A miniature two-photon microscope circumvents previous limitations of this technology and now allows imaging of activity not only in the soma but also in the dendrites and spines of neurons in freely moving animals engaged in a wide spectrum of behaviors.
Conventional brain imaging in animal models with multiphoton microscopy usually requires that the animal has its head fixed to a very large table microscope, which severely limits the types of behaviors that can be studied.
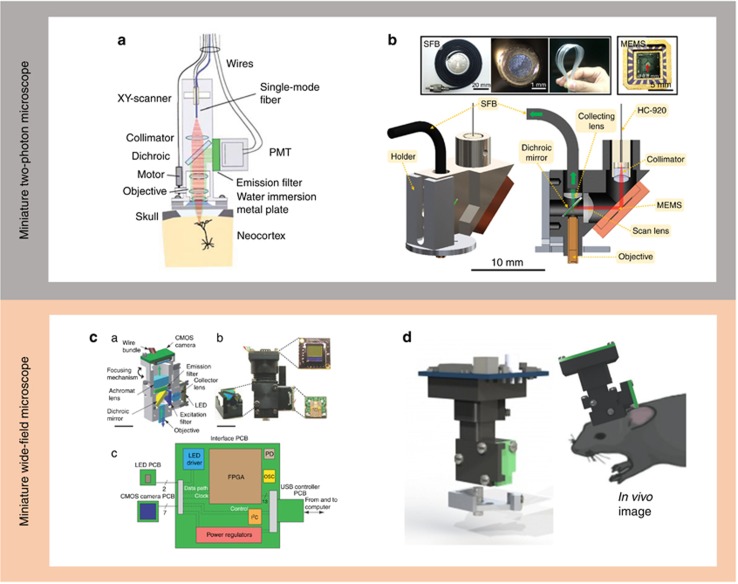
To address this limitation, recently, Schnitzer and colleagues at Stanford University1, 2 developed a complementary technique that involves head-mounted microscopes for fluorescent imaging, leveraging the power of ultrasensitive fluorescent proteins that are widely used for imaging neuronal activity (GCaMP6, Figure 1c). Inspired by Schnitzer’s invention, our laboratory helped construct an open-source, head-mounted, integrated miniature fluorescence microscope1, 2 (the UCLA miniscope) with associated hardware and software to image in vivo calcium transients using the calcium indicator GCaMP6f (Figure 1d)5. We recently used these head-mounted microscopes to study CA1 neuronal ensembles encoding multiple memories over time6.
Figure 1.
Miniaturized head-mounted microscopes. (a) Miniature two-photon microscope by Denk et al. in which the PMY is directly mounted on the microscope3; (b) miniature two-photon microscope by Cheng et al. weighing only 2.2 g4; (c) miniature wide-field microscope by Schnitzer et al.1, 2; (d) the UCLA miniature wide-field miniscope5.
These head-mounted microscopes for fluorescent imaging, however, have limitations regarding the types of structures and events that can be visualized. For example, with these microscopes, imaging is limited to somas since it is virtually impossible to resolve smaller structures such as dendrites. To address these limitations, in 2001, Denk and colleagues designed a head-mounted two-photon microscope (Figure 1a)3. Although this was an important conceptual development, this microscope and others that followed have had a very limited impact in neuroscience due to technological problems that limited their use. This is due to several problems, including optical limitations of the fibers used, which cannot deliver 920-nm femtosecond laser pulses to a living brain without significant distortion that precludes useful imaging, inadequate sampling rates and significant motion artifacts. Because of these limitations, stable imaging of calcium dynamics in dendritic spines of freely behaving animals, for example, has remained beyond the capability of available approaches.
To address these problems, Weijian Zong (now at HHMI), Liangyi Chen and Peace Cheng at Peking University developed a fast, high-resolution and miniaturized two-photon microscope capable of resolving activity in single spines in freely moving mice4. Weighing only approximately 2 g, this miniaturized two-photon microscope can image with a very high spatiotemporal resolution, and its nifty miniature scanner allows for random-access imaging and fast free-line scanning. A key development that enabled this remarkable approach was the design of a hollow-core photonic crystal fiber, developed by Aimin Wang, that is capable of delivering 920-nm femtosecond laser pulses with essentially no nonlinear pulse-broadening. This allows the miniaturized two-photon microscope to perform comparably to its big, bench-top cousins. The authors tested their miniature microscope in a wide range of behavioral paradigms, and they produced beautiful GCaMP6f imaging of activity not only in the soma but also in dendrites and spines. Importantly, the imaging was not degraded by the unpredictable, often sudden and vigorous head and body movements of the animal, thus circumventing a serious limitation of previous designs.
By every standard, this microscope represents a fundamental development that will change the way neuroscientists image cellular and subcellular structures in actively moving animals. It also inspires further efforts to design optical systems geared toward miniaturized imaging systems. The potential impact of this microscope goes beyond imaging of neuronal and dendritic activity. Systems neuroscience is primed to enter a new era, in which imaging of complex biological events in identified cells and subcellular structures of cell ensembles will bring us closer than ever before to understanding the very organizing principles underlying complex behaviors. This marvelous invention just brought us a step closer to that goal.
Footnotes
The author declares no conflict of interest.
References
- Cai DJ, Aharoni D, Shuman T, Shobe J, Biane J et al. A shared neural ensemble links distinct contextual memories encoded close in time. Nature 2016; 534: 115–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen TW, Wardill TJ, Sun Y, Pulver SR, Renninger SL et al. Ultrasensitive fluorescent proteins for imaging neuronal activity. Nature 2013; 499: 295–300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh KK, Burns LD, Cocker ED, Nimmerjahn A, Ziv Y et al. Miniaturized integration of a fluorescence microscope. Nat Methods 2011; 8: 871–878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helmchen F, Fee MS, Tank DW, Denk W. A miniature head-mounted two-photon microscope: high-resolution brain imaging in freely moving animals. Neuron 2001; 31: 903–912. [DOI] [PubMed] [Google Scholar]
- Ziv Y, Burns LD, Cocker ED, Hamel EO, Ghosh KK et al. Long-term dynamics of CA1 hippocampal place codes. Nat Neurosci 2013; 16: 264–266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zong WJ, Wu RL, Li ML, Hu YH, Li YJ et al. Fast high-resolution miniature two-photon microscopy for brain imaging in freely behaving mice. Nat Methods 2017; 14: 713–719. [DOI] [PubMed] [Google Scholar]