Abstract
Resistant hypertension (failure to achieve target blood pressures with three or more antihypertensive drugs including a diuretic) is an important and preventable cause of stroke. Hypertension is highly prevalent in China (>60% of persons above age 65), and only ~6% of hypertensives in China are controlled to target levels. Most strokes occur among persons with resistant hypertension; approximately half of strokes could be prevented by blood pressure control. Reasons for uncontrolled hypertension include (1) non-compliance; (2) consumption of substances that aggravated hypertension, such as excess salt, alcohol, licorice, decongestants and oral contraceptives; (3) therapeutic inertia (failure to intensify therapy when target blood pressures are not achieved); and (4) diagnostic inertia (failure to investigate the cause of resistant hypertension). In China, an additional factor is lack of availability of appropriate antihypertensive therapy in many healthcare settings. Sodium restriction in combination with a diet similar to the Cretan Mediterranean or the DASH (Dietary Approaches to Stop Hypertension) diet can lower blood pressure in proportion to the severity of hypertension. Physiologically individualised therapy for hypertension based on phenotyping by plasma renin activity and aldosterone can markedly improve blood pressure control. Renal hypertension (high renin/high aldosterone) is best treated with angiotensin receptor antagonists; primary aldosteronism (low renin/high aldosterone) is best treated with aldosterone antagonists (spironolactone or eplerenone); and hypertension due to overactivity of the renal epithelial sodium channel (low renin/low aldosterone; Liddle phenotype) is best treated with amiloride. The latter is far more common than most physicians suppose.
Keywords: resistant hypertension, sodium, salt, diet, renin, aldosterone, personalized medicine
Many interventions are combined to reduce recurrent stroke by ~80%.1 Of these, the three most important are probably smoking cessation, a Mediterranean diet and blood pressure control.1 Smoking cessation and diet are discussed in other articles in this issue of the journal. While smoking cessation and diet are the responsibility of the patient, with the help of the physician, probably the most important intervention that is mainly the responsibility of the physician is to achieve good blood pressure control. Resistant hypertension, defined sometimes as failure to achieve target blood pressure levels despite three drugs including a diuretic, has been estimated to account for ~20% of hypertension in the past, and increases with age. Causes of resistant hypertension are summarised in box 1.
Box 1. Causes of resistant hypertension.
-
Non-compliance
-
Consumption of substances that aggravate hypertension
Salt, licorice, non-steroidal anti-inflammatory drugs*, excess alcohol, oral contraceptives, decongestants.
-
Therapeutic inertia
Failure to intensify therapy when blood pressure is not controlled.
-
Diagnostic inertia
Failure to investigate the cause of resistant hypertension.
*Except for sulindac.15
A recent study in the UK estimated a prevalence of resistant hypertension of 10%–11% among apparently compliant patients aged 70 and older.2 A Swedish population-based study reported that 88% of patients with stroke had uncontrolled hypertension, and that ~45% of all strokes could be attributed to uncontrolled hypertension.3 Achieving blood pressure control in 72% of the population around London, Canada, reduced stroke by half, between 1977 and 1984.4 How that was accomplished will be described below.
In China there is a major problem with stroke, and a major problem with uncontrolled hypertension. The age-standardised/sex-standardised prevalence of hypertension is 37.2%; only 36% of hypertensives are aware of it, only 22.9% are on treatment and only 5.7% are controlled.5 Above age 65, ~60% of the population are hypertensive and the control rates are very low5 (figure 1). In part this may be due to unavailability of effective antihypertensive medications. Among 3362 primary healthcare sites (2758 rural, 604 urban), 8.1% stocked no antihypertensive medications, only 33.8% stocked all four classes in common use and only 32.7% of all sites stocked high-value medications. Village clinics in the western region of China had the lowest availability. Few high-value medications were prescribed (11.2% of all prescription records).6 The cost of medication is a major barrier to blood pressure control, but it should be noted that there is great underprescription of diuretics, particularly given that much of hypertension in China is driven by an inordinately high intake of salt. The most common medication prescribed is a calcium channel antagonist (55.2% of patients); only 1% of patients were prescribed hydrochlorothiazide (probably the most cost-effective antihypertensive drug), only 1.3% were prescribed indapamide and only 4.5% were prescribed any diuretic.6 This inappropriate prescribing pattern represents a major opportunity for improvement.
Figure 1.
Prevalence, awareness, treatment and control of hypertension in China. Data are shown stratified by age and sex. (Reproduced with permission from Elsevier: Lu et al. 5)
In rural areas of China, stroke mortality between 2003 and 2013 increased from 111/100 000 to 150/100 000 population per year. This was nearly three times the mortality from coronary artery disease (which increased in rural areas from 16/100 000 to 67/100 000 per year). (The steeper increase in coronary mortality, probably attributable largely to changes in diet with prosperity, is discussed in the article on diet in stroke prevention in this issue.)
What is to be done about this? First, blood pressure must be measured systematically in all patients, at virtually every healthcare encounter. A programme of public education similar to the ‘Know your number’ programme in the USA some 20 years ago should be mounted, and patients encouraged to measure their blood pressure regularly at home if they have ever been told their blood pressure was elevated, or if there is a family history of hypertension or stroke. Healthcare institutions should be required to have on hand at least a minimum list of medications for hypertension; a body such as the Chinese Hypertension Society should draw up a list of essential medications for hypertension, such as those listed in table 1. Physicians and other healthcare providers with the authority to prescribe medications should be expected to treat hypertension routinely, and the government should consider subsidising the cost of an approved list of antihypertensive medications; combination medications would probably be best tolerated and more cost-effective, with better adherence.
Table 1.
Essential medications for hypertension that should be available in all healthcare centres in China
| Class | Drug |
| Diuretic | For example, hydrochlorothiazide |
| ACE inhibitor | For example, lisinopril, quinapril |
| Angiotensin receptor blocker | For example, telmisartan |
| Calcium channel blocker | For example, amlodipine |
| Aldosterone antagonist | Spironolactone, eplerenone |
| Renal sodium channel blocker | Amiloride |
| Beta blocker | For example, bisoprolol* |
| Alpha blocker | For example, doxazosin |
*Not metoprolol because of its short duration of action and huge individual differences in metabolism due to copy number variants of CYP2D6.28
Non-compliance
A common cause of resistant hypertension is non-compliance. Patients may not take their medication because of cost, adverse effects or a system of health beliefs, such as that drugs are harmful, or a result of a conspiracy between the pharmaceutical industry and physicians. A particularly difficult problem is the patient who mistakenly attributes any and all symptoms to adverse effects of drugs.7 A blinded N of one crossover study with capsules containing active drug in one bottle and placebo in the other may be helpful in sorting this out, but it may be fruitless in patients whose mind is made up. If asked in a non-judgemental way (eg, how often do you forget to take your medication?), about half of patients will admit to non-compliance.8 Advice about the truly causal adverse effects of medications, as opposed to the long list of incidental adverse effects reported on the long lists in which symptoms are equally common on placebo versus active drug, may help the patient to resume medication. True adverse effects such as cough with ACE inhibitors can be avoided by switching to other medications such as an angiotensin receptor blocker (ARB). Persistence is better with medications that have fewer adverse effects, in particular with ARBs.9
Substances that patients consume that aggravate hypertension
A high salt intake has historically been a particular problem in China. Government efforts and changes in food preservation related to increased availability of refrigeration have made some progress, but survival free of hypertension is still significantly related to the ratio of sodium intake to potassium intake. Most sodium intake is from added condiments and processed foods, and the intake of processed foods has been increasing.10
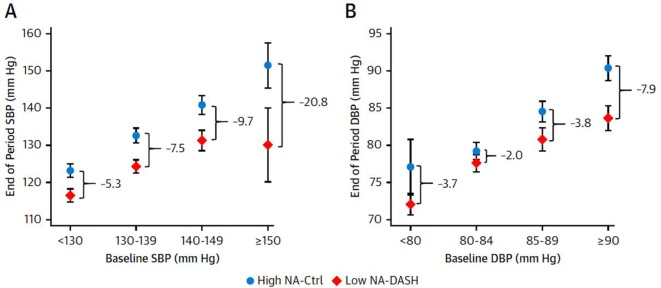
In a recent US study,11 the combination of a low sodium intake and a diet rich in fruits, vegetables and low-fat dairy products, and reduced in saturated fat and cholesterol (the Dietary Approaches to Stop Hypertension diet), markedly reduced blood pressure, and the effects were greater in patients with higher blood pressures. In patients with systolic pressure >150 mm Hg, the reduction of systolic blood pressure with this combination was 20.7 mm Hg, and with diastolic pressure >90 mm Hg, the reduction was 7.9 mm Hg (figure 2). Diet for stroke prevention is discussed in another article in this issue of the journal; essentially the recommended diet is based on the Cretan Mediterranean diet.
Figure 2.
The combined effects of low sodium and the DASH diet according to baseline blood pressure. The mean end of period SBP measurements (mm Hg) by strata of (A) baseline SBP (<130, 130–139, 140–149, >150 mm Hg) or (B) baseline DBP (<80, 80–84, 85–89, >90 mm Hg). The mean blood pressure values are presented by the high-sodium control diet (circle) or the low-sodium DASH diet (diamond). Differences between diets were determined using linear regression comparing baseline changes in systolic or diastolic blood pressure adjusted for age, female sex, black race and baseline body mass index. Bars indicate 95% CIs. Ctrl, control; DASH, Dietary Approaches to Stop Hypertension trial; DBP, diastolic blood pressure; NA, sodium; SBP, systolic blood pressure. (Reproduced with permission from Elsevier: Juraschek et al.11)
Patients with hypertension should be encouraged to reduce their salt intake towards 2–3 g per day, avoiding added salt and also limiting intake of foods containing high amounts of sodium. Sparing use of light soya sauce, more spices, herbs, ginger, lemon juice, vinegar and hot sauce can help reduce salt intake. Excess alcohol, licorice, birth control pills and non-steroidal anti-inflammatory agents (NSAIDs) may also account for resistant hypertension.
Licorice has mineralocorticoid effects causing hypertension and hypokalaemia,12 and is found in candy, tea and some tonics. Oral contraceptives cause hypertension; there may be several mechanisms. In a rat model combined hormonal therapy induced endothelial dysfunction and upregulation of the ACE-1 gene and angiotensin receptor gene13; however, combined hormonal therapy in postmenopausal women raises the level of aldosterone and lowers the level of plasma renin.14
The only NSAID that does not raise blood pressure is sulindac15; it is highly protein-bound, so very little is filtered into the renal tubule; furthermore, it is a prodrug that is oxidised in the renal tubule back to the inactive form, which does not inhibit formation of vasodilator prostaglandins.15–17
Heavy alcohol use (>3 drinks per day) doubles the risk of hypertension18 and increases the risk of non-lobar intracerebral haemorrhage.19
Therapeutic inertia
Therapeutic inertia has many sources. Busy physicians may focus on other health problems of patients with hypertension, such as diabetes, arthritis, gastrointestinal complaints, headache and so on. When a patient presents with complaints in other areas, the hypertension is not the focus of the clinic visit and may be overlooked. Furthermore, patients and physicians may be reluctant to increase the antihypertensive therapy, in the mistaken belief that white coat hypertension is benign. It is not.20 However, therapeutic inertia can be overcome, essentially by measuring blood pressure at every visit, and a policy that antihypertensive therapy will be intensified whenever target blood pressures are not achieved. In the North American Symptomatic Carotid Endarterectomy Trial,21 we essentially overcame therapeutic inertia by sending a stiff letter to the site investigator every time the medication was not increased, when a patient’s blood pressure exceeded the level specified in the protocol. This resulted in a remarkable reduction of intracranial haemorrhage to only 0.5% of strokes, at a time when 20% of strokes were due to intracerebral haemorrhage.
Diagnostic inertia
When a patient does not respond to usual antihypertensive therapy, it is crucial to consider whether the therapy is appropriate for that patient and to investigate the underlying physiology that is driving the hypertension. Failure to consider reasons why patients’ blood pressure is not controlled may be termed ‘Diagnostic inertia’.22
Among patients with resistant hypertension, an approach that is efficacious is what I call ‘Physiologically Individualized Therapy’ (PhysRx), based on phenotyping by plasma renin activity and aldosterone. This is a form of personalised medicine. An early variant of this was recommended by Laragh in the early 1970s, based on plasma renin activity.23 The results described above (a 50% reduction of stroke in our community)4 were the result of a programme of improved detection and treatment of hypertension mounted by the Department of Family Medicine, in combination with my hypertension clinic, basing therapy on stimulated plasma renin levels. Patients with a high salt intake (very common in China) have low plasma renin levels if unstimulated. By giving a dose of furosemide (0.5 mg/kg) before drawing blood for measurement of plasma renin activity (PRA), the range of plasma levels is spread out, with normal and high PRA levels moving up so that low and suppressed plasma renin can be appreciated.24 I gave oral furosemide to patients living locally 4 hours before blood draws; for those coming from several hours away, we gave intravenous furosemide 30 min before the blood draw. In patients who are already taking a diuretic, ACE inhibitor or ARB, it may not be necessary to give furosemide, as these drugs all stimulate plasma renin activity.o
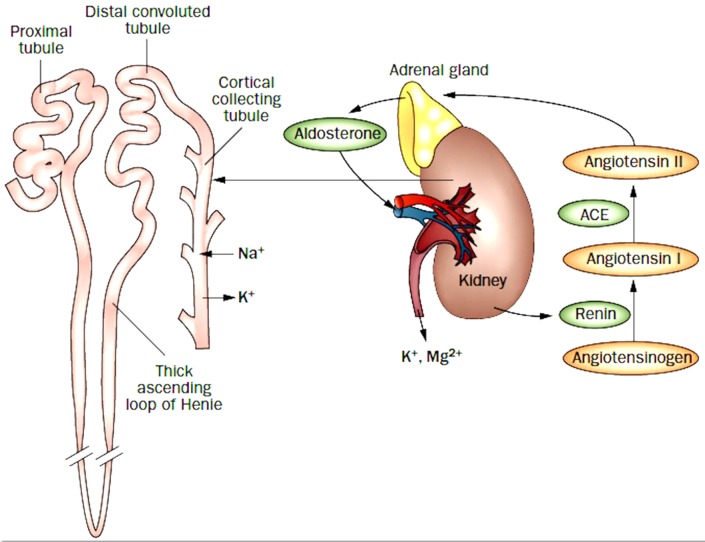
Egan and colleagues25 carried out a randomised trial of therapy based on plasma renin activity, but did not measure plasma aldosterone. In 77 patients randomised to usual care versus renin-based therapy, they reported lower systolic pressures, a trend to improved blood pressure control and a greater reduction of medication needed among patients with volume hypertension (ie, low renin hypertension). Measuring PRA is useful, but it is important to measure stimulated plasma aldosterone. Once rare causes of hypertension are eliminated (such as pheochromocytoma, adult coarctation of the aorta, and licorice ingestion), there are essentially three types of hypertension, all understood by studying the renin/angiotensin/aldosterone axis (figure 3). (1) Renal hypertension: when the kidney senses low blood pressure it releases renin, activating the formation of angiotensin and in turn release of aldosterone. Patients with renal hypertension, renal cysts, obstruction, stones, renovascular hypertension and parenchymal renal disease including hypertensive nephrosclerosis (which might be thought of as microvascular renovascular hypertension) have a high PRA and secondary hyperaldosteronism. (2) Adrenocortical hypertension: when there is hyperplasia of the adrenal cortex with overproduction of aldosterone, salt and water retention suppresses PRA, so plasma aldosterone is high and PRA is low. (3) Liddle phenotype: when there is a mutation of the renal tubular epithelial sodium channel (ENac, SCNBB1), or a mutation of genes that affect the function of ENaC, there is salt and water retention, which suppresses both PRA and aldosterone. As shown in table 2, the treatment is different for these three types of hypertension. Renal hypertension, with high renin and secondary aldosteronism, is best treated with an angiotensin receptor blocker (ARB). Primary aldosteronism, with low renin and high aldosterone, is best treated with an aldosterone antagonist. Spironolactone can be used for women, but spironolactone in adequate doses for primary hyperaldosteronism usually causes gynecomastia and mastalgia in men, so eplerenone is preferred. Abnormalities of the ENac or variants that affect the function of ENac (low renin/low aldosterone; Liddle phenotype) are specifically treated with amiloride. There are several variants of Liddle syndrome (mutations of ENaC; SCNBB1) and variants of several other genes that affect the function of ENaC (GRK, NEDD4L, CYP4A11, NPPA, UMOD), resulting in Liddle phenotype.26 27
Figure 3.
The physiology of therapy for resistant hypertension. When the kidney senses low blood pressure, renin is released, leading to production of angiotensin II. In turn, angiotensin II activates production of aldosterone by the adrenal cortex, which leads to salt and water retention. Under healthy conditions, this system is turned off once normal body water levels are restored. Impairment of this system can cause hypertension. Renal hypertension is associated with high renin and secondary hyperaldosteronism; in primary hyperaldosteronism the salt and water retention suppresses renin production, so aldosterone levels are high with low renin levels. Abnormalities of the renal tubular epithelial sodium channel cause salt and water retention, suppressing both renin and aldosterone. To identify the best primary therapy for patients with resistant hypertension, the physiological mechanism underlying the blood pressure elevation must be identified (table 1). (Reproduced with permission from Nature: Spence29).
Table 2.
Physiologically individualised therapy for resistant hypertension based on phenotyping by plasma renin activity and aldosterone
| Primary hyperaldosteronism (usually due to bilateral adrenocortical hyperplasia) |
Liddle phenotype (mutations of ENac and of other genes affecting the function of ENac) |
Renal/renovascular | |
| Renin | Low* | Low | High |
| Aldosterone | High* | Low | High |
| Primary treatment | Aldosterone antagonist (spironolactone or eplerenone) Amiloride for men where eplerenone is not available (rarely surgery)‡ |
Amiloride | Angiotensin receptor blocker or renin inhibitor† (rarely revascularisation) |
*Levels of plasma renin and aldosterone must be interpreted in the light of the medication the patient is taking at the time of sampling. In a patient taking an angiotensin receptor blocker (which would elevate renin and lower aldosterone), a plasma renin that is in the low normal range for that laboratory, with a plasma aldosterone in the high normal range, probably represents primary hyperaldosteronism, for the purposes of adjusting medical therapy.
†ACE inhibitors are less effective because of aldosterone escape via non-ACE pathways such as chymase and cathepsin.
‡It should be stressed that this approach is suitable for tailoring medical therapy in resistant hypertensives; further investigation would be required to justify adrenalectomy or renal revascularisation.
(Reproduced with permission from Elsevier: Spence30).
A clinical trial in Africa provides evidence that physiologically individualised therapy for resistant hypertension as described above markedly improved blood pressure control. Patients in three hypertension clinics, in Nigeria, Kenya and South Africa, whose blood pressure was not controlled to <140 mm Hg systolic or <90 mm Hg diastolic were allocated to usual care versus physiologically individualised therapy based on the algorithm described in table 2. In Kenya this approach had no effect; patients attended clinic less frequently, compliance was less and amiloride was not available. ‘When only the sites in Nigeria and South Africa were considered, systolic control was obtained in 15.0% of UC vs. 78.6% of PhysRx (P<0.0001), diastolic control in 45.0% vs 71.4% (P=0.04), and control of both in 15.0% vs. 66.7% (P=0.0001). If only the Nigerian site (where patients were randomized to the two treatment strategies) is considered, systolic control was obtained in 15% of UC vs 85% of PhysRx (P=0.0001), and diastolic control in 45% vs 75% (P=0.11)’.26 In this trial, the biggest difference in medication between the usual care arm and the PhysRx arm was in the prescription of amiloride: 19%of patients allocated to PhysRx vs 2.8% on usual care (P=0.02). A randomised substudy of this approach is planned in China.
Conclusion
Resistant hypertension is a major cause of stroke and can be overcome. Measures that can markedly improve blood pressure control include sodium restriction in combination with a diet similar to the Cretan Mediterranean diet, and physiologically individualised therapy based on renin/aldosterone phenotyping. Improved availability of effective medications in all healthcare settings in China will be needed to achieve a marked improvement in blood pressure control and thereby a marked reduction in stroke.
Acknowledgments
None
Footnotes
Contributors: JDS wrote this paper.
Funding: This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient consent: Not required.
Provenance and peer review: Commissioned; externally peer reviewed.
Data sharing statement: No additional data is available.
Guest chief editor: J David Spence
References
- 1. Hackam DG, Spence JD. Combining multiple approaches for the secondary prevention of vascular events after stroke: a quantitative modeling study. Stroke 2007;38:1881–5. 10.1161/STROKEAHA.106.475525 [DOI] [PubMed] [Google Scholar]
- 2. Sinnott SJ, Smeeth L, Williamson E, et al. Trends for prevalence and incidence of resistant hypertension: population based cohort study in the UK 1995-2015. BMJ 2017;358:j3984 10.1136/bmj.j3984 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Li C, Engström G, Hedblad B, et al. Blood pressure control and risk of stroke: a population-based prospective cohort study. Stroke 2005;36:725–30. 10.1161/01.STR.0000158925.12740.87 [DOI] [PubMed] [Google Scholar]
- 4. Spence JD. Antihypertensive drugs and prevention of atherosclerotic stroke. Stroke 1986;17:808–10. 10.1161/01.STR.17.5.808 [DOI] [PubMed] [Google Scholar]
- 5. Lu J, Lu Y, Wang X, et al. Prevalence, awareness, treatment, and control of hypertension in China: data from 1·7 million adults in a population-based screening study (China PEACE Million Persons Project). Lancet 2017;390:2549–58. 10.1016/S0140-6736(17)32478-9 [DOI] [PubMed] [Google Scholar]
- 6. Su M, Zhang Q, Bai X, et al. Availability, cost, and prescription patterns of antihypertensive medications in primary health care in China: a nationwide cross-sectional survey. Lancet 2017;390:2559–68. 10.1016/S0140-6736(17)32476-5 [DOI] [PubMed] [Google Scholar]
- 7. Spence JD, Dresser GK. Overcoming Challenges With Statin Therapy. J Am Heart Assoc 2016;5:e002497 10.1161/JAHA.115.002497 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Haynes RB, Taylor DW, Sackett DL, et al. Can simple clinical measurements detect patient noncompliance? Hypertension 1980;2:757–64. 10.1161/01.HYP.2.6.757 [DOI] [PubMed] [Google Scholar]
- 9. Marentette MA, Gerth WC, Billings DK, et al. Antihypertensive persistence and drug class. Can J Cardiol 2002;18:649–56. [PubMed] [Google Scholar]
- 10. Du S, Neiman A, Batis C, et al. Understanding the patterns and trends of sodium intake, potassium intake, and sodium to potassium ratio and their effect on hypertension in China. Am J Clin Nutr 2014;99:334–43. 10.3945/ajcn.113.059121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Juraschek SP, Miller ER. 3rd, Weaver CM, Appel LJ. Effects of Sodium Reduction and the DASH Diet in Relation to Baseline Blood Pressure. J Am Coll Cardiol 2017;70:2841–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Penninkilampi R, Eslick EM, Eslick GD. The association between consistent licorice ingestion, hypertension and hypokalaemia: a systematic review and meta-analysis. J Hum Hypertens 2017;31:699–707. 10.1038/jhh.2017.45 [DOI] [PubMed] [Google Scholar]
- 13. Olatunji LA, Seok YM, Igunnu A, et al. Combined oral contraceptive-induced hypertension is accompanied by endothelial dysfunction and upregulated intrarenal angiotensin II type 1 receptor gene expression. Naunyn Schmiedebergs Arch Pharmacol 2016;389:1147–57. 10.1007/s00210-016-1272-0 [DOI] [PubMed] [Google Scholar]
- 14. Ahmed AH, Gordon RD, Ward G, et al. Effect of Combined Hormonal Replacement Therapy on the Aldosterone/Renin Ratio in Postmenopausal Women. J Clin Endocrinol Metab 2017;102:2329–34. 10.1210/jc.2016-3851 [DOI] [PubMed] [Google Scholar]
- 15. Wong DG, Spence JD, Lamki L, et al. Effect of non-steroidal anti-inflammatory drugs on control of hypertension by beta-blockers and diuretics. Lancet 1986;1:997–1001. 10.1016/S0140-6736(86)91271-7 [DOI] [PubMed] [Google Scholar]
- 16. Ciabattoni G, Cinotti GA, Pierucci A, et al. Effects of sulindac and ibuprofen in patients with chronic glomerular disease. Evidence for the dependence of renal function on prostacyclin. N Engl J Med 1984;310:279–83. 10.1056/NEJM198402023100502 [DOI] [PubMed] [Google Scholar]
- 17. Cibattoni G, Boss AH, Patrignani P, et al. Effects of sulindac on renal and extrarenal eicosanoid synthesis. Clin Pharmacol Ther 1987;41:380–3. 10.1038/clpt.1987.44 [DOI] [PubMed] [Google Scholar]
- 18. Klatsky AL, Gunderson E. Alcohol and hypertension: a review. J Am Soc Hypertens 2008;2:307–17. 10.1016/j.jash.2008.03.010 [DOI] [PubMed] [Google Scholar]
- 19. Chen CJ, Brown WM, Moomaw CJ, et al. Alcohol use and risk of intracerebral hemorrhage. Neurology 2017;88:2043–51. 10.1212/WNL.0000000000003952 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Spence JD. Dilemmas in diagnosing and managing hypertension: is white coat hypertension benign? Can J Cardiol 2015;31:580–2. 10.1016/j.cjca.2015.02.031 [DOI] [PubMed] [Google Scholar]
- 21. Barnett HJ, Taylor DW, Eliasziw M, et al. Benefit of carotid endarterectomy in patients with symptomatic moderate or severe stenosis. North American Symptomatic Carotid Endarterectomy Trial Collaborators. N Engl J Med 1998;339:1415–25. 10.1056/NEJM199811123392002 [DOI] [PubMed] [Google Scholar]
- 22. Spence JD, Rayner BL. J Curve and Cuff Artefact, and Diagnostic Inertia in Resistant Hypertension. Hypertension 2016;67:32–3. 10.1161/HYPERTENSIONAHA.115.06562 [DOI] [PubMed] [Google Scholar]
- 23. Laragh JH, Baer L, Brunner HR, et al. Renin, angiotensin and aldosterone system in pathogenesis and management of hypertensive vascular disease. Am J Med 1972;52:633–52. 10.1016/0002-9343(72)90054-X [DOI] [PubMed] [Google Scholar]
- 24. Wallach L, Nyarai I, Dawson KG. Stimulated renin: a screening test for hypertension. Ann Intern Med 1975;82:27–34. 10.7326/0003-4819-82-1-27 [DOI] [PubMed] [Google Scholar]
- 25. Egan BM, Basile JN, Rehman SU, et al. Plasma Renin test-guided drug treatment algorithm for correcting patients with treated but uncontrolled hypertension: a randomized controlled trial. Am J Hypertens 2009;22:792–801. 10.1038/ajh.2009.63 [DOI] [PubMed] [Google Scholar]
- 26. Akintunde A, Nondi J, Gogo K, et al. Physiological Phenotyping for Personalized Therapy of Uncontrolled Hypertension in Africa. Am J Hypertens 2017;30:923–30. 10.1093/ajh/hpx066 [DOI] [PubMed] [Google Scholar]
- 27. Jones ES, Spence JD, Mcintyre AD, et al. High Frequency of Variants of Candidate Genes in Black Africans with Low Renin-Resistant Hypertension. Am J Hypertens 2017;30:478–83. 10.1093/ajh/hpw167 [DOI] [PubMed] [Google Scholar]
- 28. Spence JD. A single fixed-dose combination for all patients is bad medicine. Can J Cardiol 2014;30:513–6. 10.1016/j.cjca.2014.01.018 [DOI] [PubMed] [Google Scholar]
- 29. Spence JD. Secondary stroke prevention. Nat Rev Neurol 2010;6:477–86. 10.1038/nrneurol.2010.114 [DOI] [PubMed] [Google Scholar]
- 30. Spence JD. Lessons from Africa: the importance of measuring plasma renin and aldosterone in resistant hypertension. Can J Cardiol 2012;28:254–7. 10.1016/j.cjca.2011.11.010 [DOI] [PubMed] [Google Scholar]