- Wu X, Liu H, Liu J, Haley KN, Treadway JA, Larson JP, Ge N, Peale F, Bruchez MP. Immunofluorescent labeling of cancer marker Her2 and other cellular targets with semiconductor quantum dots. Nat Biotechnol. 2003;21:41–46. doi: 10.1038/nbt764. [DOI] [PubMed] [Google Scholar]
- Xiao Z, Loughlin F, George GN, Howlett GJ, Wedd AG. C-terminal domain of the membrane copper transporter Ctr1 from Saccharomyces cerevisiae binds four Cu(I) ions as a cuprous-thiolate polynuclear cluster: sub-femtomolar Cu(I) affinity of three proteins involved in copper trafficking. J Am Chem Soc. 2004;126:3081–3090. doi: 10.1021/ja0390350. [DOI] [PubMed] [Google Scholar]
- Xu X-Y, Tian X-G, Cai L-G, Xu Z-L, Lei H-T, Wang H, Sun Y-M. Molecularly imprinted polymer based surface plasmon resonance sensors for detection of Sudan dyes. Anal Methods. 2014;6:3751–3757. doi: 10.1039/C3AY42230E. [DOI] [Google Scholar]
- Yao J, Yang M, Duan Y. Chemistry, biology, and medicine of fluorescent nanomaterials and related systems: new insights into biosensing, bioimaging, genomics, diagnostics, and therapy. Chem Rev. 2014;114:6130–6178. doi: 10.1021/cr200359p. [DOI] [PubMed] [Google Scholar]
- Zeng H, Du XW, Singh SC, Kulinich SA, Yang S, He J, Cai W. Nanomaterials via laser ablation/irradiation in liquid: a review. Adv Funct Mater. 2012;22:1333–1353. doi: 10.1002/adfm.201102295. [DOI] [Google Scholar]
- Zhou J, Yang Y, Zhang CY. Toward biocompatible semiconductor quantum dots: from biosynthesis and bioconjugation to biomedical application. Chem Rev. 2015;115:11669–11717. doi: 10.1021/acs.chemrev.5b00049. [DOI] [PubMed] [Google Scholar]
- Zhou M, Chen X, Xu Y, Qu J, Jiao L, Zhang H, Chen H, Chen X. Sensitive determination of Sudan dyes in foodstuffs by Mn–ZnS quantum dots. Dyes Pigments. 2013;99:120–126. doi: 10.1016/j.dyepig.2013.04.027. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.