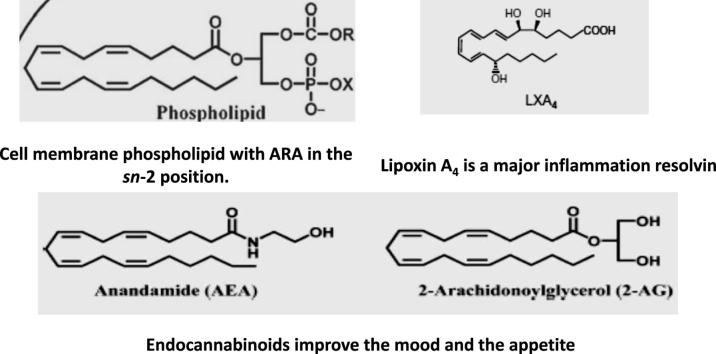
Graphical abstract
Keywords: Arachidonic acid, Ion channels, Schistosomicide, Endotumoricide, Lipoxin A4, Endocannabinoids
Abstract
It is time to shift the arachidonic acid (ARA) paradigm from a harm-generating molecule to its status of polyunsaturated fatty acid essential for normal health. ARA is an integral constituent of biological cell membrane, conferring it with fluidity and flexibility, so necessary for the function of all cells, especially in nervous system, skeletal muscle, and immune system. Arachidonic acid is obtained from food or by desaturation and chain elongation of the plant-rich essential fatty acid, linoleic acid. Free ARA modulates the function of ion channels, several receptors and enzymes, via activation as well as inhibition. That explains its fundamental role in the proper function of the brain and muscles and its protective potential against Schistosoma mansoni and S. haematobium infection and tumor initiation, development, and metastasis. Arachidonic acid in cell membranes undergoes reacylation/deacylation cycles, which keep the concentration of free ARA in cells at a very low level and limit ARA availability to oxidation. Metabolites derived from ARA oxidation do not initiate but contribute to inflammation and most importantly lead to the generation of mediators responsible for resolving inflammation and wound healing. Endocannabinoids are oxidation-independent ARA derivatives, critically important for brain reward signaling, motivational processes, emotion, stress responses, pain, and energy balance. Free ARA and metabolites promote and modulate type 2 immune responses, which are critically important in resistance to parasites and allergens insult, directly via action on eosinophils, basophils, and mast cells and indirectly by binding to specific receptors on innate lymphoid cells. In conclusion, the present review advocates the innumerable ARA roles and considerable importance for normal health.
Introduction
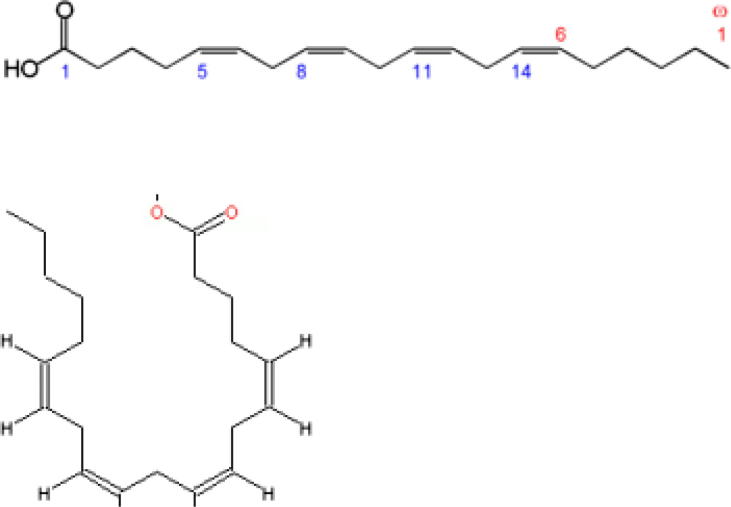
Arachidonic acid (ARA) is a 20-carbon chain fatty acid with four methylene-interrupted cis double bonds, the first with respect to the methyl end (omega, ω or n) is located between carbon 6 and 7. Hence, ARA belongs to the omega-6 (n-6) polyunsaturated fatty acids (PUFA), is designated as 20:4ω-6, with a biochemical nomenclature of all-cis-5,8,11,14-eicosatetraenoic acid, and usually assumes a hairpin configuration (Fig. 1) [1].
Fig. 1.
Arachidonic acid structure showing linear and hairpin configuration.
Arachidonic acid is obtained from food such as poultry, animal organs and meat, fish, seafood, and eggs [2], [3], [4], [5], and is incorporated in phospholipids in the cells' cytosol, adjacent to the endoplasmic reticulum membrane that is studded with the proteins necessary for phospholipid synthesis and their allocation to the diverse biological membranes [6]. Of note, glycerophospholipids are composed of a glycerol backbone esterified to two hydrophobic fatty acids tails at sn- (stereospecifically numbered) 1 and 2 position and a hydrophilic head-group at sn 3. The membrane and cytosolic phospholipids of mammalian cells and tissues are rich in ARA, usually localized in the glycerol backbone sn-2 position. Platelets, mononuclear cells, neutrophils, liver, brain and muscle have up to 25% phospholipid fatty acids as ARA [7]. Arachidonic acid participates in the Lands cycle, a membrane phospholipids' reacylation/deacylation cycle, which serves to keep the concentration of free ARA in cells at a very low level [8]. Since ARA is a fundamental constituent of cell structure, it will particularly be needed for during development and growth and upon severe or widespread cell damage and injury.
Another ARA source, so important for herbivores and vegetarians, is linoleic acid, also an omega-6, 18 carbond PUFA that contains only two cis- double bonds (18:2ω-6). Linoleic acid is an essential fatty acid for animals because they cannot synthesize it, in contrast to plants, which can synthesize it from oleic acid. Linoleic acid is abundant in many nuts, fatty seeds and their derived vegetable oils [5]. It is converted in animals cells cytosol to ARA, docosatetraenoic acid (22:4ω-6) and other fatty acids by step-wise desaturation and chain elongation. Linoleic acid conversion to ARA is, however, low. Linoleic acid is readily oxidized by delta 6-desaturase to γ-linolenic acid (18:3-n6), but several factors such as aging, nutrition, smoking impair the activity of the enzyme. Gamma linolenic elongation step to dihomo-γ-linolenic acid (20:3-n6) is rapid; yet, it is oxidized by delta-5 desaturase to yield ARA at a small percentage because delta-5 desaturase prefers the n-3 to n-6 fatty acids [9], [10], [11], [12], [13].
Arachidonic acid production
The filamentous fungus, Mortierella, especially of the species alpina (http://eol.org/collections/119317) is considered a predominant source for preparation of ARA on the industrial scale [14], [15], [16], [17], [18], [19], [20], [21], [22]. Additionally, ARA can be in vitro synthesized from 5-hexyn-1-ol as described in detail by Prakash et al. [23].
Arachidonic acid physiological functions
Cell membrane fluidity
Arachidonic acid four cis double bonds endow it with mobility and flexibility conferring flexibility, fluidity and selective permeability to membranes [24], [25]. ARA control of membrane fluidity influences the function of specific membrane proteins involved in cellular signaling [24], [25] and plays a fundamental role in maintenance of cell and organelle integrity and vascular permeability [26]. These properties might explain ARA critical role in neuron function, brain synaptic plasticity, and long-term potentiation in the hippocampus [27], [28], [29], [30], [31].
Ion channels
Non-esterified, free ARA affects neuronal excitability and synaptic transmission via acting on most voltage-gated ion (Nav, Kv, Cav, Clv, proton Hv) channels, responsible for regulating the electric activity of excitable tissues, such as the brain, heart and muscles. Ion channels are large families of integral membrane proteins that form a selective pore for ions to cross the lipid bilayer, via undergoing conformational changes in response to alteration in the cell transmembrane electrical potential. These channels gate passage of specific ions and thus control the propagation of nerve impulses, muscle contraction, and hormone secretion [32], [33], [34], [35], [36], [37], [38], [39]. The homologous mon-, di- or tetrameric subunits of ARA-sensitive voltage-gated channels are composed of four transmembrane helices spanning the cell membrane lipid bilayer (S1-S4) making up the voltage-sensor domain, and/or 2 transmembrane segments constituting the central ion-conducting pore [34], [35], [39]. The gating charges are situated on helix S4, a positively charged voltage sensor, which responds to changes in voltage across the membrane by inducing movements of the helix relative to the remainder of the protein or the movement of the positive charges through the membrane toward the extracellular side [34], [35], [36]. Since S4 is in contact with the lipid bilayer, the ARA lipophilic, flexible acyl chain can position its carboxylate negative charge onto the voltage sensor, and modulate its activity, likely shifting the voltage dependency of activation via channel-activating electrostatic interactions [37], [38], [39].
Free ARA evoked K+ channel opening in neurons of the rat visual cortex, thus suggesting the existence of an ARA-activated type of K+ channel, which may play a critical role in modulating cortical neuronal excitability [40], [41]. Arachidonic acid was previously reported to directly activate K+ channels in gastric, pulmonary artery, and vascular smooth muscle cells, and cardiac atrial cells likely via interacting with the ion channel protein itself [40], [41], [42], [43]. Conversely, ARA is known to suppress the Kv4 family of voltage-dependent K+ channels, in a direct, fast, potent, and partially reversible mode [44]. The activity of the large-conductance Ca2+- dependent K+ (BK) channels, which control diverse functions in the central nervous system such as sleep and neural regulation of the heart, is increased up to 4 folds by ARA, consequent to direct interaction with the channel protein [43], [45]. Conversely, ARA inhibited intermediate conductance, Ca++-activated K+ channels, which play crucial roles in agonist-mediated transepithelial Cl− secretion across airway and intestinal epithelia, via interacting with the pore-lining amino acids (aa) threonine (aa 250) and valine (aa 275) [46]. Background, non-voltage-dependent two pore domain K(+) channels, which play an essential role in setting the neuronal membrane potential and potential duration are opened by ARA, and not its metabolites, provided the carboxyl end is not substituted with an alcohol or methyl ester [47], [48]. Additionally, ARA was reported to inhibit the ATP- sensitive K+ channel in cardiac myocytes almost completely, while activated the ATP-insensitive K+ channel [49].
Free, non-esterified ARA prevents ischemia-induced heart arrhythmia, a major cause of sudden cardiac death in humans, by modulating the activity of cardiac Na+ channels, the major class of ion channels that determine cardiac excitability, causing a reduction in the electrical excitability and/or automaticity of cardiac myocytes [50]. Sodium channels consist of a large functional subunit and a smaller subunit, which interacts with a regulatory segment. Arachidonic acid, but not its metabolic products, was shown to voltage-dependent modulate muscle Na+ channel currents, displaying both activation and inhibitory effects depending on the depolarizing potential [51]. Arachidonic acid also displayed both activation and inhibitory effects on different Cl- channels, widely distributed especially in epithelial tissues, and thus, mediate increase or block of Cl- ions permeation [52], [53], [54], [55]. The double bonds and hydrophilic head were recently reported to be responsible for the ARA mediated dramatic increases in proton current amplitude through the voltage-gated proton (Hv) channel. The latter lacks a pore domain but allows passage of proton through the center of each voltage sensor domain [39] and supports the rapid production of reactive oxygen species (ROS) in phagocytes through regulation of pH and membrane potential [56].
Receptors and enzymes
Exogenous or endogenously produced ARA was discovered to greatly enhance the functional activity of ligand-gated ion channels, namely the γ-amino butyric acid receptor (GABA-R) located on the neuronal membrane, via modulating the GABA-R interaction characteristics with its ligands [57], [58], [59], [60]. Free ARA exposure essentially led to inhibiting the muscle and neuronal nicotinic acetylcholine receptor (nAChR), an integral membrane protein deeply embedded in the postsynaptic region, with two agonist binding sites and a central ion pore. The receptor inhibition resulted from ARA displacing lipids from their sites in the plasma membrane and direct acting as antagonist at the PUFA-protein interface [60], [61], [62], [63].
Activation of eosinophils, neutrophils, and macrophages elicits powerful respiratory burst associated with reduction of molecular oxygen to superoxide via activation of the NADPH oxidase complex, which consists of five proteins residing in resting cells in the cytosol or membrane of intracellular vesicles, and in activated cells are assembled on the cell membrane [26], [64]. Generated ROS induce membrane depolarization and cytoplasm pH decrease, thus restricting NADPH activity. Concentrations of ARA of 5–10 µM added to neutrophils enhanced NADPH oxidase stimulation due to ARA-mediated activation of the Hv channel, modulation of the membrane potential and pH, and efflux of the H+ ions generated together with the superoxide anion, O2− [56], [65], [66], [67].
PUFA, especially ARA, are documented activators of membrane-associated, magnesium-dependent, neutral sphingomyelinases (nSMase) [68], [69], [70], [71], [72], [73]. ARA was recently documented as activator of Schistosoma mansoni and S. haematobium tegument-associated neutral sphingomyelinase in a dose-dependent manner, eventually leading to their attrition in vitro and in vivo [74], [75], [76], [77].
Cell death
Free ARA levels are kept very low in cells as uncontrolled accumulation of unesterified ARA decisively impaired cell survival via induction of apoptosis [8]. The apoptotic effect was attributed to free ARA and not its metabolites as it was recorded in the absence of lipoxygenase or cyclooxygenase enzyme activity, and was speculated to be associated with oxidative stress and/or changes in membrane fluidity [6], [25], [26], [78], [79], [80], [81]. Indeed, Pompeia et al. reported that the cytotoxicity of arachidonic acid is undeniable, but may well be one of its fundamental functions in vivo [81]. ARA concentrations of 50–100 µM are cytotoxic to most cell lines in vitro. In the majority of models 1–10 µM ARA is necessary to elicit any biological response but some activities require 100–300 µM [25]. This indicates that ARA apoptotic and physiological levels overlap and it is very possible that ARA cytotoxicity occurs in vivo because under some pathologic conditions, human plasma ARA levels can increase from 0.1–50 μM to 100 and up to 500 μM [81].
A most needed nutritional supplement
Newborns
Polyunsaturated fatty acids (PUFAs), especially ARA, affect the function of numerous ion channels, the activity of various enzymes and are implicated in cell apoptosis, necrosis and death, events of critical importance during embryogenesis, thereby have significant physiological and pharmacological impact on the health of newborns [39], [40], [41], [42]. ARA and docosahexaenoic acid (DHA, 22:6 ω3) are important components of human milk but are lacking in cow milk and most commercial infant formula in developing countries [82]. Due to its importance in development especially of the central nervous system and retina [82], [83], [84], the Food and Agricultural Organization (FAO)/World Health Organization (WHO) recommended that infant formula, unless specifically added, should be supplemented with ARA [85]. Decreased postnatal ARA and DHA blood levels in premature infants were found to be associated with neonatal morbidities, while adding DHA and ARA to preterm-infant formulas led to improved visual acuity, visual attention and cognitive development [82], [83], [84], [85]. The ARA levels in human milk and ARA requirements, essentiality in pre- and neonatal life and during development, and inclusion in infant formulas have recently been reviewed [86], [87], [88], challenged and discussed [89].
Neurological disorders
ARA does not only influence cell membrane fluidity and the activity of ion channels, especially in the brain, it constitutes together with DHA 20% of the human brain dry weight, concentrated in the neurons outer membrane and in the myelin sheath [90]. Additionally, positron emission tomography was used to show that the brain of human healthy volunteers consumes ARA at a rate of 17.8 mg/d [91]. Accordingly, ARA was recommended for management of central nervous system, visual and auditory damage in preterm infants via supporting neurovascular membrane integrity [92]. Children with autism had lower levels of blood PUFA, especially ARA, than normal children [93], and showed notable improvement after dietary PUFA intake [94]. In the elderly too, ARA supplementation improved cognitive functions [31], perhaps via increasing the proliferation of neural stem/progenitor cells or newborn neurons and general hippocampal neurogenesis [30]. The charged ARA displayed beneficial effects on epileptic seizures and cardiac arrhythmia by electrostatically affecting the kV channel's voltage sensor, thus regulating neuronal excitability [37], [38].
Exercise
In skeletal muscles, ARA has been found to make up to 15–17% of total fatty acids, thus explaining why ARA supplementation affected body composition, muscle function and power output in strength-training individuals [86], [95], [96]. It is also possible that ARA modulates neuromuscular signaling through its incorporation into cell membranes, and/or increases neurotransmitter firing from nerve cells [91].
Schistosomicidal action
The first evidence relating PUFA to schistosomes came from the ability of corn oil to expose hitherto unavailable surface membrane antigens of Schistosoma mansoni lung-stage larvae to specific antibody binding, thus allowing serologic visualization [97]. Further studies indicated that among PUFA, ARA (10 µM, 30 min) was the most effective in allowing specific antibody binding to otherwise hidden surface membrane antigens of S. mansoni and Schistosoma haematobium lung-stage schistosomula [74]. Of importance, exposure to 20 µM ARA for 30 min elicited surface membrane disintegration and attrition of the schistosomula [74]. Studies aiming at clarifying these observations led to identification of surface membrane sphingomyelin (SM) instrumental role in schistosome immune evasion. Controlled SM hydrolysis by parasite tegument-associated neutral sphingomyelinase (nSMase) allows entry of nutrients but not host molecules >600 Da or antibodies. Excessive nSMase activation and consequent SM hydrolysis elicits exposure of surface membrane antigens and eventual larval death. ARA is a major nSMase activator. Accordingly, it was straightforward to predict that ARA possesses potentially potent schistosomicidal activity [75], [77], [86], [98], [99].
All adult worms of S. mansoni and S. haematobium exposed to 2.5 mM ARA in the presence of 100% fetal calf serum showed extensive damage, disorganization, and degeneration of the tegument and the subtegumental musculature followed by death of all worms within 5 h [100]. Pure ARA and different ARA formulations elicited notable, reproducible, and safe schistosomicidal activity against larval, juvenile and adult S. mansoni and S. haematobium infection of inbred and outbred mice and hamsters [100], [101]. The ability of ARA to control infection with S. mansoni was demonstrated in Egyptian children. The chemotherapeutic activity of ARA and praziquantel (PZQ) was equally high in low infection settings and equally low in children with heavy infection living in high endemicity areas. The highest cure was consistently achieved in children with light or heavy infection when ARA was combined with PZQ [77], [86], [102], [103].
A breakthrough regarding the usefulness of ARA in defense against schistosomes came from the demonstration of association between resistance of the water-rat, Nectomys squamipes to repeated infection with S. mansoni and accumulation of ARA in liver cells [104]. This pioneering study prompted us to examine the relation between susceptibility and resistance of rodents to S. mansoni or S. haematobium infection and ARA levels in serum and lung and liver cells before and weekly after infection. The results strongly suggested that ARA is a potent “natural” schistosomicide, and may be considered an endoschistosomicide [105].
The schistosomicidal action of ARA is based on excessive hydrolysis of parasite surface membrane SM. Interestingly, Miltefosine, a hexa-decyl-phosphocholine, which interferes with proper biosynthesis of SM, was recently documented as a potent schistosomicide in vitro and in vivo [106].
Tumoricidal potential
Reports decades earlier indicated that PUFA, and especially free unesterified ARA possess tumoricidal activity in vitro and in vivo [107]. The most important and consistent studies documenting the tumoricidal action of PUFA, namely ARA were reported by Undurti Das and Colleagues [108], [109], [110], [111], [112], [113], [114], [115], who advocated ARA as a potential anti-cancer drug [108]. Thus, ARA was reported to kill tumor cells selectively in vitro via eliciting cell surface membrane lipid peroxidation, which can be blocked by vitamin E, uric acid, glutathione peroxidase and superoxide dismutase [109]. Free ARA was found to inhibit the in vitro growth of human cervical carcinoma (HELA) cells and methyl cholanthrene-induced sarcoma cells. Free ARA augmented the generation of superoxide anion and lipid peroxidation in the tumor cells indicating a possible correlation between the ability of unesterified PUFA to augment free radicals and their tumoricidal action [110], [111]. Moreover, free unesterified ARA, independently of its metabolites, displayed cytotoxic action on both vincristine-sensitive (KB-3-1) and resistant (KB-Ch(R)-8-5) cancer cells in vitro that appeared to be a free-radical dependent process [112]. At concentrations of 100–200 µM, ARA was more effective than mesotrexate in in vitro suppression of gastric carcinoma cells, as a result of lipid peroxidation processes [113], and inhibited proliferation of human prostate cancer and human prostate epithelial cells, independently of free radicals generation [114]. ARA-mediated apoptosis of colon cancer cells appeared to be essentially due to loss of mitochondrial membrane, accumulation of ROS, and caspase-3 and caspase-9 activation [115]. Accordingly, it was concluded that ARA suppresses proliferation of normal and tumor cells by a variety of mechanisms that may partly depend on the type(s) of cell(s) being tested and the way ARA is handled by the cells [111], [112]. A contradictory effect of ARA on tumorigenesis was observed in mice with a germline mutation in the adenomatous polyposis coli gene [116].
We have recently proposed that ARA may inhibit proliferation and elicit death of tumor cells via its activating impact on cell membrane-associated neutral sphingomyelinase (nSMase) and increased outer leaflet SM hydrolysis [68], [69], [70], [71], [72], [73], [74], [75], [76]. Disruption of the tight SM-based hydrogen bond network around cancer cells may allow contact inhibition processes to proceed and cell proliferation to stop [77], whereby the primary SM catabolite, ceramide released following SM hydrolysis is a renowned secondary messenger involved in programmed cell death [117]. It is of importance to recall that Miltefosine, which has been approved for the treatment of breast cancer metastasis, and is currently used for the treatment of cutaneous metastases of mammary carcinoma significantly inhibits SM biosynthesis in human hepatoma and other tumor cells [106], [118], [119], [120], [121]. The likely mechanism of action of this phospholipid analogue is inhibition of phosphatidylcholine biosynthesis, thus hindering SM metabolism, and substantially increasing the levels of ceramide [120], [121].
Arachidonic acid metabolites physiological roles
The four cis double bonds of ARA mediate its propensity to react with molecular oxygen through the actions of three types of oxygenases: cyclooxygenase (COX), lipoxygenase (LOX), and cytochrome P450, leading to the generation of inflammatory bioactive lipids or eicosanoids (prostanoids and leukotrienes). However, dietary ARA is a poor substrate for oxidation [11] and ARA processing occurs only following release from cell membrane by phospholipase A2 [122], [123]. Moreover, ARA reacylation is very significant in cells whereby a large portion of the ARA that is released by phospholipase A2 is rapidly incorporated back into phospholipids and a minor portion only converted into oxygenated metabolites [8]. Additionally, several, if not all, ARA metabolites have a considerable role in maintaining normal health via regulating innumerable physiologic processes [122], [123].
Resolution of inflammation
Not only free ARA, but its metabolites, prostaglandins (PG), namely PGF2a, PGE2, and PGI2 display essential roles in skeletal muscle development and growth by controlling proliferation, differentiation, migration, fusion and survival of myoblasts [123]. Indeed, eicosanoids produced from ARA tend to promote muscle growth during and after physical activity in healthy humans. Yet, the major action of ARA metabolites is promotion of acute inflammatory response, characterized by the production of pro-inflammatory mediators such as PGE2 and PGI2, followed by a second phase in which lipid mediators with pro-resolution activities may be generated. Resolution of inflammation is no more considered a passive process, but rather an active programmed response regulated by mediators with pro-resolving capacity, prominent among which is ARA-derived lipoxin A4 [124], [125]. Lipoxin A4 stimulates cessation of neutrophil infiltration, enhances macrophage uptake of apoptotic cells in pre-clinical animal models [124], [125], [126], [127], [128], [129], [130], attenuates leukotriene C4-induced bronchoconstriction in asthmatic subjects, decreases eczema severity and duration and improves patients’ quality of life via inhibiting the activity of innate lymphoid cells type 2 [131], [132].
Lipoxin A4 (1 nM) was also reported to attenuate adipose inflammation, decreasing interleukin (IL)- 6 and increasing IL-10 expression in aged mice [129]. Recently, lipoxin A4 encapsulated in poly-lactic-co-glycolic acid microparticles displayed considerable healing effects in topical treatment of dorsal rat skin lesions, provided interaction with its specific receptor on skin cells [130]. Other ARA metabolites, notably PGE2, PGI2 and leukotriene B4 and leukotriene D4 readily promote wound healing via regulating the production of angiogenic factors and endothelial cell functions [133], and inducing stem cells' proliferation and angiogenic potential [134]. Furthermore, lipoxin A4 was reported to have anti-diabetic potential via inhibiting IL-6, tumor necrosis factor and ROS generation [135], [136].
Endocannabinoids
Endocannabinoids are so termed because they activate the same G protein-coupled, cannabinoid receptors (CB1 and CB2) as delta-9-tetrahydrocannabinol, the active component of marijuana (Cannabis sativa). Endocannabinoids are important modulators of brain reward signaling, motivational processes, emotion, stress responses, pain and energy balance [137], [138], [139], [140], [141]. The endocannabinoids, N-arachidonoyl-ethanolamine and 2-arachidonoylglycerol, are ARA-derived. ARA Trans-acylase-catalyzed transfer of ARA from the sn-1 position of phospholipids to the nitrogen atom of phosphatidylethanolamine generates N-arachidonoyl-phosphatidylethanolamine (NAPE). NAPE can be hydrolyzed to arachidonylethanolamine (anandamide, AEA) in a one-step reaction catalyzed by NAPE-specific phospholipase D, or two-steps reaction catalyzed by a phospholipase C and a phosphatase. NAPE can be converted to anandamide via two further synthetic pathways [137, Fig. 2]. The importance of anandamide can be inferred from the redundancy of its precursor conversion pathways. 2-arachidonoylglycerol (2-AG) is produced from the hydrolysis of diacylglycerols (DAGs) containing arachidonate in the 2 position, catalyzed by a DAG lipase that is selective for the sn-1 position [137, Fig. 3].
Interaction of ARA-derived endocannabinoids with their specific receptors generate signals, which control neural processes that underpin key aspects of social behavior whereby endocannabinoid signaling dysregulation is associated with social impairment related to neuropsychiatric disorders [138], [139]. Endocannabinoid-mediated signaling, especially in the brain, modulates a variety of pathophysiological processes, including appetite, pain and mood, whereby inhibition of endocannabinoid degradation is predicted to be instrumental in reducing pain and anxiety [140], [141]. Additionally, anandamide appeared to modulate human sperm motility [142] and improve renal functions and chronic inflammatory disorders of the gastrointestinal tract by regulating gut homeostasis, gastrointestinal motility, visceral sensation, and inflammation [143], [144], [145].
Roles in type 2 immune responses
Allergens, cysteine peptidases and numerous helminth-derived excretory-secretory products disrupt the epithelial or endothelial barriers, eliciting release of the type 2 immunity master cytokines and alarmins, TSLP (thymic stromal lymphopoietin), interleukin (IL)-25 and IL-33 [98], [99]. These cytokines bind to receptors on innate lymphoid cells 2 (ILC2), tissue-resident sentinels, mainly found in the skin and at mucosal surfaces of intestine and lungs. Cytokine-receptors' interactions result into signals that induce ILC2 recruitment, proliferation and activation. The activated ILC2 produce type 2 cytokines, principally IL-5 and IL-13, which are instrumental in the recruitment and activation of eosinophils, basophils, and mast cells [146], [147], [148]. Major basic proteins, proteases, histamine, heparin, type 2 cytokines, and reactive ROS are not only produced inducing the various signs of inflammation, ARA is furthermore released from the activated cell membrane and oxidized to inflammatory metabolites (see review by Hanna and Hafez [149]). The ARA-derived metabolites are the road to generation of resolvins that help in resolving inflammation and wound and lesion healing [129], [130], [131], [132], [133], [134], [135], [136]. Of considerable importance is the discovery that ILC2 share with airway and gut smooth muscle cells, and/or epithelial cells, eosinophils, mast cells, macrophages, dendritic cells, and T helper 2 (Th2) lymphocytes surface membrane receptors for ARA-derived metabolites. Chemoattractant receptor-homologous molecule expressed on Th2 cells (CRTH2) is a receptor for prostaglandin D2, CysLTR for cysteinyl leukotrienes D4 and E4 and ALXR for lipoxin A4. Prostaglandin D2, leukotriene D4 and E4 stimulate, while lipoxin A4 inhibits ILC-2 expansion and effector functions [131], [132], [150], [151], [152], [153], [154], [155], thus, implicating ARA metabolites as secondary inducers of type 2 immune responses' amplification, regulation and memory in airway and gut hyperresponsiveness and repair, and resistance to parasites [156].
Conclusions
In conclusion, it is recommended to monitor and supplement serum ARA levels in pregnant women, infants, children and the elderly in poor rural settings as dietary ARA is safe, being a poor substrate for beta-oxidation and is critically essential for the development and optimal performance of the nervous system, especially the brain and cognitive functions, the skeletal muscle and immune systems. Additionally, ARA promotes and regulates type 2 immune responses against intestinal and blood flukes and may well represent an invaluable endoschistsomicide and endotumoricide.
Conflict of interest
The authors have declared no conflict of interest.
Compliance with Ethics Requirements
This article does not contain any studies with human or animal subjects.
Biographies

Hatem Tallima, graduated from the American University in Cairo (AUC) in year 2000, cum laude in Chemistry, and obtained his PhD degree in Biochemistry from the Faculty of Science, Cairo University, year 2006. He has 37 publications in international, peer-reviewed journals, h index 15 and more than 500 citations. He teaches Organic and Biochemistry at AUC and has contributed to the development of a drug and a vaccine against schistosomiasis in the Immunology Laboratories, Faculty of Science, Cairo University.

Rashika El Ridi, PhD, D.Sc., is Professor of Immunology at the Zoology Department, Faculty of Science, Cairo University, Cairo 12613, Egypt. Tel.: Lab (00202) 3567 6708; Home: (00202) 3337 0102; Mobile: 0109/5050888; E-mail: rashika@sci.cu.edu.eg and rashika_elridi@yahoo.com. Her responsibilities involved teaching immunology and molecular immunology to pre- and post-graduate students; and has directed research in immunology funded by NIH, Sandoz Gerontological Foundation, Schistosomiasis Research Project (SRP), the Egyptian Academy of Scientific Research and Technology; the International Centre for Genetic Engineering and Biotechnology; the World Health Organization; the Arab Foundation for Science and Technology; the Egyptian Science and Technology Development Fund (STDF), supervised 65 M.Sc. and 35 PhD. Theses, and published 94 papers in international, peer-reviewed journals. Obtained for these continuous efforts the State Award of Excellence in High-Tech Sciences, 2002, and 2010; the Cairo University Award for Recognition in Applied Sciences, 2002, the D.Sc. degree in Immunobiology, 2004, and the L’Oreal-Unesco Prize for Women in Science, 2010.
Footnotes
Peer review under responsibility of Cairo University.
References
- 1.Martin S.A., Brash A.R., Murphy R.C. The discovery and early structural studies of arachidonic acid. J Lipid Res. 2016;57(7):1126–1132. doi: 10.1194/jlr.R068072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Li D., Ng A., Mann N.J., Sinclair A.J. Contribution of meat fat to dietary arachidonic acid. Lipids. 1998;33(4):437–440. doi: 10.1007/s11745-998-0225-7. [DOI] [PubMed] [Google Scholar]
- 3.Taber L., Chiu C.H., Whelan J. Assessment of the arachidonic acid content in foods commonly consumed in the American diet. Lipids. 1998;33(12):1151–1157. doi: 10.1007/s11745-998-0317-4. [DOI] [PubMed] [Google Scholar]
- 4.Komprda T., Zelenka J., Fajmonová E., Fialová M., Kladroba D. Arachidonic acid and long-chain n-3 polyunsaturated fatty acid contents in meat of selected poultry and fish species in relation to dietary fat sources. J Agric Food Chem. 2005;53(17):6804–6812. doi: 10.1021/jf0504162. [DOI] [PubMed] [Google Scholar]
- 5.Abedi E., Sahari M.A. Long-chain polyunsaturated fatty acid sources and evaluation of their nutritional and functional properties. Food Sci Nutr. 2014;2(5):443–463. doi: 10.1002/fsn3.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Vance J.E. Eukaryotic lipid-biosynthetic enzymes: the same but not the same. Trends Biochem Sci. 1998;23(11):423–428. doi: 10.1016/s0968-0004(98)01297-3. [DOI] [PubMed] [Google Scholar]
- 7.Calder P.C. Dietary arachidonic acid: harmful, harmless or helpful? Br J Nutr. 2007;98(3):451–453. doi: 10.1017/S0007114507761779. [DOI] [PubMed] [Google Scholar]
- 8.Pérez R., Matabosch X., Llebaria A., Balboa M.A., Balsinde J. Blockade of arachidonic acid incorporation into phospholipids induces apoptosis in U937 promonocytic cells. J Lipid Res. 2006;47(3):484–491. doi: 10.1194/jlr.M500397-JLR200. [DOI] [PubMed] [Google Scholar]
- 9.Zhou L., Xu N., Nilsson A. Tissue uptake and interconversion of plasma unesterified 14C linoleic acid in the guinea pig. Biochim Biophys Acta. 1997;1349(3):197–210. doi: 10.1016/s0005-2760(97)00131-8. [DOI] [PubMed] [Google Scholar]
- 10.Zhou L., Vessby B., Nilsson A. Quantitative role of plasma free fatty acids in the supply of arachidonic acid to extrahepatic tissues in rats. J Nutr. 2002;132(9):2626–2631. doi: 10.1093/jn/132.9.2626. [DOI] [PubMed] [Google Scholar]
- 11.Sprecher H. The roles of anabolic and catabolic reactions in the synthesis and recycling of polyunsaturated fatty acids. Prostaglandins Leukot Essent Fatty Acids. 2002;67(2–3):79–83. doi: 10.1054/plef.2002.0402. [DOI] [PubMed] [Google Scholar]
- 12.Huang Y.-W., Huang C.-Y. Gamma linolenic acid (GLA) In: Shahidi F., editor. Nutraceutical and specialty lipids and their co-products. Taylor and Francis; Florida: 2006. pp. 169–184. [chapter 10] [Google Scholar]
- 13.Wiktorowska-Owczarek A., Berezińska M., Nowak J.Z. PUFAs: structures, metabolism and functions. Adv Clin Exp Med. 2015;24(6):931–941. doi: 10.17219/acem/31243. [DOI] [PubMed] [Google Scholar]
- 14.Yamada H., Shimizu S., Shinmen Y. Production of arachidonic acid by Mortierella elongata 1S-5. Agri Biol Chem. 1987;51(3):785–790. [Google Scholar]
- 15.Aki T., Nagahata Y., Ishihara K., Tanaka Y., Morinaga T., Higashiyama K. Production of arachidonic acid by a filamentous fungus, Mortierella alliacea strain YN-15. J Am Oil Chem Soc. 2001;78:599–604. [Google Scholar]
- 16.Sakuradani E., Shimizu S. Single cell oil production by Mortierella alpina. J Biotechnol. 2009;144(1):31–36. doi: 10.1016/j.jbiotec.2009.04.012. [DOI] [PubMed] [Google Scholar]
- 17.Nisha A., Rastogi N.K., Venkateswaran G. Optimization of media components for enhanced arachidonic acid production by Mortierella alpina under submerged cultivation. Biotechnol Bioprocess Eng. 2011;16:229–237. [Google Scholar]
- 18.Wu W.J., Zhang A.H., Peng C., Ren L.J., Song P., Yu Y.D. An efficient multi-stage fermentation strategy for the production of microbial oil rich in arachidonic acid in Mortierella alpina. Bioresour Bioprocess. 2017;4(1):8. doi: 10.1186/s40643-017-0138-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Grima E., Perez J., Camacho F., Medina A., Gimcnez A., Lopez Alonso D. The production of polyunsaturated fatty acids by microalgae: from strain selection to product purification. Process Biochem. 1995;30:711–719. [Google Scholar]
- 20.Zhu M., Zhou P.P., Yu L.J. Extraction of lipids from Mortierella alpina and enrichment of arachidonic acid from the fungal lipids. Bioresour Technol. 2002;84(1):93–95. doi: 10.1016/s0960-8524(02)00028-7. [DOI] [PubMed] [Google Scholar]
- 21.You J.-Y., Peng C., Liu X., Ji X.-J., Lu J., Tong Q. Enzymatic hydrolysis and extraction of arachidonic acid rich lipids from Mortierella alpina. Bioresour Technol. 2011;102(10):6088–6094. doi: 10.1016/j.biortech.2011.01.074. [DOI] [PubMed] [Google Scholar]
- 22.Ji X.J., Ren L.J., Nie Z.K., Huang H., Ouyang P.K. Fungal arachidonic acid-rich oil: research, development and industrialization. Crit Rev Biotechnol. 2014;34(3):197–214. doi: 10.3109/07388551.2013.778229. [DOI] [PubMed] [Google Scholar]
- 23.Prakash C., Saleh S., Sweetman B.J., Taber D.F., Blair I.A. A synthon for C-20 trideuterated eicosanoids: preparation of [2H3]-arachidonic acid. J Labelled Comp Radiopharm. 1989;27(5):539–551. [Google Scholar]
- 24.Pompéia C., Lopes L.R., Miyasaka C.K., Procópio J., Sannomiya P., Curi R. Effect of fatty acids on leukocyte function. Braz J Med Biol Res. 2000;33(11):1255–1268. doi: 10.1590/s0100-879x2000001100001. [DOI] [PubMed] [Google Scholar]
- 25.Brash A.R. Arachidonic acid as a bioactive molecule. J Clin Invest. 2001;107(11):1339–1345. doi: 10.1172/JCI13210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Beck R., Bertolino S., Abbot S.E., Aaronson P.I., Smirnov S.V. Modulation of arachidonic acid release and membrane fluidity by albumin in vascular smooth muscle and endothelial cells. Circ Res. 1998;83(9):923–931. doi: 10.1161/01.res.83.9.923. [DOI] [PubMed] [Google Scholar]
- 27.Söderberg M., Edlund C., Kristensson K., Dallner G. Fatty acid composition of brain phospholipids in aging and in Alzheimer's disease. Lipids. 1991;26(6):421–425. doi: 10.1007/BF02536067. [DOI] [PubMed] [Google Scholar]
- 28.Kotani S., Nakazawa H., Tokimasa T., Akimoto K., Kawashima H., Toyoda-Ono Y. Synaptic plasticity preserved with arachidonic acid diet in aged rats. Neurosci Res. 2003;46(4):453–461. doi: 10.1016/s0168-0102(03)00123-8. [DOI] [PubMed] [Google Scholar]
- 29.Fukaya T., Gondaira T., Kashiyae Y., Kotani S., Ishikura Y., Fujikawa S. Arachidonic acid preserves hippocampal neuron membrane fluidity in senescent rats. Neurobiol Aging. 2007;28(8):1179–1186. doi: 10.1016/j.neurobiolaging.2006.05.023. [DOI] [PubMed] [Google Scholar]
- 30.Tokuda H., Kontani M., Kawashima H., Kiso Y., Shibata H., Osumi N. Differential effect of arachidonic acid and docosahexaenoic acid on age-related decreases in hippocampal neurogenesis. Neurosci Res. 2014;88:58–66. doi: 10.1016/j.neures.2014.08.002. [DOI] [PubMed] [Google Scholar]
- 31.Tokuda H., Kontani M., Kawashima H., Akimoto K., Kusumoto A., Kiso Y. Arachidonic acid-enriched triacylglycerol improves cognitive function in elderly with low serum levels of arachidonic acid. J Oleo Sci. 2014;63(3):219–227. doi: 10.5650/jos.ess13195. [DOI] [PubMed] [Google Scholar]
- 32.Swartz K.J. Opening the gate in potassium channels. Nat Struct Mol Biol. 2004;11(6):499–501. doi: 10.1038/nsmb0604-499. [DOI] [PubMed] [Google Scholar]
- 33.Swartz K.J. Towards a structural view of gating in potassium channels. Nat Rev Neurosci. 2004;5(12):905–916. doi: 10.1038/nrn1559. [DOI] [PubMed] [Google Scholar]
- 34.Villarroel A., Schwarz T.L. Inhibition of the Kv4 (Shal) family of transient K+ currents by arachidonic acid. J Neurosci. 1996;16(8):2522–2532. doi: 10.1523/JNEUROSCI.16-08-02522.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yazdi S., Stein M., Elinder F., Andersson M., Lindahl E. The Molecular basis of polyunsaturated fatty acid interactions with the Shaker voltage-gated potassium channel. PLoS Comput Biol. 2016;12(1):e1004704. doi: 10.1371/journal.pcbi.1004704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yellen G. The moving parts of voltage-gated ion channels. Q Rev Biophys. 1998;31(3):239–295. doi: 10.1017/s0033583598003448. [DOI] [PubMed] [Google Scholar]
- 37.Börjesson S.I., Hammarström S., Elinder F. Lipoelectric modification of ion channel voltage gating by polyunsaturated fatty acids. Biophys J. 2008;95(5):2242–2253. doi: 10.1529/biophysj.108.130757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Börjesson S.I., Parkkari T., Hammarström S., Elinder F. Electrostatic tuning of cellular excitability. Biophys J. 2010;98(3):396–403. doi: 10.1016/j.bpj.2009.10.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Elinder F., Liin S.I. Actions and mechanisms of polyunsaturated fatty acids on voltage-gated ion channels. Front Physiol. 2017;8:43. doi: 10.3389/fphys.2017.00043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Horimoto N., Nabekura J., Ogawa T. Arachidonic acid activation of potassium channels in rat visual cortex neurons. Neuroscience. 1997;77(3):661–671. doi: 10.1016/s0306-4522(96)00490-3. [DOI] [PubMed] [Google Scholar]
- 41.Ordway RW, Walsh JV Jr, Singer JJ. Arachidonic acid and other fatty acids directly activate potassium channels in smooth muscle cells. Science 1989;244(4909):1176–9. [DOI] [PubMed]
- 42.Ordway R.W., Singer J.J., Walsh J.V., Jr. Direct regulation of ion channels by fatty acids. Trends Neurosci. 1991;14(3):96–100. doi: 10.1016/0166-2236(91)90069-7. [DOI] [PubMed] [Google Scholar]
- 43.Kirber M.T., Ordway R.W., Clapp L.H., Walsh J.V., Jr, Singer J.J. Both membrane stretch and fatty acids directly activate large conductance Ca(2+)-activated K+ channels in vascular smooth muscle cells. FEBS Lett. 1992;297(1–2):24–28. doi: 10.1016/0014-5793(92)80319-c. [DOI] [PubMed] [Google Scholar]
- 44.Kuang Q., Purhonen P., Hebert H. Structure of potassium channels. Cell Mol Life Sci. 2015;72(19):3677–3693. doi: 10.1007/s00018-015-1948-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Denson D.D., Wang X., Worrell R.T., Eaton D.C. Effects of fatty acids on BK channels in GH(3) cells. Am J Physiol Cell Physiol. 2000;279(4):C1211–C1219. doi: 10.1152/ajpcell.2000.279.4.C1211. [DOI] [PubMed] [Google Scholar]
- 46.Hamilton K.L., Syme C.A., Devor D.C. Molecular localization of the inhibitory arachidonic acid binding site to the pore of hIK1. J Biol Chem. 2003;278(19):16690–16697. doi: 10.1074/jbc.M212959200. [DOI] [PubMed] [Google Scholar]
- 47.Patel A.J., Lazdunski M., Honoré E. Lipid and mechano-gated 2P domain K(+) channels. Curr Opin Cell Biol. 2001;13(4):422–428. doi: 10.1016/s0955-0674(00)00231-3. [DOI] [PubMed] [Google Scholar]
- 48.Kim D. Fatty acid-sensitive two-pore domain K+ channels. Trends Pharmacol Sci. 2003;24(12):648–654. doi: 10.1016/j.tips.2003.10.008. [Review] [DOI] [PubMed] [Google Scholar]
- 49.Kim D., Duff R.A. Regulation of K+ channels in cardiac myocytes by free fatty acids. Circ Res. 1990;67(4):1040–1046. doi: 10.1161/01.res.67.4.1040. [DOI] [PubMed] [Google Scholar]
- 50.Kang J.X., Leaf A. Prevention of fatal cardiac arrhythmias by polyunsaturated fatty acids. Am J Clin Nutr. 2000;71(1 Suppl):202S–207S. doi: 10.1093/ajcn/71.1.202S. [DOI] [PubMed] [Google Scholar]
- 51.Gu H., Fang Y.J., He Y.L., Sun J., Zhu J., Mei Y.A. Modulation of muscle rNaV1.4 Na+ channel isoform by arachidonic acid and its non-metabolized analog. J Cell Physiol. 2009;219(1):173–182. doi: 10.1002/jcp.21664. [DOI] [PubMed] [Google Scholar]
- 52.Tewari K.P., Malinowska D.H., Sherry A.M., Cuppoletti J. PKA and arachidonic acid activation of human recombinant ClC-2 chloride channels. Am J Physiol Cell Physiol. 2000;279(1):C40–C50. doi: 10.1152/ajpcell.2000.279.1.C40. [DOI] [PubMed] [Google Scholar]
- 53.Cuppoletti J., Tewari K.P., Sherry A.M., Kupert E.Y., Malinowska D.H. ClC-2 Cl- channels in human lung epithelia: activation by arachidonic acid, amidation, and acid-activated omeprazole. Am J Physiol Cell Physiol. 2001;281(1):C46–C54. doi: 10.1152/ajpcell.2001.281.1.C46. [DOI] [PubMed] [Google Scholar]
- 54.Linsdell P. Inhibition of cystic fibrosis transmembrane conductance regulator chloride channel currents by arachidonic acid. Can J Physiol Pharmacol. 2000;78(6):490–499. [PubMed] [Google Scholar]
- 55.Zhou J.J., Linsdell P. Molecular mechanism of arachidonic acid inhibition of the CFTR chloride channel. Eur J Pharmacol. 2007;563(1–3):88–91. doi: 10.1016/j.ejphar.2007.02.048. [DOI] [PubMed] [Google Scholar]
- 56.Kawanabe A., Okamura Y. Effects of unsaturated fatty acids on the kinetics of voltage-gated proton channels heterologously expressed in cultured cells. J Physiol. 2016;594(3):595–610. doi: 10.1113/JP271274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Nielsen M., Witt M.R., Thøgersen H. [3H]diazepam specific binding to rat cortex in vitro is enhanced by oleic, arachidonic and docosahexenoic acid isolated from pig brain. Eur J Pharmacol. 1988;146(2–3):349–353. doi: 10.1016/0014-2999(88)90315-9. [DOI] [PubMed] [Google Scholar]
- 58.Witt M.R., Westh-Hansen S.E., Rasmussen P.B., Hastrup S., Nielsen M. Unsaturated free fatty acids increase benzodiazepine receptor agonist binding depending on the subunit composition of the GABAA receptor complex. J Neurochem. 1996;67(5):2141–2145. doi: 10.1046/j.1471-4159.1996.67052141.x. [DOI] [PubMed] [Google Scholar]
- 59.Witt M.R., Poulsen C.F., Lükensmejer B., Westh-Hansen S.E., Nabekura J., Akaike N. Structural requirements for the interaction of unsaturated free fatty acids with recombinant human GABAA receptor complexes. Ann N Y Acad Sci. 1999;868:697–700. doi: 10.1111/j.1749-6632.1999.tb11349.x. [DOI] [PubMed] [Google Scholar]
- 60.Antollini SS, Barrantes FJ. Fatty acid regulation of voltage- and ligand-gated ion channel function. Front Physiol 2016;7:573 [eCollection 2016. Review]. [DOI] [PMC free article] [PubMed]
- 61.Antollini S.S., Barrantes F.J. Unique effects of different fatty acid species on the physical properties of the torpedo acetylcholine receptor membrane. J Biol Chem. 2002;277(2):1249–1254. doi: 10.1074/jbc.M106618200. [DOI] [PubMed] [Google Scholar]
- 62.Nievas G.A., Barrantes F.J., Antollini S.S. Conformation-sensitive steroid and fatty acid sites in the transmembrane domain of the nicotinic acetylcholine receptor. Biochemistry. 2007;46(11):3503–3512. doi: 10.1021/bi061388z. [DOI] [PubMed] [Google Scholar]
- 63.Fernández Nievas G.A., Barrantes F.J., Antollini S.S. Modulation of nicotinic acetylcholine receptor conformational state by free fatty acids and steroids. J Biol Chem. 2008;283(31):21478–21486. doi: 10.1074/jbc.M800345200. [DOI] [PubMed] [Google Scholar]
- 64.Pompéia C., Cury-Boaventura M.F., Curi R. Arachidonic acid triggers an oxidative burst in leukocytes. Braz J Med Biol Res. 2003;36(11):1549–1560. doi: 10.1590/s0100-879x2003001100013. [DOI] [PubMed] [Google Scholar]
- 65.Henderson L.M., Thomas S., Banting G., Chappell J.B. The arachidonate-activatable, NADPH oxidase-associated H+ channel is contained within the multi-membrane-spanning N-terminal region of gp91-phox. Biochem J. 1997;325(Pt 3):701–705. doi: 10.1042/bj3250701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Henderson L.M. Role of histidines identified by mutagenesis in the NADPH oxidase-associated H+ channel. J Biol Chem. 1998;273(50):33216–33223. doi: 10.1074/jbc.273.50.33216. [DOI] [PubMed] [Google Scholar]
- 67.Shiose A., Sumimoto H. Arachidonic acid and phosphorylation synergistically induce a conformational change of p47phox to activate the phagocyte NADPH oxidase. J Biol Chem. 2000;275(18):13793–13801. doi: 10.1074/jbc.275.18.13793. [DOI] [PubMed] [Google Scholar]
- 68.Jayadev S., Linardic C.M., Hannun Y.A. Identification of arachidonic acid as a mediator of sphingomyelin hydrolysis in response to tumor necrosis factor alpha. J Biol Chem. 1994;269(8):5757–5763. [PubMed] [Google Scholar]
- 69.Robinson B.S., Hii C.S., Poulos A., Ferrante A. Activation of neutral sphingomyelinase in human neutrophils by polyunsaturated fatty acids. Immunology. 1997;91(2):274–280. doi: 10.1046/j.1365-2567.1997.d01-2227.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Hofmann K., Tomiuk S., Wolff G., Stoffel W. Cloning and characterization of the mammalian brain-specific, Mg2+-dependent neutral sphingomyelinase. Proc Natl Acad Sci USA. 2000;97(11):5895–5900. doi: 10.1073/pnas.97.11.5895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Huwiler A., Kolter T., Pfeilschifter J., Sandhoff K. Physiology and pathophysiology of sphingolipid metabolism and signaling. Biochim Biophys Acta. 2000;1485(2–3):63–99. doi: 10.1016/s1388-1981(00)00042-1. [DOI] [PubMed] [Google Scholar]
- 72.Worgall T.S., Johnson R.A., Seo T., Gierens H., Deckelbaum R.J. Unsaturated fatty acid-mediated decreases in sterol regulatory element-mediated gene transcription are linked to cellular sphingolipid metabolism. J Biol Chem. 2002;277(6):3878–3885. doi: 10.1074/jbc.M102393200. [DOI] [PubMed] [Google Scholar]
- 73.Marchesini N., Hannun Y.A. Acid and neutral sphingomyelinases: roles and mechanisms of regulation. Biochem Cell Biol. 2004;82(1):27–44. doi: 10.1139/o03-091. [DOI] [PubMed] [Google Scholar]
- 74.Tallima H., Salah M., El-Ridi R. In vitro and in vivo effects of unsaturated fatty acids on Schistosoma mansoni and S. haematobium lung-stage larvae. J Parasitol. 2005;91(5):1094–1102. doi: 10.1645/GE-514R.1. [DOI] [PubMed] [Google Scholar]
- 75.El Ridi R., Tallima H. Equilibrium in lung schistosomula sphingomyelin breakdown and biosynthesis allows very small molecules, but not antibody, to access proteins at the host-parasite interface. J Parasitol. 2006;92(4):730–737. doi: 10.1645/GE-745R1.1. [DOI] [PubMed] [Google Scholar]
- 76.Tallima H., Al-Halbosiy M.F., El Ridi R. Enzymatic activity and immunolocalization of Schistosoma mansoni and Schistosoma haematobium neutral sphingomyelinase. Mol Biochem Parasitol. 2011;178(1–2):23–28. doi: 10.1016/j.molbiopara.2011.04.003. [DOI] [PubMed] [Google Scholar]
- 77.El Ridi R., Tallima H., Migliardo F. Biochemical and biophysical methodologies open the road for effective schistosomiasis therapy and vaccination. Biochim Biophys Acta. 2017;1861(Pt B):3613–3620. doi: 10.1016/j.bbagen.2016.03.036. [DOI] [PubMed] [Google Scholar]
- 78.Chilton F.H., Surette M.E., Winkler J.D. Arachidonate-phospholipid remodeling and cell proliferation. Adv Exp Med Biol. 1996;416:169–172. [PubMed] [Google Scholar]
- 79.Surette M.E., Fonteh A.N., Bernatchez C., Chilton F.H. Perturbations in the control of cellular arachidonic acid levels block cell growth and induce apoptosis in HL-60 cells. Carcinogenesis. 1999;20(5):757–763. doi: 10.1093/carcin/20.5.757. [DOI] [PubMed] [Google Scholar]
- 80.Cao Y., Pearman A.T., Zimmerman G.A., McIntyre T.M., Prescott S.M. Intracellular unesterified arachidonic acid signals apoptosis. Proc Natl Acad Sci USA. 2000;97(21):11280–11285. doi: 10.1073/pnas.200367597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Pompeia C., Lima T., Curi R. Arachidonic acid cytotoxicity: can arachidonic acid be a physiological mediator of cell death? Cell Biochem Funct. 2003;21(2):97–104. doi: 10.1002/cbf.1012. [Review] [DOI] [PubMed] [Google Scholar]
- 82.Innis S.M. Impact of maternal diet on human milk composition and neurological development of infants. Am J Clin Nutr. 2014;99(3):734S–741S. doi: 10.3945/ajcn.113.072595. [DOI] [PubMed] [Google Scholar]
- 83.Crawford M.A., Costeloe K., Ghebremeskel K., Phylactos A., Skirvin L., Stacey F. Are deficits of arachidonic and docosahexaenoic acids responsible for the neural and vascular complications of preterm babies? Am J Clin Nutr. 1997;66(4 Suppl):1032S–1041S. doi: 10.1093/ajcn/66.4.1032S. [DOI] [PubMed] [Google Scholar]
- 84.Senanayake S.P.J.N., Jaouad Fichtali J. Single-cell oils as sources of nutraceutical and specialty lipids: processing technologies and applications. In: Shahidi F., editor. Nutraceutical and specialty lipids and their co-products. Taylor and Francis; Florida: 2006. pp. 252–280. [chapter 16] [Google Scholar]
- 85.WHO and FAO joint consultation Fats and oils in human nutrition. Nutr Rev. 1995;53(7):202–205. doi: 10.1111/j.1753-4887.1995.tb01552.x. [DOI] [PubMed] [Google Scholar]
- 86.Tallima H., Hadley K., El Ridi R. Praziquantel and arachidonic acid combination. Innovative approach to the treatment of Schistosomiasis. In: Amidou S., editor. An overview of tropical diseases. InTech; Rijeka: 2015. pp. 145–172. [Google Scholar]
- 87.Hadley K.B., Ryan A.S., Forsyth S., Gautier S., Salem N., Jr. The Essentiality of arachidonic acid in infant development. Nutrients. 2016;8(4):216. doi: 10.3390/nu8040216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Robinson D.T., Martin C.R. Fatty acid requirements for the preterm infant. Semin Fetal Neonatal Med. 2017;22(1):8–14. doi: 10.1016/j.siny.2016.08.009. [DOI] [PubMed] [Google Scholar]
- 89.EFSA Panel on Dietetic Products Nutrition and Allergies. Scientific Opinion on the essential composition of infant and follow-on formulae. EFSA J. 2014;12(7):3760. [Google Scholar]
- 90.Salem N., Jr, Wegher B., Mena P., Uauy R. Arachidonic and docosahexaenoic acids are biosynthesized from their 18-carbon precursors in human infants. Proc Natl Acad Sci USA. 1996;93(1):49–54. doi: 10.1073/pnas.93.1.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Rapoport S.I. Arachidonic acid and the brain. J Nutr. 2008;138(12):2515–2520. doi: 10.1093/jn/138.12.2515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Crawford M.A., Golfetto I., Ghebremeskel K., Min Y., Moodley T., Poston L. The potential role for arachidonic and docosahexaenoic acids in protection against some central nervous system injuries in preterm infants. Lipids. 2003;38(4):303–315. doi: 10.1007/s11745-003-1065-1. [Review] [DOI] [PubMed] [Google Scholar]
- 93.Meguid N.A., Atta H.M., Gouda A.S., Khalil R.O. Role of polyunsaturated fatty acids in the management of Egyptian children with autism. Clin Biochem. 2008;41(13):1044–1048. doi: 10.1016/j.clinbiochem.2008.05.013. [DOI] [PubMed] [Google Scholar]
- 94.Yui K., Koshiba M., Nakamura S., Kobayashi Y. Effects of large doses of arachidonic acid added to docosahexaenoic acid on social impairment in individuals with autism spectrum disorders: a double-blind, placebo-controlled, randomized trial. J Clin Psychopharmacol. 2012;32(2):200–206. doi: 10.1097/JCP.0b013e3182485791. [DOI] [PubMed] [Google Scholar]
- 95.Baur L.A., O'Connor J., Pan D.A., Kriketos A.D., Storlien L.H. The fatty acid composition of skeletal muscle membrane phospholipid: its relationship with the type of feeding and plasma glucose levels in young children. Metabolism. 1998;47(1):106–112. doi: 10.1016/s0026-0495(98)90202-5. [DOI] [PubMed] [Google Scholar]
- 96.Salem N.M., Lin Y.H., Moriguchi T., Lim S.Y., Salem N., Jr, Hibbeln J.R. Distribution of omega-6 and omega-3 polyunsaturated fatty acids in the whole rat body and 25 compartments. Prostaglandins Leukot Essent Fatty Acids. 2015;100:13–20. doi: 10.1016/j.plefa.2015.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.El Ridi R., Mohamed S.H., Tallima H. Incubation of Schistosoma mansoni lung-stage schistosomula in corn oil exposes their surface membrane antigenic specificities. J Parasitol. 2003;89(5):1064–1067. doi: 10.1645/GE-3122RN. [DOI] [PubMed] [Google Scholar]
- 98.El Ridi R.A., Tallima H.A. Novel therapeutic and prevention approaches for schistosomiasis: review. J Adv Res. 2013;4(5):467–478. doi: 10.1016/j.jare.2012.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Othman A., El Ridi R. Schistosomiasis. In: Bruschi F., editor. Helminth infections and their impact on global public health. Springer; Heidelberg: 2014. pp. 49–92. [Google Scholar]
- 100.El Ridi R., Aboueldahab M., Tallima H., Salah M., Mahana N., Fawzi S. In vitro and in vivo activities of arachidonic acid against Schistosoma mansoni and Schistosoma haematobium. Antimicrob Agents Chemother. 2010;54(8):3383–3389. doi: 10.1128/AAC.00173-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.El Ridi R., Tallima H., Salah M., Aboueldahab M., Fahmy O.M., Al-Halbosiy M.F. Efficacy and mechanism of action of arachidonic acid in the treatment of hamsters infected with Schistosoma mansoni or Schistosoma haematobium. Int J Antimicrob Agents. 2012;39(3):232–239. doi: 10.1016/j.ijantimicag.2011.08.019. [DOI] [PubMed] [Google Scholar]
- 102.Selim S., El Sagheer O., El Amir A., Barakat R., Hadley K., Bruins M.J. Efficacy and safety of arachidonic acid for treatment of Schistosoma mansoni-infected children in Menoufiya, Egypt. Am J Trop Med Hyg. 2014;91(5):973–981. doi: 10.4269/ajtmh.14-0328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Barakat R., Abou El-Ela N.E., Sharaf S., El Sagheer O., Selim S., Tallima H. Efficacy and safety of arachidonic acid for treatment of school-age children in Schistosoma mansoni high-endemicity regions. Am J Trop Med Hyg. 2015;92(4):797–804. doi: 10.4269/ajtmh.14-0675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Amaral K.B., Silva T.P., Malta K.K., Carmo L.A., Dias F.F., Almeida M.R. Natural Schistosoma mansoni infection in the wild reservoir Nectomys squamipes leads to excessive lipid droplet accumulation in hepatocytes in the absence of liver functional impairment. PLoS ONE. 2016;11(11):e0166979. doi: 10.1371/journal.pone.0166979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Hanna V.S., Gawish A., Abou El Dahab M., Tallima H., El Ridi R. Is arachidonic acid an endoschistosomicide? J Adv Res. 2018;11:81–89. doi: 10.1016/j.jare.2018.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.El-Faham M.H., Eissa M.M., Igetei J.E., Amer E.I., Liddell S., El-Azzouni M.Z. Treatment of Schistosoma mansoni with miltefosine in vitro enhances serological recognition of defined worm surface antigens. PLoS Negl Trop Dis. 2017;11(8):e0005853. doi: 10.1371/journal.pntd.0005853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Siegel I., Liu T.L., Yaghoubzadeh E., Keskey T.S., Gleicher N. Cytotoxic effects of free fatty acids on ascites tumor cells. J Natl Cancer Inst. 1987;78(2):271–277. [PubMed] [Google Scholar]
- 108.Das U.N. Gamma-linolenic acid, arachidonic acid, and eicosapentaenoic acid as potential anticancer drugs. Nutrition. 1990;6(6):429–434. [PubMed] [Google Scholar]
- 109.Das U.N. Tumoricidal action of cis-unsaturated fatty acids and their relationship to free radicals and lipid peroxidation. Cancer Lett. 1991;56(3):235–243. doi: 10.1016/0304-3835(91)90008-6. [DOI] [PubMed] [Google Scholar]
- 110.Sagar P.S., Das U.N. Cytotoxic action of cis-unsaturated fatty acids on human cervical carcinoma (HeLa) cells in vitro. Prostaglandins Leukot Essent Fatty Acids. 1995;53(4):287–299. doi: 10.1016/0952-3278(95)90129-9. [DOI] [PubMed] [Google Scholar]
- 111.Ramesh G., Das U.N. Effect of cis-unsaturated fatty acids on Meth-A ascitic tumour cells in vitro and in vivo. Cancer Lett. 1998;123(2):207–214. doi: 10.1016/s0304-3835(97)00426-6. [DOI] [PubMed] [Google Scholar]
- 112.Das U.N., Madhavi N. Effect of polyunsaturated fatty acids on drug-sensitive and resistant tumor cells in vitro. Lipids Health Dis. 2011;10:159. doi: 10.1186/1476-511X-10-159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Dai J., Shen J., Pan W., Shen S., Das U.N. Effects of polyunsaturated fatty acids on the growth of gastric cancer cells in vitro. Lipids Health Dis. 2013;12:71. doi: 10.1186/1476-511X-12-71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Meng H., Shen Y., Shen J., Zhou F., Shen S., Das U.N. Effect of n-3 and n-6 unsaturated fatty acids on prostate cancer (PC-3) and prostate epithelial (RWPE-1) cells in vitro. Lipids Health Dis. 2013;12:160. doi: 10.1186/1476-511X-12-160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Zhang C., Yu H., Shen Y., Ni X., Shen S., Das U.N. Polyunsaturated fatty acids trigger apoptosis of colon cancer cells through a mitochondrial pathway. Arch Med Sci. 2015;11(5):1081–1094. doi: 10.5114/aoms.2015.54865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Petrik M.B., McEntee M.F., Chiu C.H., Whelan J. Antagonism of arachidonic acid is linked to the antitumorigenic effect of dietary eicosapentaenoic acid in Apc(Min/+) mice. J Nutr. 2000;130(5):1153–1158. doi: 10.1093/jn/130.5.1153. [DOI] [PubMed] [Google Scholar]
- 117.Arenz C., Giannis A. Synthesis of the first selective irreversible inhibitor of neutral sphingomyelinase. Angew Chem Int Ed Engl. 2000;39(8):1440–1442. doi: 10.1002/(sici)1521-3773(20000417)39:8<1440::aid-anie1440>3.0.co;2-r. [DOI] [PubMed] [Google Scholar]
- 118.Colombo D.T., Tran L.K., Speck J.J., Reitz R.C. Comparison of hexadecyl phosphocholine with fish oil as an antitumor agent. J Lipid Mediat Cell Signal. 1997;17(1):47–63. doi: 10.1016/s0929-7855(97)00020-5. [DOI] [PubMed] [Google Scholar]
- 119.Wieder T., Orfanos C.E., Geilen C.C. Induction of ceramide-mediated apoptosis by the anticancer phospholipid analog, hexadecylphosphocholine. J Biol Chem. 1998;273(18):11025–11031. doi: 10.1074/jbc.273.18.11025. [DOI] [PubMed] [Google Scholar]
- 120.Jiménez-López J.M., Carrasco M.P., Marco C., Segovia J.L. Hexadecylphospho-choline disrupts cholesterol homeostasis and induces the accumulation of free cholesterol in HepG2 tumour cells. Biochem Pharmacol. 2006;71(8):1114–1121. doi: 10.1016/j.bcp.2005.08.001. [DOI] [PubMed] [Google Scholar]
- 121.Marco C., Jiménez-López J.M., Ríos-Marco P., Segovia J.L., Carrasco M.P. Hexadecylphosphocholine alters nonvesicular cholesterol traffic from the plasma membrane to the endoplasmic reticulum and inhibits the synthesis of sphingomyelin in HepG2 cells. Int J Biochem Cell Biol. 2009;41(6):1296–1303. doi: 10.1016/j.biocel.2008.11.004. [DOI] [PubMed] [Google Scholar]
- 122.Korotkova M., Jakobsson P.J. Persisting eicosanoid pathways in rheumatic diseases. Nat Rev Rheumatol. 2014;10(4):229–241. doi: 10.1038/nrrheum.2014.1. [DOI] [PubMed] [Google Scholar]
- 123.Korotkova M., Lundberg I.E. The skeletal muscle arachidonic acid cascade in health and inflammatory disease. Nat Rev Rheumatol. 2014;10(5):295–303. doi: 10.1038/nrrheum.2014.2. [DOI] [PubMed] [Google Scholar]
- 124.Esser-von Bieren J. Immune-regulation and functions of eicosanoid lipid mediators. Biol Chem. 2017;398(11):1177–1191. doi: 10.1515/hsz-2017-0146. [DOI] [PubMed] [Google Scholar]
- 125.Maderna P., Godson C. Lipoxins: resolutionary road. Br J Pharmacol. 2009;158(4):947–959. doi: 10.1111/j.1476-5381.2009.00386.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Serhan C.N., Chiang N. Resolution phase lipid mediators of inflammation: agonists of resolution. Curr Opin Pharmacol. 2013;13:632–640. doi: 10.1016/j.coph.2013.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Serhan C.N. Pro-resolving lipid mediators are leads for resolution physiology. Nature. 2014;510(7503):92–101. doi: 10.1038/nature13479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Serhan C.N., Chiang N., Dalli J. The resolution code of acute inflammation: novel pro-resolving lipid mediators in resolution. Semin Immunol. 2015;27(3):200–215. doi: 10.1016/j.smim.2015.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Börgeson E., McGillicuddy F.C., Harford K.A., Corrigan N., Higgins D.F., Maderna P. Lipoxin A4 attenuates adipose inflammation. FASEB J. 2012;26(10):4287–4294. doi: 10.1096/fj.12-208249. [DOI] [PubMed] [Google Scholar]
- 130.Reis M.B., Pereira P.A.T., Caetano G.F., Leite M.N., Galvão A.F., Paula-Silva F.W.G. Lipoxin A4 encapsulated in PLGA microparticles accelerates wound healing of skin ulcers. PLoS ONE. 2017;12(7):e0182381. doi: 10.1371/journal.pone.0182381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Barnig C., Levy B.D. Lipoxin A4: a new direction in asthma therapy? Expert Rev Clin Immunol. 2013;9(6):491–493. doi: 10.1586/eci.13.36. [DOI] [PubMed] [Google Scholar]
- 132.Barnig C., Cernadas M., Dutile S., Liu X., Perrella M.A., Kazani S. Lipoxin A4 regulates natural killer cell and type 2 innate lymphoid cell activation in asthma. Sci Transl Med. 2013;5(174):174ra26. doi: 10.1126/scitranslmed.3004812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Pozzi A., Zent R. Regulation of endothelial cell functions by basement membrane- and arachidonic acid-derived products. Wiley Interdiscip Rev Syst Biol Med. 2009;1(2):254–272. doi: 10.1002/wsbm.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Berry E., Liu Y., Chen L., Guo A.M. Eicosanoids: emerging contributors in stem cell-mediated wound healing. Prostaglandins Other Lipid Mediat. 2017;132:17–24. doi: 10.1016/j.prostaglandins.2016.11.001. [DOI] [PubMed] [Google Scholar]
- 135.Das U.N. Arachidonic acid and lipoxin A4 as possible endogenous anti-diabetic molecules. Prostaglandins Leukot Essent Fatty Acids. 2013;88(3):201–210. doi: 10.1016/j.plefa.2012.11.009. [DOI] [PubMed] [Google Scholar]
- 136.Das U.N. Is there a role for bioactive lipids in the pathobiology of diabetes mellitus? Front Endocrinol (Lausanne) 2017;8:182. doi: 10.3389/fendo.2017.00182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Bisogno T., Maccarrone M. Endocannabinoid signaling and its regulation by nutrients. BioFactors. 2014;40(4):373–380. doi: 10.1002/biof.1167. [DOI] [PubMed] [Google Scholar]
- 138.Wei D., Lee D., Li D., Daglian J., Jung K.M., Piomelli D. A role for the endocannabinoid 2-arachidonoyl-sn-glycerol for social and high-fat food reward in male mice. Psychopharmacology. 2016;233(10):1911–1919. doi: 10.1007/s00213-016-4222-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Wei D., Allsop S., Tye K., Piomelli D. Endocannabinoid signaling in the control of social behavior. Trends Neurosci. 2017;40(7):385–396. doi: 10.1016/j.tins.2017.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Piomelli D. The endocannabinoid system: a drug discovery perspective. Curr Opin Investig Drugs. 2005;6(7):672–679. [PubMed] [Google Scholar]
- 141.More surprises lying ahead The endocannabinoids keep us guessing. Neuropharmacology. 2014;76(Pt B):228–234. doi: 10.1016/j.neuropharm.2013.07.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Amoako A.A., Marczylo T.H., Marczylo E.L., Elson J., Willets J.M., Taylor A.H. Anandamide modulates human sperm motility: implications for men with asthenozoospermia and oligoasthenoteratozoospermia. Hum Reprod. 2013;28(8):2058–2066. doi: 10.1093/humrep/det232. [DOI] [PubMed] [Google Scholar]
- 143.Alhouayek M., Muccioli G.G. The endocannabinoid system in inflammatory bowel diseases: from pathophysiology to therapeutic opportunity. Trends Mol Med. 2012;18(10):615–625. doi: 10.1016/j.molmed.2012.07.009. [DOI] [PubMed] [Google Scholar]
- 144.Izzo A.A., Muccioli G.G., Ruggieri M.R., Schicho R. Endocannabinoids and the digestive tract and bladder in health and disease. Handb Exp Pharmacol. 2015;231:423–447. doi: 10.1007/978-3-319-20825-1_15. [DOI] [PubMed] [Google Scholar]
- 145.Moradi H., Oveisi F., Khanifar E., Moreno-Sanz G., Vaziri N.D., Piomelli D. Increased renal 2-arachidonoylglycerol level is associated with improved renal function in a mouse model of acute kidney injury. Cannabis Cannabinoid Res. 2016;1(1):218–228. doi: 10.1089/can.2016.0013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Klose C.S., Artis D. Innate lymphoid cells as regulators of immunity, inflammation and tissue homeostasis. Nat Immunol. 2016;17(7):765–774. doi: 10.1038/ni.3489. [DOI] [PubMed] [Google Scholar]
- 147.Tait Wojno E.D., Artis D. Emerging concepts and future challenges in innate lymphoid cell biology. J Exp Med. 2016;213(11):2229–2248. doi: 10.1084/jem.20160525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Halim T.Y. Group 2 innate lymphoid cells in disease. Int Immunol. 2016;28(1):13–22. doi: 10.1093/intimm/dxv050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Hanna V.S., Hafez E.A. Synopsis of arachidonic acid metabolism- A review. J Adv Res. 2018;11:23–32. doi: 10.1016/j.jare.2018.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Doherty T.A., Khorram N., Lund S., Mehta A.K., Croft M., Broide D.H. Lung type 2 innate lymphoid cells express cysteinyl leukotriene receptor 1, which regulates TH2 cytokine production. J Allergy Clin Immunol. 2013;132(1):205–213. doi: 10.1016/j.jaci.2013.03.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Xue L., Salimi M., Panse I., Mjösberg J.M., McKenzie A.N., Spits H. Prostaglandin D2 activates group 2 innate lymphoid cells through chemoattractant receptor-homologous molecule expressed on TH2 cells. J Allergy Clin Immunol. 2014;133(4):1184–1194. doi: 10.1016/j.jaci.2013.10.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Wojno E.D., Monticelli L.A., Tran S.V., Alenghat T., Osborne L.C., Thome J.J. The prostaglandin D₂ receptor CRTH2 regulates accumulation of group 2 innate lymphoid cells in the inflamed lung. Mucosal Immunol. 2015;8(6):1313–1323. doi: 10.1038/mi.2015.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Xue L., Fergusson J., Salimi M., Panse I., Ussher J.E., Hegazy A.N. Prostaglandin D2 and leukotriene E4 synergize to stimulate diverse TH2 functions and TH2 cell/neutrophil crosstalk. J Allergy Clin Immunol. 2015;135(5):1358–1366. doi: 10.1016/j.jaci.2014.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Salimi M., Stöger L., Liu W., Go S., Pavord I., Klenerman P. Cysteinyl leukotriene E4 activates human group 2 innate lymphoid cells and enhances the effect of prostaglandin D2 and epithelial cytokines. J Allergy Clin Immunol. 2017;140(4):1090–1100. doi: 10.1016/j.jaci.2016.12.958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Lund S.J., Portillo A., Cavagnero K., Baum R.E., Naji L.H., Badrani J.H. Leukotriene C4 potentiates IL-33-induced group 2 innate lymphoid cell activation and lung inflammation. J Immunol. 2017;199(3):1096–1104. doi: 10.4049/jimmunol.1601569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Halim T.Y., Hwang Y.Y., Scanlon S.T., Zaghouani H., Garbi N., Fallon P.G. Group 2 innate lymphoid cells license dendritic cells to potentiate memory TH2 cell responses. Nat Immunol. 2016;17(1):57–64. doi: 10.1038/ni.3294. [DOI] [PMC free article] [PubMed] [Google Scholar]