Abstract
Background
Acute pulmonary embolism may be ruled out by combining nonhigh clinical probability and a normal D‐dimer level. Both antiplatelet drugs and HMG‐CoA reductase inhibitors (statins) have been associated with effects on thrombus formation, potentially influencing D‐dimer levels in this setting, leading to a higher rate of false‐negative tests. Therefore, we determined whether D‐dimer levels in patients with suspected pulmonary embolism are affected by concomitant use of antiplatelet drugs and/or statins and evaluated whether the effect of antiplatelet drugs or statins might affect diagnostic accuracy.
Materials and methods
We performed a posthoc analysis in the YEARS diagnostic study, comparing age‐ and sex‐adjusted D‐dimer levels among users of antiplatelet drugs, statins and nonusers. We then reclassified patients within the YEARS algorithm by developing a model in which we adjusted D‐dimer cut‐offs for statin use and evaluated diagnostic accuracy.
Results
We included 156 statins users, 147 antiplatelet drugs users and 726 nonusers of either drugs, all with suspected pulmonary embolism. Use of antiplatelet drugs did not have a significant effect, whereas statin use was associated with 15% decrease in D‐dimer levels (95% CI, −28% to −0.6%). An algorithm with lower D‐dimer thresholds in statin users yielded lower specificity (0.42 compared to 0.33) with no difference in false‐negative tests.
Conclusions
We conclude that use of statins but not of antiplatelet agents is associated with a modest decrease in D‐dimer levels. Adjusting D‐dimer cut‐offs for statin use did, however, not result in a safer diagnostic strategy in our cohort.
Keywords: fibrin fragment D, hydroxymethylglutaryl‐CoA reductase inhibitors, platelet aggregation inhibitors, pulmonary embolism, venous thromboembolism
1. INTRODUCTION
D‐dimer levels have a central role in the diagnostic work‐up of venous thromboembolism (VTE). Guidelines recommend combining clinical decision rules and a D‐dimer test to identify patients in whom pulmonary embolism (PE) or deep venous thrombosis may be ruled out without performing imaging tests (high level of evidence).1, 2 Recently, the YEARS algorithm, incorporating a variable D‐dimer cut‐off dependent on the pretest probability based on 3 clinical variables (Figure 1), has been proven to be safe and compatible with clinical practice.3
Figure 1.
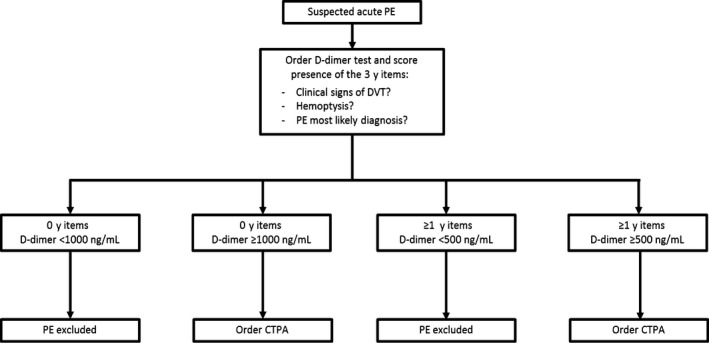
YEARS algorithm¹. CTPA, computed‐tomography pulmonary angiography; DVT, deep vein thrombosis; PE, pulmonary embolism; ¹ Figure derived with permission of publisher from: van der Hulle T, Cheung WY, Kooij S, et al Simplified diagnostic management of suspected pulmonary embolism (the YEARS study): a prospective, multicentre, cohort study. Lancet 2017;390(10091):289‐297
D‐dimers are fibrin degradation fragments, generated after fibrinolysis of a blood clot by the sequential action of thrombin, factor XIIIa and plasmin.4 The D‐dimer level in blood can be influenced by many factors such as age, active malignancy, infection, pregnancy or use of anticoagulants.5, 6, 7, 8 Other drugs that affect thrombus formation and therefore may influence D‐dimer levels as well are antiplatelet drugs and HMG‐CoA reductase inhibitors, more commonly known as statins. Antiplatelet drugs can delay thrombin generation and in general inhibit blood coagulation.9 Statins inhibit a variety of platelet factors and decrease tissue factor activity.10, 11 Through the latter mechanism, a smaller amount of factor X is activated and generation of thrombin is depleted. These effects might explain that statins may exert cardiovascular protective effects that are independent of LDL‐cholesterol lowering, the so‐called pleiotropic effects.12 Indeed, most clinical studies on this subject have found that statin therapy, especially treatment with lipophilic statins (simvastatin, atorvastatin or fluvastatin), does lower D‐dimer levels whereas antiplatelet therapy does not seem to have an effect. 13 Although most studies are retrospective and susceptible to bias due to other factors influencing D‐dimer level.13, 14, 15, 16, 17, 18, 19 Studies on the effect of statins and antiplatelet therapy on the sensitivity of D‐dimer used as diagnostic test in patients with suspected PE are lacking.
To test the hypothesis that D‐dimer cut‐offs may need to be adjusted in statin and/or antiplatelet therapy users, we performed a posthoc analysis of the YEARS diagnostic study.3 D‐dimer levels were compared among users and nonusers of antiplatelet drug and/or statin. Further, we evaluated the rate of false‐negative D‐dimer tests among users of statins, as well as the sensitivity and specificity of D‐dimer thresholds adjusted to statin use.
2. METHODS
2.1. Study design
This was a posthoc analysis within the YEARS study, of which the design was previously described in detail (Netherlands Trial Registry number NTR4193).3 Briefly, 3465 patients with suspected acute PE were included between 5 October 2013 and 9 July 2015 in a prospective multicentre cohort outcome study evaluating the safety and accuracy of the YEARS diagnostic algorithm. In this algorithm, patients were managed by combining simultaneous assessment of a clinical decision rule consisting of 3 items (clinical signs of deep vein thrombosis (DVT), haemoptysis, PE most likely diagnosis) and a D‐dimer test. In patients without YEARS items and D‐dimer <1000 ng/mL, or ≥1 YEARS items and D‐dimer <500 ng/mL, PE was considered excluded without further imaging. All other patients underwent computed‐tomography pulmonary‐angiography (CTPA; Figure 1).
D‐dimer levels were measured upon presentation of the patient, using automated high‐sensitive quantitative D‐dimer assays (according to local practice Vidas D‐dimer Exclusion®; Biomerieux, Marcy‐L’Étoile, France; STA‐LIA®; DiagnosticaStago, Asnieres, France; Innovance®; Siemens, Marburg, Germany). At study inclusion, the following baseline characteristics were assessed: age, sex, body mass index (BMI), smoking status, malignancy, hypertension, CRP level, previous VTE and present use of antiplatelet drugs and statins.
Follow‐up consisted of a scheduled outpatient visit or telephone interview after 3 months. At this visit, information was obtained on complaints suggestive of VTE. In case of clinically suspected VTE during follow‐up, objective diagnostic tests were required, including CTPA for suspected PE and compression ultrasonography for suspected deep vein thrombosis.
2.2. Patients
Hospitalized patients as well as patient visiting the emergency department with clinically suspected acute (first or recurrent) PE were eligible for inclusion in the YEARS study if they were 18 years of age or older. Exclusion criteria were as follows: treatment with therapeutic doses of anticoagulants initiated ≥24 hours prior to eligibility assessment, life expectancy less than 3 months, geographic inaccessibility precluding follow‐up, pregnancy or allergy to intravenous contrast agent. In two of the participating hospitals—the Erasmus University Medical Center and the Leiden University Medical Center—we also obtained data about use of antiplatelet drugs and statins. For that reason, we restricted the present posthoc analysis to patients evaluated in the latter 2 hospitals.
2.3. Study aim and endpoints
The primary aim of this study was to assess the effect of antiplatelet drugs and statins on D‐dimer levels in patients suspected of PE. The secondary aim of this study was to evaluate whether the effect of antiplatelet drugs or statins might affect diagnostic accuracy.
The primary endpoint of this study was the difference in D‐dimer levels among users and nonusers of antiplatelet drugs and statins. The secondary endpoints of this study were the proportion of patients categorized differently in the YEARS algorithm and the change in diagnostic accuracy and diagnostic failure rate (VTE related death, VTE or lost to follow‐up) when the effect of statins on D‐dimer levels is taken into account. Reporting of this study conforms to the STROBE statement and the broader EQUATOR guidelines.20
2.4. Statistical analysis
D‐dimer levels were log‐transformed to normalize the distribution. Multiple linear regression analyses were performed with adjustment for age, sex, current smoking, use of antiplatelet drugs, use of statins, current smoking and history of VTE according to different stepwise regression models. The contribution of these variables to the changes in D‐dimer levels was determined by comparing the explained variance from the different multivariable linear regression analyses. Due to missing data in 397 (43%) patients, we could not adjust for BMI. The variables that explained most of the change in D‐dimer were then identified. We calculated change in D‐dimer levels for each drug exposure by exponentiation of the correlation coefficient and their corresponding 95% confidence intervals (95% CI). A two‐sided P‐value of .05 was considered to indicate statistical significance. All statistical analyses were carried out using “IBM SPSS Statistics for Windows, version 21 (IBM Corp., Armonk, N.Y., USA).”
For the secondary outcome analysis, we tested the amount of patients that should have been managed differently in the YEARS algorithm when the effect of antiplatelet drugs and/or statins on D‐dimer levels was taken into account. Therefore, we developed a model to reclassify antiplatelet drugs and/or statin users by adjusting the D‐dimer cut‐offs. The cut‐offs were adjusted based on the expected change in D‐dimer levels, as derived from the regression analysis, and were inserted posthoc in the YEARS algorithm. Further, we evaluated diagnostic accuracy by comparing the diagnostic failure rate (VTE related death, VTE or lost to follow‐up) with and without this reclassification of antiplatelet drugs and/or statin users in the YEARS algorithm.
3. RESULTS
Within the YEARS study, 925 patients were included in this posthoc analysis. Mean age of these patients was 54 years (standard deviation (SD) 17), and mean BMI was 27 kg/m2 (SD 5.7); 369 of them (39.9%) were male, and 178 (19.2%) current smokers. Following the YEARS algorithm, 395 (42.7%) patients scored no YEARS items, 479 (51.8%) scored 1, 48 (5.2%) scored 2, and 3 (0.3%) patients scored 3 items. PE was diagnosed by CTPA at baseline in 16.1% of the patients not using an antiplatelet drug or a statin, compared to 17.1% of the patients using either drug. Among the group of patients in whom PE was excluded using the YEARS algorithm, only 2 developed a PE during follow‐up and in 1 patient, PE could not be excluded as cause of death. None of these patients used antiplatelet drugs or statins. In total, 5 patients were lost to follow‐up and four of them did not use either drug.
The mean D‐dimer level was 1666 ng/mL (SD 1642 ng/mL) and the median 985 ng/mL (interquartile range 479‐1203 ng/mL). Of all patients, 22% used an antiplatelet agent and/or statin. More than half of them (n = 104) were using both drugs, 43 patients were only using antiplatelet drugs and 52 patients only statins. In 72% of the statin users, a lipophilic statin (simvastatin, atorvastatin or fluvastatin) had been prescribed. Median D‐dimer values were 912 ng/mL (interquartile range 573‐1941 ng/mL) in lipophilic statin users compared to 1050 ng/mL (558‐1540 ng/mL) in hydrophilic statin (rosuvastatin and pravastatin) users, but this difference was nonsignificant. Compared to nonusers, antiplatelet drug and/or statin users were older, had a higher BMI, smoked less, and the proportion of male gender, previous VTE and the presence of hypertension was higher (Table 1). There was no difference in the median level of CRP or the presence of malignancy between users and nonusers of antiplatelet drug and/or statins.
Table 1.
General characteristics of patients
| No use of statins and antiplatelet drugs (n = 726) | Only statins (n = 52) | Only antiplatelet drugs (n = 43) | Statins and antiplatelet drugs (n = 104) | Statins and/or antiplatelet drugsa , b (n = 199) | P‐valuec | |
|---|---|---|---|---|---|---|
| Male (%) | 271 (37.3) | 21 (40.4) | 21 (48.8) | 56 (53.8) | 98 (49.2) | .002 |
| Age | 48.7 (17.2) | 61.7 (12.0) | 69.6 (14.7) | 67.5 (11.9) | 66.4 (12.9) | <.001 |
| Body mass index (kg m¯²) | 26.0 (5.4) | 30.1 (6.5) | 26.5 (5.9) | 28.8 (5.6) | 28.6 (6.0) | <.001 |
| CRP (mg/L) | 9 (3‐32) | 10 (3‐26) | 7 (3‐29) | 7 (3‐19) | 7 (3‐22) | .89 |
| Hypertension | 108 (14.9) | 33 (63.5) | 19 (45.2) | 67 (65.0) | 119 (60.4) | <.001 |
| Current smoker | 154 (21.9) | 6 (11.8) | 6 (14.0) | 12 (11.7) | 24 (12.2) | .003 |
| Prior VTE | 71 (9.8) | 11 (21.2) | 8 (18.6) | 11 (10.6) | 30 (15.1) | .03 |
| Malignancy | 104 (14.3) | 6 (11.5) | 8 (18.6) | 11 (10.6) | 25 (12.6) | .52 |
| PE at CTPA | 117 (16.1) | 13 (25.0) | 10 (23.3) | 11 (10.6) | 34 (17.1) | .45 |
| Death of any reason during follow‐up | 44 (6.1) | 1 (1.9) | 2 (4.7) | 2 (1.9) | 5 (2.5) | NA |
| PE during follow‐up | 2 (0.3) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | NA |
| Lost to follow‐up | 1 (0.1) | 0 (0) | 0 (0) | 0 (0) | 1 (0.5) | NA |
CTPA, computed‐tomography pulmonary‐angiography; PE, pulmonary embolism; VTE, venous thromboembolism. Data are shown as mean values (SD); numbers (percentages) or median (interquartile ranges). Due to missing data, n = 529 for body mass index; n = 897 for CRP; n = 920 for hypertension; n = 901 for smoking; n = 924 for prior VTE; n = 924 for malignancy.
antiplatelet drugs: acetylsalicylic acid n = 136; clopidogrel n = 6; other n = 3; unknown n = 2;
statins: simvastatin n = 73; rosuvastatin n = 22; atorvastatin n = 36; pravastatin n = 19; fluvastatin n = 3; unknown n = 3.
P‐value between groups no use vs use of statins and/or antiplatelet drugs; Pearson chi‐square test for categorical variables; Mann Whitney U test for continuous variables.
3.1. The effect of antiplatelet drugs and statins on D‐dimer levels
We estimated the proportion of the variation (R² value) and change in plasma D‐dimer levels explained by age, sex, current smoking, use of antiplatelet drugs, use of statins, history of VTE in different linear regression models (Table 2). The change in plasma levels of D‐dimer was explained for 18.3% by age, sex, use of antiplatelet drugs and use of statins. Notably, age explained most of the proportion of variation (17.8%) and current smoking added a significant 0.8% of the explained variance to this model.
Table 2.
Effect on D‐dimers according to the different linear regression models
| Independent factors used in linear regression model | Proportion of variance explained (%)a | Adjusted % change in D‐dimer in users for statins (95% CI)b | Adjusted % change in D‐dimer for antiplatelet drug users (95% CI)b |
|---|---|---|---|
| Age | 17.8 | Not applicable | Not applicable |
| Age and use of statins | 18.2 | −15% (−28%; −1.4%)c | Not applicable |
| Age, sex, use of statins | 18.2 | −15% (−28%; −0.6%)c | Not applicable |
| Age, sex, use of lipophilic statins | 18.6 | −12.5% (−27%; 5%) | Not applicable |
| Age, sex, use of statins, use of antiplatelet drugs | 18.3 | −12% (−27%; 7.5%) | −7.6% (−25%;13%) |
| Age, sex, use of statins, use of antiplatelet drugs, current smoker | 19.1 | −11% (−27%; 8.0%) | −7.6% (−25%;13%) |
| Age, sex, use of statins, use of antiplatelet drugs, current smoker, history of VTE | 19.3 | −11% (−27%; 7.6%) | −7.0% (−24%;14%) |
R² value.
adjusted change in D‐dimer values calculated by exponentiation of the correlation coefficient and their corresponding 95% confidence intervals (95% CI).
significant P‐value < .05.
When adjusted for age, sex and use of statins, use of antiplatelet drugs resulted in a decrease of 7.6% (95% CI, −25% to 13%). In this similar regression model, use of statins resulted in a 12% (95% CI, −27% to 7.5%) decrease of D‐dimer values. Both these correlations were not significant. Yet, when only adjusted for age and sex, use of statins was significantly correlated and was associated with a reduction of D‐dimer values by 15% (95% CI, −28% to −0.6%). A subgroup regression analysis with inclusion of only lipophilic statin users and nonstatin users and adjustment for age and sex did not result in a significant correlation (12.5% decrease, 95% CI, −27% to 5%).
3.2. Model for reclassifying statin users within the YEARS algorithm
As antiplatelet drugs did not show any significant effect on D‐dimers, we only calculated adjusted D‐dimer cut‐offs for statin users. This calculation was based on the results of the linear regression model containing age, sex and use of statins. Adhering to this model, we adjusted the D‐dimer cut‐off to be 15% lower for patients using statins. This resulted in an adjusted cut‐off determined at 850 ng/mL instead of 1000 ng/mL for patients having no YEARS criteria. For patients having 1 or more YEARS criteria, the adjusted cut‐off was determined at 425 ng/mL instead of 500 ng/mL.
3.3. Performance of YEARS algorithm after reclassification of statin users
In the original YEARS algorithm, we found that 56 statin users (35.9%) were managed without CTPA. None of these patients was lost to follow‐up, no VTE or VTE‐related death was detected. When we tested the algorithm using the adjusted cut‐offs, thus incorporating the effects of statins on D‐dimer levels, we found that 12 patients were reclassified to the group needing a CTPA to rule out PE. In these patients, we did not detect any additional diagnostic failures during follow‐up, in 1 patient, CTPA was conducted for other reasons, showing no PE (Table 3). After reclassification of these 12 patients, specificity decreased from 0.42‐0.33, while sensitivity did not change.
Table 3.
Characteristics of statin users reclassified according to lower adjusted D‐dimer level cut‐offs
| Patient | Sex (M/F) | Age | YEARS criteria (0‐3) | D‐dimer (ng/mL) | Use of antiplatelet drugs (yes/no) | Diagnostic failuresa (yes/no) | Death during follow‐up or lost to follow‐up (yes/no) |
|---|---|---|---|---|---|---|---|
| 1 | M | 79 | 1 | 442 | Yes | No | No |
| 2 | F | 70 | 1 | 450 | No | No | No |
| 3 | M | 76 | 1 | 468 | Yes | No | No |
| 4 | M | 62 | 1 | 470 | Yes | No | No |
| 5 | M | 72 | 1 | 480 | No | No | No |
| 6 | M | 62 | 1 | 492 | No | No | No |
| 7 | M | 67 | 0 | 860 | Yes | No | No |
| 8 | F | 54 | 0 | 890 | No | No | No |
| 9 | M | 77 | 0 | 910 | Yes | No | No |
| 10 | F | 74 | 0 | 913 | No | No | No |
| 11 | M | 85 | 0 | 930 | Yes | No | No |
| 12 | M | 75 | 0 | 990 | Yes | Nob | No |
Reclassification of statin users having no YEARS and D‐dimer ≥ 850 and <1000 ng/mL or statin users having YEARS ≥ 1 and D‐dimer ≥ 425 and <500 ng/mL.
Diagnostic failures are defined by venous thromboembolism during follow‐up, VTE‐related death during follow‐up or lost to follow‐up.
CTPA (computed‐tomography pulmonary‐angiography) conducted during follow‐up showing no pulmonary embolism.
4. DISCUSSION
Our study results showed that in our population of patients suspected of having a PE, statins decreased D‐dimer levels but antiplatelet drugs did not. Adjusting D‐dimer cut‐offs for statin use did, however, not result in a safer diagnostic strategy. Based on our study, there is no need for adjusting D‐dimer cut‐off values for statin users. Moreover, age explained the largest proportion of the D‐dimer variance, underlining that age has a higher impact on D‐dimer levels than antiplatelet drugs and statins.
Nowadays, diagnostic algorithms for detecting PE are sensitive, but overall 3‐month diagnostic failure rate is still not reduced to zero.21, 22, 23 This is, to our best knowledge, the first study to test the effect of statins and antiplatelet drugs on the sensitivity of D‐dimer tests for the diagnosis of acute VTE. The reported effect of statins on D‐dimer levels differs between previous studies. Both the results of a review and a meta‐analysis suggested a decrease in plasma D‐dimer levels with the use of statins.13, 15 One small intervention study in patients with hypercholesterolaemia for example did not show a significant change in D‐dimer levels after start of treatment with statins whereas another study including type 2 diabetes patients found a significant reduction of approximately 8% in D‐dimer levels.18, 24 The studies included in this review and meta‐analysis concerned different patient populations, not taking into account concurrent antiplatelet therapy.25, 26 This is dissimilar to our study, in which we analysed the effect of antiplatelet drugs and statins separately. Our study results are best compared with those of a study by Adams et al14 comparing haemostatic factor levels between statin users and nonusers. In a cohort of 6814 healthy men and women age 45‐84 years, participants using statins had a 9% lower D‐dimer level after adjusting for age, race/ethnicity, education, income, hormone replacement therapy and major cardiovascular risk factors. The main difference with our study is that this was a cohort study in a healthy population, whereas in our study, D‐dimers levels were determined in the clinical setting of suspected PE. Another difference is that we could not adjust for all cardiovascular risk factors (including BMI and diabetes status), race/ethnicity, education, income and hormone replacement therapy, as these data were not fully collected at baseline. Notably, in our subgroup analysis with inclusion of only lipophilic statin users and nonusers of statins, we could not detect a significant correlation with D‐dimer levels, however, we might have been underpowered.
The evidence for the absence of effect of antiplatelet drugs on D‐dimers is compellingly illustrated in 2 studies in healthy male volunteers using acetylsalicylic acid.16, 27 One of these studies was a randomized placebo controlled trial in 30 healthy volunteers. Also, Kamath et al17 showed that D‐dimer levels in patients with atrial fibrillation treated with aspirin were not lower than in those given no treatment. A study in patients on peritoneal dialysis likewise found no effect on D‐dimer levels in those using low‐dose aspirin for 8 weeks.28
A methodological strength of this posthoc analysis in the YEARS population is that we studied a real‐world cohort of patients with a near complete follow‐up. Also, using the different linear regression models, the change in proportion of the variation explained by each variable and in particular statins could be extracted. Having analysed this population in daily practice using a clinical approach, we expect that results can be easily interpreted and translated to clinical care.
Limitations of our study include that this reports on a posthoc finding of a large outcome study, in which extensive data on baseline medication use were only available for two of the participating centres. For this reason, the secondary endpoints of our study are likely underpowered and we cannot rule out a relevant effect of statin use in clinical practice with sufficient certainty. Nevertheless, our study offers currently the best available evidence evaluating the diagnostic accuracy of a clinical diagnostic algorithm on PE in statin users explicitly. Also, we expect that a separate prospective study designed and powered to address these endpoints specifically will probably not be performed in future because of lack of financial incentives. Another limitation of our study is that CTPA results of the reclassified statin users were not available and the change in D‐dimer in statin users was used for modification of the D‐dimer cut‐off in the same population. Also, the adjusted cut‐offs were calculated posthoc and management decisions were based on the original cut‐offs. Therefore, extrapolation of our findings to other populations or healthcare settings warrants caution. Further, we used different quantitative D‐dimer assays and were unable to stratify the results by assay, as patient‐level information on the assay used was not available. However, all were contemporary, well‐validated high‐sensitive quantitative D‐dimer assays with similar fixed cut‐off levels.29
Based on our results, we conclude that use of statins is associated with a 15% decrease in D‐dimer levels. In our cohort, this was not associated with an increase in false‐negative test results, compared to nonusers. Nevertheless, further validation in a larger clinical cohort would be needed, however, we consider it unlikely that a prospective study designed and powered to address all endpoints separately will be performed in future.
CONFLICT OF INTEREST
None of the authors reports a conflict of interests with regard to this manuscript.
AUTHORS' CONTRIBUTIONS
SS and MJHAK designed the study. S.S, TvdH, JSB, LMvdP, FAK and MVH collected data and managed the study with support and input from all other authors. S.S analysed the data, which were interpreted by all other authors. S.S, TvG, JV and MJHAK wrote the first draft of the manuscript, which was reviewed, modified and approved by all other authors.
ACKNOWLEDGEMENTS
Suzanne Schol‐Gelok received a grant support of the Dutch Society for Clinical Pharmacology & Biopharmacy.
Schol‐Gelok S, van der Hulle T, Biedermann JS, et al. Clinical effects of antiplatelet drugs and statins on D‐dimer levels. Eur J Clin Invest. 2018;48:e12944 10.1111/eci.12944
REFERENCES
- 1. Konstantinides SV, Torbicki A, Agnelli G, et al. 2014 ESC guidelines on the diagnosis and management of acute pulmonary embolism. Eur Heart J. 2015;36:2642. [DOI] [PubMed] [Google Scholar]
- 2. Raja AS, Greenberg JO, Qaseem A, et al. Evaluation of patients with suspected acute pulmonary embolism: best practice advice from the clinical guidelines committee of the american college of physicians. Ann Intern Med. 2015;163:701‐711. [DOI] [PubMed] [Google Scholar]
- 3. van der Hulle T, Cheung WY, Kooij S, et al. Simplified diagnostic management of suspected pulmonary embolism (the YEARS study): a prospective, multicentre, cohort study. Lancet. 2017;390:289‐297. [DOI] [PubMed] [Google Scholar]
- 4. Adam SS, Key NS, Greenberg CS. D‐dimer antigen: current concepts and future prospects. Blood. 2009;113:2878‐2887. [DOI] [PubMed] [Google Scholar]
- 5. Douma RA, van Sluis GL, Kamphuisen PW, et al. Clinical decision rule and D‐dimer have lower clinical utility to exclude pulmonary embolism in cancer patients. Explanations and potential ameliorations. Thromb Haemost. 2010;104:831‐836. [DOI] [PubMed] [Google Scholar]
- 6. Harb TS, Zareba W, Moss AJ, et al. Association between inflammatory markers, hemostatic, and lipid factors in postinfarction patients. Am J Cardiol. 2003;91:1120‐1123. [DOI] [PubMed] [Google Scholar]
- 7. Chabloz P, Reber G, Boehlen F, Hohlfeld P, de Moerloose P. TAFI antigen and D‐dimer levels during normal pregnancy and at delivery. Br J Haematol. 2001;115:150‐152. [DOI] [PubMed] [Google Scholar]
- 8. Crop MJ, Siemes C, Berendes P, van der Straaten F, Willemsen S, Levin MD. Influence of C‐reactive protein levels and age on the value of D‐dimer in diagnosing pulmonary embolism. Eur J Haematol. 2014;92:147‐155. [DOI] [PubMed] [Google Scholar]
- 9. Szczeklik A, Krzanowski M, Gora P, Radwan J. Antiplatelet drugs and generation of thrombin in clotting blood. Blood. 1992;80:2006‐2011. [PubMed] [Google Scholar]
- 10. Owens AP 3rd, Mackman N. The antithrombotic effects of statins. Annu Rev Med. 2014;65:433‐445. [DOI] [PubMed] [Google Scholar]
- 11. Violi F, Calvieri C, Ferro D, Pignatelli P. Statins as antithrombotic drugs. Circulation. 2013;127:251‐257. [DOI] [PubMed] [Google Scholar]
- 12. Oesterle A, Laufs U, Liao JK. Pleiotropic effects of statins on the cardiovascular system. Circ Res. 2017;120:229‐243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Sahebkar A, Serban C, Mikhailidis DP, et al. Association between statin use and plasma d‐dimer levels: a systematic review and meta‐analysis of randomised controlled trials. Thromb Haemost. 2015;114:546‐557. [DOI] [PubMed] [Google Scholar]
- 14. Adams NB, Lutsey PL, Folsom AR, et al. Statin therapy and levels of hemostatic factors in a healthy population: the Multi‐Ethnic study of atherosclerosis. J Thromb Haemost. 2013;11:1078‐1084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Squizzato A, Romualdi E, Ageno W. Why should statins prevent venous thromboembolism? A systematic literature search and a call for action. J Thromb Haemost. 2006;4:1925‐1927. [DOI] [PubMed] [Google Scholar]
- 16. Derhaschnig U, Schweeger‐Exeli I, Marsik C, Cardona F, Minuz P, Jilma B. Effects of aspirin and NO‐aspirin (NCX 4016) on platelet function and coagulation in human endotoxemia. Platelets. 2010;21:320‐328. [DOI] [PubMed] [Google Scholar]
- 17. Kamath S, Blann AD, Chin BSP, et al. A study of platelet activation in atrial fibrillation and the effects of antithrombotic therapy. Eur Heart J. 2002;23:1788‐1795. [DOI] [PubMed] [Google Scholar]
- 18. Van De Ree MA, De Maat MP, Kluft C, Meinders AE, Princen HM, Huisman MV. Decrease of hemostatic cardiovascular risk factors by aggressive vs. conventional atorvastatin treatment in patients with Type 2 diabetes mellitus. J Thromb Haemost. 2003;1:1753‐1757. [DOI] [PubMed] [Google Scholar]
- 19. Wada H, Mori Y, Kaneko T, et al. Hypercoagulable state in patients with hypercholesterolemia: effects of pravastatin. Clin Ther. 1992;14:829‐834. [PubMed] [Google Scholar]
- 20. Simera I, Moher D, Hoey J, Schulz KF, Altman DG. A catalogue of reporting guidelines for health research. Eur J Clin Invest. 2010;40:35‐53. [DOI] [PubMed] [Google Scholar]
- 21. van Belle A, Buller HR, Huisman MV, et al. Effectiveness of managing suspected pulmonary embolism using an algorithm combining clinical probability, D‐dimer testing, and computed tomography. JAMA. 2006;295:172‐179. [DOI] [PubMed] [Google Scholar]
- 22. Righini M, Van Es J, Den Exter PL, et al. Age‐adjusted D‐dimer cutoff levels to rule out pulmonary embolism: the ADJUST‐PE study. JAMA. 2014;311:1117‐1124. [DOI] [PubMed] [Google Scholar]
- 23. Douma RA, Mos IC, Erkens PM, et al. Performance of 4 clinical decision rules in the diagnostic management of acute pulmonary embolism: a prospective cohort study. Ann Intern Med. 2011;154:709‐718. [DOI] [PubMed] [Google Scholar]
- 24. Bolaman Z, Kadikoylu G, Özgel N, Yenisey C. Effects of atorvastatin on coagulation parameters and homocysteine in patients with primary hypercholesterolemia. J Natl Med Assoc. 2006;98:1273‐1277. [PMC free article] [PubMed] [Google Scholar]
- 25. Dangas G, Badimon JJ, Smith DA, et al. Pravastatin therapy in hyperlipidemia: effects on thrombus formation and the systemic hemostatic profile. J Am Coll Cardiol. 1999;33:1294‐1304. [DOI] [PubMed] [Google Scholar]
- 26. Walter T, Szabo S, Suselbeck T, et al. Effect of atorvastatin on haemostasis, fibrinolysis and inflammation in normocholesterolaemic patients with coronary artery disease: a post hoc analysis of data from a prospective, randomized, double‐blind study. Clin Drug Investig. 2010;30:453‐460. [DOI] [PubMed] [Google Scholar]
- 27. Kim KM, Kim H, Chi HS, Park JS, Kim SB. Comparison of antiplatelet potency of sarpogrelate, aspirin, and beraprost in healthy volunteers according to in‐vitro closure time. Blood Coagul Fibrinolysis. 2010;21:262‐265. [DOI] [PubMed] [Google Scholar]
- 28. Kim SB, Lee SK, Min WK, Chi HS, Park JS. Lack of effects of low‐dose aspirin on high sensitivity C‐reactive protein, hemostatic factors, and troponin T in CAPD patients. Peritoneal Dial Int. 2002;22:721‐723. [PubMed] [Google Scholar]
- 29. Oude Elferink RF, Loot AE, Van De Klashorst CG, Hulsebos‐Huygen M, Piersma‐Wichers M, Oudega R. Clinical evaluation of eight different D‐dimer tests for the exclusion of deep venous thrombosis in primary care patients. Scand J Clin Lab Invest. 2015;75:230‐238. [DOI] [PubMed] [Google Scholar]


