Abstract
Background/aims
The contribution of inflammation to tendinopathy has been debated in the scientific literature. Several factors may contribute to this lack of clarity, including inconsistent definitions of inflammation. We hypothesised that the adoption and/or rejection of a causal link between inflammation and tendinopathy varied as a function of the ‘inflammatory component’ (eg, immune cell and molecular mediators included in published reviews).
Methods
Twenty data items were collected from each review to determine conclusions about the role of inflammation in tendinopathy, specific definitions of the ‘inflammatory component,’ quality of the review and other potential correlates. Associations between correlates and a review’s conclusion about the role of inflammation in tendinopathy were tested using binomial logistic regression. The database searches retrieved 2261 unique publications: 137 fulfilled inclusion criteria after full text screenings.
Results
There has been little support for an inflammatory component to tendinopathy until recently (2012–2015). Prior to 2012, the majority of published reviews did not discuss monocytes, macrophages or lymphocytes in tendinopathy; rather they focused on the lack of neutrophils, often referred to as ‘the inflammatory infiltrate’, or immune cells were not discussed. Reviews including monocytes and lymphocytes in their discussions were 5.23 times more likely to conclude inflammation was important than reviews that did not, p<0.001.
Conclusions
Data collected show growing support for an inflammatory component to tendinopathy, particularly among high-quality reviews and those that used more robust definitions of inflammation. This finding may have implications for explaining dissonance in the literature regarding a causal role for inflammation in the pathogenesis of tendinopathy.
Keywords: Tendinopathy, Tendinosis, Immunology, inflammation
What are new findings?
This study is the first to conduct a quantitative analysis of published reviews about the pathogenesis of tendinopathy with the aim to understand reasons for discordant theories about the role of inflammation in tendinopathy.
Results revealed growing support for an inflammatory component to tendinopathy, particularly during the past 3–6 years, which paralleled a rise in more robust definitions of inflammatory components.
This study suggests that dissonance in the scientific literature may be addressed by standardisation of definitions of inflammation.
Potential impact on clinical practice?
Further characterisation of the phenotypes of tendon cells, monocytes, macrophages and lymphocytes may reveal novel mechanisms of tendon pathology and facilitate discussion and discovery of therapeutic options for tendinopathy.
New therapeutic approaches are required to target inflammation in tendon disease. Such therapeutics could potentially be used as preventative measures to stunt the progression of tendon pathology, often resulting in partial tears or full rupture in the clinical setting.
Introduction
The role of inflammation in the pathogenesis of tendinopathy has been a topic of debate since the mid-late 20th century. Prior to the turn of the 21st century, inflammation was believed to contribute to the pain and pathology associated with non-acute, non-ruptured diseased tendons.1–3 This is often referred to as the ‘tendinitis era’; treatments used for tendinopathy during this time were traditional anti-inflammatories, such as non-steroidal anti-inflammatory drugs and corticosteroids.4 However, as anti-inflammatory regimens for tendon dysfunction were shown as ineffective in treating pathology and histological studies of diseased tendons indicated an absence of ‘inflammatory cells’, the term ‘tendinitis’ and its associated inflammatory paradigm were increasingly considered misleading.1–3 5–7 The orthopaedic community shifted their attention towards other explanatory paradigms and novel treatment modalities. Thus, tendinopathy was predominantly characterised as a 'degenerative' condition devoid of inflammation since the early 2000s (tendinosis).5 8–11 The shift in disease paradigms may have been an oversimplification of inflammation’s role in tendinopathy, thus impeding the discovery of novel inflammatory mechanisms in tendinopathy.10 12 13
Few data have examined reasons for shifts in causal theories. Previous research has assessed the role of inflammation in tendinopathy directly through basic and clinical studies or by conducting qualitative reviews. To our knowledge, this is the first study to systematically search and evaluate published reviews’ conclusions about the role of inflammation in tendinopathy and to evaluate potential correlates of those conclusions through comprehensive data collection and statistical analysis.
We hypothesised that equipoise in the literature on the role of inflammation in tendinopathy may have been due to specific factors, such as inconsistent definitions of the inflammatory component and the quality of the review. The purpose of this study was to conduct a quantitative analysis of published reviews to examine potential paradigm shifts and predictors of conclusions regarding the role of inflammation in tendinopathy. Our specific objectives were to evaluate: (1) trends and paradigm shifts from inception to 2015 about the perception of inflammation in tendinopathy, (2) if the adoption and/or rejection of a causal theory between inflammation and tendinopathy varied as a function of the characterisation of the ‘inflammatory component’ (what immune cell types, mechanisms and/or molecular mediators are included in discussions), (3) if the adoption and/or rejection of a causal theory between inflammation and tendinopathy varied as a function of the quality of the review and (4) other correlates of an article’s conclusion about the role of inflammation in tendinopathy. Other correlates included the type of journal the review was published in, the journal’s impact factor and the number of citations per year the review has received.
Methods
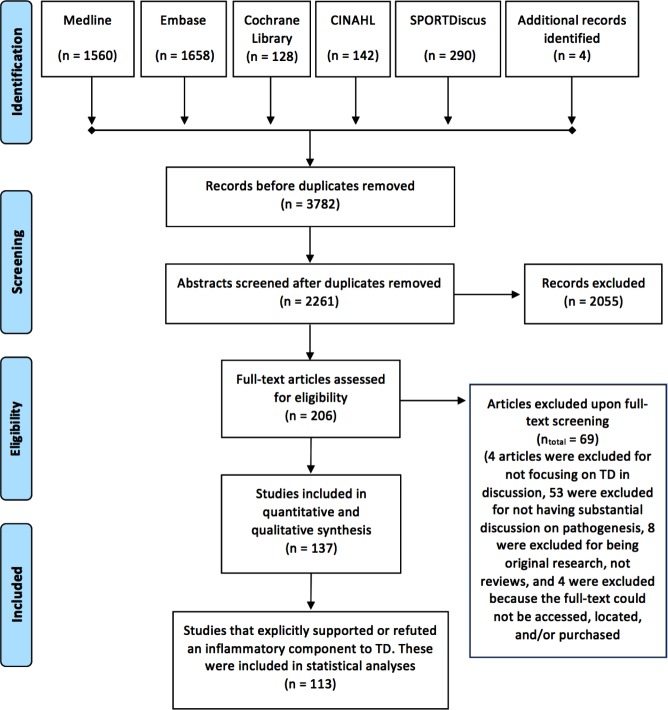
This manuscript follows the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRIMSA) guidelines.14 Search strategies and terms are included in online supplementary appendix 1, and the PRIMSA flow diagram is displayed in figure 1.
Figure 1.
Preferred Reporting Items for Systematic Reviews and Meta-Analyses flow chart for the identification and screening of reviews.
Search strategy and eligibility criteria
A systematic literature search was conducted on 15 December 2015 using the Medline (OVID), Embase (OVID), Cochrane Database of Systematic Reviews (Wiley), CINAHL (EbscoHost) and SPORTDiscus (EbscoHost) databases to identify published literature related to the pathogenesis of tendinopathy. The search terms used included variations of ‘tendinitis,’ OR ‘tendinosis,’ OR ‘tendinopathy,’ OR ‘tendon disease / rupture / injury // tear’ searched in the title or abstract fields and using appropriate Mesh or Emtree or other database-specific controlled vocabulary headings. These terms were combined with relevant search filters for identifying systematic reviews. Detailed search strategies for each database are included in online supplementary appendix 1. Publications were limited to reviews in the English language if necessary. The literature search did not include a specific search for non-indexed reviews, dissertations or conference proceedings. After abstract and full-text screening, citation lists of included studies were manually searched to identify additional reviews for screening.
bmjsem-2017-000332supp001.docx (34KB, docx)
Once the literature search was completed, results for each database were compiled into Endnote7 for title and abstract screening. Duplicates were removed and two reviewers (MJM and MSR) independently screened titles and abstracts for inclusion/exclusion criteria. We included reviews that included human, animal or other models and met the following criteria: (1) the publication was a review and not an original research paper; (2) the primary purpose of the publication was to discuss the pathogenesis of tendinopathy, including potential causes or risk factors for tendinopathy, pathways associated with the development of tendinopathy, histological characteristics of tendinopathic tissue or reasons for the failure of healing in diseased tendons and (3) the publication could be found in the English language.
Publications were excluded from the study during abstract screening if: (1) it focused on non-pathological, acute injuries where the tear/rupture was clearly due to a traumatic event rather than tears/ruptures due to sustained pathology; (2) it was a case study and/or series or (3) it did not have a clear intent to focus its discussion on the pathogenesis, causes, risk factors or characteristics of diseased tendons, but is rather a brief overview of tendinopathy (eg, ‘what’s new in tendinopathy’). Publications were excluded from the study during full-text screening if: (1) the full-text could not be obtained; (2) the publication was not substantially engaging with pathogenesis; (3) the publication was not explicitly discussing pathological tendon tissue or (4) the publication included other musculoskeletal diseases (eg, ligament and/or rheumatoid diseases) into the discussions and/or conclusions without a primary focus on tendinopathy.
Data items and collection process
The inclusion/exclusion criteria, data items and methods of analysis were specified in a written protocol prior to the systematic search. Multiple pilot studies were used to generate, test and refine relevant data items and collection protocol for the systematic review. After abstracts and full texts were independently screened for inclusion/exclusion criteria, classification and quality data were extracted from all included reviews and documented in an Excel spreadsheet. Twenty data items were collected for each review, including categorical data regarding the review’s conclusions about the role of inflammation in tendinopathy.
Judgement regarding a review’s conclusions on a causal role for inflammation in tendinopathy was determined systematically. Each publication was placed into one of several predefined categories: (1) the review determines that the pathogenesis of tendinopathy is multifactorial without mentioning ‘degeneration’ or ‘inflammation’; (2) the review determines that tendinopathy is degenerative with an absence of a chronic inflammatory component; (3) the review determines that inflammation is a contributing factor in tendon pathology; (4) the review discusses inflammation and/or degeneration in the content of tendon pathology but the author’s conclusion about either’s importance is not readily apparent and (5) other/not applicable/expert panel determines categorisation is unclear. To be categorised as ‘for’ or ‘against’ the inflammatory concept, the review must unambiguously state one side or the other. An example of a paper that was categorised as ‘for’ inflammation was published by Rees et al, who stated “More modern research tools have confirmed the presence of inflammatory cells including macrophages and lymphocytes in chronic tendinopathy.”10 An example of a paper that was categorised as ‘against’ inflammation was published by Magnan et al, who stated “Achilles tendinopathy is a degenerative, not an inflammatory, condition.”15
Similar to how a review’s conclusions on inflammation were determined, the types of immune cells discussed, whether signalling molecules/effectors are included and the role they play in pathogenesis of tendinopathy were systematically documented. In addition, the type of journal the review was published in, its impact factor, the number of citations the review received and the year of publication were documented. Finally, a 12-question assessment on the methodological quality/depth of scientific rigour of each review was conducted. The assessment was adapted from the validated quality of assessment tool for systematic reviews, ‘Assessment of Multiple Systematic Reviews’ to evaluate reviews of a wide range of quality, including narrative/qualitative reviews.16
For each categorical data item, a list of responses was developed, piloted and internally corroborated to provide an appropriate balance between specificity and generalisability. All data items were collected for included reviews using standardised extraction sheets. During final data extraction, extraction sheets were printed and responses were determined and documented. Once data collection was complete, data were transferred from the physical extraction sheets into Microsoft Excel. For any given question, if it was unclear what response to choose this was noted for further review by an expert panel of scientists who would determine the response: SGD, SJS and AJC.
All data were collected by MJM. Additionally, all data items were independently extracted by SGD and SJS from a random sample of included reviews (n=10). Results from the random sample were compared among MJM, SGD and SJS to statistically assess reproducibility of all outcomes. Any outcomes without sufficient reproducibility were excluded from the analysis in this review.
Synthesis of results and statistical analyses
All graphs and statistical analyses were performed in SPSS or GraphPad Prism 7 (GraphPad Software). Frequency distributions were performed for all data abstracted; appropriate categories were collapsed or stratified for ease of data interpretation and display. Temporal trends in select outcomes were evaluated using basic proportions of categorisation (displayed via histograms). Stratified analyses were conducted for specific questions and predictors of the conclusion that inflammation plays a causal role in tendinopathy. Associations between potential correlates and a review’s conclusion about the role of inflammation in tendinopathy were tested using binomial logistic regression. Only reviews that explicitly supported or refuted an inflammatory component to tendinopathy were included in statistical analyses. Omnibus tests of model coefficients (x2) were performed to ensure that variance in the data set was significantly greater than unexplained variance. Statistical significance was set at p<0.05.
Results
Study selection
The literature search retrieved a total of 3782 results (figure 1). After removal of 1521 duplicates, 2261 unique publications remained. Screening of the titles and abstracts revealed 206 reviews of the pathogenesis of tendinopathy eligible for inclusion. Further assessment of eligibility through a screening of the full texts led to the exclusion of 69 papers: 4 for having discussions centred around a range of musculoskeletal diseases rather than focusing on tendinopathy, 53 for not having substantial discussion on pathogenesis, 8 were excluded for being original research and not review articles and 4 were excluded because the full text could not be accessed, located and/or purchased. This left 137 reviews meeting our criteria for inclusion, a list of which is available in online supplementary appendix 2. Only reviews that explicitly supported or refuted an inflammatory component to tendinopathy were included in statistical analyses to minimise misclassification bias and produce the most robust analyses (n=113).
General characteristics of included reviews
Of the 137 reviews that met inclusion criteria, 62% (n=85) had specific aims and discussions on the pathogenesis tendinopathy: the remaining 52 were narrative-style overviews on the pathogenesis of tendinopathy. Specific reviews discussed range of topics including: (1) the role of mechanical factors in tendinopathy, (2) matrix protein dysregulation in tendinopathy, (3) inflammatory mediator involvement in tendinopathy, (4) drug-induced tendinopathy, (5) neuronal regulation and involvement in tendinopathy, (6) vascularity and its role in tendinopathy, (7) histological findings of diseased tendons and (8) the association of metabolic disorders/adiposity with tendinopathy. 13.1% (n=18) of included reviews conducted a systematic search and analysis of published literature. There were no reviews that conducted a meta-analysis.
Among included reviews, 86.1% (n=118) were categorised as low quality (quality score ≤4), 9.5% (n=13) were moderate quality (4<quality score<9) and 4.4% (n=6) were high quality (quality score ≥9). Only nine reviews formally assessed the methodological quality of their primary literature, all of which were systematic reviews. Although most of our included reviews were of low quality, there has been an increase in the number of moderate-quality to high-quality reviews over time.
Temporal trends in the specificity and inclusion of an ‘inflammatory component’ to tendinopathy
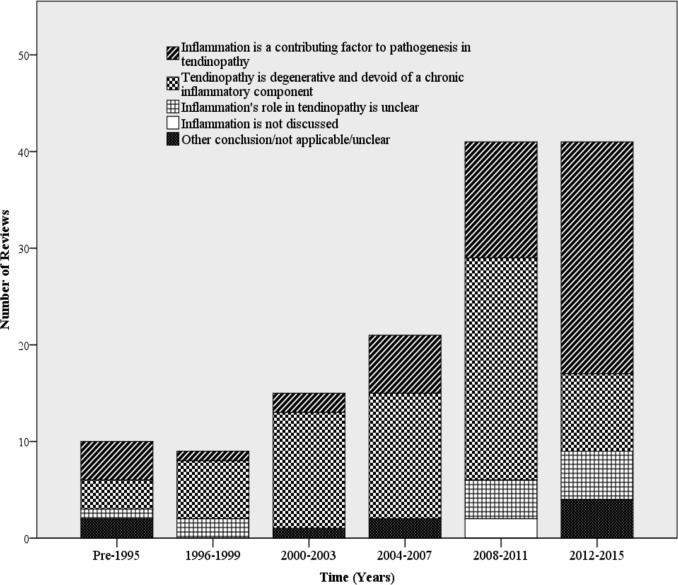
Of included reviews, 47.4% (n=65) concluded that tendinopathy is primarily degenerative with little to no chronic inflammatory component and 35.8% (n=49) concluded that inflammation plays a role in tendinopathy. 8.8% (n=12) discussed a potential for inflammation to contribute to the pathogenesis of tendinopathy but remained sceptical or their conclusions could not be easily determined. 8% (n=11) of included reviews’ conclusions did not fit into one of these categories: three discussed the pathogenesis of tendinopathy without any mention of inflammation or discussion on degenerative theories.17–19 Three did not discuss pathogenesis but evaluated risk factors of tendinopathy.20–22 Four reviews discussed or evaluated the association between fluoroquinolone use and tendinopathy/rupture; however, they did not substantially engage with the inflammation versus degeneration debate.23–26 The final review whose conclusions did not fit into a predefined category systematically assessed the association between tendinopathy and diabetes mellitus but did not discuss the inflammation versus degeneration debate.27
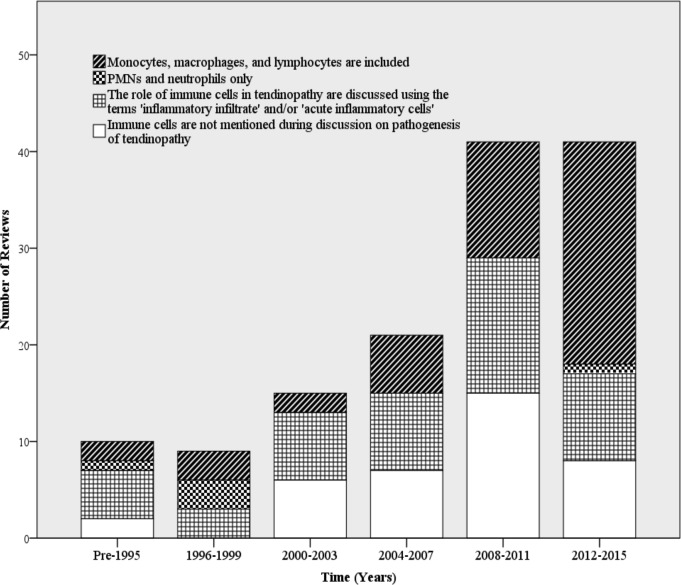
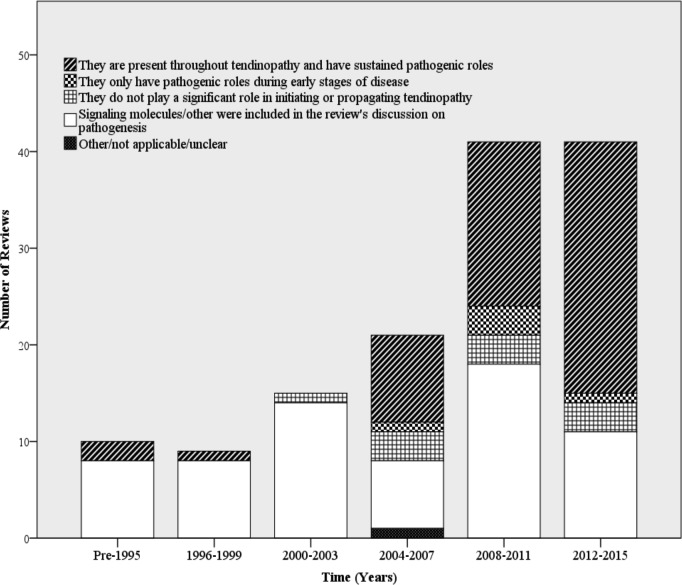
Although a significant portion of the literature concluded no inflammatory component to tendinopathy, the proportion of reviews supporting an inflammatory component increased substantially between 2012 and 2015 (figure 2). Previous discussions focused on the lack of an ‘inflammatory infiltrate’, referring to the known absence of neutrophils in diseased tendon tissue. However, more recent discussions on the pathogenesis of tendinopathy have included potential roles for monocytes, macrophages and lymphocytes to contribute to tendinopathy (figure 3). There has also been a surge in the interest of signalling molecules, molecular effectors and inflammatory activation pathways in initiating and propagating tendinopathy (figure 4). Prior to 2004, there were little to no reviews that included these effectors as an integral part of the discussion and/or conclusion about the role of inflammation in tendinopathy.
Figure 2.
Time trends in conclusions about the role of inflammation in tendinopathy (intervals of 3 years).
Figure 3.
Time trends in types of immune cells included in discussions about the role of inflammation in tendinopathy (intervals of 3 years). PMNs, polymorphonuclear neutrophils.
Figure 4.
Time trends in conclusions about the role of signalling molecules and or molecular effectors on tendinopathy (intervals of 3 years).
Potential correlates of the conclusion that inflammation has a role in tendinopathy: cells and molecules incorporated into discussions
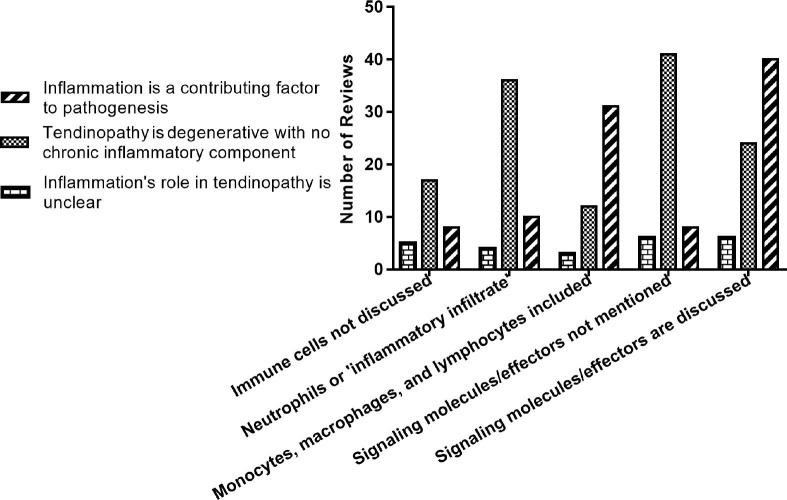
Conclusions about the role of inflammation in tendinopathy were stratified by immune cell type and inclusion of signalling molecules/effectors to determine if associations were present (figure 5). Reviews that discussed neutrophils or an ‘inflammatory infiltrate’ largely concluded that there is not an inflammatory component to tendinopathy. The same trend was present in reviews that did not discuss any immune cell type. However, reviews that included discussions on monocytes and lymphocytes largely concluded that there is an inflammatory component to tendinopathy. There was little difference on the types of immune cells discussed in reviews whose conclusion about the role of inflammation was ambiguous or not clearly stated.
Figure 5.
Conclusions about the role of inflammation according to immune cells and molecules discussed.
Similar associations exist for the inclusion or exclusion of signalling molecules/effectors and conclusions about inflammation in tendinopathy. To be categorised as ‘including’ these molecules, a review could have discussed any cytokine, activation pathway or molecular effector with a focus on its role in initiating and/or propagating pathology. They did not have to be proinflammatory or anti-inflammatory pathways or molecules to be included; however, most reviews that were categorised as including these in their discussions focused on inflammatory cytokines and activations pathways (eg, cyclo-oxygenase, nuclear factor-κβ, prostaglandin E1, prostaglandin E2, interleukin 6, interleukin 10, tumour necrosis factor-α). Reviews that did not include these molecules/effectors largely concluded no inflammatory component to tendinopathy. Reviews that did include these molecules mostly supported an inflammatory paradigm; however, a sizeable portion of these reviews were ambivalent.
A binary logistic regression was performed to ascertain any effects inclusion of monocytes, macrophages, lymphocytes and signalling molecules on the likelihood that a review concludes inflammation plays a role in tendinopathy. Only reviews that explicitly supported or refuted an inflammatory paradigm to tendinopathy were included in the model (n=113). The logistic regression model was statistically significant: x2(2)=35.4, p<0.001. The model explained 36.1% (Nagelkerke R2) of the variance in conclusions about the role of inflammation in tendinopathy and correctly classified 75.2% of cases. Reviews including monocytes and lymphocytes in their discussions were 5.23 times more likely to conclude that inflammation was important than reviews that did not, p<0.001. Reviews that included signalling molecules, effectors and activation pathways in their discussions on pathogenesis were 4.92 times more likely to conclude that inflammation is important than reviews that did not, p=0.003.
Potential correlates of the conclusion that inflammation has a role in tendinopathy: quality indicators
Conclusions about the role of inflammation in tendinopathy were stratified by the reviews’ total quality score and whether the review was a systematic review of the literature. There were very few systematic reviews that met inclusion criteria (n=18) and discussed the inflammation versus degeneration paradigm (n=13). Of the included systematic reviews that discussed the inflammation debate, the majority supported an inflammatory component: eight reviews supported an inflammatory component, two reviews rejected it and three reviews’ conclusions about inflammation were ambiguous.
A binary logistic regression was performed to ascertain the effects of these quality indicators on the likelihood that a review concludes inflammation plays a role in tendinopathy. Again, only reviews that explicitly supported or refuted an inflammatory paradigm to tendinopathy were included in the model (n=113). The logistic regression model was statistically significant: x2(3)=31.852, p<0.001. The model explained 33% (Nagelkerke R2) of the variance in conclusions about the role of inflammation in tendinopathy and correctly classified 75.2% of cases. Increasing quality score was associated with an increased likelihood to support an inflammatory component to tendinopathy, p=0.015. However, if the review was systematic it was not significantly more likely to support an inflammatory paradigm. This is likely due to the low number and wide range in the quality of systematic reviews (range 3–10, median=5).
Potential correlates of the conclusion that inflammation has a role in tendinopathy: journal type, impact factor and number of citations
Included reviews’ conclusions about the role of inflammation in tendinopathy were stratified by journal type, journal impact factor and number of citations per year to determine if any associations were present. Most included reviews were published in either clinical (n=50) or hybrid (n=39) orthopaedic journals. Only five reviews were published in basic science orthopaedic journals. The rest of included reviews were published in non-orthopaedic journals: 19 in basic science journals, 12 in clinical journals and 12 in other types. Among most journal types no given conclusion was vastly over-represented. However, in clinical journals (orthopaedic and non-specific), the 'degenerative' hypothesis represented the majority of conclusions. In non-orthopaedic basic science journals, the majority of published reviews supported an inflammatory hypothesis. Journal impact factor did not correlate to any given conclusion. The number of citations per year did not correlate with any given conclusion, however, the top five reviews (>33 citations/year) all concluded there was no inflammatory component to tendinopathy.
A binary logistic regression was performed to ascertain any effects journal type, impact factor and number of citations per year had on the likelihood that a review concludes inflammation plays a role in tendinopathy. Only reviews that explicitly supported or refuted an inflammatory paradigm to tendinopathy were included in the model (n=113). The logistic regression model was statistically significant: x2(7)=16.348, p=0.022. The model explained 18.2% (Nagelkerke R2) of the variance in conclusions about the role of inflammation in tendinopathy and correctly classified 65.2% of cases. If the review was published in a basic science journal it was 12 times more likely to support an inflammatory component to tendinopathy than if it was not (p=0.042). Journal impact factor and number of citations per year were not associated with any given conclusion about the role of inflammation in tendinopathy.
Discussion
This systematic review and quantitative analysis of published reviews prior to 2016 documented growing support for an inflammatory paradigm in the literature. This paralleled an increased discussion of monocytes, macrophages, lymphocytes, signalling molecules and growth factors. The adoption and/or rejection of a causal link between inflammation and tendinopathy was significantly associated with characterisation of the ‘inflammatory component’ in tendinopathy: reviews with a robust definition of inflammation were five times more likely to conclude a causal association. Reviews that limited discussion to neutrophils were significantly more likely to refute an inflammatory component to tendinopathy. We also documented that reviews of higher quality or those that included critical analyses of relevant primary literature were more likely to conclude inflammation is important in tendinopathy.
Our results are consistent with reviews that have suggested that the current ‘degenerative’ disease paradigm minimises inflammation’s complex role in tendinopathy by precluding all inflammatory pathways and cells on the pretext that an ‘inflammatory infiltrate’ is absent.9 28 29 The transition away from ‘tendinitis’ to ‘tendinosis’ has been described as an oversimplification of inflammation’s complex role in tendinopathy, which potentially acts through a variety of immune and stromal cell types, activation pathways, cytokines and growth factors.10 12 13 Our results support these notions: prior to 2011, most reviews refuted an inflammatory component to tendinopathy and did so by citing an absence of neutrophils in diseased tendon tissues. Reviews have only recently begun to include a greater variety of cell types and signalling molecules into their discussions on tendinopathy. This increased interrogation of leucocytes and cytokines in tendinopathy has led to most reviews concluding that inflammation may play a key role in pathology. Of note, recent studies have identified that resident stromal fibroblasts from diseased human tendons show a proinflammatory phenotype.28–30 Activated stromal fibroblasts are known to release chemokines that promote retention of immune cells in inflamed musculoskeletal soft tissues.31 These findings imply that non-immune cells are also implicated in tendon inflammatory processes, and that understanding interplay between immune cells and stromal populations is critical to advancing knowledge of disease mechanism.
When analysing the causes of tendinopathy it is important to appreciate the anatomical features of these tissues: the tendon proper is surrounded by tissues known as the paratenon and epitenon. The extent to which inflammation is present or not present in each layer or the tendon proper should be determined separately in order to fully understand the potential role of inflammation in tendinopathy. In addition, tissue inflammation signatures are known to change between temporal phases of acute and chronic inflammation.28–30 32 Tendinopathy has a multifactorial aetiology: ageing, overuse, genetic, epigenetic, and inflammation all play roles in its course. In this review, we showed that there is dissonance in the literature surrounding the presence and/or potential roles of these immune cells/mediators in pathology. Further work is required to characterise the phenotypes of cells populating diseased tendons.
This research may provide evidence useful for the orthopaedic community to standardise the ‘inflammatory component’ to tendinopathy. Standardisation of what constitutes and ‘inflammatory component’ may help to facilitate discussions of inflammation’s role in tendinopathy among scientists, and advance the discovery and dissemination of novel pathways in the pathogenesis of tendinopathy that could lead to the development of effective therapeutic strategies to reduce tendon disabilities.
Limitations
Our review was designed to be generalisable: reviews of all levels of quality and focus were included to present an accurate representation of consensus over time. The literature search design was fairly broad and was limited to publications in the English language. Additionally, data collected on a review’s conclusions, discussions and classification of the ‘inflammatory component’ in tendinopathy were categorised into predefined groups. These groups were sufficiently general to provide intuitive data at the cost of precision: we did not collect specific data on the exact immune cells and/or mediators discussed. Due to this, our statistical model was more generalisable and representative of contemporary opinions in the medical literature but was less specific due to the use of categorised groups.
There is the potential for misclassification bias. This potential bias was minimised by piloting all our data extractions to assess reproducibility and validity. Additionally, we reduced error by having an independent panel categorise data that was deemed not sufficiently reproducible. Our overall agreement was excellent, suggesting that misclassification bias was unlikely to have a significant impact on our conclusions.
We wanted both narrative and systematic reviews in this study since both provide unique contributions, contexts and discussions to the debate surrounding inflammation in tendinopathy. Additionally, we wanted to design a way to determine their quality using the same standardised tool. Our quality assessment, which was modified from existing validated tools, presents a limitation. Despite this, we believe it allowed us to differentiate subtle differences between the quality of the included reviews while also giving systematic reviews the weight they deserve.
Acknowledgments
MJM acknowledges the Harvard University Francis H Burr, Class of 1909 Scholarship Prize Fund and the National Collegiate Athletic Association Post Graduate Scholarship Award for financial support to complete his degree at Oxford.
Footnotes
Contributors: MJM, SJS, SGD and AJC formulated and designed the study. MJM and SK designed and conducted the systematic search. MJM and MSR reviewed all abstracts for inclusion/exclusion criteria. MJM read full texts for inclusion/exclusion criteria, extracted all data from included full-texts, conducted statistical analyses and authored the manuscript. MJM, SJS and SGD designed and performed the validation study for our data extraction sheets. All authors contributed in the editing and approved the final manuscript.
Funding: SGD received funding from Arthritis Research UK grant 20506 and an Oxford UCB Prize Fellowship in Biomedical Sciences. SJS received funding from Arthritis Research UK grants 20087. Work from NDORMS is supported by the Oxford NIHR Biomedical Research Centre (BRC).
Competing interests: None declared.
Patient consent: Not required.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data sharing statement: The results of the data extraction for each individual article included in the study are compiled onto an Excel spreadsheet. This information may be used for future studies; however, if any reader is interested in acquiring some or all these data, inquiries can be sent to moscam2@gmail.com.
References
- 1. Khan KM, Cook JL, Bonar F, et al. . Histopathology of common tendinopathies. Update and implications for clinical management. Sports Med 1999;27:393–408. [DOI] [PubMed] [Google Scholar]
- 2. Khan KM, Cook JL, Kannus P, Maffulli N, et al. . Time to abandon the "tendinitis" myth. BMJ 2002;324:626–7. 10.1136/bmj.324.7338.626 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Khan KM, Maffulli N. Tendinopathy: an Achilles' heel for athletes and clinicians. Clin J Sport Med 1998;8:151–4. [PubMed] [Google Scholar]
- 4. Weiler JM. Medical modifiers of sports injury. The use of nonsteroidal anti-inflammatory drugs (NSAIDs) in sports soft-tissue injury. Clin Sports Med 1992;11:625–44. [PubMed] [Google Scholar]
- 5. Aström M, Rausing A. Chronic Achilles tendinopathy. A survey of surgical and histopathologic findings. Clin Orthop Relat Res 1995;316:151–64. [PubMed] [Google Scholar]
- 6. Jozsa LG, Kannus P. Human tendons: anatomy, physiology, and pathology. Champaign, IL: Human Kinetics, 1997. [Google Scholar]
- 7. Puddu G, Ippolito E, Postacchini F. A classification of Achilles tendon disease. Am J Sports Med 1976;4:145–50. 10.1177/036354657600400404 [DOI] [PubMed] [Google Scholar]
- 8. Hashimoto T, Nobuhara K, Hamada T. Pathologic evidence of degeneration as a primary cause of rotator cuff tear. Clin Orthop Relat Res 2003;415:111–20. 10.1097/01.blo.0000092974.12414.22 [DOI] [PubMed] [Google Scholar]
- 9. Movin T, Gad A, Reinholt FP, et al. . Tendon pathology in long-standing achillodynia. Biopsy findings in 40 patients. Acta Orthop Scand 1997;68:170–5. 10.3109/17453679709004002 [DOI] [PubMed] [Google Scholar]
- 10. Rees JD, Stride M, Scott A. Tendons--time to revisit inflammation. Br J Sports Med 2014;48:1553–7. 10.1136/bjsports-2012-091957 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Rees JD, Wilson AM, Wolman RL. Current concepts in the management of tendon disorders. Rheumatology 2006;45:508–21. 10.1093/rheumatology/kel046 [DOI] [PubMed] [Google Scholar]
- 12. Dean BJ, Franklin SL, Carr AJ. A systematic review of the histological and molecular changes in rotator cuff disease. Bone Joint Res 2012;1:158–66. 10.1302/2046-3758.17.2000115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Dean BJ, Gettings P, Dakin SG, et al. . Are inflammatory cells increased in painful human tendinopathy? A systematic review. Br J Sports Med 2016;50:1–6. 10.1136/bjsports-2015-094754 [DOI] [PubMed] [Google Scholar]
- 14. Moher D, Liberati A, Tetzlaff J, et al. . PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 2009;151:264–9. 10.7326/0003-4819-151-4-200908180-00135 [DOI] [PubMed] [Google Scholar]
- 15. Magnan B, Bondi M, Pierantoni S, et al. . The pathogenesis of Achilles tendinopathy: A systematic review. Foot and Ankle Surgery 2014;20:154–9. 10.1016/j.fas.2014.02.010 [DOI] [PubMed] [Google Scholar]
- 16. Shea BJ, Grimshaw JM, Wells GA, et al. . Development of AMSTAR: a measurement tool to assess the methodological quality of systematic reviews. BMC Med Res Methodol 2007;7:1–7. 10.1186/1471-2288-7-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Bureau N. Calcific Tendinopathy of the Shoulder. Semin Musculoskelet Radiol 2013;17:080–4. 10.1055/s-0033-1333941 [DOI] [PubMed] [Google Scholar]
- 18. Laseter JT, Russell JA. Anabolic steroid-induced tendon pathology: a review of the literature. Med Sci Sports Exerc 1991;23:1–3. [PubMed] [Google Scholar]
- 19. Uhthoff HK, Sarkar K. Calcifying tendinitis. Int Orthop 1978;2:187–93. 10.1007/BF00266076 [DOI] [Google Scholar]
- 20. Blanco I, Krähenbühl S, Schlienger RG. Corticosteroid-associated tendinopathies: an analysis of the published literature and spontaneous pharmacovigilance data. Drug Saf 2005;28:633–43. 10.2165/00002018-200528070-00005 [DOI] [PubMed] [Google Scholar]
- 21. Stephenson AL, Wu W, Cortes D, et al. . Tendon Injury and Fluoroquinolone Use: A Systematic Review. Drug Saf 2013;36:709–21. 10.1007/s40264-013-0089-8 [DOI] [PubMed] [Google Scholar]
- 22. van der Worp H, van Ark M, Roerink S, et al. . Risk factors for patellar tendinopathy: a systematic review of the literature. Br J Sports Med 2011;45:446–52. 10.1136/bjsm.2011.084079 [DOI] [PubMed] [Google Scholar]
- 23. Kaleagasioglu F, Olcay E. Fluoroquinolone-induced tendinopathy: etiology and preventive measures. Tohoku J Exp Med 2012;226:251–8. 10.1620/tjem.226.251 [DOI] [PubMed] [Google Scholar]
- 24. Khaliq Y, Zhanel GG. Fluoroquinolone-associated tendinopathy: a critical review of the literature. Clin Infect Dis 2003;36:1404–10. 10.1086/375078 [DOI] [PubMed] [Google Scholar]
- 25. Lewis T, Cook J. Fluoroquinolones and tendinopathy: a guide for athletes and sports clinicians and a systematic review of the literature. J Athl Train 2014;49:422–7. 10.4085/1062-6050-49.2.09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Melhus Åsa. Fluoroquinolones and tendon disorders. Expert Opin Drug Saf 2005;4:299–309. 10.1517/14740338.4.2.299 [DOI] [PubMed] [Google Scholar]
- 27. Shi L, Rui YF, Li G. Alterations of tendons in patients with diabetes mellitus: A systematic review. Int Ortho 2015;39:1465–73. [DOI] [PubMed] [Google Scholar]
- 28. Dakin SG, Martinez FO, Yapp C, et al. . Inflammation activation and resolution in human tendon disease. Sci Transl Med 2015;7:311ra173–13. 10.1126/scitranslmed.aac4269 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Dakin SG, Buckley CD, Al-Mossawi MH, et al. . Persistent stromal fibroblast activation is present in chronic tendinopathy. Arthritis Res Ther 2017;19:16 10.1186/s13075-016-1218-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Dakin SG, Newton J, Martinez FO, et al. . Chronic inflammation is a feature of Achilles tendinopathy and rupture. Br J Sports Med 2018;52:359–67. 10.1136/bjsports-2017-098161 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Patel R, Filer A, Barone F, et al. . Stroma: fertile soil for inflammation. Best Pract Res Clin Rheumatol 2014;28:565–76. 10.1016/j.berh.2014.10.022 [DOI] [PubMed] [Google Scholar]
- 32. Mosca MJ, Carr AJ, Snelling SJB, et al. . Differential expression of alarmins-S100A9, IL-33, HMGB1 and HIF-1α in supraspinatus tendinopathy before and after treatment. BMJ Open Sport Exerc Med 2017;3:e000225 10.1136/bmjsem-2017-000225 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjsem-2017-000332supp001.docx (34KB, docx)