Abstract
The objective of this study was to investigate the physicochemical and textural properties of heat-induced gels prepared with pork muscles irradiated with gamma rays, electron beams, and X-rays. Pork muscles were irradiated at 5 kGy using the different irradiation sources, and heat-induced gels were prepared from the irradiated pork muscles at a protein concentration of 5 mg/mL. Each irradiation treatment produced lower water-holding capacity, protein solubility, apparent viscosity, hardness, springiness, cohesiveness, gumminess, and chewiness than the control gel prepared with non-irradiated pork muscle (P < 0.05). In addition, gamma irradiation was more influential than electron or X-ray irradiation on the negative impacts on water-holding capacity and texture of heat-induced gels. Therefore, this study suggests that the irradiation source could be one of the significant factors affecting gelling properties of irradiated meat.
Keywords: X-ray, Electron-beam, Gamma-ray, Ionizing radiation, Myofibrillar protein, Gelling properties
Introduction
Food irradiation is an effective and safe preservation method to extend the shelf-life of fresh meat and processed meat products for long-term storage, thereby controlling food-borne pathogen growth [1]. Currently, irradiation up to 10 kGy is generally known to be safe without adverse impacts on the nutritional and sensory properties of food [2]. The Codex Alimentarius Commission [2] has approved three ionizing sources (gamma ray, electron beam (e-beam), and X-ray) for food irradiation.
The gel-forming ability of muscle proteins, which is mostly related to salt-soluble myofibrillar protein, is one of the functional properties responsible for quality characteristics of processed meat products [3]. The muscle proteins entail partial protein denaturation during thermal processes, which results in the formation of a three-dimensional gel network. In particular, myofibrillar proteins have an important role in forming heat-induced gels [3]. However, the heat-induced gelation of salt-soluble proteins extracted from pork muscles irradiated with different ionizing sources has not yet been studied.
Therefore, the objective of this study was to compare the physicochemical and textural properties of heat-induced gels prepared with salt-soluble proteins extracted from pork muscles irradiated with gamma rays, electron beams, and X-rays.
Materials and methods
Irradiation procedure
Pork muscles (M. biceps femoris, M. semitendinosus, and M. semimembranosus) purchased from a local market were vacuum-packaged in nylon/polyethylene bags (thickness of the samples was 1.5 cm) and irradiated at an absorbed dose level of 5 kGy using the three irradiation sources; gamma rays, electron beams, and X-rays. The dose rate was fixed at 5 kGy/h for all methods of irradiation. Actual doses were determined using radiochromic dosimetry films at two different positions (top and bottom) of each sample.
Gel preparation
Gel preparation from irradiated pork muscles was performed according to McCord et al. [4]. Ground pork muscles were homogenized with two volumes of 0.58 M saline solution (0.49 M NaCl, 17.8 mM Na5P3O10, and 1 mM NaN3, pH 8.3, 2 °C) for 30 s in a food blender. The homogenate was made to stand in a 2 °C refrigerator for 1 h and was centrifuged at 12,000×g (2 °C) for 1 h (Supra 25K High speed refrigerated centrifuge, Hanil Science Industrial, Seoul, Korea). The supernatant containing salt-soluble proteins was filtered through three layers of cheesecloth and was diluted to 5% of the protein concentration using a saline solution (pH 6.0). The salt-soluble protein solution was placed in glass gelling tubes (diameter of 20 mm). To remove the air bubbles from the solution, the gelling tubes were centrifuged at 800×g for 15 min (4 °C). Before heating, all samples were equilibrated in a 20 °C water bath for 10 min, heated to 90 °C at 1.75 °C/min, and held at 90 °C for 20 min. The heated tubes were cooled in a 4 °C water bath for gelation.
Physicochemical and textural analyses
The pH value of the gel was measured using an electronic pH meter. The water-holding capacity (WHC) of the heat-induced gel was determined by the method used by Kocher and Foegeding [5]. CIE L* (lightness), a* (redness), and b* (yellowness) were measured using a colorimeter. Protein solubility was determined according to the method used by Joo et al. [6]. The apparent viscosity of the salt-soluble protein solution was determined in a rotational viscometer [7]. The textural properties of heat-induced gels were determined using a texture analyzer [8].
Statistical analysis
A total of three independent replicate batches was performed. Data were analyzed using the general linear model (GLM) procedure of Statistical Analysis Software (SAS) package [9]. Duncan’s multiple range test (P < 0.05) was conducted to separate differences between treatment means.
Results and discussion
The physicochemical properties of heat-induced gels prepared from pork muscles irradiated with gamma rays, electron beams, and X-rays are shown in Table 1. The pH values of the control gel (6.42) prepared with non-irradiated pork muscles were significantly (P < 0.05) lower than those prepared with irradiated pork muscles (6.46–6.52). In particular, the heat-induced gel prepared with gamma-irradiated pork muscles resulted in the highest pH among all treatments (P < 0.05). Similarly, Choi et al. [1] reported that the pH of heat-induced gels prepared from gamma-irradiated chicken increased with increasing irradiation dose level (0–10 kGy).
Table 1.
Physicochemical properties of heat-induced gels prepared with pork myofibrillar proteins irradiated with gamma rays, electron beams, and X-rays
| Parameters | Control (non-irradiation) | Gamma-ray | Electron-beam | X-ray |
|---|---|---|---|---|
| pH | 6.42 ± 0.03c | 6.52 ± 0.02a | 6.46 ± 0.03b | 6.47 ± 0.02b |
| WHC (Water-holding capacity, %) | 74.35 ± 0.38a | 73.00 ± 0.17b | 73.12 ± 0.33b | 72.67 ± 0.31c |
| Color characteristics | ||||
| CIE L* (lightness) | 78.62 ± 0.11d | 82.61 ± 0.26a | 81.27 ± 0.72b | 80.29 ± 0.54c |
| CIE a* (redness) | 0.24 ± 0.05a | −0.21 ± 0.06bc | −0.25 ± 0.10b | −0.18 ± 0.03c |
| CIE b* (yellowness) | 6.08 ± 0.16c | 6.44 ± 0.25a | 6.20 ± 0.60b | 6.32 ± 0.09b |
| Protein solubility (mg/g) | ||||
| Sarcoplasmic proteins | 2.52 ± 0.12a | 2.17 ± 0.14b | 2.15 ± 0.05b | 2.10 ± 0.13b |
| Myofibrillar proteins | 5.40 ± 0.26a | 4.18 ± 0.35c | 4.76 ± 0.17b | 4.84 ± 0.29b |
| Total proteins | 7.92 ± 0.27a | 6.35 ± 0.37c | 6.91 ± 0.12b | 6.94 ± 0.29b |
All values are the mean ± standard deviation of three replicates
a–d Means within a row with different letters are significantly different (P < 0.05)
The WHC of the control gel was significantly (P < 0.05) higher than that of all samples prepared with irradiated pork muscles, whereas the heat-induced gel prepared from gamma-irradiated pork muscles showed the lowest WHC. Choi et al. [1] suggested that the reduced WHC of heat-induced gel prepared from gamma-irradiated chicken was likely due to protein denaturation because of irradiation. Thus, the result of the WHC in the current study could imply that gamma irradiation might cause more critical impact on salt-soluble pork proteins.
Significant differences in the CIE L* value (lightness), a* value (redness), and b* value (yellowness) of heat-induced gel were observed because of different irradiation sources (P < 0.05). The heat-induced gel prepared with irradiated pork muscles had significantly (P < 0.05) higher lightness and yellowness but lower redness than the control gel, regardless of the irradiation source. Choi et al. [1] have also reported that the values of lightness and yellowness for all irradiated samples are higher than those of the control, whereas the redness of gel in the control was the highest (P < 0.05).
The solubilities of sarcoplasmic, myofibrillar, and total proteins of heat-induced gels prepared from irradiated pork muscles were significantly (P < 0.05) lower than those of the control. A similar result was reported by Choi et al. [1], who found that the protein solubility (myofibrillar and total proteins) of heat-induced gels prepared from gamma-irradiated chicken was higher than that of non-irradiated chicken. In addition, the heat-induced gel prepared with gamma-irradiated pork muscles showed a lower myofibrillar protein solubility than that prepared with electron or X-ray irradiated pork muscles (P < 0.05). Liu et al. [10] reported that salt-soluble myofibrillar proteins, such as myosin and actin, play an important role in the formation of continuous three-dimensional networks, which could immobilize water in heat-induced gels. Wang et al. [3] reported that the solubility of muscle proteins could indicate the degree of muscle protein denaturation occurring during meat processing. Thus, the reduced myofibrillar protein solubility in the gel prepared with gamma irradiated pork muscle could be one of the evidences for the decreased WHC.
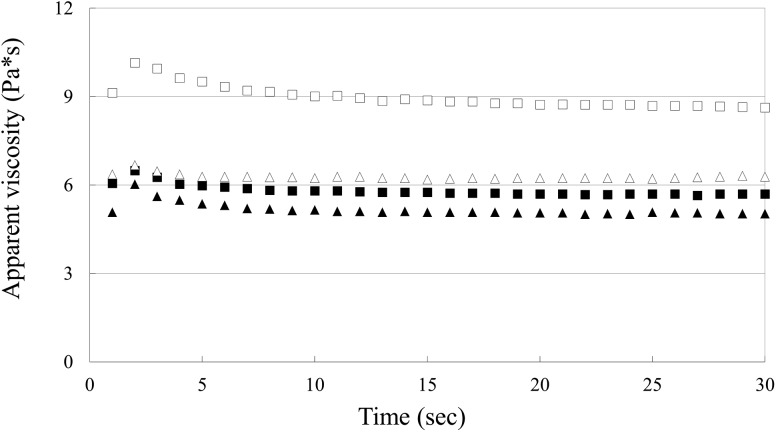
The apparent viscosity of salt-soluble protein solution prepared with pork muscles irradiated with gamma rays, electron beams, and X-rays is shown in Fig. 1. All treatments showed thixotropic behavior, i.e., their apparent viscosities decreased with increasing rotation time. However, the apparent viscosities of samples prepared with irradiated pork muscles were significantly (P < 0.05) lower than that of control. In addition, the sample prepared with gamma irradiated pork musclead slightly lower apparent viscosity compared to those prepared with electron-beam or X-ray irradiated pork muscles. Ciesla et al. [11] indicated that protein cross-linking could contribute to an increase in the apparent viscosity of the protein solution. Thus, this result indicates that irradiation might undermine the cross-linking of salt-soluble pork proteins, and this phenomenon was remarkably observed using gamma irradiation.
Fig. 1.
Apparent viscosity of the salt-soluble protein solution prepared with pork muscles irradiated with gamma rays, electron beams, and X-rays. (open square) control (non-irradiation); (filled triangle) gamma-ray; (open triangle) E-beam; (filled square) X-ray
Results on the texture profile analysis of heated gel prepared with differently irradiated salt-soluble proteins are shown in Table 2. All textural parameters of heat-induced gels prepared with irradiated pork muscles were significantly (P < 0.05) lower than those of the control, except for cohesiveness. While the highest cohesiveness was found in the control gel, the cohesiveness of heat-induced gel prepared with X-ray irradiated pork muscles was comparable with that of control (P > 0.05). These results were in agreement with those reported by Choi et al. [1]. They reported that the decreased values of hardness, cohesiveness, gumminess, and chewiness could be attributed to the denaturation of salt-soluble protein caused by gamma irradiation.
Table 2.
Textural characteristics of heat-induced gels prepared with pork myofibrillar proteins irradiated with gamma rays, electron beams, and X-rays
| Parameters | Control (non-irradiation) | Gamma-ray | Electron-beam | X-ray |
|---|---|---|---|---|
| Hardness (N) | 0.98 ± 0.12a | 0.70 ± 0.11b | 0.69 ± 0.08b | 0.53 ± 0.09c |
| Springiness | 0.96 ± 0.07a | 0.84 ± 0.06b | 0.86 ± 0.05b | 0.83 ± 0.13b |
| Cohesiveness | 0.49 ± 0.04a | 0.41 ± 0.03b | 0.36 ± 0.09c | 0.45 ± 0.11ab |
| Gumminess (N) | 0.48 ± 0.09a | 0.29 ± 0.09b | 0.25 ± 0.08b | 0.23 ± 0.11b |
| Chewiness (N) | 0.46 ± 0.08a | 0.24 ± 0.07b | 0.22 ± 0.07b | 0.19 ± 0.08b |
All values are the mean ± standard deviation of three replicates
a–c Means within a row with different letters are significantly different (P < 0.05)
In conclusion, this current study indicates that 5 kGy irradiation on pork muscles decreased the WHC and deteriorated textural properties of the heat-induced gel prepared with the irradiated pork muscles, which was likely due to the muscle protein denaturation supported by reductions in myofibrillar protein solubility and apparent viscosity. In addition, gamma irradiation was more influential on the negative impact on the WHC and texture of the heat-induced gel, when compared to electron-beam or X-ray irradiation. Therefore, this study suggests that the irradiation source can be one of the significant factors affecting gelling properties of irradiated meat.
Acknowledgements
This research was supported High Value-added Food Technology Development Program (314068-3) by the Ministry of Agriculture, Food and Rural Affairs (Republic of Korea).
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflict of interest.
References
- 1.Choi YS, Kim HW, Hwang KE, Song DH, Jeong TJ, Seo KW, Kim YB, Kim CJ. Effects of gamma irradiation on physicochemical properties of heat-induced gel prepared with chicken salt-soluble proteins. Radiat. Phys. Chem. 2015;106:16–20. doi: 10.1016/j.radphyschem.2014.06.029. [DOI] [Google Scholar]
- 2.CAC (Codex Alimentarius Commission). Codex general standard for irradiated foods. CODEX STAN 106-1983. Rev.1-2003 (2003).
- 3.Wang SF, Smith DM, Steffe JF. Effect of pH on the dynamic rhelogical properties of chicken breast salt-soluble protein during heat-induced gelation. Poult. Sci. 1990;69:2220–2227. doi: 10.3382/ps.0692220. [DOI] [Google Scholar]
- 4.McCord A, Smyth AB, O’Neill EE. Heat-induced gelation properties of salt-soluble muscle proteins as affected by non-meat proteins. J. Food Sci. 1998;63:580–583. doi: 10.1111/j.1365-2621.1998.tb15789.x. [DOI] [PubMed] [Google Scholar]
- 5.Kocher PN, Foegeding EA. Microcentrifuge-based method for measuring water-holding of protein gels. J. Food Sci. 1993;58:1040–1046. doi: 10.1111/j.1365-2621.1993.tb06107.x. [DOI] [Google Scholar]
- 6.Joo ST, Kauffman RG, Kim BC, Park GB. The relationship of sarcoplasmic and myofibrillar protein solubility to colour and water-holding capacity in porcine longissimus muscle. Meat Sci. 1999;52:291–297. doi: 10.1016/S0309-1740(99)00005-4. [DOI] [PubMed] [Google Scholar]
- 7.Park SY, Wang SH, Chin KB, Kim YD. Rheological properties of the mixture and heat-induced gel prepared from pork salt soluble protein in combined with soluble chitooligosaccharide and chitosan. Korean J. Food Sci. Technol. 2004;36:594–597. [Google Scholar]
- 8.Bourne MC. Texture profile analysis. Food Technol. 1978;32:62–66. [Google Scholar]
- 9.SAS . SAS user’s guide: Basic statistical analysis. NC, USA: Statistical Analysis Systems Institute; 2011. [Google Scholar]
- 10.Liu R, Zhao SM, Xiong SB, Xie BJ, Qin LH. Role of secondary structures in the gelation of porcine myosin at different pH values. Meat Sci. 2008;80:632–639. doi: 10.1016/j.meatsci.2008.02.014. [DOI] [PubMed] [Google Scholar]
- 11.Ciesla K, Salmieri S, Lacroix M, Tien Le C. Gamma irradiation influence on physical properties of milk proteins. Radiat. Phys. Chem. 2004;71:93–97. doi: 10.1016/j.radphyschem.2004.04.068. [DOI] [Google Scholar]