Abstract
Myeloid-derived suppressor cells (MDSCs) are a prominent component of the pro-tumoral response. The phenotype of and mechanisms used by MDSCs is heterogeneous and requires more precise characterization in gastric cancer (GC) patients. Here, we have identified a novel subset of CD45+CD33lowCD11bdim MDSCs in the peripheral blood of GC patients compared to healthy individuals. CD45+CD33lowCD11bdim MDSCs morphologically resembled neutrophils and expressed high levels of the neutrophil marker CD66b. Circulating CD45+CD33lowCD11bdim MDSCs effectively suppressed CD8+ T cells activity through the inhibition of CD8+ T cell proliferation and interferon-γ (IFN-γ) and granzyme B (GrB) production. The proportion of CD45+CD33lowCD11bdim MDSCs also negatively correlated with the proportion of IFN-γ+CD8+ T cell in the peripheral blood of GC patients. GC patient serum-derived IL-6 and IL-8 activated and induced CD45+CD33lowCD11bdim MDSCs to express arginase I via the PI3K-AKT signaling pathway. This pathway contributed to CD8+ T cell suppression as it was partially rescued by the blockade of the IL-6/IL-8-arginase I axis. Peripheral blood CD45+CD33lowCD11bdim MDSCs, as well as IL-6, IL-8, and arginase I serum levels, positively correlated with GC progression and negatively correlated with overall patient survival. Altogether, our results highlight that a subset of neutrophilic CD45+CD33lowCD11bdim MDSCs is functionally immunosuppressive and activated via the IL-6/IL-8-arginase I axis in GC patients.
Introduction
Gastric cancer (GC) is the fourth most common cancer worldwide. GC patients frequently present with advanced stage disease, which has a poor prognosis and low survival rate1. The immune system of cancer patients is often perturbed by pro-tumorigenic signals from the tumor microenvironment. Counter to this, natural killer cells and T cells act as a critical component of anti-tumor immunity, in particular tumor-specific effector CD8+ T cells, which directly induce tumor cell cytotoxicity. Effector CD8+ T cell activity, however, is inhibited during the development and metastatic progression of GC2. This effect may still be amenable to immunomodulation, however, as tumor-specific CD8+ T cells from the peripheral blood of GC patients can still exert cytotoxicity following stimulation by peptide-pulsed cells in vitro3. Understanding the factors driving CD8+ T cell suppression is therefore critical for the most effective clinical modulation of anti-tumor immunity.
Immunosuppressive myeloid cells were first described in the 1980s in cancer patients4. A large body of evidence now exists on their immunosuppressive effects during cancer progression, with emphasis on their heterogeneous phenotypes and mechanisms of action. In humans, myeloid-derived suppressor cells (MDSCs) are broadly classified as either neutrophilic or MO MDSCs, and are phenotypically identified as being CD11b+CD15+CD66b+CD33+CD14− or CD11b+CD15−CD33+CD14+HLA-DR-/low, respectively5–8. In our previous studies, we observed a subset of immunosuppressive myeloid cells in the peripheral blood of GC patients. This myeloid subset was CD66+ but CD33lowCD11bdim in surface phenotype, rather than being typically CD11b+CD33+. A negative correlation was also observed between the proportions of CD33lowCD11bdim myeloid cells versus CD8+ T cells in the peripheral blood of GC patients. We thus hypothesized that the GC-selective CD33lowCD11bdim myeloid subset identified might function as MDSCs, and thereby detrimentally influence the progression of GC.
MDSCs are recruited by pro-inflammatory signals from the tumor microenvironment and exert their immunosuppressive activities through the upregulation of arginase I, iNOS, indoleamine 2, 3 deoxygenase (IDO), nitric oxide (NO), and reactive oxygen species (ROS)9,10. Arginase I is a highly conserved enzyme that metabolizes host L-arginine11 from the extracellular environment, results in decreased expression of the TCRζ-chain of CD3, and then impaired proliferation and cytokine production of T lymphocytes12. Human neutrophilic MDSCs are known to upregulate arginase I to inhibit CD8+ T cell activity13, while pro-inflammatory cytokine such as IL-6 and IL-8 were reported to regulate the expression or exocytosis of arginase I14,15. We then hypothesized that the CD45+CD33lowCD11bdim myeloid subset function as suppressive cells through arginase I and regulated by these pro-inflammatory factors.
In this study, we further characterized the prevalence, phenotype, and function of CD45+CD33lowCD11bdim MDSCs identified in peripheral blood of GC patients. We found that the CD45+CD33lowCD11bdim MDSCs exhibited a CD66b neutrophilic phenotype, and that increased frequencies correlated with tumor stage and decreased overall survival in GC patients. We further demonstrated that this subset suppressed CD8+ T cells IFN-γ and granzyme B production via IL-6-induced and/or IL-8-induced arginase I production. Suppression of CD8+ T cell activity could be partially rescued upon blockade of the IL-6/IL-8-arginase I axis. In conclusion, CD8+ T cell-mediated immunotherapy in GC patients may require the modulation of suppressive CD45+CD33lowCD11bdim MDSCs to be maximally effective, in particular through the blockade of the IL-6/IL-8-arginase I axis.
Results
Neutrophilic CD45+CD33lowCD11bdim myeloid cells are increased in the peripheral blood of GC patients
We first confirmed that CD45+CD33+CD11b+ myeloid cells were significantly increased in the peripheral blood of GC patients compared to healthy donors, in concordance with other reports16,17. Interestingly, we also identified a myeloid cell subset, which was CD45+CD33lowCD11bdim and uniquely appeared in the peripheral blood of GC patients compared to healthy donors (Fig. 1a). As MDSCs are frequently found in cancer patients, we analyzed for the potential correlation between the frequency of CD45+CD33lowCD11bdim myeloid cells and IFN-γ-producing CD8+ T cells in GC patients, and found it to be significantly inversed (Fig. 1b). We further characterized this CD45+CD33lowCD11bdim myeloid cell subset using known human neutrophilic or MO MDSC surface markers, and found that they expressed high levels of the neutrophil marker CD66b and exhibited a multi-lobar nucleus characteristic of neutrophils (Fig. 1c, d). We also examined the CD45+CD33lowCD11bhigh myeloid cells subset, which also highly expressed CD66b (Supplementary Figure 1). We thus identified a novel GC patient selective subset of neutrophilic CD45+CD33lowCD11bdim myeloid cells in peripheral blood.
Fig. 1. Peripheral blood CD45+CD33lowCD11bdim myeloid cells with a neutrophilic morphology are increased in patients with GC.
a Surface expression of CD33 versus CD11b on CD45+ gated cells from the peripheral blood of GC patients or healthy donors as analyzed by flow cytometry. b Correlation between the percentages of peripheral blood CD45+CD33lowCD11bdim myeloid cell and IFN-γ-producing CD8+ T cells in GC patients. c Neutrophil marker CD66b surface expression on CD45+ gated CD33lowCD11bdim myeloid cells from the peripheral blood of GC patients. d Wright staining of sorted peripheral blood CD45+CD33lowCD11bdim myeloid cells from a GC patient. (red: CD45+CD33+CD11bhigh cell; blue: CD45+CD33lowCD11bhigh cell; green: CD45+CD33lowCD11bdim cell)
Peripheral blood CD45+CD33lowCD11bdim myeloid cells suppress CD8+ T cell activity in vitro
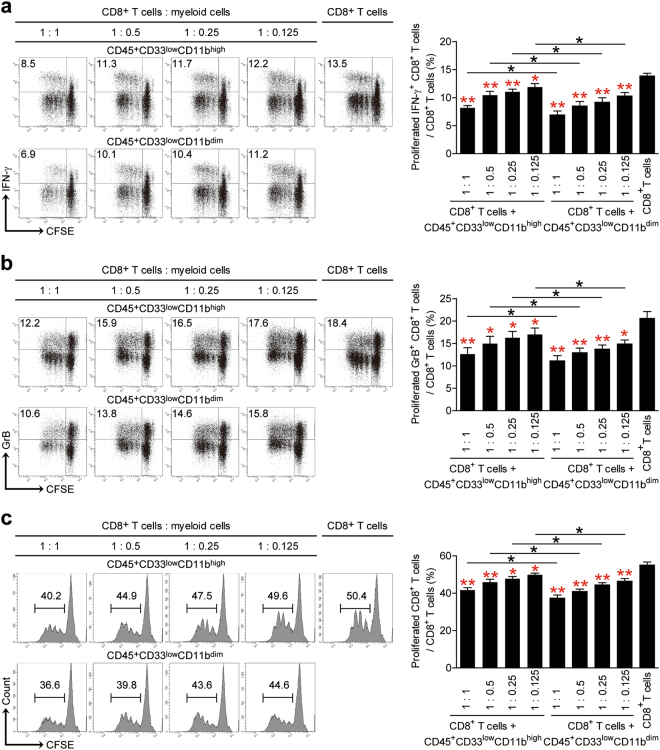
To evaluate the function of the CD45+CD33lowCD11bdim and also CD45+CD33lowCD11bhigh myeloid cell subsets, we sorted them from the peripheral blood of GC patients by fluorescence-activated cell sorting (FACS) and then co-cultured them with purified CD8+ T cells at different ratios for 5 days. We used typical regulatory T cells as positive control (data not shown), and found that both myeloid cell subsets suppressed CD8+ T cell proliferation and IFN-γ and GrB production upon allogeneic antigen stimulation (Fig. 2a–c). CD45+CD33lowCD11bdim myeloid cells were marginally more suppressive of CD8+ T cell activity compared to CD45+CD33lowCD11bhigh myeloid cells (Fig. 2a–c). Furthermore, CD45+CD33lowCD11bdim cell frequency positively correlated with patient tumor size and stage (Supplementary Figure 2). Altogether, these findings suggested that CD45+CD33lowCD11bdim myeloid cells behaved as MDSCs.
Fig. 2. Peripheral blood CD45+CD33lowCD11bdim myeloid cells suppress CD8+ T cell activity.
CFSE-labeled peripheral CD8+ T cells from healthy donors were co-cultured for 5 days with CD45+CD33lowCD11bhigh or CD45+CD33lowCD11bdim myeloid cells from autologous GC patients at different ratios. a CD8+ T cell proliferation, b IFN-γ and c granzyme B production was quantified by flow cytometry with data representative of three independent experiments (n = 3). *p < 0.05; **p < 0.01, and n.s, p > 0.05 for comparisons between respective groups (black) or compared with CD8+T cell control (red)
CD45+CD33lowCD11bdim myeloid cells suppress CD8+ T cell activity via arginase I production
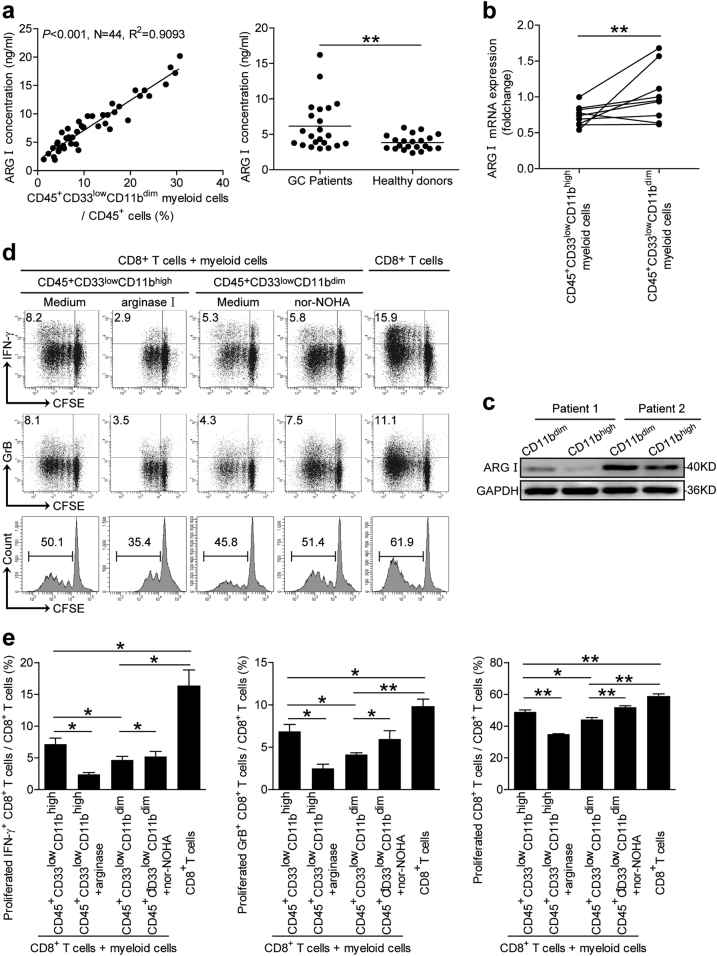
CD8+ T cell suppression can be mediated via cell-to-cell contact (i.e., engagement of checkpoint receptors) or soluble factor release (i.e., ROS, NO, and arginase I). Human neutrophilic MDSCs are known to upregulate arginase I to inhibit CD8+ T cell activity13. We thus investigated whether CD8+ T cell suppression was dependent on arginase I activity in CD45+CD33lowCD11bdim myeloid cells. The concentration of serum arginase I was increased in GC patients compared to healthy donors (Fig. 3a); and a positive correlation was found between serum arginase I concentration and the frequency of peripheral blood CD45+CD33lowCD11bdim myeloid cells (Fig. 3a). We then investigated the capacity of arginase I production by two myeloid subsets, and found that the CD45+CD33lowCD11bdim myeloid cells produce more arginase I both in mRNA and protein levels, when compared to the CD45+CD33lowCD11bhigh myeloid cells (Fig. 3b, c). To affirm whether arginase I operates in this process, we added arginase I or an arginase inhibitor nor-NOHA to CD45+CD33lowCD11bhigh/CD8+ T cell or CD45+CD33lowCD11bdim/CD8+ T cell co-cultures separately. Interestingly, arginase I incubation enhanced CD45+CD33lowCD11bhigh myeloid cell suppression of CD8+ T cell activity in terms of IFN-γ or GrB production (Fig. 3d, e). Additional of nor-NOHA inhibited this suppressive effect (Fig. 3d, e). Clinically, serum arginase I concentration positively correlated with tumor size and tumor stage (Supplementary Figure 3). These results indicated that the CD45+CD33lowCD11bdim myeloid cell subset suppressed CD8+ T cell activity at least partly through arginase I production.
Fig. 3. Peripheral blood CD45+CD33lowCD11bdim myeloid cells suppress CD8+T cell function via arginase I (ARG I) production.
a The correlation between the percentage of peripheral blood CD45+CD33lowCD11bdim myeloid cells and the serum concentration of arginase I was analyzed in GC patients. Serum arginase I concentration was also analyzed in GC patients compared to healthy donors. b, c Arginase I expression in CD45+CD33lowCD11bhigh myeloid cells and CD45+CD33lowCD11bdim myeloid cells by quantitative real-time PCR and western blot, respectively. d, e CFSE-labeled peripheral CD8+ T cells from healthy donors were co-cultured for 5 days with either autologous peripheral blood CD45+CD33lowCD11bhigh myeloid cells from GC patients in the presence or absence of arginase I, or autologous CD45+CD33lowCD11bdim myeloid cells from GC patients in the presence or absence of nor-NOHA. CD8+ T cell proliferation and IFN-γ and granzyme B production experiments are representative of three independent experiments (n = 3). *p < 0.05; **p < 0.01, and n.s, p > 0.05 between respective groups. ARG I arginase I
IL-6-induced and IL-8-induced CD45+CD33lowCD11bdim myeloid cell arginase I production via the PI3K-AKT signaling pathway
IL-6 and IL-8 have been reported to promote the production of arginase I in neutrophils14,15,18. We therefore first measured serum levels of IL-6 and IL-8 in GC patients, and found that it was increased compared to healthy donors (Fig. 4a). Serum IL-6 and IL-8 levels positively correlated with GC tumor size and tumor stage (Supplementary Figure 4 and 5). We also found a positive correlation between serum IL-6 and IL-8 levels and the proportion of peripheral blood CD45+CD33lowCD11bdim myeloid cells (Fig. 4b). A positive correlation also existed between serum IL-6 or IL-8 levels and serum arginase I levels in GC patients (Fig. 4b). We next investigated whether GC patient derived serum IL-6 and/or IL-8 could stimulate the production and secretion of arginase I in CD45+CD33lowCD11bdim myeloid cells. To test this, we added recombinant human IL-6 (rhIL-6) and/or IL-8 (rhIL-8) to CD45+CD33lowCD11bdim myeloid cells and incubated for 24 h. Increased arginase I production was detected upon rhIL-6 and/or rhIL-8 addition (Fig. 4c). Arginase I levels conversely decreased in a reciprocal experiment in which neutralizing anti-IL-6 and IL-8 Abs were added to the cultured CD45+CD33lowCD11bdim myeloid cells in the presence of 50% GC patient serum (Fig. 4c).
Fig. 4. IL-6 and IL-8 induces CD45+CD33lowCD11bdim myeloid cell production of arginase I via PI3K-AKT signaling.
a Serum IL-6 and IL-8 concentration levels in GC patients compared to healthy donors. b Correlations between the proportions of peripheral blood CD45+CD33lowCD11bdim myeloid cells or serum arginase I concentration and IL-6 or IL-8 serum concentrations in GC patients. c Arginase I expression and production in CD33lowCD11bdim myeloid cells exposed to recombinant IL-6 and/or IL-8, or 50% GC patient serum in the presence or absence of IL-6 and/or IL-8 neutralizing abs as analyzed by western blot and ELISA, respectively. d Arginase I expression in CD45+CD33lowCD11bdim myeloid cells exposed to 50% GC patient serum in the presence or absence of FLLL32 (STAT3 phosphorylation inhibitor), Wortmannin (PI3K inhibitor), or SB203580 (MAPK inhibitor; top panel). AKT and p-AKT expression levels in CD45+CD33lowCD11bdim myeloid cells exposed to IL-6 and/or IL-8 (middle panel), or 50% GC patient serum in the presence or absence of IL-6 and/or IL-8 neutralizing abs (bottom panels) as analyzed by western blot. *p < 0.05; **p < 0.01, and n.s, p > 0.05. ARG I arginase I
To investigate which signaling pathway(s) could mediate CD45+CD33lowCD11bdim myeloid cell production of arginase I, we pretreated CD45+CD33lowCD11bdim myeloid cells with a range of MDSC signaling pathway inhibitors before incubation in 50% GC patient serum. Interestingly, decreased arginase I production was observed following pretreatment with the PI3K inhibitor Wortmannin (Fig. 4d). AKT (Ser473) phosphorylation was increased when CD45+CD33lowCD11bdim myeloid cells were stimulated with rhIL-6 and rhIL-8 (Fig. 4d), and decreased following the addition of neutralizing anti-IL-6 and IL-8 Abs to 50% GC patient serum (Fig. 4d). Altogether, these results indicate that IL-6 and IL-8 can induce CD45+CD33lowCD11bdim myeloid cells to increase arginase I production via the PI3K-AKT signaling pathway.
Blockade of the IL-6/IL-8-arginase I pathway in CD45+CD33lowCD11bdim myeloid cells reduces the degree of CD8+ T cell suppression in vitro
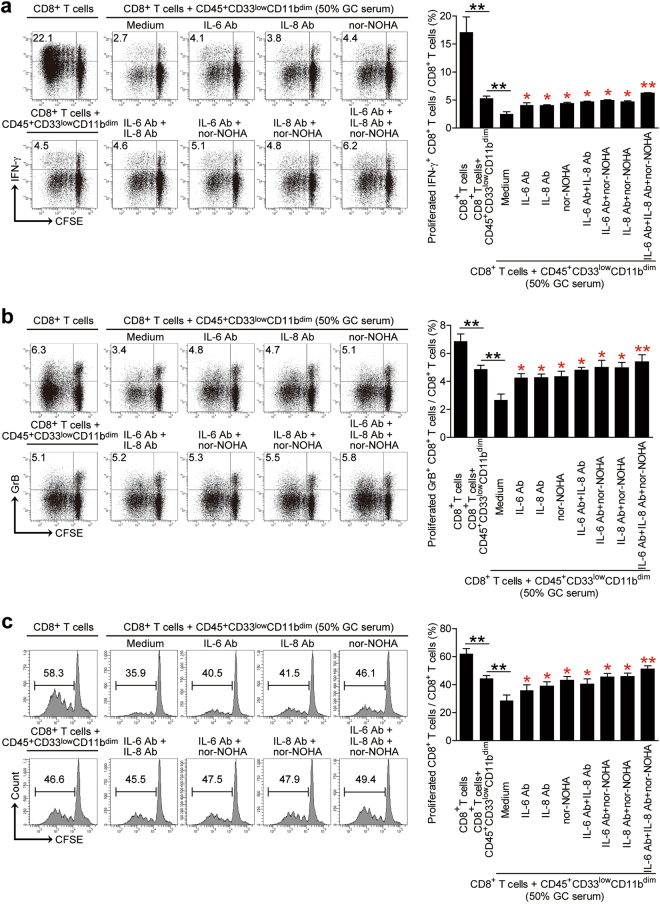
Neutralizing anti-IL-6 and/or IL-8 Abs and an arginase I inhibitor were added separately or altogether following 50% GC patient serum addition to the CD45+CD33lowCD11bdim myeloid and CD8+ T cell co-cultures. Addition of CD45+CD33lowCD11bdim myeloid cells alone or the presence of 50% GC patient serum in the co-culture decreased CD8+ T cells proliferation and IFN-γ or GrB production (Fig. 5a–c). Blockade of the IL-6/IL-8-arginase I axis attenuated the degree of reduced CD8+ T cell proliferation and IFN-γ or GrB production (Fig. 5a–c). These findings further confirm the involvement of the IL-6/IL-8-arginase I axis in CD45+CD33lowCD11bdim myeloid cell-mediated CD8+ T cell suppression.
Fig. 5. IL-6/IL-8-arginase I blockade reduces CD45+CD33lowCD11bdim myeloid cell-mediated CD8+ T cell suppression.
CFSE-labeled peripheral blood CD8+ T cells from healthy donors were co-cultured for 5 days with peripheral blood CD45+CD33lowCD11bdim myeloid cells from GC patients in the presence or absence of 50% GC patient serum. A proportion of samples were either co-cultured in the presence or absence of IL-6 and/or IL-8 neutralizing abs and/or nor-NOHA, and the proportion of a IFN-γ expressing, b granzyme B expressing and c proliferating CD8+ T cells quantitated by flow cytometry. Data representative of three independent experiments (n = 3). *p < 0.05; **p < 0.01, and n.s, p > 0.05 between respective groups (black) or compared with media control (red)
CD45+CD33lowCD11bdim myeloid cell frequency correlates with increased tumor progression and decreased patient survival
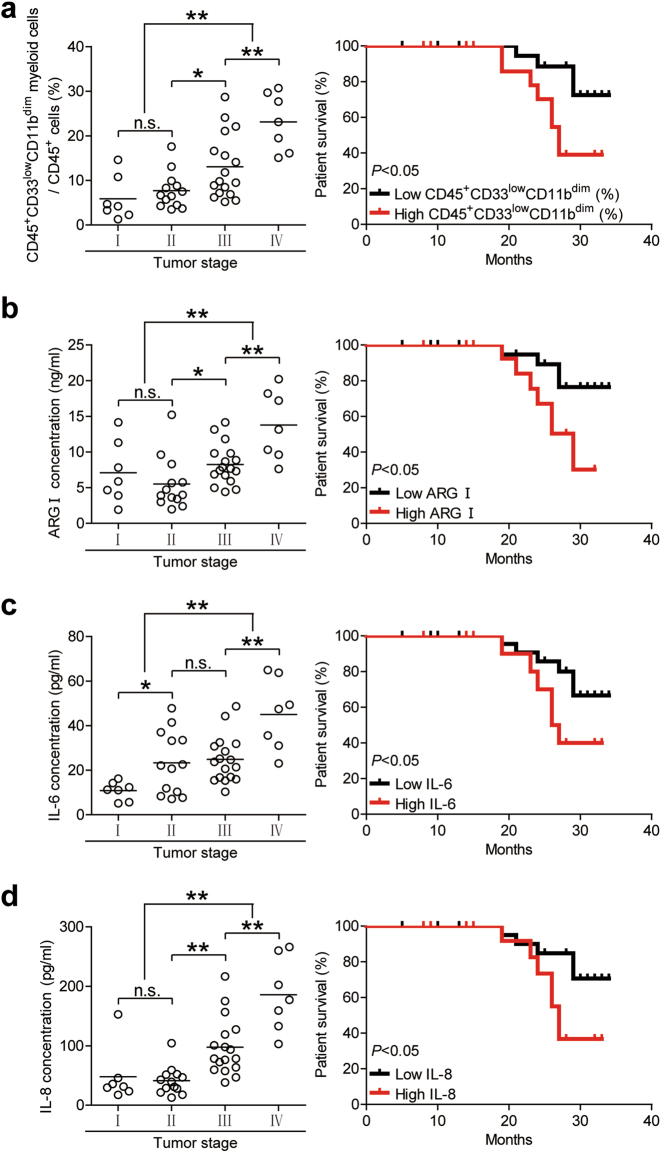
To further investigate whether CD45+CD33lowCD11bdim myeloid cells were a clinically relevant biomarker of GC, we staged GC patients according to the TNM classification system, and found that peripheral blood CD45+CD33lowCD11bdim myeloid cell frequency was positively associated with advanced stage GC progression (Fig. 6a). A comparison of GC patients based on high (≥9.2% median level) versus low (<9.2%) CD45+CD33lowCD11bdim myeloid cell frequencies showed that the 35-month survival rate was significantly lower in patients with a higher CD45+CD33lowCD11bdim myeloid cell frequency (Fig. 6a). A similar trend was observed when GC patients were separated by high (≥7.52 ng/ml) or low (<7.52 ng/ml) serum arginase I levels (Fig. 6b). We also evaluated the prognostic value of serum IL-6 and IL-8 levels for GC patient survival. The 35-month survival rate was also significantly lower in patients with higher IL-6 (≥ 22.37 pg/ml) or IL-8 (≥ 61.61 pg/ml) serum levels (Fig. 6c, d). Altogether, our findings demonstrate that peripheral blood CD45+CD33lowCD11bdim MDSCs suppress CD8+ T cell activity via the IL-6/IL-8-arginase I axis and are positively associated with GC tumor progression and negatively associated with overall patient survival.
Fig. 6. Peripheral blood CD45+CD33lowCD11bdim myeloid cell frequency, and IL-6, IL-8, and arginase I levels correlate with GC tumor progression and decreased patient survival.
Peripheral blood (a) CD45+CD33lowCD11bdim myeloid cell percentages (b) arginase I concentration, (c) IL-6 concentration, and (d) IL-8 concentration were measured and categorized by patient TNM stage. Kaplan-Meier plots were calculated for overall patient survival according to (a) median peripheral blood CD45+CD33lowCD11bdim myeloid cell percentage (9.2%) or (b) median arginase I concentration (7.52 ng/ml) or (c) median IL-6 concentration (22.37 pg/ml) or (d) median IL-8 concentration (61.61 pg/ml). *p < 0.05; **p < 0.01; n.s, p > 0.05. ARG arginase I
Discussion
MDSCs are a phenotypically and functionally heterogeneous population categorized by their ability to functionally suppress T cell activity in cancer patients and tumor-bearing mice6,10. In mice, MDSCs are phenotypically CD11b+Gr1+ like neutrophils and monocytes during homeostasis and classified as either polymorphonuclear (PMN)-MDSCs or monocytic (MO)-MDSCs. Murine PMN-MDSCs suppressed CD8+ antigen-specific T cell activity via ROS production, while MO-MDSCs suppressed CD8+ T cell activity non-specifically through arginase I and NO production19–21. In humans, MDSCs are also classified into PMN-MDSCs or MO-MDSCs. The neutrophil marker CD66b, a member of the carcinoembryonic antigen-like glycoprotein family, has also been reported as a marker of MDSCs in patients with head and neck cancer22, renal cell carcinoma23, and autoimmune diseases such as systemic lupus erythematosus24. In GC patients, we identified CD45+CD11b+CD33+ classical MDSCs as well as two additional subsets of myeloid cells, one is CD45+CD33lowCD11bdim and the other is CD45+CD33lowCD11high. However, in the peripheral blood of healthy donors, we only identified CD45+CD33lowCD11high cells (Fig. 1a). We therefore suspect that the appearance of CD45+CD33lowCD11bdim cells may be associated with GC. We then compared the functions of the two cell populations. Both CD45+CD33lowCD11bdim cells and CD45+CD33lowCD11high cells suppressed CD8+ T cell activity, although the CD45+CD33lowCD11bdim cells showed a stronger suppress activity (Fig. 2). These results indicated that both subsets function as myeloid suppressor cells in the circulation. It is possible that the CD45+ CD33lowCD11high cells may suppress autoantigen reactive T cells in healthy donors and play a role in preventing or limiting autoimmune diseases; however the CD45+CD33lowCD11bdim cells may function in the peripheral blood of GC patients to promote an immunosuppressive tumor microenvironment. We therefore focused on CD45+CD33lowCD11bdim MDSCs.
CD45+CD33lowCD11bdim MDSCs express CD66b, they also possess multi-lobar nuclei, further indicative of a granulocytic rather than MO phenotype22,23(Fig. 1c, d). Since CD45+CD33lowCD11bdim MDSCs were detectable in peripheral blood from GC patients but healthy donors, it is likely for them to have been selectively expanded by factors derived from the GC microenvironment. In lieu with this, we found that CD45+CD33lowCD11bdim MDSCs significantly suppressed CD8+ T cell activity in vitro and also correlated positively in frequency with GC stage and negatively with overall patient survival (Fig. 6). To our knowledge, this is the first description of a CD45+CD33lowCD11bdim subset of PMN-MDSCs in GC patients.
Aside from phagocytosis, mature granulocytes primarily exert their functions through the release of primary, secondary, and tertiary granules. We therefore speculated whether CD8+ T cell suppression by CD45+CD33lowCD11bdim MDSCs was dependent on the secretion of soluble factors, similar to classical MDSC subsets. MDSCs are recruited by pro-inflammatory signals from the tumor microenvironment or during autoimmune disease and other pathologic conditions. Functionally, they exert their immunosuppressive activities through the upregulation of arginase I, iNOS, IDO, NO, and ROS9,10. Since MDSC frequency positively correlates with serum arginase I levels24 and arginase I is constitutively expressed in human granulocytes25, we hypothesized whether granulocytic CD45+CD33lowCD11bdim MDSCs could suppress CD8+ T cells via arginase I production. A positive correlation was found between serum arginase I levels and the frequency of peripheral blood CD45+CD33lowCD11bdim MDSCs in our cohort of GC patients, serum arginase I levels were increased in GC patients compared to healthy donors (Fig. 3a). Arginase I metabolizes L-arginine from the extracellular environment11. L-arginine depletion by MDSCs has been reported to inhibit the re-expression of the CD3 TCRζ-chain and impair antigen-specific CD8+ and CD4+ T cell proliferation12. CD45+CD33lowCD11bdim MDSCs also inhibited CD8+ T cell proliferation and IFN-γ or GrB production in vitro, in accordance with MDSC-mediated immunosuppression. Granzymes are stored in granules in cytotoxic lymphocytes26,27 and released upon stimulation28,29. However, production of IFN-γ starts from the activation of STAT1 and T-bet, through transcription and translation, which takes 8–11 h to reach the peak30. Therefore the production of IFN-γ by CD45+CD33lowCD11bdim MDSCs after treatment was likely not yet completely restored when compared with granzymes (Fig. 5).
Numerous serum or tumor-derived pro-inflammatory factors have been implicated in MDSC survival and/or activity. Increased levels of serum IL-6 and IL-8 are commonly observed in cancer patients, and are positively correlated with the number of infiltrating MDSCs in breast cancer31. IL-6 induces neutrophil elastase release32, while IL-8 induces neutrophil exocytosis of arginase I in non-small cell lung cancer patients15. In our study, we also found a positive correlation between serum IL-6 and IL-8 concentrations and the frequency of peripheral blood CD45+CD33lowCD11bdim MDSCs in GC patients (Fig. 4a). IL-6 and IL-8 serum concentrations also positively correlated with the concentration of serum arginase I in GC patients (Fig. 4a). In our study, GC patient derived-serum IL-6 and IL-8 both stimulated arginase I production and release in CD45+CD33lowCD11bdim MDSCs. Besides, the gene expression profile of human PMN-MDSCs includes mTOR, MAPK, and other related signaling pathways13,33. We found that the activation of CD45+CD33lowCD11bdim MDSCs were through the PI3K/Akt pathway (Fig. 4d), which has crosstalk with mTOR signaling pathway, and may also induce direct activation or phosphorylation of mTOR.
Our findings indicate a role for the IL-6/IL-8-arginase I axis in CD45+CD33lowCD11bdim MDSCs-mediated CD8+ T cell immunosuppression in GC patients (Fig. 7). GC patient serum-derived IL-6 and IL-8 activates the PI3 kinase signaling pathway in CD45+CD33lowCD11bdim MDSCs to induce arginase I production and release, thereby inhibiting the activity of CD8+ T cells. Overall, our study re-emphasizes the heterogeneous spectrum of MDSC subsets identified in cancer patients, and suggests that the IL-6/IL-8-arginase I axis is a potential therapeutic target for the ablation of MDSC-mediated immunosuppression in GC patients.
Fig. 7. Peripheral blood CD45+CD33lowCD11bdim myeloid cells suppress CD8+ T cells through the IL-6/IL-8-arginase I (ARG I) axis in GC patients.

Circulating IL-6 and IL-8 induces the secretion of arginase I from CD45+CD33lowCD11bdim myeloid cells via the PI3K-AKT signaling pathway
Materials and methods
Patients samples
GC patients who underwent surgical resection without prior radiotherapy or chemotherapy treatment between January 2015 and December 2017 were enrolled at the Southwest Hospital of Army Medical University. GC patients with concurrent autoimmune diseases, infectious diseases, or multiple primary cancers were excluded from the study. Peripheral blood samples from 45 healthy volunteers were used as controls. The clinical stages of tumors were determined according to the TNM classification system of International Union Against Cancer (Edition 7). Individual GC patient characteristics are provided in Supplementary Table 1. This study was reviewed and approved by the Ethics Committee of the Southwest Hospital of Army Medical University, and written informed consent for study enrollment and blood collections obtained from all individuals.
Antibodies and other reagents
Antibodies (Abs) used for flow cytometry were anti-IFN-γ-PE-Cy7 (BD Pharmingen); anti-CD66b-FITC, anti-CD33-PE, anti-CD45-PE-Cy7, anti-CD14-PerCP-Cy5.5, anti-HLA-DR-Alexa Fluor 647 and CD8-PerCP-Cy5.5 (eBioscience); anti-perforin-PE, and anti-granzyme B-APC (Biolegend). Abs used for western blotting were anti-human arginase I (R&D Systems) and horseradish peroxidase (HRP)-conjugated secondary ab (Zhongshan Biotechnology). Abs used for IL-6/IL-8 blockade studies were: anti-human IL-6 (Biolegend) and anti-human IL-8 (R&D Systems). The chemical arginase I inhibitor nor-NOHA (Santa Cruz) was also used. Purified anti-CD3 and anti-CD28 Abs (Biolegend) and recombinant human IL-2 (PeproTech) were also used for T cell studies.
Recombinant human IL-6, IL-8, and arginase I (Biolegend) were used for CD45+CD33lowCD11bdim activation studies. Drug inhibitors studies involved the STAT3 inhibitor FLLL32 (Medco Biosciences), PI3K inhibitor Wortmannin, and MAPK inhibitor SB203580 (Calbiochem).
Flow cytometry
A 100 µl of peripheral blood was incubated with primary ab cocktails for 30 min at 4 °C in the dark. Erythrocytes were lysed with the FACS lysing solution for 10 min in the dark. Samples were centrifuged for 5 min at 300 g and the supernatant was discarded. The cell pellet was re-suspended in PBS and flow cytometric analysis performed on the BD FACS Canto II using FACS Diva version 6.1.2 (BD Biosciences).
For intracellular cell staining (ICS), cells were stimulated for 5 h with the leukocyte activation cocktail with BD Golgiplug (BD Pharmingen). ICS was performed after cell fixation and permeabilization, using the Perm/Wash solution (BD Pharmingen) as per manufacturer’s protocols. Cells were analyzed using the FACSCanto II with FACS Diva version 6.1.2 (BD Biosciences).
Fluorescence-activated cell sorting
A 20 ml of peripheral blood was stained with anti-human CD45, anti-human CD33 and anti-human CD11b Ab, and erythrocytes lysed as per above. The cell pellet was then suspended in PBS at a concentration of 5 × 106 cells/ml. CD45+CD33lowCD11bhigh cells and CD45+CD33lowCD11bdim cells were sorted by FACS using the FACS Aria II (BD Biosciences) and collected in RPMI 1640 containing 10% FCS.
Myeloid and T cell co-cultures
Peripheral blood mononuclear cells (PBMCs) were isolated by density gradient centrifugation with Ficoll-Paque Plus (GE Healthcare). 105 magnetic bead-purified peripheral CD8+ T cells from PBMCs (STEMCELL Technologies) were labeled with carboxyfluoresceinsuccinimidyl ester (CFSE; eBioscience) and cultured in RPMI 1640 containing 10% FCS with recombinant human IL-2 (20 IU/ml), with pre-coated anti-CD3 (2 μg/ml) and anti-CD28 (1 μg/ml) abs on the plate.
CD45+CD33lowCD11bhigh and CD45+CD33lowCD11bdim myeloid cells were sorted as described above and added with CD8+ T cells at a 1:1, 1:2, 1:4, or 1:8 ratio. In some experiments, autologous human serum was added into the co-culture system (50% serum) and/or neutralizing abs against IL-6 (10 μg/ml), IL-8 (10 μg/ml), and arginase I (25 μM) or nor-NOHA (250 μM) also added to the co-culture system. After 5 days incubation, cell pellets resuspended in PBS for flow cytometry.
Western blot analysis
Total proteins from vehicle control or IL-6, IL-8, GC serum, or drug inhibitor pretreated CD45+CD33lowCD11bdim myeloid cells were extracted with Pierce reagent. Western blot assays were performed on 10–15% SDS-PAGE gels using equivalent amounts of cell lysate proteins per sample. Proteins were transfer onto a PVDF membrane and blocked with 3% BSA. Human arginase I was detected with anti-arginase I abs and human phosphorylated AKT-ser473 or total AKT were detected with anti-p-AKT or anti-AKT abs, followed by incubation with HRP-conjugated secondary abs and visualization using the SuperSignal® West Dura Extended Duration Substrate kit (Thermo Fisher Scientific).
RNA extraction and quantitative real-time PCR
Total RNA of CD45+CD33lowCD11bhigh cells and CD45+CD33lowCD11bdim cells were extracted with TRIzol reagent. The RNA quality was measured with NANO2000, and samples were reversed transcript to double-stranded cDNA with PrimeScriptTM RT reagent Kit (TaKaRa). Quantitative Real-time PCR was performed using the primers listed in Supplementary Table 2 on the IQ5 (Bio-Rad) with the Real-time PCR Master Mix (Toyobo). Expression of arginase I was normalized against human GAPDH, and the results were calculated by the ΔΔCt method and expressed as fold change.
ELISA
ELISA was performed on serum or the supernatant of cell cultures. Human IL-6 (eBioscience), IL-8 (eBioscience), IFN-γ (Biolegend) and arginase I (Cusabio Biotech) were detected. The concentration of serum IL-6, IL-8, and arginase I and T cell culture-derived IFN-γ was determined using Enzyme-linked immunosorbent assay (ELISA) kits according to manufacturer’s instructions.
Statistical analysis
Flow cytometry analysis was performed using Flowjo software or BD FACS Diva software. All results are displayedas mean ± SEM, with each data point representing individual patients or patient samples. Statistical analysis performed with the SPSS statistical software (version 13.0). The Student t test or Mann-Whitney U test was used to analyze the difference between the mean of two groups depending on the nature of sample variance. Pearson correlation analysis and linear regression analysis were used to calculate correlations. Overall patient survival was defined as the interval between the date of surgery and date of death or last clinical follow-up, whichever occurred earlier. Cumulative survival time was calculated by the Kaplan-Meier method, with survival measured in months and the log-rank test applied for comparisons between two groups. A two-sided p value of <0.05 was considered statistically significant.
Electronic supplementary material
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2016YFC1302200) and grant of National Natural Science Foundation of China (81670510).
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Edited by T. Kaufmann
Electronic supplementary material
Supplementary Information accompanies this paper at (10.1038/s41419-018-0803-7).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Xiao-lan Guo, Phone: (86)0817-2282052, Email: alan5200@hotmail.com.
Yuan Zhuang, Phone: (86)023-68752315, Email: yuanzhuang1983@yahoo.com.
References
- 1.Torre LA, et al. Global cancer statistics, 2012. CA Cancer J. Clin. 2015;65:87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 2.Vuillefroy de Silly R, Dietrich PY, Walker PR. Hypoxia and antitumor CD8(+) T cells: An incompatible alliance? Oncoimmunology. 2016;5:e1232236. doi: 10.1080/2162402X.2016.1232236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wang Y, et al. Prostatic acid phosphatase as a target molecular in specific immunotherapy for patients with nonprostate adenocarcinoma. J. Immunother. 2005;28:535–541. doi: 10.1097/01.cji.0000175490.26937.22. [DOI] [PubMed] [Google Scholar]
- 4.Young MR, Newby M, Wepsic HT. Hematopoiesis and suppressor bone marrow cells in mice bearing large metastatic Lewis lung carcinoma tumors. Cancer Res. 1987;47:100–105. [PubMed] [Google Scholar]
- 5.Gabrilovich DI, Nagaraj S. Myeloid-derived suppressor cells as regulators of the immune system. Nat. Rev. Immunol. 2009;9:162–174. doi: 10.1038/nri2506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhuang Y, et al. CD8(+) T cells that produce interleukin-17 regulate myeloid-derived suppressor cells and are associated with survival time of patients with gastric cancer. Gastroenterology. 2012;143:951–962 e958. doi: 10.1053/j.gastro.2012.06.010. [DOI] [PubMed] [Google Scholar]
- 7.Ding Y, et al. CD40 controls CXCR5-induced recruitment of myeloid-derived suppressor cells to gastric cancer. Oncotarget. 2015;6:38901–38911. doi: 10.18632/oncotarget.5644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Shen J, Chen X, Wang Z, Zhang G, Chen W. Downregulation of CD40 expression contributes to the accumulation of myeloid-derived suppressor cells in gastric tumors. Oncol. Lett. 2014;8:775–780. doi: 10.3892/ol.2014.2174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.De Sanctis F, et al. MDSCs in cancer: conceiving new prognostic and therapeutic targets. Biochim. Biophys. Acta. 2016;1865:35–48. doi: 10.1016/j.bbcan.2015.08.001. [DOI] [PubMed] [Google Scholar]
- 10.Fujimura T, Mahnke K, Enk AH. Myeloid derived suppressor cells and their role in tolerance induction in cancer. J. Dermatol. Sci. 2010;59:1–6. doi: 10.1016/j.jdermsci.2010.05.001. [DOI] [PubMed] [Google Scholar]
- 11.Bronte V, Zanovello P. Regulation of immune responses by L-arginine metabolism. Nat. Rev. Immunol. 2005;5:641–654. doi: 10.1038/nri1668. [DOI] [PubMed] [Google Scholar]
- 12.Rodriguez PC, et al. Arginase I production in the tumor microenvironment by mature myeloid cells inhibits T-cell receptor expression and antigen-specific T-cell responses. Cancer Res. 2004;64:5839–5849. doi: 10.1158/0008-5472.CAN-04-0465. [DOI] [PubMed] [Google Scholar]
- 13.Veglia F, Perego M, Gabrilovich D. Myeloid-derived suppressor cells coming of age. Nat. Immunol. 2018;19:108–119. doi: 10.1038/s41590-017-0022-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schmok E, et al. Suppressor of cytokine signaling 3 in macrophages prevents exacerbated interleukin-6-dependent arginase-1 activity and early permissiveness to experimental tuberculosis. Front. Immunol. 2017;8:1537. doi: 10.3389/fimmu.2017.01537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Rotondo R, et al. IL-8 induces exocytosis of arginase 1 by neutrophil polymorphonuclears in nonsmall cell lung cancer. Int. J. Cancer. 2009;125:887–893. doi: 10.1002/ijc.24448. [DOI] [PubMed] [Google Scholar]
- 16.Wang L, et al. Increased myeloid-derived suppressor cells in gastric cancer correlate with cancer stage and plasma S100A8/A9 proinflammatory proteins. J. Immunol. 2013;190:794–804. doi: 10.4049/jimmunol.1202088. [DOI] [PubMed] [Google Scholar]
- 17.Gabitass RF, Annels NE, Stocken DD, Pandha HA, Middleton GW. Elevated myeloid-derived suppressor cells in pancreatic, esophageal and gastric cancer are an independent prognostic factor and are associated with significant elevation of the Th2 cytokine interleukin-13. Cancer Immunol. Immunother. 2011;60:1419–1430. doi: 10.1007/s00262-011-1028-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jiang M, et al. Interleukin-6 trans-signaling pathway promotes immunosuppressive myeloid-derived suppressor cells via suppression of suppressor of cytokine signaling 3 in breast cancer. Front. Immunol. 2017;8:1840. doi: 10.3389/fimmu.2017.01840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Noman MZ, et al. PD-L1 is a novel direct target of HIF-1alpha, and its blockade under hypoxia enhanced MDSC-mediated T cell activation. J. Exp. Med. 2014;211:781–790. doi: 10.1084/jem.20131916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Youn JI, Nagaraj S, Collazo M, Gabrilovich DI. Subsets of myeloid-derived suppressor cells in tumor-bearing mice. J. Immunol. 2008;181:5791–5802. doi: 10.4049/jimmunol.181.8.5791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Movahedi K, et al. Identification of discrete tumor-induced myeloid-derived suppressor cell subpopulations with distinct T cell-suppressive activity. Blood. 2008;111:4233–4244. doi: 10.1182/blood-2007-07-099226. [DOI] [PubMed] [Google Scholar]
- 22.Trellakis S, et al. Granulocytic myeloid-derived suppressor cells are cryosensitive and their frequency does not correlate with serum concentrations of colony-stimulating factors in head and neck cancer. Innate. Immun. 2013;19:328–336. doi: 10.1177/1753425912463618. [DOI] [PubMed] [Google Scholar]
- 23.Rodriguez PC, et al. Arginase I-producing myeloid-derived suppressor cells in renal cell carcinoma are a subpopulation of activated granulocytes. Cancer Res. 2009;69:1553–1560. doi: 10.1158/0008-5472.CAN-08-1921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wu H, et al. Arginase-1-dependent promotion of TH17 differentiation and disease progression by MDSCs in systemic lupus erythematosus. Sci. Transl. Med. 2016;8:331ra340. doi: 10.1126/scitranslmed.aae0482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Munder M, et al. Arginase I is constitutively expressed in human granulocytes and participates in fungicidal activity. Blood. 2005;105:2549–2556. doi: 10.1182/blood-2004-07-2521. [DOI] [PubMed] [Google Scholar]
- 26.Bovenschen N, Kummer JA. Orphan granzymes find a home. Immunol. Rev. 2010;235:117–127. doi: 10.1111/j.0105-2896.2010.00889.x. [DOI] [PubMed] [Google Scholar]
- 27.de Poot SA, Bovenschen N. Granzyme M: behind enemy lines. Cell Death Differ. 2014;21:359–368. doi: 10.1038/cdd.2013.189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Desiree A. Anthony DMA, Watt SallyV, Trapani JosephA, Smyth. MarkJ. Functional dissection of the granzyme family: cell death and inflammation. Immunol. Rev. 2010;235:14. doi: 10.1111/j.0105-2896.2010.00907.x. [DOI] [PubMed] [Google Scholar]
- 29.Chowdhury D, Lieberman J. Death by a thousand cuts: granzyme pathways of programmed cell death. Annu. Rev. Immunol. 2008;26:389–420. doi: 10.1146/annurev.immunol.26.021607.090404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.B. Sander SC, Moller E. Interleukin 4 and interferon gamma production in restimulated CD4+and CD8+cells indicates memory type responsiveness. Scand. J. Immunol. 1991;33:11. doi: 10.1111/j.1365-3083.1991.tb01774.x. [DOI] [PubMed] [Google Scholar]
- 31.Xu M, et al. Interactions between interleukin-6 and myeloid-derived suppressor cells drive the chemoresistant phenotype of hepatocellular cancer. Exp. Cell Res. 2017;351:142–149. doi: 10.1016/j.yexcr.2017.01.008. [DOI] [PubMed] [Google Scholar]
- 32.Bank U, et al. Effects of interleukin-6 (IL-6) and transforming growth factor-beta (TGF-beta) on neutrophil elastase release. Inflammation. 1995;19:83–99. doi: 10.1007/BF01534383. [DOI] [PubMed] [Google Scholar]
- 33.Zhang C, et al. The mTOR signal regulates myeloid-derived suppressor cells differentiation and immunosuppressive function in acute kidney injury. Cell Death Dis. 2017;8:e2695. doi: 10.1038/cddis.2017.86. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.