Abstract
The notion that one common pathogenic pathway could account for the various clinically distinguishable, typically late-onset neurodegenerative diseases might appear unlikely given the plethora of diverse primary causes of neurodegeneration. On the contrary, an autoinflammatory pathogenic mechanism allows diverse genetic and environmental factors to converge into a common chain of causality. Inflammation has long been known to correlate with neurodegeneration. Until recently this relationship was seen as one of consequence rather than cause—with inflammatory cells and events acting to ‘clean up the mess’ after neurological injury. This explanation is demonstrably inadequate and it is now clear that inflammation is at the very least, rate-limiting for neurodegeneration (and more likely, a principal underlying cause in most if not all neurodegenerative diseases), protective in its initial acute phase, but pernicious in its latter chronic phase.
Introduction
Research to define the aetiology of neurodegeneration has been dominated for decades by the hypothesis that aggregation of amyloid-β is the principal basis of Alzheimer’s disease and related conditions. The disappointing results (1,2) of recent clinical trials of interventions targeting amyloid-β aggregation have shifted the focus to alternative hypotheses. One new hypothesis invokes inflammation as a causal upstream driver of neurodegeneration. Inflammation is activated by a diverse array of stimuli and typically correlates with neurodegeneration, but to date has largely been viewed as protective, rather than disease-causing. Recent genetic evidence strongly implicates inflammation as the primary initiating lesion. Various inflammatory response components and their regulators are common amongst identified genetic susceptibility loci. Inflammatory pathway genes are activated in affected tissues and elevated inflammatory signals occur early in disease onset and precede neurodegeneration, independently of any infectious aetiology. Genetic and pharmacological ablation studies in animal models of several neurodegenerative diseases demonstrate that inflammation is required for pathology. We argue that this evidence is highly compelling for Alzheimer’s and many related conditions, and indeed it seems likely that inflammation will emerge as pivotal in the full range of neurodegenerative diseases. The research effort should now focus on evaluating interventions that target pro-inflammatory mediators and their eliciting factors and regulatory networks.
Systemic Inflammatory Activation
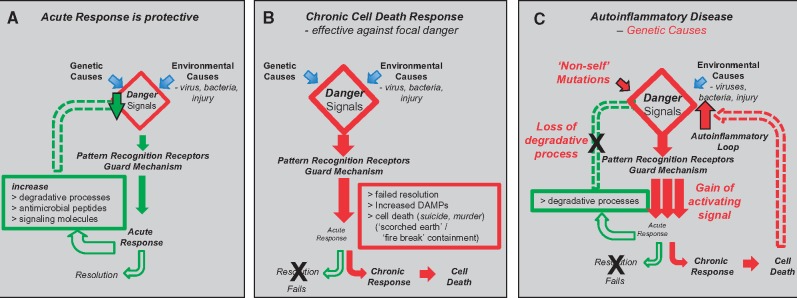
Inflammatory activation provides the body’s front-line defence and quality surveillance system. Surveillance occurs both autonomously within the intracellular environment as well as by cells that patrol the extra-cellular environment for threats, damage and dysfunction. The initial acute inflammatory response is protective—directed toward degrading the threat, repairing the damage, correcting the defect and restoring homeostasis. Persistent activation leads to a chronic response that includes focal cell death as a containment strategy, denying potential pathogens and pre-cancerous cells the ability to replicate and spread (Box 1 and Fig. 1).
Figure 1.
Inflammatory acute and chronic responses and autoinflammatory disease. (A) Acute phase inflammatory response: (i) activated by trigger molecules/danger signals, (ii) produces antimicrobial peptides and signalling molecules (cytokines), (iii) increase degradative processes (including various forms of autophagy and integrated stress response) and (iv) directed towards resolution and restoration of homeostasis. (B) Chronic phase inflammatory response: (i) failed resolution by acute phase and (ii) focal cell death (autonomous and non-cell autonomous) and subsequent containment and resolution as ‘pesudo-tumour’ or granuloma. (C) Autoinflammatory disease: (i) genetic lesions activate inflammation by initiating or amplifying innate immune pathways by a number of mechanisms, including the direct induction of danger signals, or the induction of downstream pathways independently of pattern recognition receptors (PRRs) or as an exaggerated response to PRRs; (ii) cell death products also add to danger signals, dependant upon means of cell death; (iii) two functionally distinct forms of cell death—non-lytic (apoptosis—typical in development) and lytic (e.g. necroptosis, pyroptosis, ferroptosis), differ in release of trigger molecules (damage associated molecular patterns) by lytic cell death that contribute to danger signal load and perpetuate inflammatory response and its spread; and (iv) environmental trauma (infection, injury) can account for instigation of autoinflammatory loop by sensitizing cells above a threshold required inflammatory activation.
Rapid evolution and high genetic variation is a common property of the host-pathogen arms race, as each side seeks to discover new forms of advantage that can include beneficial symbioses. Variation in resistance and susceptibility therefore promotes competitive fitness in a host population (e.g. sampling and discriminating potentially harmful from beneficial microbes) and a likely reason why some components of the inflammatory response are themselves pathogen restriction factors (a noteworthy example is Ref(2)P that is a viral restriction factor in Drosophila (3) while mutations in its human orthologue, known as Sequestosome 1 or p62, are a cause of familial ALS) (4). This is one of now many genes linked with human neurodegenerative disease genes that have roles in the activation or resolution of inflammation (Tables 1 and 2).
Table 1.
Human neurodegenerative disease genes with roles in inflammatory activation
| Gene | Disease | Role in inflammation | References |
|---|---|---|---|
| Disease causing mutations | |||
| ADAR1 | Aicardi-Goutieres syndrome | RNA modification (‘non-self’ to ‘self’) | (5,6) |
| Bilateral striatal necrosis | (5,6) | ||
| MDA5 (IFIH1) | Aicardi-Goutieres syndrome | RNA ‘non-self’ Pattern Recognition Receptor | (5,6) |
| RNAseH subunits | Aicardi-Goutieres syndrome | RNA/DNA hybrid resolution, induce cGAS/STING | (7) |
| SamHD1 | Aicardi-Goutieres syndrome | Retroviral restriction factor | (8) |
| TREX | Aicardi-Goutieres syndrome | Degrades DNA to prevent spontaneous inflammatory disease | (9) |
| TBK1 | Amyotrophic lateral sclerosis | Transduction of TLR signal with optineurin | (10) |
| Optineurin | Amyotrophic lateral sclerosis | Role in TBK1 signal transduction | (11) |
| P62 (sequestosome 1) | Amyotrophic lateral sclerosis | Ablation prevents mitophagy, enhances inflammasome activation | (4) |
| Progranulin | Amyotrophic lateral sclerosis | TLR9 co-factor—deficiency promotes microglial pruning | (12) |
| TDP43 | Amyotrophic lateral sclerosis | RNA Stress Granule, granulophagy | (13) |
| FUS | Amyotrophic lateral sclerosis | RNA Stress Granule, granulophagy | (13) |
| VCP | Amyotrophic lateral sclerosis | RNA Stress Granule transport to autophagosome | (14) |
| CHMP2B | Amyotrophic lateral sclerosis | Granulophagy (autophagosome-lysosome fusion) | (14) |
| Profilin-1 | Amyotrophic lateral sclerosis | RNA stress granule dynamics, granulophagy | (15) |
| Amyloid-β | Familial Alzheimer’s disease | Antimicrobial peptide, damage-associated molecular pattern | (16) |
| Tau | Fronto-temporal dementia | Mitochondrial transport, mitophagy | (17) |
| α-synuclein | Parkinsons disease | Mitophagy | (18) |
| PINK | Parkinsons disease | Mitophagy | (18) |
| Parkin | Parkinsons disease | Mitophagy | (18) |
| LRRK2 | Parkinsons disease | Mitophagy | (18) |
| DJ-1 | Parkinsons disease | Mitophagy | (18) |
| GBA | Parkinsons disease | Mitophagy | (18) |
| ATPA13A2 | Parkinsons disease | Mitophagy | (18) |
| EXOSCIII | Ponto-cerebellar hypoplasia | SKIVL (RNA exosome) ‘non-self’ RNA degradation | (19) |
| TSEN2, 15, 34, 54 | Ponto-cerebellar hypoplasia | tRNA splicing endonucleases | (20,21) |
| TBC1D23 | Ponto-cerebellar hypoplasia | between XBP1 and MYD88, TRIF in TLR4-NFKB1 pathway | (22) |
| Risk-factor alleles | |||
| PLCG2 | Alzheimer’s disease | Phospholipase Cγ2 ⋙ signal transduction | (23) |
| TREM2 | Alzheimer’s disease | Microglial protein ⋙ ApoE4 | (24) |
| APOE | Alzheimer’s disease | Ligand for TREM2? | (25) |
| TOMM40 | Alzheimer’s disease | Mitochondrial unfolded protein response? ⋙ mitophagy | (26,27) |
| IL-33 | Alzheimer’s disease | Cytokine signals | (28) |
| Glucocerebrosidase | Parkinson’s disease | Diminished lysosome function ⋙ autophagy | (29) |
| Ataxin2 | Amyotrophic lateral sclerosis | RNA Stress Granule dynamics ⋙ granulophagy | (30) |
Table 2.
Inflammatory response pathway genes required for symptoms in animal models of human neurodegenerative diseases
| Gene/target | Disease model | References |
|---|---|---|
| Toll-related receptors | Drosophila model of expanded CAG repeat diseases | (31) |
| Toll-Like Receptor 4 | mouse model of amyotrophic lateral sclerosis (SOD1) | (32) |
| MyD88 | mouse model of spino-cerebellar ataxia (SCA6) | (33) |
| cGAS, STING | rat model of ataxia telangiactasia | (34) |
| relish (NFκB) | Drosophila model of ataxia telangiactasia | (35) |
| Ref(2)P (p62) | Drosophila model of polyglutamine diseases | (36) |
| p62 | zebrafish model of amyotrophic lateral sclerosis (C9orf72 repeat RNA) | (37) |
| Toll-Like Receptor 2, 3, 4 | mouse model of Huntington’s disease | (38) |
| HMGB1 | rat model of Parkinson’s disease | (39) |
| TREM2 | mouse model of tau fronto-temporal dementia | (40) |
| eIF-2α | mouse model of tau fronto-temporal dementia | (41) |
| eIF-2α | mouse model of prion disease | (41) |
| eIF-2α | mouse model of traumatic brain injury | (42) |
| eIF-2α | Drosophila model of amyotrophic lateral sclerosis (TDP-43) | (43) |
Autoinflammatory diseases were first recognized twenty years ago and are defined as the presence of acute or chronic inflammation without the high-titer autoantibodies or antigen-specific T cells seen in classic autoimmune diseases, and without the necessity for infection. They include various monogenic disorders that have revealed new regulatory pathways in the inflammatory response and its resolution (44). Systemic autoinflammatory diseases are typically episodic, such that individuals carrying genetic causes of these diseases can go for extensive periods without symptoms. The likely explanation is that incidental exposure to an environmental ‘second-hit’ to elicit an inflammatory response is required for at-risk individuals to develop clinical manifestations. Geographic variation in disease-susceptibility alleles is also consistent with the possibility of localized selective environmental factor(s) (e.g. familial Mediterranean fever).
Autoinflammatory Hallmarks in Neurodegeneration
Genetic lesions in components of the inflammatory response are the origin of inherited autoinflammatory diseases—indeed the characterization of these genes has been a major source of discovery and definition of the innate inflammatory pathways (44). While common pathways can be involved, the diseases exhibit distinctive symptoms and tissue sensitivity, indicating that different cell lineages have diverse rate-limiting components in the underlying pathways. Lessons from systemic autoinflammatory diseases indicate that a plausible mechanism for neuronal cell death in the neurodegenerative diseases is recurrent or chronic inflammation, which mediates cell death after a transition from a protective acute inflammatory response (44,45). It is noteworthy in this regard that amyloid deposition is not unique to neurodegeneration but is also associated with at least some of the systemic autoinflammatory diseases (e.g. serum amyloid A deposits in FMF). Consistent with this notion, amyloid-β deposition has recently been shown in a mouse model of AD to be mediated by the RAGE (receptor of advanced glycation end-products) pattern recognition receptor of the innate surveillance system (46). Furthermore, a functional role for amyloid-β peptide in the innate inflammatory response is evident in its ability to protect against disseminating microbial infection in mouse and worm models of Alzheimer’s disease (16).
A direct connection between systemic autoinflammatory disease and neurodegenerative disease has recently been made. Specific mutations in the PLCG2 gene cause two distinct systemic autoimmune (PLAID) and autoinflammatory (APLAID) phenotypes (47,48). Remarkably, genetic variation in the PLCG2 gene is also a risk factor for Alzheimer’s disease (23).
While focal at its onset, an unresolved and persistent acute inflammatory response may give rise to feed-forward signals (IL-1β and TNF)—cytokines that are met in adjacent cells by higher endogenous sensitivity to inflammatory activation due to the genetic lesion. This elevated chronic inflammatory response resists the normal resolution phase, leading to events including cell death that typically spread beyond the initial lesion. Such systemic autoinflammatory cycles, including alleviation of neurological systems, have been successfully broken by treating affected individuals with inhibitors of IL-1β or TNF (44).
Inflammation in the central nervous system
Neuroinflammation in the central nervous system (CNS) can arise in response to multiple causes and has both distinct and common elements with its systemic counterpart. Focal traumatic brain injury (TBI) (49) activates an inflammatory response that is protective in its early acute phase—facilitating remodelling and recovery. Indeed general suppression of inflammation in TBI affected individuals is disadvantageous.
While experimental models of TBI are typically sterile, natural instances of brain damage are unlikely to be sterile and therefore, as well as promoting tissue repair, inflammatory activation would be a prophylactic defense against opportunistic infection. The increased risk of neurodegenerative disease in individuals who have survived an episode of sepsis indicates that systemic components of the inflammatory response also contribute to sensitization in the CNS (50). Indeed, gut microbiota influence neurodegeneration in a mouse model of Parkinson’s disease (51) and microbes have been proposed as the common causal agent for AD (52). Pathogens provide a source of multiple trigger/danger molecules (pathogen associated molecular patterns—PAMPs) of widely variant structure. Therefore, like their systemic autoinflammatory disease counterparts, the onset of CNS autoinflammatory neurodegeneration may be facilitated by PAMPs elevating the trigger molecule pool above a threshold for chronic inflammatory activation.
Age is the greatest risk factor for neurodegenerative diseases and indeed inflammation in the brain increases with age (53). Somatic mutation in neurons increases with age and is more prevalent in neurodegenerative disease (54). Such mutations provide a source of truncated proteins that stimulate the unfolded protein response and thereby account for age-dependant increase in inflammatory activation.
Neuroinflammation involves many cell lineages
The neurovascular unit (NVU) is the principal multicellular organizational unit in the CNS (Box 2). Each of the cell types in the NVU has distinct functions. Resident macrophage microglia and recruited peripheral immune cells participate in the inflammatory response. Such observations have contributed to the view that the neuroinflammatory response is simply a process to clear debris and recover homeostasis (55,56). However genetic variants in the microglia protein TREM2 (24) as well as PLCG2 and ABI3 (23) are risk factors for Alzheimer’s disease, indicating that microglia-mediated innate inflammation is rate-limiting for the disease.
However, neurodegeneration is not necessarily a cell-autonomous process. There is ample evidence for multiple cells in the NVU having roles to play in non-cell autonomous neuronal cell death (57). Astrocytes are determinants of disease progression in mouse models of inherited ALS (58). Astrocytes that are activated by microglia, in turn kill neurons (59). Signalling molecules (IL-1α, TNF and C1q) between microglia and astrocytes indicate that the CNS has some unique components and others shared with the systemic inflammatory system. Endothelial cells activate neutrophils to acquire cytotoxic capability (60) however the signals involved are not yet known. Indeed, expression of mutant androgen receptor in muscle cells is able to drive nerve cell degeneration in mouse models of Kennedy’s disease (spino-bulbar muscular atrophy) (61). Cell-cell communication is therefore a typical requirement for neurodegeneration and while the signals involved have not all been discovered, those that are well characterized are typically cytokines and other signals of the innate inflammatory response.
Ischemic stroke is a risk factor for vascular dementia and Alzheimer’s disease (62). Neurovascular changes occur prior to motor neuron degeneration in a mouse ALS model (63) and recently depletion of coagulation factor XII has been shown to ameliorate brain pathology in AD model mice (64). Like many contributors to the innate inflammatory response fibrin has multiple functions. In addition to its beneficial role in clot formation, fibrin attracts cells that help with wound healing. However, like other danger molecules, its persistence leads to chronic inflammatory activation that contributes to axonal damage in neuroinflammatory disease (65). Compartmentalization is another means of harbouring/exposing danger signals. Various forms of ‘lytic’ cell death lead to the release of damage associated molecular patterns (DAMPs/alarmins) including molecules such as HMGB1 and the S100 proteins, as well as amyloid-β, that also acts as an antimicrobial peptide (16). This assortment of DAMPs then bind to multiple PRRs in adjacent cells and in so doing spread the inflammatory activation (66,67). Multiplicity of activation signals is a common feature of this alarm system, presumably to override pathogenic inhibition of any single pathway. It also explains why pharmacological inhibition of a single DAMP signal (such as amyloid-β) is ineffective in preventing inflammatory-mediated cell death (1,2,66).
Inflammation precedes neurodegeneration
Consistent with a causal role, inflammatory activation precedes neuronal cell death with increased circulating cytokines found in presymptomatic Huntington’s disease (68), a finding replicated and extended with increased microglial activation as well as cytokines in premanifest Huntington’s disease carriers (69). In addition, inflammatory genes are activated in the cells destined to undergo neurodegeneration. This includes interferon-regulated genes induced in myotonic dystrophy DM1 and DM2 cataracts (70), increased inflammatory gene expression in HD brain (71), inflammatory gene signatures of sporadic ALS motor neurons (72). The observation that protein aggregates and filaments are associated with Alzheimer’s disease has led to this disease sometimes being referred to as a ‘prion-like’ disease. Indeed the prion-like spread of protein aggregates has been noted for multiple neurodegenerative diseases (73). It is therefore noteworthy that inflammatory activation precedes neurodegeneration in a mouse retinal nerve model of prion disease (74), as well as a mouse model of AD (75).
Thus inflammation is activated at the right time and place to be the causal common mechanism for neurodegenerative diseases. Its characteristics are also consistent with the various genetic and environmental determinants of neurodegenerative diseases. Its genetic or pharmacological ablation in animal models of neurodegenerative diseases typically results in rescue of disease phenotypes—indicating that inflammatory activation is required for pathogenesis (Table 2).
Monogenic Contributions to Neurodegeneration
Like their systemic counterparts, numerous elements of the inflammatory response are sites for familial causes of neurodegenerative disease (Table 1). Inflammatory activation appears to be threshold dependant and also the subject of feed-forward (sensitization) as well as feed-back (inhibition) mechanisms. Genetic lesions fall into three reasonably distinct categories (Fig. 1C). Those that increase the abundance of trigger molecules can do so directly through mutations that impact ‘non-self’ to ‘self’ modification of endogenous molecules (31). Alternatively, mutations in degradative pathways can lead to an increase in steady-state levels of these danger signal molecules through their reduced turn-over (Table 1). Thirdly, mutations that result in increased activation signals from the pattern recognition of trigger molecules will also enhance inflammation.
Non-self mutations and direct increases in trigger molecules
‘Non-self’ nucleic acid is a common trigger molecule arising from invading pathogens that signals potential to cause harm. Non-self RNA and dsDNA are detected by various pattern recognition receptors to initiate inflammation. Modification of endogenous molecules is a common means of distinguishing self from non-self molecules, including various forms of RNA modification (31). Aberrant recognition of self RNA after insufficient modification is implicated as an upstream driver of RNA repeat disorders that elicit neurodegenerative diseases. Notably, loss of function mutations in the RNA editing enzyme ADAR1 are a cause of Aicardi-Goutieres syndrome—a congenital disease that includes neurodegeneration as one of its symptoms, with affected newborns having the appearance of being subjected to in utero inflammatory injury (5,6). The absence of ADAR1 editing renders certain endogenous RNAs (including Alu element double-stranded RNAs) to be recognized as non-self by the pattern recognition receptor IFIH1 that then activates inflammation (76–80). Indeed, gain-of-function mutations in IFIH1 are also a cause of Aicardi-Goutieres syndrome, while deletion of IFIH1 rescues the phenotype caused by ADAR1 loss in mouse models (81).
‘Non-self’ molecular patterns in proteins can also be afforded by tertiary structure. Prions adopt a structure that is recognized by pattern recognition receptors that then activate inflammation (74). Furthermore the seeding of such protein aggregate structures in neurodegeneration has been proposed as a means of their spread during disease progression (73).
Elevated inflammatory activation signal transduction pathways
A shift to elevated responsiveness to endogenous RNA can also trigger neuroinflammation. Gain-of-function mutations in the MDA-5 (IFIH1) pattern recognition receptor are a cause of Aicardi-Goutieres syndrome by enhancing the affinity of the receptor for its RNA ligands (5,6) and therefore increasing the resultant activation signal. Loss-of-function mutations in proteins that are negative regulators of inflammatory activation signalling give rise to familial ALS. TBK1 and optineurin participate in toll-like receptor (TLR) signal transduction that contributes to interferon response pathway (10,11). Progranulin is a TLR9 co-factor and its deficiency promotes microglial pruning (12).
Loss of RNA degradation pathways
RNA stress granules and the integrated stress response
Cellular stress response and inflammatory activation are integrated in various ways and include the management of mRNAs and their selective translation and degradation. Cellular stress causes a cessation in translation, the dissociation of polysomes and transport of mRNAs to RNA stress granules. If the stress is relieved the mRNAs are released for translation. If not, the RNA stress granules are trafficked to autophagosomes for degradation. These RNA stress granules are also the sites of viral RNA degradation. The antiviral RNA defense mechanism involves the binding of multiple RIG-I-like pattern recognition receptors along single molecules of RNA to form a filament structure that is then bound by mitochondrial antiviral signalling protein (MAVS) (82,83). A length requirement for the RNA molecule appears related to the need for such filaments to exceed a threshold for MAVS binding and/or activation (83,84). Mutations in several protein components of RNA stress granules and their trafficking are causes of familial ALS (Table 1). Plurality and integration are typical features of these responses. In stressed cells RNA can also be subject to degradation by non-sense mediated (NMD) decay (85) and in RNA exosomes. There is interplay between these RNA degradation pathways, with NMD regulating integrated stress response components.
The endogenous RNAs recognized by IFIH1 include not only Alu dsRNA but also products of the endoplasmic reticulum unfolded protein response (ER-UPR) (86). The ER-UPR is orchestrated by three proteins PERK, IRE-1 and ATF6. IRE-1 encodes a ribonuclease activity that cleaves XBP-1 mRNA in two places. Religation (by a tRNA ligase) results in an mRNA that encodes XBP-1s transcription factor that in turn stimulates expression of response genes including interferon-β (87). This XBP-1 mRNA activation process has been detected in multiple neurodegenerative diseases (88). IRE-1 cleavage of other mRNAs not only attenuates protein translation but generates unusual RNA 3′ ends that are ligands for the IFIH1 pattern recognition receptor thereby activating the inflammatory response (86). These endogenous non-self RNAs are in turn degraded by the SKIVL RNA exosome. Notably loss-of-function mutation in EXOSC3 from this SKIVL complex is a cause of inherited ponto-cerebellar hypoplasia (PCH1B) (19) indicating that this degradative pathway is crucial in avoiding the build up of RNAs that result in neurodegenerative disease (Table 1). Other genetic causes of PCH include mutations in genes involved in tRNA splicing and synthesis, while another PCH causing gene, TBC1D23, acts downstream of MYD88 and TRIF and upstream of XBP1 in the TLR4-NFKB1 pathway, consistent with a role in inflammatory activation (89). The IRE-1 pool of RNA cleavage products is also thought to contribute to the proximal cause of Aicardi-Goutieres syndrome (5,6).
The ER-UPR is in turn part of the larger integrated stress response (ISR) that includes sentinals for ER stress, viral infection, amino acid deprivation and heme deprivation (90). From each of these pathways one of four kinases, PERK, PKR, GCN2 and HRI is able to phosphorylate a unique serine in the translation initiation factor eIF2α. This eIF2α phosphorylation diminishes protein synthesis, while allowing preferential translation of specific mRNAs. The phosphorylation and de-phosphorylation of eIF2α is therefore a common integral element. Indeed eIF2α is also a central factor in RNA stress granule regulation.
Expanded repeat RNAs are not only ligands for pattern recognition receptors (31) but also the subject of non-canonical translation (repeat-associated non-AUG RAN translation) giving rise to repeat polypeptides that activate the integrated stress response (91). Therefore, somewhat analogous to RNA viruses, multiple gene products from expanded repeat disease loci are able to instigate inflammatory activation.
Mitochondria and autophagy
Mitochondria are not only central to homeostasis but along with NF-κB, are also key monitors and mediators of inflammatory activation (92). NF-κB is a transcription factor that transduces signals from certain TLRs. NF-κB targets include competing pathways mediated by p62 (sequestosome 1) and inflammasomes. p62 is a signalling scaffold and adaptor that binds damaged macromolecules (including mitochondria) that have been targeted by ubiquitination for degradation by autophagosomes. Autophagy is a major recycling system for a wide range of cellular molecules and structures including mitochondria (mitophagy) and RNA stress granules (granulophagy) as well as foreign pathogens (xenophagy) (26). Pink1 and parkin are two proteins responsible for the identification and tagging of damaged mitochondria by ubiquitination. LRRK2 controls mitochondrial fission (and fusion) that enables mitophagy of discrete parts of the mitochondrial complex. Multiple familial Parkinsons disease genes (α-synuclein, PINK1, Parkin, LRRK2, DJ-1, GBA and ATP13A2) have roles in mitophagy (18,93), while mutations in p62 are a cause of amyotrophic lateral sclerosis (4). Furthermore the protein TOMM40 is gate-keeper of Pink1 mitochondrial import. The TOMM40 gene is located within the APOE block of linkage disequilibrium that confers susceptibility to Alzheimer’s disease and its variation is a better predictor than APOE of age of disease onset (27).
In the alternative pathway NF-κB-induced inflammasomes activate gasdermin D, an extremely potent pore-forming cytolytic protein that mediates cell death (92,94–97). Damaged mitochondria also contribute to inflammasome activation, so their removal by mitophagy limits the activity of this pathway. Defects in mitophagy therefore enhance cell death. While these pathways have been characterized in systemic macrophages they appear to be relevant to the processes in their CNS counterparts, the microglia.
Mitochondria are also the sites of RNA stress granule/RNA pattern recognition receptor filament interaction with MAVS/IPS-1. This assembly leads to TRAFs/IKK mediated interferon activation and cell death unless degraded by autophagy (98). As noted previously, Ref(2)P, the Drosophila orthologue of p62, is a viral restriction factor, with multiple alleles conferring variable resistance/sensitivity to viral infection (3). Genetic variation in components of pathways dependent upon autophagy can thereby account for variable susceptibility to environmental triggers that unless degraded, can lead to cell death.
Clinical Implications
Together this assembled evidence demonstrates that endogenous inflammatory triggers and/or genetic mutations that accelerate, impede or redirect inflammatory processes, act in many major neurodegenerative diseases. This builds a strong rationale for invoking a shared autoinflammatory mechanism as central and causal to neurodegenerative diseases. This hypothesis must now be evaluated in genetic mouse models and clinical cohorts to build the evidence that inflammation is not just a bystander, but necessary and sufficient for neurodegenerative disease.
The pressing imperative for novel therapeutic strategies stands to be advanced by this fresh approach. Recent animal studies reveal the inflammation-driven integrated stress response pathway is an effective target for intervention in several neurodegenerative conditions. Excitingly, pharmacological inhibition of eIF2α activity was effective in relieving symptoms in a Drosophila model of amyotrophic lateral sclerosis and mouse models of prion disease, tau fronto-temporal dementia and traumatic brain injury (41–43). Although the long-term consequences of ablation of the integrated stress response are as yet unclear, this is just the tip of the iceberg of potential targets in the inflammatory cascade. The initial promise offered by these studies now justifies concerted research effort to explore redeployment of existing therapies for autoinflammatory conditions, and new drug targets, for clinical potential in the enormous and growing burden of neurodegenerative diseases.
Outstanding questions
What is/are the mechanism(s) for transition from the acute (protective) to chronic (pernicious) phase of inflammation? The transition between these two phases would appear an ideal therapeutic target. Will prolonging the acute phase and/or boosting capacity to resolve inflammation in the transition to chronic inflammatory-mediated cell death, delay onset or slow progression of neurodegenerative diseases? What systemic factor(s)/signals is/are responsible for increased risk after episodes of sepsis or trauma? Age is the greatest risk factor for neurodegenerative disease. CNS inflammation is known to increase with age—what are the targets to limit the impact of age and are there protective interventions to delay age-related onset and progression rate? Is it possible to inhibit neurodegeneration in humans by manipulating a single inflammatory pathway, or would it be necessary to modulate multiple pathways to obtain a clinical response? At what point in the neurodegenerative process in humans would it be too late to intervene?
Box 1. Inflammatory activation components (a partial inventory)
Trigger molecules/Danger signals
PAMPs—pathogen associated molecular patterns—microbial foreign ‘non-self’ molecules
DAMPs – danger associated molecular patterns – endogenous signals
dAMPs (alarmins) – damage associated molecular patterns – signals released by lytic cell death
HAMPs – homeostasis associated molecular patterns – signals induced by altered cellular physiology
Pattern recognition receptors (bind and transduce danger signals)
Toll-like receptors (TLRs) – bind wide variety of molecules
RIG-I-like receptors (RLRs) – recognize ‘non-self’ RNA
cGAS/STING – recognizes ‘non-self’ DNA
RAGE – recognizes advanced glycation end-products
Degradative pathways
Autophagy (includes mitophagy, granulophagy, xenophagy)
Integrated Stress Response (includes Unfolded Protein Response)
Non-sense mediated decay
Danger signal transduction pathways
MyD88
TRIF
NFκB
Mitochondrial Activator Viral Signalling (MAVS)
Inflammasomes, caspases
Signalling and defence molecules
Cytokines (includes interleukins, interferons, tumour necrosis factor)
Damage Associated Molecular Patterns (alarmins) – includes HMG-B1, amyloid-β, S100 proteins
Antimicrobial Peptides – includes amyloid-β, serum amyloid A
Cell death mechanisms
Non-lytic (Apoptosis) – protective
Lytic (Necroptosis, Pyroptosis, Ferroptosis, others?) – pernicious (release damage associated molecular patterns)
Box 2. Cells of the Neurovascular Unit
Smooth muscle cells - control localized blood flow
Endothelial cells - comprise the vasculature, blood-brain barrier integrity. Activate neutrophils as they traverse the blood-brain barrier
Pericytes - gate-keepers of the blood brain barrier, regulate endothelial cell function including molecular and cellular transport across the blood-brain barrier
Oligodendrocytes myelin sheath, axonal remodelling and remyelination
Neurons specific function to the region of the CNS, synaptic and dendritic plasticity
Astrocytes - monitor and maintain neuron synapses, activated by microglia
Microglia - resident macrophages, myeloid origin from yolk sac, environmental surveillance, constantly sampling, maintain homeostasis, perform synaptic pruning, phagocytose dead cells (99).
Neutrophils - by invitation, attracted by cytokines to location of inflammation, infiltration facilitated by pericytes, activated by endothelial cells, contribute to inflammatory response and resolution (60).
Acknowledgements
The authors dedicate this article to the memory of two inspiring leaders in this field—Allen D. Roses and Ben A. Barres. We are also grateful to Laura Hewson, Mary-Claire King, Shane Liddelow, Louise O’Keefe, Ann Saunders and Andrew Scott for reading and comments on drafts of this manuscript. We also thank Elma Affliki, Katerina Akassoglou, Ivona Aksentijevich, Bernhard Baune, Jae Jin Chae, Kurt Fischbeck, Massimo Gadina, John Hayball, Kim Hemsley, Michael Karin, Adam Labadorf, Michael Lardelli, Kalpana Manthiram, Dorian McGavern, Avi Nath, John O'Shea, Elaine Remmers, Bryan Smith, Nathan Staff and Bryan Traynor for helpful discussions.
Conflict of Interest statement. None declared.
Funding
This work was supported in part by grant funding from the National Health and Medical Research Council of Australia Project Grant APP1069348 and a Study Leave grant from The University of Adelaide to R.I.R. to undertake a sabbatical at the National Institutes of Health, Bethesda, USA.
References
- 1. Honig L.S., Vellas B., Woodward M., Boada M., Bullock R., Borrie M., Hager K., Andreasen N., Scarpini E., Liu-Seifert H. (2018) Trial of solanezumab for mild dementia due to Alzheimer’s disease. N. Engl. J. Med., 378, 321–330. [DOI] [PubMed] [Google Scholar]
- 2. Murphy M.P. (2018) Amyloid-beta solubility in the treatment of Alzheimer’s disease. N. Engl. J. Med., 378, 391–392. [DOI] [PubMed] [Google Scholar]
- 3. Carré-Mlouka A., Gaumer S., Gay P., Petitjean A.M., Coulondre C., Dru P., Bras F., Dezélée S., Contamine D. (2007) Control of sigma virus multiplication by the Ref(2)P gene of Drosophila melanogaster: an in vivo study of the PB1 domain of Ref(2)P. Genetics, 176, 409–419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Fecto F., Yan J., Vemula S.P., Liu E., Yang Y., Chen W., Zheng J.G., Shi Y., Siddique N., Arrat H. (2011) SQSTM1 mutations in familial and sporadic amyotrophic lateral sclerosis. Arch. Neurol., 68, 1440–1446. [DOI] [PubMed] [Google Scholar]
- 5. Crow Y.J., Chase D.S., Lowenstein Schmidt J., Szynkiewicz M., Forte G.M., Gornall H.L., Oojageer A., Anderson B., Pizzino A., Helman G.. et al. (2015) Characterization of human disease phenotypes associated with mutations in TREX1, RNASEH2A, RNASEH2B, RNASEH2C, SAMHD1, ADAR, and IFIH1. Am. J. Med. Genet. A, 167, 296–312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Crow Y.J., Manel N. (2015) Aicardi-Goutieres syndrome and the type I interferonopathies. Nat. Rev. Immunol., 15, 429–440. [DOI] [PubMed] [Google Scholar]
- 7. Mackenzie KarenJ., Carroll P.aula., Lettice L.aura., Tarnauskaitė Žygimantė., Reddy K.aalak., Dix F.lora., Revuelta A.ilsa., Abbondati E.rika., Rigby RachelE., Rabe B.jörn.. et al. (2016) Ribonuclease H2 mutations induce a cGAS/STING-dependent innate immune response. EMBO J., 35, 831–844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Sze A., Olagnier D., Lin R., van Grevenynghe J., Hiscott J. (2013) SAMHD1 host restriction factor: a link with innate immune sensing of retrovirus infection. J. Mol. Biol., 425, 4981–4994. [DOI] [PubMed] [Google Scholar]
- 9. Grieves J.L., Fye J.M., Harvey S., Grayson J.M., Hollis T., Perrino F.W. (2015) Exonuclease TREX1 degrades double-stranded DNA to prevent spontaneous lupus-like inflammatory disease. Proc. Natl. Acad. Sci. USA., 112, 5117–5122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Freischmidt A., Wieland T., Richter B., Ruf W., Schaeffer V., Müller K., Marroquin N., Nordin F., Hübers A., Weydt P.. et al. (2015) Haploinsufficieny of TBK1 causes familial ALS and fronto-temporal dementia. Nat. Neurosci., 18, 631–636. [DOI] [PubMed] [Google Scholar]
- 11. Tanishima M., Takashima S., Honda A., Yasuda D., Tanikawa T., Ishii S., MaruYama T. (2017) Identification of optineurin as an interleukin-1 receptor-associated kinase 1-binding protein and its role in regulation of MyD88-dependent signaling. J. Biol. Chem., 292, 17250–17257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Lui H., Zhang J., Makinson S.R., Cahill M.ichelle K., Kelley K.evin W., Huang H.sin-Y.i., Shang Y.ulei., Oldham M.ichael C., Martens L.auren H.erl., Gao F.uying.. et al. (2016) Progranulin deficiency promotes circuit-specific synaptic pruning by microglia via complement activation. Cell, 165, 921–935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Ito D., Suzuki N. (2011) Conjoint pathologic cascades mediated by ALS/FTLD-U linked RNA-binding proteins TDP-43 and FUS. Neurology, 77, 1636–1643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Talbot K., Ansorge D. (2006) Recent advances in the genetics of amyotrophic lateral sclerosis and frontotemporal dementia: common pathways in neurodegenerative disease. Hum. Mol. Genet., 15, R182–R187. [DOI] [PubMed] [Google Scholar]
- 15. Figley M.D., Bieri G., Kolaitis R.M., Taylor J.P., Gitler A.D. (2014) Profilin 1 associates with stress granules and ALS-linked mutations alter stress granule dynamics. J. Neurosci., 34, 8083–8097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Kumar D.K.V., Choi S.H., Washicosky K.J., Eimer W.A., Tucker S., Ghofrani J., Lefkowitz A., McColl G., Goldstein L.E., Tanzi R.E., Moir R.D. (2016) Amyloid-β peptide protects against microbial infection in mouse and worm models of Alzheimer’s disease. Sci. Transl. Med., 8, 340ra72.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Iqbal K., Liu F., Gong C.-X. (2016) Tau and neurodegenerative disease: the story so far. Nat. Rev. Neurol., 12, 15–27. [DOI] [PubMed] [Google Scholar]
- 18. Zhang H., Duan C., Yang H. (2015) Defective autophagy in Parkinson’s disease: lessons from genetics. Mol. Neurobiol., 51, 89–104. [DOI] [PubMed] [Google Scholar]
- 19. Wan J., Yourshaw M., Mamsa H., Rudnik-Schöneborn S., Menezes M.P., Hong J.E., Leong D.W., Senderek J., Salman M.S., Chitayat D.. et al. (2012) Mutations in the RNA exosome component gene EXOSC3 cause pontocerebellar hypoplasia and spinal motor neuron degeneration. Nat. Genet., 44, 704–708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Budde B.S., Namavar Y., Barth P.G., Poll-The B.T., Nürnberg G., Becker C., van Ruissen F., Weterman M.A., Fluiter K., te Beek E.T.. et al. (2008) tRNA splicing endonuclease mutations cause pontocerebellar hypoplasia. Nat. Genet., 40, 1113–1118. [DOI] [PubMed] [Google Scholar]
- 21. Breuss M.W., Sultan T., James K.N., Rosti R.O., Scott E., Musaev D., Furia B., Reis A., Sticht H., Al-Owain M.. et al. (2016) Autosomal-recessive mutations in the tRNA splicing endonuclease subunit TSEN15 cause pontocerebellar hypoplasia and progressive microcephaly. Am. J. Hum. Genet., 99, 228–235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Ivanova E.L., Mau-Them F.T., Riazuddin S., Kahrizi K., Laugel V., Schaefer E., de Saint Martin A., Runge K., Iqbal Z., Spitz M.A.. et al. (2017) Homozygous truncating variants in TBC1D23 cause pontocerebellar hypoplasia and alter cortical development. Am. J. Hum. Genet., 101, 428–440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Sims R., van der Lee S.J., Naj A.C., Bellenguez C., Badarinarayan N., Jakobsdottir J., Kunkle B.W., Boland A., Raybould R., Bis J.C.. et al. (2017) Rare coding variants in PLCG2, ABI3, and TREM2 implicate microglial-mediated innate immunity in Alzheimer’s disease. Nat. Genet., 49, 1373–1384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Jonsson T., Stefansson H., Steinberg S., Jonsdottir I., Jonsson P.V., Snaedal J., Bjornsson S., Huttenlocher J., Levey A.I., Lah J.J.. et al. (2013) Variant of TREM2 associated with the risk of Alzheimer’s disease. N. Engl. J. Med., 368, 107–116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Krasemann S.usanne., Madore C.harlotte., Cialic R.on., Baufeld C.aroline., Calcagno N.arghes., El Fatimy R.achid., Beckers L.ien., O’Loughlin E.laine., Xu Y.ang., Fanek Z.ain.. et al. (2017) The TREM2-APOE pathway drives the transcriptional phenotype of dysfunctional microglia in neurodegenerative diseases. Immunity, 47, 566–581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Klionsky D.J., Abdelmohsen K., Abe A., Abedin M.J., Abeliovich H., Acevedo Arozena A., Adachi H., Adams C.M., Adams P.D., Adeli K., Adhihetty P.J.. et al. (2016) Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy, 12, 1–222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Roses A.D., Lutz M.W., Amrine-Madsen H., Saunders A.M., Crenshaw D.G., Sundseth S.S., Huentelman M.J., Welsh-Bohmer K.A., Reiman E.M. (2010) A TOMM40 variable-length polymorphism predicts the age of late-onset Alzheimer’s disease. Pharmacogenomics J., 10, 375–384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Chapuis J., Hot D., Hansmannel F., Kerdraon O., Ferreira S., Hubans C., Maurage C.A., Huot L., Bensemain F., Laumet G.. et al. (2009) Transcriptomic and genetic studies identify IL-33 as a candidate gene for Alzheimer’s disease. Mol. Psychiatry, 14, 1004–1016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Sardi S.P., Clarke J., Kinnecom C., Tamsett T.J., Li L., Stanek L.M., Passini M.A., Grabowski G.A., Schlossmacher M.G., Sidman R.L.. et al. (2011) CNS expression of glucocerebrosidase corrects α-synuclein pathology and memory in a mouse model of Gaucher-related synucleinopathy. Proc. Natl. Acad. Sci. USA., 108, 12101–12106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Sproviero W., Shatunov A., Stahl D., Shoai M., van Rheenen W., Jones A.R., Al-Sarraj S., Andersen P.M., Bonini N.M., Conforti F.L.. et al. (2017) ATXN2 trinucleotide repeat length correlates with risk of ALS. Neurobiol. Aging, 51, 178.e1–178.e9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Samaraweera S.E., O'Keefe L.V., Price G.R., Venter D.J., Richards R.I. (2013) Distinct roles for Toll and autophagy pathways in double-stranded RNA toxicity in a Drosophila model of expanded repeat neurodegenerative diseases. Hum. Mol. Genet., 22, 2811–2819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Lee J.Y., Lee J.D., Phipps S., Noakes P.G., Woodruff T.M. (2015) Absence of toll-like receptor 4 (TLR4) extends survival in the hSOD1 G93A mouse model of amyotrophic lateral sclerosis. J. Neuroinflammation, 12, 90.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Aikawa T., Mogushi K., Iijima-Tsutsui K., Ishikawa K., Sakurai M., Tanaka H., Mizusawa H., Watase K. (2015) Loss of MyD88 alters neuroinflammatory response and attenuates early Purkinje cell loss in a spinocerebellar ataxia type 6 mouse model. Hum. Mol. Genet., 24, 4780–4791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Quek H., Luff J., Cheung K., Kozlov S., Gatei M., Lee C.S., Bellingham M.C., Noakes P.G., Lim Y.C., Barnett N.L.. et al. (2017) A rat model of ataxia-telangiectasia: evidence for a neurodegenerative phenotype. Hum. Mol. Genet., 26, 109–123. [DOI] [PubMed] [Google Scholar]
- 35. Petersen A.J., Katzenberger R.J., Wassarman D.A. (2013) The innate immune response transcription factor relish is necessary for neurodegeneration in a Drosophila model of ataxia-telangiectasia. Genetics, 194, 133–142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Saitoh Y.N., Fujikake N., Okamoto Y., Popiel H.A., Hatanaka Y., Ueyama M., Suzuki M., Gaumer S., Murata M., Wada K., Nagai Y. (2015) p62 plays a protective role in the autophagic degradation of polyglutamine oligomers in polyglutamine disease model flies. J. Biol. Chem., 290, 1442–1453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Swinnen B., Bento-Abreu A., Gendron T.F., Boeynaems S., Bogaert E., Nuyts R., Timmers M., Scheveneels W., Hersmus N., Wang J.. et al. (2018) A zebrafish model for C9orf72 ALS reveals RNA toxicity as a pathogenic mechanism. Acta Neuropathol. doi:10.1007/s00401-017-1796-5. [DOI] [PubMed] [Google Scholar]
- 38. Griffioen K., Mattson M.P., Okun E. (2018) (2018) Deficiency of Toll-like receptors 2, 3 or 4 extends life expectancy in Huntington’s disease mice. Heliyon, 4, e00508.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Sasaki T., Liu K., Agari T., Yasuhara T., Morimoto J., Okazaki M., Takeuchi H., Toyoshima A., Sasada S., Shinko A.. et al. (2016) Anti-high mobility group box 1 antibody exerts neuroprotection in a rat model of Parkinson’s disease. Exp. Neurol., 275, 220–231. [DOI] [PubMed] [Google Scholar]
- 40. Leyns C.E.G., Ulrich J.D., Finn M.B., Stewart F.R., Koscal L.J., Remolina Serrano J., Robinson G.O., Anderson E., Colonna M., Holtzman D.M. (2017) TREM2 deficiency attenuates neuroinflammation and protects against neurodegeneration in a mouse model of tauopathy. Proc. Natl. Acad. Sci. USA., 114, 11524.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Halliday M., Radford H., Zents K.A.M., Molloy C., Moreno J.A., Verity N.C., Smith E., Ortori C.A., Barrett D.A., Bushell M., Mallucci G.R. (2017) Repurposed drugs targeting eIF2a-P-mediated translational repression prevent neurodegeneration in mice. Brain, 140, 1768–1783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Chou A., Krukowski K., Jopson T., Zhu P.J., Costa-Mattioli M., Walter P., Rosi S. (2017) Inhibition of the integrated stress response reverses cognitive deficits after traumatic brain injury. Proc. Natl. Acad. Sci. USA., 114, E6420–E6426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Kim H.-J., Raphael A.R., LaDow E.S., McGurk L., Weber R.A., Trojanowski J.Q., Lee V.M., Finkbeiner S., Gitler A.D., Bonini N.M. (2014) Therapeutic modulation of eIF2α phosphorylation rescues TDP-43 toxicity in amyotrophic lateral sclerosis disease models. Nat. Genet., 46, 152–160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Manthiram K., Zhou Q., Aksentijevich I., Kastner D.L. (2017) The monogenic autoinflammatory diseases define new pathways in human innate immunity and inflammation. Nat. Immunol., 18, 832–842. [DOI] [PubMed] [Google Scholar]
- 45. Richards R.I., Robertson S.A., O'Keefe L.V., Fornarino D., Scott A., Lardelli M., Baune B.T. (2016) The enemy within: innate surveillance-mediated cell death, the common mechanism of neurodegenerative disease. Front. Neurosci., 10, 193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Fang F., Yu Q., Arancio O., Chen D., Gore S.S., Yan S.F., Yan S.S. (2018) RAGE mediates Aβ accumulation in a mouse model of Alzheimer’s disease via modulation of β- and γ-secretase activity. Hum. Mol. Genet. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Ombrello M., Remmers E.F., Sun G., Freeman A.F., Datta S., Torabi-Parizi P., Subramanian N., Bunney T.D., Baxendale R.W., Martins M.S. (2012) Cold urticaria, immunodeficiency, and autoimmunity related to PLCG2 deletions. N. Engl. J. Med., 366, 330–338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Zhou Q., Lee G.S., Brady J., Datta S., Katan M., Sheikh A., Martins M.S., Bunney T.D., Santich B.H., Moir S.. et al. (2012) A hypermorphic missense mutation in PLCG2, encoding phospholipase Cγ2, causes a dominantly inherited autoinflammatory disease with immunodeficiency. Am. J. Hum. Genet., 91, 713–720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Russo M.V., McGavern D.B. (2016) Inflammatory neuroprotection following traumatic brain injury. Science, 353, 783–785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Widmann C.N., Heneka M.T. (2014) Long-term cerebral consequences of sepsis. Lancet Neurol., 13, 630–636. [DOI] [PubMed] [Google Scholar]
- 51. Sampson T.R., Debelius J.W., Thron T., Janssen S., Shastri G.G., Ilhan Z.E., Challis C., Schretter C.E., Rocha S., Gradinaru V.. et al. (2016) Gut microbiota regulate motor deficits and neuroinflammation in a model of Parkinson's disease. Cell, 167, 1469–1480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Itzhaki R.F., Lathe R., Balin B.J., Ball M.J., Bearer E.L., Braak H., Bullido M.J., Carter C., Clerici M., Cosby S.L.. et al. (2016) Microbes and Alzheimer’s disease. J. Alzheimer’s Dis., 51, 979–984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Cribbs D.H., Berchtold N.C., Perreau V., Coleman P.D., Rogers J., Tenner A.J., Cotman C.W. (2012) Extensive innate immune gene activation accompanies brain aging, increasing vulnerability to cognitive decline and neurodegeneration: a microarray study. J. Neuroinflammation, 9, 179.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Lodato M.A., Rodin R.E., Bohrson C.L., Coulter M.E., Barton A.R., Kwon M., Sherman M.A., Vitzthum C.M., Luquette L.J., Yandava C.N.. et al. (2017) Aging and neurodegeneration are associated with increased mutations in single human neurons. Science, 10.1126/science.aao4426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Ransohoff R.M. (2016) How neuroinflammation contributes to neurodegeneration. Science, 353, 777–783. [DOI] [PubMed] [Google Scholar]
- 56. Ransohoff R.M. (2016) Surprises from the sanitary engineers Nature. doi: 10.1038/nature17881. [DOI] [PubMed]
- 57. Ilieva H., Polymenidou M., Cleveland D.W. (2009) Non-cell autonomous toxicity in neurodegenerative disorders: ALS and beyond. J. Cell Biol., 187, 761–772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Yamanaka K., Chun S.J., Boillee S., Fujimori-Tonou N., Yamashita H., Gutmann D.H., Takahashi R., Misawa H., Cleveland D.W. (2008) Astrocytes as determinants of disease progression in inherited amyotrphic lateral sclerosis. Nat. Neurosci., 11, 251–253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Liddelow S.A., Guttenplan K.A., Clarke L.E., Bennett F.C., Bohlen C.J., Schirmer L., Bennett M.L., Münch A.E., Chung W.S., Peterson T.C.. et al. (2017) Neurotoxic reactive astrocytes are induced by activated microglia. Nature, 541, 481–487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Liu X., Dong H., Wang M., Gao Y., Zhang T., Hu G., Duan H., Mu X. (2016) IL-1α-induced microvascular endothelial cells promote neutrophil killing by increasing MMP-9 concentration and lysozyme activity. Immunol. Res., 64, 133–142. [DOI] [PubMed] [Google Scholar]
- 61. Cortes C.J., Ling S.C., Guo L.T., Hung G., Tsunemi T., Ly L., Tokunaga S., Lopez E., Sopher B.L., Bennett C.F.. et al. (2014) Muscle expression of mutant androgen receptor accounts for systemic and motor neuron disease phenotypes in spinal and bulbar muscular atrophy. Neuron, 82, 295–307., (2014) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Vijayan M., Reddy P.H. (2016) Stroke, vascular dementia, and Alzheimer’s disease: molecular Links. J. Alzheimer’s Dis., 54, 427–443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Zhong Z., Deane R., Ali Z., Parisi M., Shapovalov Y., O'Banion M.K., Stojanovic K., Sagare A., Boillee S., Cleveland D.W., Zlokovic B.V. (2008) ALS-causing SOD1 mutants generate vascular changes prior to motor neuron degeneration. Nat. Neurosci., 11, 420–422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Chen Z.-L., Revenko A.S., Singh P., MacLeod A.R., Norris E.H., Strickland S. (2017) Depletion of coagulation factor XII ameliorates brain pathology and cognitive impairment in Alzheimer disease mice. Blood, 129, 2547–2556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Davalos D., Ryu J.K., Merlini M., Baeten K.M., Le Moan N., Petersen M.A., Deerinck T.J., Smirnoff D.S., Bedard C., Hakozaki H.. et al. (2012) Fibrinogen-induced perivascular microglial clustering is required for the development of axonal damage in neuroinflammation. Nat. Commun., 3, 1227.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Clark I.A., Vissel B. (2015) Amyloid β: one of three danger-associated molecules that are secondary inducers of the proinflammatory cytokines that mediate Alzheimer’s disease. Br. J. Pharmacol., 172, 3714–3727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Wallach D., Kang T.B., Dillon C.P., Green D.R. (2016) Programmed necrosis in inflammation: toward identification of the effector molecules. Science, 352, aaf2154. /dx.doi. org/10.1126/science.aaf2154 [DOI] [PubMed] [Google Scholar]
- 68. Bjorkqvist M., Wild E.J., Thiele J., Silvestroni A., Andre R., Lahiri N., Raibon E., Lee R.V., Benn C.L., Soulet D.. et al. (2008) A novel pathogenic pathway of immune activation detectable before clinical onset in Huntington's disease. J. Exp. Med., 205, 1869–1877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Politis M. (2015) Increased central microglial activation associated with peripheral cytokine levels in premanifest Huntington's disease gene carriers. Neurobiol. Dis., 83, 115–121. [DOI] [PubMed] [Google Scholar]
- 70. Rhodes J.D., Lott M.C., Russell S.L., Moulton V., Sanderson J., Wormstone I.M., Broadway D.C. (2012) Activation of the innate immune response and interferon signalling in myotonic dystrophy type 1 and type 2 cataracts. Hum. Mol. Genet., 21, 852–862. [DOI] [PubMed] [Google Scholar]
- 71. Labadorf A., Hoss A.G., Lagomarsino V., Latourelle J.C., Hadzi T.C., Bregu J., MacDonald M.E., Gusella J.F., Chen J.F., Akbarian S.. et al. (2015) RNA sequence analysis of human Huntington disease brain reveals an extensive increase in inflammatory and developmental gene expression. PLoS One, 10, e0143563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Batra R., Hutt K., Vu A., Rabin S.J., Baughn M.W., Libby R.T., Hoon S., Ravits J., Yeo G.W. (2016) Gene expression signatures of sporadic ALS motor neuron populations. BioRxiv, 4, 74. [Google Scholar]
- 73. Polymenidou M., Cleveland D.W. (2012) Prion-like spread of protein aggregates in neurodegeneration. J. Exp. Med., 209, 889–893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. West Greenlee M.H., Lind M., Kokemuller R., Mammadova N., Kondru N., Manne S., Smith J., Kanthasamy A., Greenlee J. (2016) Temporal resolution of misfolded prion protein transport accumulation, glial activation, and neuronal death in the retinas of mice inoculated with scrapie. Am. J. Pathol., 186, 2302–2309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Boza-Serrano A., Yang Y., Paulus A., Deierborg T. (2018) Innate immune alterations are elicited in microglial cells before plaque deposition in the Alzheimer’s disease mouse model 5xFAD. Sci. Rep., 8, 1550.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Mannion N.M., Greenwood S.M., Young R., Cox S., Brindle J., Read D., Nellåker C., Vesely C., Ponting C.P., McLaughlin P.J.. et al. (2014) The RNA-editing enzyme ADAR1 controls innate immune responses to RNA. Cell Rep., 9, 1482–1494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. George C.X., Ramaswami G., Li B., Samuel C.E. (2016) Editing of cellular self-RNAs by adenosine deaminase ADAR1 suppresses innate immune stress responses. J. Biol. Chem., 291, 6158–6168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Ahmad S., Mu X., Yang F., Greenwald E., Park J.W., Jacob E., Zhang C.-Z., Hur S. (2018) Breaching self-tolerance to Alu duplex RNA underlies MDA5-mediated inflammation. Cell, 172, 797–810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Chung H., Calis J.J.A., Wu X., Sun T., Yu Y., Sarbanes S.L., Dao Thi V.L., Shilvock A.R., Hoffmann H.-H., Rosenberg B.R., Rice C.M. (2018) Human ADAR1 prevents endogenous RNA from triggering translational shutdown. Cell, 172, 811–824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Uggenti C., Crow Y.J. (2018) Sort yourself out. Cell, 172, 640–642. [DOI] [PubMed] [Google Scholar]
- 81. Liddicoat B.J., Chalk A.M., Walkley C.R. (2016) ADAR1, inosine and the immune sensing system: distinguishing self from non-self. WIREs RNA, 7, 157–172. [DOI] [PubMed] [Google Scholar]
- 82. Paro S., Imler J.L., Meignin C. (2015) Sensing viral RNAs by Dicer/RIG-I like ATPases across species. Curr. Opin. Immunol., 32, 106–113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Yoneyama M., Onomoto K., Jogi M., Akaboshi T., Fujita T. (2015) Viral RNA detection by RIG-I-like receptors. Curr. Opin. Immunol., 32, 48–53. [DOI] [PubMed] [Google Scholar]
- 84. Kato H., Takeuchi O., Mikamo-Satoh E., Hirai R., Kawai T., Matsushita K., Hiiragi A., Dermody T.S., Fujita T., Akira S. (2008) Length-dependent recognition of double-stranded ribonucleic acids by retinoic acid: inducible gene-I and melanoma differentiation-associated gene 5. J. Exp. Med., 205, 1601–1610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Goetz A.E., Wilkinson M. (2017) Stress and the nonsense-mediated RNA decay pathway. Cell. Mol. Life Sci., 74, 3509–3531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Janssens S., Pulendran B., Lambrecht B.N. (2014) Emerging functions of the unfolded protein response in immunity. Nat. Immunol., 15, 910–919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Hu F., Yu X., Wang H., Zuo D., Guo C., Yi H., Tirosh B., Subjeck J.R., Qiu X., Wang X.Y. (2011) ER stress and its regulator X-box-binding protein-1 enhance polyIC-induced innate immune response in dendritic cells. Eur. J. Immunol., 41, 1086–1097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Dunys J., Duplan E., Checler F. (2014) The transcription factor X-box binding protein-1 in neurodegenerative diseases. Mol. Neurodegener., 9, 35.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. De Arras L., Yang I.V., Lackford B., Riches D.W., Prekeris R., Freedman J.H., Schwartz D.A., Alper S. (2012) Spatiotemporal inhibition of innate immunity signaling by the Tbc1d23 RAB-GAP. J. Immunol., 188, 2905–2913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Pakos-Zebrucka K., Koryga I., Mnich K., Ljujic M., Samali A., Gorman A.M. (2016) The integrated stress response. EMBO Rep., 17, 1374–1395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Green K.M., Glineburg M.R., Kearse M.G., Flores B.N., Linsalata A.E., Fedak S.J., Goldstrohm A.C., Barmada S.J., Todd P.K. (2017) RAN translation at C9orf72-associated repeat expansions is selectively enhanced by the integrated stress response. Nat. Commun. doi: 10.1038/s41467-017-02200-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Zhong Z., Umemura A., Sanchez-Lopez E., Liang S., Shalapour S., Wong J., He F., Boassa D., Perkins G., Ali S.R.. et al. (2016) NF-kB restricts inflammasome activation via elimination of damaged mitochondria. Cell, 164, 896–910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Ryan B.J., Hoek S., Fon E.A., Wade-Martins R. (2015) Mitochondrial dysfunction and mitophagy in Parkinson's: from familial to sporadic disease. Trends Biochem. Sci., 40, 200–210. [DOI] [PubMed] [Google Scholar]
- 94. Kayagaki N., Stowe I.B., Lee B.L., O’Rourke K., Anderson K., Warming S., Cuellar T., Haley B., Roose-Girma M., Phung Q.T.. et al. (2015) Caspase-11 cleaves gasdermin D for non-canonical inflammasome signalling. Nature, 526, 666–671. [DOI] [PubMed] [Google Scholar]
- 95. Shi J., Zhao Y., Wang K., Shi X., Wang Y., Huang H., Zhuang Y., Cai T., Wang F., Shao F. (2015) Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death. Nature, 526, 660–665. [DOI] [PubMed] [Google Scholar]
- 96. Ding J., Wang K., Liu W., She Y., Sun Q., Shi J., Sun H., Wang D.C., Shao F. (2016) Pore-forming activity and structural autoinhibition of the gasdermin family. Nature, 535, 111–116. [DOI] [PubMed] [Google Scholar]
- 97. Aglietti R.A., Estevez A., Gupta A., Ramirez M.G., Liu P.S., Kayagaki N., Ciferri C., Dixit V.M., Dueber E.C. (2016) GsdmD p30 elicited by caspase-11 during pyroptosis forms pores in membranes. Proc. Natl. Acad. Sci. USA., 113, 7858–7863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Yoneyama M., Jogi M., Onomoto K. (2016) Regulation of antiviral innate immune signaling by stress-induced RNA granules. J. Biochem., 159, 279–286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Cherry J.D., Olschowka J.A., O'Banion M.K. (2014) Neuro-inflammation and M2 microglia: the good, the bad, and the inflamed. J. Neuroinflammation, 11, 98.. [DOI] [PMC free article] [PubMed] [Google Scholar]