Abstract
MADS-box genes have been demonstrated to participate in a number of processes in tomato development, especially fruit ripening. In this study, we reported a novel MADS-box gene, SlMBP15, which is implicated in fruit ripening. Based on statistical analysis, the ripening time of SlMBP15-silenced tomato was delayed by 2–4 days compared with that of the wild-type (WT). The accumulation of carotenoids and biosynthesis of ethylene in fruits were decreased in SlMBP15-silenced tomato. Genes related to carotenoid and ethylene biosynthesis were greatly repressed. SlMBP15 can interact with RIN, a MADS-box regulator affecting the carotenoid accumulation and ethylene biosynthesis in tomato. In addition, SlMBP15-silenced tomato produced dark green leaves, and its plant height was reduced. The gibberellin (GA) content of transgenic plants was lower than that of the WT and GA biosynthesis genes were repressed. These results demonstrated that SlMBP15 not only positively regulated tomato fruit ripening but also affected the morphogenesis of the vegetative organs.
Keywords: SlMBP15, MADS-box, gibberellin, ripening, ethylene
Introduction
MADS-box genes encode a group of important transcription factors that are widely distributed in plants, animals, and fungi (Messenguy and Dubois, 2003). In 1990, the studies on the MADS-box related to its essential role in floral organ development were published (Sommer et al., 1990; Yanofsky et al., 1990). Subsequently, the famous ABC model for floral organ construction was proposed based on extensive genetic and molecular studies (Coen and Meyerowitz, 1991). Improvements in this research introduced new regulators and extended the ABC model into the ABCDE model (Colombo et al., 1995; Pelaz et al., 2001). In Arabidopsis, the A-class genes include APETALA1 (AP1) and APETALA2 (AP2). The B-class genes contain APETALA3 (AP3) and PISTILLATA (PI). The C- and D- class genes all belong to the AGAMOUS (AG) clade, while the E-class genes come from the SEPALLATA (SEP) clade. All these genes belong to the MADS-box except for AP2 and its homologs (Theissen et al., 2000). So far, in tomato, the function of most of the genes contained in the ABCDE model has been well described. In addition, some other MADS-box genes that influence floral organ development have been identified. For example, our former study showed that the MADS-box gene SlAGL6 affected the development of the sepal and petal (Yu et al., 2017). Overexpression of SlFYFL delayed sepal senescence, (Xie et al., 2014) while overexpressing the AGL15 clade gene SlMBP11 delayed perianth senescence in tomato (Guo et al., 2017). Furthermore, TM8 overexpression in tomato plants generated anomalous stamens with poorly viable pollen (Daminato et al., 2014). A recent study suggested the tomato MADS-box gene SlMBP21 is a novel factor involved in organ size control (Li et al., 2017).
In addition to floral organ identity determination, MADS-box genes also take part in inflorescence development in tomato. The MADS-box gene JOINTLESS (J) suppresses sympodial meristem identity in inflorescence meristems (Szymkowiak and Irish, 2006). The AP1/FUL gene MACROCALYX (MC)-regulates inflorescence determinacy (Vrebalov et al., 2002). In addition, a lack of MC produced an indeterminate inflorescence in tomato (Nakano et al., 2012). A recent study revealed that MC exerts the function of regulating inflorescence development through an interaction with SFT and J (Yuste-Lisbona et al., 2016). Additionally, current research revealed three novel MADS-box genes that influenced inflorescence architecture (Soyk et al., 2017).
Tomato MADS-box genes are also involved in plant vegetative growth. Previous research showed that overexpression of SlFYFL delayed leaf senescence (Xie et al., 2014). The AP1/FUL transcription factor gene MBP20 is involved in tomato leaf development, and overexpression of a dominant-negative form of MBP20 led to leaf simplification and partly suppressed the increased leaf complexity of plants (Burko et al., 2013). Apart from tomato leaf development, MADS-box genes are also considered to affect tomato plant vegetative growth in other ways. For example, tomato plants with increased SlMBP11 levels displayed reduced plant height, leaf size and internode length; highly branched growth from each leaf axil; and an increased number of nodes and leaves (Guo et al., 2017). In tomato, there are some studies on the regulation of abscission zone (AZ) formation by MADS-box family members. The deletion of the MADS-box gene JOINTLESS leads to the failure of activation of pedicel AZ development in tomato plants (Mao et al., 2000). Similarly, the MC-suppressed transformants produced by the introduction of an MC antisense construct displayed an absence of the AZ structure or a barely visible, knuckle-like structure. Further studies revealed that MC interacts physically with JOINTLESS and that the resulting heterodimer acquires a specific DNA-binding activity (Nakano et al., 2012). Subsequently, the SEP clade gene SlMBP21 was identified as an additional transcription activator factor for the development of AZ in tomato and SlMBP21 may form higher-order protein complexes with JOINTLESS and MC (Liu et al., 2014).
It has been proved that MADS-box transcription factors are involved in abiotic stress response in some plant species such as Arabidopsis (Lee et al., 2005), rice (Lee et al., 2008, 2011) and wheat (Tardif et al., 2007). However, to date, only SlMBP11 and SlMBP8 have been reported to function in abiotic stress response. SlMBP11 functions as a positive regulator in response to salt stress (Guo et al., 2016). Tomato plants with repressed FLC/MAF clade gene SlMBP8 became more tolerant to drought and salt stress (Yin et al., 2017b).
In addition to the functions we described above, tomato MADS-box genes mediate the following processes: cuticle development (Gimenez et al., 2015; Garceau et al., 2017), parthenocarpy (Ampomah-Dwamena et al., 2002; Klap et al., 2016), fruit ripening and so on. Among these processes, the most notable role for MADS-box genes is regulating fruit ripening. In 2002, a tomato ripening-inhibitor (rin) mutant with fruits that failed to ripen and enlarged sepals was reported (Vrebalov et al., 2002). Gene repression and mutant complementation demonstrated that RIN regulates tomato fruit ripening (Vrebalov et al., 2002). However, a RIN-knockout mutation did not repress the initiation of ripening, and the fruits of the mutant showed moderate red coloring (Ito et al., 2017). Recent research re-evaluated the rin mutant and declared that RIN is not required for the initiation of ripening and the rin is not a null mutation but a gain-of-function mutation that produces a protein that actively represses fruit ripening (Ito et al., 2017). In addition to RIN, many other MADS-box genes were confirmed to participate in the regulation of tomato fruit ripening. Tomato plants with reduced TAGL1 mRNA produced a fruit-maturation-inhibition phenotype with reduced carotenoids and ethylene (Vrebalov et al., 2009). Moreover, two tomato FRUITFULL (FUL) clade genes also play roles in fruit ripening. Dual suppression of FUL1 (TDR4) and FUL2 (MBP7) substantially inhibited fruit ripening by blocking ethylene biosynthesis and decreasing carotenoid accumulation (Wang et al., 2014). These two genes also regulate fruit ripening in an ethylene-independent way (Bemer et al., 2012). In our laboratory, some MADS-box genes regulating fruit ripening have been investigated to explore the mechanism of tomato ripening. In tomato, SlMADS1 acts as a negative regulator of fruit ripening and regulates the expression of carotenoid and ethylene biosynthesis genes (Dong et al., 2013). Overexpression of the tomato MADS-box gene SlFYFL delayed fruit ripening by 3–5 days (Xie et al., 2014), and suppression of SlMBP8 accelerated fruit ripening and induced the expression of carotenoid- and ethylene- biosynthesis genes (Yin et al., 2017a).
In previous research, we revealed the function of SlMBP8, one of the FLC/MAF clade members. However, the roles of the other members, SlMBP15 and SlMBP25, in tomato are still unclear. Previous studies showed that SlMBP15 and SlMBP25 are expressed in both vegetative and floral tissues (Hileman et al., 2006), which indicates that they probably play important roles in various developmental processes. Here, we characterized SlMBP15 (accession number: NCBI: XM_004252663 / SGN: Solyc12g087810.2; Supplementary Table S1), and the expression pattern analysis showed that SlMBP15 accumulation in tomato fruits of different stages was greater than that in other organs, while in fruit-ripening mutants, SlMBP15 was repressed on the whole. Therefore, we hypothesized that SlMBP15 may modulate tomato fruit ripening. To confirm this, we generated transgenic tomato with SlMBP15 repressed by RNAi (RNA interference). We show herein that the altered expression of SlMBP15 delayed fruit ripening providing strong evidence for our hypothesis. In addition, the morphology of the vegetative organs of the transgenic plants was different from that of the wild-type (WT).
Materials and Methods
Plant Materials and Growth Conditions
Tomato plants (Solanum lycopersicon Mill. cv. Ailsa Craig AC++) were used as the WT in this study. Transgenic and the wild-type plants were grown in the greenhouse conditions (16 h-day/8 h-night cycle, 25/18°C day/night temperature, 80% humidity, and 250 μmolm-2 s-1 light intensity). The experiments in this study were also repeated in our experimental farm in the natural condition. Two generations of the transgenic tomato plants were used in this study. Plants of the first generation (T0) came from the tissue culture, and the second generation (T1) from the seeds. For gene expression analysis, flowers were tagged at anthesis and the ripening stages of tomato fruits were divided according to the days post anthesis (dpa) and fruit color. In the wild-type, we defined the fruit of 20 dpa as IMG (Immature green) and 35 dpa as MG (Mature Green), which was characterized as being green and shiny with no obvious color change. The fruit with a color change from green to yellow was defined as B (Breaker) while B + 4 and B + 7 were fruits of 4 days after breaker and 7 days after breaker, respectively. Tomato pericarp of 0.5 cm wide along the equator was used as fruit sample. All the harvested samples were immediately frozen in liquid nitrogen and stored at -80°C. Three individual plants for each line were used for biological replicates in all the treatments.
Isolation and Sequence Analysis of SlMBP15
Total RNA of all plant tissues was extracted from three biological replicates using the RNAiso Plus (Takara) according to the manufacturer’s instructions. Then 1 μg RNA was used to synthesize the first strand cDNA by reverse transcription polymerase chain reaction (M-MLV reverse transcriptase, Takara, China) with tailed oligo d(T)18 primer (5′ GCT GTC AAC GAT ACG CTA CGT AAC GGC ATG ACA GTG TTT TTT TTT TTT TTT TTT 3′). To clone the full-length of SlMBP15 gene, the PCR was employed using primers SlMBP15-F (5′ GAA ATC CAG GTG GCA GAG CAC 3′) and SlMBP15-R (5′GCT GTC AAC GAT ACG CTA CGT AAC G 3′), and 1–2 μl cDNA was used as PCR template. The PCR reaction was performed using high fidelity Prime STARTMHS DNA polymerase (Takara) following the procedure below: 94°C for 5 min followed by 35 cycles of 30 s at 94°C, 30 s at 56°C and 30 s at 72°C, and a final extension at 72°C for 10 min. The amplified product was tailed using DNA A-Tailing kit (Takara) and linked to pMD18-T vector (Takara). Positive colonies were picked out via Escherichia coli JM109 transformation and confirmed by sequencing (BGI, China). The multiple sequence alignment of SlMBP15 with other MADS-box proteins was performed using DNAMAN 5.2.2 software. All accession numbers of proteins used in multiple sequence alignment and phylogenetic analysis were listed in Supplementary Table S2.
Construction of the SlMBP15 RNAi Vector and Plant Transformation
In order to repress the expression of the SlMBP15 gene, a RNAi construct was assembled using the pBIN19 vector (Fray et al., 1994; Frisch et al., 1995). Firstly, the 320 bp SlMBP15 specific DNA fragment used in the hairpin was amplified using the primers SlMBP15-F (5′ GAA ATC CAG GTG GCA GAG CAC 3′) and SlMBP15-R (5′ GGA AAG TCA GCG AAA TCC GTA 3′) tailed with Kpn I, Cla I and Xho I, Xba I restriction cutting site at the 5′ end, respectively. Secondly, the purified products were digested with Xho I/Kpn I and inserted into pHANNIBAL vector (Wesley et al., 2001) plasmid at Xho I/Kpn I restriction site in the sense orientation, while the products tailed with Xba I/Cla I restriction enzyme in the antisense orientation. Thirdly, the double-stranded RNA expression unit, containing the cauliflower mosaic virus (CaMV) 35S promoter, SlMBP15 fragment in the sense orientation, PDK intron, SlMBP15 fragment in the antisense orientation and the OCS terminator was digested by SacI/XbaI and linked into plant binary vector pBIN19. At last, the generated binary plasmid was used for the transformation to tomato cotyledon explants by Agrobacterium tumefaciens (strain LBA4404) (Chen et al., 2004). The tissue culture plants were selected by kanamycin (80 mg l-1) and then analyzed by PCR with the primers NPTII-F (5′GAC AAT CGG CTG CTC TGA 3′) and NPTII-R (5′AAC TCC AGC ATG AGA TCC 3′) to determine the presence of T-DNA.
Quantitative Real-Time PCR (qPCR) Analysis
Quantitative real-time PCR was performed using the SYBR® Premix Ex Taq II kit (Takara, China) in a 10 μl total reaction volume (5.0 μl 2 × SYBR Premix Ex Taq, 1.0 μl primers, 1.0 μl cDNA, 3.0 μl ddH2O). All reactions were performed using a two-step method: 95°C for 30 s, followed by 40 cycles of 95°C for 5 s, and 60°C for 30 s. To remove the effect of genomic DNA and the template from environment, NTC (no template control) and NRT (no reverse transcription control) were performed. Additionally, three replications for each sample were used and the standard curves were run simultaneously. Tomato CAC gene was selected as internal standard (Exposito-Rodriguez et al., 2008). The primers SlMBP15-Q-F and SlMBP15-Q-R (Supplementary Table S3) were used to determine the expression levels of SlMBP15 in wild-type, Nr (never ripe), rin (ripening inhibitor) and transgenic lines. The relative gene expression was calculated using the 2-ΔΔCT method (Livak and Schmittgen, 2001). All primers used for qPCR were listed in Supplementary Table S3.
Measurement of Carotenoid Contents
Tomato carotenoid was extracted from the pericarp using a modified protocol from a previous report (Forth and Pyke, 2006). 1.0 g sample was milled into powder with liquid nitrogen. Then 10 ml of 60:40 (v/v) hexane-acetone was added and total carotenoids of the wild-type and RNAi lines fruits were extracted. The extract was centrifuged at 4000 g for 5 min and the absorbance of supernatant was measured at 450 nm. The carotenoid content was calculated with the following equations: total carotenoids (mg ml-1) = (OD450)/0.25. Three independent experiments were performed for each sample.
Measurement of GA3 and Fruit Ethylene Release
The stem from 4-week-old tomato plants was collected and frozen in liquid nitrogen. GA3 was extracted and purified using GA3 kit (GA-4-Y Comin Biotechnology Co., Ltd., China) according to the manufacturer’s instructions. Then the concentration of endogenous bioactive gibberellin (GA3) were measured by HPLC (High Performance Liquid Chromatography).
The ethylene released by fruit was measured at B, B + 4 and B + 7 stages. Fruits were placed in airtight jars for 24 h at 28°C. 1 mL gas sample of the headspace was injected into a Hewlett-Packard 5890 series II gas chromatograph equipped with a flame ionization detector. The samples were compared with reagent grade ethylene standards of known concentration and normalized for fruit weight (Chung et al., 2010). Three biological replicates and three technical replications were adopted for the data analysis.
Yeast Two-Hybrid Assay
The yeast two-hybrid assay was performed using the MATCHMAKER GAL4 Two-Hybrid System III according to the manufacturer’s protocol (Clontech). The ORF of SlMBP15 was amplified by PCR with the primer SlMBP15(Y)-F (5′ GGC GAA TTC GGG CTT TTA ATC GGC GAA AAA 3′) and SlMBP15(Y)-R (5′ CGC GGA TCC CGA CGA ATA CGA CGA TAA TCA 3′). The PCR product was digested using EcoRI and SalI, and it was cloned into the pGBKT7 bait vector by the EcoRI/SalI site. The recombined vector was named pGBKT7-SlMBP15 and transformed into yeast strain Y2HGold. The Y2HGold with bait was plated on SDO (SD medium -Trp) and TDO (SD medium -Trp, -His, and -adenine) to test the self-activation of pGBKT7-SlMBP15. In parallel, the ORF of SlMADS-RIN was amplified by primers SlRIN(Y)-F (5′CCG GAA TTC ATG GGT AGA GGG AAA GTA GA3′) and SlRIN(Y)-R (5′CGC GGA TCC TCA TAG ATG TTT ATT CAT3′). The product was cloned into the pGADT7 vector and transformed into yeast strain Y187. Subsequently, the Y2HGold with bait and the Y187 with prey were cultured together in 2 × YPDA medium for 24 h. Then, the culture was grown on DDO (SD medium lacking Trp and Leu) to screen the diploids containing prey and bait. After 2–5 days, fresh diploid cells were plated on QDO/X [SD medium lacking Trp, Leu, His, and adenine with X-a-Gal (5-bromo-4-chloro-3-indolyl-a-D-galactopyranoside)] to test if SlMBP15 interacts with SlMADS-RIN. The colonies developing blue color were picked out and cultivated again on plates with QDO/X medium. The plates were incubated at 30°C for 3–7 days. The empty prey and bait vectors were used as controls to remove false positive, respectively. At the same time, the positive control and negative control were cultured. The assay was repeated at least three times with fresh transformants.
Statistical Analysis
The data in this study was presented as mean ± standard deviation. The significant difference between wild-type and transgenic lines was analyzed by Student’s t-test (P < 0.05). The measurement values came from the means of at least three biological replicates.
Results
Sequence and Expression Analysis of SlMBP15
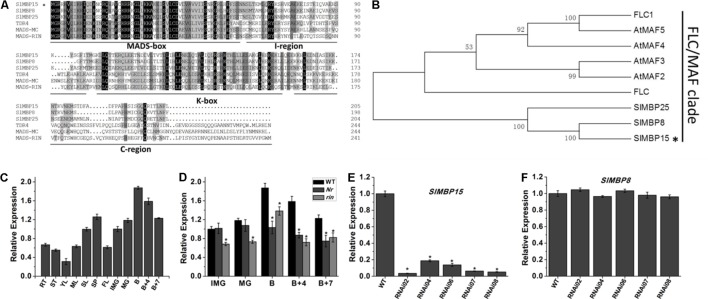
We conducted the alignment with the gene sequence of SlMBP15 by Basic Local Alignment Search Tool (BLAST)1. The result showed that there are three transcript variants (Supplementary Table S4). Even with some nucleotide difference, the transcript variant X3 and X4 translate the same protein. However, except for SlMBP15, the two proteins encoded by three transcript variants do not contain complete MADS-box domain (Supplementary Figure S1). We also carried out sequence alignment between SlMBP15 and other MADS-box proteins belonging to MIKCC type using DNAMAN 5.2.2 software. The result displayed that SlMBP15 contained four typical domains (Figure 1A) which are shared by other MIKCC proteins and the MADS-box domain was highly conserved. In addition, the phylogenic analysis showed that SlMBP15 belongs to FLC/MAF clade (Figure 1B).
FIGURE 1.
Sequence, phylogenic and expression analysis of SlMBP15. (A) Multiple sequence alignment of SlMBP15 with other MADS-proteins. (B) Phylogenic analysis of SlMBP15 and other MADS-box proteins. The phylogenic tree was constructed by the neighbor-joining method, bootstrap analysis of 1,000 replicates. SlMBP15 is marked with asterisk. Accession numbers of other proteins contained in phylogenic analysis are listed in Supplementary Table S3. (C) Expression profile of SlMBP15 in WT. RT, root; ST, stem; YL, young leaf; ML, mature leaf; SL, senescent leaf; SP, sepal; FL, flower; IMG, immature green fruit; MG, mature green fruit; B, breaker fruit; B + 4, 4 days after breaker; B + 7, 7 days after breaker. (D) Expression pattern of SlMBP15 in fruits of wild-type Nr and rin mutants. (E) Relative expression of SlMBP15 in wild-type and transgenic tomato plants. (F) Relative expression of SlMBP8 in wild-type and transgenic lines. Asterisks indicate a significant difference (P < 0.05).
In order to explore the expression profile of SlMBP15, we designed specific qPCR primers and detected the expression of SlMBP15 in various tissues of wild-type tomato. Furthermore, the transcription of SlMBP15 was also investigated in fruits at series developmental stages in wild-type, Nr and rin mutants. In wild-type tomato, the constitutive expression of SlMBP15 indicated it may participate in the developmental processes in multiple tissues and organs of tomato (Figure 1C). In the wild-type tomato fruits, the expression of SlMBP15 rose gradually along with the fruit development process, and the highest accumulation appeared at B stage. After that, SlMBP15 was declined. The expression of SlMBP15 decreased with the maturity of tomato fruit, which implied that SlMBP15 may play important roles in fruit ripening. Therefore, we further compared the expression of SlMBP15 in Nr and rin mutants with the wild-type. The transcript level of SlMBP15 in Nr and rin was lower in fruits at almost all stages than that in the wild-type (Figure 1D), which suggested that SlMBP15 may be regulated by RIN and Nr, and probably function downstream of these genes.
Down-Regulation of SlMBP15 Generated Dark Green Leaves in Tomato Plants
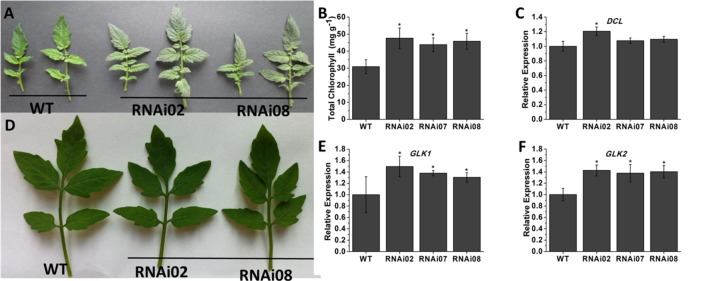
To explore the biological functions of SlMBP15 in tomato, we generated transgenic tomato lines with the SlMBP15 gene silenced. In total, we obtained 5 transgenic lines in which SlMBP15 was well repressed, and the lines RNAi02, RNAi07 and RNAi08 were used for subsequent experiments because they showed the lowest expression levels of SlMBP15 (Figure 1E). The expression of SlMBP8, the gene sequence shares the highest homology with SlMBP15, was not affected in the transgenic lines (Figure 1F). According to our observations, the compound leaves of transgenic plants were darker green in color than those of the wild-type both in field (Figure 2A) and in pots (Figure 2D). Then, we examined the total chlorophyll content, and the results demonstrated that the leaves of the three transgenic lines contained more chlorophyll than those of the wild-type, which was consistent with the previous observation (Figure 2B). Furthermore, we detected the transcript level of genes involved in the pathway of chlorophyll biosynthesis. As a result, a chloroplast development-related gene, DCL (DEFECTIVE CHLOROPLASTS AND LEAVES) (Keddie et al., 1996), and two photosystem-related genes GLK1 (Golden 2-like 1) and GLK2 (Powell et al., 2012), were examined. GLK1 and GLK2 determine the capacity for light-stimulated photosynthesis by controlling chloroplast formation (Powell et al., 2012). In tomato, co-suppression of GLK1 resulted in pale leaves with reduced chlorophyll whereas co-suppression of GLK2 resulted in fruits without a green shoulder (Nguyen et al., 2014). The tomato dcl mutation produces albino areas with abnormal palisade cells; moreover, the chloroplasts from proplastids are blocked (Keddie et al., 1996). qPCR results showed that GLK1 and GLK2 were significantly upregulated in the three transgenic lines, (Figures 2E,F) while DCL showed little change (Figure 2C).
FIGURE 2.
Phenotype of leaf color in SlMBP15-silenced lines. (A) The leaves detached from tomato plants grown in field. (B) The leaves detached from tomato plants grown in pots. (C) Total chlorophyll content in leaves. (D–F) Expression of genes related to chlorophyll biosynthesis. Each value represents the mean ± SE of three biological replicates. Asterisks indicate a significant difference (P < 0.05).
Silencing SlMBP15 Inhibited Plant Growth and GA Biosynthesis
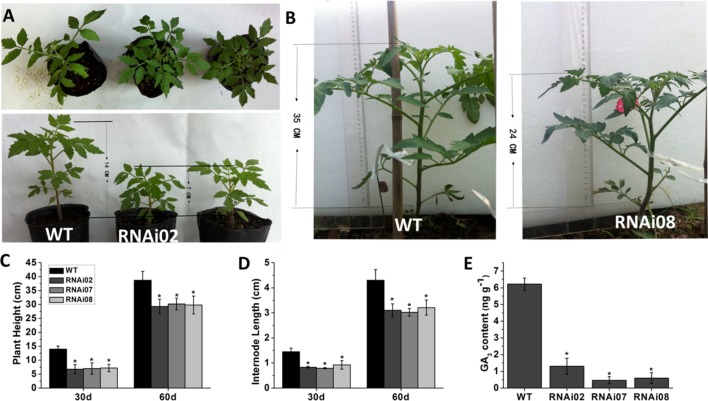
In addition to having a different leaf color, the transgenic plants were shorter than the wild-type plants. We also measured the plant height and internode length of each transgenic line and the wild-type. On the 30th day from germination, the transgenic plants were 7 cm shorter than the wild-type (Figures 3A,C). A month later, the transgenic plants still remained 11 cm shorter than the wild-type plants (Figures 3B,C). The internode length of the transgenic lines was also shorter than that of the wild-type plants (Figure 3D).
FIGURE 3.
Phenotype of wild-type and transgenic plant stems. (A) Tomato plants of 30-day-old. (B) Tomato plants of 60-day-old. (C) Plant height of plants at 30 and 60-day-old, respectively. (D) The internode length of plants at 30 and 60-day-old, respectively. (E) The GA3 content of wild-type and transgenic lines.
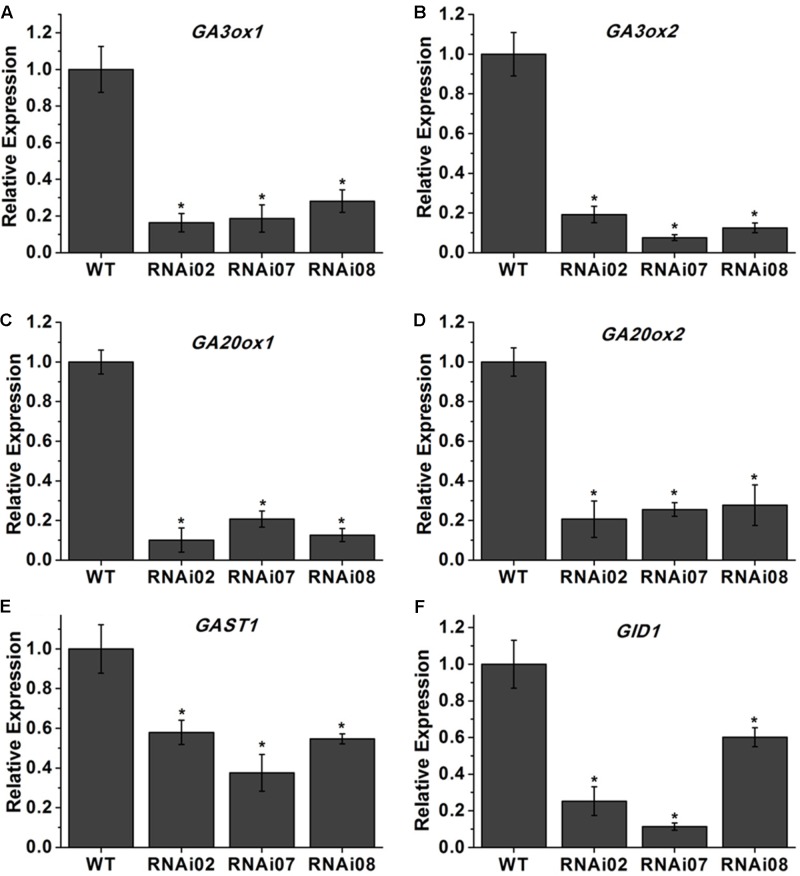
Previous research showed that GAs play an important role in stem elongation (Olszewski et al., 2002). Here, we measured GA3 content, and the result showed that the GA3 level in transgenic lines was significantly lower than that in the wild-type (Figure 3E). We further detected some genes known to be involved in GA biosynthesis. The GA20oxs act as important biosynthesis enzymes determining the GA concentration in many species, while the GA3oxs function in the final step of producing bioactive GAs (GA1, GA3, GA4, and GA7) (Yamaguchi, 2008). The transcription of GAST1 (tomato gibberellin-stimulated transcripts 1) can be dramatically induced by GA (Shi and Olszewski, 1998). GID2 (GIBBERELLIN INSENSITIVE DWARF1) is a receptor of GAs that is important for the perception and transduction of GA signaling (Ueguchi-Tanaka et al., 2007; Murase et al., 2008). In this study, the transcriptional level of all genes mentioned above was reduced (Figure 4). These results indicated that SlMBP15 regulated the biosynthesis and response of GAs.
FIGURE 4.
Relative expression of genes involved in GA biosynthesis. (A–F) The expression levels of GA3ox1, GA3ox1, GA20ox1, GA20ox2, GAST1 and GID1 in wild-type and transgenic lines. Each value represents the mean ± SE of three biological replicates. Asterisks indicate a significant difference (P < 0.05).
Repression of SlMBP15 Delayed Tomato Fruit Ripening
The downregulation of SlMBP15 not only led to the difference in plant morphology at the vegetative growth stage but also inhibited tomato fruit ripening. To study the impact of SlMBP15 on tomato fruit ripening, the number of days from anthesis to ripening was recorded. Statistical analysis of the data showed that the ripening time of the transgenic lines was delayed by 2–4 days compared with that of the wild-type (Table 1).
Table 1.
Days from anthesis to breaker stage for wild-type and SlMBP15-silenced lines.
| Tomato lines | Days |
|---|---|
| Wild-type | 38.0 ± 0.50 |
| RNAi02 | 42.6 ± 0.56∗ |
| RNAi07 | 41.2 ± 0.38∗ |
| RNAi08 | 40.4 ± 0.43∗ |
Values represent means ± SD in days for at least 20 fruits of each line. Asterisks indicate a significant difference (P < 0.05).
Inhibition of SlMBP15 Down-Regulated the Carotenoid Content
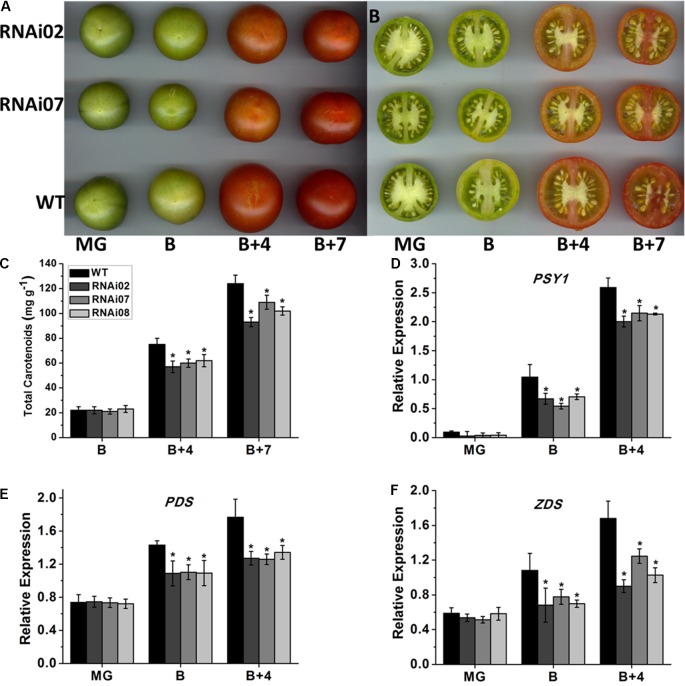
Previous studies have demonstrated that tomato fruit ripening is accompanied by color alteration due to the continuous accumulation of carotenoids (Fraser et al., 1994). According to our observations, the pericarp color of SlMBP15-silenced lines at the B + 4 and B + 7 stages was lighter than that of the wild-type (Figures 5A,B). The test of carotenoid content in tomato pericarps demonstrated that, at the B + 4 and B + 7 stages, the tomato fruits of transgenic lines accumulated fewer carotenoids than those of the wild-type, which was in agreement with the lighter-color phenotype of transgenic tomato fruits (Figure 5C).
FIGURE 5.
The accumulation of carotenoid in wild-type and transgenic lines. (A,B) The fruits of wild-type and transgenic lines at different stages. (C) The carotenoid content in fruits. (D–F) Relative expression of genes participating in carotenoid biosynthesis. Each value represents the mean ± SE of three biological replicates. Asterisks indicate a significant difference (P < 0.05).
It is known that the biosynthesis of carotenoids is regulated by a group of genes, among which, PSY1 (phytoene synthase 1), PDS (phytoene desaturase) and ZDS (ζ-carotene desaturase) are important regulators (Hirschberg, 2001). To further confirm the underlying causes that led to the reduced accumulation of carotenoids in SlMBP15-silenced tomato fruits, the three genes were detected in tomato pericarps. qPCR analysis showed a remarkably lower expression level of PSY1, PDS and ZDS in the transgenic lines than in the wild-type at the B and B + 4 stages (Figures 5D–F). These results revealed that silencing SlMBP15 inhibited the biosynthesis of carotenoids.
Deficiency of SlMBP15 Led to a Reduction in Ethylene Content
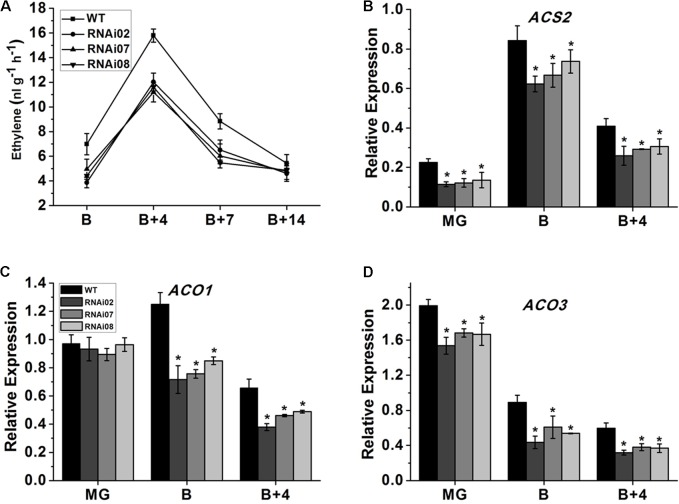
Ethylene is considered to be involved in the ripening of climacteric fruit (Alexander and Grierson, 2002) and regulates the accumulation of carotenoids in tomato fruit (Maunders et al., 1987). Therefore, we measured the ethylene production of wild-type and SlMBP15-silenced tomato fruits at different stages. As shown in Figure 6A, the repression of SlMBP15 decreased the ethylene production at the B, B + 4, and B + 7 stages.
FIGURE 6.
The ethylene content measurement and biosynthesis related gene expression. (A) The ethylene content produced by fruits of wild-type and transgenic lines. (B–D) Relative transcript level of genes regulating ethylene biosynthesis. Each value represents the mean ± SE of three biological replicates. Asterisks indicate a significant difference (P < 0.05).
Subsequently, we carried out qPCR to further investigate the underlying reasons for the alteration in ethylene biosynthesis. The ethylene biosynthesis-related genes ACS (1-aminocyclopropane-1-carboxylatesynthase)2 (Adams and Yang, 1979), ACO (1-aminocyclopropane-1-carboxylate oxidase)1 and ACO3 (Alexander and Grierson, 2002) were detected in the pericarps of the wild-type and each transgenic line at the MG, B and B + 4 stages. As shown in Figures 6B–D the mRNA levels of ACS2 and ACO3 were distinctly lower in all stages we examined while that of ACO1 was downregulated only in the B and B + 4 stages. In sum, SlMBP15 downregulation repressed ethylene biosynthesis in tomato fruit.
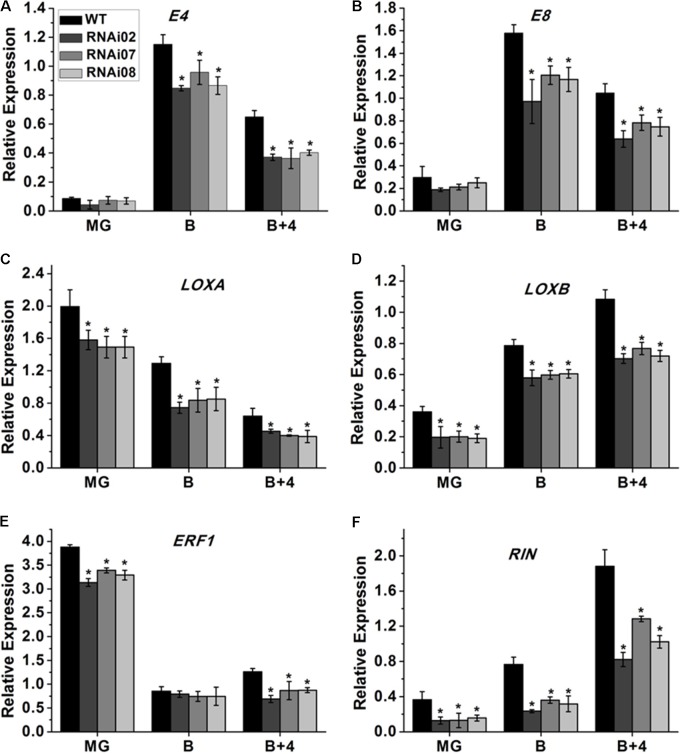
SlMBP15 Influenced the Expression of Ripening Associated Genes
Tomato ripening is a complicated process that involves a series of changes in physiology and biochemistry. A large number of genes are involved alone or together to regulate the process. Some genes have been identified to be essential for tomato fruit ripening. Here, we also performed qPCR to study the expression of genes in the pericarp that modulate tomato ripening. The qPCR analysis showed that the transcription levels of E4, E8 and ERF1, which also serve as ethylene-responsive genes (Lincoln et al., 1987; Lincoln and Fischer, 1988; Penarrubia et al., 1992), were all downregulated in the transgenic lines (Figures 7A,B,E). Additionally, the fruit-specific lipoxygenase genes LOXA and LOXB (Griffiths et al., 1999) were also inhibited after silencing SlMBP15 (Figures 7C,D). RIN is a MADS-box regulator that affects the color of tomato fruit during ripening (Ito et al., 2017). In the SlMBP15-silenced transgenic lines, the transcript level of RIN was also reduced (Figure 7F). These results indicated that repression of SlMBP15 has an impact on the expression of ripening associated genes.
FIGURE 7.
Detection of genes regulating tomato fruit ripening. (A–F) Relative expression of E4, E8, LOXA, LOXB, ERF1, and RIN in RNAi lines and the wild type. Each value represents the mean ± SE of three biological replicates. Asterisks indicate a significant difference (P < 0.05).
SlMBP15 Interacted With RIN in Yeast
RIN is a key regulator of the ripening-gene expression network, with 100s of gene targets controlling changes in color, flavor, texture and taste during tomato fruit ripening (Fujisawa et al., 2013). Previous research suggested that MADS-box proteins usually function by interacting with each other to form higher-order complexes (Leseberg et al., 2008). In our study, the expression of the MADS-box gene RIN was inhibited in SlMBP15-silenced tomato. Therefore, we considered whether SlMBP15 interacts with RIN. To confirm this hypothesis, we performed a yeast two-hybrid assay. Figure 8 shows that the yeast grown on DDO/X medium turned blue, as did the positive control, which indicates that SlMBP15 interacts with SlMADS-RIN in yeast.
FIGURE 8.
Yeast two-hybrid assay for SlMBP15 and RIN. SDO, SD medium lacking Trp; TDO, SD medium lacking Trp, His, and Ade; QDO, SD medium lacking Trp, Leu, His, and Ade; QDO/X, SD medium lacking Trp, Leu, His, and Ade with X-a-Gal. Numbered wedges are as follows: 1, pGBKT7-MBP15 and pGADT7-RIN (interaction of SlMBP15 and SlMADS-RIN); 2, pGBKT7-53 and pGADT7-T (positive control); 3, pGBKT7-Lam and pGADT7-T (negative control); 4, pGBKT7-MBP15 (autoactivation assay); 5, pGBKT7 and pGADT7-RIN (empty bait vector); 6, pGBKT7-MBP15 and pGADT7 (empty prey vector).
Discussion
Since the 1990s, many studies on the MADS-box have been carried out, and many MADS-box genes have been isolated and characterized. Until now, MADS-box genes have been proved to participate in many events during the plant life cycle. However, as a transcription factor family with more than 100 members (investigated by our laboratory, not published), only a small number of MADS-box members have been well studied. Here, we elucidated the function of a FLC/MAF clade gene, SlMBP15. The expression profile predicted by Multi-Plant eFP Browser 2.02 showed that the expression of SlMBP15 in fruits was higher than in other organs and that the expression in fruits after the B stages was higher than that of IMG and MG, which is consistent with our results (Supplementary Table S5). Recently, Shinozaki et al. (2018) showed the expression pattern of genes in individual cell and tissue types. It seems that the expression of SlMBP15 has no change in the total pericarp during fruit development and ripening, which differed from our results (Supplementary Figure S2). However, the expression patterns obtain in two different ways were all detected in cv. Heinz while our expression pattern and wild-type tomato plant materials were from cv. Ailsa Craig AC++. This probably caused the difference in the expression of SlMBP15. The expression profile in the wild-type and ripening-inhibited mutants implied that SlMBP15 may play a direct or indirect role in tomato fruit ripening (Figures 1C,D). Therefore, the SlMBP15-silenced transgenic tomato was generated for further functional study.
SlMBP15 Affects Plant Vegetative Growth in Tomato Plants
Previous research showed that MADS-box genes are associated with morphogenesis of almost all organs and throughout the entire plant life cycle (Guo et al., 2016). In our study, the SlMBP15-silenced plants were shorter than the wild-type plants, and the phenotype was further confirmed by plant height measurements (Figures 3A–D). It has been reported that overexpression of SlMBP11 can also lead to reduced plant height due to a reduction in GA biosynthesis (Guo et al., 2017). Hence, we supposed that the variation in GA level may result in the dwarf phenotype in SlMBP15-silenced lines. To test this hypothesis, we examined the GA content in the wild-type and transgenic lines, and the result confirmed our hypothesis (Figure 3E). Moreover, we studied the GA biosynthesis and transduction-related genes to further explore the molecular mechanism. The qPCR result showed that GA-biosynthesis genes (GA3ox1, GA3ox2, GA20ox1 and GA20ox2), a GA-induced gene (GAST1) and a GA-receptor gene (GID2) were all inhibited in the transgenic lines compared with the wild-type (Figure 4). These results indicated that SlMBP15 influences the plant height and internode length in tomato by modulating GA biosynthesis and transduction. In addition, the leaves of SlMBP15-silenced transgenic lines were darker green than those of the wild-type, and the accumulation of chlorophyll and the transcription of GLK1 and GLK2 were elevated, which demonstrated that SlMBP15 probably influenced the accumulation of chlorophyll in the leaf by regulating the chloroplast formation controlled by GLK1 and GLK2.
In the introduction, we mentioned that some tomato MADS-box genes have been considered to influence different aspects of plant vegetative growth, such as leaf development, plant height, lateral organ development and AZ formation. Here, we identified SlMBP15 as a novel regulator of tomato vegetative growth via its influence on plant height and leaf color. Tomato plants overexpressing SlMBP11 and with silenced SlMBP15 showed the same phenotype of plant height and similar gene expression trends for GA-biosynthesis genes. These results indicate that the MADS-box gene does have an effect on the development of the tomato stem. In addition, in this regulatory process, some members function positively, while some function negatively. They cooperate to keep a balance during the development process.
SlMBP15 Acts as a Promoter of Tomato Fruit Ripening
It has been proved that a large amount of ethylene is required at the beginning of fruit ripening (Bleecker and Kende, 2000) and that the accumulation of carotenoids is a sign of tomato fruit ripening. In previous studies, several MADS-box genes that regulated fruit ripening were proved to affect the carotenoid accumulation and ethylene production. For example, SlMADS1 functions as an inhibitor in tomato fruit ripening, and repression of this gene increased ethylene production and carotenoid accumulation (Dong et al., 2013). However, the pattern is the opposite in TAGL1-reduced tomato, in which carotenoid and ethylene content were both decreased (Vrebalov et al., 2009), which indicates that TAGL1 acts as a promoter of tomato fruit ripening. In SlFYFL-overexpressing tomato, fruit ripening was delayed by inhibiting carotenoid accumulation and ethylene production (Xie et al., 2014).
In our study, the accumulation of carotenoid was significantly reduced in the pericarp of SlMBP15-silenced fruits at B + 4 and B + 7 compared with the wild-type (Figure 5C). PSY1 is a major regulator of metabolic flux toward downstream carotenoids, which is induced by ethylene during fruit ripening (Fray and Grierson, 1993). The PDS and ZDS are two enzymes that work downstream of PSY1. The qPCR results revealed that the transcription of these three genes involved in carotenoid biosynthesis was all remarkably reduced in SlMBP15-silenced lines (Figures 5D–F), which revealed that SlMBP15 regulates the biosynthesis of carotenoids. In addition, the ethylene production in the transgenic lines was also repressed. The ACS and ACO are two rate-limiting enzymes for the ethylene biosynthesis. The ACS catalyzes the conversion of SAM (s-adenosyl-l-methionine) to ACC (aminocyclopropane-1-carboxylic acid), while the ACO modulate the conversion of ACC to ethylene (Kende, 1993). It has been reported that repression of ACS2 inhibited tomato fruit ripening (Oeller et al., 1991). Besides, ACO1 and ACO3 trigger fruit ripening in tomato (Alexander and Grierson, 2002). The significantly decreased expression of ACS1, ACO1 and ACO3 (Figures 6B–D) indicated that the silencing of SlMBP15 suppressed the ethylene biosynthesis in fruit.
Silencing SlMBP15 also down-regulated the transcription of ripening-associated genes including E4, E8, LOXA, LOXB, ERF1 and RIN. E4 and E8 are well-known and important ethylene response factors involved in fruit ripening (Lincoln and Fischer, 1988). The LOXs are enzymes that catalyze the lipoxygenase pathway to transform linoleic and linolenic acids into 9-hydroperoxides and 13-hydroperoxides (Chen et al., 2004) which are involved in the volatile production that is key for tomato flavor. Moreover, LOXB is a fruit-specific lipoxygenase that is induced by ethylene (Griffiths et al., 1999). ERF1 is an ethylene-responsive factor, while RIN is a MADS-box gene that affects the accumulation of carotenoids in tomato (Ito et al., 2017). The expression level of these genes was reduced in the SlMBP15-silenced lines. Taken together, these results indicated that SlMBP15 acts as a promoter of fruit ripening. However, owing to the complex process of fruit ripening controlled by MADS-box genes, more experiments are needed to figure out whether SlMBP15 plays direct or indirect roles in tomato fruit ripening.
RIN was considered as a major regulator that is essential for the induction of ripening, although recent research indicated that RIN does not play such a crucial role in fruit ripening and that the substantial inhibition of fruit ripening due to the rin mutation is caused by the gain-of-function of RIN-MC chimera (Ito et al., 2017); it is true that RIN modulates ethylene synthesis, inhibits fruit coloring and regulates the expression of many ripening-associated genes (Lin et al., 2008; Martel et al., 2011; Qin et al., 2012; Ito et al., 2017). MADS-box proteins usually function through the formation of heterodimers, homodimers, or higher-order complexes with each other (Favaro et al., 2002) to perform diverse functions. In this study, the ethylene production in tomato fruit was downregulated, and the fruit color turned lighter in the SlMBP15-silenced lines. Moreover, SlMBP15 regulated the expression of ripening-associated genes. Therefore, we carried out a yeast two-hybrid assay to examine whether SlMBP15 interacts with RIN. The result provided evidence for the interaction between SlMBP15 and RIN. In the previous study, some genes that mediate tomato fruit ripening were proved to interact with RIN such as SlMADS1 (Dong et al., 2013), SlFYFL (Xie et al., 2014), FUL1, FUL2 (Leseberg et al., 2008), TAGL1 (Martel et al., 2011) and SlMBP8 (Yin et al., 2017a). We conclude that SlMBP15 may take part in regulating the expression of carotenoid biosynthesis, ethylene biosynthesis and ripening-associated genes by cooperating with RIN.
The results showed that the MADS-box contributes substantially to the complex regulatory process of tomato fruit ripening. MADS-box genes participate in fruit ripening, but the roles they play and the positions they occupy in the regulatory network are different. Some MADS-box genes promote tomato fruit ripening, while some inhibit it. In the study of SlMBP15, it is interesting that the other FLC/MAF clade gene, SlMBP8, which is highly homologous to SlMBP15, is also involved in tomato fruit ripening (Yin et al., 2017a); however, unlike SlMBP15, SlMBP8 inhibits ripening. We speculated that SlMB15 may have been produced by SlMBP8 duplication and divergence, or vice versa and they probably work in opposite directions in the same regulatory pathway.
In this study, we identified a novel fruit ripening promoter that belongs to the MADS-box family. SlMBP15 regulates the transcription of genes involved in carotenoid and ethylene biosynthesis. SlMBP15 also affects the biosynthesis of chlorophyll and GA. Its function in tomato fruit ripening has been revealed here, but the higher-level developmental regulatory mechanism remains to be discovered. Since repression of SlMBP15 delays fruit ripening, it may be beneficial for extending the shelf life of tomato, which is a character with application potential.
Author Contributions
GC and ZH designed and managed the research work and improved the manuscript. WY, BT, YW, CL, and YZ performed the experiments. XY wrote the manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The reviewer JB and handling Editor declared their shared affiliation.
Funding. This work was supported by National Natural Science Foundation of China (Nos. 30600044 and 31572129) and the Opening Foundation of Science and Technology of Henan Province (172106000055).
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpls.2018.00938/full#supplementary-material
Alignment of the protein sequences of SlMBP15 and the transcript variants.
The predicted expression profile of SlMBP15 in tissues and cells in tomato fruit.
Gene information for SlMBP15.
The accession numbers of proteins contained in multiple sequence alignment and phylogenic analysis.
Primers used for Quantitative PCR analysis.
Accession numbers of SlMBP15 and the transcript variants.
Predicted expression profile of SlMBP15.
References
- Adams D. O., Yang S. F. (1979). Ethylene biosynthesis - identification of 1-Aminocyclopropane-1-carboxylic acid as an intermediate in the conversion of methionine to ethylene. Proc. Natl. Acad. Sci. U.S.A. 76 170–174. 10.1073/pnas.76.1.170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexander L., Grierson D. (2002). Ethylene biosynthesis and action in tomato: a model for climacteric fruit ripening. J. Exp. Bot. 53 2039–2055. 10.1093/jxb/erf072 [DOI] [PubMed] [Google Scholar]
- Ampomah-Dwamena C., Morris B. A., Sutherland P., Veit B., Yao J. L. (2002). Down-regulation of TM29, a tomato SEPALLATA homolog, causes parthenocarpic fruit development and floral reversion. Plant Physiol. 130 605–617. 10.1104/pp.005223 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bemer M., Karlova R., Ballester A. R., Tikunov Y. M., Bovy A. G., Wolters-Arts M., et al. (2012). The tomato FRUITFULL homologs TDR4/FUL1 and MBP7/FUL2 regulate ethylene-independent aspects of fruit ripening. Plant Cell 24 4437–4451. 10.1105/tpc.112.103283 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bleecker A. B., Kende H. (2000). Ethylene: a gaseous signal molecule in plants. Annu. Rev. Cell Dev. Biol. 16 1–18. 10.1146/annurev.cellbio.16.1.1 [DOI] [PubMed] [Google Scholar]
- Burko Y., Shleizer-Burko S., Yanai O., Shwartz I., Zelnik I. D., Jacob-Hirsch J., et al. (2013). A role for APETALA1/FRUITFULL transcription factors in tomato leaf development. Plant Cell 25 2070–2083. 10.1105/tpc.113.113035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen G. P., Hackett R., Walker D., Taylor A., Lin Z. F., Grierson D. (2004). Identification of a specific isoform of tomato lipoxygenase (TomloxC) involved in the generation of fatty acid-derived flavor compounds. Plant Physiol. 136 2641–2651. 10.1104/pp.104.041608 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung M. Y., Vrebalov J., Alba R., Lee J., Mcquinn R., Chung J. D., et al. (2010). A tomato (Solanum lycopersicum) APETALA2/ERF gene, SlAP2a, is a negative regulator of fruit ripening. Plant J. 64 936–947. 10.1111/j.1365-313X.2010.04384.x [DOI] [PubMed] [Google Scholar]
- Coen E. S., Meyerowitz E. M. (1991). The war of the whorls: genetic interactions controlling flower development. Nature 353 31–37. 10.1038/353031a0 [DOI] [PubMed] [Google Scholar]
- Colombo L., Franken J., Koetje E., Van Went J., Dons H. J., Angenent G. C., et al. (1995). The petunia MADS box gene FBP11 determines ovule identity. Plant Cell 7 1859–1868. 10.1105/tpc.7.11.1859 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daminato M., Masiero S., Resentini F., Lovisetto A., Casadoro G. (2014). Characterization of TM8, a MADS-box gene expressed in tomato flowers. BMC Plant Biol. 14:319. 10.1186/s12870-014-0319-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong T., Hu Z., Deng L., Wang Y., Zhu M., Zhang J., et al. (2013). A tomato MADS-box transcription factor, SlMADS1, acts as a negative regulator of fruit ripening. Plant Physiol. 163 1026–1036. 10.1104/pp.113.224436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Exposito-Rodriguez M., Borges A. A., Borges-Perez A., Perez J. A. (2008). Selection of internal control genes for quantitative real-time RT-PCR studies during tomato development process. BMC Plant Biol. 8:131. 10.1186/1471-2229-8-131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Favaro R., Immink R. G. H., Ferioli V., Bernasconi B., Byzova M., Angenent G. C., et al. (2002). Ovule-specific MADS-box proteins have conserved protein-protein interactions in monocot and dicot plants. Mol. Genet. Genomics 268 152–159. 10.1007/s00438-002-0746-6 [DOI] [PubMed] [Google Scholar]
- Forth D., Pyke K. A. (2006). The suffulta mutation in tomato reveals a novel method of plastid replication during fruit ripening. J. Exp. Bot. 57 1971–1979. 10.1093/jxb/erj144 [DOI] [PubMed] [Google Scholar]
- Fraser P. D., Truesdale M. R., Bird C. R., Schuch W., Bramley P. M. (1994). Carotenoid biosynthesis during tomato fruit-development. Plant Physiol. 105 405–413. 10.1104/pp.105.1.405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fray R. G., Grierson D. (1993). Identification and genetic-analysis of normal and mutant phytoene synthase genes of tomato by sequencing, complementation and co-suppression. Plant Mol. Biol. 22 589–602. 10.1007/BF00047400 [DOI] [PubMed] [Google Scholar]
- Fray R. G., Wallace A. D., Grierson D. (1994). Identification of unexplained DNA fragments within the T-DNA borders of the bin-19 plant transformation vector. Plant Mol. Biol. 25 339–342. 10.1007/BF00043863 [DOI] [PubMed] [Google Scholar]
- Frisch D. A., Harrishaller L. W., Yokubaitis N. T., Thomas T. L., Hardin S. H., Hall T. C. (1995). Complete sequence of the binary vector bin-19. Plant Mol. Biol. 27 405–409. 10.1007/BF00020193 [DOI] [PubMed] [Google Scholar]
- Fujisawa M., Nakano T., Shima Y., Ito Y. (2013). A large-scale identification of direct targets of the tomato MADS box transcription factor RIPENING INHIBITOR reveals the regulation of fruit ripening. Plant Cell 25 371–386. 10.1105/tpc.112.108118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garceau D. C., Batson M. K., Pan I. L. (2017). Variations on a theme in fruit development: the PLE lineage of MADS-box genes in tomato (TAGL1) and other species. Planta 246 313–321. 10.1007/s00425-017-2725-5 [DOI] [PubMed] [Google Scholar]
- Gimenez E., Dominguez E., Pineda B., Heredia A., Moreno V., Lozano R., et al. (2015). Transcriptional activity of the MADS box ARLEQUIN/TOMATO AGAMOUS-LIKE1 gene is required for cuticle development of tomato fruit. Plant Physiol. 168 1036–1048. 10.1104/pp.15.00469 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffiths A., Barry C., Alpuche-Solis A. G., Grierson D. (1999). Ethylene and developmental signals regulate expression of lipoxygenase genes during tomato fruit ripening. J. Exp. Bot. 50 793–798. 10.1093/jxb/50.335.793 [DOI] [Google Scholar]
- Guo X. H., Chen G. P., Cui B. L., Gao Q., Guo J. E., Li A. Z., et al. (2016). Solanum lycopersicum agamous-like MADS-box protein AGL15-like gene, SlMBP11, confers salt stress tolerance. Mol. Breed. 36 1–15. 10.1007/s11032-016-0544-1 [DOI] [Google Scholar]
- Guo X. H., Chen G. P., Naeem M., Yu X. H., Tang B. Y., Li A. Z., et al. (2017). The MADS-box gene SlMBP11 regulates plant architecture and affects reproductive development in tomato plants. Plant Sci. 258 90–101. 10.1016/j.plantsci.2017.02.005 [DOI] [PubMed] [Google Scholar]
- Hileman L. C., Sundstrom J. F., Litt A., Chen M., Shumba T., Irish V. F. (2006). Molecular and phylogenetic analyses of the MADS-box gene family in tomato. Mol. Biol. Evol. 23 2245–2258. 10.1093/molbev/msl095 [DOI] [PubMed] [Google Scholar]
- Hirschberg J. (2001). Carotenoid biosynthesis in flowering plants. Curr. Opin. Plant Biol. 4 210–218. 10.1016/S1369-5266(00)00163-1 [DOI] [PubMed] [Google Scholar]
- Ito Y., Nishizawa-Yokoi A., Endo M., Mikami M., Shima Y., Nakamura N., et al. (2017). Re-evaluation of the rin mutation and the role of RIN in the induction of tomato ripening. Nat. Plants 3 866–874. 10.1038/s41477-017-0041-5 [DOI] [PubMed] [Google Scholar]
- Keddie J. S., Carroll B., Jones J. D., Gruissem W. (1996). The DCL gene of tomato is required for chloroplast development and palisade cell morphogenesis in leaves. EMBO J. 15 4208–4217. [PMC free article] [PubMed] [Google Scholar]
- Kende H. (1993). Ethylene biosynthesis. Annu. Rev. Plant Physiol. Plant Mol. Biol. 44 283–307. 10.1146/annurev.pp.44.060193.001435 [DOI] [Google Scholar]
- Klap C., Yeshayahou E., Bolger A. M., Arazi T., Gupta S. K., Shabtai S., et al. (2016). Tomato facultative parthenocarpy results from SlAGAMOUS-LIKE 6 loss of function. Plant Biotechnol. J. 15 634–647. 10.1111/pbi.12662 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee B. H., Henderson D. A., Zhu J. K. (2005). The Arabidopsis cold-responsive transcriptome and its regulation by ICE1. Plant Cell 17 3155–3175. 10.1105/tpc.105.035568 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee I., Seo Y. S., Coltrane D., Hwang S., Oh T., Marcotte E. M., et al. (2011). Genetic dissection of the biotic stress response using a genome-scale gene network for rice. Proc. Natl. Acad. Sci. U.S.A. 108 18548–18553. 10.1073/pnas.1110384108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee S., Woo Y. M., Ryu S. I., Shin Y. D., Kim W. T., Park K. Y., et al. (2008). Further characterization of a rice AGL12 group MADS-box gene, OsMADS26. Plant Physiol. 147 156–168. 10.1104/pp.107.114256 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leseberg C. H., Eissler C. L., Wang X., Johns M. A., Duvall M. R., Mao L. (2008). Interaction study of MADS-domain proteins in tomato. J. Exp. Bot. 59 2253–2265. 10.1093/jxb/ern094 [DOI] [PubMed] [Google Scholar]
- Li N., Huang B. W., Tang N., Jian W., Zou J., Chen J., et al. (2017). The MADS-box gene SlMBP21 regulates sepal size mediated by ethylene and auxin in tomato. Plant Cell Physiol. 58 2241–2256. 10.1093/pcp/pcx158 [DOI] [PubMed] [Google Scholar]
- Lin Z. F., Hong Y. G., Yin M. G., Li C. Y., Zhang K., Grierson D. (2008). A tomato HD-Zip homeobox protein, LeHB-1, plays an important role in floral organogenesis and ripening. Plant J. 55 301–310. 10.1111/j.1365-313X.2008.03505.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lincoln J. E., Cordes S., Read E., Fischer R. L. (1987). Regulation of gene-expression by ethylene during lycopersicon-esculentum (Tomato) fruit-development. Proc. Natl. Acad. Sci. U.S.A. 84 2793–2797. 10.1073/pnas.84.9.2793 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lincoln J. E., Fischer R. L. (1988). Diverse mechanisms for the regulation of ethylene-inducible gene-expression. Mol. Gen. Genet. 212 71–75. 10.1007/BF00322446 [DOI] [PubMed] [Google Scholar]
- Liu D., Wang D., Qin Z., Zhang D., Yin L., Wu L., et al. (2014). The SEPALLATA MADS-box protein SLMBP21 forms protein complexes with JOINTLESS and MACROCALYX as a transcription activator for development of the tomato flower abscission zone. Plant J. 77 284–296. 10.1111/tpj.12387 [DOI] [PubMed] [Google Scholar]
- Livak K. J., Schmittgen T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2(T)(-Delta Delta C) method. Methods 25 402–408. 10.1006/meth.2001.1262 [DOI] [PubMed] [Google Scholar]
- Mao L., Begum D., Chuang H. W., Budiman M. A., Szymkowiak E. J., Irish E. E., et al. (2000). JOINTLESS is a MADS-box gene controlling tomato flower abscission zone development. Nature 406 910–913. 10.1038/35022611 [DOI] [PubMed] [Google Scholar]
- Martel C., Vrebalov J., Tafelmeyer P., Giovannoni J. J. (2011). The tomato MADS-box transcription factor RIPENING INHIBITOR interacts with promoters involved in numerous ripening processes in a COLORLESS NONRIPENING-dependent manner. Plant Physiol. 157 1568–1579. 10.1104/pp.111.181107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maunders M. J., Holdsworth M. J., Slater A., Knapp J. E., Bird C. R., Schuch W., et al. (1987). Ethylene stimulates the accumulation of ripening-related messenger-RNAs in tomatoes. Plant Cell Environ. 10 177–184. [Google Scholar]
- Messenguy F., Dubois E. (2003). Role of MADS box proteins and their cofactors in combinatorial control of gene expression and cell development. Gene 316 1–21. 10.1016/S0378-1119(03)00747-9 [DOI] [PubMed] [Google Scholar]
- Murase K., Hirano Y., Sun T. P., Hakoshima T. (2008). Gibberellin-induced DELLA recognition by the gibberellin receptor GID1. Nature 456 459–463. 10.1038/nature07519 [DOI] [PubMed] [Google Scholar]
- Nakano T., Kimbara J., Fujisawa M., Kitagawa M., Ihashi N., Maeda H., et al. (2012). MACROCALYX and JOINTLESS interact in the transcriptional regulation of tomato fruit abscission zone development. Plant Physiol. 158 439–450. 10.1104/pp.111.183731 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nguyen C. V., Vrebalov J. T., Gapper N. E., Zheng Y., Zhong S. L., Fei Z. J., et al. (2014). Tomato GOLDEN2-LIKE transcription factors reveal molecular gradients that function during fruit development and ripening. Plant Cell 26 585–601. 10.1105/tpc.113.118794 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oeller P. W., Wong L. M., Taylor L. P., Pike D. A., Theologis A. (1991). Reversible inhibition of tomato fruit senescence by antisense RNA. Science 254 437–439. 10.1126/science.1925603 [DOI] [PubMed] [Google Scholar]
- Olszewski N., Sun T. P., Gubler F. (2002). Gibberellin signaling: biosynthesis, catabolism, and response pathways. Plant Cell 14 (Suppl.), S61–S80. 10.1105/tpc.010476 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelaz S., Tapia-Lopez R., Alvarez-Buylla E. R., Yanofsky M. F. (2001). Conversion of leaves into petals in Arabidopsis. Curr. Biol. 11 182–184. 10.1016/S0960-9822(01)00024-0 [DOI] [PubMed] [Google Scholar]
- Penarrubia L., Aguilar M., Margossian L., Fischer R. L. (1992). An antisense gene stimulates ethylene hormone production during tomato fruit ripening. Plant Cell 4 681–687. 10.1105/tpc.4.6.681 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Powell A. L. T., Nguyen C. V., Hill T., Cheng K. L., Figueroa-Balderas R., Aktas H., et al. (2012). Uniform ripening encodes a golden 2-like transcription factor regulating tomato fruit chloroplast development. Science 336 1711–1715. 10.1126/science.1222218 [DOI] [PubMed] [Google Scholar]
- Qin G., Wang Y., Cao B., Wang W., Tian S. (2012). Unraveling the regulatory network of the MADS box transcription factor RIN in fruit ripening. Plant J. 70 243–255. 10.1111/j.1365-313X.2011.04861.x [DOI] [PubMed] [Google Scholar]
- Shi L. F., Olszewski N. E. (1998). Gibberellin and abscisic acid regulate GAST1 expression at the level of transcription. Plant Mol. Biol. 38 1053–1060. 10.1023/A:1006007315718 [DOI] [PubMed] [Google Scholar]
- Sommer H., Beltran J. P., Huijser P., Pape H., Lonnig W. E., Saedler H., et al. (1990). Deficiens, a homeotic gene involved in the control of flower morphogenesis in Antirrhinum majus: the protein shows homology to transcription factors. EMBO J. 9 605–613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soyk S., Lemmon Z. H., Oved M., Fisher J., Liberatore K. L., Park S. J., et al. (2017). Bypassing negative epistasis on yield in tomato imposed by a domestication gene. Cell 169 1142–1155.e12. 10.1016/j.cell.2017.04.032 [DOI] [PubMed] [Google Scholar]
- Shinozaki Y., Nicolas P., Fernandez-Pozo N., Ma Q. Y., Evanich D. J., Shi Y. N., et al. (2018). High-resolution spatiotemporal transcriptome mapping of tomato fruit development and ripening. Nat. Commun, 9:364. 10.1038/s41467-017-02782-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szymkowiak E. J., Irish E. E. (2006). JOINTLESS suppresses sympodial identity in inflorescence meristems of tomato. Planta 223 646–658. 10.1007/s00425-005-0115-x [DOI] [PubMed] [Google Scholar]
- Tardif G., Kane N. A., Adam H., Labrie L., Major G., Gulick P., et al. (2007). Interaction network of proteins associated with abiotic stress response and development in wheat. Plant Mol. Biol. 63 703–718. 10.1007/s11103-006-9119-6 [DOI] [PubMed] [Google Scholar]
- Theissen G., Becker A., Di Rosa A., Kanno A., Kim J. T., Munster T., et al. (2000). A short history of MADS-box genes in plants. Plant Mol. Biol. 42 115–149. 10.1023/A:1006332105728 [DOI] [PubMed] [Google Scholar]
- Ueguchi-Tanaka M., Nakajima M., Katoh E., Ohmiya H., Asano K., Saji S., et al. (2007). Molecular interactions of a soluble gibberellin receptor, GID1, with a rice DELLA protein, SLR1, and gibberellin. Plant Cell 19 2140–2155. 10.1105/tpc.106.043729 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vrebalov J., Pan I. L., Arroyo A. J., Mcquinn R., Chung M., Poole M., et al. (2009). Fleshy fruit expansion and ripening are regulated by the Tomato SHATTERPROOF gene TAGL1. Plant Cell 21 3041–3062. 10.1105/tpc.109.066936 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vrebalov J., Ruezinsky D., Padmanabhan V., White R., Medrano D., Drake R., et al. (2002). A MADS-box gene necessary for fruit ripening at the tomato ripening-inhibitor (rin) locus. Science 296 343–346. 10.1126/science.1068181 [DOI] [PubMed] [Google Scholar]
- Wang S., Lu G., Hou Z., Luo Z., Wang T., Li H., et al. (2014). Members of the tomato FRUITFULL MADS-box family regulate style abscission and fruit ripening. J. Exp. Bot. 65 3005–3014. 10.1093/jxb/eru137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wesley S. V., Helliwell C. A., Smith N. A., Wang M. B., Rouse D. T., Liu Q., et al. (2001). Construct design for efficient, effective and high-throughput gene silencing in plants. Plant J. 27 581–590. 10.1046/j.1365-313X.2001.01105.x [DOI] [PubMed] [Google Scholar]
- Xie Q., Hu Z., Zhu Z., Dong T., Zhao Z., Cui B., et al. (2014). Overexpression of a novel MADS-box gene SlFYFL delays senescence, fruit ripening and abscission in tomato. Sci. Rep. 4:4367. 10.1038/srep04367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamaguchi S. (2008). Gibberellin metabolism and its regulation. Annu. Rev. Plant Biol. 59 225–251. 10.1146/annurev.arplant.59.032607.092804 [DOI] [PubMed] [Google Scholar]
- Yanofsky M. F., Ma H., Bowman J. L., Drews G. N., Feldmann K. A., Meyerowitz E. M. (1990). The protein encoded by the Arabidopsis homeotic gene agamous resembles transcription factors. Nature 346 35–39. 10.1038/346035a0 [DOI] [PubMed] [Google Scholar]
- Yin W., Hu Z., Cui B., Guo X., Hu J., Zhu Z., et al. (2017a). Suppression of the MADS-box gene SlMBP8 accelerates fruit ripening of tomato (Solanum lycopersicum). Plant Physiol. Biochem. 118 235–244. 10.1016/j.plaphy.2017.06.019 [DOI] [PubMed] [Google Scholar]
- Yin W., Hu Z. L., Hu J. T., Zhu Z. G., Yu X. H., Cui B. L., et al. (2017b). Tomato (Solanum lycopersicum) MADS-box transcription factor SlMBP8 regulates drought, salt tolerance and stress-related genes. Plant Growth Regul. 83 55–68. 10.1007/s10725-017-0283-2 [DOI] [Google Scholar]
- Yu X. H., Chen G. P., Guo X. H., Lu Y., Zhang J. L., Hu J. T., et al. (2017). Silencing SlAGL6, a tomato AGAMOUS-LIKE6 lineage gene, generates fused sepal and green petal. Plant Cell Rep. 36 959–969. 10.1007/s00299-017-2129-9 [DOI] [PubMed] [Google Scholar]
- Yuste-Lisbona F. J., Quinet M., Fernandez-Lozano A., Pineda B., Moreno V., Angosto T., et al. (2016). Characterization of vegetative inflorescence (mc-vin) mutant provides new insight into the role of MACROCALYX in regulating inflorescence development of tomato. Sci. Rep. 6:18796. 10.1038/srep18796 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Alignment of the protein sequences of SlMBP15 and the transcript variants.
The predicted expression profile of SlMBP15 in tissues and cells in tomato fruit.
Gene information for SlMBP15.
The accession numbers of proteins contained in multiple sequence alignment and phylogenic analysis.
Primers used for Quantitative PCR analysis.
Accession numbers of SlMBP15 and the transcript variants.
Predicted expression profile of SlMBP15.