Abstract
The von Willebrand receptor complex, which is composed of the glycoproteins Ibα and Ibβ, V and IX, plays an essential role in the earliest steps in hemostasis. Over the last four decades, it has become apparent that loss of function of any one of three of the genes encoding these glycoproteins namely, GP1BA, GP1BB and GP9, leads to autosomal recessive macrothrombocytopenia complicated by bleeding. A small number of variants in GP1BA have been reported to cause a milder and dominant form of macrothrombocytopenia but only two tentative reports exists of such a variant in GP1BB. By analyzing data from a collection of over 1,000 genome-sequenced patients with a rare bleeding and/or platelet disorder, we have identified a significant association between rare monoallelic variants in GP1BB and macrothrombocytopenia. In order to strengthen our findings, we sought further cases in two additional collections in the UK and Japan. Across 18 families exhibiting phenotypes consistent with autosomal dominant inheritance of macrothrombocytopenia, we report on 27 affected cases carrying one of 9 rare variants in GP1BB.
Introduction
The earliest step in hemostasis is the tethering of platelets to the damaged endothelium through their cell surface receptor for von Willebrand factor (VWF). The glycoproteins (GPs) Ibα and Ibβ, in association with GPV and GPIX, form the transmembrane receptor complex on platelets for VWF1,2. The leucine-rich repeat (LRR) domain of GPIbα contains the binding site for VWF while the β chain contributes to the surface expression of the complex, which, by phosphorylating its intracellular domain, also participates in downstream signaling. GPIbβ is synthesized from a 1.5 kb mRNA transcribed from GP1BB and is highly expressed in megakaryocytes but absent from other blood cell progenitors3. The mRNA encodes a 206 amino acid long transmembrane protein with a 22 kD molecular mass and an extracellular LRR domain4–6.
Bernard and Soulier were the first to describe a patient with a rare autosomal recessive syndrome (Bernard-Soulier syndrome; BSS) characterized by giant platelets with concomitant thrombocytopenia complicated by severe bleeding7. Platelets from patients with this syndrome do not agglutinate in response to ristocetin and show a subtly reduced aggregation response to thrombin. Over four decades, 45, 39 and 28 variants have been identified in GP1BA, GP1BB and GP9, respectively, that, if present as compound heterozygotes or homozygotes, cause the VWF receptor to be absent or reduced in function8. Only four variants in GP1BA with dominant effects have been reported, one being the “Bolzano variant”9, and a further three observed in three isolated pedigrees8. Only two variants in GP1BB have been reported as being possibly responsible for dominant macrothrombocytopenia. One encoding Y113C was identified in two Japanese families and led to initial speculation that variants in the subunits of the GPIb/IX complex could span a spectrum of platelet and bleeding phenotypes10,11. Another, encoding R42C, was observed in only one Japanese patient and data from family or functional studies supporting an association with a dominant phenotype were lacking12. There are no reports of variants in GP9 having a dominant effect, and BSS cases due to variants in GP5 do not exist because GPV is not required for receptor expression13.
We have found a significant statistical association between rare monoallelic non-synonymous variants in GP1BB and macrothrombocytopenia. Family history and co-segregation data from 17 pedigrees, in which 26 affected cases carry one of 9 rare variants in GP1BB, are consistent with autosomal dominant inheritance.
Study Design
Study population
The study population comprises a discovery collection and two validation collections. The discovery collection consists of cases with an assumed inherited bleeding or platelet disorder (BPD) of unknown molecular aetiology or their relatives enrolled in the NIHR BioResource after providing informed written consent14. The BioResource cohort was enrolled over a 4-year period and is composed of 1,542 patients with a BPD or their close relatives, with a further 5,422 patients with other rare inherited disorders or their close relatives (https://bioresource.nihr.ac.uk/rare-diseases/clinicians/). The latter collection contains data from 75 patients with thrombocytopenia assessed with the ThromboGenomics diagnostic platform15 and 301 probands from a Japanese collection16 referred over an 8-year period for a suspected diagnosis of inherited macrothrombocytopenia and their relatives.
Data analysis
Coding of clinical and laboratory phenotypes with Human Phenotype Ontology (HPO) terms and collection of numerical and family history data were performed as described previously14. Variants from high-throughput sequencing were called and annotated using Isaac (Illumina, Inc; whole-genome sequencing) or as described previously15 (capture-based sequencing). We used phenotype similarity regression17 (SimReg) to identify statistical associations between presence of a variant affecting protein sequence in a gene and similarity to a latent HPO-coded phenotype in the discovery collection, subsequently corroborated using Fisher’s exact test. The variants obtained by high-throughput DNA sequencing of the probands were genotyped by Sanger sequencing in relatives who agreed to participate in this study. The variants identified in all members of the Japanese collection were obtained by Sanger sequencing. Where possible, complete blood counts (CBCs) were obtained using automated hematology analyzers, VWF receptor expression levels on platelets were determined by cytometry and platelet function was tested by light transmission aggregometry. Macrothrombocytopenia was deemed present if the platelet count was below 150 x 109/l, the mean platelet volume above 12fl or if there was clear platelet anisocytosis with a subset of platelets being abnormally large.
Results and Discussion
We identified a strong statistical association between presence of rare non-synonymous monoallelic DNA variants (population allele frequency less than 1/10,000) in GP1BB and macrothrombocytopenia in the discovery collection (SimReg posterior probability = 0.93 with inferred characteristic phenotype preferentially included the HPO term “Increased mean platelet volume”, Fisher’s p = 2.10 x 10-6). All eight probands with macrothrombocytopenia from the discovery collection had a family history suggestive of autosomal dominant inheritance. This mode of transmission was corroborated by results from co-segregation studies (Figure 1; p = 1.95x10-3). Systematic review of rare variants in these eight cases within the 15 established genes implicated in macrothrombocytopenia18, including GP1BA and GP9, did not reveal any alternative variants that could plausibly explain the platelet phenotype. We searched for further cases in the validation collection and identified four further probands in the ThromboGenomics dataset and six further probands in the Japanese collection, who carried a variant specific to people of Japanese ancestry (Figure 1).
Figure 1.
Graphical representation of the 18 pedigrees with autosomal dominant inheritance of macrothrombocytopenia in the discovery and validation collections. No genome-wide excess relatedness among individuals subjected to high-throughput sequencing from different pedigrees sharing the same variant (pedigrees B and I, C and J, D and E and H and L) could be detected by genetic analysis,25 although the shared variants in GP1BB may have been co-inherited from a distant common ancestor. Filled symbols: macrothrombocytopenia, grey symbols: unknown, blank symbols: normal platelet count and volume and absence of macrothrombocytes. Squares: males, circles: females, +/M: heterozygous, +/+: wildtype. A second variant encoding L175P was identified by Sanger sequencing in K-3 absent from K-6 which might influence PLT and MPV by affecting the cytoskeleton. The probability of the genotyping results under the null hypothesis of random segregation, conditional on the genotypes of the index cases, is 0.59 = 1.95x10-3.
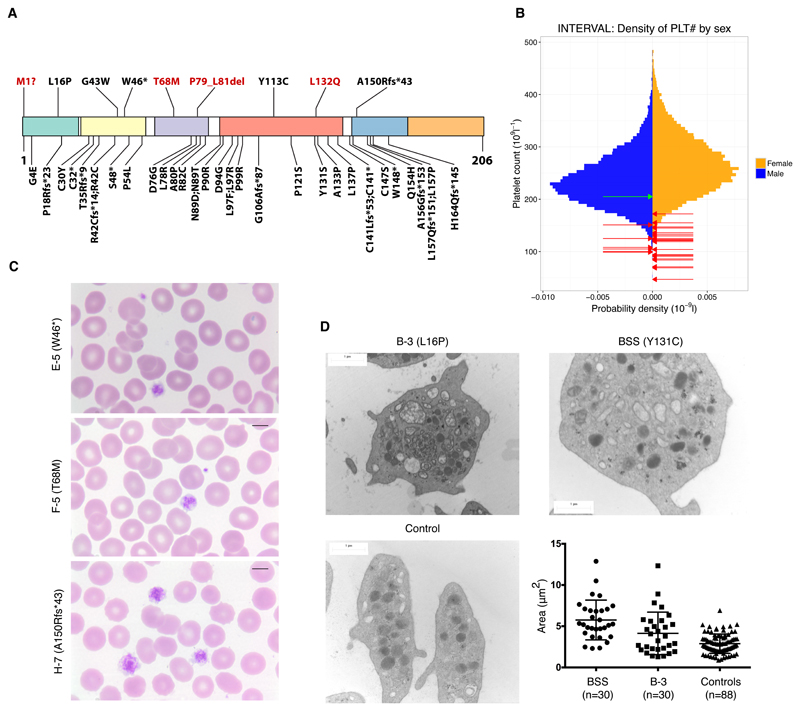
Altogether, we identified nine unique variants in GP1BB, one resulting in a disruption of the canonical methionine start codon, one encoding a premature stop at residue 46, five missense variants at L16P, G43W, T68M, Y113C and L132Q, a three-amino acid deletion P79_L81del, and a frameshift in the codon for residue A150 leading to an alternative reading frame with a premature stop after 43 further residues (Figure 2A). Assessment of the potential effect of the missense and in-frame variants on GPIbβ structure, supported by prior work by McEwan et al,19 is available in Supplemental Table 2 and Supplemental Figure 1. Only the variant encoding G43W is present in the ExAC database20 of over 60,000 exome-sequenced individuals, where it is heterozygous in just one individual. It is noteworthy that the variant underlying the premature stop at residue 46, the variant leading to a frameshift at residue 150 and the missense variants encoding L16P, G43W and Y113C have been previously implicated in BSS8,10,21,22. We reason that the effects of these five variants in heterozygosity, except for Y113C10,11, went unnoticed because the counts of the parents of these BSS probands were not measured or were not deemed notably low in isolation from other monoallelic cases.
Figure 2.
A: Localization and consequence of the nine distinct rare variants identified in GP1BB (shown above the diagram) in the context of all 39 variants published previously, excluding the 22q11.2 deletion. Novel variants are shown in red. Known variants were obtained from a recent review8, two publications on BSS published since the review26,27 and the article reporting a single case with dominant macrothrombocytopenia possibly due to a heterozygous variant encoding R42C12. The variant encoding T68M was observed in an unaffected relative of a non-BPD rare disease patient for whom a CBC was unavailable. Only the variant encoding G43W is present (in one individual) in the ExAC database. The domains shown in colors, from left to right, are signal peptide (1–25), LRR N-terminal (27–55), LRR (60–83), LRR C-terminal (89–143), transmembrane domain (148–172) and cytoplasmic domain (173–206). B: Gender-stratified histograms of platelet count measurements obtained using a Sysmex hematology analyzer from 48,345 blood donors from the INTERVAL randomized controlled trial28 after adjustment for technical artifacts. The red arrows superimposed upon the histograms indicate the gender and values for patients with one of the variants in GP1BB reported herein. For individuals I-3 and L-3 with a range of platelet counts, the midpoints were used. The green arrow indicates the gender and value for individual E-1, who is homozygous for the wildtype allele. Any superimposed arrows have been nudged slightly along the vertical axis so they can be distinguished. C: images of blood films from cases E-5, F-5 and H-7 with different variants in GP1BB (encoding W46*, T68M and A150Rfs*43). Original magnification, x100; May-Grünwald-Giemsa stain. D: images show representative electron micrographs of platelets from patient B-3, a patient with BSS (homozygous for a variant encoding Y131C) and an unaffected control, while the dot plot shows area measurements of 30 randomly selected platelets from a BSS patient, 30 from case B-3 and 88 from three controls (30+30+28 platelets) with similar distributions. There was a significant difference amongst the means of the three groups (p<0.0001, one-way analysis of variance). There was a significant difference between the mean area of platelets from the BSS case and patient B-3 (p=0.0021), between the BSS case and controls (p<0.0001) and between B-3 and controls (p=0.004); Tukey's method for multiple comparisons.
The count and mean volume of platelets of the 18 probands was 107.9 x109/l (range 47–172 x109/l) and 12.74 fl (range 10.7–14.3fl), respectively (Figure 2B, Supplemental Table 1). In several cases, May-Grünwald-Giemsa stained blood smears revealed platelet anisocytosis with a subset of platelets being abnormally large, however blood films were not available for all cases (Figure 2C, Supplemental Table 1). Electron micrographs of the platelets of B-3 show significantly increased platelet surface area that is intermediate between healthy controls and BSS cases but includes a small number of giant platelets (Figure 2D). Thus, the mean platelet volumes obtained by automated CBC analysis may obscure an increased spread of platelet volumes. Bleeding diathesis, including menorrhagia, epistaxis, spontaneous bleeding and post-partum bleeding, was reported in nine out of 23 females but none of the eight males for whom platelet count and bleeding phenotype information was available, which suggests that the overall propensity to bleeding is, at most, only marginally increased in these patients relative to the general population. There was a reduction of at least 30% of GPIbα on platelets as measured by flow cytometry in cases from eight of the nine families for which measurements were available (Supplemental Table 1).
Each VWF receptor complex has four GPIbβ molecules, which are covalently bonded with two copies of GPIbα through cysteines at residues 147 and 526/527 respectively23 and non-convalently paired with two molecules each of GPV and GPIX24. This highly uneven stoichiometry may explain why a single allele encoding a mutated GPIbβ molecule may exert a dominant negative effect on the function of the VWF complex. It could also exacerbate haploinsufficiency in the cases with disruption of the methionine start codon and truncation at residue 46. It is important to note that while the overall association between monoallelic variants in GP1BB and macrothrombocytopenia is robust, it is possible that some of the nine variants reported here are individually not causal. Therefore further work and the continued sharing of genotype and phenotype data from many patients with macrothrombocytopenia will be required to establish the molecular consequences of these variants definitively.
Supplementary Material
Key point.
Variants in GP1BB cause autosomal dominant macrothrombocytopenia
Acknowledgements
We thank research nurse Amy Frary at the University of Cambridge and Alice Glaser at Imperial College London for their contributions to the clinical phenotyping of NIHR BioResource participants. The NIHR BioResource–Rare Diseases is funded by the National Institute for Health Research of England (NIHR, www.nihr.ac.uk; award number RG65966). Research in the Ouwehand laboratory is supported by the British Heart Foundation, European Commission, Medical Research Council, NIHR and the Wellcome Trust and also receives support from National Health Service Blood and Transplant. C.L. and S.K.W. are supported by Medical Research Council Clinical Training Fellowships (MR/K023489/1). M.A.L. acknowledges support from the NIHR Imperial College Biomedical Research Centre. We thank Drs Ryoji Kobayashi, Masaaki Noguchi, Koji Miyazaki, Junichi Watanabe, Yoshiyuki Ogawa and Tomohiro Kajiguchi for providing patient samples.
Footnotes
Authorship
S.S. and E.T. wrote the paper with assistance from J.C.S., M.A.L., K.F. and W.H.O.; J.C.S. maintained pedigrees, managed clinical data and performed genotyping; S.K.W., A.K., C.L., P.N., K.P., K.P, D.J.P, C.R., M.A.L., D.P.H., R.C.T., A.D.M. N.B., K.F. and S.K. provided samples and clinical data; E.G.H. conducted structure analysis; C.J.P. and K.S. managed the NIHR BioResource sequencing pipeline; S.P. coordinated the NIHR BioResource–Rare Diseases BPD project; K.D. and I.S. managed ThromboGenomics; D.H. supervised S.S.; S.K. managed the Japanese collection; W.J.A. analyzed the INTERVAL dataset; K.F. analyzed microscopy data; D.G. analyzed data under the supervision of E.T.
Disclosure of Conflicts of Interest
None to declare
References
- 1.Ozaki Y, Asazuma N, Suzuki-Inoue K, Berndt MC. Platelet GPIb-IX-V-dependent signaling. J Thromb Haemost. 2005;3(8):1745–1751. doi: 10.1111/j.1538-7836.2005.01379.x. [DOI] [PubMed] [Google Scholar]
- 2.Lopez JA, Andrews RK, Afshar-Kharghan V, Berndt MC. Bernard-Soulier syndrome. Blood. 1998;91(12):4397–4418. [PubMed] [Google Scholar]
- 3.Watkins Na, Gusnanto A, de Bono B, et al. A HaemAtlas: characterizing gene expression in differentiated human blood cells. Blood. 2009;113(19):e1–9. doi: 10.1182/blood-2008-06-162958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kelly MD, Essex DW, Shapiro SS, et al. Complementary DNA cloning of the alternatively expressed endothelial cell glycoprotein Ib beta (GPIb beta) and localization of the GPIb beta gene to chromosome 22. J Clin Invest. 1994;93(6):2417–2424. doi: 10.1172/JCI117249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lopez JA, Chung DW, Fujikawa K, et al. The alpha and beta chains of human platelet glycoprotein Ib are both transmembrane proteins containing a leucine-rich amino acid sequence. Proc Natl Acad Sci U S A. 1988;85(7):2135–2139. doi: 10.1073/pnas.85.7.2135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zieger B, Hashimoto Y, Ware J. Alternative expression of platelet glycoprotein Ib(beta) mRNA from an adjacent 5’ gene with an imperfect polyadenylation signal sequence. J Clin Invest. 1997;99(3):520–525. doi: 10.1172/JCI119188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bernard J, Soulier JP. Sur une nouvelle variété de dystrophie thrombocytaire-hémorragipare congénitale. Semin Hop Paris. 1948;24:3217. [PubMed] [Google Scholar]
- 8.Savoia A, Kunishima S, De Rocco D, et al. Spectrum of the mutations in Bernard-Soulier syndrome. Hum Mutat. 2014;35(9):1033–1045. doi: 10.1002/humu.22607. [DOI] [PubMed] [Google Scholar]
- 9.De Marco L, Mazzucato M, Fabris F, et al. Variant Bernard-Soulier Syndrome Type Bolzano. J Clin Invest. 1990;86(July):25–31. doi: 10.1172/JCI114692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kunishima S, Lopez JA, Kobayashi S, et al. Missense mutations of the glycoprotein (GP) Ib beta gene impairing the GPIb alpha/beta disulfide linkage in a family with giant platelet disorder. Blood. 1997;89(7):2404–2412. [PubMed] [Google Scholar]
- 11.Kurokawa Y, Ishida F, Kamijo T, et al. A missense mutation (Tyr88 to Cys) in the platelet membrane glycoprotein Ibbeta gene affects GPIb/IX complex expression--Bernard-Soulier syndrome in the homozygous form and giant platelets in the heterozygous form. Thromb Haemost. 2001;86:1249–1256. [PubMed] [Google Scholar]
- 12.Kunishima S, Naoe T, Kamiya T, Saito H. Novel heterozygous missense mutation in the platelet glycoprotein Ibβ gene associated with isolated giant platelet disorder. Am J Hematol. 2001;68(4):249–255. doi: 10.1002/ajh.10000. [DOI] [PubMed] [Google Scholar]
- 13.Lopez JA, Leung B, Reynolds CC, et al. Efficient plasma membrane expression of a functional platelet glycoprotein Ib-IX complex requires the presence of its three subunits. J Biol Chem. 1992;267(18):12851–12859. [PubMed] [Google Scholar]
- 14.Westbury SK, Turro E, Greene D, et al. Human phenotype ontology annotation and cluster analysis to unravel genetic defects in 707 cases with unexplained bleeding and platelet disorders. Genome Medicine. 2015;7(1):36. doi: 10.1186/s13073-015-0151-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Simeoni I, Stephens JC, Hu F, et al. A comprehensive high-throughput sequencing test for the diagnosis of inherited bleeding, thrombotic and platelet disorders. Blood. 2016;127(23):2791–2804. doi: 10.1182/blood-2015-12-688267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kunishima S, Okuno Y, Yoshida K, et al. ACTN1 mutations cause congenital macrothrombocytopenia. Am J Hum Genet. 2013;92:431–438. doi: 10.1016/j.ajhg.2013.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Greene D, BioResource N, Richardson S, Turro E. Phenotype Similarity Regression for Identifying the Genetic Determinants of Rare Diseases. Am J Hum Genet. 2016;98(3):490–499. doi: 10.1016/j.ajhg.2016.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lentaigne C, Freson K, Laffan MA, et al. Inherited platelet disorders: toward DNA-based diagnosis. Blood. 2016;127(23):2814–23. doi: 10.1182/blood-2016-03-378588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.McEwan PA, Yang W, Carr KH, et al. Quaternary organization of GPIb-IX complex and insights into Bernard-Soulier syndrome revealed by the structures of GPIbbeta and a GPIbbeta/GPIX chimera. Blood. 2011;118(19):5292–5301. doi: 10.1182/blood-2011-05-356253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lek M, Karczewski KJ, Minikel EV, et al. Analysis of protein-coding genetic variation in 60,706 humans. Nature. 2016;536(7616):285–291. doi: 10.1038/nature19057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Moran N, Morateck PA, Deering A, et al. Surface expression of glycoprotein Iba is dependent on glycoprotein Ibb: evidence from a novel mutation causing Bernard-Soulier syndrome. Blood. 2000;96(2):532–539. [PubMed] [Google Scholar]
- 22.Kunishima S, Sako M, Yamazaki T, Hamaguchi M, Saito H. Molecular genetic analysis of a variant Bernard-Soulier syndrome due to compound heterozygosity for two novel glycoprotein Ibβ mutations. Eur J Haematol. 2006;77(6):501–512. doi: 10.1111/j.0902-4441.2006.t01-1-EJH2817.x. [DOI] [PubMed] [Google Scholar]
- 23.Luo S-Z, Mo X, Afshar-Kharghan V, et al. Glycoprotein Ibalpha forms disulfide bonds with 2 glycoprotein Ibbeta subunits in the resting platelet. Blood. 2007;109(2):603–609. doi: 10.1182/blood-2006-05-024091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dong JF, Sae-Tung G, Lopez JA. Role of glycoprotein V in the formation of the platelet high-affinity thrombin-binding site. Blood. 1997;89(12):4355–4363. [PubMed] [Google Scholar]
- 25.Astle WJ, Balding DJ. Population Structure and Cryptic Relatedness in Genetic Association Studies. Stat Sci. 2009;24(4):451–471. [Google Scholar]
- 26.Bragadottir G, Birgisdottir ER, Gudmundsdottir BR, et al. Clinical phenotype in heterozygote and biallelic Bernard-Soulier syndrome--a case control study. Am J Hematol. 2015;90(2):149–155. doi: 10.1002/ajh.23891. [DOI] [PubMed] [Google Scholar]
- 27.Qiao J, Davis AK, Morel-Kopp M-C, et al. Low levels of CD9 coincidental with a novel nonsense mutation in glycoprotein Ibbeta in a patient with Bernard-Soulier syndrome. Ann Hematol. 2015;94(12):2069–2071. doi: 10.1007/s00277-015-2473-1. [DOI] [PubMed] [Google Scholar]
- 28.Moore C, Sambrook J, Walker M, et al. The INTERVAL trial to determine whether intervals between blood donations can be safely and acceptably decreased to optimise blood supply: study protocol for a randomised controlled trial. Trials. 2014;15:363. doi: 10.1186/1745-6215-15-363. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.