Abstract

The nickel PNP pincer complex (iPrPNP)NiPh (iPrPNP = κP,κN,κP-N(CH2CH2PiPr2)2) was prepared by reacting (iPrPNP)NiBr with PhMgCl or deprotonating [(iPrPNHP)NiPh]Y (iPrPNHP = κP,κN,κP-HN(CH2CH2PiPr2)2; Y = Br, PF6) with KOtBu. The byproducts of the PhMgCl reaction were identified as [(iPrPNHP)NiPh]Br and (iPrPNP′)NiPh (iPrPNP′ = κP,κN,κP-N(CH=CHPiPr2)(CH2CH2PiPr2)). The methyl analog (iPrPNP)NiMe was synthesized from the reaction of (iPrPNP)NiBr with MeLi, although it was contaminated with (iPrPNP′)NiMe due to ligand oxidation. Protonation of (iPrPNP)NiX (X = Br, Ph, Me) with various acids, such as HCl, water, and MeOH, was studied in C6D6. Nitrogen protonation was shown to be the most favorable process, producing a cationic species [(iPrPNHP)NiX]+ with the NH moiety hydrogen-bonded to the conjugate base (i.e., Cl–, HO–, or MeO–). Protonation of the Ni–C bond was observed at room temperature with (iPrPNP)NiMe, whereas at 70 °C with (iPrPNP)NiPh, both resulting in [(iPrPNHP)NiCl]Cl as the final product. Protonation of (iPrPNP)NiBr was complicated by site exchange between Br– and the conjugate base and by the degradation of the pincer complexes. Indene, which lacks hydrogen-bonding capability, was unable to protonate (iPrPNP)NiPh and (iPrPNP)NiMe, despite being more acidic than water and MeOH. Neutral and cationic nickel pincer complexes involved in this study, including (iPrPNP′)NiBr, (iPrPNP)NiPh, (iPrPNP′)NiPh, (iPrPNP)NiMe, [(iPrPNHP)NiPh]Y (Y = Br, PF6, BPh4), [(iPrPNHP)NiPh]2[NiCl4], [(iPrPNHP)NiMe]Y (Y = Cl, Br, BPh4), [(iPrPNHP)NiBr]Br, and [(iPrPNHP)NiCl]Cl, were characterized by X-ray crystallography.
Introduction
The advances of metal–ligand bifunctional catalysis have provided the momentum for the recent surge in developing base metal-catalyzed (de)hydrogenation reactions.1−9 The key to the success of these reactions is often attributed to ligand participation in proton-transfer steps, which helps activate substrates while deemphasizing the roles that metals need to play. One particularly effective and popular ligand system has the general formula HN(CH2CH2PR2)2 (abbreviated here as RPNHP),10 which is commonly referred to as PNP pincer when the ligand adopts the meridional coordination mode. Over the past few years, Ni,11−14 Co,11,15−21 Fe,22−30 and Mn complexes31−33 bearing the PNP pincer ligands or their deprotonated form [N(CH2CH2PR2)2]− (abbreviated here as RPNP) have been successfully produced, many of which have shown excellent catalytic activity for hydrogenation,16,21,25−42 dehydrogenation,16,22,34,35,43−52 and related transfer hydrogenation reactions.20,53 Although the presence of the NH group is not always necessary,53 especially for the reduction of olefins12,16 and CO2,54−56 its function in many (de)hydrogenation processes has been well established.16,26,38,44,57 As illustrated by a generalized mechanism for carbonyl reduction (Scheme 1; for clarity, other ligands are removed), H+/H– transfer from nitrogen/metal to the O=C bond generates the alcohol product and an amido complex, which activates H2 to close the catalytic cycle. The reverse process, namely, alcohol dehydrogenation, proceeds via H+/H– transfer from the alcohol molecule to the amido complex. Although the key features have been captured, the missing piece of the mechanistic picture is an adduct of the alcohol molecule and the amido species linked by a hydrogen-bonding interaction. According to density functional theory calculations,34,43,58 such an intermediate represents a local minimum in energy and plays a critical role in H2 activation (for hydrogenation) or elimination (for dehydrogenation).57,59 Isolation or even spectroscopic observation of this specific intermediate has proven to be challenging or may not even be possible, yet understanding its bonding and reactivity is essential for the further development of the catalytic reactions.
Scheme 1. Hydrogen Transfer in (De)Hydrogenation Reactions Catalyzed by PNP Pincer Complexes.
In this work, we chose the nickel PNP pincer system as the platform to probe the hydrogen-bonding interaction in question. We simplified the research problem by focusing on the protonation of (iPrPNP)NiX (X = Br, Ph, Me) with various Brønsted acids, including methanol and water (eq 1). In particular, products resulting from methanol protonation should mimic the alcohol adduct depicted in Scheme 1. In addition to examining different acids, we varied the nickel-bound ligand X with an objective to study its effect on the protonation reaction.
 |
1 |
Results and Discussion
Synthesis and Characterization of Nickel Phenyl Complexes
The synthesis of the nickel bromide complex (iPrPNP)NiBr (1) was already reported by the Arnold group in 2011.11 We thus turned our initial attention to the target phenyl complex (iPrPNP)NiPh (2). Group 10 metal phenyl complexes supported by a pincer ligand are often readily prepared by treating the corresponding metal halide complexes with PhLi60−63 or PhMgX (X = Cl, Br).64−66 Applying this synthetic strategy to the nickel PNP pincer system is, however, nontrivial. The room-temperature reaction of 1 with PhLi did not go to completion unless more than 3 equiv of PhLi were added, in which case a complicated mixture was obtained. Similar phenomena were observed when Ph2Zn was employed in place of PhLi. Using PhMgCl as the source of the phenyl group also resulted in multiple products but needed only 1 equiv of the Grignard reagent to fully convert 1. Fortunately, the solubility difference of the products in pentane made separation possible (see Experimental Section for details). In addition to the anticipated complex 2, a cationic nickel phenyl complex [(iPrPNHP)NiPh]Br ([2H]Br) and a neutral nickel phenyl complex bearing a dehydrogenated PNP pincer ligand (complex 2′) were isolated (eq 2). Control experiments indicated that when exposed to air, both 1 and 2 decomposed rapidly, resulting in a mixture of a cationic species, a complex with a dehydrogenated PNP pincer ligand, and other unidentified species. Efforts were thus made to avoid contact with oxygen and moisture during the synthesis of 2. Unfortunately, [2H]Br and 2′ remained present in the crude product.
 |
2 |
Schneider and co-workers have previously mentioned low selectivity for the reaction between [(iPrPNHP)PdCl]PF6 and a phenyl Grignard reagent.67 They have therefore developed an alternate route to [(iPrPNHP)PdPh]PF6 using (TMEDA)PdPhI (TMEDA = Me2NCH2CH2NMe2), iPrPNHP, and AgPF6. Additionally, they have shown that deprotonation of [(iPrPNHP)PdPh]PF6 by KOtBu provides the neutral phenyl complex (iPrPNP)PdPh in good yield. We found that this strategy could also be successfully implemented using (TMEDA)NiPhI as the starting material for the synthesis of [(iPrPNHP)NiPh]PF6 ([2H]PF6) and 2 (Scheme 2). Alternatively, 2 could be isolated from deprotonation of [2H]Br with KOtBu.
Scheme 2. Independent Synthesis of Complex 2.
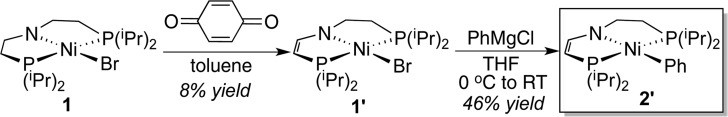
To confirm the formation of 2′, a nickel bromide complex supported by the dehydrogenated PNP pincer ligand (complex 1′; see Scheme 3) was prepared first following the procedure established for the dehydrogenation of (tBuPNP)CoCl,19 although the isolated yield was low (8%). Synthesis of 2′ from 1′ and PhMgCl was relatively straightforward, reflecting higher stability of 1′ and 2′ toward oxygen and moisture. Previous studies of (iPrPNP)Ru(PMe3)H have suggested that the PNP pincer ligand, when dehydrogenated, can be restored in the presence of H2 (1 bar).68,69 In our work, exposing a 95:5 mixture of 1 and 1′ (in C6D6) to 2 bar of H2 over a period of 5 days showed no sign of hydrogenation of 1′ to 1. The phenyl complex 2′ also resisted hydrogenation under similar conditions.
Scheme 3. Independent Synthesis of Complex 2′.
The nickel phenyl complexes described above were characterized by NMR spectroscopy and elemental analysis. Consistent with the symmetries of the molecules, 2 displays two sets of CH3 resonances in its 1H and 13C{1H} NMR spectra, whereas 2′, [2H]Br, and [2H]PF6 each show four different sets of CH3 resonances. The higher symmetry of 2 is further manifested by the observation of only one carbon resonance for the CH groups of the pincer ligand. In contrast, two pincer CH resonances are located for 2′, [2H]Br, and [2H]PF6. As expected, the 31P{1H} NMR spectrum of 2′ displays an AB quartet for the two inequivalent phosphorus nuclei with a large coupling constant of 259.7 Hz.
The structures of 2, 2′, [2H]Br, and [2H]PF6 were further established by X-ray crystallography. The dehydrogenated PNP pincer ligand in 2′ is evident from a relatively short C(1)–C(2) distance of 1.392(3) Å (Figure 1 and Table 1), which is best described as a C=C bond.13,19 Its C(3)–C(4) bond [1.462(3) Å], although shorter than the C(1)–C(2) [1.517(2) Å] and C(3)–C(4) [1.514(2) Å] bonds in 2 (Figure 2), is most consistent with a single bond. The phenyl group in 2 and 2′ adopts a conformation almost perpendicular to the coordination plane; the dihedral angles between the least-square planes P(1)–P(2)–N(1)–Ni and C(17) through C(22) were measured to be 76.34(4) and 75.60(5)° for 2 and 2′, respectively. The sum of angles about the nitrogen (∑N) is 354.05° for 2 and 359.49° for 2′, suggesting that the nitrogen lone-pair electrons are delocalized into the neighboring π-system and/or nickel. The cations of [2H]Br (Figure 3) and [2H]PF6 (Figure 4) are almost structurally identical to each other with only slight differences in Ni–P bond lengths and P–Ni–P angle (Table 1). Like 2 and 2′, the phenyl group in [2H]Br and [2H]PF6 is nearly perpendicular to the coordination plane, presumably to minimize the steric clash with the isopropyl groups. The Ni–N bonds of [2H]Br [1.985(2) Å] and [2H]PF6 [1.999(3) Å] are substantially longer than those of 2 [1.8932(11)Å] and 2′ [1.9139(14) Å], which reflects the difference between an amine and an amido donor. Perhaps the most notable structural feature of [2H]Br is the hydrogen-bonding interaction (Table S1) between the NH group and the counterion, bromide [NH···Br = 3.370(3) Å]. It is interesting to note that weaker hydrogen bonds also exist in [2H]PF6 between the NH moiety and the fluorine atoms of PF6– [NH···F = 3.021(7) Å] (Figure 4 and Table S1). The extent of hydrogen-bonding interaction in the solid state was probed by IR spectroscopy, which showed a broad band at 3351 cm–1 for [2H]Br and a sharp band at 3249 cm–1 in the case of [2H]PF6, both assigned to the N–H bond stretch.
Figure 1.

Oak Ridge thermal ellipsoid plot (ORTEP) drawing of (iPrPNP′)NiPh (2′) at the 50% probability level. Hydrogen atoms except those attached to the pincer backbone are omitted for clarity.
Table 1. Selected Bond Lengths (Å) and Angles (deg) for the Nickel Phenyl Complexes.
| (iPrPNP)NiPh (2) | (iPrPNP′)NiPh (2′) | [(iPrPNHP)NiPh]Br ([2H]Br) | [(iPrPNHP)NiPh]PF6 ([2H]PF6) | [(iPrPNHP)NiPh]BPh4 ([2H]BPh4)a | [(iPrPNHP)NiPh]2[NiCl4] ([2H]2[NiCl4])b | |
|---|---|---|---|---|---|---|
| Ni–N(1) | 1.8932(11) | 1.9139(14) | 1.985(2) | 1.999(3) | 1.997(2) | 1.9806(18) |
| Ni–C(17) | 1.9163(13) | 1.9066(16) | 1.909(3) | 1.900(3) | 1.903(2) | 1.906(2) |
| Ni–P(1) | 2.1513(4) | 2.1743(5) | 2.1801(8) | 2.1960(6) | 2.1780(8) | 2.1812(5) |
| Ni–P(2) | 2.1492(4) | 2.1675(5) | 2.1769(8) | 2.1960(6) | 2.1827(7) | 2.1760(6) |
| C(1)–C(2) | 1.517(2) | 1.392(3) | 1.511(4) | 1.521(3) | 1.512(4) | 1.518(3) |
| C(3)–C(4) | 1.514(2) | 1.462(3) | 1.514(4) | 1.521(3) | 1.518(4) | 1.506(4) |
| P(1)–Ni–P(2) | 172.006(15) | 171.250(19) | 173.87(3) | 168.05(4) | 174.55(3) | 172.04(2) |
| C(17)–Ni–N(1) | 179.22(5) | 178.21(7) | 179.71(12) | 175.85(13) | 178.88(11) | 177.26(9) |
| dihedral anglec | 76.34(4) | 75.60(5) | 75.45(8) | 84.99(2) | 75.43(7) | 79.92(5) |
[2H]BPh4 crystallizes as [2H]BPh4·3/2THF.
[2H]2[NiCl4] crystallizes as [2H]2[NiCl4]·4C6D6·1/2H2O.
Angle between the least-square planes P(1)–P(2)–N(1)–Ni and C(17) through C(22).
Figure 2.

ORTEP drawing of (iPrPNP)NiPh (2) at the 50% probability level. Hydrogen atoms are omitted for clarity.
Figure 3.

ORTEP drawing of [(iPrPNHP)NiPh]Br ([2H]Br) at the 50% probability level. Hydrogen atoms except the one attached to the nitrogen are omitted for clarity.
Figure 4.

ORTEP drawing of [(iPrPNHP)NiPh]PF6 ([2H]PF6) at the 50% probability level. Hydrogen atoms except the one attached to the nitrogen are omitted for clarity. The PF6 counterion is disordered in the fluorine atoms.
Synthesis of the cationic nickel phenyl complex devoid of hydrogen bonds is possible. This was accomplished by a salt metathesis reaction between [2H]Br and NaBPh4, as illustrated in eq 3. The product [2H]BPh4, when recrystallized in tetrahydrofuran (THF)-pentane, provides a structure without hydrogen-bonding interaction (Figure 5) despite the fact that THF molecules are present in the crystal lattice. In contrast, our previous crystallographic analysis of an iron PNP pincer complex trans-(iPrPNHP)Fe(CO)Br2 showed that a co-crystallized THF molecule was hydrogen-bonded to the NH group.25 Nevertheless, the lack of hydrogen bonds in [2H]BPh4 appears to have a negligible impact on key angles and distances (Table 1) as well as the orientation of the phenyl group. One might have anticipated that without the N–H···X interaction, the nitrogen is less amido like, resulting in a longer Ni–N bond. The IR spectrum of [2H]BPh4 (solid) shows a sharp N–H stretching band at 3181 cm–1, further supporting that hydrogen bonds are absent in this molecule.
 |
3 |
Figure 5.

ORTEP drawing of [(iPrPNHP)NiPh]BPh4 ([2H]BPh4) at the 50% probability level. THF molecules (co-crystallized with [2H]BPh4) and all hydrogen atoms except the one attached to the nitrogen are omitted for clarity.
Because the reaction of 1 with PhMgCl (eq 2) involves two different halides, there is a possibility that the counterion for the cationic phenyl complex could be a mixture of Br– and Cl–. However, both elemental analysis and X-ray study (barring selective crystallization of one compound) support the [2H]Br formulation. Pure [2H]Cl, which was obtained from protonation of 2 by HCl (eq 4), gives similar but distinctively different NMR spectra. In particular, the NH resonance is shifted from 5.35 ppm for [2H]Br (in CD3CN) to 6.25 ppm for [2H]Cl, consistent with a stronger hydrogen bond formed by a more electronegative halide. The equilibrium for the halide exchange (eq 5, approached from both directions) apparently lies far from the [2H]Br/MgCl2 side, likely driven by the more negative Gibbs free energy of formation for MgCl2.70
 |
4 |
 |
5 |
The aforementioned cationic nickel phenyl complexes are soluble in polar solvents, such as acetonitrile and acetone. In benzene, the ones with a strong hydrogen-bonding interaction ([2H]Cl and [2H]Br) are sparsely soluble, whereas the ones with weak or no hydrogen-bonding interaction ([2H]PF6 and [2H]BPh4) are insoluble. Interestingly, a concentrated solution of [2H]Cl in C6D6 left under an argon atmosphere for 2 weeks yielded some single crystals, which were analyzed by X-ray crystallography as [2H]2[NiCl4] (Figure 6). As summarized in Table 1, the structural parameters of the cation are virtually the same as those of other cationic nickel phenyl complexes. The chlorides of [NiCl4]2– are all hydrogen-bonded, two with the NH moieties [NH···Cl = 3.327(2) Å] (Table S1) and the other two with co-crystallized water molecules [OH···Cl = 3.321(5) Å].
Figure 6.
ORTEP drawing of [(iPrPNHP)NiPh]2[NiCl4] ([2H]2[NiCl4]) at the 50% probability level. Co-crystallized C6D6 and H2O molecules and all hydrogen atoms, except the one attached to the nitrogen, are omitted for clarity.
In solution, the rotation of the phenyl group appears to be restricted, as judged by NMR spectroscopy. Complex [2H]PF6 or [2H]BPh4 dissolved in CD3CN exhibited two different resonances for the ortho carbons. Although [2H]Cl or [2H]Br in CD3CN showed only one ortho carbon resonance as a triplet, the solution in C6D6 gave a 1H NMR spectrum with two aromatic hydrogen resonances attributable to the ortho hydrogens.
Synthesis and Characterization of Nickel Methyl Complexes
Complex (iPrPNP)NiMe (3) has been previously prepared by treating [(iPrPNHP)NiBr]Br with 2.5 equiv of MeLi.71 Our attempts to repeat this synthesis were, however, unsuccessful; other synthetic routes (for analogous complexes65,72−78) were thus pursued. Under the conditions used for the synthesis of 2 (eq 2), 1 reacted sluggishly with MeMgBr, as inferred by the persistent green characteristic of 1. Extending the reaction time (from 1 to 17 h) eventually led to the disappearance of the green color, although NMR analysis of the crude product revealed a very complicated mixture. A more successful route involved the reaction of 1 with MeLi in diethyl ether (eq 6), which was previously used for the synthesis of (CyPNP)NiMe bearing cyclohexyl groups as the phosphorus substituents.12 The isolated material was confirmed as the desired compound 3 although contaminated with a small amount of byproduct 3′. The percentage of 3′ varied from batch to batch (5–20%), likely due to the high sensitivity of 3 toward the adventitious O2 present during the synthesis. The amount of cationic nickel methyl species was negligible (<1%). The 31P{1H} NMR spectrum of the isolated material shows a singlet at 68.81 ppm for 3 (in C6D6) along with a small AB quartet at 62.71 and 49.86 ppm (JAB = 275.4 Hz) for 3′. As expected for 3, the nickel-bound methyl group appears in the upfield region, featuring triplets at −0.59 (JP–H = 8.6 Hz) and −26.32 ppm (JP–C = 25.6 Hz) for the proton and carbon resonances, respectively.
 |
6 |
Cationic nickel methyl complexes [3H]Br and [3H]Cl were prepared by generating 3 in situ followed by protonation with MeOH (Scheme 4). Instead of bearing MeO– as the counterion, the isolated products contain the original halide hydrogen-bonded to the NH moiety (vide infra). The byproduct LiOMe is presumably stabilized by MeOH through solvation. The halide can be removed via a salt metathesis reaction with NaBPh4, as demonstrated by a successful synthesis of [3H]BPh4 from 1.
Scheme 4. Synthesis of Cationic Nickel Methyl Complexes.
The 1H and 13C{1H} NMR spectra of [3H]Cl, [3H]Br, and [3H]BPh4 (in CD3CN) show more splittings than 3 (in C6D6) due to reduced symmetry. Chemical shifts for the three cations differ only slightly. The most significantly shifted resonances are the NH hydrogen observed as a broad triplet ([3H]Cl, 5.61 ppm; [3H]Br, 4.92 ppm; [3H]BPh4, 3.57 ppm) and NiCH3 carbon also seen as a triplet ([3H]Cl, −24.27 ppm; [3H]Br, −24.04 ppm; [3H]BPh4, −23.73 ppm). The trend for the NH chemical shifts can be rationalized by the extent of hydrogen-bonding interaction, which follows the order [3H]Cl > [3H]Br > [3H]BPh4.
The four nickel methyl complexes (3, [3H]Cl, [3H]Br, and [3H]BPh4) in their solid state were studied by X-ray crystallography (Figure 7). As summarized in Table 2, there is very little structural variation in the cation of [3H]Cl, [3H]Br, and [3H]BPh4. Similar to the phenyl series, an hydrogen-bonding interaction (Table S1) is present between the NH moiety and Cl–/Br– [NH···Cl = 3.140(2) Å, NH···Br = 3.010(16) Å] but absent when the counterion is BPh4–. This was further supported by IR spectroscopy, which revealed a very broad N–H stretching band for [3H]Cl (3393 cm–1) and [3H]Br (3399 cm–1), whereas a relatively sharp band for [3H]BPh4 (3189 cm–1). Compared to the neutral methyl complex 3, the Ni–N and Ni–P bonds of the cationic species are elongated by 0.09–0.11 and 0.02–0.05 Å, respectively. Without the proton, the amido nitrogen of 3 is nearly flat (∑N = 353.05°), similar to that of the phenyl complex 2 (∑N = 354.05°) but less planar than that observed for the complexes supported by the dehydrogenated PNP pincer ligand (2′, ∑N = 359.49°; 1′, ∑N = 359.1 or 359.7°; see Supporting Information for details). Interestingly, the related palladium complexes (iPrPNP)PdX (X = Cl, ∑N = 337.4°; X = Me, ∑N = 345.7°)67 as well as the nickel bromide complex 1 (∑N = 348.30 or 348.35°)11 were reported to have a more pyramidal amido nitrogen.
Figure 7.
ORTEP drawings of nickel methyl complexes at the 50% probability level. Hydrogen atoms except the one attached to the nitrogen are omitted for clarity.
Table 2. Selected Bond Lengths (Å) and Angles (deg) for Nickel Methyl Complexes.
| (iPrPNP)NiMe (3) | [(iPrPNHP)NiMe]Cl ([3H]Cl) | [(iPrPNHP)NiMe]Br ([3H]Br) | [(iPrPNHP)NiMe]BPh4 ([3H]BPh4) | |
|---|---|---|---|---|
| Ni–N(1) | 1.8943(17) | 1.9897(13) | 1.9866(14) | 2.0081(12) |
| Ni–C(17) | 1.968(2) | 1.9488(17) | 1.9756(18) | 1.9608(15) |
| Ni–P(1) | 2.1471(6) | 2.1806(4) | 2.1737(4) | 2.1814(4) |
| Ni–P(2) | 2.1504(6) | 2.1744(4) | 2.1810(5) | 2.1976(4) |
| C(1)–C(2) | 1.516(3) | 1.518(2) | 1.517(2) | 1.519(2) |
| C(3)–C(4) | 1.519(3) | 1.520(2) | 1.518(2) | 1.517(2) |
| P(1)–Ni–P(2) | 169.25(2) | 172.998(17) | 172.966(18) | 173.884(16) |
| C(17)–Ni–N(1) | 171.23(10) | 178.10(8) | 178.10(8) | 177.49(6) |
Protonation of the Nickel PNP Pincer Complexes
Since the nickel halide complexes lack acid-labile Ni–C bonds, for simplicity, their protonation reactions were investigated first (Scheme 5). As anticipated, adding 1 equiv of HCl (2 M solution in Et2O) to the chloride complex 4 provided [4H]Cl quantitatively as a result of nitrogen protonation. No hydride species was observed during the reaction. Protonation of the metal is likely to be kinetically and thermodynamically unfavorable,79−81 as it would require a geometric reorganization at nickel and a formal oxidation of Ni(II) to Ni(IV). The product [4H]Cl was synthesized independently by mixing NiCl2 with iPrPNHP and was characterized by NMR spectroscopy and elemental analysis. IR studies showed that the N–H stretching band was too broad to be accurately located. An N–H···Cl interaction is observed in the crystal structure (NH···Cl = 3.109(8) and 3.152(8) Å for the two independent molecules, Table S1). Furthermore, [4H]Cl displays a broad proton resonance at 7.88 ppm (in CDCl3) or 9.79 ppm (in C6D6), which is in the region for an NH group participating in strong hydrogen-bonding interaction.82 Protonation of the bromide complex 1 in C6D6 with HBF4·Et2O also yielded a cationic complex exclusively. The NH resonance of the product ([1H]BF4) was found at 4.44 pm, implying weak hydrogen-bonding interaction. By comparison, the NH resonance of [1H]Br, which forms hydrogen bonds between the NH group and the bromide (NH···Br = 3.331(15) and 3.301(15) Å for the two independent molecules), appears at 8.74 ppm (in C6D6). Protonation of 1 with 1 equiv of HCl is more complicated because of facile Cl–/Br– exchange at the nickel and NH sites. Comparing the NMR data of the cationic complexes described in this study (nine total) suggests that for these cationic species, the 31P chemical shift is primarily determined by what is bound to nickel. This allowed us to determine the ratio of [4H]X/[1H]X to be 2:1, consistent with the notion that nickel forms a stronger bond with a harder donor atom (i.e., Cl as opposed to Br).83,84
Scheme 5. Protonation of Nickel Halide Complexes.
Complex 1 is surprisingly sensitive to water. When treated with 5 equiv of degassed H2O, the solution of 1 in C6D6 exhibited a broad 31P resonance at 66.53 ppm (85%) along with a sharp 31P resonance at 50.15 ppm (15%). Assuming that the former is a weighted average of the chemical shifts of 1 (68.33 ppm) and its protonated form [1H]+ (58.2 ppm), we estimated 15% of 1 being converted to [1H]OH (Scheme 6) or perhaps [1H]OH(H2O)n, in which a more elaborate hydrogen-bonding network is established with water. The resonance at 50.15 ppm is tentatively assigned to an isomer of [1H]OH with a Br–/OH– swap. Isolating this minor species was not possible because the mixture decomposed readily by releasing the free PNP pincer ligand (−1.04 ppm). It is possible that the nickel hydroxide species forms a dimer bridged by the OH groups. In fact, related nickel hydroxide complexes bearing a pincer ligand are known to be unstable.85,86
Scheme 6. Reaction of 1 with H2O.
The reaction of 1 with MeOH (1–10 equiv) was quite similar, forming products consistent with [(iPrPNHP)NiBr]OMe and its isomer [(iPrPNHP)NiOMe]Br as well as the free PNP pincer ligand. The 1H NMR spectrum of the reaction mixture also displayed an emerging triplet at −19.16 ppm (JP–H = 62.0 Hz), suggesting the formation of a nickel hydride species. It is possible that [(iPrPNHP)NiOMe]Br proceeds via β-hydride elimination to yield [(iPrPNHP)NiH]Br.87
Nickel–carbon bonds are susceptible to cleavage by acids.88 Complex 2 specifically may undergo protonation of the phenyl group in parallel with or in competition with protonation of the nitrogen atom. Considering that a coordinating anion such as Cl– is needed to stabilize nickel following phenyl protonation, we used HCl as the acid for the protonation study. Judging from the NMR spectra, the reaction between 2 and HCl (2 M solution in Et2O) in a 1:1 stoichiometry led rapidly and cleanly to [2H]Cl (Scheme 7). There was no evidence suggesting that a neutral or cationic nickel chloride complex was produced. Phenyl protonation would eventually take place if 2 equiv of HCl were initially added to 2; however, this process was shown to be extremely slow at room temperature, converting ∼50% of the newly formed [2H]Cl to [4H]Cl over a period of 3 weeks. Raising the temperature to 70 °C accelerated the second protonation step, resulting in full conversion to [4H]Cl within 24 h. As one might have expected, using a substoichiometric amount of HCl (e.g., 0.5 equiv) generated a mixture of 2 and [2H]Cl. Interestingly, the phosphorus resonances of these two species are significantly broad, presumably due to a rapid interconversion. In an attempt to test if [4H]Cl was generated via elimination of Ph–H from [2H]Cl followed by protonation of the resulting nickel chloride complex 4, pure [2H]Cl was dissolved in C6D6 and CD3CN (for easier detection of benzene) and then heated to 70 °C. In both cases, no Ph–H elimination was observed after monitoring the samples for 3 days.
Scheme 7. Protonation of 2 by HCl.
Treatment of 2 with a weak acid, such as MeOH or H2O, provided a rapidly equilibrated mixture of 2 and a cationic nickel phenyl complex. As illustrated by the NMR titration of 2 with MeOH (Figure 8), >10 equiv of MeOH are required for complete protonation of 2. Unlike the bromide complex 1, the mixture of 2 and H2O (5 equiv) in C6D6 showed no sign of degradation to the free PNP pincer ligand. This can be explained by the fact that the phenyl group is unable to dissociate from nickel (or protonated by H2O). Hydrogen-bonding interaction is critical to the protonation process. Because our reactions were carried out in C6D6, pKa values measured in dimethyl sulfoxide cannot be used to predict the extent of protonation. For instance, indene (pKa = 20.1),89 which is more acidic than MeOH (pKa = 29.0) and H2O (pKa = 31.4)90 but lacks hydrogen-bonding capability, failed to protonate 2.
Figure 8.
31P{1H} NMR spectra of 2 (in C6D6) with various amounts of MeOH added.
Replacing the phenyl group with a more basic methyl group should enhance the reactivity of the Ni–C bond toward acids. Indeed, protonation of complex 3 by HCl (2 M solution in Et2O) was shown to proceed via two different routes (Scheme 8): nitrogen protonation first (route 1) or methyl protonation first (route 2). With a large excess of HCl (>2 equiv), 3 was fully converted to [4H]Cl with a concomitant release of CH4. When 3 was treated with only 1 equiv of HCl, [3H]Cl was obtained as the major product (75%) along with 4 observed as the minor product (25%). Control experiments showed that CH4 elimination from [3H]Cl was not feasible even at 70 °C, ruling out the possibility that 4 was generated from [3H]Cl. A separate NMR experiment established a rapid protonation of [3H]Cl by HCl to give [4H]Cl and CH4. Evidently, [4H]Cl is more acidic than [3H]Cl because the equilibrium in eq 7 heavily favors [3H]Cl and 4 (tested from both directions). Given these results, one might propose that 3 undergoes nitrogen protonation first, followed by methyl protonation to yield [4H]Cl (route 1), which in turn reacts with the remaining 3 to give [3H]Cl and 4. However, this mechanism would require protonation of the [3H]Cl methyl group by HCl and protonation of the 3 nitrogen atom by [4H]Cl to be faster than direct methyl protonation of 3 by HCl, which seems unlikely. It was noted that adding 2 equiv of HCl to 3 in C6D6 caused a color change of the solution from orange to yellow (for [4H]Cl) via a transient green color (for 4). This further supports the viability of route 2 by which methyl protonation precedes nitrogen protonation, although it is still kinetically less competitive than route 1. The protonation study was also carried out using 0.5 equiv of HCl, which yielded a mixture of 3, [3H]Cl, and 4. In contrast to the sharp phosphorous resonance attributed to 4, the spectral lines for 3 and [3H]Cl appeared quite broad, implying a rapid process that allows these two species to interconvert.
 |
7 |
Scheme 8. Two Parallel Pathways for the Protonation of 3 by HCl.

Complex 3 behaves similarly to phenyl complex 2 when reacting with weak acids. Addition of MeOH or H2O to a solution of 3 in C6D6 resulted in partial protonation of 3 to a cationic nickel methyl species. NMR titration experiments indicated that a complete protonation of 3 was accomplished with ∼7 equiv of MeOH (Figure S49), supporting the notion that 3 is more basic than 2. The protonation result is not surprising, given the success of making cationic nickel methyl complexes from in situ generated 3 dissolved in MeOH (Scheme 4). Once again, despite being more acidic than MeOH and H2O, indene exhibited no reactivity toward 3.
Conclusions
In this work, we have shown the propensity of (iPrPNP)NiX (X = Br, Ph, Me) to undergo nitrogen protonation, even in the presence of a weak Brønsted acid, such as water or MeOH. The reaction is primarily driven by the hydrogen-bonding interaction formed between the NH group and the conjugate base. Halides, which could come from the employed Brønsted acid (e.g., HCl) or the starting pincer complex (e.g., (iPrPNP)NiBr), appear to form the strongest interaction with the NH group. Thus, the synthesis of (iPrPNP)NiPh and (iPrPNP)NiMe from (iPrPNP)NiBr is always plagued by the formation of a cationic byproduct bearing N–H···Br bonds (eq 2), likely due to nitrogen protonation by adventitious water, followed by OH–/Br– exchange. Similarly, the degradation of (iPrPNP)NiBr in the presence of water (Scheme 6) or MeOH is initiated by nitrogen protonation and facilitated by the exchange of the nickel-bound Br– with OH– or MeO–. The nitrogen protonation and halide exchange steps can, however, be utilized to make [(iPrPNHP)NiMe]X (X = Cl, Br) from (iPrPNP)NiX and MeLi following MeOH addition (Scheme 4). The Ni–C bond in (iPrPNP)NiPh and (iPrPNP)NiMe becomes labile only when a strong acid, such as HCl, is added, and in the former case, an elevated temperature of 70 °C is required to cleave the Ni–Csp2 bond (Scheme 7). We believe our results have important implications in the understanding of the stability of catalysts containing an RPNHP- or RPNP-type pincer ligand, especially for reactions that either use or generate alcohols and for processes that contain water and/or halides. Our future work in this area will be focused on other PNP pincer systems to examine the generality of our observations. As far as catalytic reactions are concerned, it will also be interesting to see if replacing the nitrogen with a less electronegative donor (e.g., phosphorus) can improve the stability of the catalysts.
Experimental Section
General Comments
Unless otherwise noted, all organometallic/inorganic compounds were prepared and handled under an argon atmosphere using standard Schlenk and inert-atmosphere box techniques. Dry and oxygen-free solvents (THF, CH2Cl2, diethyl ether, toluene, and pentane) were collected from an Innovative Technology solvent purification system and used throughout the experiments. Acetone-d6 was purchased from Cambridge Isotope Laboratories (CIL) and used as received. Benzene-d6 (purchased from CIL) was dried over Na-benzophenone and distilled under an argon atmosphere. Acetonitrile-d3 (purchased from CIL), acetonitrile, and acetone were dried over CaH2 and distilled under an argon atmosphere. Methanol was dried over 4 Å molecular sieves and then degassed by bubbling argon through it for 30 min to 1 h. All other reagents were used as received from commercial sources. iPrPNHP,91,92 (iPrPNP)NiBr (1),11 and [(iPrPNHP)NiBr]Br ([1H]Br)11 were prepared as described in the literature. 1H, 13C{1H}, and 31P{1H} NMR spectra were recorded on a Bruker Avance 400 MHz spectrometer. Chemical shift values in 1H and 13C{1H} NMR spectra were referenced internally to the residual solvent resonances. Assignments were made based on DEPT-135 and 1H–13C HSQC spectra. 31P{1H} NMR spectra were referenced externally to 85% H3PO4 (0 ppm). Infrared spectra were recorded on a Thermo Scientific Nicolet 6700 Fourier transform infrared (FT-IR) spectrometer or a PerkinElmer Spectrum Two FT-IR spectrometer equipped with a smart orbit diamond-attenuated total reflectance (ATR) accessory.
Synthesis of (iPrPNP)NiPh (2), [(iPrPNHP)NiPh]Br ([2H]Br), and (iPrPNP′)NiPh (2′) from (iPrPNP)NiBr (1) and PhMgCl
Under an argon atmosphere, a 2.0 M solution of PhMgCl in THF (500 μL, 1.0 mmol) was added dropwise to a Schlenk flask (flask #1) containing a solution of 1 (443 mg, 1.0 mmol) in 5 mL of THF chilled at 0 °C. The resulting mixture was warmed to room temperature, stirred at that temperature for 1 h, and then filtered into Schlenk flask #2, from which the solvent was removed under vacuum. The residue in flask #2 was treated with 10 mL of pentane, resulting in a suspension, which was filtered into Schlenk flask #3. The filtrate (in flask #3) was dried under vacuum to give 2 as a yellow solid (219 mg, 50% yield). The solid left in flask #2 was rinsed with pentane (collected in Schlenk flask #4) until the pentane wash was colorless. The combined pentane washes in flask #4 were dried under vacuum to yield a yellow solid. Recrystallization of this solid with pentane or acetone at −25 °C yielded orange-yellow rod-shaped crystals of 2′ (20 mg, 5% yield). The solid obtained after exhaustive pentane washes (in flask #2) was dissolved in acetone and then filtered through a pad of Celite to give a pale yellow solution. This solution was dried under vacuum, and the residue was suspended in a minimum amount of toluene. Several drops of acetone were added to the toluene suspension until a clear solution was obtained. The toluene–acetone solution was carefully layered with pentane and kept at −25 °C to yield colorless plate-shaped crystals of [2H]Br (147 mg, 28% yield).
Characterization Data of 2
1H NMR (400 MHz, C6D6, δ): 7.70 (d, 3JH–H = 7.2 Hz, ArH, 2H), 7.11 (t, 3JH–H = 7.4 Hz, ArH, 2H), 6.95 (t, 3JH–H = 7.0 Hz, ArH, 1H), 3.23–3.15 (m, NCH2, 4H), 1.84–1.79 (m, PCH(CH3)2, 4H), 1.76–1.71 (m, PCH2, 4H), 1.07–0.98 (m, PCH(CH3)2, 24H). 13C{1H} NMR (101 MHz, C6D6, δ): 156.09 (t, JP–C = 28.6 Hz, ArC), 140.88 (s, ArC), 125.76 (s, ArC), 120.97 (s, ArC), 59.10 (s, NCH2), 25.06 (t, JP–C = 9.8 Hz, PCH2), 23.33 (t, JP–C = 11.1 Hz, PCH(CH3)2), 18.30 (s, CH3), 17.48 (s, CH3). 31P{1H} NMR (162 MHz, C6D6, δ): 64.40 (s). Anal. Calcd for C22H41NP2Ni: C, 60.03; H, 9.39; N, 3.18. Found: C, 59.75; H, 9.47; N, 3.18.
Characterization Data of [2H]Br
1H NMR (400 MHz, CD3CN, δ): 7.33 (d, 3JH–H = 7.6 Hz, ArH, 2H), 6.92 (t, 3JH–H = 7.4 Hz, ArH, 2H), 6.75 (t, 3JH–H = 7.2 Hz, ArH, 1H), 5.35 (t, broad, 3JH–H = 11.0 Hz, NH, 1H), 3.18–3.03 (m, NCH2, 2H), 2.45–2.34 (m, NCH2, 2H), 2.22–2.15 (m, PCH(CH3)2, 2H), 2.10–2.00 (m, PCH(CH3)2 + PCH2, 6H), 1.28–1.23 (m, PCH(CH3)2, 12H), 1.06–0.95 (m, PCH(CH3)2, 12H). 13C{1H} NMR (101 MHz, CD3CN, δ): 144.64 (t, JP–C = 29.4 Hz, ArC), 138.67 (t, JP–C = 3.5 Hz, ArC), 127.32 (s, ArC), 122.78 (s, ArC), 51.99 (t, JP–C = 4.5 Hz, NCH2), 23.75 (t, JP–C = 11.9 Hz, PCH(CH3)2), 22.98 (t, JP–C = 9.4 Hz, PCH2), 22.67 (t, JP–C = 13.1 Hz, PCH(CH3)2), 18.43 (s, CH3), 18.33 (s, CH3), 17.68 (s, CH3), 17.27 (s, CH3). 31P{1H} NMR (162 MHz, CD3CN, δ): 48.41 (s). 31P{1H} NMR (162 MHz, C6D6, δ): 48.44 (s). Selected data from ATR-IR (solid): νN–H = 3351 cm–1 (broad). Anal. Calcd for C22H42NP2BrNi: C, 50.71; H, 8.12; N, 2.69. Found: C, 50.84; H, 7.93; N, 2.50.
Characterization Data of 2′
1H NMR (400 MHz, C6D6, δ): 7.71 (d, 3JH–H = 7.2 Hz, ArH, 2H), 7.42 (d, J = 42.8 Hz, CH, 1H), 7.14 (t, 3JH–H = 7.2 Hz, ArH, 2H), 6.96 (t, 3JH–H = 7.2 Hz, ArH, 1H), 3.41 (br, CH, 1H), 3.14 (dt, 3JP–H = 18.4 Hz, 3JH–H = 6.9 Hz, NCH2, 2H), 2.09–1.99 (m, PCH(CH3)2, 2H), 1.76–1.66 (m, PCH(CH3)2, 2H), 1.30 (dt, 2JP–H = 9.2 Hz, 3JH–H = 4.0 Hz, PCH2, 2H), 1.23 (dd, 3JP–H = 13.8 Hz, 3JH–H = 6.9 Hz, CH3, 6H), 1.14 (dd, 3JP–H = 15.4 Hz, 3JH–H = 7.5 Hz, CH3, 6H), 0.95–0.89 (m, CH3, 12H). 13C{1H} NMR (101 MHz, C6D6, δ): 167.67 (dd, JP–C = 22.4 and 4.8 Hz, CH), 153.92 (dd, JP–C = 30.4 and 29.2 Hz, ArCipso), 139.59 (t, JP–C = 3.2 Hz, ArC), 126.14 (t, JP–C = 2.1 Hz, ArC), 121.22 (t, JP–C = 2.0 Hz, ArC), 65.80 (d, JP–C = 40.8 Hz, CH), 55.13 (dd, JP–C = 7.4 and 2.6 Hz, NCH2), 24.28 (d, JP–C = 18.7 Hz, PCH2), 23.43 (dd, JP–C = 25.8 and 1.2 Hz, PCH(CH3)2), 22.79 (dd, JP–C = 19.6 and 1.9 Hz, PCH(CH3)2), 18.25 (d, JP–C = 3.7 Hz, CH3), 18.06 (d, JP–C = 4.2 Hz, CH3), 17.71 (d, JP–C = 0.9 Hz, CH3), 17.36 (s, CH3). 31P{1H} NMR (162 MHz, C6D6, δ): 58.97 (AB, JAB = 259.7 Hz), 48.51 (AB, JAB = 259.7 Hz). Anal. Calcd for C22H39NP2Ni: C, 60.30; H, 8.97; N, 3.20. Found: C, 61.20; H, 9.02; N, 2.79.
Synthesis of (iPrPNP′)NiBr (1′)
Under an argon atmosphere, 1 (111 mg, 0.25 mmol) and 1,4-benzoquinone (27 mg, 0.25 mmol) were mixed with 3 mL of toluene at room temperature and stirred for 24 h. The resulting black suspension was filtered, and the solvent in the filtrate was removed under vacuum. The product was extracted into pentane (5 mL × 3). Green crystals of 1′ were obtained from a concentrated pentane solution kept at −25 °C (8.5 mg, 8% yield). 1H NMR (400 MHz, C6D6, δ): 6.76 (ddd, J = 45.1, 4.9, and 1.5 Hz, CH, 1H), 3.32–3.30 (m, CH, 1H), 2.79 (dt, 3JP–H = 21.2 Hz, 3JH–H = 6.8 Hz, NCH2, 2H), 2.25–2.15 (m, PCH(CH3)2, 2H), 2.00–1.90 (m, PCH(CH3)2, 2H), 1.64 (dd, 3JP–H = 16.0 Hz, 3JH–H = 6.8 Hz, PCH(CH3)2, 6H), 1.41 (dd, 3JP–H = 15.2 Hz, 3JH–H = 7.2 Hz, PCH(CH3)2, 6H), 1.30 (dd, 3JP–H = 13.6 Hz, 3JH–H = 7.2 Hz, PCH(CH3)2, 6H), 0.94–1.02 (m, PCH(CH3)2 + PCH2, 8H). 13C{1H} NMR (101 MHz, C6D6, δ): 168.50 (dd, JP–C = 22.9 and 4.4 Hz, CH), 68.25 (d, JP–C = 40.9 Hz, CH), 57.44 (dd, JP–C = 8.7 and 2.0 Hz, NCH2), 25.15 (dd, JP–C = 25.2 and 2.5 Hz, PCH(CH3)2), 23.86 (dd, JP–C = 19.2 and 2.4 Hz, PCH(CH3)2), 22.23 (d, JP–C = 19.3 Hz, PCH2), 19.16 (d, JP–C = 3.9 Hz, CH3), 19.02 (d, JP–C = 4.2 Hz, CH3), 18.05 (s, CH3), 17.78 (s, CH3). 31P{1H} NMR (162 MHz, C6D6, δ): 63.58 (AB, JAB = 299.2 Hz), 57.87 (AB, JAB = 299.2 Hz). Anal. Calcd for C16H34NP2BrNi: C, 43.58; H, 7.77; N, 3.18. Found: C, 43.82; H, 7.96; N, 3.12.
Independent Synthesis of (iPrPNP′)NiPh (2′) from (iPrPNP′)NiBr (1′)
Under an argon atmosphere, a 2.0 M solution of PhMgCl in THF (50 μL, 0.10 mmol) was added dropwise to a solution of 1′ (37 mg, 0.084 mmol) in 5 mL of THF chilled at 0 °C. The resulting mixture was stirred at room temperature for 1 h, after which the solvent was removed under vacuum. The residue was dissolved in pentane and filtered through a pad of Celite. The filtrate was pumped to dryness, giving the desired product as a yellow solid (17 mg, 46%).
Synthesis of (TMEDA)Ni(Ph)I
This compound was prepared following a procedure similar to the one used for the synthesis of (TMEDA)Ni(Ph)Cl.93 Under an argon atmosphere, a solution of TMEDA (280 μL, 1.9 mmol) in iodobenzene (2.2 mL, 20 mmol) was added to a Schlenk flask containing Ni(COD)2 (420 mg, 1.5 mmol). The resulting mixture was stirred at room temperature for 6 h, followed by layering with pentane (10 mL). The mixture was then allowed to stand at room temperature overnight, resulting in a precipitate, which was collected by filtration, washed with pentane (10 mL × 3), and dried under vacuum. The product was isolated as a pink solid (500 mg, 88% yield). 1H NMR (400 MHz, THF-d8, δ): 7.47 (br, ArH, 2H), 6.62 (br, ArH, 2H), 6.43 (br, ArH, 1H), 2.58–2.13 (m, CH3 + CH2, 16H). 13C{1H} NMR (101 MHz, THF-d8, δ): 146.32 (br, ArC), 139.63 (s, ArC), 124.65 (s, ArC), 121.05 (s, ArC), 61.11 (br, NCH2), 57.39 (br, NCH2), 49.51 (br, CH3), 48.68 (br, CH3). Anal. Calcd for C12H21N2INi: C, 38.04; H, 5.59; N, 7.39. Found: C, 37.25; H, 5.68; N, 7.31.
Synthesis of [(iPrPNHP)NiPh]PF6 ([2H]PF6)
Under an argon atmosphere, a solution of iPrPNHP (159 mg, 0.52 mmol) in 5 mL of THF was added dropwise to (TMEDA)Ni(Ph)I (197 mg, 0.52 mmol) dissolved in 20 mL of THF. The resulting mixture was stirred at room temperature for 30 min, giving a yellow precipitate. The solid was collected by filtration, washed with diethyl ether (5 mL × 3), and dried under vacuum. The isolated complex (268 mg, 0.47 mmol) was dissolved in 20 mL of CH2Cl2, followed by dropwise addition of a solution of AgPF6 (143 mg, 0.56 mmol) in 10 mL of CH2Cl2. The resulting suspension was stirred at room temperature for 15 min and then filtered, and the solid was washed with diethyl ether (5 mL × 2). The filtrate combined with the ether wash was concentrated under vacuum to ∼3 mL, and the product precipitated after adding diethyl ether and pentane (5 mL each). The precipitate was collected by filtration, washed with diethyl ether (5 mL × 2), and dried under vacuum. The desired product was isolated as a yellow solid (238 mg, 78% yield over two steps). 1H NMR (400 MHz, CD3CN, δ): 7.31 (d, 3JH–H = 7.6 Hz, ArH, 2H), 6.96 (t, 3JH–H = 7.4 Hz, ArH, 2H), 6.79 (t, 3JH–H = 7.4 Hz, ArH, 1H), 3.62 (t, broad, 3JH–H = 13.8 Hz, NH, 1H), 3.22–3.09 (m, NCH2, 2H), 2.58–2.45 (m, NCH2, 2H), 2.30–2.18 (m, PCH(CH3)2, 2H), 2.18–2.06 (m, PCH(CH3)2 + PCH2, 4H), 1.97–1.88 (m, PCH2, 2H), 1.31–1.21 (m, PCH(CH3)2, 12H), 1.05–0.98 (m, PCH(CH3)2, 12H). 1H NMR (400 MHz, acetone-d6, δ): 7.38 (d, 3JH–H = 7.2 Hz, ArH, 2H), 6.96 (t, 3JH–H = 7.4 Hz, ArH, 2H), 6.77 (t, 3JH–H = 7.4 Hz, ArH, 1H), 4.42 (t, broad, 3JH–H = 11.8 Hz, NH, 1H), 3.41–3.27 (m, NCH2, 2H), 2.77–2.66 (m, NCH2, 2H), 2.35–2.23 (m, PCH(CH3)2 + PCH2, 4H), 2.20–2.15 (m, PCH(CH3)2, 2H), 2.10–2.02 (m, PCH2, 2H), 1.38–1.33 (m, PCH(CH3)2, 6H), 1.32–1.26 (m, PCH(CH3)2, 6H), 1.11–1.04 (m, PCH(CH3)2, 12H). 13C{1H} NMR (101 MHz, acetone-d6, δ): 143.42 (t, JP–C = 29.3 Hz, ArC), 138.59–138.53 (m, ArC), 138.40–138.33 (m, ArC), 127.64 (s, ArC), 123.16 (t, JP–C = 1.7 Hz, ArC), 52.52 (t, JP–C = 4.5 Hz, NCH2), 23.93 (t, JP–C = 12.0 Hz, PCH(CH3)2), 22.95 (t, JP–C = 9.0 Hz, PCH2), 22.50 (t, JP–C = 13.0 Hz, PCH(CH3)2), 18.38 (s, CH3), 18.01 (s, CH3), 17.34 (s, CH3), 17.31 (s, CH3). 31P{1H} NMR (162 MHz, acetone-d6, δ): 46.42 (s, PiPr2), −143.72 (sept, 1JP–F = 708.2 Hz, PF6). 31P{1H} NMR (162 MHz, CD3CN, δ): 47.64 (s, PiPr2), −142.84 (sept, 1JP–F = 706.6 Hz, PF6). Selected data from ATR-IR (solid): νN–H = 3249 cm–1. Anal. Calcd for C22H42NP3F6Ni: C, 45.08; H, 7.22; N, 2.39. Found: C, 45.02; H, 7.18; N, 2.41.
Independent Synthesis of (iPrPNP)NiPh (2) from [(iPrPNHP)NiPh]Br ([2H]Br) or [(iPrPNHP)NiPh]PF6 ([2H]PF6)
From [2H]Br: Under an argon atmosphere, a solution of KOtBu (5.6 mg, 0.050 mmol) in 3 mL of THF kept at −78 °C was added to [2H]Br (26 mg, 0.050 mmol) suspended in 5 mL of THF, which was also chilled at −78 °C. The resulting mixture was stirred at that temperature for 15 min. The volatiles were removed under vacuum and the product was extracted with pentane (5 mL × 3). Complex 2 was isolated as a yellow solid after removing pentane under vacuum (14.8 mg, 67% yield). From [2H]PF6: Under an argon atmosphere, a solution of KOtBu (16 mg, 0.14 mmol) in 5 mL of THF was added to [2H]PF6 (82 mg, 0.14 mmol) suspended in 10 mL of THF, which was chilled at −78 °C. The resulting suspension was stirred at that temperature for 15 min. The volatiles were removed under vacuum and the product was extracted with pentane (5 mL × 3). Complex 2 was isolated as a yellow solid after removing pentane under vacuum (29 mg, 47% yield).
Synthesis of [(iPrPNHP)NiPh]BPh4 ([2H]BPh4)
Under an argon atmosphere, a solution of NaBPh4 (82 mg, 0.24 mmol) in 2 mL of CH3OH was added dropwise to [2H]Br (100 mg, 0.19 mmol) dissolved in 2 mL of CH3OH. The mixture was stirred at room temperature for 30 min, giving a yellow precipitate. The solid was filtered off, washed with diethyl ether (5 mL × 2), and dried under vacuum. The desired product was isolated as a yellow solid (87 mg, 60% yield). 1H NMR (400 MHz, CD3CN, δ): 7.32–7.28 (m, ArH, 10H), 7.00 (t, 3JH–H = 7.4 Hz, ArH, 8H), 6.94 (t, 3JH–H = 7.2 Hz, ArH, 2H), 6.85 (t, 3JH–H = 7.0 Hz, ArH, 4H), 6.77 (t, 3JH–H = 7.2 Hz, ArH, 1H), 3.77 (t, broad, 3JH–H = 10.4 Hz, NH, 1H), 3.17–3.03 (m, NCH2, 2H), 2.52–2.42 (m, NCH2, 2H), 2.24–2.16 (m, PCH(CH3)2, 2H), 2.13–2.01 (m, PCH(CH3)2 + PCH2, 4H), 1.92–1.83 (m, PCH2, 2H), 1.29–1.19 (m, PCH(CH3)2, 12H), 1.03–0.96 (m, PCH(CH3)2, 12H). 13C{1H} NMR (101 MHz, CD3CN, δ): 164.68 (q, 1JB–C = 49.5 Hz, ArC), 143.52 (t, JP–C = 29.5 Hz, ArC), 138.63 (t, JP–C = 2.2 Hz, ArC), 138.44 (t, JP–C = 3.5 Hz, ArC), 136.62 (q, JB–C = 1.2 Hz, ArC), 127.54 (br, ArC), 126.50 (q, JB–C = 2.7 Hz, ArC), 123.05 (t, JP–C = 2.0 Hz, ArC), 122.67 (s, ArC), 52.13 (t, JP–C = 4.5 Hz, NCH2), 23.82 (t, JP–C = 12.1 Hz, PCH(CH3)2), 22.86 (t, JP–C = 9.0 Hz, PCH2), 22.47 (t, JP–C = 13.1 Hz, PCH(CH3)2), 18.27 (s, CH3), 17.98 (s, CH3), 17.30 (s, CH3), 17.23 (s, CH3). 31P{1H} NMR (162 MHz, CD3CN, δ): 47.72 (s). Selected data from ATR-IR (solid): νN–H = 3181 cm–1. Anal. Calcd for C46H62NBP2Ni·1.5C4H8O (crystals grown from THF–pentane): C, 71.90; H, 8.59; N, 1.61. Found: C, 71.28; H, 8.25; N, 1.87.
Synthesis of [(iPrPNHP)NiPh]Cl ([2H]Cl)
At room temperature under an argon atmosphere, a 2.0 M solution of HCl in diethyl ether (75 μL, 0.15 mmol) was added dropwise to 2 (54 mg, 0.12 mmol) dissolved in 5 mL of diethyl ether. The resulting suspension was stirred for 30 min. The volatiles were then removed under vacuum, and the residue was washed with pentane (5 mL × 3). After drying under vacuum, the product was isolated as a greenish yellow solid (57 mg, 97% yield). 1H NMR (400 MHz, CD3CN, δ): 7.34 (d, 3JH–H = 6.0 Hz, ArH, 2H), 6.91 (t, 3JH–H = 7.4 Hz, ArH, 2H), 6.74 (t, 3JH–H = 7.0 Hz, ArH, 1H), 6.25 (t, broad, 3JH–H = 10.0 Hz, NH, 1H), 3.14–3.04 (m, NCH2, 2H), 2.46–2.36 (m, NCH2, 2H), 2.21–1.97 (m, PCH(CH3)2 + PCH2, 8H), 1.30–1.22 (m, PCH(CH3)2, 12H), 1.08–1.02 (m, PCH(CH3)2, 6H), 1.01–0.95 (m, PCH(CH3)2, 6H). 13C{1H} NMR (101 MHz, CD3CN, δ): 145.18 (t, JP–C = 29.6 Hz, ArC), 138.82 (t, JP–C = 3.0 Hz, ArC), 127.31 (s, ArC), 122.76 (s, ArC), 52.13 (t, JP–C = 4.7 Hz, NCH2), 23.81 (t, JP–C = 11.9 Hz, PCH(CH3)2), 23.07 (t, JP–C = 9.2 Hz, PCH2), 22.72 (t, JP–C = 13.0 Hz, PCH(CH3)2), 18.55 (s, CH3), 18.41 (s, CH3), 17.83 (s, CH3), 17.34 (s, CH3). 31P{1H} NMR (162 MHz, CD3CN, δ): 48.76 (s). 31P{1H} NMR (162 MHz, C6D6, δ): 48.92 (s). Selected data from ATR-IR (solid): νN–H = 3360 cm–1 (broad).
Synthesis of (iPrPNP)NiMe (3)
At room temperature under an argon atmosphere, a 1.6 M solution of MeLi in diethyl ether (188 μL, 0.30 mmol) was added dropwise to a solution of 1 (111 mg, 0.25 mmol) in 5 mL of diethyl ether, resulting in a color change from dark green to orange. After the addition, the reaction mixture was stirred at room temperature for another 5 min. The volatiles were removed under vacuum, and the residue was extracted with pentane (5 mL × 2). The pentane extracts were concentrated under vacuum, giving 3 as an orange solid (72 mg, 76% yield). The 31P{1H} NMR spectrum suggested that the product was contaminated with a small amount (∼5%) of the dehydrogenated species, (iPrPNP′)NiMe (3′). 1H NMR (400 MHz, C6D6, δ): 3.23–3.15 (m, NCH2, 4H), 1.94–1.87 (m, PCH(CH3)2, 4H), 1.75–1.71 (m, PCH2, 4H), 1.27–1.21 (m, PCH(CH3)2, 12H), 1.11–1.06 (m, PCH(CH3)2, 12H), −0.59 (t, 3JP–H = 8.6 Hz, NiCH3, 3H). 13C{1H} NMR (101 MHz, C6D6, δ): 58.75 (t, broad, NCH2), 25.19 (t, JP–C = 9.8 Hz, PCH2), 23.64 (t, JP–C = 10.3 Hz, PCH(CH3)2), 19.25 (t, JP–C = 2.1 Hz, PCH(CH3)2), 17.85 (s, PCH(CH3)2), −26.32 (t, 2JP–C = 25.6 Hz, NiCH3). 31P{1H} NMR (162 MHz, C6D6, δ): 68.81 (s). Elementary analyses were unsatisfactory despite repeated trials. This is likely due to the extremely high sensitivity of this compound toward even a trace amount of O2. Coincidently, the data matched those of 3 with two oxygen atoms incorporated. Anal. Calcd for C17H39NO2P2Ni (3 + O2): C, 49.78; H, 9.58; N, 3.42. Found: C, 49.62; H, 9.86; N, 3.42.
Characterization Data of 3′
Selected 1H NMR data (400 MHz, C6D6, δ): 7.47 (dd, J = 41.8 and 5.0 Hz, CH, 1H), −0.35 (t, 3JP–H = 8.6 Hz, NiCH3, 3H). 31P{1H} NMR (162 MHz, C6D6, δ): 62.71 (AB, JAB = 275.4 Hz), 49.86 (AB, JAB = 275.4 Hz).
Synthesis of [(iPrPNHP)NiMe]Br ([3H]Br)
At room temperature under an argon atmosphere, a 1.6 M solution of MeLi in diethyl ether (188 μL, 0.30 mmol) was added dropwise to a solution of 1 (111 mg, 0.25 mmol) in 5 mL of diethyl ether. After addition, the mixture was stirred at room temperature for another 5 min. The volatiles were removed under vacuum, and the residue was treated with 3 mL of methanol, which resulted in the formation of a small amount of black particles. The reaction mixture was stirred at room temperature for 15 min, and methanol was removed under vacuum. The residue was suspended in 20 mL of toluene (or 3 mL acetonitrile) and filtered through a pad of Celite to remove the black particles. The filtrate was pumped to dryness, washed with pentane (5 mL × 3), and dried under vacuum. The desired product was isolated as a yellow solid (60 mg, 52% yield). 1H NMR (400 MHz, CD3CN, δ): 4.92 (t, broad, NH, 1H), 3.11–2.94 (m, NCH2, 2H), 2.34–2.20 (m, PCH(CH3)2 + NCH2, 6H), 1.98–1.93 (m, PCH2, 4H), 1.36–1.24 (m, PCH(CH3)2, 24H), −0.57 (t, 3JP–H = 9.0 Hz, NiCH3, 3H). 13C{1H} NMR (101 MHz, CD3CN, δ): 51.87 (t, JP–C = 4.7 Hz, NCH2), 25.11 (t, JP–C = 11.3 Hz, PCH(CH3)2), 23.14 (t, JP–C = 12.4 Hz, PCH(CH3)2), 23.00 (t, JP–C = 9.3 Hz, PCH2), 19.44 (t, JP–C = 1.5 Hz, PCH(CH3)2), 19.34 (t, JP–C = 2.2 Hz, PCH(CH3)2), 18.43 (s, PCH(CH3)2), 17.91 (s, PCH(CH3)2), −24.04 (t, JP–C = 23.8 Hz, NiCH3). 31P{1H} NMR (162 MHz, CD3CN, δ): 51.69 (s). 31P{1H} NMR (162 MHz, C6D6, δ): 51.86 (s). Selected data from ATR-IR (solid): νN–H = 3399 cm–1 (broad). Anal. Calcd for C17H40NP2BrNi: C, 44.48; H, 8.78; N, 3.05. Found: C, 44.58; H, 8.71; N, 3.07.
Synthesis of (iPrPNP)NiCl (4)
Under an argon atmosphere, a 1.6 M solution of n-BuLi in hexanes (750 μL, 1.2 mmol) was added dropwise to a chilled (−78 °C) solution of iPrPNHP (305 mg, 1.0 mmol) in 5 mL of THF. The reaction mixture was stirred first at −78 °C for 30 min and then at room temperature for 1.5 h. In a separate flask, a mixture of NiCl2 (194 mg, 1.5 mmol) and 15 mL of THF was stirred at room temperature for 2 h and then cooled to −78 °C. The lithiated ligand iPrPNP was slowly added while the temperature was maintained at −78 °C. The resulting suspension was stirred at −78 °C for 30 min and then at room temperature for 16 h. The volatiles were removed under vacuum, and the residue was treated with 20 mL of pentane and then filtered through a pad of Celite. The filtrate was concentrated under vacuum and recrystallized at −25 °C. The desired product was isolated as dark green crystals (73 mg, 18% yield). The 31P{1H} NMR spectrum suggested that the product was contaminated with a small amount (∼2%) of the dehydrogenated species, (iPrPNP′)NiCl (4′). 1H NMR (400 MHz, C6D6, δ): 2.59–2.49 (m, NCH2, 4H), 2.10–2.01 (m, PCH(CH3)2, 4H), 1.55–1.50 (m, PCH(CH3)2, 12H), 1.38–1.33 (m, PCH2, 4H), 1.16–1.11 (m, PCH(CH3)2, 12H). 13C{1H} NMR (101 MHz, C6D6, δ): 60.70 (t, JP–C = 6.5 Hz, NCH2), 23.59 (t, JP–C = 10.0 Hz, PCH(CH3)2), 22.03 (t, JP–C = 10.7 Hz, PCH2), 19.17 (t, JP–C = 1.9 Hz, CH3), 17.76 (s, CH3). 31P{1H} NMR (162 MHz, C6D6, δ): 66.09 (s). Anal. Calcd for C16H36NP2ClNi: C, 48.22; H, 9.10; N, 3.51. Found: C, 48.14; H, 9.30; N, 3.47.
Characterization Data of 4′
Selected 1H NMR data (400 MHz, C6D6, δ): 6.72 (ddd, J = 45.2, 5.2, and 1.2 Hz, CH, 1H), 3.28 (t, J = 3.8 Hz, CH, 1H), 2.76 (dt, 3JP–H = 21.2 Hz, 3JH–H = 6.8 Hz, NCH2, 2H). 31P{1H} NMR (162 MHz, C6D6, δ): 60.87 (AB, JAB = 307.8 Hz), 55.02 (AB, JAB = 307.8 Hz).
Synthesis of [(iPrPNHP)NiMe]Cl ([3H]Cl)
At room temperature under an argon atmosphere, a 1.6 M solution of MeLi in diethyl ether (188 μL, 0.30 mmol) was added dropwise to a solution of 4 (100 mg, 0.25 mmol) in 5 mL of diethyl ether. After addition, the mixture was stirred at room temperature for 5 min. The volatiles were removed under vacuum, and the residue was treated with 3 mL of methanol, which resulted in the formation of a small amount of black particles. The reaction mixture was stirred for 15 min, and methanol was removed under vacuum. The resulting solid was treated with 3 mL of acetonitrile and filtered through a pad of Celite to remove the black particles. The filtrate was pumped to dryness, and the residue was washed with pentane (5 mL × 3) and then dried under vacuum. The product was isolated as a yellow solid (75 mg, 72% yield). 1H NMR (400 MHz, CD3CN, δ): 5.61 (br, NH, 1H), 3.06–2.96 (m, NCH2, 2H), 2.30–2.21 (m, PCH(CH3)2 + NCH2, 6H), 1.37–1.25 (m, PCH2 + PCH(CH3)2, 28H), −0.58 (t, 3JP–H = 8.8 Hz, NiCH3, 3H). 13C{1H} NMR (101 MHz, CD3CN, δ): 51.91 (t, JP–C = 4.7 Hz, NCH2), 25.03 (t, JP–C = 11.2 Hz, PCH(CH3)2), 23.09 (t, JP–C = 12.4 Hz, PCH(CH3)2), 22.96 (t, JP–C = 9.8 Hz, PCH2), 19.41 (br, PCH(CH3)2), 19.27 (t, JP–C = 2.4 Hz, PCH(CH3)2), 18.37 (s, PCH(CH3)2), 17.87 (s, PCH(CH3)2), −24.27 (t, JP–C = 23.9 Hz, NiCH3). 31P{1H} NMR (162 MHz, CD3CN, δ): 51.85 (s). 31P{1H} NMR (162 MHz, C6D6, δ): 51.97 (s). Selected data from ATR-IR (solid): νN–H = 3393 cm–1 (broad). Anal. Calcd for C17H40NP2ClNi: C, 49.25; H, 9.72; N, 3.38. Found: C, 48.04; H, 9.59; N, 3.31.
Synthesis of [(iPrPNHP)NiMe]BPh4 ([3H]BPh4)
At room temperature under an argon atmosphere, a 1.6 M solution of MeLi in diethyl ether (188 μL, 0.30 mmol) was added dropwise to a solution of 1 (111 mg, 0.25 mmol) in 5 mL of diethyl ether. After addition, the mixture was stirred at room temperature for another 5 min. The volatiles were removed under vacuum, and the residue was treated with 3 mL of methanol, which resulted in the formation of a small amount of black particles. A solution of NaBPh4 (103 mg, 0.30 mmol) in 3 mL of methanol was then added dropwise, and the resulting suspension was stirred at room temperature for 30 min, after which methanol was removed under vacuum. The residue was suspended in 3 mL of acetonitrile and filtered through a pad of Celite to remove the black particles. The filtrate was pumped to dryness, washed with pentane (5 mL × 3), and dried under vacuum. The desired product was isolated as a yellow solid (115 mg, 66% yield). 1H NMR (400 MHz, CD3CN, δ): 7.29–7.25 (br, ArH, 8H), 7.00 (t, 3JH–H = 7.4 Hz, ArH, 8H), 6.84 (t, 3JH–H = 7.2 Hz, ArH, 4H), 3.57 (t, broad, 3JH–H = 11.6 Hz, NH, 1H), 3.08–3.00 (m, NCH2, 2H), 2.37–2.19 (m, PCH(CH3)2 + NCH2, 6H), 2.03–1.98 (m, PCH2, 2H), 1.84–1.75 (m, PCH2, 2H), 1.34–1.22 (m, PCH(CH3)2, 24H), −0.53 (t, 3JP–H = 8.8 Hz, NiCH3, 3H). 13C{1H} NMR (101 MHz, CD3CN, δ): 164.72 (q, 1JB–C = 49.6 Hz, ArC), 136.66 (br, ArC), 126.52 (q, JB–C = 2.8 Hz, ArC), 122.69 (s, ArC), 51.89 (t, JP–C = 4.5 Hz, NCH2), 25.09 (t, JP–C = 11.3 Hz, PCH(CH3)2), 22.93 (t, JP–C = 12.2 Hz, PCH(CH3)2), 22.88 (t, JP–C = 9.1 Hz, PCH2), 19.30 (br, PCH(CH3)2), 19.07 (t, JP–C = 1.9 Hz, PCH(CH3)2), 18.36 (s, PCH(CH3)2), 17.64 (s, PCH(CH3)2), −23.73 (t, JP–C = 23.7 Hz, NiCH3). 31P{1H} NMR (162 MHz, CD3CN, δ): 51.24 (s). Selected data from ATR-IR (solid): νN–H = 3189 cm–1. Anal. Calcd for C41H60NBP2Ni: C, 70.51; H, 8.66; N, 2.01. Found: C, 70.70; H, 8.80; N, 2.04.
Synthesis of [(iPrPNHP)NiCl]Cl ([4H]Cl)
Under an argon atmosphere, a solution of the ligand iPrPNHP (380 mg, 1.25 mmol) in 5 mL of THF was added to NiCl2 (161 mg, 1.24 mmol) suspended in 10 mL of THF. The resulting mixture was stirred at room temperature for 24 h. The solvent was then removed under vacuum, and the residue was extracted with CH2Cl2 (10 mL × 3). The combined CH2Cl2 fractions were concentrated under vacuum, layered with diethyl ether, and kept at −25 °C for crystallization. The product was isolated as orange crystals (73 mg, 14% yield). 1H NMR (400 MHz, CDCl3, δ): 7.88 (br, NH, 1H), 3.06 (tt, J = 19.2 and 3.9 Hz, NCH2, 2H), 2.41–2.17 (m, PCH(CH3)2 + PCH2 + NCH2, 8H), 1.69–1.60 (m, PCH(CH3)2 + PCH2, 8H), 1.58–1.54 (m, PCH(CH3)2, 6H), 1.54–1.46 (m, PCH(CH3)2, 6H), 1.44–1.39 (m, PCH(CH3)2, 6H). 13C{1H} NMR (101 MHz, CDCl3, δ): 54.97 (t, JP–C = 5.0 Hz, NCH2), 24.34 (t, JP–C = 11.2 Hz, PCH(CH3)2), 23.73 (t, JP–C = 12.6 Hz, PCH(CH3)2), 20.53 (t, JP–C = 9.3 Hz, PCH2), 19.45 (s, CH3), 18.92 (s, CH3), 18.06 (s, CH3), 17.95 (s, CH3). 31P{1H} NMR (162 MHz, CDCl3, δ): 55.25 (s). 31P{1H} NMR (162 MHz, C6D6, δ): 55.40 (s). For both solid and solution samples, the νN–H band was absent from the IR spectra. Anal. Calcd for C16H37NP2Cl2Ni: C, 44.18; H, 8.57; N, 3.22. Found: C, 44.18; H, 8.57; N, 3.24.
Protonation of Nickel PNP Pincer Complexes with an Acid
Under an argon atmosphere, a neutral nickel pincer complex (1–4, 0.010 or 0.020 mmol) was dissolved in ∼0.5 mL of C6D6 and placed in a J. Young NMR tube. To the solution was added an appropriate amount of acid (HCl as a 2.0 M solution in Et2O, HBF4·Et2O, H2O, or MeOH), as described in the Results and Discussion section. The progress of the reaction was monitored by 1H and 31P{1H} NMR spectroscopy.
Acknowledgments
The authors acknowledge the National Science Foundation (CHE-1464734) and the University of Cincinnati (University Research Council Graduate Student Research Fellowship to N.P.N.W.) for support of this research. Crystallographic data were collected on a Bruker Apex-II CCD diffractometer (funded by NSF-MRI grant CHE-0215950), a Bruker D8 Venture Photon-II diffractometer (funded by NSF-MRI grant CHE-1625737), or through the SCrALS (Service Crystallography at Advanced Light Source) Program at Beamline 11.3.1 at the Advanced Light Source (ALS), Lawrence Berkeley National Laboratory (supported by the U.S. Department of Energy, Office of Basic Energy Sciences, under contract No. DE-AC02- 05CH11231).
Supporting Information Available
The Supporting Information is available free of charge on the ACS Publications website at DOI: 10.1021/acsomega.8b00777.
Complete characterization data, including structural parameters of the compounds characterized by X-ray diffraction (PDF)
X-ray crystallographic information for 1′, [1H]Br, 2, 2′, [2H]Br, [2H]PF6, [2H]BPh4·3/2THF, [2H]2[NiCl4]·4C6D6·1/2H2O, 3, [3H]Cl, [3H]Br, [3H]BPh4, [4H]Cl, and (TMEDA)NiPhI (CIF)
The authors declare no competing financial interest.
Supplementary Material
References
- Morris R. H. Exploiting Metal–Ligand Bifunctional Reactions in the Design of Iron Asymmetric Hydrogenation Catalysts. Acc. Chem. Res. 2015, 48, 1494–1502. 10.1021/acs.accounts.5b00045. [DOI] [PubMed] [Google Scholar]
- Zell T.; Milstein D. Hydrogenation and Dehydrogenation Iron Pincer Catalysts Capable of Metal–Ligand Cooperation by Aromatization/Dearomatization. Acc. Chem. Res. 2015, 48, 1979–1994. 10.1021/acs.accounts.5b00027. [DOI] [PubMed] [Google Scholar]
- Chakraborty S.; Bhattacharya P.; Dai H.; Guan H. Nickel and Iron Pincer Complexes as Catalysts for the Reduction of Carbonyl Compounds. Acc. Chem. Res. 2015, 48, 1995–2003. 10.1021/acs.accounts.5b00055. [DOI] [PubMed] [Google Scholar]
- Li Y.-Y.; Yu S.-L.; Shen W.-Y.; Gao J.-X. Iron-, Cobalt-, and Nickel-Catalyzed Asymmetric Transfer Hydrogenation and Asymmetric Hydrogenation of Ketones. Acc. Chem. Res. 2015, 48, 2587–2598. 10.1021/acs.accounts.5b00043. [DOI] [PubMed] [Google Scholar]
- Werkmeister S.; Neumann J.; Junge K.; Beller M. Pincer-Type Complexes for Catalytic (De)Hydrogenation and Transfer (De)Hydrogenation Reactions: Recent Progress. Chem. – Eur. J. 2015, 21, 12226–12250. 10.1002/chem.201500937. [DOI] [PubMed] [Google Scholar]
- Bernskoetter W. H.; Hazari N. Reversible Hydrogenation of Carbon Dioxide to Formic Acid and Methanol: Lewis Acid Enhancement of Base Metal Catalysts. Acc. Chem. Res. 2017, 50, 1049–1058. 10.1021/acs.accounts.7b00039. [DOI] [PubMed] [Google Scholar]
- Garbe M.; Junge K.; Beller M. Homogeneous Catalysis by Manganese-Based Pincer Complexes. Eur. J. Org. Chem. 2017, 4344–4362. 10.1002/ejoc.201700376. [DOI] [Google Scholar]
- Dub P. A.; Gordon J. C. Metal–Ligand Bifunctional Catalysis: The “Accepted” Mechanism, the Issue of Concertedness, and the Function of the Ligand in Catalytic Cycles Involving Hydrogen Atoms. ACS Catal. 2017, 7, 6635–6655. 10.1021/acscatal.7b01791. [DOI] [Google Scholar]
- Filonenko G. A.; van Putten R.; Hensen E. J. M.; Pidko E. A. Catalytic (De)Hydrogenation Promoted by Non-precious Metals – Co, Fe and Mn: Recent Advances in an Emerging Field. Chem. Soc. Rev. 2018, 47, 1459–1483. 10.1039/C7CS00334J. [DOI] [PubMed] [Google Scholar]
- Schneider S.; Meiners J.; Askevold B. Cooperative Aliphatic PNP Amido Pincer Ligands – Versatile Building Blocks for Coordination Chemistry and Catalysis. Eur. J. Inorg. Chem. 2012, 2012, 412–429. 10.1002/ejic.201100880. [DOI] [Google Scholar]
- Rozenel S. S.; Kerr J. B.; Arnold J. Metal Complexes of Co, Ni and Cu with the Pincer Ligand HN(CH2CH2PiPr2)2: Preparation, Characterization and Electrochemistry. Dalton Trans. 2011, 40, 10397–10405. 10.1039/c1dt10599j. [DOI] [PubMed] [Google Scholar]
- Vasudevan K. V.; Scott B. L.; Hanson S. K. Alkene Hydrogenation Catalyzed by Nickel Hydride Complexes of an Aliphatic PNP Pincer Ligand. Eur. J. Inorg. Chem. 2012, 4898–4906. 10.1002/ejic.201200758. [DOI] [Google Scholar]
- Schneck F.; Finger M.; Tromp M.; Schneider S. Chemical Non-Innocence of an Aliphatic PNP Pincer Ligand. Chem. – Eur. J. 2017, 23, 33–37. 10.1002/chem.201604407. [DOI] [PubMed] [Google Scholar]
- Tamizmani M.; Sivasankar C. Synthesis, Characterization and Catalytic Application of Some Novel PNP-Ni(II) Complexes: Regio-selective [2 + 2 + 2] Cycloaddition Reaction of Alkyne. J. Organomet. Chem. 2017, 845, 82–89. 10.1016/j.jorganchem.2017.02.039. [DOI] [Google Scholar]
- Zhang G.; Scott B. L.; Hanson S. K. Mild and Homogeneous Cobalt-Catalyzed Hydrogenation of C=C, C=O, and C=N Bonds. Angew. Chem., Int. Ed. 2012, 51, 12102–12106. 10.1002/anie.201206051. [DOI] [PubMed] [Google Scholar]
- Zhang G.; Vasudevan K. V.; Scott B. L.; Hanson S. K. Understanding the Mechanisms of Cobalt-Catalyzed Hydrogenation and Dehydrogenation Reactions. J. Am. Chem. Soc. 2013, 135, 8668–8681. 10.1021/ja402679a. [DOI] [PubMed] [Google Scholar]
- Camp C.; Naested L. C. E.; Severin K.; Arnold J. N–N Bond Cleavage in a Nitrous Oxide–NHC Adduct Promoted by a PNP Pincer Cobalt(I) Complex. Polyhedron 2016, 103, 157–163. 10.1016/j.poly.2015.09.001. [DOI] [Google Scholar]
- Rozenel S. S.; Padilla R.; Arnold J. Chemistry of Reduced Monomeric and Dimeric Cobalt Complexes Supported by a PNP Pincer Ligand. Inorg. Chem. 2013, 52, 11544–11550. 10.1021/ic4018882. [DOI] [PubMed] [Google Scholar]
- Lagaditis P. O.; Schluschaß B.; Demeshko S.; Würtele C.; Schneider S. Square-Planar Cobalt(III) Pincer Complex. Inorg. Chem. 2016, 55, 4529–4536. 10.1021/acs.inorgchem.6b00369. [DOI] [PubMed] [Google Scholar]
- Fu S.; Chen N.-Y.; Liu X.; Shao Z.; Luo S.-P.; Liu Q. Ligand-Controlled Cobalt-Catalyzed Transfer Hydrogenation of Alkynes: Stereodivergent Synthesis of Z- and E-Alkenes. J. Am. Chem. Soc. 2016, 138, 8588–8594. 10.1021/jacs.6b04271. [DOI] [PubMed] [Google Scholar]
- Mills M. R.; Barnes C. L.; Bernskoetter W. H. Influences of Bifunctional PNP-Pincer Ligands on Low Valent Cobalt Complexes Relevant to CO2 Hydrogenation. Inorg. Chem. 2018, 57, 1590–1597. 10.1021/acs.inorgchem.7b02931. [DOI] [PubMed] [Google Scholar]
- Alberico E.; Sponholz P.; Cordes C.; Nielsen M.; Drexler H.-J.; Baumann W.; Junge H.; Beller M. Selective Hydrogen Production from Methanol with a Defined Iron Pincer Catalyst under Mild Conditions. Angew. Chem., Int. Ed. 2013, 52, 14162–14166. 10.1002/anie.201307224. [DOI] [PubMed] [Google Scholar]
- Koehne I.; Schmeier T. J.; Bielinski E. A.; Pan C. J.; Lagaditis P. O.; Bernskoetter W. H.; Takase M. K.; Würtele C.; Hazari N.; Schneider S. Synthesis and Structure of Six-Coordinate Iron Borohydride Complexes Supported by PNP Ligands. Inorg. Chem. 2014, 53, 2133–2143. 10.1021/ic402762v. [DOI] [PubMed] [Google Scholar]
- Fillman K. L.; Bielinski E. A.; Schmeier T. J.; Nesvet J. C.; Woodruff T. M.; Pan C. J.; Takase M. K.; Hazari N.; Neidig M. L. Flexible Binding of PNP Pincer Ligands to Monomeric Iron Complexes. Inorg. Chem. 2014, 53, 6066–6072. 10.1021/ic5004275. [DOI] [PubMed] [Google Scholar]
- Chakraborty S.; Dai H.; Bhattacharya P.; Fairweather N. T.; Gibson M. S.; Krause J. A.; Guan H. Iron-Based Catalysts for the Hydrogenation of Esters to Alcohols. J. Am. Chem. Soc. 2014, 136, 7869–7872. 10.1021/ja504034q. [DOI] [PubMed] [Google Scholar]
- Bornschein C.; Werkmeister S.; Wendt B.; Jiao H.; Alberico E.; Baumann W.; Junge H.; Junge K.; Beller M. Mild and Selective Hydrogenation of Aromatic and Aliphatic (Di)Nitriles with a Well-Defined Iron Pincer Complex. Nat. Commun. 2014, 5, 4111. 10.1038/ncomms5111. [DOI] [PubMed] [Google Scholar]
- Schneck F.; Assmann M.; Balmer M.; Harms K.; Langer R. Selective Hydrogenation of Amides to Amines and Alcohols Catalyzed by Improved Iron Pincer Complexes. Organometallics 2016, 35, 1931–1943. 10.1021/acs.organomet.6b00251. [DOI] [Google Scholar]
- Elangovan S.; Wendt B.; Topf C.; Bachmann S.; Scalone M.; Spannenberg A.; Jiao H.; Baumann W.; Junge K.; Beller M. Improved Second Generation Iron Pincer Complexes for Effective Ester Hydrogenation. Adv. Synth. Catal. 2016, 358, 820–825. 10.1002/adsc.201500930. [DOI] [Google Scholar]
- Lange S.; Elangovan S.; Cordes C.; Spannenberg A.; Jiao H.; Junge H.; Bachmann S.; Scalone M.; Topf C.; Junge K.; Beller M. Selective Catalytic Hydrogenation of Nitriles to Primary Amines Using Iron Pincer Complexes. Catal. Sci. Technol. 2016, 6, 4768–4772. 10.1039/C6CY00834H. [DOI] [Google Scholar]
- Smith N. E.; Bernskoetter W. H.; Hazari N.; Mercado B. Q. Synthesis and Catalytic Activity of PNP-Supported Iron Complexes with Ancillary Isonitrile Ligands. Organometallics 2017, 36, 3995–4004. 10.1021/acs.organomet.7b00602. [DOI] [Google Scholar]
- Elangovan S.; Topf C.; Fischer S.; Jiao H.; Spannenberg A.; Baumann W.; Ludwig R.; Junge K.; Beller M. Selective Catalytic Hydrogenations of Nitriles, Ketones, and Aldehydes by Well-Defined Manganese Pincer Complexes. J. Am. Chem. Soc. 2016, 138, 8809–8814. 10.1021/jacs.6b03709. [DOI] [PubMed] [Google Scholar]
- Fu S.; Shao Z.; Wang Y.; Liu Q. Manganese-Catalyzed Upgrading of Ethanol into 1-Butanol. J. Am. Chem. Soc. 2017, 139, 11941–11948. 10.1021/jacs.7b05939. [DOI] [PubMed] [Google Scholar]
- Kulkarni N. V.; Brennessel W. W.; Jones W. D. Catalytic Upgrading of Ethanol to n-Butanol via Manganese-Mediated Guerbet Reaction. ACS Catal. 2018, 8, 997–1002. 10.1021/acscatal.7b03653. [DOI] [Google Scholar]
- Chakraborty S.; Lagaditis P. O.; Förster M.; Bielinski E. A.; Hazari N.; Holthausen M. C.; Jones W. D.; Schneider S. Well-Defined Iron Catalysts for the Acceptorless Reversible Dehydrogenation-Hydrogenation of Alcohols and Ketones. ACS Catal. 2014, 4, 3994–4003. 10.1021/cs5009656. [DOI] [Google Scholar]
- Chakraborty S.; Brennessel W. W.; Jones W. D. A Molecular Iron Catalyst for the Acceptorless Dehydrogenation and Hydrogenation of N-Heterocycles. J. Am. Chem. Soc. 2014, 136, 8564–8567. 10.1021/ja504523b. [DOI] [PubMed] [Google Scholar]
- Xu R.; Chakraborty S.; Bellows S. M.; Yuan H.; Cundari T. R.; Jones W. D. Iron-Catalyzed Homogeneous Hydrogenation of Alkenes under Mild Conditions by a Stepwise, Bifunctional Mechanism. ACS Catal. 2016, 6, 2127–2135. 10.1021/acscatal.5b02674. [DOI] [Google Scholar]
- Elangovan S.; Garbe M.; Jiao H.; Spannenberg A.; Junge K.; Beller M. Hydrogenation of Esters to Alcohols Catalyzed by Defined Manganese Pincer Complexes. Angew. Chem., Int. Ed. 2016, 55, 15364–15368. 10.1002/anie.201607233. [DOI] [PubMed] [Google Scholar]
- Werkmeister S.; Junge K.; Wendt B.; Alberico E.; Jiao H.; Baumann W.; Junge H.; Gallou F.; Beller M. Hydrogenation of Esters to Alcohols with a Well-Defined Iron Complex. Angew. Chem., Int. Ed. 2014, 53, 8722–8726. 10.1002/anie.201402542. [DOI] [PubMed] [Google Scholar]
- Fairweather N. T.; Gibson M. S.; Guan H. Homogeneous Hydrogenation of Fatty Acid Methyl Esters and Natural Oils under Neat Conditions. Organometallics 2015, 34, 335–339. 10.1021/om5011337. [DOI] [Google Scholar]
- Rezayee N. M.; Samblanet D. C.; Sanford M. S. Iron-Catalyzed Hydrogenation of Amides to Alcohols and Amines. ACS Catal. 2016, 6, 6377–6383. 10.1021/acscatal.6b01454. [DOI] [Google Scholar]
- Jayarathne U.; Zhang Y.; Hazari N.; Bernskoetter W. H. Selective Iron-Catalyzed Deaminative Hydrogenation of Amides. Organometallics 2017, 36, 409–416. 10.1021/acs.organomet.6b00816. [DOI] [Google Scholar]
- Yuwen J.; Chakraborty S.; Brennessel W. W.; Jones W. D. Additive-Free Cobalt-Catalyzed Hydrogenation of Esters to Alcohols. ACS Catal. 2017, 7, 3735–3740. 10.1021/acscatal.7b00623. [DOI] [Google Scholar]
- Bielinski E. A.; Förster M.; Zhang Y.; Bernskoetter W. H.; Hazari N.; Holthausen M. C. Base-Free Methanol Dehydrogenation Using a Pincer-Supported Iron Compound and Lewis Acid Co-catalyst. ACS Catal. 2015, 5, 2404–2415. 10.1021/acscatal.5b00137. [DOI] [Google Scholar]
- Bellows S. M.; Chakraborty S.; Gary J. B.; Jones W. D.; Cundari T. R. An Uncanny Dehydrogenation Mechanism: Polar Bond Control over Stepwise or Concerted Transition States. Inorg. Chem. 2017, 56, 5519–5524. 10.1021/acs.inorgchem.6b01800. [DOI] [PubMed] [Google Scholar]
- Zhang G.; Hanson S. K. Cobalt-Catalyzed Acceptorless Alcohol Dehydrogenation: Synthesis of Imines from Alcohols and Amines. Org. Lett. 2013, 15, 650–653. 10.1021/ol303479f. [DOI] [PubMed] [Google Scholar]
- Bielinski E. A.; Lagaditis P. O.; Zhang Y.; Mercado B. Q.; Würtele C.; Bernskoetter W. H.; Hazari N.; Schneider S. Lewis Acid-Assisted Formic Acid Dehydrogenation Using a Pincer-Supported Iron Catalyst. J. Am. Chem. Soc. 2014, 136, 10234–10237. 10.1021/ja505241x. [DOI] [PubMed] [Google Scholar]
- Andérez-Fernández M.; Vogt L. K.; Fischer S.; Zhou W.; Jiao H.; Garbe M.; Elangovan S.; Junge K.; Junge H.; Ludwig R.; Beller M. A Stable Manganese Pincer Catalyst for the Selective Dehydrogenation of Methanol. Angew. Chem., Int. Ed. 2017, 56, 559–562. 10.1002/anie.201610182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glüer A.; Förster M.; Celinski V. R.; auf der Günne J. S.; Holthausen M. C.; Schneider S. Highly Active Iron Catalyst for Ammonia Borane Dehydrocoupling at Room Temperature. ACS Catal. 2015, 5, 7214–7217. 10.1021/acscatal.5b02406. [DOI] [Google Scholar]
- Sharninghausen L. S.; Mercado B. Q.; Crabtree R. H.; Hazari N. Selective Conversion of Glycerol to Lactic Acid with Iron Pincer Precatalysts. Chem. Commun. 2015, 51, 16201–16204. 10.1039/C5CC06857F. [DOI] [PubMed] [Google Scholar]
- Peña-López M.; Neumann H.; Beller M. Iron(II) Pincer-Catalyzed Synthesis of Lactones and Lactams through a Versatile Dehydrogenative Domino Sequence. ChemCatChem 2015, 7, 865–871. 10.1002/cctc.201402967. [DOI] [Google Scholar]
- Lane E. M.; Uttley K. B.; Hazari N.; Bernskoetter W. Iron-Catalyzed Amide Formation from the Dehydrogenative Coupling of Alcohols and Secondary Amines. Organometallics 2017, 36, 2020–2025. 10.1021/acs.organomet.7b00258. [DOI] [Google Scholar]
- Anke F.; Han D.; Klahn M.; Spannenberg A.; Beweries T. Formation of High-Molecular Weight Polyaminoborane by Fe Hydride Catalysed Dehydrocoupling of Methylamine Borane. Dalton Trans. 2017, 46, 6843–6847. 10.1039/C7DT01487B. [DOI] [PubMed] [Google Scholar]
- Zhang G.; Hanson S. K. Cobalt-Catalyzed Transfer Hydrogenation of C=O and C=N Bonds. Chem. Commun. 2013, 49, 10151–10153. 10.1039/c3cc45900d. [DOI] [PubMed] [Google Scholar]
- Zhang Y.; MacIntosh A. D.; Wong J. L.; Bielinski E. A.; Williard P. G.; Mercado B. Q.; Hazari N.; Bernskoetter W. H. Iron Catalyzed CO2 Hydrogenation to Formate Enhanced by Lewis Acid Co-catalysts. Chem. Sci. 2015, 6, 4291–4299. 10.1039/C5SC01467K. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spentzos A. Z.; Barnes C. L.; Bernskoetter W. H. Effective Pincer Cobalt Precatalysts for Lewis Acid Assisted CO2 Hydrogenation. Inorg. Chem. 2016, 55, 8225–8233. 10.1021/acs.inorgchem.6b01454. [DOI] [PubMed] [Google Scholar]
- Jayarathne U.; Hazari N.; Bernskoetter W. H. Selective Iron-Catalyzed N-Formylation of Amines using Dihydrogen and Carbon Dioxide. ACS Catal. 2018, 8, 1338–1345. 10.1021/acscatal.7b03834. [DOI] [Google Scholar]
- Sonnenberg J. F.; Wan K. Y.; Sues P. E.; Morris R. H. Ketone Asymmetric Hydrogenation Catalyzed by P-NH-P′ Pincer Iron Catalysts: An Experimental and Computational Study. ACS Catal. 2017, 7, 316–326. 10.1021/acscatal.6b02489. [DOI] [Google Scholar]
- Qu S.; Dai H.; Dang Y.; Song C.; Wang Z.-X.; Guan H. Computational Mechanistic Study of Fe-Catalyzed Hydrogenation of Esters to Alcohols: Improving Catalysis by Accelerating Precatalyst Activation with a Lewis Base. ACS Catal. 2014, 4, 4377–4388. 10.1021/cs501089h. [DOI] [Google Scholar]
- Friedrich A.; Drees M.; Schmedt auf der Günne J.; Schneider S. Highly Stereoselective Proton/Hydride Exchange: Assistance of Hydrogen Bonding for the Heterolytic Splitting of H2. J. Am. Chem. Soc. 2009, 131, 17552–17553. 10.1021/ja908644n. [DOI] [PubMed] [Google Scholar]
- Johansson R.; Jarenmark M.; Wendt O. F. Insertion of Carbon Dioxide into (PCP)PdII-Me Bonds. Organometallics 2005, 24, 4500–4502. 10.1021/om0505561. [DOI] [Google Scholar]
- Polukeev A. V.; Kuklin S. A.; Petrovskii P. V.; Peregudova S. M.; Smol’yakov A. F.; Dolgushin F. M.; Koridze A. A. Synthesis and Characterization of Fluorophenylpalladium Pincer Complexes: Electronic Properties of Some Pincer Ligands Evaluated by Multinuclear NMR Spectroscopy and Electrochemical Studies. Dalton Trans. 2011, 40, 7201–7209. 10.1039/c1dt10446b. [DOI] [PubMed] [Google Scholar]
- Martínez-Prieto L. M.; Melero C.; del Río D.; Palma P.; Cámpora J.; Álvarez E. Synthesis and Reactivity of Nickel and Palladium Fluoride Complexes with PCP Pincer Ligands. NMR-Based Assessment of Electron-Donating Properties of Fluoride and Other Monoanionic Ligands. Organometallics 2012, 31, 1425–1438. 10.1021/om2009793. [DOI] [Google Scholar]
- Adhikary A.; Krause J. A.; Guan H. How Does the Carbon Hybridization Impact the Rate of Metal-Carbon Bond Cleavage by an Alkyne? A Case Study with POCOP-Pincer Ligated Palladium Hydrocarbyl Complexes. J. Organomet. Chem. 2017, 845, 151–156. 10.1016/j.jorganchem.2017.04.024. [DOI] [Google Scholar]
- Liang L.-C.; Chien P.-S.; Huang Y.-L. Intermolecular Arene C-H Activation by Nickel(II). J. Am. Chem. Soc. 2006, 128, 15562–15563. 10.1021/ja065505p. [DOI] [PubMed] [Google Scholar]
- Castonguay A.; Beauchamp A. L.; Zargarian D. Preparation and Reactivities of PCP-Type Pincer Complexes of Nickel. Impact of Different Ligand Skeletons and Phosphine Substituents. Organometallics 2008, 27, 5723–5732. 10.1021/om8005454. [DOI] [Google Scholar]
- Venkanna G. T.; Tammineni S.; Arman H. D.; Tonzetich Z. J. Synthesis, Characterization, and Catalytic Activity of Nickel(II) Alkyl Complexes Supported by Pyrrole–Diphosphine Ligands. Organometallics 2013, 32, 4656–4663. 10.1021/om400630q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marziale A. N.; Herdtweck E.; Eppinger J.; Schneider S. Palladium N(CH2CH2PiPr2)2-Dialkylamides: Synthesis, Structural Characterization, and Reactivity. Inorg. Chem. 2009, 48, 3699–3709. 10.1021/ic802286u. [DOI] [PubMed] [Google Scholar]
- Käß M.; Friedrich A.; Drees M.; Schneider S. Ruthenium Complexes with Cooperative PNP Ligands: Bifunctional Catalysts for the Dehydrogenation of Ammonia-Borane. Angew. Chem., Int. Ed. 2009, 48, 905–907. 10.1002/anie.200805108. [DOI] [PubMed] [Google Scholar]
- Friedrich A.; Drees M.; Käss M.; Herdtweck E.; Schneider S. Ruthenium Complexes with Cooperative PNP-Pincer Amine, Amido, Imine, and Enamido Ligands: Facile Ligand Backbone Functionalization Processes. Inorg. Chem. 2010, 49, 5482–5494. 10.1021/ic100358m. [DOI] [PubMed] [Google Scholar]
- Rumble J. R., Ed.; CRC Handbook of Chemistry and Physics, 98th ed.; Taylor & Francis: Boca Raton, FL, 2018; pp 5–24. [Google Scholar]
- Strautmann J.; Paciello R.; Schaub T.; Schierle-Arndt K.; Löffler D.; Wilmer H.; Eickemeyer F.; Blasberg F.. Process for the Generation of Thin Inorganic Films. WO 22016/020208 A12016.
- Ozerov O. V.; Guo C.; Fan L.; Foxman B. M. Oxidative Addition of N-C and N-H Bonds to Zerovalent Nickel, Palladium, and Platinum. Organometallics 2004, 23, 5573–5580. 10.1021/om0493513. [DOI] [Google Scholar]
- Castonguay A.; Sui-Seng C.; Zargarian D.; Beauchamp A. L. Syntheses and Reactivities of New PCsp3P Pincer Complexes of Nickel. Organometallics 2006, 25, 602–608. 10.1021/om050844x. [DOI] [Google Scholar]
- Liang L.-C.; Chien P.-S.; Lin J.-M.; Huang M.-H.; Huang Y.-L.; Liao J.-H. Amido Pincer Complexes of Nickel(II): Synthesis, Structure, and Reactivity. Organometallics 2006, 25, 1399–1411. 10.1021/om050943a. [DOI] [Google Scholar]
- Pandarus V.; Zargarian D. New Pincer-Type Diphosphinito (POCOP) Complexes of Nickel. Organometallics 2007, 26, 4321–4334. 10.1021/om700400x. [DOI] [PubMed] [Google Scholar]
- Salah A. B.; Zargarian D. The Impact of P-Substituents on the Structures, Spectroscopic Properties, and Reactivities of POCOP-Type Pincer Complexes of Nickel(II). Dalton Trans. 2011, 40, 8977–8985. 10.1039/c1dt10381d. [DOI] [PubMed] [Google Scholar]
- Adhikary A.; Schwartz J. R.; Meadows L. M.; Krause J. A.; Guan H. Interaction of Alkynes with Palladium POCOP-Pincer Hydride Complexes and Its Unexpected Relation to Palladium-Catalyzed Hydrogenation of Alkynes. Inorg. Chem. Front. 2014, 1, 71–82. 10.1039/C3QI00073G. [DOI] [Google Scholar]
- Salah A.; Corpet M.; Khan N. u.-H.; Zargarian D.; Spasyuk D. M. Synthesis of Unsymmetrical 5,6-POCOP′-Type Pincer Complexes of Nickel(II): Impact of Nickelacycle Size on Structures and Spectroscopic Properties. New J. Chem. 2015, 39, 6649–6658. 10.1039/C5NJ00807G. [DOI] [Google Scholar]
- Jessop P. G.; Morris R. H. Reactions of Transition Metal Dihydrogen Complexes. Coord. Chem. Rev. 1992, 121, 155–284. 10.1016/0010-8545(92)80067-2. [DOI] [Google Scholar]
- Heinekey D. M.; Oldham W. J. Jr. Coordination Chemistry of Dihydrogen. Chem. Rev. 1993, 93, 913–926. 10.1021/cr00019a004. [DOI] [Google Scholar]
- Papish E. T.; Rix F. C.; Spetseris N.; Norton J. R.; Williams R. D. Protonation of CpW(CO)2(PMe3)H: Is the Metal or the Hydride the Kinetic Site?. J. Am. Chem. Soc. 2000, 122, 12235–12242. 10.1021/ja002395s. [DOI] [Google Scholar]
- Friedrich A.; Ghosh R.; Kolb R.; Herdtweck E.; Schneider S. Iridium Olefin Complexes Bearing Dialkylamino/amido PNP Pincer Ligands: Synthesis, Reactivity, and Solution Dynamics. Organometallics 2009, 28, 708–718. 10.1021/om800423u. [DOI] [Google Scholar]
- Zhang J.; Adhikary A.; King K. M.; Krause J. A.; Guan H. Substituent Effects on Ni–S Bond Dissociation Energies and Kinetic Stability of Nickel Arylthiolate Complexes Supported by a Bis(phosphinite)-based Pincer Ligand. Dalton Trans. 2012, 41, 7959–7968. 10.1039/c2dt30407d. [DOI] [PubMed] [Google Scholar]
- Li H.; Meng W.; Adhikary A.; Li S.; Ma N.; Zhao Q.; Yang Q.; Eberhardt N. A.; Leahy K. M.; Krause J. A.; Zhang J.; Chen X.; Guan H. Metathesis Reactivity of Bis(phosphinite) Pincer Ligated Nickel Chloride, Isothiocyanate and Azide Complexes. J. Organomet. Chem. 2016, 804, 132–141. 10.1016/j.jorganchem.2015.12.038. [DOI] [Google Scholar]
- Castonguay A.; Beauchamp A. L.; Zargarian D. New Derivatives of PCP-Type Pincer Complexes of Nickel. Inorg. Chem. 2009, 48, 3177–3184. 10.1021/ic801641u. [DOI] [PubMed] [Google Scholar]
- Hao J.; Mougang-Soumé B.; Vabre B.; Zargarian D. On the Stability of a POCsp3OP-Type Pincer Ligand in Nickel(II) Complexes. Angew. Chem., Int. Ed. 2014, 53, 3218–3222. 10.1002/anie.201310386. [DOI] [PubMed] [Google Scholar]
- Eberhardt N. A.; Guan H. Nickel Hydride Complexes. Chem. Rev. 2016, 116, 8373–8426. 10.1021/acs.chemrev.6b00259. [DOI] [PubMed] [Google Scholar]
- Jongbloed L. S.; García-López D.; van Heck R.; Siegler M. A.; Carbó J. J.; van der Vlugt J. I. Arene C(sp2)-H Metalation at NiII Modeled with a Reactive PONCPh Ligand. Inorg. Chem. 2016, 55, 8041–8047. 10.1021/acs.inorgchem.6b01162. [DOI] [PubMed] [Google Scholar]
- Bordwell F. G.; Drucker G. E. Acidities of Indene and Phenyl-, Diphenyl-, and Triphenylindenes. J. Org. Chem. 1980, 45, 3325–3328. 10.1021/jo01304a037. [DOI] [Google Scholar]
- Olmstead W. N.; Margolin Z.; Bordwell F. G. Acidities of Water and Simple Alcohols in Dimethyl Sulfoxide Solution. J. Org. Chem. 1980, 45, 3295–3299. 10.1021/jo01304a032. [DOI] [Google Scholar]
- Dixon J. T.; Grove J. J. C.; Wasserscheid P.; McGuinness D. S.; Hess F. M.; Maumela H.; Morgan D. H.; Bollmann A.. Trimerisation and Oligomerisation of Olefins Using a Chromium Based Catalyst. WO 2003/053890 A12003.
- Abdur-Rashid K.Transfer Hydrogenation Processes and Catalysts. US 2005/0107638 A12005.
- Marshall W. J.; Grushin V. V. Activation of Chlorobenzene with Ni(0) N,N-Chelates — A Remarkably Profound Effect of a Minuscule Change in Ligand Structure. Can. J. Chem. 2005, 83, 640–645. 10.1139/v05-022. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.