Abstract
Hydrogen sulfide (H2S), an endogenous gasotransmitter, mediated a variety of biological processes through multiple signaling pathways, and aberrant H2S metabolism has been associated with mesenchymal stem cell (MSC) dysfunction. Here we employed the small interfering RNA treatment for cystathionine β-synthase (CBS), cystathionine γ-lyase, the main enzymes to synthesize H2S, and CBS-knockout mice to analyze the effect of H2S on dental pulp homeostasis. We showed that H2S deficiency attenuated dental pulp stem cell (DPSC) osteogenic/dentinogenic differentiation in vitro and in vivo with enhanced cell proliferation. Mechanically, H2S facilitated the transient receptor potential action channel subfamily V member 1-mediated calcium (Ca2+) influx, which subsequently activated the β-catenin pathway. While H2S deficiency decreased Ca2+, resulting in glycogen synthase kinase-3β-mediated β-catenin degradation, which controls proliferation and differentiation of DPSCs. Consistently, H2S-deficient mice displayed disturbed pattern of dental pulp and less dentin formation. In this study, we identified a previously unknown mechanism by which H2S regulates DPSC lineage determination and dental pulp homeostasis.
Subject terms: Stem cells, Cell signalling
Introduction
Hydrogen sulfide (H2S) could freely diffuse through cell membranes to elicit various cellular events. H2S is synthesized by three enzymes, namely cystathionine β-synthase (CBS), cystathionine γ-lyase (CSE), and 3-mercaptopyruvate sulfurtransferase (MPST) from l-cysteine1. Alternations of H2S metabolism have been linked to osteoporosis and T cell-related immune disorders2,3. Endogenous H2S is essential to maintain differentiation and proliferation of neural stem cells and human-induced pluripotent stem cells, and to restore the function of endothelial progenitor cell and mesenchymal stem cells (MSCs) to ensure bone homeostasis4,5. Our previous studies showed that H2S deficiency resulted in bone marrow MSC impairment caused by H2S deficiency reduced sulfhydration of combined Ca2+ transient receptor potential (TRP)-mediated (transient receptor potential action channel subfamily V member 3 (TRPV3), TRPV6, and TRPM4) aberrant Ca2+ influx6. H2S donor was also reported to prevent trabecular bone loss induced by ovariectomy via increased Wnt ligands Wnt16, Wnt2b, Wnt6, and Wnt10b in the bone marrow7.
The oral cavity is abundant with a plethora of bacteria, some of which are well known to produce H2S. When the dynamic ecological equilibrium in the biofilm is disturbed, bacteria may elicit oral diseases such as caries, gingivitis, and periodontitis8. Several studies have indicated the role of H2S in dental stem cells9. Dental pulp stem cells (DPSCs), isolated from dental pulp, were reported to be highly proliferative and capable of differentiating into a variety of cells including odontoblasts, osteoblasts, adipocytes, and neural cells. DPSCs could be induced to generate bone and dentin and play an important role in maintaining dental pulp and dentin homeostasis10. Recently, studies showed that H2S increased DPSC hepatic differentiation11,12. Exogenous H2S induced DPSC apoptosis by activating mitochondrial pathway apoptosis13. H2S may also be involved in periodontal ligament stem cell proliferation and differentiation and orthodontic tooth movement14–17. However, it remains unclear as to how H2S molecularly maintains homeostasis of DPSCs and dental pulp.
In this study, we showed that the gasotransmitter H2S is essential to maintain DPSC function via TRPV1-mediated Ca2+ influx-stimulated glycogen synthase kinase-3β (GSK3β)/β-catenin pathway.
Results
DPSCs expressed CBS, CSE, and produced H2S
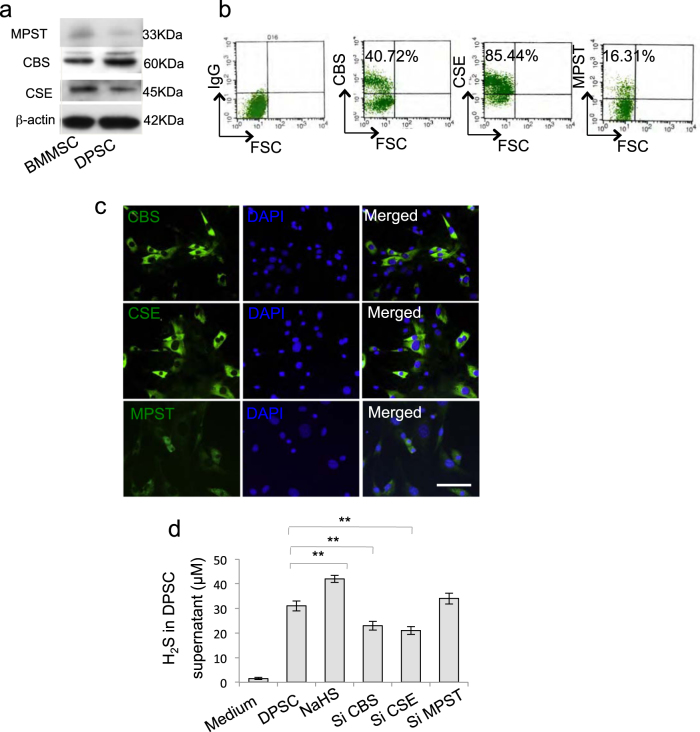
Aberrant H2S metabolism has been linked to MSC dysfunction; here we revealed that DPSCs expressed CBS, CSE, and ubiquitous enzyme MPST, the main catalyzed enzymes of H2S synthesis (Fig. 1a, c and Supplementary Figure 1). Meanwhile, DPSCs produced H2S in the culture supernatant, which was downregulated by CBS small interfering RNA (siRNA) or CSE siRNA treatment but not MPST siRNA treatment (Fig. 1d), and H2S concentration was upregulated by H2S donor NaHS treatment (Fig. 1d).
Figure 1. Human dental pulp stem cells (DPSCs) produced H2S.
a Western blot analysis showed that DPSCs expressed CBS, CSE, and MPST as BMMSCs. b, c DPSCs expressed CBS, CSE, and MPST assessed by flow cytometry (b) and immunofluorescence staining (c). d DPSCs produced H2S in the culture supernatant. Knockdown of CBS or CSE, but not MPST, by siRNA decreased the production of H2S by DPSCs, while NaHS treatment could upregulate H2S concentration. **P < 0.01; scale bar: 20 μm. All experimental data were verified in at least three independent experiments
H2S maintained DPSC function
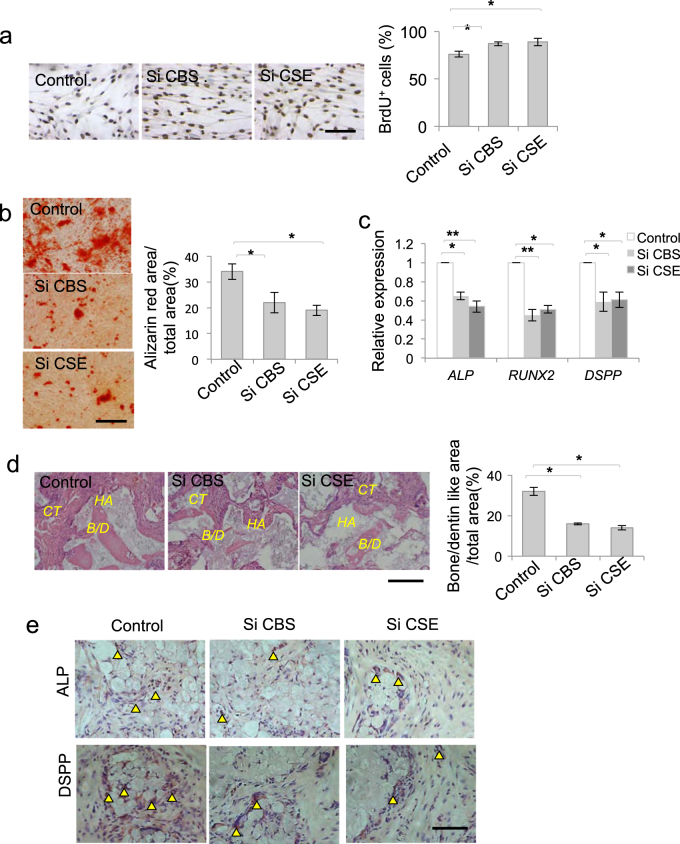
To analyze the role of H2S on DPSC function, we used CBS or CSE siRNA to treat DPSCs and found that DPSC proliferation was increased after CBS or CSE siRNA treatment (Fig. 2a and Supplementary Figure 2a). CBS or CSE siRNA treatment inhibited DPSC osteogenic/dentinogenic differentiation, accompanied by decreased expression of osteogenesis-related genes RUNX2 and ALP, and dentin sialophosphoprotein (DSPP) (Fig. 2b, c). CSE, but not CBS, siRNA treatment decreased the adipogenic differentiation of DPSCs, along with decreased expression of adipogenesis-related genes LPL and PPARγ (Supplementary Figure 2b, 2c). When DPSCs were subcutaneously implanted into immunocompromised mice using hydroxyapatite/tricalcium phosphate (HA/TCP) as a carrier, bone/dentin-like hard tissue formation were observed. After CBS or CSE siRNA treatment, bone/dentin-like hard tissue formation of DPSCs was decreased (Fig. 2d). Moreover, ALP-positive and DSPP-positive cells were detected in the tissues formed by DPSC transplantation in vivo, while ALP-positive and DSPP-positive cells were decreased after CBS or CSE siRNA treatment (Fig. 2e). These data implied that DPSCs could differentiate into functional odontoblast-like cells and osteoblast in vitro and in vivo, which was attenuated by H2S deficiency.
Figure 2. H2S was required to maintain homeostasis of DPSCs.
a DPSC proliferation was increased after CBS or CSE siRNA treatment, as assessed by BrdU proliferation assay (a). b, c CBS or CSE siRNA treatment decreased DPSC osteogenic/dentiogenic differentiation, as assessed by Alizarin red staining, with decreased expression of RUNX2, ALP, and DSPP analyzed by qPCR. d When DPSCs were subcutaneously implanted into immunocompromised mice using HA/TCP as a carrier, bone/dentin-like hard tissue formation of DPSCs was decreased after CBS or CSE siRNA treatment. e ALP-positive and DSPP-positive cells were detected in the tissue formed by DPSC in vivo transplantation. CBS or CSE siRNA treatment decreased ALP-positive and DSPP-positive cells compared to the control ones. *P < 0.05, **P < 0.01; scale bar: 100 μm (a, b, d), 50 μm (e). HA HA/TCP, CT connect tissue, B/D bone/dentin. All experimental data were verified in at least three independent experiments
H2S activated TRPV1-mediated Ca2+ influx
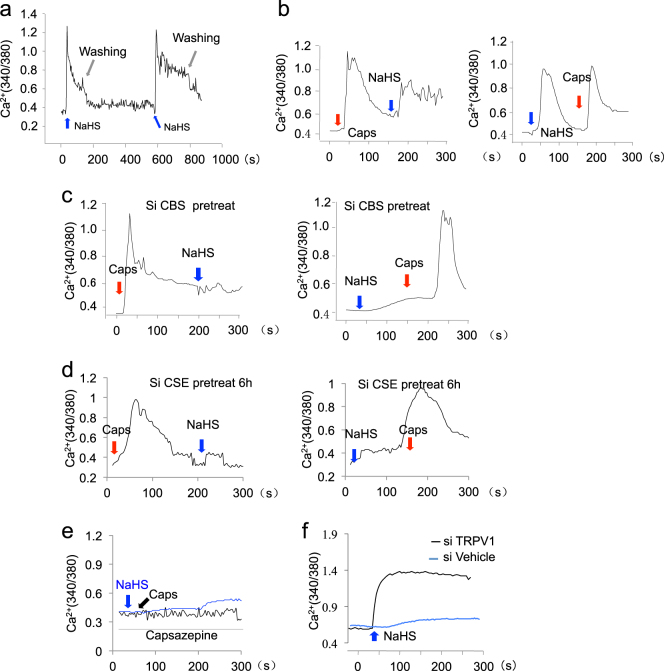
Emerging evidences showed that H2S plays crucial role in maintaining calcium homeostasis in endothelial cells, cardiomyocytes, and MSCs18. Here we found that H2S donor NaHS treatment induced Ca2+ influx in DPSCs (Fig. 3a). The TRPV subfamily is Ca2+-permeable cation channel, which could be activated by different stimuli. Our previous study revealed that combined TRPV3, TRPV6, and TRPM4 Ca2+ channels mediated Ca2+ influx-promoted osteogenic differentiation of BMMSCs6. First, we used TRPV3, TRPV6, and TRPM4 siRNAs individually or in combination to treat DPSCs and found that inhibition of these Ca2+ channels could not block NaHS-induced Ca2+ influx (Supplementary Figure 3). Studies found that TRPV1 was expressed in osteoblasts and was important in maintaining osteoblast differentiation ability19. Here we revealed that capsaicin, which acts on the TRPV1 receptor in nociceptive neurons to activate calcium influx, induced Ca2+ influx in DPSCs. NaHS treatment induced similar Ca2+ influx as capsaicin in DPSCs (Fig. 3b). When DPSCs were pretreated with CBS or CSE siRNA treatment, attenuated NaHS, but not capsaicin, triggered Ca2+ influx (Fig. 3c, d). When DPSCs were pretreated with capsazepine, a competitive antagonist of TRPV1, the Ca2+ influx induced both by NaHS and capsaicin were blocked, which indicated that TRPV1 channels play a crucial role in mediated H2S-induced Ca2+ influx in DPSCs (Fig. 3e). Furthermore, we found that knockdown of TRPV1 by siRNA blocked NaHS-triggered Ca2+ influx in DPSCs (Fig. 3f), verifying that H2S stimulates TRPV1 channel to trigger Ca2+ influx in DPSCs.
Figure 3. H2S activated TRPV1-mediated Ca2+ influx.
a NaHS treatment could induce Ca2+ influx in DPSCs, as assessed by Ca2+ levels using the ratio of emission in response to excitation at 340 and 380 nm on an Olympus Optical IX71 microscope. b NaHS treatment could induce Ca2+ influx in DPSCs as capsaicin treatment. c Capsaicin, but not NaHS, could induce Ca2+ influx in CBS siRNA-pre-treated DPSCs. d Capsaicin, but not NaHS, could induce Ca2+ influx in CSE siRNA-pre-treated DPSCs. e Both capsaicin-induced and NaHS-induced Ca2+ influx was blocked by capsazepine treatment. f Knockdown of TRPV1 by siRNA could partially block the Ca2+ influx induced by NaHS treatment. Caps capsaicin. All experimental data were verified in at least three independent experiments
TRPV1-mediated Ca2+ influx was required to maintain DPSC function
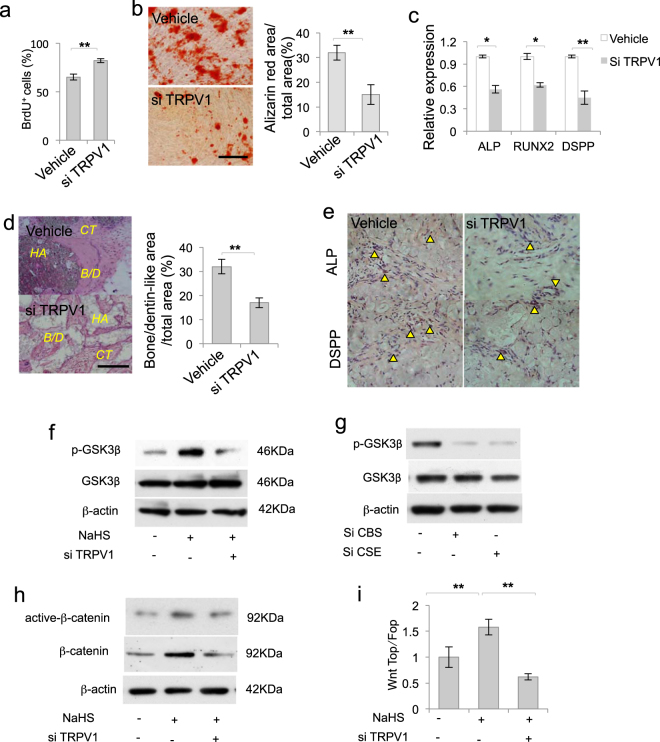
To verify the role of TRPV1-mediated Ca2+ influx in DPSCs, we analyzed DPSC function after TRPV1 knockdown and revealed that DPSC proliferation was increased after TRPV1 knockdown (Fig. 4a). DPSC osteogenic/dentinogenic differentiation was decreased, along with downregulated expression of RUNX2, ALP, and DPSS after TRPV1 knockdown by siRNA (Fig. 4b, c). Moreover the in vivo bone/dentin-like hard tissue formed by DPSC transplantation also decreased after TRPV1 knockdown, which was consistent with H2S deficiency. (Fig. 4d). Consistently, less ALP-positive and DSPP-positive cells were detected in the tissues formed by DPSC transplantation after TRPV1 siRNA treatment (Fig. 4e). These studies indicate that TRPV1-mediated Ca2+ influx play crucial roles in maintaining DPSC function.
Figure 4. TRPV1-mediated Ca2+ influx was required to maintain DPSC function.
a DPSC proliferation was increased after TRPV1 knockdown, as assessed by BrdU proliferation assay. b, c DPSC osteogenic/dentinogenic differentiation was decreased after TRPV1 knockdown, as assessed by Alizarin red staining, with decreased expression of RUNX2, ALP, and DSPP analyzed by qPCR. d Bone/dentin-like hard tissue formation of DPSCs in vivo decreased after TRPV1 knockdown by siRNA. e ALP-positive and DSPP-positive cells detected in tissues formed by DPSC transplantation were decreased after TRPV1 knockdown. f NaHS treatment upregulated p-GSK3β expression, and knockdown of TRPV1 by siRNA partially blocked NaHS-induced GSK3β phosphorylation. g CBS or CSE siRNA treatment downregulated p-GSK3β expression. h H2S donor NaHS treatment activated β-catenin expression, which was partially blocked after TRPV1 knockdown. i NaHS treatment enhanced TOPflash activity in DPSCs, which was blocked after TRPV1 siRNA treatment, as assessed by luciferase activity. NaHS treatment failed to alter FOPflash activity. HA HA/TCP, CT connect tissue, B/D bone/dentin. *P < 0.05, **P < 0.01; scale bar: 100 μm (b, d), 50 μm (e). All experimental data were verified in at least three independent experiments
H2S activated β-catenin signaling
Next, we analyzed the underlying mechanisms of how Ca2+ influx regulated DPSC function. We used H2S donor NaHS to treat DPSCs and found that NaHS treatment elevated phosphorylation of GSK3β, while knockdown of TRPV1 by siRNA blocked NaHS-induced phosphorylated-GSK3β (p-GSK3β) elevation (Fig. 4f). Consistently, we revealed that CBS or CSE siRNA treatment downregulated p-GSK3β expression (Fig. 4g). β-Catenin signaling plays crucial roles in determining stem cell lineage differentiation1. We found that H2S donor NaHS treatment increased β-catenin and active-β-catenin expression, which was blocked by TRPV1 knockdown (Fig. 4h). Next, we investigated whether H2S affected β-catenin transcriptional activity by using luciferase assay analyzed by the ratio of TOPflash and FOPflash. The results showed that H2S donor NaHS treatment increased pathway activity. TRPV1 siRNA treatment attenuated NaHS-induced pathway activity elevation (Fig. 4i). These data implied that H2S gasotransmitter pathway activated β-catenin signaling in DPSCs.
H2S-mediated β-catenin signaling contributed to dentin formation
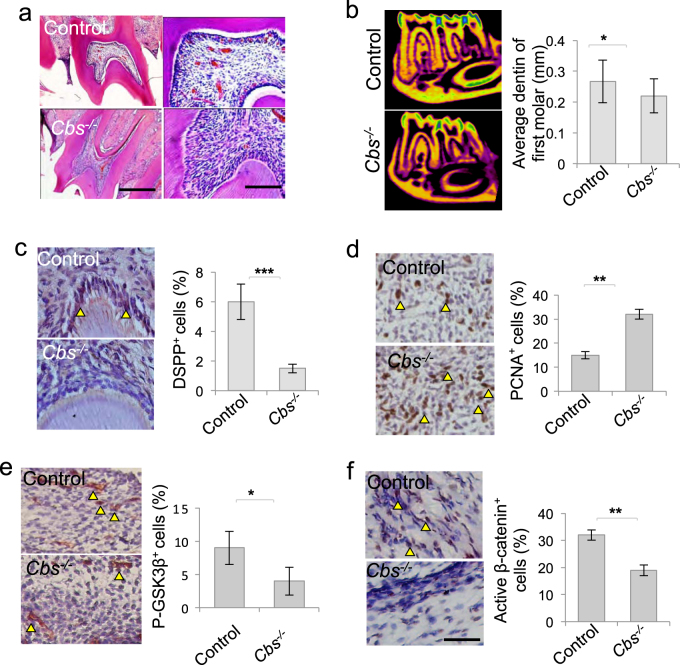
In order to verify the role of β-catenin cascade in maintaining dental pulp homeostasis, we employed H2S-deficient (Cbs−/−) mice and found that Cbs−/− mice dental pulp showed disturbed pattern with increased cell infiltration than control ones (Fig. 5a). The average thickness of dentin was decreased in Cbs−/− mice with decreased DSPP-positive cells in the dental pulp compared to control ones (Fig. 5b, c). Furthermore, proliferative PCNA+ cells increased in Cbs−/− mice dental pulp (Fig. 5d), while p-GSK3β+ and active-β-catenin+ cells decreased in Cbs−/− mice dental pulp (Fig. 5e, f), which to some extent verifies that H2S-mediated β-catenin signaling is important in maintaining dentin formation and DPSC function in vivo.
Figure 5. β-Catenin signaling were decreased in Cbs−/− dental pulp.
a Cbs−/− mice dental pulp showed more cells than control ones. b The average dentin thickness of mandible first molar is less in Cbs−/− mice than control ones, as assessed by micro-CT. c DSPP+ cells decreased in Cbs−/− mice dental pulp compared to control ones. d PCNA+ cells increased in Cbs−/− mice dental pulp. e, f p-GSK3β+ and active-β-catenin cells decreased in Cbs−/− mice dental pulp. *P < 0.05, **P < 0.01, ***P < 0.001, scale bar: 200 μm left panel, 100 μm right panel (a), 1 mm (b), 20 μm (c–f). All experimental data were verified in at least three independent experiments
Discussion
H2S regulates several physiological and pathological processes via diverse mechanisms. In this study, we showed that H2S is essential to maintain DPSC osteogenic and dentinogenic differentiation. We used siRNA to knockdown CBS and CSE, the main enzymes that catalyzed H2S production in DPSCs, and found that DPSC proliferation was elevated. There were more PCNA+ cells infiltrated in Cbs−/− mice dental pulp, indicating that H2S is required to restrict aberrant proliferation of stem cells, which is consistent with previous reports15. Furthermore, we found that the dentin thickness in Cbs−/− mice decreased compared to the control ones, which was consistent with the decreased osteogenic and dentinogenic differentiation of H2S deficiency DPSCs in vitro and in vivo. These results expand the physiological roles of H2S in MSCs from bone to dental pulp and dentin homeostasis.
Calcium channels were one of main cellular events that may contribute H2S-mediated molecular responses20,21. The L-type and T-type Ca2+ channels and TRP channels are reported to be the alternative targets of H2S. TRP channels were involved in diverse cellular functions such as gene expression, proliferation, differentiation, migration, and apoptosis. It is reported that H2S triggers TRPV1 to mediate chloride secretion in acute pancreatitis and colon22,23. In this study, we found that H2S donors mimicked the effect of capsaicin in the neurons, leading to the Ca2+ flux entry in DPSCs. Ca2+ influx entry then triggered GSK3β/β-catenin cascade to regulate DPSC proliferation and differentiation. Furthermore, the study revealed that TRPV1 was essential to maintain DPSC proliferation and differentiation. When TRPV1 was knocked down by siRNA in DPSCs, the proliferation of DPSCs was elevated and osteogenic/dentinogenic differentiation of DPSCs was inhibited, which firstly implied the role of TRPV1 in maintaining MSC capacity. Our previous study reported that H2S-triggered Ca2+ influx is required to maintain bone and MSC homeostasis. The study revealed that combined TRPV3, TRPV6, and TRPM4 Ca2+ channels mediated Ca2+ influx to promote osteogenic differentiation of BMMSCs6. In this study, we found that the proliferation and differentiation capacity of DPSCs could not be attenuated by TRPV3, TRPV6, and TRPM4 Ca2+ channels blockage. These differences may attribute that DPSCs were derived from the neural crest and mesoderm during development, which were more accessible to differentiate into dentin-like tissue rather than the bone compared to BMMSCs. Functionally, DPSCs also easily differentiated into neuronal and glial cells and acted as neuronal cells24,25. These studies shed light on the different molecular target induced by H2S to illustrate the unique properties of MSCs derived from different tissues
Variety of signal pathways may contribute to that H2S fine-tune its effects on different tissues and cells. H2S may trigger the nuclear translocation of nuclear factor-κB (NF-κB) and affect the activity of numerous kinases including p38 mitogen-activated protein kinase, extracellular signal-regulated kinase, and Akt signaling. H2S is able to promote angiogenesis and vascular remodeling via the PI3K/Akt/survivin axis in endothelial cells by augmenting phosphorylation of ERK and p3820,21. H2S deceases pro-inflammatory genes involved in cardiac ischemic/reperfusion injury via inhibiting the nuclear translocation of NF-κB26. In BMMSCs, H2S-induced Ca2+ influx promoted β-catenin activity via the PKC/ERK pathway to maintain bone homeostasis6. However, we found that H2S treatment failed to alter ERK signaling in DPSCs (data were not shown). We showed that TRPV1-mediated Ca2+ influx entry facilitates the phosphorylation of GSK3β to disrupt the “destruction complex.” β-Catenin accumulates in the cytoplasm and then translocates into the nucleus, which promoted DPSC osteogenic differentiation. H2S deficiency decreased TRPV1-mediated Ca2+ influx entry, leading to decreased phosphorylation of GSK3β. These data indicated that the response of different stem cells to H2S may be regulated by different signaling pathways.
Cytoplasmic β-catenin was degraded, resulting in DPSC osteogenic differentiation deficiency. In vivo, Cbs−/− mice dental pulp showed elevated cell infiltration and proliferation and decreased GSK3β/β-catenin signaling. Moreover, the dentin formation of Cbs−/− mice was less than control ones. These data suggest that H2S-mediated GSK3β/β-catenin cascade is important to maintain DPSC function and dental pulp homeostasis. These results shed light on the different biology and mechanism of H2S targeting on different MSCs and tissue homeostasis.
Conclusion
Our study reveals that the levels of H2S are essential to maintain Ca2+ homeostasis via TRPV1 Ca2+ channel in DPSCs. H2S deficiency results in Ca2+ influx/GSK3β/β-catenin cascade response in DPSCs, leading to differentiation deficiency of DPSCs and dentin formation disorders (Supplementary Figure 4).
Materials and methods
Mice
C57BL/6 (JAX #000664), B6.129P2-Cbstm1Unc/J (JAX #002461) Cbs+/− mice were purchased from Jackson lab. Female immunocompromised mice (beige nude/nude XIDIII) were purchased from Harlan Laboratories. All animal experiments were performed under institutionally approved protocols for the use of animal research at the University of Pennsylvania (IACUC# 805478) and Peking University (#LA2012-65).
Antibodies and chemicals
Antibodies
Anti-active-β-catenin and β-catenin antibodies were purchased from Millipore (Temecula, CA, USA). Anti-CD105-PE, anti-CD146-PE, anti-CD90-PE, anti-CD34-PE, and anti-CD45-PE antibodies were purchased from BD Bioscience (San Jose, CA, USA). Unconjugated anti-GSK3β and anti-phospho-GSK3β antibodies were purchased from Cell Signaling Inc. (San Francisco, CA, USA). Anti-β-actin antibody was purchased from Sigma-Aldrich Corporation (St. Louis, MO, USA). Unconjugated anti-CBS, CSE, DSPP (DSP) and TRPV1 were purchased from Abcam Inc. (Cambridge, MA, USA).
Chemicals
siRNA for CBS (hydroxylamine), CSE (dl-propargylglycine), TRPV1, TRPV3, TRPV6, and TRPM4 were purchased from Santa Cruz. H2S donor NaHS was purchased from Sigma-Aldrich Corporation (St. Louis, MO, USA). Capsaicin and capsazepine were purchased from EMD Millipore (Billerica, MA, USA). The concentrations of capsaicin and capsazepine used for the treatment were 1 μM.
Isolation and culture of human bone DPSCs
Dental pulp from human extracted wisdom teeth were gently separated, minced, and digested with 2 mg/ml collagenase type I (Worthington Biochemical, Freehold, NJ, USA) and 4 mg/ml dispase II (Roche Diagnostics, Indianapolis, IN, USA) in phosphate-buffered saline for 1 h at 37 °C. Then, the cells were passed through a 70-μm strainer (BD Biosciences, Franklin Lakes, NJ, USA) to get single cells. The single-cell suspensions were cultured with α-minimum essential medium (α-MEM) supplemented with 15% fetal bovine serum (FBS), 100 mM l-ascorbic acid-2-phosphate, 2 mM l-glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin and passaged, as previously reported27.
Human bone marrow MSCs BMMSC culture
Human bone marrow aspirates from healthy adult donors (20–35 years of age) were purchased from AllCells (Emeryville, CA, USA). Human bone marrow progenitor cells were enriched using fresh human bone marrow by RosetteSep® Human Bone Marrow Progenitor Cell Pre-Enrichment Cocktail (STEMCELL Technologies, Vancouver, BC, Canada) following the manufacturer’s instructions. The enriched single-cell suspensions were cultured with α-MEM supplemented with 15% FBS, 100 mM l-ascorbic acid-2-phosphate, 2 mM l-glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin and passaged, as previously reported27.
Cell proliferation assay
DPSCs were passaged to a 4-well chamber slide (Nunc, 1 × 104 per well) and incubated for 12 h at 37 °C in a 5% CO2 condition. Then, the cells were assayed for bromodeoxyuridine (BrdU) incorporation, as per the manufacturer’s instruction. BrdU+ and total cell numbers were counted in five images per subject. The BrdU+ cells were indicated as a percentage of BrdU+ cells and the total cell number.
DPSC-mediated hard tissue formation in vivo
Approximately 4.0 × 106 DPSCs were mixed with HA/TCP ceramic particles (40 mg, Zimmer Inc., Warsaw, IN, USA) as a carrier and subcutaneously implanted into the dorsal surface of 8- to 10-week-old immunocompromised mice (n = 4). At 8 weeks post-implantation, the implants were harvested, fixed in 4% paraformaldehyde, and then decalcified with 5% EDTA, followed by paraffin embedding. The 5 μm paraffin sections were stained with hematoxylin and eosin and analyzed by NIH ImageJ. The newly formed mineralized tissue area in each field was calculated and shown as a percentage of the total tissue area.
Osteogenic/dentinogenic differentiation assay
Osteogenic/dentinogenic differentiation is performed as described in the Supplementary materials.
Adipogenic differentiation assay
Adipogenic differentiation is performed as described in the Supplementary materials.
Immunofluorescent staining
DPSCs were seeded on a 4-well chamber slide and incubated for 12 h at 37 °C in a 5% CO2 condition. The detailed method of immunofluorescent staining is described in the Supplementary materials.
Immunohistochemical staining
Immunohistochemical staining is performed as described in the Supplementary materials.
Western blot
Western blot is performed as described in the Supplementary materials.
Quantitative PCR
For quantitative PCR (qPCR) analysis, total RNA was extracted with RNeasy Mini kit (Qiagen, Valencia, CA, USA), and complementary DNA was prepared using a SuperScript® III Reverse Transcriptase (RT) kit (Invitrogen). qPCR was carried out using SYBR® Green Supermix (Bio-Rad) on a Bio-Rad CFX96 Real Time System, as indicated by the manufacturer’s protocol.
Measurement of Ca2+ image
DPSCs (1 × 105) were seeded onto 60 mm culture dishes and cultured for 24 h at 37 °C in 5% CO2. Then, the cells were loaded with fura-2 AM (Invitrogen) and incubated for 1 h at 37 °C in 5% CO2 in the dark. Ca2+ levels were measured from the ratio of emission in response to excitation at 340 and 380 nm on an Olympus Optical IX71 microscope.
Measurement of H2S
Cell culture supernatants were mixed with 0.25 ml Zn acetate (1%) and 0.45 ml water for 10 min at room temperature. TCA (10%; 0.25 μl) was then added and centrifuged (14,000 × g for 10 min at 4 °C). The supernatant was then collected and mixed with N,N-dimethyl-p-phenylenediamine sulfate (20 μM) in 1.2 M HCl and FeCl3 (30 μM) in 1.2 mol/l HCl. After 20 min, absorbance was measured at 650 nm.
In vitro transcription assays
β-Catenin-mediated transcriptional activity was measured by the luciferase activity by using TOPflash and FOPflash described previously6. DPSCs (1 × 105) were seeded onto a 24-well culture plate and cultured for 24 h. TOPflash or FOPflash (10 ng/well) and renilla luciferase (4 ng/well) were transfected into DPSCs using a Lipofectamine™ RNAiMAX kit (Invitrogen). After 15 h, the plates were assayed for firefly luciferase and renilla luciferase activities using the Dual-Luciferase® Reporter Assay System (Promega).
Micro computed tomography
The mandible of mice was scanned with a micro-computed tomography (micro-CT) system (Inveon MMCT, Berlin, Germany) at 80 kV, 500 µA, and an image voxel size of 18 µm. The hemi-mandibles were excised (n = 6 per group). The hemi-mandible samples were directed parallel to the occlusal plane and scanned for 2.70 mm. The thickness of the first molar was measured with the DataViewer software (ver. 1.4.3; SkyScan).
Statistical analysis
P values were analyzed from two-tailed Student’s t test for the difference between two groups or one-way analysis of variance to compare the difference from more than two groups using the SPSS 18.0 software. P values <0.05 were considered significant.
Supplementary information
Acknowledgements
This work was supported by Supported by Beijing Natural Science Foundation (7182182), the Young Elite Scientist Sponsorship Program by CAST (2017QNRC001), International Science & Technology Cooperation Program of China No. 2015DFB30040 (to Y.Z.), the National Science Foundations of China, No. 81470717 (to Y.Z.), 81600865 (to R.Y.), and 81570953 (to Y.S. Z.).
Authors' contributions
R.Y.: conception and design, collection and assembly of data, data analysis and interpretation, final approval of manuscript; Y.L., T.Y.: collection data and interpretation, final approval of manuscript. Y.H.Z., Y.S. Z., S.S.: critically edited the manuscript, financial support, final approval of manuscript.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
AE Sayan.
Edited by A.E. Sayan
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
7/10/2019
Due to a technical error, content intended for publication in Volume 4 (2018) published in Volume 5 (2019). The content has been moved into the correct volume, and the citation information was updated accordingly.
Contributor Information
Yongsheng Zhou, Phone: +86-10-8219-5370, Email: kqzhouysh@hsc.pku.edu.cn.
Yanheng Zhou, Phone: +86-10-8219-5527, Email: yanhengzhou@vip.163.com.
Supplementary information
The online version of this article (10.1038/s41420-018-0071-4) contains supplementary material, which is available to authorized users.
References
- 1.Li J, Pei M. Cell senescence: a challenge in cartilage engineering and regeneration. Tissue Eng. Part B. 2012;18:270–287. doi: 10.1089/ten.teb.2011.0583. [DOI] [PubMed] [Google Scholar]
- 2.Yang R, et al. Hydrogen sulfide promotes Tet1- and Tet2-mediated Foxp3 demethylation to drive regulatory T cell differentiation and maintain immune homeostasis. Immunity. 2015;43:251–263. doi: 10.1016/j.immuni.2015.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Oh SA, Li MO. TETs link hydrogen sulfide to immune tolerance. Immunity. 2015;43:211–213. doi: 10.1016/j.immuni.2015.08.001. [DOI] [PubMed] [Google Scholar]
- 4.Wang Z, et al. l-Cysteine promotes the proliferation and differentiation of neural stem cells via the CBS/H(2)S pathway. Neuroscience. 2013;237:106–117. doi: 10.1016/j.neuroscience.2012.12.057. [DOI] [PubMed] [Google Scholar]
- 5.Zhao Y, Wei H, Kong G, Shim W, Zhang G. Hydrogen sulfide augments the proliferation and survival of human induced pluripotent stem cell-derived mesenchymal stromal cells through inhibition of BKCa. Cytotherapy. 2013;15:1395–1405. doi: 10.1016/j.jcyt.2013.06.004. [DOI] [PubMed] [Google Scholar]
- 6.Liu Yi, Yang Ruili, Liu Xibao, Zhou Yu, Qu Cunye, Kikuiri Takashi, Wang Songlin, Zandi Ebrahim, Du Junbao, Ambudkar Indu S., Shi Songtao. Hydrogen Sulfide Maintains Mesenchymal Stem Cell Function and Bone Homeostasis via Regulation of Ca2+ Channel Sulfhydration. Cell Stem Cell. 2014;15(1):66–78. doi: 10.1016/j.stem.2014.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Grassi Francesco, Tyagi Abdul Malik, Calvert John W, Gambari Laura, Walker Lindsey D, Yu Mingcan, Robinson Jerid, Li Jau-Yi, Lisignoli Gina, Vaccaro Chiara, Adams Jonathan, Pacifici Roberto. Hydrogen Sulfide Is a Novel Regulator of Bone Formation Implicated in the Bone Loss Induced by Estrogen Deficiency. Journal of Bone and Mineral Research. 2015;31(5):949–963. doi: 10.1002/jbmr.2757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Aas JA, Paster BJ, Stokes LN, Olsen I, Dewhirst FE. Defining the normal bacterial flora of the oral cavity. J. Clin. Microbiol. 2005;43:5721–5732. doi: 10.1128/JCM.43.11.5721-5732.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhang JH, Dong Z, Chu L. Hydrogen sulfide induces apoptosis in human periodontium cells. J. Periodontal Res. 2010;45:71–78. doi: 10.1111/j.1600-0765.2009.01202.x. [DOI] [PubMed] [Google Scholar]
- 10.Miura M, et al. SHED: stem cells from human exfoliated deciduous teeth. Proc. Natl. Acad. Sci. USA. 2003;100:5807–5812. doi: 10.1073/pnas.0937635100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ishkitiev N, et al. Hydrogen sulfide increases hepatic differentiation in tooth–pulp stem cells. J. Breath Res. 2012;6:017103. doi: 10.1088/1752-7155/6/1/017103. [DOI] [PubMed] [Google Scholar]
- 12.Okada M, et al. Hydrogen sulphide increases hepatic differentiation of human tooth–pulp stem cells compared with human bone marrow stem cells. Int. Endod. J. 2014;47:1142–1150. doi: 10.1111/iej.12262. [DOI] [PubMed] [Google Scholar]
- 13.Kobayashi C, et al. Hydrogen sulfide causes apoptosis in human pulp stem cells. J. Endod. 2011;37:479–484. doi: 10.1016/j.joen.2011.01.017. [DOI] [PubMed] [Google Scholar]
- 14.Liu F, et al. Force-induced H2S by PDLSCs modifies osteoclastic activity during tooth movement. J. Dent. Res. 2017;96:694–702. doi: 10.1177/0022034517690388. [DOI] [PubMed] [Google Scholar]
- 15.Su Y, et al. Physiologic levels of endogenous hydrogen sulfide maintain the proliferation and differentiation capacity of periodontal ligament stem cells. J. Periodontol. 2015;86:1276–1286. doi: 10.1902/jop.2015.150240. [DOI] [PubMed] [Google Scholar]
- 16.Pu, H. & Hua, Y. Hydrogen sulfide regulates bone remodeling and promotes orthodontic tooth movement. Mol. Med. Rep. 6, 9415–9422 (2017). [DOI] [PMC free article] [PubMed]
- 17.Aykan A, et al. The effects of hydrogen sulfide on adipocyte viability in human adipocyte and adipocyte-derived mesenchymal stem cell cultures under ischemic conditions. Ann. Plast. Surg. 2015;75:657–665. doi: 10.1097/SAP.0000000000000595. [DOI] [PubMed] [Google Scholar]
- 18.Munaron L, Avanzato D, Moccia F, Mancardi D. Hydrogen sulfide as a regulator of calcium channels. Cell Calcium. 2013;53:77–84. doi: 10.1016/j.ceca.2012.07.001. [DOI] [PubMed] [Google Scholar]
- 19.He LH, et al. TRPV1 deletion impaired fracture healing and inhibited osteoclast and osteoblast differentiation. Sci. Rep. 2017;7:42385. doi: 10.1038/srep42385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wang R. Physiological implications of hydrogen sulfide: a whiff exploration that blossomed. Physiol. Rev. 2012;92:791–896. doi: 10.1152/physrev.00017.2011. [DOI] [PubMed] [Google Scholar]
- 21.Li L, Rose P, Moore PK. Hydrogen sulfide and cell signaling. Annu. Rev. Pharmacol. Toxicol. 2011;51:169–187. doi: 10.1146/annurev-pharmtox-010510-100505. [DOI] [PubMed] [Google Scholar]
- 22.Xu GY, et al. The endogenous hydrogen sulfide producing enzyme cystathionine-beta synthase contributes to visceral hypersensitivity in a rat model of irritable bowel syndrome. Mol. Pain. 2009;5:44. doi: 10.1186/1744-8069-5-44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nishimura S, et al. Hydrogen sulfide as a novel mediator for pancreatic pain in rodents. Gut. 2009;58:762–770. doi: 10.1136/gut.2008.151910. [DOI] [PubMed] [Google Scholar]
- 24.Yang R, Yu T, Zhou Y. Interplay between craniofacial stem cells and immune stimulus. Stem Cell Res. Ther. 2017;8:147. doi: 10.1186/s13287-017-0607-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gronthos S, Mankani M, Brahim J, Robey PG, Shi S. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc. Natl. Acad. Sci. USA. 2000;97:13625–13630. doi: 10.1073/pnas.240309797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Miller TW, et al. Hydrogen sulfide is an endogenous potentiator of T cell activation. J. Biol. Chem. 2012;287:4211–4221. doi: 10.1074/jbc.M111.307819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shi S, et al. Bone formation by human postnatal bone marrow stromal stem cells is enhanced by telomerase expression. Nat. Biotechnol. 2002;20:587–591. doi: 10.1038/nbt0602-587. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.