Abstract
The Phaseolus vulgaris (common bean), a worldwide vegetable of high consumption, can act as a nutritional supplement in the diet of oversized individuals to reduce weight. Studies have demonstrated the existence of molecules capable of inhibiting the breakdown of carbohydrates via inhibition of both α-amylases and glycosidases. Here, we describe a novel property of the Phaseolus vulgaris: inhibition of thrombotic cardiovascular events. Using assays to test platelet aggregation and secretion, and flow cytometry against the surface expression of P-Selectin. We show that bean extracts significantly reduced adenosine 5′-diphosphate and arachidonic acid induced-platelet aggregation. The mechanism underlying such effect appears to be mediated by AKT, since AKT hypo-phosphorylation decreases the “inside out” activation of platelets. In sum, our results support the hypothesis that common beans are nutritional ingredients that help reduce the risk of cardiovascular diseases associated with platelet hyper-reactivity.
Keywords: Phaseolus vulgaris, extracts, platelets aggregation, cardiovascular diseases, AKT
INTRODUCTION
Cardiovascular diseases (CVD) are the leading cause of death in the world, ~30% of all deaths are due to vascular affections (1) (17.3 million/year), a number expected to increase by 2030 (2). Atherosclerosis is among the most frequent causes of CVD. In the arterial walls, the accumulation of lipids (atheroma) that contain cholesterol, inflammatory cells, collagen, and elastin (3) stimulate the proliferation and inflammation of the endothelium. Over time, the atheroma, via interactions with extracellular matrix factors, promotes the recruitment of new platelets. The accumulation of disc-shaped fragments at the site of arterial damage further increases the inflammatory process that consequently interferes with blood flow (4). Thus, aggregation of platelets represents the cellular bridge between inflammation and thrombosis (4). Indeed, pharmacological agents that block platelet activity significantly reduce the formation of thrombotic and inflammatory events (5), key steps in the progression of CVDs. As a consequence, platelet aggregation inhibitors are widely used as primary and secondary prevention methods against ischemic insults (6,7).
There are lifestyle factors that help reduce the risk of CVDs. For example, regular exercise and avoiding the consumption of tobacco are two factors that significantly reduce the formation of atheromas (8,9). Other lifestyle factor considered to decrease CVD risk is the consumption of fruits and vegetables (10). In fact, there is consensus that the beneficial effect of a balanced diet is not limited to the nutritional content, but it provides factors that protect arteries against toxic agents that increase the risk of CVDs (11). In here, we examine the group of leguminous plants that have edible seeds and are consumed in five continents as essential components of the diet. Particularly, we studied the common bean (Phaseolus vulgaris) that is consumed either as dry or fresh grain (12).
In Chile, this legume (locally called poroto) is consumed in three vegetative stages. The first is when the grain is not yet possible to separate from the pod, its color is green (green beans) and is consumed mainly in salads and typical dishes; the second form is the fresh grain, this is harvested when the grain has reached its final maturity, is separated from the pod and is consumed mainly in traditional cooking, this grain still conserves the water it obtained from the plant (fresh beans); the third presentation is stored grain, the grain is harvested at the end of their maturation and drying to be stored without risk of contamination, this type of grain is consumed in the winter season.
Previous studies have shown the beneficial properties of bean consumption as antioxidant (13,14), appetite inhibitor (15,16), and carbohydrate metabolism (16,17). Here, we provide evidence indicating that Phaseolus vulgaris inhibits platelet aggregation. Overall, our aim is to transform diet guides in detailed food catalogues that help people choose the appropriate types and amounts of foods that favor a healthy condition.
MATERIALS AND METHODS
All protocols were approved by the Ethics Committee of University of Talca (registry number 2017005) in accordance with the Declaration of Helsinki (approved by the 18th World Medical Assembly in Helsinki, Finland, 1964).
Plant samples
Phaseolus vulgaris varieties at different growing seasons (green beans, with green pods and green grain; fresh beans, shelled) were collected and processed as indicated below. All beans were obtained from the Regional Supply Center of Talca (CREA).
Aqueous extract
The aqueous beans extract was prepared as follows: beans (1 kg) were washed with pure water, macerated with a blender (Oster) and dissolved in water. The homogenate was filtered with gauze, then cotton wool, and Whatman paper (pore size 0.45 μm). The extract was lyophilized (FreeZone 6, Labconco, Kansas City, MO, USA) and stored (−70°C).
Methanol extract
Beans were washed with water, macerated as above and mixed in 95% methanol (1/1 w/v ratio; Sigma-Aldrich Co., St. Louis, MO, USA). The mixture was sonicated (Transsonic 700/H, Elma Schmidbauer GmbH, Singen, Germany) for 5 min and filtered (Wathman paper, 0.45 μm). To remove most of the solvent, the mixture was placed on a rotary evaporator (RE 111-B461, Büchi Labortechnik AG, Hendrik-Ido-Ambacht, The Netherlands), then lyophilized (Freezone 6, Labconco) and stored at −70°C.
The chemical composition of the bean extracts was analyzed by the Institute of Chemistry at Universidad of Talca according to standardized methods (Association of Official Analytical Chemists) (18).
Determination of total phenols concentration
The total amount of phenols in each bean extract was determined by a modified version of the Folin-Ciocalteu method (19). Briefly, 0.1 mL of a given extract was mixed with 0.5 mL of the Folin-Ciocalteu phenol reagent (Merck, Darmstadt, Germany). After 5 min of incubation, 0.5 mL of sodium carbonate (Na2CO3; 10% w/v) was added. Then, after 15 min at room temperature (RT), the absorbance (A) of the homogenate was measured at 640 nm (SPECTRO-UV11, mrclab, Haifa, Israel). The total concentration of phenols in the extract was expressed as mg of gallic acid equivalents/g dry weight.
Determination of antioxidant activity
Quantification of 2,2-diphenyl-1-picrylhydrazyl (DPPH) (Fluka Chemie GmbH, Buchs, Switzerland) was measured using the method of von Gadow et al. (20). Briefly, 0.1 mL of the bean extracts were mixed with 2 mL of DPPH (20 mg/L) and incubated for 8 min (RT). The absorbance of the gallic acid at different concentrations was used as a standard, and the DPPH capture expressed as mg of chromogenic acid equivalents/g fresh weight (Table 1).
Table 1.
Chemical analysis and profile of antioxidants present in aqueous and methanolic extracts of fresh beans
| Fresh beans | ||
|---|---|---|
|
|
||
| Aqueous | Methanolic | |
| Humidity (%) | 0 | 0 |
| Ashes (%) | 7.8 | 7.9 |
| Protein (%) | 26 | 33 |
| Total fat (%) | 10 | 3.6 |
| ENN (%) | 55.2 | 55.4 |
| Total fiber (%) | 1 | 1 |
| Total phenols (GAE/g dry weight) | 5.4 | 4.1 |
| DPPH (CAE/g fresh weight) | 5.0 | 4.8 |
GAE, gallic acid equivalents; CAE, chromogenic acid equivalents.
Preparation of platelets
Platelets were prepared as previously described (21). Blood samples were obtained from 3 healthy adult volunteers (BD Vacutainer®, BD Biosciences, Franklin Lakes, NJ, USA), none of which took non-steroidal anti-inflammatory drugs for at least 8 days prior to the study (informed consent was obtained from participants). Samples were collected in falcon tubes containing sodium citrate 3.2% (ratio blood/anticoagulant=9/1 v/v). After centrifugation of the citrated blood, at 250 g for 10 min, a platelet rich plasma (PRP) was obtained. After counting the platelets in a hematological counter (Bayer Advia 60, Diamond Diagnostics, Holliston, MA, USA), PRPs were washed in 4-(2-hydroxyethyl)-1-piperazineethane-sulfonic acid (HEPES)-Tyrode buffer plus PGE1 (120 nmol/L) and re-suspended in HEPES-Tyrode buffer to a final dilution of 200×106 platelets/mL. Platelets were maintained at 4°C for 2 h.
Determination of platelet aggregation and adenosine triphosphate (ATP) secretion
Platelet aggregation and ATP secretion were monitored by a turbidometric method using light transmission in an aggregometer (22) (Chrono-Log, Havertown, PA, USA). Briefly, aliquots of 480 μL of PRPs were placed in the aggregometer and incubated with 20 μL of either saline (control) or bean extract (1 mg/mL). Following 3 min of incubation, 20 μL of the platelet aggregation agonists adenosine 5′-diphosphate (ADP) (8 μM) or arachidonic acid (AA) (1 mM) were added to the samples. The aggregation assays were quantified after 6 min, at the maximum height of the curve. To quantify ATP secretion, 50 μL of luciferin/luciferase was added to the samples 2 min prior the activation assays. Platelet aggregation and ATP secretion [maximal amplitude (%)] were calculated using the AGGRO/LINK software (Chrono-Log). The platelet aggregation and ATP secretion inhibitory activities were expressed as percent inhibition compared with that measured for the vehicle alone (saline). All measurements were performed from three separate platelet donors.
Measurement of P-selectin surface expression
Measurements of P-selectin surface expression were achieved by modifying platelet activation previously described by Frojmovic et al. (23). Briefly, 480 mL of platelets (200×106 platelets/mL) were pre-incubated with either 20 μL of vehicle (dimethyl sulfoxide) or bean extract (1 mg/mL) for 3 min. After 6 min of stimulation at 37°C with ADP 8 μM, platelets were kept at 4°C. To determine P-selectin expression, 50 μL of the platelet sample was incubated with anti-CD62-PE and anti-CD61-FITC, and maintained for 25 min in the dark. After a quench-dilution step (<4-fold), samples were analyzed in an Accuri C6 flow cytometer (BD Biosciences). Distinct platelet populations were gated on cell size using forward vs. side scatter, the CD61 positivity separated to distinguish it from the electronic noise. Light scatter and fluorescence channels were set at logarithmic gain, and 5,000 events per sample were analyzed. Fluorescence intensities of differentially stained populations were expressed as a mean channel value using the BD Accuri C6 Software (BD Biosciences).
Western blotting
Platelets (200×106 platelets/mL) were pre-incubated with either the vehicle (0.9% NaCl) or bean extracts (1 mg/mL) for 10 min. Then platelets were lysed with 0.2 mL of lysis buffer in ice (30 min), and heated at 95°C (10 min). Equal quantities of total protein (30 mg) were subjected to SDS-PAGE, and transferred under reduced conditions to a nitrocellulose membrane. To detect AKT1 phosphorylation, a polyclonal antibody against anti-phospho-AKT1 (AKT1, phosphor S473, Abnova, Taipei, Taiwan) was used. A monoclonal antibody against anti-γ-tubulin [γ-tubulin monoclonal antibody (4D11), Thermo Scientific, Waltham, MA, USA] was used as the control. After incubation with a second antibody conjugated to horseradish peroxidase [peroxidase-conjugated AffiniPure goat anti-rabbit IgG (H+L), Jackson Immunoresearch Laboratories Inc., West Grove, PA, USA], samples were developed via chemiluminescence (western blotting substrate, Thermo Scientific) under a radiographic film. Bands were analyzed using ImageJ plugins. All measurements were performed from 6 separate platelet donors.
Statistical analysis
Data were analyzed using SPSS version 17.0 (SPSS Inc., Chicago, IL, USA) and expressed as mean±standard error of mean (SEM). Results were expressed as percent of aggregation over the vehicle (considered as 100%). Statistically significant differences were analyzed by paired or unpaired t-test and one-way analysis of variance (ANOVA) followed by Tukey’s post-hoc test. P-values <0.05 were considered significant.
RESULTS
Inhibitory effect of bean extracts on platelet aggregation activity
Platelet aggregation was achieved by the use of two agonists, ADP or AA. As shown in Table 2, both the aqueous and methanolic bean extracts obtained from fresh beans showed anti-platelet action. In the presence of ADP 8 μM, the aqueous and methanolic bean extracts inhibited platelet aggregation by 28±3% and 49±5%, respectively (P<0.01). A similar effect was observed when 1 mM AA was used as the activator of platelet aggregation. The % of inhibition were 28±4% (P<0.05) and 35±3% (P<0.01) for the aqueous and methanolic bean extracts respectively. When aqueous and methanolic extracts obtained from green beans were studied, similar anti-platelet aggregation effects were observed. Overall, the results indicate that both fresh and green varieties have equivalent activity.
Table 2.
Inhibition of platelet aggregation activity
| Vegetative stage | Extract (1 mg/mL) | Maximum platelet aggregation (%) | |
|---|---|---|---|
|
| |||
| ADP 8 μM | AA 1 mM | ||
| Green beans | Aqueous | 85±3 | 80±2 |
| Methanolic | 82±5 | 79±5 | |
| Fresh beans | Aqueous | 64±3* | 60±4* |
| Methanolic | 45±5** | 54±3** | |
| Control | 89±4 | 83±5 | |
Results were expressed as % of inhibition from the maximum platelet aggregation. Two activators: ADP (8 μM) and AA (1 mM) were used to induce maximum platelet aggregation. Saline 0.9% was used as control.
P<0.05;
P<0.01.
Effects of bean extracts on platelet secretion
Platelets contain important inflammatory/hemostatic mediators within their granules. As a result of the activation, platelets release the granules to the extracellular space. To determine whether aqueous and methanolic bean extracts modulate platelet secretion, we measured the levels of ATP in the extracellular media. Our results (Table 3) indicate that the methanolic extract obtained from fresh beans (1 mg/mL) inhibited the secretion of ATP mediated by ADP (8 μM) and AA (1 mM). The maximum blockades of ATP secretion were 34±11% and 29±13% (P<0.05) for ADP and AA, respectively. Similar results were obtained when methanolic extracts obtained from green beans were used. The maximum inhibitions of ATP secretion were 24±9% and 28±14% for ADP and AA, respectively. Interestingly, the aqueous extracts of both varieties, fresh and green beans, showed no significant inhibitory effects on platelet secretion.
Table 3.
Inhibition of platelet secretion
| Vegetative stage | Extract (1 mg/mL) | Platelet secretion (%) | |
|---|---|---|---|
|
| |||
| ADP 8 μM | AA 1 mM | ||
| Green beans | Aqueous | 90±2 | 94±6 |
| Methanolic | 68±9* | 65±14* | |
| Fresh beans | Aqueous | 79±18 | 88±13 |
| Methanolic | 59±11* | 64±13* | |
| Control | 90±9 | 90±2 | |
ATP levels were measured after 5 min of incubation with the respective bean extracts. Results were expressed as % of inhibition from the maximal platelet secretion. ADP (8 μM) and AA (1 mM) were used as agonists of platelet aggregation. Saline 0.9% was used as control.
P<0.05.
Effects of bean extracts on platelet P-selectin expression
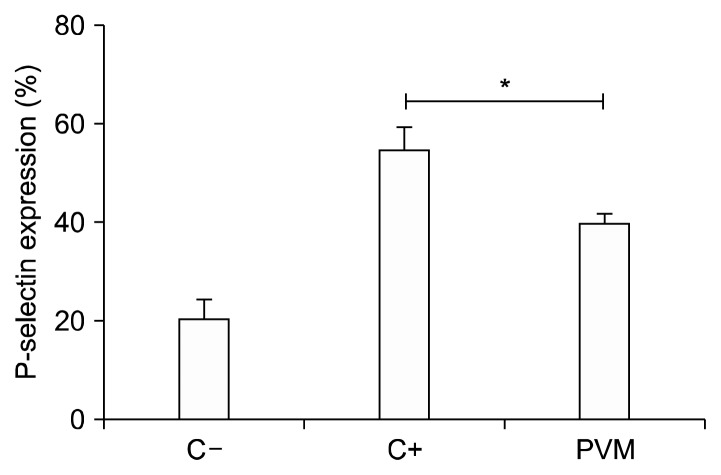
Expression of P-selectin on the membrane of activated platelets is one of the most widely used markers to evidence the degree of platelet activation. This same parameter determined in vitro in human platelets of healthy volunteers is useful to determine the degree of reactivity of the platelets on exposure to an agonist in the presence or absence of the different extracts and thus determine if the extracts are able to reduce the activation of platelets. Because most favorable results were presented to the methanol extract of bean stew, this was subjected to tests to determine its effect on the expression of the P-selectin molecule actively involved in platelet activity. As shown in Fig. 1, the shelled bean methanol extract at a concentration of 1 mg/mL induced a decrease in the expression of P-selectin mediated ADP 8 μM almost 15% (P<0.05). Thus, the shelled bean methanolic extract inhibits platelet activation, decreasing the expression of P-selectin.
Fig. 1.
P-selectin expression on the platelets surface. C−, control saline; C+, adenosine 5′-diphosphate (ADP) 8 μM; PVM, ADP 8 μM+methanolic extracts of green beans. *P<0.05.
Methanolic bean extracts alter the phosphorylation state of AKT-1
To investigate the plausible mechanism by which bean extracts inhibited platelet activation and aggregation, we evaluated the phosphorylation of AKT1, a key intermediary in intracellular platelet signaling. Fig. 2 shows a decrease in phosphorylation of AKT (S473) using methanolic fresh bean extract to 1 mg/mL, using it as agonist ADP 8 μM aggregation. This result by western blot shows that the methanol extract of Phaseolus vulgaris is capable of inhibiting AKT phosphorylation, blocking subsequent platelet activation and aggregation.
Fig. 2.
Western blots analysis of P-Akt in platelets treated with vehicle and methanolic extracts of Phaseolus vulgaris. Adenosine 5′-diphosphate (ADP) (4 μM) was used to activate platelets aggregation. FB, fresh beans. Detection of α-tubulin was used as a control for protein loading and image analysis.
DISCUSSION
The mortality associated with CVD directly relates to the prevalence in the population (24). To promote a healthy lifestyle, and an appropriate diet in the population, it is essential to encourage consumption of foods rich in fruits and vegetables. It is necessary to create an educated “nutritional catalog” that relates the benefits of a healthy diet with factors that reduce CVD risks.
One of the main pathophysiological events involved in CVDs is the aggregation of platelets, a central phenomenon in thrombosis and atherogenesis (25,26).
In the case of Phaseolus vulgaris, with the results presented in Table 1 and 2, it is shown that Phaseolus vulgaris has an antiplatelet effect and that this effect is dependent upon the state of maturation of the grain. Although Phaseolus vulgaris has multiple effects on human health, there are no reports about its specific effect on platelets. To our knowledge, this study is the first to describe this direct and effect dependent on the state of maturation of the beans on the activity of platelets.
In order to partially characterize the mechanism by which Phaseolus vulgaris exerts its anti-platelet effect, it was necessary to study the methanol extracts, and the ability of these to suppress platelet secretion.
P-selectin plays an essential role in platelet function, also considered important in thrombotic events (27–29). P-selectin is a cell adhesion glycoprotein in resting platelets found in the alpha granules (30). When platelets are activated, due to secretory mechanisms of surrounding cells as histamine and thrombin, P-selectin is translocated to the membrane and binds to its ligand, promoting aggregation (30). The effects against expression of this adhesion protein were evident in the platelets administered prior to the methanol extracts.
Similar results on the activity of platelets and platelet P-selectin expression versus the presence of ADP were obtained using other vegetables and fruits, such as tomatos (31,32). The proposed reduction in P-selectin on the platelet membrane relaxation mechanism is the cAMP levels in platelets by modulating the activity of PKA (27).
The AKT/PKB signaling protein is fundamental in signaling from the inside out and from the outside in, triggered by the activation of platelets (33,34). Our results demonstrate that the mechanism of action of shelled bean extracts involves the inhibition of AKT phosphorylation at residue (S473). This may explain the effect on activation, aggregation, and secretion in treated platelets. This effect takes on a central importance in the context of developing new strategies that aim to lower the high rates of death from ischemic CVD.
A study by Vaiyapuri et al. (35) showed that nobiletin, a flavonoid, has the ability to inhibit platelet function. There is a growing interest in the search for extracts or compounds of natural origin that are capable of reducing the risk factors of CVD, this is mainly due to the low adherence of pharmacological therapies and a large number of undesirable side effects presented by groups pharmacological agents such as statins. Current pharmacotherapy, although it has managed to counteract the formation of atheromatous plaques in individuals at risk, has increased thrombotic events due, among other aspects, to the endothelial erosion that they produce. These clinical data motivate the search for clinical therapies of natural origin that together with reducing risk factors, do not increase thrombotic events secondarily. It should be noted that extracts of Phaseolus vulgaris are used today in the reduction of body weight in overweight and obese individuals. These extracts could be an alternative to reduce the side effects of pharmacological therapies or be alone therapies tending to reduce the risk factors.
ACKNOWLEDGEMENTS
The authors thank Dr. Juan Carlos Tapia, Universidad of Talca, for helping in discussion and writing of this paper.
Footnotes
AUTHOR DISCLOSURE STATEMENT
The authors declare no conflict of interest.
REFERENCES
- 1.Mendis S, Puska P, Norrving B, editors. WHO. Global atlas on cardiovascular disease prevention and control: policies, strategies and interventions. World Health Organization; Geneva, Switzerland: 2011. pp. 4–8. [Google Scholar]
- 2.Smith SC, Jr, Collins A, Ferrari R, Holmes DR, Jr, Logstrup S, McGhie DV, Ralston J, Sacco RL, Stam H, Taubert K, Wood DA, Zoghbi WA World Heart Federation; American Heart Association; American College of Cardiology Foundation; European Heart Network; European Society of Cardiology. Our time: a call to save preventable death from cardiovascular disease (heart disease and stroke) J Am Coll Cardiol. 2012;60:2343–2348. doi: 10.1016/j.jacc.2012.08.962. [DOI] [PubMed] [Google Scholar]
- 3.Mimura J, Itoh K. Role of Nrf2 in the pathogenesis of atherosclerosis. Free Radic Biol Med. 2015;88:221–232. doi: 10.1016/j.freeradbiomed.2015.06.019. [DOI] [PubMed] [Google Scholar]
- 4.Willeit J, Kiechl S. Biology of arterial atheroma. Cerebrovasc Dis. 2000;10:1–8. doi: 10.1159/000047599. [DOI] [PubMed] [Google Scholar]
- 5.Grove EL, Würtz M, Thomas MR, Kristensen SD. Antiplatelet therapy in acute coronary syndromes. Expert Opin Pharmacother. 2015;16:2133–2147. doi: 10.1517/14656566.2015.1079619. [DOI] [PubMed] [Google Scholar]
- 6.Wang W, Zhang L, Liu W, Zhu Q, Lan Q, Zhao J. Antiplatelet agents for the secondary prevention of ischemic stroke or transient ischemic attack: a network meta-analysis. J Stroke Cerebrovasc Dis. 2016;25:1081–1089. doi: 10.1016/j.jstrokecerebrovasdis.2016.01.026. [DOI] [PubMed] [Google Scholar]
- 7.Bhatt DL, Bonaca MP, Bansilal S, Angiolillo DJ, Cohen M, Storey RF, Im K, Murphy SA, Held P, Braunwald E, Sabatine MS, Steg PG. Reduction in ischemic events with ticagrelor in diabetic patients with prior myocardial infarction in PEGASUS-TIMI 54. J Am Coll Cardiol. 2016;67:2732–2740. doi: 10.1016/j.jacc.2016.03.529. [DOI] [PubMed] [Google Scholar]
- 8.Metkus TS, Jr, Baughman KL, Thompson PD. Exercise prescription and primary prevention of cardiovascular disease. Circulation. 2010;121:2601–2604. doi: 10.1161/CIRCULATIONAHA.109.903377. [DOI] [PubMed] [Google Scholar]
- 9.Karmali KN, Lloyd-Jones DM, Berendsen MA, Goff DC, Jr, Sanghavi DM, Brown NC, Korenovska L, Huffman MD. Drugs for primary prevention of atherosclerotic cardiovascular disease: an overview of systematic reviews. JAMA Cardiol. 2016;1:341–349. doi: 10.1001/jamacardio.2016.0218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yu E, Rimm E, Qi L, Rexrode K, Albert CM, Sun Q, Willett WC, Hu FB, Manson JE. Diet, lifestyle, biomarkers, genetic factors, and risk of cardiovascular disease in the nurses’ health studies. Am J Public Health. 2016;106:1616–1623. doi: 10.2105/AJPH.2016.303316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Carbajal A, Ortega R. La dieta mediterránea como modelo de dieta prudente y saludable. Rev Chil Nutr. 2001;28:224–236. [Google Scholar]
- 12.Ulloa JA, Ulloa PR, Ramírez JCR, Rangel BEU. El frijol (Phaseolus vulgaris): su importancia nutricional y como fuente de fitoquímicos. Revista Fuente Año. 2011;3:5–9. [Google Scholar]
- 13.Frassinetti S, Gabriele M, Caltavuturo L, Longo V, Pucci L. Antimutagenic and antioxidant activity of a selected lectin-free common bean (Phaseolus vulgaris L.) in two cell-based models. Plant Foods Hum Nutr. 2015;70:35–41. doi: 10.1007/s11130-014-0453-6. [DOI] [PubMed] [Google Scholar]
- 14.Reyes-Martínez A, Almaraz-Abarca N, Gallardo-Velázquez T, González-Elizondo Mdel S, Herrera-Arrieta Y, Pajarito-Ravelero A, Alanís-Bañuelos RE, Torres-Morán MI. Evaluation of foliar phenols of 25 Mexican varieties of common bean (Phaseolus vulgaris L.) as antioxidants and varietal markers. Nat Prod Res. 2014;28:2158–2162. doi: 10.1080/14786419.2014.930855. [DOI] [PubMed] [Google Scholar]
- 15.Zaru A, Maccioni P, Riva A, Morazzoni P, Bombardelli E, Gessa GL, Carai MA, Colombo G. Reducing effect of a combination of Phaseolus vulgaris and Cynara scolymus extracts on operant self-administration of a chocolate-flavoured beverage in rats. Phytother Res. 2013;27:258–263. doi: 10.1002/ptr.4814. [DOI] [PubMed] [Google Scholar]
- 16.Spadafranca A, Rinelli S, Riva A, Morazzoni P, Magni P, Bertoli S, Battezzati A. Phaseolus vulgaris extract affects glycometabolic and appetite control in healthy human subjects. Br J Nutr. 2013;109:1789–1795. doi: 10.1017/S0007114512003741. [DOI] [PubMed] [Google Scholar]
- 17.Maccioni P, Colombo G, Riva A, Morazzoni P, Bombardelli E, Gessa GL, Carai MA. Reducing effect of a Phaseolus vulgaris dry extract on operant self-administration of a chocolate-flavoured beverage in rats. Br J Nutr. 2010;104:624–628. doi: 10.1017/S0007114510001017. [DOI] [PubMed] [Google Scholar]
- 18.Helrich KC. Official methods of analysis of the AOAC. 15th ed. Vol. 2. Association of Official Analytical Chemists; Gaithersburg, MD, USA: 1990. pp. 685–1213. [Google Scholar]
- 19.Coseteng MY, Lee CY. Changes in apple polyphenoloxidase and polyphenol concentrations in relation to degree of browning. J Food Sci. 1987;52:985–989. doi: 10.1111/j.1365-2621.1987.tb14257.x. [DOI] [Google Scholar]
- 20.von Gadow A, Joubert E, Hansmann CF. Comparison of the antioxidant activity of aspalathin with that of other plant phenols of rooibos tea (Aspalathus linearis), α-tocopherol, BHT, and BHA. J Agric Food Chem. 1997;45:632–638. doi: 10.1021/jf960281n. [DOI] [Google Scholar]
- 21.Rodríguez-Azúa R, Treuer A, Moore-Carrasco R, Cortacáns D, Gutiérrez M, Astudillo L, Fuentes E, Palomo I. Effect of tomato industrial processing (different hybrids, paste, and pomace) on inhibition of platelet function in vitro, ex vivo, and in vivo. J Med Food. 2014;17:505–511. doi: 10.1089/jmf.2012.0243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Born GVR, Cross MJ. The aggregation of blood platelets. J Physiol. 1963;168:178–195. doi: 10.1113/jphysiol.1963.sp007185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Frojmovic MM, Wong T, van de Ven T. Dynamic measurements of the platelet membrane glycoprotein IIb-IIIa receptor for fibrinogen by flow cytometry. Biophys J. 1991;59:828–837. doi: 10.1016/S0006-3495(91)82295-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.O’Donnell CJ, Elosua R. Factores de riesgo cardiovascular. Perspectivas derivadas del Framingham Heart Study. Rev Esp Cardiol. 2008;61:299–310. doi: 10.1157/13116658. [DOI] [PubMed] [Google Scholar]
- 25.Barrett NE, Holbrook L, Jones S, Kaiser WJ, Moraes LA, Rana R, Sage T, Stanley RG, Tucker KL, Wright B, Gibbins JM. Future innovations in anti-platelet therapies. Br J Pharmacol. 2008;154:918–939. doi: 10.1038/bjp.2008.151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Massberg S, Brand K, Grüner S, Page S, Müller E, Müller I, Bergmeier W, Richter T, Lorenz M, Konrad I, Nieswandt B, Gawaz M. A critical role of platelet adhesion in the initiation of atherosclerotic lesion formation. J Exp Med. 2002;196:887–896. doi: 10.1084/jem.20012044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Minamino T, Kitakaze M, Asanuma H, Tomiyama Y, Shiraga M, Sato H, Ueda Y, Funaya H, Kuzuya T, Matsuzawa Y, Hori M. Endogenous adenosine inhibits P-selectin-dependent formation of coronary thromboemboli during hypoperfusion in dogs. J Clin Invest. 1998;101:1643–1653. doi: 10.1172/JCI635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Burger PC, Wagner DD. Platelet P-selectin facilitates atherosclerotic lesion development. Blood. 2003;101:2661–2666. doi: 10.1182/blood-2002-07-2209. [DOI] [PubMed] [Google Scholar]
- 29.Lefer DJ, Flynn DM, Buda AJ. Effects of a monoclonal antibody directed against P-selectin after myocardial ischemia and reperfusion. Am J Physiol. 1996;270:H88–H98. doi: 10.1152/ajpheart.1996.270.1.H88. [DOI] [PubMed] [Google Scholar]
- 30.Vestweber D, Blanks JE. Mechanisms that regulate the function of the selectins and their ligands. Physiol Rev. 1999;79:181–213. doi: 10.1152/physrev.1999.79.1.181. [DOI] [PubMed] [Google Scholar]
- 31.O’Kennedy N, Crosbie L, Whelan S, Luther V, Horgan G, Broom JI, Webb DJ, Duttaroy AK. Effects of tomato extract on platelet function: a double-blinded crossover study in healthy humans. Am J Clin Nutr. 2006;84:561–569. doi: 10.1093/ajcn/84.3.561. [DOI] [PubMed] [Google Scholar]
- 32.Dutta-Roy AK, Crosbie L, Gordon MJ. Effects of tomato extract on human platelet aggregation in vitro. Platelets. 2001;12:218–227. doi: 10.1080/09537100120058757. [DOI] [PubMed] [Google Scholar]
- 33.Guidetti GF, Canobbio I, Torti M. PI3K/Akt in platelet integrin signaling and implications in thrombosis. Adv Biol Regul. 2015;59:36–52. doi: 10.1016/j.jbior.2015.06.001. [DOI] [PubMed] [Google Scholar]
- 34.Angiolillo DJ, Ferreiro JL. Platelet adenosine diphosphate P2Y12 receptor antagonism: benefits and limitations of current treatment strategies and future directions. Rev Esp Cardiol. 2010;63:60–76. doi: 10.1016/S0300-8932(10)70010-5. [DOI] [PubMed] [Google Scholar]
- 35.Vaiyapuri S, Roweth H, Ali MS, Unsworth AJ, Stainer AR, Flora GD, Crescente M, Jones CI, Moraes LA, Gibbins JM. Pharmacological actions of nobiletin in the modulation of platelet function. Br J Pharmacol. 2015;172:4133–4145. doi: 10.1111/bph.13191. [DOI] [PMC free article] [PubMed] [Google Scholar]