Abstract
The number of bariatric surgeries being performed worldwide has markedly risen. While the improvement in obesity-associated comorbidities after bariatric surgery is well-established, very little is known about its impact on cancer risk. The peripheral lymphocyte micronucleus test is a widely used method for the monitoring of chromosomal damage levels in vivo, and micronucleus frequency positively correlates with cancer risk. Therefore, the aim of this study was to compare the micronucleus frequency before and after bariatric surgery in obese subjects. Peripheral blood mononuclear cells were collected from 45 obese subjects before and at two time-points after bariatric surgery (6 and 12 months) to assess spontaneous micronucleus frequency. Consistent with the increased cancer risk previously shown, bariatric surgery-induced weight loss led to a significant reduction in lymphocyte micronucleus frequency after 12 months. Interestingly, comorbidities such as type 2 diabetes mellitus and metabolic syndrome further seemed to have an impact on the lymphocyte micronucleus frequency. Our findings may indicate a successful reduction of cancer risk in patients following weight loss caused by bariatric surgery.
Introduction
Obesity is one of the most pressing health challenges of the 21st century. According to the WHO, over 600 million adults around the world suffer from obesity, which is closely associated with other chronic diseases such as type 2 diabetes mellitus (T2DM), hypertension, non-alcoholic fatty liver disease (NAFLD) and cardiovascular disease1. The clustering of obesity, insulin resistance and dyslipidemia in individuals has been referred to as metabolic syndrome2. There are reports of increased cancer risk among obese individuals, with T2DM itself representing an independent risk factor. Since 80% of T2DM patients are overweight or obese, cancer risk becomes even more important for the subgroup of patients with metabolic syndrome3.
Conservative weight loss regimens are limited in their efficiency, making bariatric surgery the superior therapeutic option - especially for morbid obesity and related comorbidities4,5. Indeed, various follow-up studies have demonstrated the successful remission of T2DM associated with bariatric surgery-induced weight loss as well as a reduction in mortality6–8. Similarly, the Swedish Obese Subjects (SOS) study and the Utah Cancer Registry (UCR) reported a decrease in cancer-related mortality after bariatric surgery8–10. However, elevated (colon) cancer incidences have also been reported11.
Genomic instability is one of the driving forces for carcinogenesis. Micronuclei, chromatin containing structures in the cytoplasm, are a good marker of genomic damage and are a subtype of chromosomal aberrations12,13. The micronucleus test is a well-established method for the detection of genotoxicity. For human biomonitoring, peripheral blood-derived lymphocytes are the most often used cells14,15. Numerous studies have reported increased micronucleus frequency in peripheral blood-derived lymphocytes among cancer patients and population studies indicated a correlation between cancer risk and micronucleus frequency, supporting a positive association with cancer risk16–24. An increase of micronuclei has been found in obesity25–28, but the impact of weight loss on genomic damage has not been investigated so far.
Therefore, the purpose of this study was to analyze the possible influence of weight loss induced by bariatric surgery on chromosomal damage as detected by micronucleus formation.
Materials and Methods
Materials
Unless stated otherwise, chemicals were purchased from Sigma Aldrich Germany (Munich, Germany). Cell culture media and reagents were purchased from PAA Laboratories (Pasching, Austria) and Life Technologies (Darmstadt, Germany). Fetal bovine serum (FBS) was from Biochrom (Berlin, Germany). Microscope slides were from Asisstent (Sondheim, Germany) and methanol was from thgeyer (Renningen, Germany). Gel Green was purchased from Biotrend (Köln, Germany).
Methods
Studied Subjects
Blood samples were collected from 45 morbidly obese (38 female and 7 male) subjects before, 6 and 12 months after the bariatric surgery. The bariatric surgery was performed in the Surgery Department, University Hospital Wuerzburg. Sample collection was conducted in the Surgery and Endocrinology Departments, University Hospital Wuerzburg and all participants gave written informed consent. This study was approved by the Ethics Committee of the University of Wuerzburg (Study No: 186/14). All experiments were performed in accordance with relevant guidelines and regulations.
Insulin resistance had been determined prior to this study by HOMA-IR (homeostasis model assessment-insulin resistance) and the presence of a metabolic syndrome was defined according to the current definition of the international diabetes federation consensus worldwide definition of the metabolic syndrome29.
Blood sampling and isolation of mononuclear cells
Blood samples were obtained from 45 obese subjects with a BMI of 51.02 ± 0.94 kg/m2 and an age of 44.37 ± 1.62 years. Sampling was performed before and after (about 6 and 12 months) bariatric surgery. Whole blood was collected into commercially available EDTA tubes. Peripheral blood mononuclear cells (PBMCs) were isolated by density gradient centrifugation on histopaque. Blood samples were layered on histopaque surface (1:1) and were centrifuged at room temperature 1600rpm for 30 min. After centrifugation, the so-called “buffy coat” containing mononuclear cells was pipetted into a 15 ml tube and washed two times with RPMI 1640 medium (containing 1% FBS, 1% L-glutamine and penicillin) at 12000 rpm for 10 min at room temperature (RT). Isolated PBMCs were used for micronucleus assessment. Blood parameters were determined by the central laboratory of the University Hospital Wuerzburg.
Cytokinesis block micronucleus assay
After isolation, PBMCs were washed two times with RPMI medium (containing 1% FBS, 1% L-glutamine and penicillin) and cell number was determined by a cell counting chamber. 1 × 106 cells/ml were cultivated with 1 µg/ml phytohaemagglutinin (PHA) for 44 hours at 37 °C with 5% CO2. Subsequently, 3 µg/ml cytochalasin B was added for another 24 hours to yield binucleated cells. On the next day, cell number was determined and cells were diluted to 300,000 cells/ml. One hundred µl of this cell suspension was placed on to a microscope slide (2 slides for each patient 30,000 cells/slide) by cytospin centrifugation (at 1000 rpm for 5 min) and fixed in methanol at −20 °C for at least two hours.
Shortly before staining, two slides for each patient were air dried under the fume hood and stained with gel green (1:100 diluted in bidistilled water) and mounted with Dabco (1, 4- diazabicyclo[2.2.2]octane) for microscopic evaluation. For each patient, two replicate slides and 1000 binucleated cells per slide were scored for micronuclei. As a quality criteria of the lymphocyte culture, proliferation index was assessed. 1000 cells from two replicate slides were scored for the assessment of the proliferation index (PI) for the categories of mono- (MoN), bi- (BN) and multi-nucleated (MuN) cells together with apoptosis, mitosis, nucleoplasmic bridges and nuclear buds15. The calculation of the PI was done according to the following formula. The frequency of apoptosis and mitosis were represented as per mill (%o). The frequency of nucleoplasmic bridges and nuclear buds were less than 1% (data not shown).
Statistics
Statistical analysis was performed using SPSS 22 software. Data are presented as Mean ± SEM. Normality of the data was checked with the Shapiro-Wilk test and according to the results either a t-test or the Wilcoxon test (for independent samples Mann-Whitney) was performed to check for significant differences between individual groups. Results were considered significant with p ≤ 0.05.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
Results
Patient blood biochemistry, comorbidities and weight loss
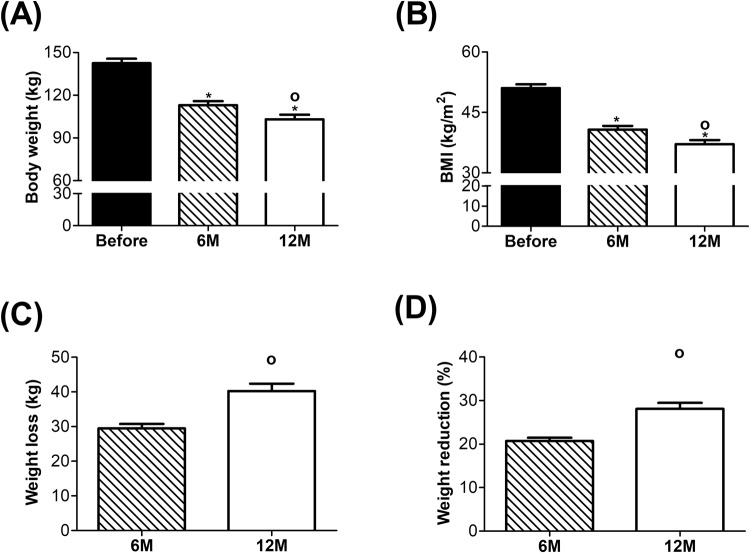
At baseline, obese study participants were at an average age of 44.37 ± 1.62 years with a BMI of 51.02 ± 0.94 kg/m2. Six months after surgery, obese subjects lost a significant amount of excess body weight and reached an average BMI of 40.76 ± 0.92 kg/m2 (Fig. 1A,B). In the second 6-month period, subjects continued to lose a significant amount of excess body weight and reached an average BMI of 37.11 ± 1.04 kg/m2 by the end of our study Weight loss in kg and weight reduction as percent of previous body weight are shown in Fig. 1C,D, respectively. A summary for the body weight (kg), BMI, and age over the experimental time frame as well as gender distribution can be seen in Table 1.
Figure 1.
Body weight, BMI and weight loss before and after bariatric surgery. (A) Body weight in kg, (B) Body Mass Index (BMI kg/m2), (C) Weight loss in kg and (D) Weight reduction in %. Before surgery, n = 45; 6 M, n = 35; 12 M, n = 45. *p ≤ 0.05 significant vs. Before and °p ≤ 0.05 significant vs. 6 M.
Table 1.
Summary for body weight, BMI, age and gender before and after surgery.
| Groups | Body weight (kg) | BMI (kg/m2) | Age | Gender |
|---|---|---|---|---|
| Before | 142.55 ± 3.07 | 51.02 ± 0.94 | 44.37 ± 1.62 | Female: 38 Male: 7 |
| 6 M | 113.06 ± 2.74 | 40.76 ± 0.92 | — | — |
| 12 M | 103.14 ± 3.09 | 37.11 ± 1.04 | — | — |
Elevated levels of some important biochemical blood parameters were found in morbidly obese subjects before surgery and, as soon as 6 months afterwards, there were significant improvements that generally remained so for the duration of the 12-month recording period. Among these parameters, a progressive reduction in CRP levels indicated the improvement of the chronic inflammation status and the significantly reduced glycated haemoglobin and fasting glucose levels (HbA1c) pointed to the improvement of glucose homeostasis. Notably, LDL and HDL levels showed a reciprocal pattern progressively decreasing and increasing after surgery, respectively, consistent with an improvement in lipid homeostasis. Precise biochemical blood parameters of the study subjects can be seen in Table 2 and distribution of related comorbidities is shown in Table 3.
Table 2.
Biochemical blood parameters.
| Parameters | Before | After 6 months | After 12 months | Significance |
|---|---|---|---|---|
| Glucose (mg/dl) | 99.14 ± 3.90 (N = 44) |
86.52 ± 3.61 (N = 44) |
94.50 ± 5.22 (N = 42) |
*,# |
| Alkaline phosphatase (U/l) | 80.14 ± 5.41 (N = 44) |
82.07 ± 4.06 (N = 44) |
75.60 ± 3.53 (N = 42) |
∆ |
| Triglyceride (mg/dl) | 139.61 ± 10.44 (N = 33) |
110.82 ± 5.50 (N = 44) |
105.24 ± 7.22 (N = 42) |
*,# |
| Cholesterol (mg/dl) | 190.12 ± 7.11 (N = 33) |
180.27 ± 6.26 (N = 44) | 178.93 ± 6.73 (N = 42) |
*,# |
| LDL (mg/dl) | 114.15 ± 6.84 (N = 33) |
105.93 ± 5.75 (N = 44) |
97.67 ± 6.09 (N = 42) |
*,#,∆ |
| HDL (mg/dl) | 48.12 ± 9.86 (N = 33) |
52.23 ± 1.56 (N = 44) |
59.62 ± 2.07 (N = 42) |
#,∆ |
| CRP (mg/dl) | 1.18 ± 0.18 (N = 45) |
0.59 ± 0.09 (N = 44) |
0.54 ± 0.11 (N = 42) |
*,#,∆ |
| HbA1c (%) | 5.94 ± 0.18 (N = 36) |
5.42 ± 0.08 (N = 45) |
5.45 ± 0.11 (N = 42) |
*,# |
LDL: low density lipoprotein, HDL: high density lipoprotein, CRP: C-reactive protein and HbA1c: glycated hemoglobin *p ≤ 0.05 Before vs. After 6 months, #p ≤ 0.05 Before vs. After 12 months and ∆p ≤ 0.05 After 6 months vs. After 12 months.
Table 3.
Comorbidities before surgery.
| Comorbidities | Number | % |
|---|---|---|
| IR | 14/45 | 31.1 |
| T2DM | 13/45 | 28.9 |
| IDDM | 6/45 | 13.3 |
| NASH/NAFLD | 40/45 | 88.9 |
| Hypertension | 30/45 | 66.7 |
| Metabolic syndrome | 14/45 | 31.1 |
IR: insulin resistance, T2DM: type 2 diabetes mellitus, IDDM: insulin dependent diabetes mellitus, NASH: non-alcoholic steatohepatitis, NAFLD: non-alcoholic fatty liver disease.
Analysis of lymphocyte micronucleus frequency
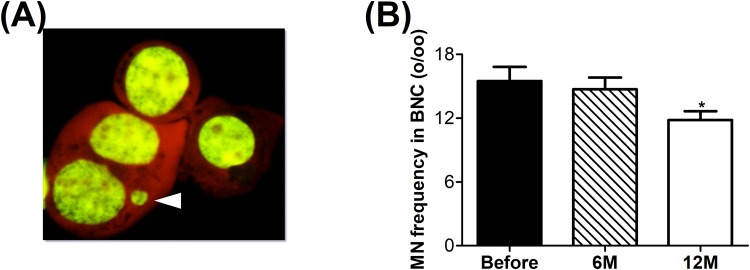
The cytokinesis blocking micronucleus test was used to determine the spontaneously accumulated chromosomal/genomic damage in lymphocytes. A representative picture of a micronucleus in a binucleated cell (BNC) can be seen in Fig. 2A. Six months after surgery, there was no significant difference in micronucleus number of obese subjects compared to before surgery. However, a significant reduction was observed 12 months after surgery (Fig. 2B). In Table 4, proliferation index, mitosis and apoptosis per mill can be found. After the weight loss a significant increase in proliferation index and mitosis were observed with a significant reduction in apoptosis.
Figure 2.
Micronucleus frequency per mill (%o) in isolated lymphocytes of obese subjects. (A) Representative picture of a micronucleus in a binucleated cell indicated by the white arrow. (B) Micronucleus frequency per mill (%o) before and after surgery. Before, n = 45; 6 M, n = 35 and 12 M, n = 45. *p ≤ 0.05 significant vs. before.
Table 4.
Proliferation Index (PI), Mitosis and Apoptosis.
| Groups | PI | Mitosis (%o) | Apoptosis (%o) |
|---|---|---|---|
| Before | 1.52 ± 0.03 | 12.87 ± 1.34 | 9.60 ± 1.47 |
| 6 M | 1.60 ± 0.03 | 20.81 ± 1.74 (*) | 6.77 ± 1.02 |
| 12 M | 1.59 ± 0.03 (#) | 18.88 ± 1.23 (#) | 5.63 ± 1.21 (#) |
*p ≤ 0.05 Before vs. After 6 months and #p ≤ 0.05 Before vs. After 12 months.
After dividing patients into subgroups according to comorbidities (insulin resistance (IR)/type 2 diabetes mellitus (T2DM), hypertension (H) and metabolic syndrome (MetS)), significant differences in micronucleus frequencies were detected (Fig. 3). The insulin resistant/T2DM group showed a significantly higher micronucleus frequency compared to the nondiabetic (ND) group. The hypertensive (H) subgroup did not show any significant difference compared to the non-hypertensive (NH) subgroup. The metabolic syndrome subgroup had a significantly higher micronucleus number compared to patients not affected by metabolic syndrome (Fig. 3A). Within these subgroups that are displayed in Fig. 3A, the small group of nondiabetic/non-hypertensive patients (n = 4) exhibited the lowest micronucleus frequency with 8.87 ± 2.81 (mean MN/1000 BNC ± SEM) and another small group of patients with insulin-dependent diabetes mellitus (n = 5) showed the highest micronucleus frequency with 25.82 ± 4.40 (mean MN/1000 BNC ± SEM) among all groups before surgery.
Figure 3.
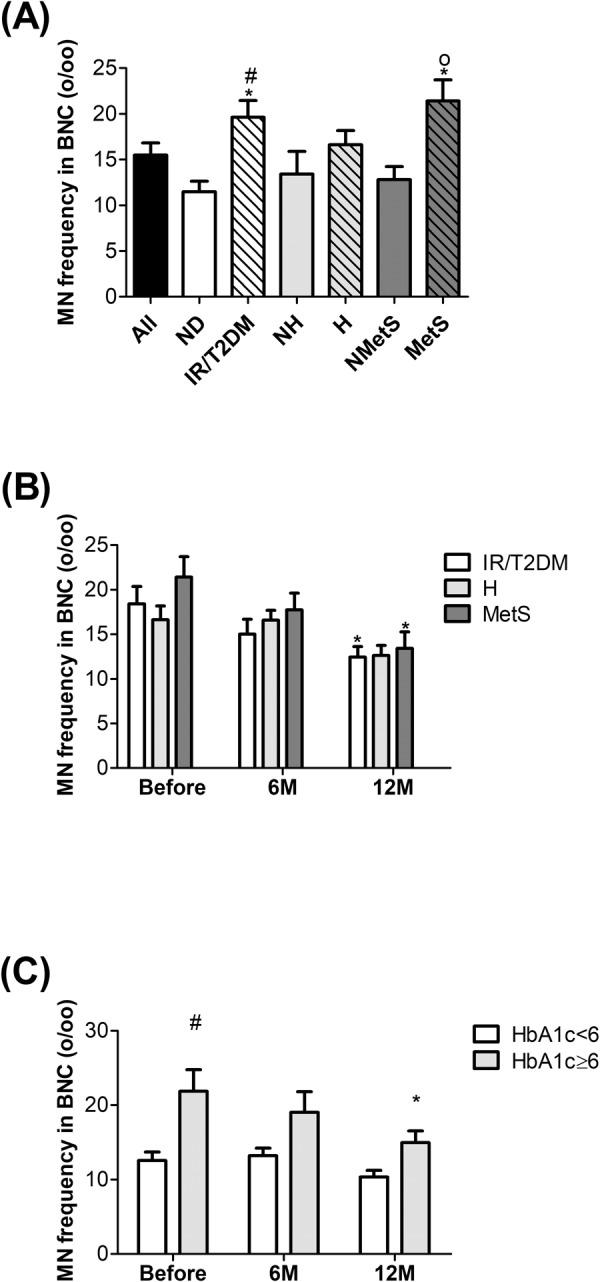
Micronucleus frequency per mill (%o) among the subgroups of obese subjects before surgery. (A) Comparison of micronucleus frequency between all patients, nondiabetic (ND), IR/T2DM, nonhypertensive (NH), hypertensive (H), nonmetabolic syndrome (NMetS) and metabolic syndrome (MetS). (B) Comparison of micronucleus frequency between IR/T2DM, hypertensive (H) and metabolic syndrome (MetS) before and after surgery. (C) Comparison of micronucleus frequency between a subgroup of HbA1c < 6 and HbA1c ≥ 6 before and after surgery. Before surgery, nAll = 45, nND = 19, nIR/T2DM = 26, nNH = 16, nH = 29, nNMetS = 31 and nMetS = 14; 6 M, nIR/T2DM = 21, nH = 21, and nMetS = 10; 12 M, nIR/T2DM = 26, nH = 29 and nMetS = 14. *p ≤ 0.05 significant vs. All or vs. BeforeIR/T2DM or vs. BeforeMetS, #p ≤ 0.05 significant vs. ND or vs. BeforeHbA1c≥6 and op ≤ 0.05 significant vs. NMetS.
In Fig. 3B, the micronucleus frequency of subgroups with IR/T2DM, hypertension and metabolic syndrome are shown before and after weight loss. Within the IR/T2DM group, we observed a significant reduction in micronucleus frequency after 12 months. Hypertensive patients did not show a significant difference. Patients with metabolic syndrome (obese, IR or T2DM and hypertensive) showed significant reduction in micronucleus number at 12 months after surgery.
Since our observation showed a strong effect in patients with metabolic syndrome and IR/T2DM, but not with hypertension, we decided to have a closer look on the effect of blood glucose levels on micronucleus formation. Therefore, glycated haemoglobin (HbA1c) was taken as a separate criteria and patients before and after surgery were divided into two groups according to their HbA1c levels (Fig. 3C). The patients with HbA1c levels ≥6 showed significantly higher micronucleus numbers than the patients with HbA1c levels <6 mg/dl. Only the patients with HbA1c levels ≥6 showed a significant reduction in micronucleus number 12 months after the surgery.
Discussion
Increased DNA damage in obese subjects compared to healthy controls has consistently been documented25–28.
In a previous study performed on obese Zucker rats, we found that increased genomic damage in kidney, liver and colon was improved after weight loss induced either by gastric bypass surgery or by chronic caloric restriction30. This prompted us to observe the genomic damage level of an obese patient group before and at two time-points during the first year after bariatric surgery. There was a clear and significant reduction in micronucleus frequency after 12 months but not after 6 months. The significant increase in proliferation index and mitosis with a significant reduction in apoptosis indicated improved fitness of the isolated lymphocytes after weight loss. It has to be kept in mind that even 12 months after bariatric surgery, patients still classify as severely obese.
To our knowledge, this is the first study in which genomic damage after bariatric surgery has been analyzed. Two previously published clinical studies did however demonstrate an effect of weight loss induced by bariatric surgery on oxidative stress. Billeter and colleagues31 demonstrated a significant reduction of insulin resistance, leptin values, inflammation and oxidative stress 24 months after bariatric surgery. Horn and colleagues32 also showed reduced oxidative stress in 16 obese women 180 days after bariatric surgery. The average BMI of the participants was 44.1 ± 6.8 kg/m2 before surgery. Even though their starting BMI was lower than that of our obese group (BMI 51.02 ± 0.94 kg/m2), the significantly reduced lipid peroxidation and carbonylated protein level of the patients after surgery were still significantly higher than those of the healthy controls. Also, while weight loss may reduce the chronic low inflammatory status of obese patients, the dramatic reduction of fat tissue may burden the body with oxidative stress-inducing molecules. Thus, the situation may be complex and may depend on the amount of fat lost and the starting BMI. At the time point of 6 months (i.e. about 180 days), our patients exhibited significantly lowered CRP (reflecting reduced inflammation), which is in accordance with the published findings on oxidative stress. However, CRP was slightly higher than the healthy reference range. In future studies, a comparison between different extents of obesity might be helpful to see whether there is threshold of final weight or amount of weight loss for reduction of the genomic damage or oxidative stress markers to a healthy control level. It would be further of interest to determine whether a chronic caloric restriction-induced weight loss without surgery can lead to reductions in DNA damage.
In addition to the excess body weight, there are several other health concerns associated with obesity including cardiovascular disease, type 2 diabetes mellitus, hypertension and non-alcoholic fatty liver disease. All these comorbidities are known for their association with oxidative stress33–37. Increased oxidative stress could damage cellular macromolecules including DNA. The most common comorbidities among our obese subjects were non-alcoholic fatty liver disease together with steatohepatitis, hypertension and T2DM/Insulin resistance. We divided the obese subjects according to some of these comorbidities and compared the micronucleus frequency among these subgroups. Since the vast majority (89%) of the subjects had either non-alcoholic fatty liver disease (NAFLD) or non-alcoholic steatohepatitis (NASH), we did not use these two comorbidities as an extra category. Patients with hypertension and T2DM/insulin resistance (IR) were referred to as metabolic syndrome subgroup. Before bariatric surgery, the T2DM/insulin resistance (IR) subgroup showed a significantly higher micronucleus frequency compared to the remaining groups. Within the diabetic patients, the small subgroup of insulin dependent patients showed a significantly higher micronucleus frequency. This is consistent with previous findings that T2DM in itself is a risk factor for cancer (3). The reduction of genomic damage in T2DM/IR patients after bariatric surgery might indicate a direct effect of insulin which we have previously shown in vitro and in rodent models in vivo38–40. The putative mechanism behind this might be that increased insulin levels cause an additional increase of ROS formation and ROS induced DNA damage via mitochondrial or NADPH oxidase activation41,42.
Interestingly, hypertension alone did not lead to any significant elevation of micronucleus formation. Nevertheless, the metabolic syndrome subgroup showed a significant increase in micronucleus frequency. This points to a synergistic effect of hypertension.
In summary, the findings from the present study support the existing knowledge about increased DNA damage in obese subjects and for the first time demonstrate the beneficial effect of bariatric surgery-induced weight loss on micronucleus frequency in humans. However, there are still some important knowledge gaps, such as whether a reduction to the genomic damage level of healthy individuals can be achieved over a longer time after surgery or with a more extensive weight loss, and whether particular methods of weight loss are superior to others. In addition, a comparison of MN frequency from volunteers with a wide range of BMI would be interesting though there was no significant correlation between MN frequency and BMI in this study. In addition to all other beneficial health improvements in severely obese patients, bariatric surgery induced weight loss lowers the genomic damage and this may represent a lower cancer risk.
Acknowledgements
We thank to Nicole Buchta, Kathrin Hohl and Lena Leppich for their excellent collaboration in collecting blood samples. This publication was funded by the German Research Foundation (DFG) and the University of Wuerzburg in the funding programme Open Access Publishing.
Author Contributions
E.E.B. performed the data analysis and wrote the manuscript. C.A. contributed to preparation and analysis of the samples. I.H. collected and summarized the clinical data. M.H. reviewed the manuscript and contributed to the discussion. F.S. and H.S. designed the study and reviewed the manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Florian Seyfried and Helga Stopper contributed equally to this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.WHO. Obesity and Overweight, Fact sheet No 311 (updated June 2016). (2013).
- 2.Huang PL. A comprehensive definition for metabolic syndrome. Disease Models & Mechanisms. 2009;2:231–237. doi: 10.1242/dmm.001180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gandhi, G. & Saini, A. K. DNA Damage and Obesity In Diabetic Patients (2013).
- 4.Picot, J. et al. The clinical effectiveness and cost-effectiveness of bariatric (weight loss) surgery for obesity: a systematic review and economic evaluation. Health technology assessment (Winchester, England)13, 1–190, 215–357, iii-iv, 10.3310/hta13410 (2009). [DOI] [PubMed]
- 5.Colquitt, J. L., Pickett, K., Loveman, E. & Frampton, G. K. Surgery for weight loss in adults. The Cochrane database of systematic reviews, Cd003641, 10.1002/14651858.CD003641.pub4 (2014). [DOI] [PMC free article] [PubMed]
- 6.Courcoulas AP, et al. Long-term Outcomes of Bariatric Surgery: A National Institutes of Health Symposium. JAMA surgery. 2014;149:1323–1329. doi: 10.1001/jamasurg.2014.2440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Adams TD, et al. Long-term mortality after gastric bypass surgery. The New England journal of medicine. 2007;357:753–761. doi: 10.1056/NEJMoa066603. [DOI] [PubMed] [Google Scholar]
- 8.Sjostrom L, et al. Effects of bariatric surgery on mortality in Swedish obese subjects. The New England journal of medicine. 2007;357:741–752. doi: 10.1056/NEJMoa066254. [DOI] [PubMed] [Google Scholar]
- 9.Adams TD, et al. Cancer Incidence and Mortality After Gastric Bypass Surgery. Obesity (Silver Spring, Md.) 2009;17:796–802. doi: 10.1038/oby.2008.610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Anveden Å, et al. Long-term incidence of female-specific cancer after bariatric surgery or usual care in the Swedish Obese Subjects Study. Gynecologic Oncology. 2017;145:224–229. doi: 10.1016/j.ygyno.2017.02.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Derogar M, et al. Increased risk of colorectal cancer after obesity surgery. Ann Surg. 2013;258:983–988. doi: 10.1097/SLA.0b013e318288463a. [DOI] [PubMed] [Google Scholar]
- 12.Bonassi S, El-Zein R, Bolognesi C, Fenech M. Micronuclei frequency in peripheral blood lymphocytes and cancer risk: evidence from human studies. Mutagenesis. 2011;26:93–100. doi: 10.1093/mutage/geq075. [DOI] [PubMed] [Google Scholar]
- 13.Bonassi S, et al. An increased micronucleus frequency in peripheral blood lymphocytes predicts the risk of cancer in humans. Carcinogenesis. 2007;28:625–631. doi: 10.1093/carcin/bgl177. [DOI] [PubMed] [Google Scholar]
- 14.Fenech M, Holland N, Chang WP, Zeiger E, Bonassi S. The HUman MicroNucleus Project—An international collaborative study on the use of the micronucleus technique for measuring DNA damage in humans. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis. 1999;428:271–283. doi: 10.1016/S1383-5742(99)00053-8. [DOI] [PubMed] [Google Scholar]
- 15.Fenech M. Cytokinesis-block micronucleus cytome assay. Nature protocols. 2007;2:1084–1104. doi: 10.1038/nprot.2007.77. [DOI] [PubMed] [Google Scholar]
- 16.El-Zein RA, et al. Cytokinesis-blocked micronucleus assay as a novel biomarker for lung cancer risk. Cancer Res. 2006;66:6449–6456. doi: 10.1158/0008-5472.CAN-06-0326. [DOI] [PubMed] [Google Scholar]
- 17.Yildirim IH, Yesilada E, Yologlu S. Micronucleus frequency in peripheral blood lymphocytes and exfoliated buccal cells of untreated cancer patients. Genetika. 2006;42:705–710. [PubMed] [Google Scholar]
- 18.Santos RA, et al. Basal levels of DNA damage detected by micronuclei and comet assays in untreated breast cancer patients and healthy women. Clin Exp Med. 2010;10:87–92. doi: 10.1007/s10238-009-0079-4. [DOI] [PubMed] [Google Scholar]
- 19.Minicucci EM, et al. DNA damage in lymphocytes and buccal mucosa cells of children with malignant tumours undergoing chemotherapy. Clinical and Experimental Medicine. 2008;8:79–85. doi: 10.1007/s10238-008-0161-3. [DOI] [PubMed] [Google Scholar]
- 20.Milosevic-Djordjevic O, Grujicic D, Vaskovic Z, Marinkovic D. High micronucleus frequency in peripheral blood lymphocytes of untreated cancer patients irrespective of gender, smoking and cancer sites. Tohoku J Exp Med. 2010;220:115–120. doi: 10.1620/tjem.220.115. [DOI] [PubMed] [Google Scholar]
- 21.Karaman A, Binici DN, Kabalar ME, Çalıkuşu Z. Micronucleus analysis in patients with colorectal adenocarcinoma and colorectal polyps. World Journal of Gastroenterology: WJG. 2008;14:6835–6839. doi: 10.3748/wjg.14.6835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cefle K, et al. Increased sister chromatid exchange frequency in young women with breast cancer and in their first-degree relatives. Cancer Genet Cytogenet. 2006;171:65–67. doi: 10.1016/j.cancergencyto.2006.06.005. [DOI] [PubMed] [Google Scholar]
- 23.Chang P, Li Y, Li D. Micronuclei levels in peripheral blood lymphocytes as a potential biomarker for pancreatic cancer risk. Carcinogenesis. 2011;32:210–215. doi: 10.1093/carcin/bgq247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Murgia E, Ballardin M, Bonassi S, Rossi AM, Barale R. Validation of micronuclei frequency in peripheral blood lymphocytes as early cancer risk biomarker in a nested case-control study. Mutat Res. 2008;639:27–34. doi: 10.1016/j.mrfmmm.2007.10.010. [DOI] [PubMed] [Google Scholar]
- 25.Karaman A, Aydin H, Geckinli B, Cetinkaya A, Karaman S. DNA damage is increased in lymphocytes of patients with metabolic syndrome. Mutat Res Genet Toxicol Environ Mutagen. 2015;782:30–35. doi: 10.1016/j.mrgentox.2015.03.009. [DOI] [PubMed] [Google Scholar]
- 26.Donmez-Altuntas H, et al. Evaluation of chromosomal damage, cytostasis, cytotoxicity, oxidative DNA damage and their association with body-mass index in obese subjects. Mutat Res Genet Toxicol Environ Mutagen. 2014;771:30–36. doi: 10.1016/j.mrgentox.2014.06.006. [DOI] [PubMed] [Google Scholar]
- 27.Andreassi MG, Barale R, Iozzo P, Picano E. The association of micronucleus frequency with obesity, diabetes and cardiovascular disease. Mutagenesis. 2011;26:77–83. doi: 10.1093/mutage/geq077. [DOI] [PubMed] [Google Scholar]
- 28.Scarpato R, et al. Nuclear damage in peripheral lymphocytes of obese and overweight Italian children as evaluated by the gamma-H2AX focus assay and micronucleus test. Faseb j. 2011;25:685–693. doi: 10.1096/fj.10-168427. [DOI] [PubMed] [Google Scholar]
- 29.Alberti KG, Zimmet P, Shaw J. Metabolic syndrome–a new world-wide definition. A Consensus Statement from the International Diabetes Federation. Diabetic medicine: a journal of the British Diabetic Association. 2006;23:469–480. doi: 10.1111/j.1464-5491.2006.01858.x. [DOI] [PubMed] [Google Scholar]
- 30.Bankoglu EE, et al. Impact of weight loss induced by gastric bypass or caloric restriction on oxidative stress and genomic damage in obese Zucker rats. Free Radic Biol Med. 2016;94:208–217. doi: 10.1016/j.freeradbiomed.2016.02.033. [DOI] [PubMed] [Google Scholar]
- 31.Billeter AT, et al. Gastric bypass simultaneously improves adipose tissue function and insulin-dependent type 2 diabetes mellitus. Langenbecks Arch Surg. 2017 doi: 10.1007/s00423-017-1601-x. [DOI] [PubMed] [Google Scholar]
- 32.Horn RC, et al. Obesity, bariatric surgery and oxidative stress. Rev Assoc Med Bras (1992) 2017;63:229–235. doi: 10.1590/1806-9282.63.03.229. [DOI] [PubMed] [Google Scholar]
- 33.Dandona P, et al. Oxidative damage to DNA in diabetes mellitus. Lancet. 1996;347:444–445. doi: 10.1016/S0140-6736(96)90013-6. [DOI] [PubMed] [Google Scholar]
- 34.Hinokio Y, et al. Oxidative DNA damage in diabetes mellitus: its association with diabetic complications. Diabetologia. 1999;42:995–998. doi: 10.1007/s001250051258. [DOI] [PubMed] [Google Scholar]
- 35.Collins AR, et al. DNA damage in diabetes: correlation with a clinical marker. Free Radic Biol Med. 1998;25:373–377. doi: 10.1016/S0891-5849(98)00053-7. [DOI] [PubMed] [Google Scholar]
- 36.Botto N, et al. Elevated levels of oxidative DNA damage in patients with coronary artery disease. Coron Artery Dis. 2002;13:269–274. doi: 10.1097/00019501-200208000-00004. [DOI] [PubMed] [Google Scholar]
- 37.Bankoglu EE, et al. Role of PTEN in Oxidative Stress and DNA Damage in the Liver of Whole-Body Pten Haplodeficient Mice. PLoS One. 2016;11:e0166956. doi: 10.1371/journal.pone.0166956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Othman EM, Hintzsche H. & Stopper, H. Signaling steps in the induction of genomic damage by insulin in colon and kidney cells. Free Radic Biol Med. 2014;68:247–257. doi: 10.1016/j.freeradbiomed.2013.12.010. [DOI] [PubMed] [Google Scholar]
- 39.Othman EM, Kreissl MC, Kaiser FR, Arias-Loza PA, Stopper H. Insulin-mediated oxidative stress and DNA damage in LLC-PK1 pig kidney cell line, female rat primary kidney cells, and male ZDF rat kidneys in vivo. Endocrinology. 2013;154:1434–1443. doi: 10.1210/en.2012-1768. [DOI] [PubMed] [Google Scholar]
- 40.Othman EM, Leyh A, Stopper H. Insulin mediated DNA damage in mammalian colon cells and human lymphocytes in vitro. Mutat Res. 2013;745-746:34–39. doi: 10.1016/j.mrfmmm.2013.03.006. [DOI] [PubMed] [Google Scholar]
- 41.Espinosa A, Garcia A, Hartel S, Hidalgo C, Jaimovich E. NADPH oxidase and hydrogen peroxide mediate insulin-induced calcium increase in skeletal muscle cells. The Journal of biological chemistry. 2009;284:2568–2575. doi: 10.1074/jbc.M804249200. [DOI] [PubMed] [Google Scholar]
- 42.Mahmoud AM, et al. Nox2 contributes to hyperinsulinemia-induced redox imbalance and impaired vascular function. Redox Biology. 2017;13:288–300. doi: 10.1016/j.redox.2017.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during the current study are available from the corresponding author on reasonable request.