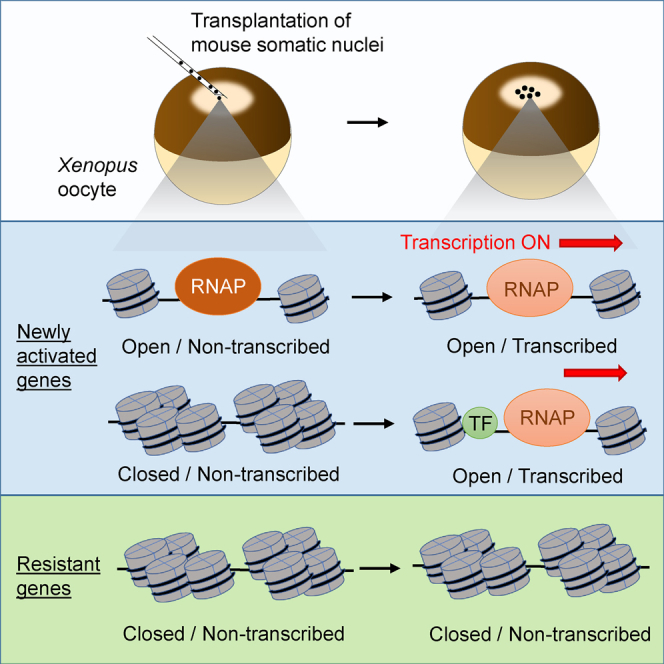
Summary
Oocytes have a remarkable ability to reactivate silenced genes in somatic cells. However, it is not clear how the chromatin architecture of somatic cells affects this transcriptional reprogramming. Here, we investigated the relationship between the chromatin opening and transcriptional activation. We reveal changes in chromatin accessibility and their relevance to transcriptional reprogramming after transplantation of somatic nuclei into Xenopus oocytes. Genes that are silenced, but have pre-existing open transcription start sites in donor cells, are prone to be activated after nuclear transfer, suggesting that the chromatin signature of somatic nuclei influences transcriptional reprogramming. There are also activated genes associated with new open chromatin sites, and transcription factors in oocytes play an important role in transcriptional reprogramming from such genes. Finally, we show that genes resistant to reprogramming are associated with closed chromatin configurations. We conclude that chromatin accessibility is a central factor for successful transcriptional reprogramming in oocytes.
Keywords: open chromatin, transcriptional activation, reprogramming, nuclear transfer
Graphical Abstract
Highlights
-
•
ATAC-seq reveals chromatin accessibility changes during reprogramming in oocytes
-
•
Genes with open promoters are preferentially activated during reprogramming
-
•
Transcription factors play a role in transcriptional reprogramming in oocytes
-
•
Closed chromatin is associated with reprogramming-resistant genes
Miyamoto et al. show genome-wide changes in chromatin accessibility during transcriptional reprogramming in oocytes using the frog nuclear transfer system. They demonstrate that donor cell chromatin states affect transcriptional reprogramming and changes in open chromatin during reprogramming are associated with specific transcription factors.
Introduction
Transcriptional activation is pivotal for cell fate changes and is modulated by the access of chromatin- and transcription-related factors to gene regulatory regions such as promoters and enhancers. Chromatin accessibility at gene regulatory regions affects transcriptional outcome. Pronounced nucleosome-depleted regions are found around transcription start sites (TSSs) of active genes (Teif et al., 2012). In the course of mouse embryonic development, open chromatin regions dynamically change, which is accompanied by altered transcriptional activities of the associated genes (Lu et al., 2016, Wu et al., 2016). Studying chromatin accessibility dynamics at gene regulatory regions during transcriptional activation provides insight into cell fate changes.
In order to examine chromatin accessibility, DNase sequencing (DNase-seq) has been widely used, which identifies genomic regions that can be cut by the DNaseI enzyme, known as DNaseI-hypersensitive sites (Boyle et al., 2008, Stalder et al., 1980, Thurman et al., 2012). Recently, the Assay for Transposase-Accessible Chromatin Sequencing (ATAC-seq) has been developed to examine open chromatin regions by taking advantage of the Tn5 transposon’s ability to preferentially insert in open chromatin regions (Buenrostro et al., 2013). ATAC-seq is a powerful tool to map open chromatin and nucleosome positions in samples of 500–50,000 cells, producing results comparable with conventional DNase-seq but requiring orders of magnitude fewer cells (Buenrostro et al., 2013). ATAC-seq methods have also been used for single cells (Buenrostro et al., 2015, Cusanovich et al., 2015).
Using the genome-wide approaches, dynamic changes in chromatin accessibility have been investigated when transcription patterns of somatic cells are reprogrammed in induced pluripotent stem (iPS) cells. In the course of reprogramming toward iPS cells, nucleosome occupancy and open chromatin regions are changed in regulatory regions, especially at the binding sites for reprogramming transcription factors (TFs) (Li et al., 2017, West et al., 2014). The binding of reprogramming TFs to chromatin is inhibited by a closed chromatin configuration marked by histone H3 lysine 9 trimethylation (H3K9me3) (Soufi et al., 2012). Furthermore, enhancing chromatin accessibility during reprogramming by knocking down the histone chaperone CAF-1 accelerates binding of TFs and the transcriptional activation of pluripotency genes (Cheloufi et al., 2015). In contrast to iPS cells, chromatin accessibility dynamics during nuclear reprogramming in oocytes have not been well elucidated on a genome-wide scale. Nuclear transfer (NT) of somatic cells to oocytes allows efficient reprogramming in gene expression, which is induced by a different mechanism from iPS cells (Jullien et al., 2011). However, it is unclear to what extent oocytes can reprogram chromatin accessibility of somatic cells.
In this study, we examined open chromatin regions using ATAC-seq before and after reprogramming of mouse somatic nuclei in Xenopus laevis oocytes (Jullien et al., 2014, Miyamoto et al., 2013). This NT system enables DNA replication- and cell division-independent reprogramming of somatic nuclei so that we can assess the direct impact of oocyte factors on chromatin accessibility. We find that chromatin states of donor cells profoundly affect transcriptional reprogramming, although oocytes have an ability to open up gene regulatory regions. We show that chromatin accessibility is a key factor influencing transcriptional reprogramming in oocytes.
Results and Discussion
Optimization and Evaluation of ATAC-Seq for Analyzing Cells Transplanted into Oocytes
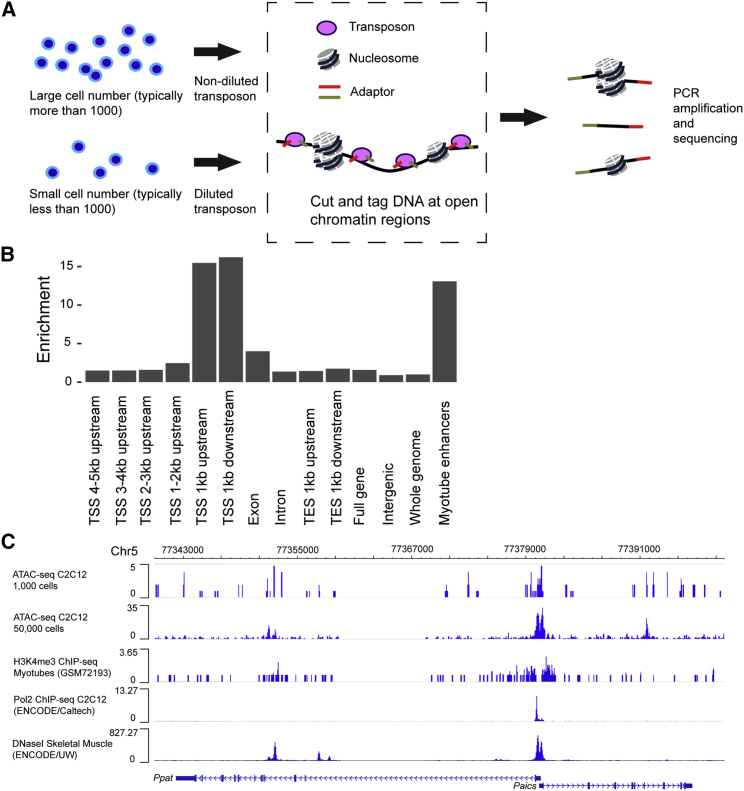
Somatic cell nuclei transferred into oocytes have different characteristics from conventional cultured cells, in that only a small number of cells can be prepared, and those cells are difficult to permeabilize. We therefore optimized ATAC-seq protocols for our study. As reported previously (Buenrostro et al., 2013), the concentration of transposon for cutting and tagging open chromatin regions was key to a successful assay. When the cell number was small (less than 1,000 cells), dilution of the transposon prevented over-digestion of template chromatin (Figures 1A and S1A). The correct transposon concentration allowed for successful production of a DNA library with periodic nucleosome peaks even from a single cell (Figure S1B). In addition, Triton X-100 was needed to permeabilize reprogrammed cells, which contain inhibitory Xenopus oocyte materials only after NT.
Figure 1.
ATAC-Seq Enables the Identification of Open Chromatin Regions
(A) The modified ATAC-seq protocol for our experiments.
(B) The genomic distribution of ATAC-seq peaks in C2C12 mouse myoblasts, representing open chromatin. The y axis represents the enrichment of peaks in each type of genomic region relative to the whole genome. Two independently prepared ATAC-seq libraries were used for the analysis.
(C) A track image of ATAC-seq from 1,000 and 50,000 cells, chromatin immunoprecipitation sequencing (ChIP-seq) of H3K4me3 (GSM72193) and RNA polymerase II (GSM915176), and DNase-seq (GSM1014189) at the Ppat and Paics genes in mouse muscle cells. Open chromatin at the TSS is adjacent to H3K4me3 marks.
After incorporating these modifications, we performed ATAC-seq using mouse C2C12 myoblasts from 50,000, 1,000, 100, 10, and 1 cell. When cell numbers were small (less than 100 cells), many reads were derived from mitochondria and the produced libraries showed characteristics of those with a low quality, such as duplicated reads and low library complexity. Hierarchical clustering of the mapped ATAC-seq reads around TSSs showed that the 1,000-cell and 50,000-cell samples clustered together while those created from 100, 10, and 1 cell did not cluster with the 1,000-cell and 50,000-cell samples (Figure S1C). For making use of sequencing data from the small cell numbers, multiplexing of a large number of libraries or referring to pre-existing chromatin accessibility maps would be a good way forward (Buenrostro et al., 2015, Cusanovich et al., 2015). These results suggest that ATAC-seq reads that cover the whole genome can only be obtained from libraries prepared from 1,000 or more cells, at least using our reported method. Therefore, we decided to prepare ATAC-seq libraries using more than 1,000 cells in subsequent experiments.
Genomic locations of ATAC-seq peaks, representing open chromatin sites, were examined in C2C12 cells, and 52,697 unique sites were found. Open chromatin regions were overrepresented within 1 kb of TSSs by 15.9-fold relative to the whole genome (p < 1E−6) and by 13.1-fold in myotube enhancers (p < 1E−6) (Figure 1B). Further, 40.0% of all TSSs and 40.3% of all myotube enhancers (p < 1E−6) contained ATAC-seq peaks. ATAC-seq peaks were 10.9-fold enriched in regions marked with histone H3 lysine 4 trimethylation (H3K4me3) on a genome-wide scale (p < 1E−6; Figure 1C, track image), in agreement with the previously noted association of H3K4me3 with open and active promoter elements (Guenther et al., 2007, Heintzman et al., 2007). These data verify that our modified ATAC-seq protocol captures characteristic open chromatin features.
ATAC-Seq Reveals Changes in Chromatin Accessibility after NT of Somatic Cells to Xenopus Oocytes
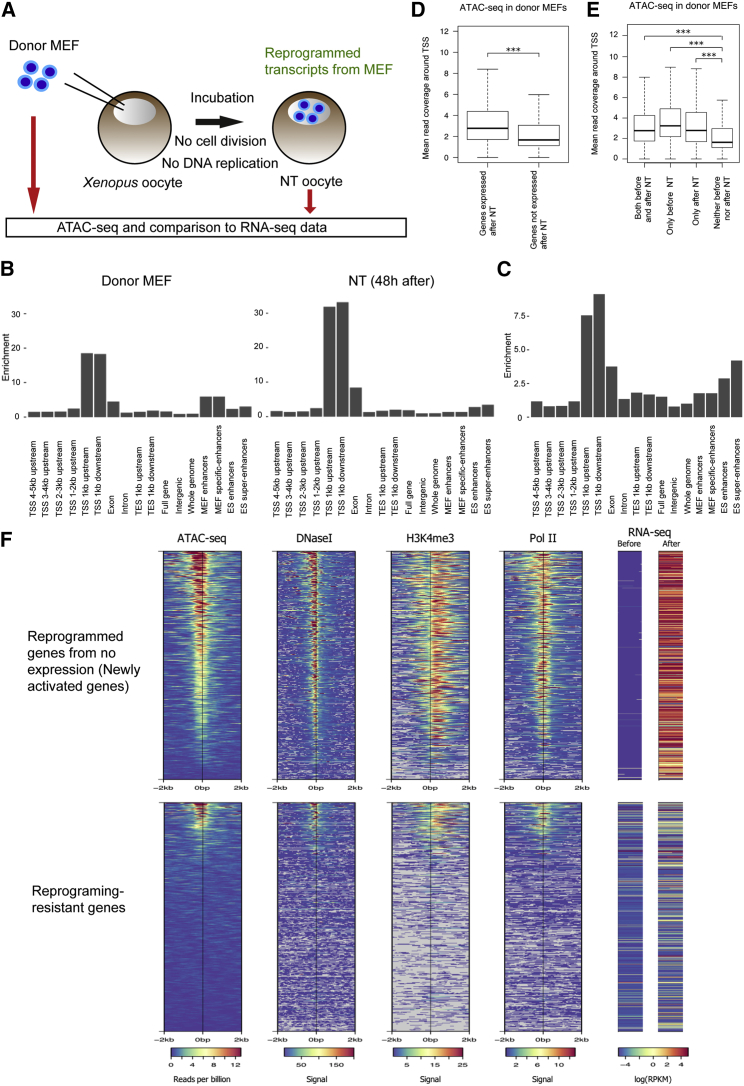
We then examined chromatin accessibility dynamics before and after transcriptional reprogramming of somatic cells in oocytes by using the modified ATAC-seq protocol. We took advantage of the direct transcriptional reprogramming system in Xenopus oocytes, in which hundreds of mouse somatic nuclei transplanted into the germinal vesicle (GV), a giant nucleus of a Xenopus oocyte, undergo extensive transcriptional reprogramming toward an oocyte-like state within 2 days, without cell divisions and DNA replication (Figure 2A) (Jullien et al., 2014). Therefore, any changes in chromatin accessibility observed in our reprograming system are accomplished in a replication-independent manner. We performed ATAC-seq on donor mouse embryonic fibroblasts (MEFs) and on MEFs 48 hr after NT, using 3,000 cells for each sample (Figure 2A). Our ATAC-seq reads of donor MEFs resembled reads from published DNase-seq of mouse 3T3 embryonic fibroblast cells (GSM1003831, Spearman correlation with our reads = 0.84) and mouse headless embryos at day 11.5 (GSM1014172, Spearman correlation with our reads = 0.85; Figure S2A). Examination of the genomic distribution of peaks showed enrichment around TSSs both before and after NT (Figure 2B). Open chromatin peaks were also enriched at enhancers for MEFs before NT, but this enrichment was not observed after NT (Figure 2B), suggesting the closing of open chromatin at MEF-specific enhancers after NT. The relative abundance of peaks for embryonic stem cell (ESC) enhancers did not increase much after NT, but the closing of enhancers was not observed, unlike MEF enhancers (Figure 2B, right two bars). These results suggest that MEFs, which have undergone NT, may use a different set of enhancers from those before NT to regulate gene transcription.
Figure 2.
Genes with Open TSSs Are Preferentially Reprogrammed upon NT to Xenopus laevis Oocytes
(A) MEFs are transplanted into the nuclei of Xenopus oocytes, reprogramming their transcription. MEFs before NT and reprogrammed MEFs were used for ATAC-seq. Two biologically independent NT experiments were performed for the subsequent analyses (10 NT oocytes, equivalent to 3,000 cells, were pooled in each experiment).
(B) The genomic distribution of ATAC-seq peaks representing open chromatin before and after NT. The y axis represents the enrichment of peaks in each type of genomic region relative to the whole genome.
(C) The genomic distribution of newly appeared ATAC-seq peaks after NT.
(D) ATAC-seq reads in donor MEFs were compared around TSSs. Genes were divided into two categories: expressed in NT oocytes and not expressed in NT oocytes. The y axis represents the mean read coverage in a 1-kb window centered on the TSS.
(E) ATAC-seq reads around TSSs in donor MEFs were compared among different gene categories: genes expressed before and after NT, those expressed only before NT, those expressed only after NT, and those expressed at neither time point.
(F) Representation of signal associated with open chromatin (ATAC-seq, DNase-seq, H3K4me3 ChIP-seq, and Pol II ChIP-seq) at TSSs of MEF genes reprogrammed in Xenopus oocytes. RNA-seq results (Jullien et al., 2014, Jullien et al., 2017) are shown at the right panel. Accession numbers for the DNase-seq, H3K4me3, and Pol II data are GSM1014172, GSM769029, and GSM918761, respectively.
∗∗∗p < 1E−6 by the Mann-Whitney U test.
We next investigated genomic regions newly opened after NT. We found that 3,146 peaks appeared only after NT; those peaks present after NT were at least 5,000 bp away from a peak in MEF before NT. Such newly appeared open chromatin regions were enriched at gene regulatory regions including TSSs and embryonic stem (ES) super-enhancers (Figure 2C). The mean distance of these newly opened chromatin regions from a TSS is 61,049 bp, compared with 23,813 bp for all peaks after NT, suggesting that newly open regions are more likely to be distant from promoters. These results suggest that dynamic changes in chromatin accessibility are induced in a replication-independent manner during oocyte-mediated reprogramming.
Open Promoters of Silent Genes Are Permissive for Transcriptional Activation in Oocytes
Distribution of open chromatin peaks clearly indicates that accessible chromatin sites are mainly located at gene regulatory regions (Figures 2B and 2C). We then investigated the relationship between open chromatin at gene regulatory regions and transcriptional activation. We took advantage of our previous RNA-seq dataset in which transcriptome of donor MEFs before and after NT was revealed (Jullien et al., 2014). Mapped ATAC-seq reads were plotted around TSSs and, as expected, genes expressed after NT showed more open chromatin than non-expressed genes in these samples (Figure S2B). Intriguingly, genes expressed after NT exhibited more open chromatin than genes not expressed after NT at TSSs in the donor cells as well (Figure 2D), suggesting that pre-existing open chromatin might affect subsequent transcriptional reprogramming. To further pursue this, we examined genes newly activated after NT. These genes showed more open chromatin at TSSs than non-expressed genes in donor cells and in NT oocytes (only after NT versus neither before nor after NT; Figures 2E and S2C). Even more strikingly, a large proportion of genes activated after NT contained open chromatin already in donor cells and were also associated with H3K4me3 and unphosphorylated RNA polymerase II (Figure 2F, upper panels). This is in contrast to genes that our previous study identified as resistant to transcriptional activation in MEFs during oocyte-mediated reprogramming (Figure 2F, lower panels; reprogramming-resistant genes are discussed below) (Jullien et al., 2017). In conclusion, genes marked at their TSSs by open chromatin and other priming-associated factors such as the loading of poised RNA polymerase II (Adelman and Lis, 2012) and H3K4me3 (Voigt et al., 2013) are prone to transcriptional activation during reprogramming in oocytes. These results also imply that pre-existing open chromatin states in donor cells affect transcriptional reprogramming, further extending our previous finding that somatic cell genes abnormally maintain their expression in Xenopus NT embryos as somatic memory genes (Hörmanseder et al., 2017).
Transcriptional Reprogramming Is Not Solely Explained by Pre-existing Open Chromatin States at Promoters
We asked whether pre-existing open chromatin is enough to induce transcriptional reprogramming. We compared open chromatin states of downregulated genes and genes activated after reprogramming. Genes downregulated after NT (genes that were only expressed in MEFs) had a higher level of open chromatin than non-expressed genes in donor cells, as expected, but strong open chromatin states were unexpectedly maintained in downregulated genes even after NT (Figure 2E; Figure S2C, only before NT). On further inspection, we observed that, while downregulated genes retained open chromatin after NT at the global level, the open regions shifted their locations slightly (Figure S2D). In fact, some motifs were more enriched in ATAC-seq peaks detected in MEFs near these downregulated genes than in peaks detected in MEFs after NT around the same genes, and vice versa (Figures S2E and S2F). As expected, the TFs with motifs more enriched in MEF peaks tended to be more expressed in MEFs and not expressed in Xenopus oocytes, while the TFs with motifs more enriched in post-NT peaks tended to be more expressed in MEFs after NT and/or in Xenopus oocytes (Figures S2E and S2F). These results suggest that oocytes utilize different TFs from somatic cells to maintain the open chromatin near genes expressed only before NT. These regions may stay open after NT as a secondary effect of the oocyte transcriptional regulators after NT, but without somatic factors, transcription from these genes is not efficiently performed even in the presence of open chromatin. This idea is in good accordance with our previous finding that the removal of somatic transcriptional machinery and the loading of the oocyte counterpart is observed after NT to oocytes (Jullien et al., 2014).
Oocyte-Mediated Reprogramming Involves Opening of Closed Chromatin
Although many genes are transcribed from pre-existing open TSSs (Figure 2F), some genes acquire open chromatin from the closed state during oocyte reprogramming. 3,146 ATAC-seq peaks were detected only in NT samples, but not in donor MEFs (Table S1), and 497 genes without an ATAC-seq peak in MEFs gained a peak after NT, such as Utf1 and Hoxc8, both of which have been shown to be reprogrammed after NT (Miyamoto et al., 2013) (Figure S3A; Table S2). We also found 1,245 ATAC-seq peaks after NT that do not overlap genes and are located at least 5 kb from a TSS or a peak before NT (Table S1). These newly formed regions of open chromatin may serve as enhancers that drive transcriptional reprogramming in this system, analogous to the enhancers that open during cellular differentiation and iPS reprogramming (Wang et al., 2011a, West et al., 2014). Newly opened regions were associated with genes related to fatty acid biosynthesis, regulation of stem cell maintenance, and protein targeting to membrane, as revealed by the Genomic Regions Enrichment of Annotations Tool (GREAT) (McLean et al., 2010) (Figure S3B). Furthermore, several motifs for binding of TFs were enriched in ATAC-seq peaks after NT relative to before NT, including GATA binding protein 3 (GATA3), retinoic acid receptor gamma (RARG), and RE1-silencing transcription factor (REST) (Figure S3C). Interestingly, REST has been shown to play a key role in NT-mediated nuclear reprogramming in porcine oocytes (Kong et al., 2016), supporting the validity of our approach.
Transcription Factors in Oocytes Are Involved in Transcriptional Reprogramming
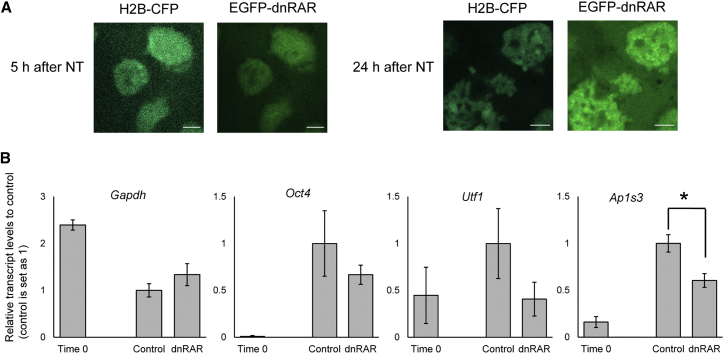
Because chromatin sites newly opened after NT are enriched with specific TF motifs (Figure S3C), we sought to test roles of TFs in oocytes for transcriptional reprogramming. Among the TFs identified, we focused on RAR because it showed one of the most significant hits and because RAR functions in Xenopus oocytes (Minucci et al., 1998). We first selected genes to test for further analyses. Pou5f1 (known as Oct4) and Utf1 have been shown to be regulated by RARs (Delacroix et al., 2010, Okazawa et al., 1991) and to be newly activated after NT to oocytes (Miyamoto et al., 2013). We also examined Ap1s3, which contains Retinoic Acid Response Element (RARE) at the TSS and is activated after NT. A RAR-α dominant-negative form (dnRAR) (Wang et al., 2011b) was overexpressed in NT oocytes. dnRAR was localized in transplanted MEF nuclei several hours after NT (Figure 3A). The overexpression of dnRAR in NT oocytes inhibited transcription from Oct4, Utf1, and Ap1s3 (1.5- to 2.5-fold decrease), whereas expression of Gapdh, a housekeeping gene, was not affected (Figure 3B). These results suggest that newly activated genes are indeed regulated by RAR. Therefore, TFs in oocytes impact transcriptional reprogramming, possibly through opening of inaccessible sites in somatic chromatin.
Figure 3.
RAR Influences Transcriptional Reprogramming in Oocytes
(A) NT oocytes overexpressed with EGFP-dnRAR and histone H2B-CFP were subjected to confocal microscopy. EGFP-dnRAR was accumulated in the injected nuclei. Scale bars indicate 5 μm.
(B) Expression of RA-regulated genes was downregulated after overexpression of EGFP-dnRAR in NT oocytes. n = 3 (time 0) or 4 (control and dnRAR). Time 0 represents NT oocytes just after NT. Error bars represent ± SEM. ∗p < 0.05 by the Student’s t test.
Closed Chromatin Is Associated with Reprogramming-Resistant Genes
We next asked whether an opening of somatic chromatin is efficiently carried out or not after NT to oocytes. Our previous study identified genes resistant to oocyte-mediated reprogramming in MEFs (Jullien et al., 2017). These genes are not activated after nuclear transplantation of MEFs to oocytes but are transcribed after transplantation of ESCs (Jullien et al., 2017). This suggests that although transcriptional activators for the MEF reprogramming-resistant genes are present in oocytes, some feature of MEFs is preventing successful activation. Interestingly, both resistant and activated genes were transcriptionally silent in donor MEFs, but the TSSs at resistant genes in MEFs were clearly more closed compared with successfully activated genes (Figure S3D). The opening of chromatin is a central factor for successful transcriptional reprogramming, but is inefficient as is evident from the presence of the MEF reprogramming-resistant genes, suggesting that the opening of closed chromatin is a barrier for reprogramming.
Chromatin Opening Facilitates Transcriptional Reprogramming
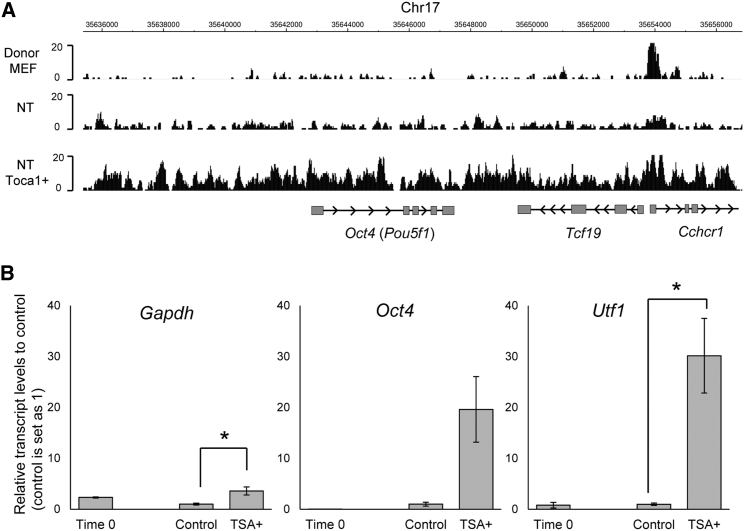
We finally tested whether accessible chromatin states enable efficient transcriptional reprogramming. We have previously reported that enhancing nuclear actin polymerization results in efficient transcriptional reprogramming (Miyamoto et al., 2011), and that nuclear actin polymerization plays a role in chromatin decondensation (Baarlink et al., 2017). Toca1/Fnbp1l, a factor that enhances actin polymerization and transcriptional reprogramming (Miyamoto et al., 2011), was overexpressed in NT oocytes and subjected to ATAC-seq. More open chromatin peaks were found in Toca1/Fnbp1l-overexpressed NT oocytes, compared with control NT oocytes (2.6-fold increase), and 2.1-fold more genes were associated with ATAC-seq peaks by Toca1/Fnbp1l overexpression. Toca1/Fnbp1l overexpression has been shown to enhance Oct4 activation (Miyamoto et al., 2011). Indeed, more ATAC-seq reads were found at the Oct4 gene locus in Toca1/Fnbp1l-overexpreessed NT oocytes than in control NT oocytes (Figure 4A), suggesting that enhanced transcriptional reprogramming is associated with the accessible chromatin state. On the other hand, genes whose transcription is not affected by Toca1/Fnbp1l overexpression (Miyamoto et al., 2011) also showed more ATAC-seq reads (Figure S4A), supporting our contention that open chromatin at TSSs is not sufficient to determine transcriptional activities. The genomic distribution of peaks found in Toca1/Fnbp1l-overexpressed NT oocytes showed enrichment around TSSs (Figure S4B), like normal NT oocytes (Figure 2B). Together, accelerated nuclear actin polymerization by Toca1/Fnbp1l overexpression seems to induce accessible chromatin globally, rather than locally, at specific loci.
Figure 4.
Efficient Transcriptional Reprogramming Is Associated with Increased Chromatin Accessibility
(A) ATAC-seq reads for donor MEFs, control NT oocytes, and NT oocytes overexpressed with Toca1/Fnbp1l around the Oct4 gene locus.
(B) TSA treatment enhances transcriptional reprogramming. NT oocytes incubated with or without 50 nM TSA for 24 hr were used for qRT-PCR. Time 0 represents NT oocytes just after NT. n = 3 (time 0) or 4 (control and dnRAR). Error bars represent ± SEM. ∗p < 0.05 by the Student’s t test.
We next treated NT oocytes with 50 nM Trichostatin A (TSA), a histone deacetylase inhibitor, which can induce the accessible chromatin state in somatic nuclei (Bui et al., 2010, Maalouf et al., 2009, Miyamoto et al., 2017). NT oocytes incubated with TSA increased Oct4 and Utf1 expression by 19.6- and 30.2-fold, respectively (Figure 4B). These results suggest that somatic chromatin is not fully opened to support transcriptional reprogramming, even after NT to oocytes, and reprogramming can be enhanced by modulating chromatin accessibility.
In summary, our study reveals genome-wide changes in chromatin accessibility in a DNA-replication-independent manner after NT of somatic cells to Xenopus oocytes. Transcriptional activation from the silenced genes in donor somatic cells can be accomplished from both pre-existing open chromatin and closed chromatin. In 56% of newly activated genes, pre-existing open chromatin peaks were found near TSSs before NT. These results indicate that primed genes are prone to transcriptional activation during oocyte-mediated reprogramming. Presumably, having open chromatin, H3K4me3, and poised RNA polymerase II (Pol II) at these genes helps ensure that these genes can be promptly and efficiently activated, compared with genes with closed chromatin. In contrast, only a few percentages of genes acquire obvious open chromatin sites after NT, implying that opening of closed chromatin is an inefficient process. Indeed, we found that a closed chromatin configuration at TSSs is a barrier to some genes successfully being activated during reprogramming. Chromatin opening related to transcriptional reprogramming is likely mediated by TFs such as RARs. Although oocytes possess many TFs and other machineries to relax chromatin (Jullien et al., 2011, Jullien et al., 2014), some experimental interventions can still boost chromatin opening and transcriptional reprogramming of somatic cells. These observations suggest that somatic nuclei acquire extremely stable chromatin signatures in the course of differentiation. Increased chromatin accessibility contributes to the destabilization of somatic chromatin features and hence successful reprogramming.
Experimental Procedures
Animals
Our work using mature Xenopus laevis females is covered under the UK Home Office Project License PPL 70/8591 or approved by the Animal Research Committee of Kindai University. Frog husbandry and all experiments were performed according to the relevant regulatory standards. For collecting oocytes, frogs were anesthetized by subcutaneous injection of 120 mg (in 400 μL) of Tricaine methanesulfonate (MS222). Subsequently, the frogs were slaughtered by exsanguination under anesthesia, followed by freezing for appropriate disposal.
ATAC-Seq
ATAC-seq was performed as described previously (Buenrostro et al., 2013) with some modifications. Cultured cells were harvested, washed with PBS, then transferred to cold lysis buffer (10 mM Tris-HCl [pH 7.4], 10 mM NaCl, 3 mM MgCl2, 0.1% IGEPAL, 0.5% Triton X-100). Triton X-100 was crucial for permeabilizing cells after NT. Cells in lysis buffer were collected by centrifugation and the cell pellet resuspended in the transposon reaction mix (25 μL 2× TD Buffer [FC-121-1030; Illumina], 2.5 μL Tn5 Transposase [FC-121-1030; Illumina], 22.5 μL of Nuclease Free H2O). A total of 50 μL of the transposon reaction was used for 50,000 cells, while 1,000 or fewer cells were incubated in 5 μL of the transposon reaction. Moreover, diluted transposon (up to 1/1,000) was used when cell numbers were 100, 10, and 1. The transposition reaction was performed at 37°C for 30 min. After the reaction, transposed DNA was purified using a Qiagen MinElute kit and used for subsequent PCR amplification. DNA from 1,000 or fewer cultured cells was directly subjected to PCR amplification without kit purification. Transposed DNA was amplified by two rounds of PCR using NEB Next High Fidelity Master Mix (M0541; New England Labs) and the Customized Nextera PCR primers in 50 μL (Buenrostro et al., 2013). The first PCR varied from 5 to 15 cycles depending on the starting cell numbers. The number of second PCR cycles was determined by qPCR using 10% of the total PCR in order to amplify the DNA in the exponential growth phase. The amplified DNA was purified using QIAGEN PCR Cleanup kit and finally resuspended in 20 μL of elution buffer. The quality of library was checked on polyacrylamide gels and quantified using the KAPA library quantification kit for Illumina sequencing platforms (KAPA Biosystems). ATAC-seq libraries were computationally processed as described in the Supplemental Experimental Procedures.
Statistical Analysis
The number of biological replicates is shown as n. In transcriptional assays by qRT-PCR, the statistical difference was calculated by the two-tailed Student’s t test. Error bars were represented as the SEM. The levels of significance were set as ∗p < 0.05. The statistical difference in mean ATAC-seq signal around groups of genes was calculated using the two-tailed Mann-Whitney U test. The statistical significance of the overlap between ATAC-seq peaks and other genomic datasets was calculated using the hypergeometric test. These statistical tests were conducted using the R software package (version 3.4). Further computational methods are described in the Supplemental Experimental Procedures.
Acknowledgments
We thank Drs. W.J. Greenleaf and H.Y. Chang for providing a detailed ATAC-seq protocol and Dr. P. Liu for the dnRAR construct. We also thank T. Kobayashi, Y. Tsukamoto, and C. Kimura for technical support. K.M. is supported by the Human Frontier Science Program (grant RGP0021/2016), JSPS KAKENHI (grants JP16H01321, JP16H01222, and JP17H05045), and Kindai University (research grant 17-II-3). The Gurdon laboratory is supported by grants from Wellcome Trust (101050/Z/13/Z) and the MRC (MR/K011022/1), and supported by the Gurdon Institute core grant from Cancer Research UK (C6946/A14492) and the Wellcome Trust (092096/Z/10/Z). K.T.N. was supported by grants from the NIH (5-R01-GM113708 and 5-U41-HG007000).
Author Contributions
K.M. conceived the experiments and wrote the manuscript. K.M., K.T.N., and J.J. designed the experiments. K.M., T.O., and J.B.G. carried out experiments. K.T.N., D.K., G.E.A., and C.R.B. performed bioinformatics analyses. K.T.N., J.J., and J.B.G. edited the manuscript. F.J.L., M.K., and J.B.G. supervised research.
Declaration of Interests
The authors declare no competing interests.
Published: July 10, 2018
Footnotes
Supplemental Information includes Supplemental Experimental Procedures, four figures, and two tables and can be found with this article online at https://doi.org/10.1016/j.celrep.2018.06.030.
Contributor Information
Kei Miyamoto, Email: kmiyamo@waka.kindai.ac.jp.
Manolis Kellis, Email: manoli@mit.edu.
John B. Gurdon, Email: j.gurdon@gurdon.cam.ac.uk.
Data and Software Availability
The accession number for the ATAC-seq data generated in this study and reported in this paper is GEO: GSE98776.
Supplemental Information
References
- Adelman K., Lis J.T. Promoter-proximal pausing of RNA polymerase II: emerging roles in metazoans. Nat. Rev. Genet. 2012;13:720–731. doi: 10.1038/nrg3293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baarlink C., Plessner M., Sherrard A., Morita K., Misu S., Virant D., Kleinschnitz E.M., Harniman R., Alibhai D., Baumeister S. A transient pool of nuclear F-actin at mitotic exit controls chromatin organization. Nat. Cell Biol. 2017;19:1389–1399. doi: 10.1038/ncb3641. [DOI] [PubMed] [Google Scholar]
- Boyle A.P., Davis S., Shulha H.P., Meltzer P., Margulies E.H., Weng Z., Furey T.S., Crawford G.E. High-resolution mapping and characterization of open chromatin across the genome. Cell. 2008;132:311–322. doi: 10.1016/j.cell.2007.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buenrostro J.D., Giresi P.G., Zaba L.C., Chang H.Y., Greenleaf W.J. Transposition of native chromatin for fast and sensitive epigenomic profiling of open chromatin, DNA-binding proteins and nucleosome position. Nat. Methods. 2013;10:1213–1218. doi: 10.1038/nmeth.2688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buenrostro J.D., Wu B., Litzenburger U.M., Ruff D., Gonzales M.L., Snyder M.P., Chang H.Y., Greenleaf W.J. Single-cell chromatin accessibility reveals principles of regulatory variation. Nature. 2015;523:486–490. doi: 10.1038/nature14590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bui H.T., Wakayama S., Kishigami S., Park K.K., Kim J.H., Thuan N.V., Wakayama T. Effect of trichostatin A on chromatin remodeling, histone modifications, DNA replication, and transcriptional activity in cloned mouse embryos. Biol. Reprod. 2010;83:454–463. doi: 10.1095/biolreprod.109.083337. [DOI] [PubMed] [Google Scholar]
- Cheloufi S., Elling U., Hopfgartner B., Jung Y.L., Murn J., Ninova M., Hubmann M., Badeaux A.I., Euong Ang C., Tenen D. The histone chaperone CAF-1 safeguards somatic cell identity. Nature. 2015;528:218–224. doi: 10.1038/nature15749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cusanovich D.A., Daza R., Adey A., Pliner H.A., Christiansen L., Gunderson K.L., Steemers F.J., Trapnell C., Shendure J. Multiplex single cell profiling of chromatin accessibility by combinatorial cellular indexing. Science. 2015;348:910–914. doi: 10.1126/science.aab1601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delacroix L., Moutier E., Altobelli G., Legras S., Poch O., Choukrallah M.A., Bertin I., Jost B., Davidson I. Cell-specific interaction of retinoic acid receptors with target genes in mouse embryonic fibroblasts and embryonic stem cells. Mol. Cell. Biol. 2010;30:231–244. doi: 10.1128/MCB.00756-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guenther M.G., Levine S.S., Boyer L.A., Jaenisch R., Young R.A. A chromatin landmark and transcription initiation at most promoters in human cells. Cell. 2007;130:77–88. doi: 10.1016/j.cell.2007.05.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heintzman N.D., Stuart R.K., Hon G., Fu Y., Ching C.W., Hawkins R.D., Barrera L.O., Van Calcar S., Qu C., Ching K.A. Distinct and predictive chromatin signatures of transcriptional promoters and enhancers in the human genome. Nat. Genet. 2007;39:311–318. doi: 10.1038/ng1966. [DOI] [PubMed] [Google Scholar]
- Hörmanseder E., Simeone A., Allen G.E., Bradshaw C.R., Figlmüller M., Gurdon J., Jullien J. H3K4 methylation-dependent memory of somatic cell identity inhibits reprogramming and development of nuclear transfer embryos. Cell Stem Cell. 2017;21:135–143.e6. doi: 10.1016/j.stem.2017.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jullien J., Pasque V., Halley-Stott R.P., Miyamoto K., Gurdon J.B. Mechanisms of nuclear reprogramming by eggs and oocytes: a deterministic process? Nat. Rev. Mol. Cell Biol. 2011;12:453–459. doi: 10.1038/nrm3140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jullien J., Miyamoto K., Pasque V., Allen G.E., Bradshaw C.R., Garrett N.J., Halley-Stott R.P., Kimura H., Ohsumi K., Gurdon J.B. Hierarchical molecular events driven by oocyte-specific factors lead to rapid and extensive reprogramming. Mol. Cell. 2014;55:524–536. doi: 10.1016/j.molcel.2014.06.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jullien J., Vodnala M., Pasque V., Oikawa M., Miyamoto K., Allen G., David S.A., Brochard V., Wang S., Bradshaw C. Gene resistance to transcriptional reprogramming following nuclear transfer is directly mediated by multiple chromatin-repressive pathways. Mol. Cell. 2017;65:873–884.e8. doi: 10.1016/j.molcel.2017.01.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kong Q.R., Xie B.T., Zhang H., Li J.Y., Huang T.Q., Wei R.Y., Liu Z.H. RE1-silencing transcription factor (REST) is required for nuclear reprogramming by inhibiting transforming growth factor β signaling pathway. J. Biol. Chem. 2016;291:27334–27342. doi: 10.1074/jbc.M116.743849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li D., Liu J., Yang X., Zhou C., Guo J., Wu C., Qin Y., Guo L., He J., Yu S. Chromatin accessibility dynamics during iPSC reprogramming. Cell Stem Cell. 2017;21:819–833.e6. doi: 10.1016/j.stem.2017.10.012. [DOI] [PubMed] [Google Scholar]
- Lu F., Liu Y., Inoue A., Suzuki T., Zhao K., Zhang Y. Establishing chromatin regulatory landscape during mouse preimplantation development. Cell. 2016;165:1375–1388. doi: 10.1016/j.cell.2016.05.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maalouf W.E., Liu Z., Brochard V., Renard J.P., Debey P., Beaujean N., Zink D. Trichostatin A treatment of cloned mouse embryos improves constitutive heterochromatin remodeling as well as developmental potential to term. BMC Dev. Biol. 2009;9:11. doi: 10.1186/1471-213X-9-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLean C.Y., Bristor D., Hiller M., Clarke S.L., Schaar B.T., Lowe C.B., Wenger A.M., Bejerano G. GREAT improves functional interpretation of cis-regulatory regions. Nat. Biotechnol. 2010;28:495–501. doi: 10.1038/nbt.1630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minucci S., Wong J., Blanco J.C., Shi Y.B., Wolffe A.P., Ozato K. Retinoid receptor-induced alteration of the chromatin assembled on a ligand-responsive promoter in Xenopus oocytes. Mol. Endocrinol. 1998;12:315–324. doi: 10.1210/mend.12.3.0074. [DOI] [PubMed] [Google Scholar]
- Miyamoto K., Pasque V., Jullien J., Gurdon J.B. Nuclear actin polymerization is required for transcriptional reprogramming of Oct4 by oocytes. Genes Dev. 2011;25:946–958. doi: 10.1101/gad.615211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyamoto K., Teperek M., Yusa K., Allen G.E., Bradshaw C.R., Gurdon J.B. Nuclear Wave1 is required for reprogramming transcription in oocytes and for normal development. Science. 2013;341:1002–1005. doi: 10.1126/science.1240376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyamoto K., Tajima Y., Yoshida K., Oikawa M., Azuma R., Allen G.E., Tsujikawa T., Tsukaguchi T., Bradshaw C.R., Jullien J. Reprogramming towards totipotency is greatly facilitated by synergistic effects of small molecules. Biol. Open. 2017;6:415–424. doi: 10.1242/bio.023473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okazawa H., Okamoto K., Ishino F., Ishino-Kaneko T., Takeda S., Toyoda Y., Muramatsu M., Hamada H. The oct3 gene, a gene for an embryonic transcription factor, is controlled by a retinoic acid repressible enhancer. EMBO J. 1991;10:2997–3005. doi: 10.1002/j.1460-2075.1991.tb07850.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soufi A., Donahue G., Zaret K.S. Facilitators and impediments of the pluripotency reprogramming factors’ initial engagement with the genome. Cell. 2012;151:994–1004. doi: 10.1016/j.cell.2012.09.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stalder J., Larsen A., Engel J.D., Dolan M., Groudine M., Weintraub H. Tissue-specific DNA cleavages in the globin chromatin domain introduced by DNAase I. Cell. 1980;20:451–460. doi: 10.1016/0092-8674(80)90631-5. [DOI] [PubMed] [Google Scholar]
- Teif V.B., Vainshtein Y., Caudron-Herger M., Mallm J.P., Marth C., Höfer T., Rippe K. Genome-wide nucleosome positioning during embryonic stem cell development. Nat. Struct. Mol. Biol. 2012;19:1185–1192. doi: 10.1038/nsmb.2419. [DOI] [PubMed] [Google Scholar]
- Thurman R.E., Rynes E., Humbert R., Vierstra J., Maurano M.T., Haugen E., Sheffield N.C., Stergachis A.B., Wang H., Vernot B. The accessible chromatin landscape of the human genome. Nature. 2012;489:75–82. doi: 10.1038/nature11232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voigt P., Tee W.W., Reinberg D. A double take on bivalent promoters. Genes Dev. 2013;27:1318–1338. doi: 10.1101/gad.219626.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang D., Garcia-Bassets I., Benner C., Li W., Su X., Zhou Y., Qiu J., Liu W., Kaikkonen M.U., Ohgi K.A. Reprogramming transcription by distinct classes of enhancers functionally defined by eRNA. Nature. 2011;474:390–394. doi: 10.1038/nature10006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang W., Yang J., Liu H., Lu D., Chen X., Zenonos Z., Campos L.S., Rad R., Guo G., Zhang S. Rapid and efficient reprogramming of somatic cells to induced pluripotent stem cells by retinoic acid receptor gamma and liver receptor homolog 1. Proc. Natl. Acad. Sci. USA. 2011;108:18283–18288. doi: 10.1073/pnas.1100893108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- West J.A., Cook A., Alver B.H., Stadtfeld M., Deaton A.M., Hochedlinger K., Park P.J., Tolstorukov M.Y., Kingston R.E. Nucleosomal occupancy changes locally over key regulatory regions during cell differentiation and reprogramming. Nat. Commun. 2014;5:4719. doi: 10.1038/ncomms5719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu J., Huang B., Chen H., Yin Q., Liu Y., Xiang Y., Zhang B., Liu B., Wang Q., Xia W. The landscape of accessible chromatin in mammalian preimplantation embryos. Nature. 2016;534:652–657. doi: 10.1038/nature18606. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.